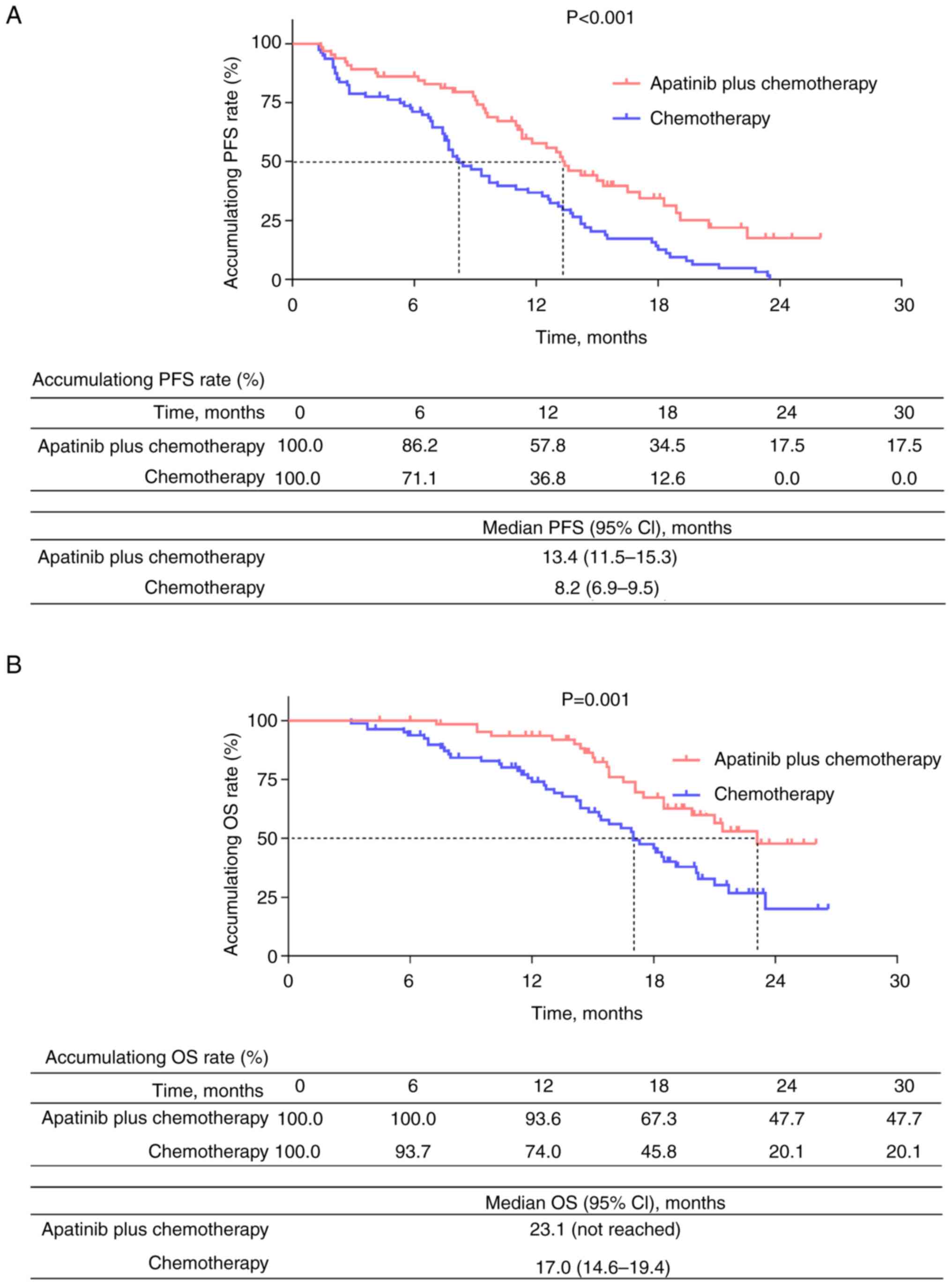
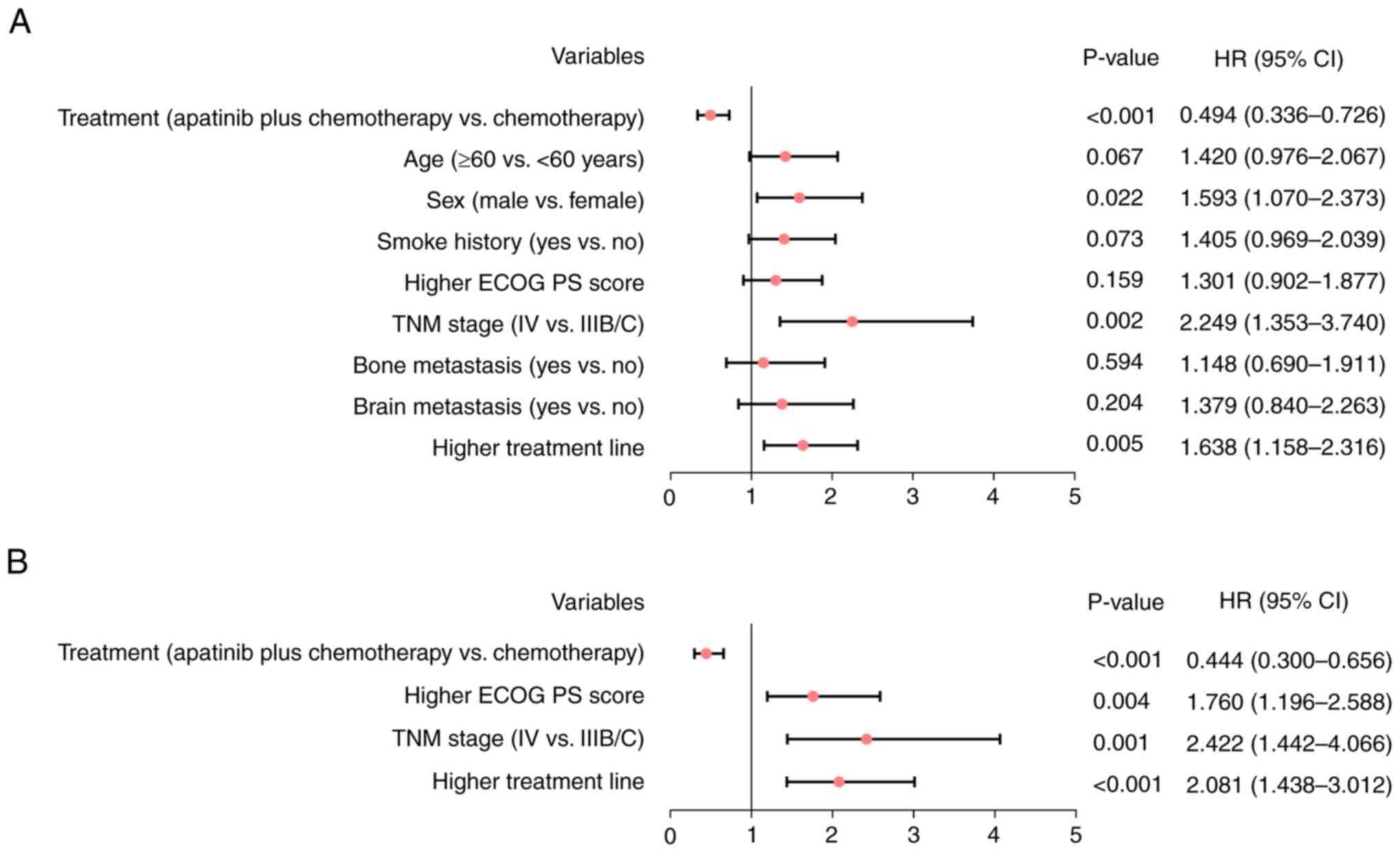
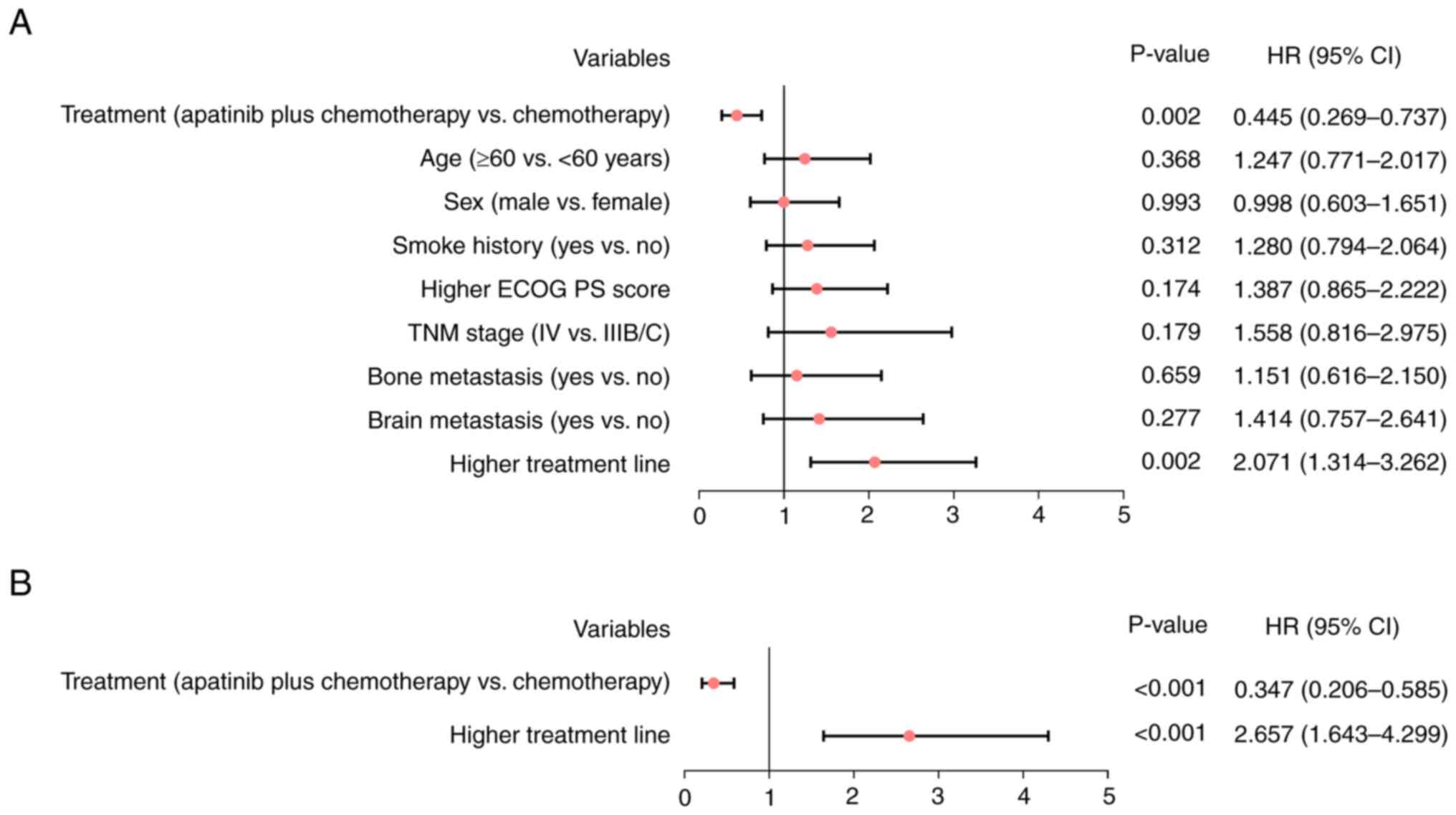
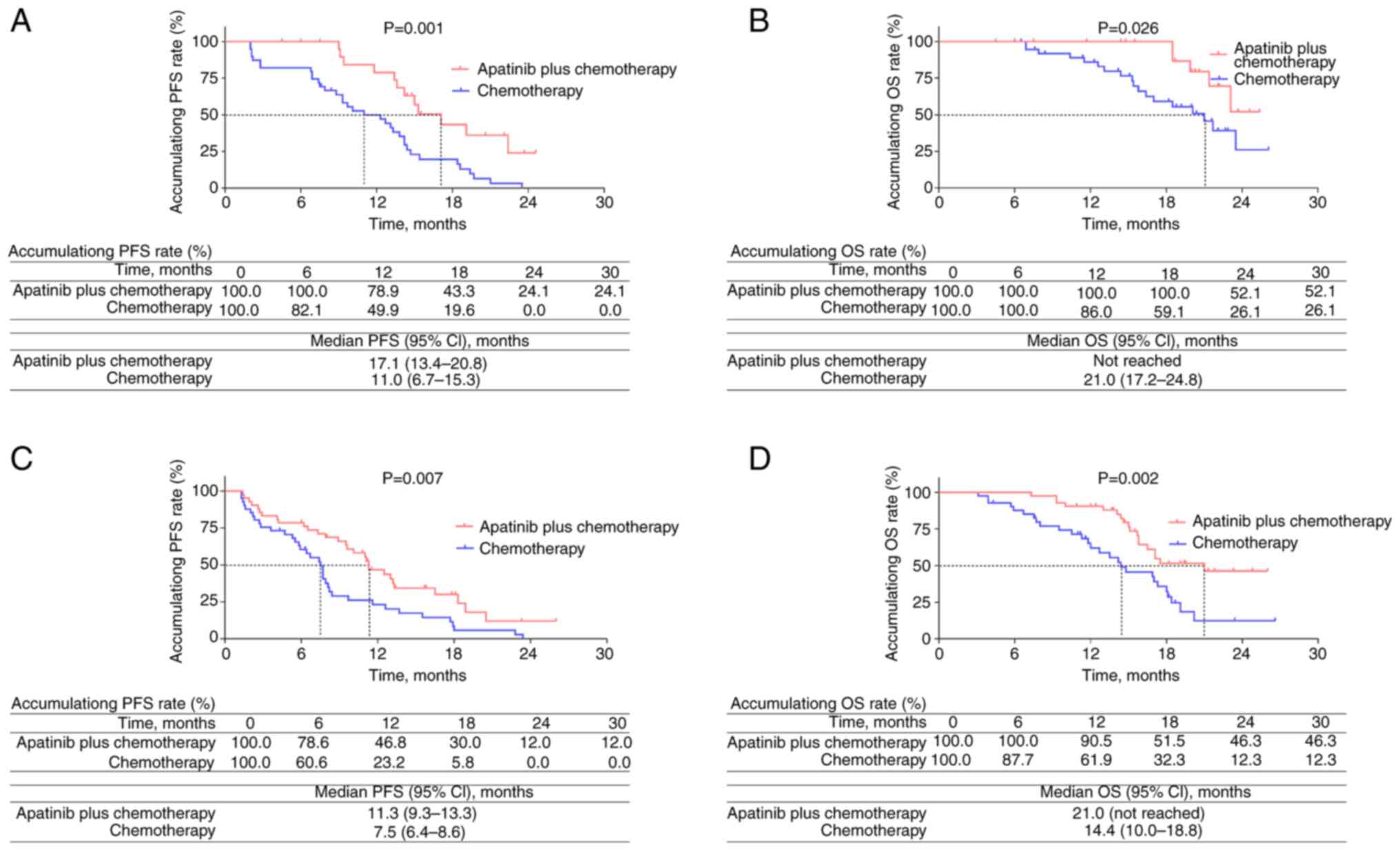
|
1
|
Seguin L, Durandy M and Feral CC: Lung
adenocarcinoma tumor origin: A guide for personalized medicine.
Cancers (Basel). 14:17592022. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chen Z, Fillmore CM, Hammerman PS, Kim CF
and Wong KK: Non-small-cell lung cancers: A heterogeneous set of
diseases. Nat Rev Cancer. 14:535–546. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Melocchi V, Dama E, Mazzarelli F, Cuttano
R, Colangelo T, Di Candia L, Lugli E, Veronesi G, Pelosi G,
Ferretti GM, et al: Aggressive early-stage lung adenocarcinoma is
characterized by epithelial cell plasticity with acquirement of
stem-like traits and immune evasion phenotype. Oncogene.
40:4980–4991. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tan AC and Tan DSW: Targeted therapies for
lung cancer patients with oncogenic driver molecular alterations. J
Clin Oncol. 40:611–625. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Karimi N and Moghaddam SJ: KRAS-Mutant
lung cancer: Targeting molecular and immunologic pathways,
therapeutic advantages and restrictions. Cells. 12:7492023.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kim SY, Kim SM, Lim S, Lee JY, Choi SJ,
Yang SD, Yun MR, Kim CG, Gu SR, Park C, et al: Modeling clinical
responses to targeted therapies by patient-derived organoids of
advanced lung adenocarcinoma. Clin Cancer Res. 27:4397–4409. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lu Y, Fan Z, Zhu SJ, Huang X, Zhuang Z, Li
Y, Deng Z, Gao L, Hong X, Zhang T, et al: A new ALK inhibitor
overcomes resistance to first- and second-generation inhibitors in
NSCLC. EMBO Mol Med. 14:e142962022. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Rusdi F, Iskandar H, Hardjianti T, Bakri
S, Udaya W, Kasim H, Arief E and Seweng A: The one year survival
rate of lung adenocarcinoma patients treated with chemotherapy or
targeted therapy. Enfermería Clínica. 30:456–460. 2020. View Article : Google Scholar
|
|
9
|
Lopez-Chavez A, Thomas A, Rajan A, Raffeld
M, Morrow B, Kelly R, Carter CA, Guha U, Killian K, Lau CC, et al:
Molecular profiling and targeted therapy for advanced thoracic
malignancies: A biomarker-derived, multiarm, multihistology phase
II basket trial. J Clin Oncol. 33:1000–1007. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cui Y, Fang W, Li C, Tang K, Zhang J, Lei
Y, He W, Peng S, Kuang M, Zhang H, et al: Development and
validation of a novel signature to predict overall survival in
‘Driver Gene-negative’ Lung Adenocarcinoma (LUAD): Results of a
multicenter study. Clin Cancer Res. 25:1546–1556. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Gao X, Chen M, Liu X, Shi Y, Liang H, Zhou
Q, Zhao J, Pan R, Zhong W, Xu Y and Wang M: Prognostic factors and
survival benefits of antitumor treatments for advanced non-small
cell lung cancer patients with central nervous system metastasis
with or without driver genes: A Chinese single-center cohort study.
Front Oncol. 12:8795542022. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hames ML, Chen H, Iams W, Aston J, Lovly
CM and Horn L: Correlation between KRAS mutation status and
response to chemotherapy in patients with advanced non-small cell
lung cancer☆. Lung Cancer. 92:29–34. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Scott LJ: Apatinib: A review in advanced
gastric cancer and other advanced cancers. Drugs. 78:747–758. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cui YJ, Liu J, Liu MM and Zhang HZ:
Observation on the clinical effect of apatinib combined with
chemotherapy in the treatment of advanced non-small cell lung
cancer. Pak J Med Sci. 37:1036–1041. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hu R, Li T, Hui K, Chen Z, Wang N, Wu X,
Ge L and Zhou L: Apatinib sensitizes chemoresistant NSCLC cells to
doxetaxel via regulating autophagy and enhances the therapeutic
efficacy in advanced and refractory/recurrent NSCLC. Mol Med Rep.
22:3935–3943. 2020.PubMed/NCBI
|
|
16
|
Yu Z, Cai X, Xu Z, He Z, Lai J, Wang W,
Zhang J, Kong W, Huang X, Chen Y, et al: Apatinib plus chemotherapy
as a second-line treatment in unresectable non-small cell lung
carcinoma: A randomized, controlled, multicenter clinical trial.
Oncologist. 25:e1640–e1649. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cainap C, Balacescu O, Cainap SS and Pop
LA: Next generation sequencing technology in lung cancer diagnosis.
Biology (Basel). 10:8642021.PubMed/NCBI
|
|
18
|
Chansky K, Detterbeck FC, Nicholson AG,
Rusch VW, Vallières E, Groome P, Kennedy C, Krasnik M, Peake M,
Shemanski L, et al: The IASLC lung cancer staging project: External
Validation of the Revision of the TNM Stage Groupings in the Eighth
Edition of the TNM Classification of Lung Cancer. J Thorac Oncol.
12:1109–1121. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ettinger DS, Wood DE, Aisner DL, Akerley
W, Bauman J, Chirieac LR, D'Amico TA, DeCamp MM, Dilling TJ,
Dobelbower M, et al: Non-Small cell lung cancer, version 5.2017,
NCCN clinical practice guidelines in oncology. J Natl Compr Canc
Netw. 15:504–535. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Freites-Martinez A, Santana N,
Arias-Santiago S and Viera A: Using the common terminology criteria
for adverse events (CTCAE – Version 5.0) to Evaluate the severity
of adverse events of anticancer therapies. Actas Dermosifiliogr
(Engl Ed). 112:90–92. 2021.(In English, Spanish). View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Azam F, Latif MF, Farooq A, Tirmazy SH,
AlShahrani S, Bashir S and Bukhari N: Performance status assessment
by using ECOG (Eastern Cooperative Oncology Group) Score for cancer
patients by oncology healthcare professionals. Case Rep Oncol.
12:728–736. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Jiang Q, Zhang NL, Ma DY, Tan BX, Hu X and
Fang XD: Efficacy and safety of apatinib plus docetaxel as the
second or above line treatment in advanced nonsquamous NSCLC: A
multi center prospective study. Medicine (Baltimore).
98:e160652019. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Xu J, Liu X, Yang S and Shi Y: Efficacy,
safety, and prognostic factors of apatinib plus platinum doublet
chemotherapy in advanced non-small cell lung cancer. J Cancer Res
Ther. 18:1425–1431. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Huang MP, Gu SZ, Huang B, Li GW, Xiong ZP,
Tang T and Zeng SN: Apatinib inhibits angiogenesis in intrahepatic
cholangiocarcinoma by regulating the vascular endothelial growth
factor receptor-2/signal transducer and activator of transcription
factor 3/hypoxia inducible factor 1 subunit alpha signaling axis.
Pharmacology. 106:509–519. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Song J, Guan Z, Song C, Li M, Gao Z and
Zhao Y: Apatinib suppresses the migration, invasion and
angiogenesis of hepatocellular carcinoma cells by blocking VEGF and
PI3K/AKT signaling pathways. Mol Med Rep. 23:4292021. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Hu Y, Jing J, Shi Y, Zhang P, Dong D, Wu
Y, Dong X, Li E and Fan Y: Apatinib inhibits pancreatic cancer
growth, migration and invasion through the PI3K/AKT and ERK1/2/MAPK
pathways. Transl Cancer Res. 10:3306–3316. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Xie C, Zhou X, Liang C, Li X, Ge M, Chen
Y, Yin J, Zhu J and Zhong C: Apatinib triggers autophagic and
apoptotic cell death via VEGFR2/STAT3/PD-L1 and ROS/Nrf2/p62
signaling in lung cancer. J Exp Clin Cancer Res. 40:2662021.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Liu ZL, Jin BJ, Cheng CG, Zhang FX, Wang
SW, Wang Y and Wu B: Apatinib resensitizes cisplatin-resistant
non-small cell lung carcinoma A549 cell through reversing multidrug
resistance and suppressing ERK signaling pathway. Eur Rev Med
Pharmacol Sci. 21:5370–5377. 2017.PubMed/NCBI
|
|
30
|
Zhang X, Xiong Y, Xia Q, Wu F, Liu L, Zhou
Y, Zeng L, Zhou C, Xia C, Jiang W, et al: Efficacy and safety of
apatinib plus vinorelbine in patients with wild-type advanced
non-small cell lung cancer after second-line treatment failure: A
nonrandomized clinical trial. JAMA Netw Open. 3:e2012262020.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wang W, He Q, Li C, Zhuang C, Zhang H,
Wang Q, Fan X, Qi M, Sun R and Yu J: Research on the mechanism and
prevention of hypertension caused by apatinib through the RhoA/ROCK
signaling pathway in a mouse model of gastric cancer. Front
Cardiovasc Med. 9:8738292022. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Cohen JB, Brown NJ, Brown SA, Dent S, van
Dorst DCH, Herrmann SM, Lang NN, Oudit GY, Touyz RM; American Heart
Association Council on Hypertension, ; et al: Cancer
therapy-related hypertension: A scientific statement from the
american heart association. Hypertension. 80:e46–e57. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Camarda N, Travers R, Yang VK, London C
and Jaffe IZ: VEGF receptor inhibitor-induced hypertension:
Emerging mechanisms and clinical implications. Curr Oncol Rep.
24:463–474. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Caletti S, Paini A, Coschignano MA, De
Ciuceis C, Nardin M, Zulli R, Muiesan ML, Salvetti M and Rizzoni D:
Management of VEGF-Targeted therapy-induced hypertension. Curr
Hypertens Rep. 20:682018. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Maitland ML, Bakris GL, Black HR, Chen HX,
Durand JB, Elliott WJ, Ivy SP, Leier CV, Lindenfeld J, Liu G, et
al: Initial assessment, surveillance, and management of blood
pressure in patients receiving vascular endothelial growth factor
signaling pathway inhibitors. J Natl Cancer Inst. 102:596–604.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Patel S, Dushenkov A, Jungsuwadee P,
Krishnaswami A and Barac A: Team-Based approach to management of
hypertension associated with angiogenesis inhibitors. J Cardiovasc
Transl Res. 13:463–477. 2020. View Article : Google Scholar : PubMed/NCBI
|