Introduction
Ovarian yolk sac tumor (OYST), also referred to as
ovarian endodermal sinus tumor, is a rare malignant tumor typically
occurring in adolescent and young adult women. OYSTs comprise ~1%
of ovarian malignancies (1,2). In
addition, OYST is a highly malignant germ cell tumor,
characteristically expressing α-fetoprotein (AFP). AFP is a useful
tumor marker for detecting malignancies such as hepatocellular
carcinoma, yolk sac tumor and a specific type of gastric carcinoma.
An elevated serum AFP level is usually detected in patient with
OYST and may facilitate diagnosis (2).
The prognosis of OYST was extremely dismal in the
past (3,4). Over the last few decades, with the
introduction of novel combination chemotherapy, the prognosis of
OYST has significantly improved; however, its prognosis remains
unsatisfactory. Furthermore, little is known regarding the
prognostic factors of OYST, due to the rarity of this type of
tumor. The number of studies that have analyzed the prognostic
factors of OYST is currently limited, while the rare published
series on OYST usually include <50 cases (2,3,5–7).
Certain studies presented evidence of the association between serum
AFP level and the prognosis of OYST. Mitchell et al
(8) reported that a shorter
relapse-free survival (RFS) was primarily observed among patients
with AFP >1,000 kU/l compared to that among patients with AFP
≤1,000 kU/l [odds ratio (OR)=0.09, 95% confidence interval (CI):
0.01–0.85]. In addition, de La Motte Rouge et al (9) demonstrated that there was a
significant association between high serum AFP levels and 5-year
overall survival (OS) of OYST (OR=0.24; 95% CI: 0.05–1.19). By
contrast, other studies failed to demonstrate that AFP was a
prognostic factor in patients with OYST (1,6,10).
Therefore, the objective of the present study was to
perform a meta-analysis of the association between serum AFP levels
and the prognosis of OYST.
Materials and methods
Literature search
A systematic review of original articles analyzing
the prognostic value of AFP for OYST was performed through
searching MEDLINE (1946 to August, 2013) and EMBASE (1980 to
August, 2013). The search terms used were ‘ovarian neoplasm’,
‘ovarian masses’, ‘ovarian lesions’ and ‘alpha fetoprotein/AFP’,
combined with the MeSH term ‘prognosis’ (disease-free survival,
relapse-free survival, overall survival, progression-free survival,
medical futility, neoplasm grading, neoplasm staging, nomograms,
pregnancy outcome, treatment outcome and treatment failure). The
search was limited to human studies and reports in languages other
than English were excluded. Moreover, the reference lists of the
available studies were reviewed to identify additional relevant
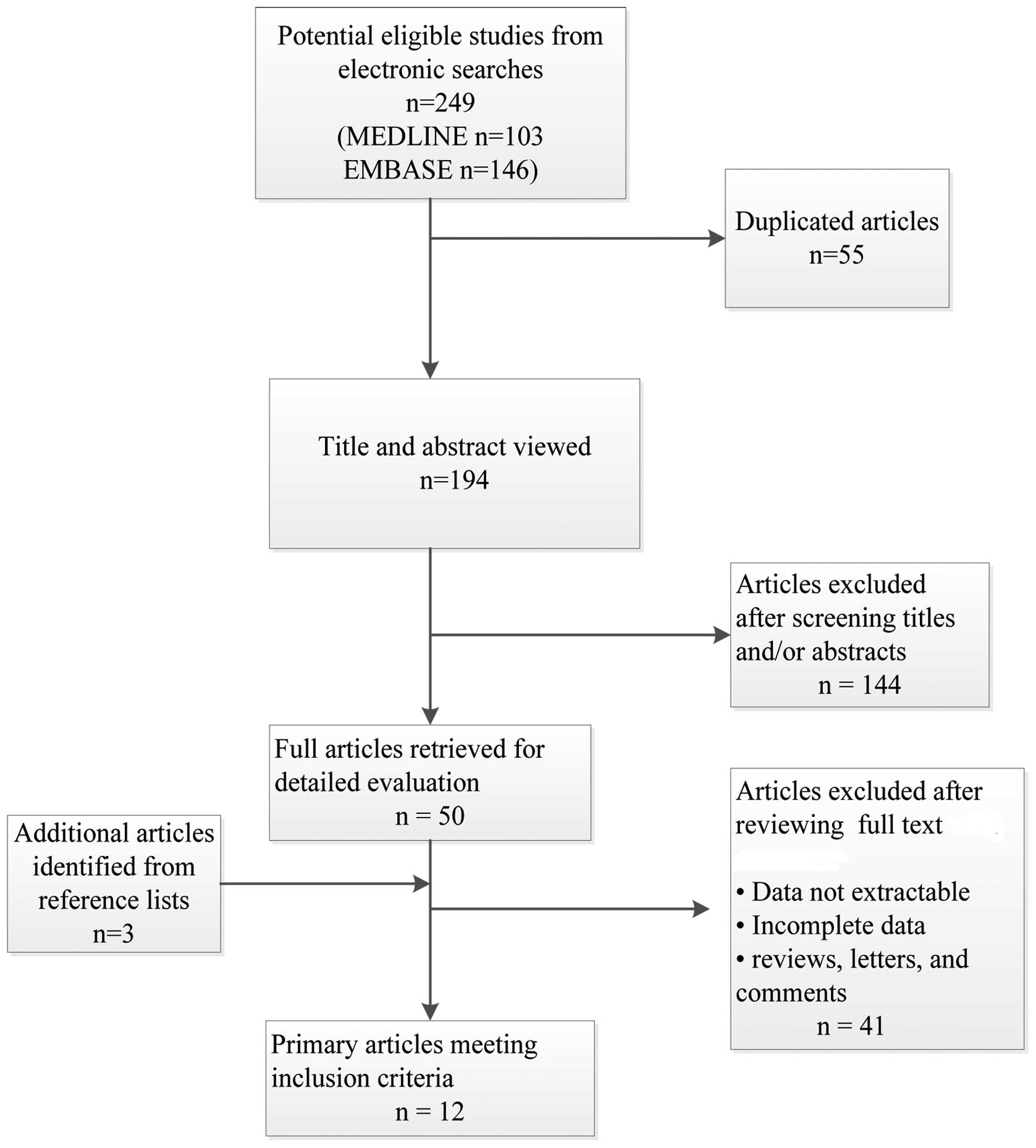
citations. The study selection process is summarized in Fig. 1.
Inclusion criteria
The inclusion criteria were as follows: i)
pathological diagnosis of YST or mixed YST; ii) serum AFP level was
obtained; iii) original article published in English; and iv)
survival data obtained, with ≥12 months of follow-up.
Exclusion criteria
The exclusion criteria were as follows: i)
incomplete data; ii) animal studies; iii) duplicate studies; iv)
reviews, letters and comments; v) low-quality articles; and vi)
results presented as abstracts only.
Data extraction
English articles meeting the inclusion criteria were
regularly assessed by pairs of reviewers. Any differences were
discussed until the two reviewers reached a consensus. We recorded
publication reference, tumor types (OYST or other tumors), mean
patient age, study design, statistical techniques, country, total
number of participants, characteristics of AFP, outcome reported
(OS and RFS), mean follow-up and confounding factors. The
characteristics of the studies included in this meta-analysis are
summarized in Table I and detailed
data for each study are provided in Table II.
 | Table ICharacteristics of the studies
included in the meta-analysis. |
Table I
Characteristics of the studies
included in the meta-analysis.
| First author | Mean age (years) | Study design | Statistical
analysis | Country | (Refs.) |
|---|
| Sell et
al | 28.2 | Retrospective | Case description | Denmark | (11) |
| Talerman et
al | 15.9 | Retrospective | Case description | Holland | (12) |
| Sessa et
al | 17.2 | Retrospective | Case description | Italy | (13) |
| Sasaki et
al | 20.1 | Retrospective | Cox proportional
hazard models | Japan | (14) |
| Nogales et
al | 58.3 | Retrospective | Case description | Spain | (15) |
| Davidoff et
al | 3.8 | Retrospective | Case description | America | (16) |
| Chen et
al | 15.1 | Retrospective
(case-control study) | Case description | China | (17) |
| Mitchell et
al | 21 | Retrospective | Log-rank test and
Cox's regression | UK | (8) |
| Nawa et
al | NK | Prospective | Logistic
regression | Japan | (6) |
| Umezu et
al | NK | Retrospective | Log-rank test | Japan | (18) |
| Tong et
al | NK | Retrospective | Cox proportional
hazard model | China | (10) |
| de La Motte Rouge
et al | NK | Retrospective | Cox proportional
hazard regression | France | (9) |
 | Table IIDetailed data of the studies included
in the meta-analysis. |
Table II
Detailed data of the studies included
in the meta-analysis.
| First author
(year) | Subjects | Characteristics of
AFP | Outcomes | Mean follow-up
(months) | Controlled
factors | (Refs.) |
|---|
| Sell et al
(1976) | 8 | Postoperative | 1-year OS and
1-year RFS | 14 | Primary surgery and
AFP determination | (11) |
| Talerman et
al (1978) | 9 | Preoperative and
postoperative | 2-year OS and
2-year RFS | 24 | Primary
surgery | (12) |
| Sessa et al
(1987) | 13 | Postoperative | 2-year RFS | 36 | Primary surgery and
combination chemotherapy | (13) |
| Sasaki et al
(1994) | 27 | Preoperative | 3-year OS | 28 | Histopathological
diagnosis and combination chemotherapy | (14) |
| Nogales et
al (1996) | 6 | Postoperative | 1-year OS and
1-year RFS | 14 | Primary
surgery | (15) |
| Davidoff et
al (1996) | 6 | Postoperative | 2-year OS and
2-year RFS | 53 | Primary surgery and
combination chemotherapy | (16) |
| Chen et al
(1997) | 6 | Postoperative | 1-year OS and
1-year RFS | 12 | Primary surgery and
combination chemotherapy. Liver metastasis | (17) |
| Mitchell et
al (1999) | 41 | Postoperative | 2-year RFS | 68 | Platinum-based
chemotherapy | (8) |
| Nawa et al
(2001) | 42 | Preoperative | 5-year OS | 60 | Primary surgery and
combination chemotherapy | (6) |
| Umezu et al
(2008) | 36 | Preoperative | 5-year OS | 60 | Primary surgery and
combination chemotherapy | (18) |
| Tong et al
(2008) | 53 | Preoperative | 5-year OS | 60 | Primary surgery and
combination chemotherapy | (10) |
| de La Motte Rouge
et al (2011) | 73 | Postoperative | 5-year OS and
5-year RFS | 71 | Primary
surgery | (9) |
Exposure measurements
We recorded the characteristics of AFP, including
value, types and standard classification from each study and
classified different groups according to the characteristics of AFP
among studies. Certain studies reported on preoperative or
postoperative AFP level for each patient, whereas others reported
the total number of patients in each group, divided by preoperative
or postoperative AFP level. We calculated the OR of higher vs.
lower AFP level by comparing the outcomes in the two groups. The
standard of higher and lower AFP level was based on the respective
standard from each study.
Study quality
The quality of each study was assessed using a
subset of items from Quality Assessment of Diagnostic Accuracy
Studies 2 (19). We estimated the
criteria as follows: type of study design, patient selection by
diagnostic protocol, exposure measurement (characteristics of AFP,
comparability of measurement across the study groups, follow-up
time, validity and reliability of the measurement method),
statistical analysis (appropriateness) and adjustment for key
confounders (primary surgery and combination chemotherapy).
Statistical analysis
Pooled ORs of AFP level with 95% CIs for OS and RFS
were obtained using the fixed- or random-effects models. The Q and
I2 statistics were used to assess heterogeneity, where
P≤0.05 or I2>50% were considered to indicate
significant heterogeneity. When the result of the Q-test and
I2 statistics suggested heterogeneity (P≤0.05 and
I2>50%), a random-effects model was used. We used
either fixed- or random-effects models in pooled estimates.
Furthermore, we preferentially presented a random-effects model
when heterogeneity was present. We also created forest plots for
the ORs from each of the studies included in the meta-analysis and
the estimation of the pooled OR. The sizes of the markers of each
OR in the plots indicated the relative weight each study
contributed to the pooled estimation. Publication bias was
estimated by funnel plots, Begg's test and a weighted Egger's test
(20). In addition, we produced
sensitivity analyses, whereby each study was omitted in turn,
reevaluating the pooled estimates under particular conditions. All
the analyses were performed using STATA software, version 12.0
(StataCorp LP, College Station, TX, USA).
Results
Study characteristics
This meta-analysis included 12 studies reporting on
19 independent estimates. There were 320 participants from 9
different countries (3 studies from Japan, 2 from China and 1 each
from Denmark, Holland, Italy, Spain, America, France and United
Kingdom). Five studies reported preoperative AFP and OS; OS was
assessed by 2- (n=1), 3- (n=1) and 5-year OS (n=3). Six studies
reported postoperative AFP and OS; OS was assessed by 1- (n=3), 2-
(n=2) and 5-year OS (n=1). Eight studies reported postoperative AFP
and RFS; RFS was assessed by 1- (n=3), 2- (n=4) and 5-year RFS
(n=1). The follow-up time ranged between 14 and 71 months.
Association between preoperative AFP
and OS in OYST
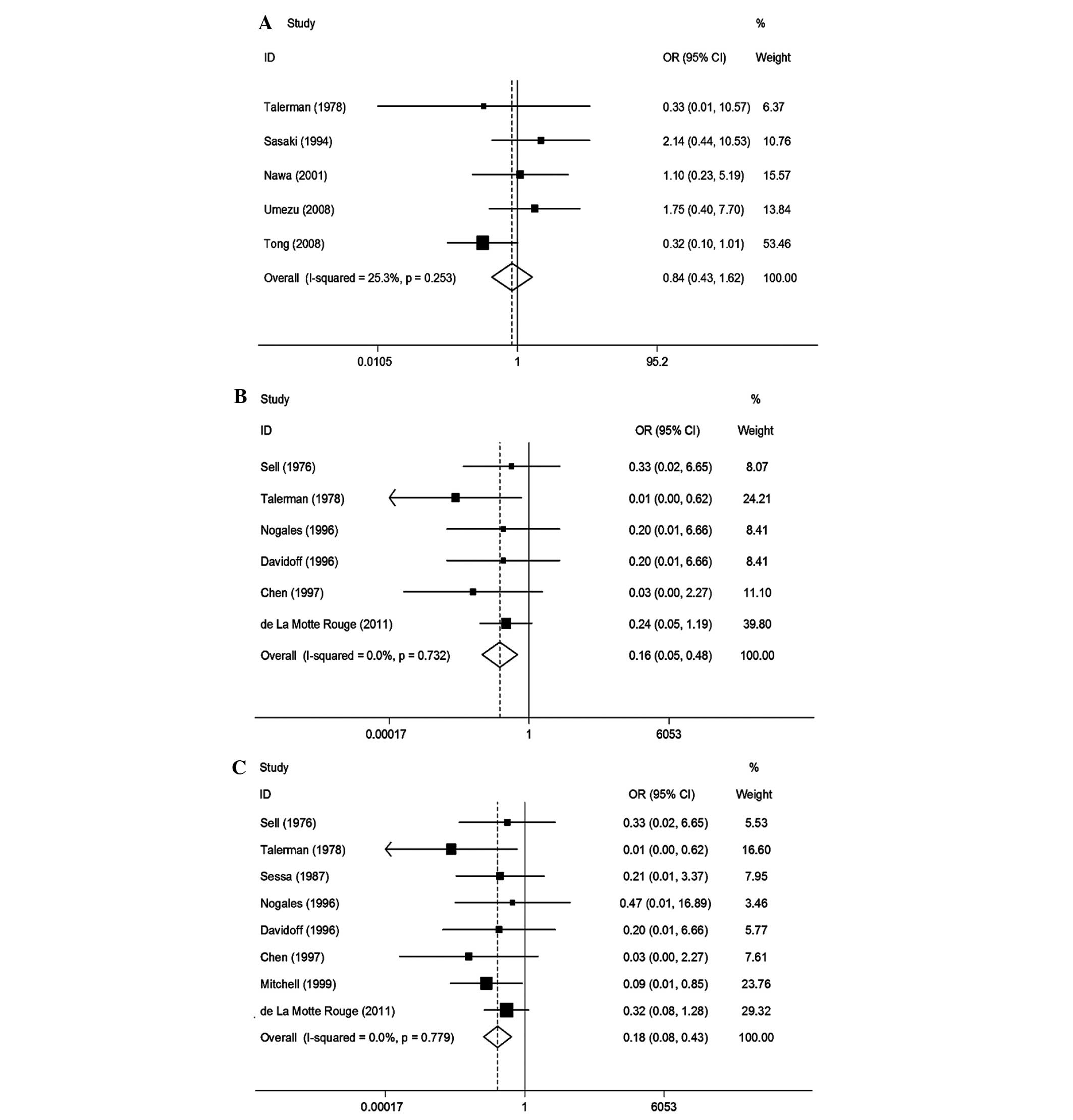
We conducted a meta-analysis for 5 studies on
preoperative AFP and OS (Table
II). Heterogeneity tests revealed no evidence of heterogeneity
among studies (Q=5.35, P=0.253, I2=25.3%). Therefore,
the fixed-effects model was used to calculate a pooled OR with 95%
CI. We failed to identify a correlation of preoperative AFP with OS
(OR=0.84, 95% CI: 0.43–1.62) in OYST. A forest plot is shown in
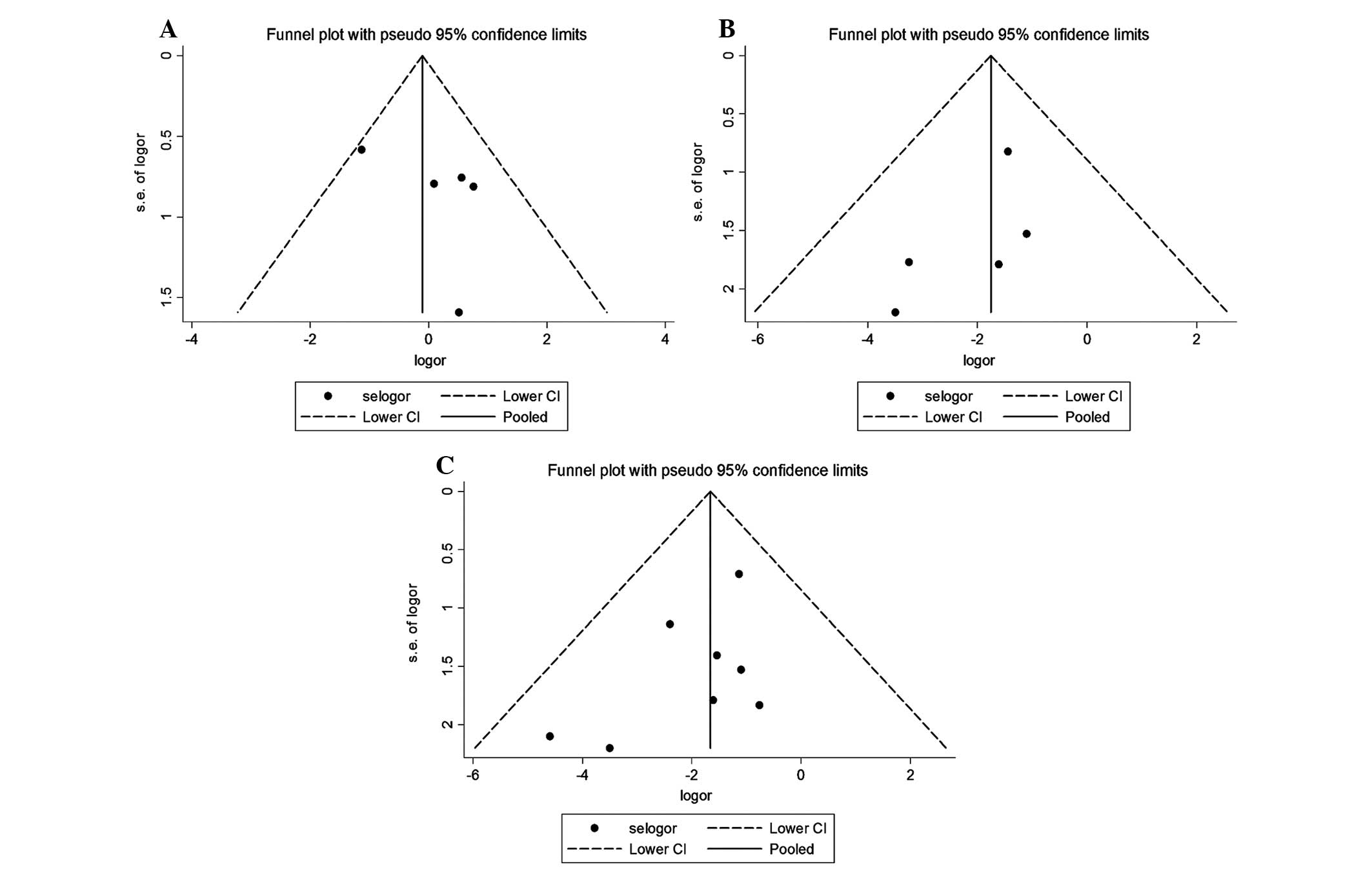
Fig. 2A. A funnel plot is shown in
Fig. 3A and it exhibits near
symmetry. The Begg's and Egger's tests indicated that publication
bias was unlikely (Zc=0.73, P=0.462; t=1.24, P=0.303). A
sensitivity analysis demonstrated that the results for preoperative
AFP and OS in these studies were not robust, due to the Umezu et
al (18) study, which
exhibited significant heterogeneity (Table III).
 | Table IIISensitivity analysis of the
associations between AFP and outcomes. |
Table III
Sensitivity analysis of the
associations between AFP and outcomes.
| AFP and
outcome | Subjects | Model | OR (95% CI) | Q | P-value | I2 (%) | (Refs.) |
|---|
| Preoperative AFP
and OS | | | | | | | |
|
Total | 167 | Fixed-effects |
0.84
(0.43–1.62) |
5.35 |
0.253 |
25.3 | |
|
Talerman et al
excluded | 9 | Fixed-effects |
0.87
(0.44–1.71) |
5.07 |
0.167 |
40.8 | (12) |
| Sasaki
et al excluded | 27 | Fixed-effects |
0.68
(0.32–1.42) |
3.72 |
0.293 |
19.4 | (14) |
| Nawa
et al excluded | 42 | Fixed-effects |
0.79
(0.38–1.64) |
5.22 |
0.157 |
42.5 | (6) |
| Umezu
et al excluded | 36 | Fixed-effects |
0.69
(0.33–1.46) |
4.16 |
0.245 |
27.8 | (18) |
| Tong
et al excluded | 53 | Fixed-effects |
1.43
(0.62–3.31) |
1.11 |
0.774 |
0.0 | (10) |
| Postoperative AFP
and OS | | | | | | | |
|
Total | 108 | Fixed-effects |
0.16
(0.05–0.48) |
2.79 |
0.732 |
0.0 | |
| Sell
et al excluded | 8 | Fixed-effects |
0.15
(0.04–0.48) |
2.54 |
0.638 |
0.0 | (11) |
|
Talerman et al
excluded | 9 | Fixed-effects |
0.21
(0.05–0.68) |
0.89 |
0.926 |
0.0 | (12) |
| Nogales
et al excluded | 6 | Fixed-effects |
0.16
(0.05–0.50) |
2.78 |
0.595 |
0.0 | (15) |
|
Davidoff et al
excluded | 6 | Fixed-effects |
0.16
(0.05–0.50) |
2.78 |
0.595 |
0.0 | (16) |
| Chen
et al excluded | 6 | Fixed-effects |
0.18
(0.06–0.55) |
2.17 |
0.705 |
0.0 | (17) |
| de La
Motte Rouge et al excluded | 73 | Fixed-effects |
0.11
(0.03–0.50) |
2.39 |
0.665 |
0.0 | (9) |
| Postoperative AFP
and RFS | | | | | | | |
|
Total | 162 | Fixed-effects |
0.18
(0.08–0.43) |
4.01 |
0.779 |
0.0 | |
| Sell
et al excluded | 8 | Fixed-effects |
0.17
(0.07–0.43) |
3.86 |
0.695 |
0.0 | (11) |
|
Talerman et al
excluded | 9 | Fixed-effects |
0.22
(0.09–0.54) |
1.94 |
0.925 |
0.0 | (12) |
| Sessa
et al excluded | 13 | Fixed-effects |
0.18
(0.07–0.45) |
4.00 |
0.676 |
0.0 | (13) |
| Nogales
et al excluded | 6 | Fixed-effects |
0.17
(0.07–0.42) |
3.75 |
0.710 |
0.0 | (15) |
|
Davidoff et al
excluded | 6 | Fixed-effects |
0.18
(0.07–0.44) |
4.01 |
0.675 |
0.0 | (16) |
| Chen
et al excluded | 6 | Fixed-effects |
0.20
(0.08–0.47) |
3.29 |
0.772 |
0.0 | (17) |
|
Mitchell et al
excluded | 41 | Fixed-effects |
0.21
(0.08–0.54) |
3.50 |
0.744 |
0.0 | (8) |
| de La
Motte Rouge et al excluded | 73 | Fixed-effects |
0.13
(0.04–0.39) |
3.07 |
0.799 |
0.0 | (9) |
Association between postoperative AFP
and OS in OYST
Six studies were included (Table II). Heterogeneity tests revealed
no evidence of heterogeneity among studies (Q=2.79, P=0.732,
I2=0.0%); therefore, the fixed-effects model was used to
assess a pooled OR with 95% CI. We observed that a high
postoperative AFP level was associated with a decrease in OS
(OR=0.16, 95% CI: 0.05–0.48). A high postoperative AFP level was
associated with a worse OS compared to a low postoperative AFP
level in patients with OYST. A forest plot is shown in Fig. 2B. A funnel plot is shown in
Fig. 3B and it exhibits near
symmetry. The Begg's and Egger's tests indicated that publication
bias was unlikely in the meta-analysis on association between
postoperative AFP and OS in these studies (Zc=1.31, P=0.189;
t=-1.47, P=0.217). A sensitivity analysis demonstrated that the
pooled estimate of the effect of postoperative AFP level on the
risk of OS was robust, as shown in Table III.
Association between postoperative AFP
and RFS in OYST
We performed a meta-analysis of 8 studies on
postoperative AFP and RFS (Table
II). Heterogeneity tests revealed no evidence of heterogeneity
among studies (Q=4.01, P=0.779, I2=0.0%); therefore, the
fixed-effects model was used to estimate a pooled OR with 95% CI.
We observed that a high postoperative AFP level was associated with
a decrease in RFS (OR=0.18, 95% CI: 0.08–0.43). A high
postoperative AFP level was associated with a worse RFS compared to
low postoperative AFP level in patients with OYST. A forest plot is
shown in Fig. 2C. A funnel plot is
shown in Fig. 3C and it exhibits
near symmetry. The Begg's and Egger's tests indicated that
publication bias was unlikely in the meta-analysis on association
between postoperative AFP and RFS in these studies (Zc=1.11,
P=0.266; t=-1.61, P=0.159). A sensitivity analysis demonstrated
that the result was robust, as shown in Table III.
Subgroup analysis of postoperative AFP
and OS
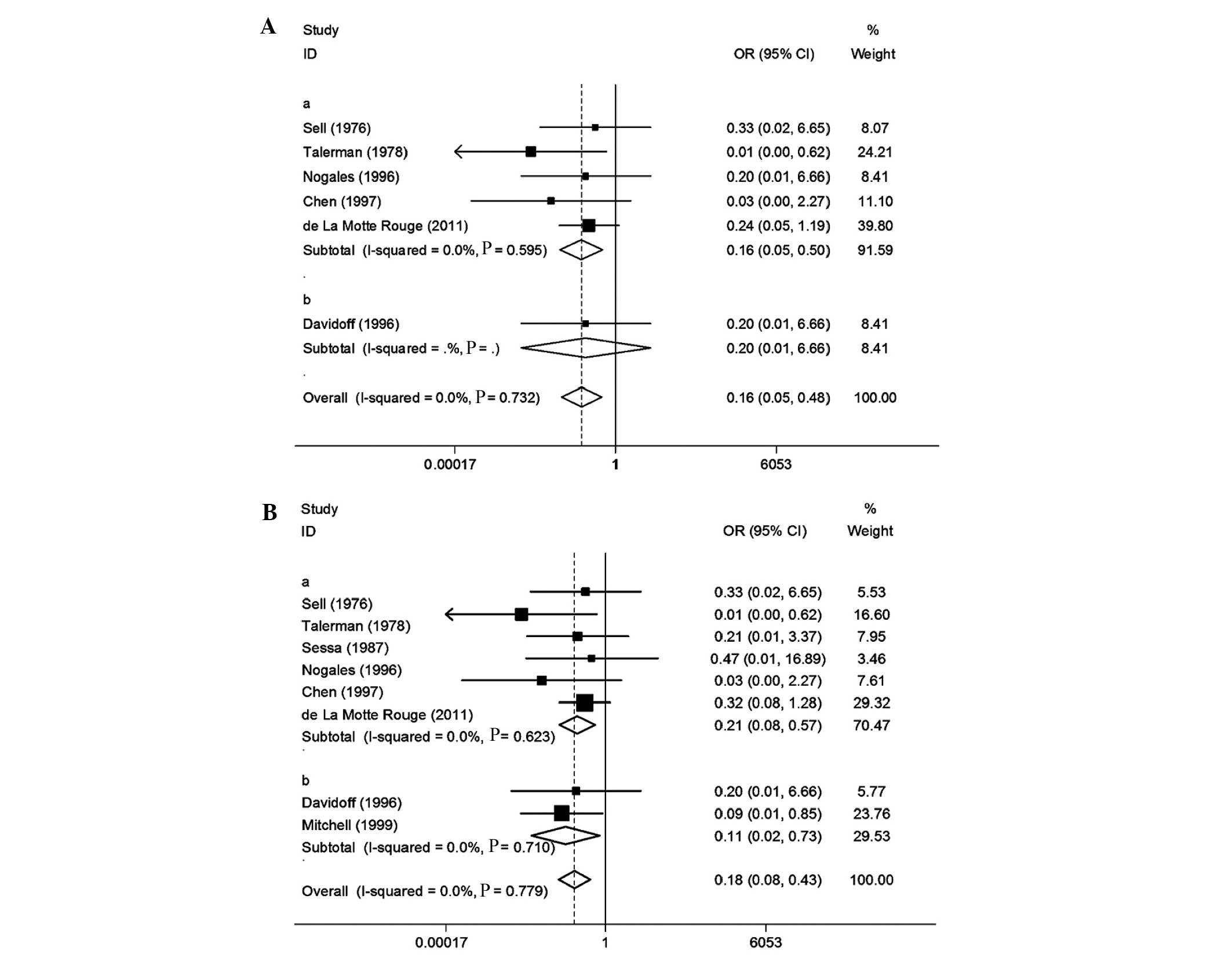
We conducted a meta-analysis of 8 studies on
postoperative AFP and OS (Table
IV). Heterogeneity tests revealed no evidence of heterogeneity
among studies (Q=2.78, P=0.595, I2=0.0%); therefore, the
fixed-effects model was used to calculate a pooled OR with 95% CI.
In the design that was used, a high postoperative AFP level was
associated with a decrease in OS (OR=0.16, 95% CI: 0.05–0.50). A
postoperative AFP level of >1,000 ng/ml was associated with a
worse OS compared to an AFP level of ≤1,000 ng/ml in patients with
OYST. A forest plot is shown in Fig.
4A. The Begg's and Egger's tests indicated that publication
bias was unlikely in the meta-analysis of the association between
postoperative AFP and OS in these studies (Zc=1.22, P=0.221;
t=-1.48, P=0.236).
 | Table IVDetailed data of the studies included
in the subgroup meta-analysis. |
Table IV
Detailed data of the studies included
in the subgroup meta-analysis.
| First author
(year) | Subjects | Characteristic of
AFP | Outcomes | Mean followed-up
(months) | Controlled
factors | (Refs.) |
|---|
| Sell et al
(1976) | 8 | Postoperative AFP
>1,000 vs. AFP ≤1,000 ng/ml | 1-year OS and
1-year RFS | 14 | Primary surgery and
AFP determination | (11) |
| Talerman et
al (1978) | 9 | Postoperative AFP
>1,000 vs. AFP ≤1,000 ng/ml | 2-year OS and
2-year RFS | 24 | Primary
surgery | (12) |
| Sessa et al
(1987) | 13 | Postoperative AFP
>1,000 vs. AFP ≤1,000 ng/ml | 2-year RFS | 36 | Primary surgery and
combination chemotherapy | (13) |
| Nogales et
al (1996) | 6 | Postoperative AFP
>1,000 vs. AFP ≤1,000 ng/ml | 1-year OS and
1-year RFS | 14 | Primary
surgery | (15) |
| Davidoff et
al (1996) | 6 | Postoperative AFP
elevated vs. AFP normal | 2-year OS and
2-year RFS | 53 | Primary surgery and
combination chemotherapy | (16) |
| Chen et al
(1997) | 6 | Postoperative AFP
>1,000 vs. AFP ≤1,000 ng/ml | 1-year OS and
1-year RFS | 12 | Primary surgery and
combination chemotherapy Liver metastasis | (17) |
| Mitchell et
al (1999) | 41 | Postoperative AFP
>1,000 vs. AFP ≤1,000 kU/l | 2-year RFS | 68 | Platinum-basedch
emotherapy | (8) |
| de La Motte Rouge
et al (2011) | 73 | Postoperative AFP
>1,000 vs. AFP ≤1,000 ng/ml | 5-year OS and
5-year RFS | 71 | Primary
surgery | (9) |
Subgroup analysis of postoperative AFP
and RFS
Eight studies were included (Table IV). Heterogeneity tests revealed
no evidence of heterogeneity among studies (Q=3.50, P=0.623,
I2=0.0%); therefore, the fixed-effects model was used to
calculate a pooled OR with 95% CI. In the design that was used, a
high postoperative AFP level was associated with a decrease in RFS
(OR=0.21, 95% CI: 0.08–0.57). A postoperative AFP level of
>1,000 ng/ml was associated with a worse RFS compared to an AFP
level of ≤1,000 ng/ml in patients with OYST. A forest plot is shown
in Fig. 4B. The Begg's and Egger's
tests indicated that publication bias was unlikely in the
meta-analysis on the association between postoperative AFP and RFS
in these studies (Zc=1.11, P=0.266; t=-1.61, P=0.159).
Discussion
OYST is a rare malignancy, occurring most commonly
in young women. AFP is an important tumor marker of yolk sac
tumors. Moreover, an elevation of the serum AFP level is usually
detected in patient with YST and is correlated with the severity of
the disease. Due to the extreme sensitivity of AFP in diagnosing
recurrence, the follow-up of the patients mainly includes measuring
AFP levels (2).
Our study demonstrated that preoperative AFP was not
a prognostic factor in patients with OYST. We failed to identify a
correlation between preoperative AFP level and OS (OR=0.84, 95% CI:
0.43–1.62) in OYST, which may be due to the fact that preoperative
AFP levels may only reflect the condition of the primary disease,
but have a limited impact on patient outcome. Surgical resection of
the tumor and chemosensitivity may be key to a successful treatment
in patients with OYST. It is generally considered that the initial
therapy for the majority of the patients should be a surgical
intervention with comprehensive staging, so that a histological
diagnosis and appropriate staging may be obtained (1,6).
Tumor stage and the amount of residual tumor significantly affect
prognosis. Certain authors have reported that tumor stage is a
significant prognostic factor (3,21,22).
Over the last few decades, fertility-sparing surgery has been the
standard management of patients desiring fertility preservation,
regardless of disease stage. Conservative surgery was not shown to
affect the survival figures (10,23,24).
OYSTs are characterized by a high malignant
potential, but are also highly chemosensitive. It was reported that
3 courses of bleomycin, etoposide and cisplatin (BEP) was the
current standard adjuvant therapy, with 4 courses of BEP
recommended in case of bulky residual disease following surgery
(8,25,26).
An effective postoperative chemotherapy regimen (BEP regimen)
following surgery is a prognostic factor at least as significant as
stage in these chemosensitive tumors (27). Our meta-analysis demonstrated that
postoperative AFP was a prognostic factor in patients with OYST.
Postoperative AFP was found to be correlated with OS and RFS. A
high postoperative AFP level was associated with a worse OS
(OR=0.16, 95% CI: 0.05–0.48) and RFS (OR=0.18, 95% CI: 0.08–0.43)
compared to a low postoperative AFP level in patients with OYST,
possibly because a high AFP level postoperatively suggests that the
tumor was not completely resected. A number of factors may lead to
incomplete tumor resection, such as inappropriate surgical
approach, high degree of tumor invasion and metastatic spread. In
general, patients with more residual tumor will exhibit higher
postoperative AFP levels. Moreover, the amount of residual tumor
was shown to be a significant prognostic factor (1,6).
To date, we demonstrated that high postoperative AFP
levels were associated with worse OS and RFS compared to low
postoperative AFP levels in patients with OYST. According to the
groups from all studies that were divided by AFP level (>1,000
vs. ≤1,000 ng/ml), except in the studies by Davidoff et al
(16) and Mitchell et al
(8), we performed a subgroup
analysis and observed that a postoperative AFP level of >1,000
ng/ml was associated with a decrease in OS (OR=0.16, 95% CI:
0.05–0.50) and RFS (OR=0.21, 95% CI: 0.08–0.57). A postoperative
AFP level of >1,000 ng/ml was associated with a worse OS and RFS
compared to an AFP level of ≤1,000 ng/ml in patients with OYST.
Accordingly, we concluded that postoperative AFP level >1,000
ng/ml was associated with poor prognosis in patients with OYST.
The sensitivity analysis demonstrated that the
results for preoperative AFP and OS were not robust, as the study
by Umezu et al (18)
exhibited significant heterogeneity. However, the Umezu et
al (18) study did not affect
the overall results. Furthermore, the results for postoperative AFP
and outcome were robust.
A number of factors, which were difficult to
evaluate, may influence the effect estimates, such as sample size,
study design, characteristics of AFP, outcomes and duration of
follow-up. Of note, there was no heterogeneity among the studies we
observed using statistical analysis. Additionally, an almost
symmetric inverted funnel shape was displayed by all the studies,
indicating that publication bias was unlikely.
There were several limitations to our meta-analysis.
First, due to the rarity of this type of tumor, our research
included only a limited amount of cases and discussed two types of
OYST (pure and mixed OYST) rather than a single type (pure OYST).
Mixed OYSTs exhibit different biological or clinical
characteristics compared to pure OYSTs, which may affect the
results. More data are required to determine the similarities and
differences between pure and mixed OYSTs. Second, the determination
of a high AFP level standard (>1,000 ng/ml) in our study was
dependent on a single measurement (which was used in most studies).
This standard may be inaccurate. In the future, a more accurate
standard must be determined based on accumulation of research
data.
In conclusion, OYST is a rare malignancy encountered
in young women and AFP is a useful tumor marker for detecting OYST.
The postoperative, but not the preoperative, AFP level was found to
be a prognostic factor in patients with OYST. High postoperative
AFP levels were associated with a decrease in OS and RFS. In
particular, a postoperative AFP level of >1,000 ng/ml was
associated with a worse OS and RFS compared to an AFP level of
≤1,000 ng/ml in patients with OYST. Therefore, a postoperative AFP
level of >1,000 ng/ml was identified as an indicator of poor
prognosis in patients with OYST.
Acknowledgements
This study was supported by the Natural Science
Foundation of Zhejiang Province of China (grant no. Y2100593).
Glossary
Abbreviations
Abbreviations:
|
AFP
|
α-fetoprotein
|
|
OYST
|
ovarian yolk sac tumor
|
|
OS
|
overall survival
|
|
OR
|
odds ratio
|
|
CI
|
confidence interval
|
|
RFS
|
relapse-free survival
|
|
BEP
|
bleomycin, etoposide and cisplatin
|
References
|
1
|
Kawai M, Kano T, Furuhashi Y, et al:
Prognostic factors in yolk sac tumors of the ovary. A
clinicopathologic analysis of 29 cases. Cancer. 67:184–192. 1991.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Dallenbach P, Bonnefoi H, Pelte MF and
Vlastos G: Yolk sac tumours of the ovary: an update. Eur J Surg
Oncol. 32:1063–1075. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kurman RJ and Norris HJ: Endodermal sinus
tumor of the ovary: a clinical and pathologic analysis of 71 cases.
Cancer. 38:2404–2419. 1976. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Jimerson GK and Woodruff JD: Ovarian
extraembryonal teratoma. I. Endodermal sinus tumor. Am J Obstet
Gynecol. 127:73–79. 1977.PubMed/NCBI
|
|
5
|
Ayhan A, Taskiran C, Bozdag G, Altinbas S,
Altinbas A and Yuce K: Endodermal sinus tumor of the ovary: the
Hacettepe University experience. Eur J Obstet Gynecol Reprod Biol.
123:230–234. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Nawa A, Obata N, Kikkawa F, et al:
Prognostic factors of patients with yolk sac tumors of the ovary.
Am J Obstet Gynecol. 184:1182–1188. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gershenson DM, Del Junco G, Herson J and
Rutledge FN: Endodermal sinus tumor of the ovary: the M.D. Anderson
experience. Obstet Gynecol. 61:194–202. 1983.PubMed/NCBI
|
|
8
|
Mitchell PL, Al-Nasiri N, A'Hern R, et al:
Treatment of nondysgerminomatous ovarian germ cell tumors: an
analysis of 69 cases. Cancer. 85:2232–2244. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
de La Motte Rouge T, Pautier P, Rey A, et
al: Prognostic factors in women treated for ovarian yolk sac
tumour: a retrospective analysis of 84 cases. Eur J Cancer.
47:175–182. 2011.PubMed/NCBI
|
|
10
|
Tong X, You Q, Li L, et al: Prognostic
factors of patients with ovarian yolk sac tumors: a study in
Chinese patients. Onkologie. 31:679–684. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sell A, Sogaard H and Norgaard-Pedersen B:
Serum alpha-fetoprotein as a marker for the effect of
post-operative radiation therapy and/or chemotherapy in eight cases
of ovarian endodermal sinus tumour. Int J Cancer. 18:574–580. 1976.
View Article : Google Scholar
|
|
12
|
Talerman A, Haije WG and Baggerman L:
Serum alpha fetoprotein (AFP) in diagnosis and management of
endodermal sinus (yolk sac) tumor and mixed germ cell tumor of the
ovary. Cancer. 41:272–278. 1978. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sessa C, Bonazzi C, Landoni F, Pecorelli
S, Sartori E and Mangioni C: Cisplatin, vinblastine, and bleomycin
combination chemotherapy in endodermal sinus tumor of the ovary.
Obstet Gynecol. 70:220–224. 1987.PubMed/NCBI
|
|
14
|
Sasaki H, Furusato M, Teshima S, et al:
Prognostic significance of histopathological subtypes in stage I
pure yolk sac tumour of the ovary. Br J Cancer. 69:529–536. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nogales FF, Bergeron C, Carvia RE, Alvaro
T and Fulwood HR: Ovarian endometrioid tumors with yolk sac tumor
component, an unusual form of ovarian neoplasm. Analysis of six
cases. Am J Surg Pathol. 20:1056–1066. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Davidoff AM, Hebra A, Bunin N, Shochat SJ
and Schnaufer L: Endodermal sinus tumor in children. J Pediatr
Surg. 31:1075–1079. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chen RJ, Huang SC, Chow SN, Hsieh CY, Lin
MC and Hsu HC: Influence of chemotherapy or liver metastasis on the
immunoelectrophoretic discrimination of alpha-fetoprotein from yolk
sac tumor. Int J Gynecol Cancer. 7:486–489. 1997. View Article : Google Scholar
|
|
18
|
Umezu T, Kajiyama H, Terauchi M, et al:
Long-term outcome and prognostic factors for yolk sac tumor of the
ovary. Nagoya J Med Sci. 70:29–34. 2008.PubMed/NCBI
|
|
19
|
Whiting PF, Rutjes AW, Westwood ME, et al:
QUADAS-2: a revised tool for the quality assessment of diagnostic
accuracy studies. Ann Intern Med. 155:529–536. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Egger M, Davey Smith G, Schneider M and
Minder C: Bias in meta-analysis detected by a simple, graphical
test. BMJ. 315:629–634. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Gershenson DM, Del Junco GD, Copeland LJ
and Rutledge FN: Mixed germ cell tumors of the ovary. Obstet
Gynecol. 64:200–206. 1984.PubMed/NCBI
|
|
22
|
Morris HH, La Vecchia C and Draper GJ:
Endodermal sinus tumor and embryonal carcinoma of the ovary in
children. Gynecol Oncol. 21:7–17. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Bakri YN, Ezzat A, Akhtar, Dohami and
Zahrani: Malignant germ cell tumors of the ovary. Pregnancy
considerations. Eur J Obstet Gynecol Reprod Biol. 90:87–91. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Low JJ, Perrin LC, Crandon AJ and Hacker
NF: Conservative surgery to preserve ovarian function in patients
with malignant ovarian germ cell tumors. A review of 74 cases.
Cancer. 89:391–398. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Williams S, Blessing JA, Liao SY, Ball H
and Hanjani P: Adjuvant therapy of ovarian germ cell tumors with
cisplatin, etoposide, and bleomycin: a trial of the Gynecologic
Oncology Group. J Clin Oncol. 12:701–706. 1994.PubMed/NCBI
|
|
26
|
Gershenson DM, Morris M, Cangir A, et al:
Treatment of malignant germ cell tumors of the ovary with
bleomycin, etoposide, and cisplatin. J Clin Oncol. 8:715–720.
1990.PubMed/NCBI
|
|
27
|
Cicin I, Saip P, Guney N, et al: Yolk sac
tumours of the ovary: evaluation of clinicopathological features
and prognostic factors. Eur J Obstet Gynecol Reprod Biol.
146:210–214. 2009. View Article : Google Scholar : PubMed/NCBI
|