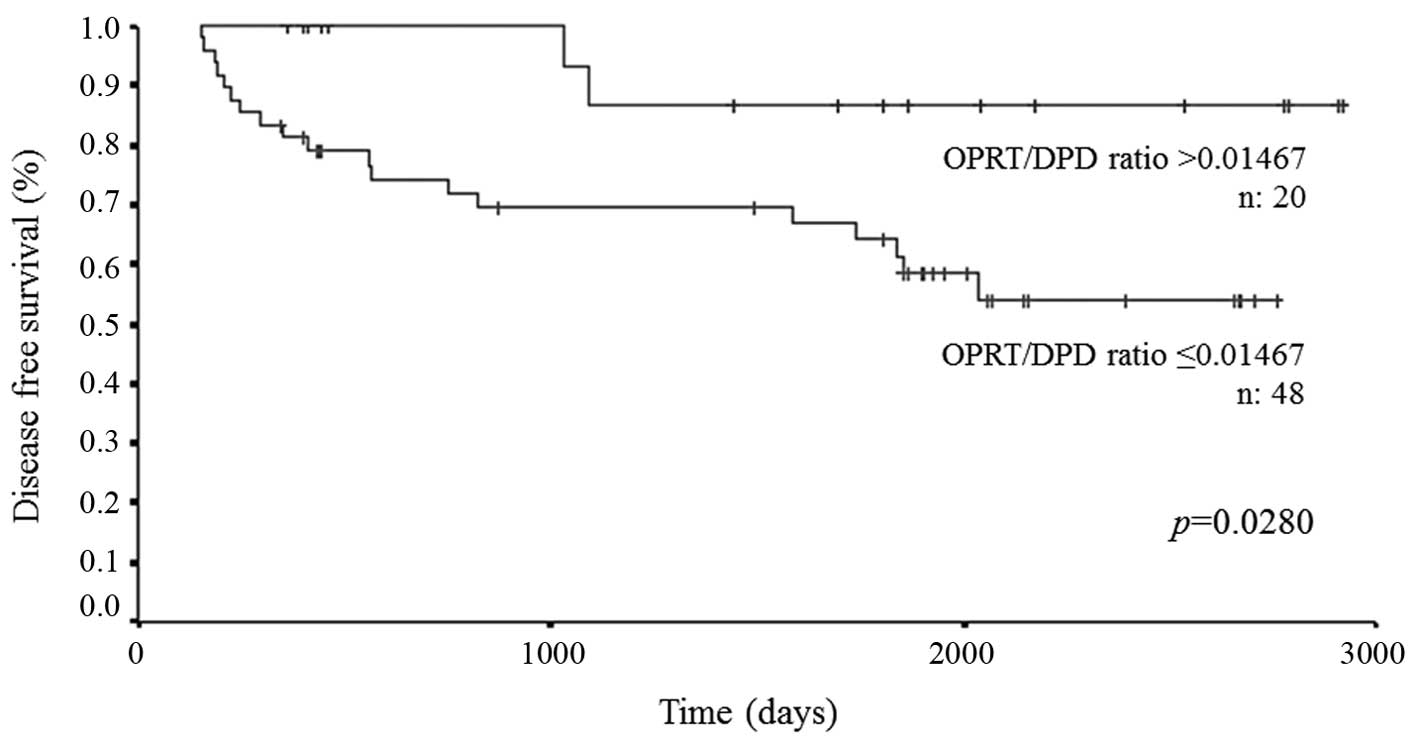
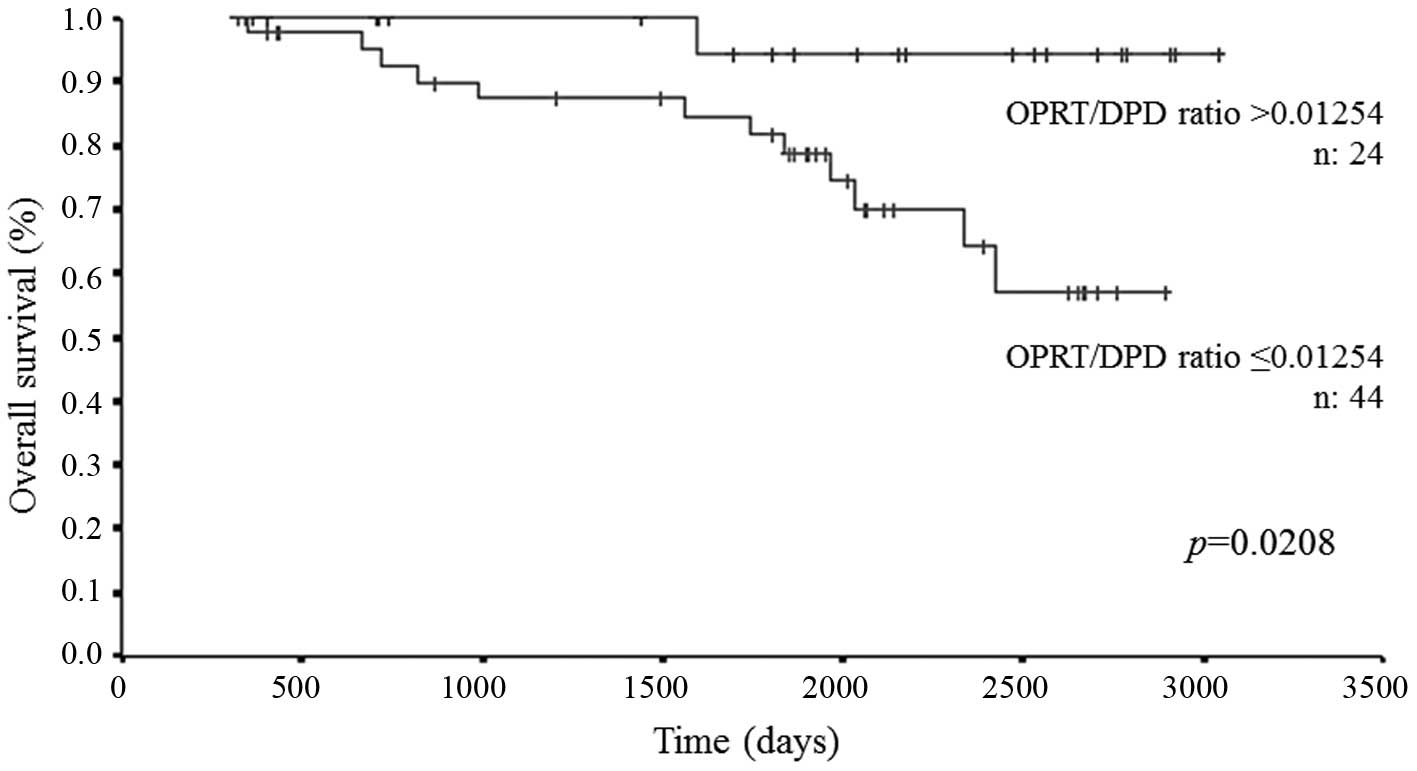
|
1
|
Bang YJ, Van Cutsem E, Feyereislova A, et
al: Trastuzumab in combination with chemotherapy versus
chemotherapy alone for treatment of HER2-positive advanced gastric
or gastro-oesophageal junction cancer (ToGA): a phase 3,
open-label, randomised controlled trial. Lancet. 376:687–697. 2010.
View Article : Google Scholar
|
|
2
|
Lièvre A, Bachet JB, Le Corre D, et al:
KRAS mutation status is predictive of response to cetuximab therapy
in colorectal cancer. Cancer Res. 66:3992–3995. 2006.PubMed/NCBI
|
|
3
|
Karapetis CS, Khambata-Ford S, Jonker DJ,
et al: K-ras mutations and benefit from cetuximab in advanced
colorectal cancer. N Engl J Med. 359:1757–1765. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Van Cutsem E, Köhne CH, Hitre E, et al:
Cetuximab and chemotherapy as initial treatment for metastatic
colorectal cancer. N Engl J Med. 360:1408–1417. 2009.PubMed/NCBI
|
|
5
|
Bokemeyer C, Bondarenko I, Makhson A, et
al: Fluorouracil, leucovorin, and oxaliplatin with and without
cetuximab in the first-line treatment of metastatic colorectal
cancer. J Clin Oncol. 27:663–671. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Folprecht G, Gruenberger T, Bechstein WO,
et al: Tumour response and secondary resectability of colorectal
liver metastases following neoadjuvant chemotherapy with cetuximab:
the CELIM randomised phase 2 trial. Lancet Oncol. 11:38–47. 2010.
View Article : Google Scholar
|
|
7
|
Hecht JR, Patnaik A, Berlin J, et al:
Panitumumab monotherapy in patients with previously treated
metastatic colorectal cancer. Cancer. 110:980–988. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Van Cutsem E, Peeters M, Siena S, et al:
Open-label phase III trial of panitumumab plus best supportive care
compared with best supportive care alone in patients with
chemotherapy-refractory metastatic colorectal cancer. J Clin Oncol.
25:1658–1664. 2007.PubMed/NCBI
|
|
9
|
Siena S, Peeters M, Van Cutsem E, et al:
Association of progression-free survival with patient-reported
outcomes and survival: results from a randomised phase 3 trial of
panitumumab. Br J Cancer. 97:1469–1474. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Weiner LM, Belldegrun AS, Crawford J, et
al: Dose and schedule study of panitumumab monotherapy in patients
with advanced solid malignancies. Clin Cancer Res. 14:502–508.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Amado RG, Wolf M, Peeters M, et al:
Wild-type KRAS is required for panitumumab efficacy in patients
with metastatic colorectal cancer. J Clin Oncol. 26:1626–1634.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Muro K, Yoshino T, Doi T, et al: A phase 2
clinical trial of panitumumab monotherapy in Japanese patients with
metastatic colorectal cancer. Jpn J Clin Oncol. 39:321–326. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lacouture ME, Mitchell EP, Piperdi B, et
al: Skin toxicity evaluation protocol with panitumumab (STEPP), a
phase II, open-label, randomized trial evaluating the impact of a
pre-emptive skin treatment regimen on skin toxicities and quality
of life in patients with metastatic colorectal cancer. J Clin
Oncol. 28:1351–1357. 2010. View Article : Google Scholar
|
|
14
|
Ochiai T, Sugitani M, Nishimura K, et al:
Correlation between 5-fluorouracil (5-FU) sensitivity as measured
by collagen gel droplet embedded culture drug sensitivity test
(CD-DST) and expression of orotate phosphoribosyl transferase
(OPRT), thymidylate synthase (TS), and dihydropyrimidine
dehydrogenase (DPD) in colorectal cancer. Gan To Kagaku Ryoho.
28:661–667. 2001.(In Japanese).
|
|
15
|
Ochiai T, Sugitani M, Nishimura K, et al:
Prognostic impact of orotate phosphoribosyl transferase (OPRT)
activity in resectable colorectal cancers (CRC) treated by
5-FU-based adjuvant chemotherapy. ASCO abstract. J Clin Oncol.
22:35742004.PubMed/NCBI
|
|
16
|
Ochiai1 T, Nishimura K, Noguchi1 H, et al:
Prognostic impact of orotate phosphoribosyl transferase among
5-fluorouracil metabolic enzymes in resectable colorectal cancers
treated by oral 5-fluorouracil-based adjuvant chemotherapy. Int J
Cancer. 118:3084–3088. 2006. View Article : Google Scholar
|
|
17
|
Kinoshita M, Kodera Y, Hibi K, et al: Gene
expression profile of 5-fluorouracil metabolic enzymes in primary
colorectal cancer: potential as predictive parameters for response
to fluorouracil-based chemotherapy. Anticancer Res. 27:851–856.
2007.
|
|
18
|
Tokunaga Y, Sasaki H and Saito T: Clinical
role of orotate phosphoribosyl transferase and dihydropyrimidine
dehydrogenase in colorectal cancer treated with postoperative
fluoropyrimidine. Surgery. 141:346–353. 2007. View Article : Google Scholar
|
|
19
|
Ishikawa M, Miyauchi T and Kashiwagi Y:
Clinical implications of thymidylate synthetase, dihydropyrimidine
dehydrogenase and orotate phosphoribosyl transferase activity
levels in colorectal carcinoma following radical resection and
administration of adjuvant 5-FU chemotherapy. BMC Cancer.
8:1882008. View Article : Google Scholar
|
|
20
|
Yamada H, Iinuma H and Watanabe T:
Prognostic value of 5-fluorouracil metabolic enzyme genes in Dukes’
stage B and C colorectal cancer patients treated with oral
5-fluorouracil-based adjuvant chemotherapy. Oncol Rep. 19:729–735.
2008.PubMed/NCBI
|
|
21
|
Koopman M, Venderbosch S, van Tinteren H,
et al: Predictive and prognostic markers for the outcome of
chemotherapy in advanced colorectal cancer, a retrospective
analysis of the phase III randomised CAIRO study. Eur J Cancer.
45:1999–2006. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Koizumi W, Tanabe S, Azuma M, et al:
Impacts of fluorouracil-metabolizing enzymes on the outcomes of
patients treated with S-1 alone or S-1 plus cisplatin for
first-line treatment of advanced gastric cancer. Int J Cancer.
126:162–170. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Jeung HC, Rha SY, Shin SJ, et al:
Predictive values of 5-fluorouracil pathway genes for S-1 treatment
in patients with advanced gastric cancer. Anticancer Drugs.
22:801–810. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Laskin JD, Evans RM, Slocum HK, Burke D
and Hakala MT: Basis for natural variation in sensitivity to
5-fluorouracil in mouse and human cells in culture. Cancer Res.
39:383–390. 1979.PubMed/NCBI
|
|
25
|
Diasio RB, Beavers TL and Carpenter JT:
Familial deficiency of dihydropyridine dehydrogenase: Biochemical
basis for familial pyrrimidinemia and severe 5-fluorouracil-
induced toxicity. J Clin Invest. 81:47–51. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Harris BE, Song R, Soong SJ and Diasio RB:
Relationship between dihydropyrimidine dehydrogenase activity and
plasma 5-fluorouracil levels with evidence for circadian variation
of enzyme activity and plasma drug levels in cancer patients
receiving 5-fluorouracil by protracted continuous infusion. Cancer
Res. 50:197–201. 1990.
|
|
27
|
Miller RG and Siegmund D: Maximally
selected Chi square statistics. Biometrics. 38:1011–1016. 1982.
View Article : Google Scholar
|
|
28
|
Halpern J: Maximally selected Chi square
statistics for small samples. Biometrics. 38:1017–1023. 1982.
View Article : Google Scholar
|
|
29
|
Yamada Y, Takahari D, Matsumoto H, et al:
Leucovorin, fluorouracil, and oxaliplatin plus bevacizumab versus
S-1 and oxaliplatin plus bevacizumab in patients with metastatic
colorectal cancer (SOFT): an open-label, non-inferiority,
randomised phase 3 trial. Lancet Oncol. 14:1278–1286. 2013.
View Article : Google Scholar
|
|
30
|
Hong YS, Park YS, Lim HY, et al: S-1 plus
oxaliplatin versus capecitabine plus oxaliplatin for first-line
treatment of patients with metastatic colorectal cancer: a
randomised, non-inferiority phase 3 trial. Lancet Oncol.
13:1125–1132. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Mochizuki I, Takiuchi H, Ikejiri K,
Nakamoto Y, et al: Safety of UFT/LV and S-1 as adjuvant therapy for
stage III colon cancer in phase III trial: ACTS-CC trial. Br J
Cancer. 106:1268–1273. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Orimo A, Gupta PB, Sgroi DC, et al:
Stromal fibroblasts present in invasive human breast carcinomas
promote tumor growth and angiogenesis through elevated SDF-1/CXCL12
secretion. Cell. 121:335–348. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Stuelten CH, DaCosta Byfield SD, Arany PR,
et al: Breast cancer cells induce stromal fibroblasts to express
MMP-9 via secretion of TNF-alpha and TGF-beta. J Cell Sci.
118:2143–2153. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Gaggioli C, Hooper S, Hidalgo-Carcedo C,
et al: Fibroblast-led collective invasion of carcinoma cells with
differing roles for RhoGTPases in leading and following cells. Nat
Cell Biol. 9:1392–1400. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
35
|
Allavena P, Sica A, Solinas G, Porta C and
Mantovani A: The inflammatory micro-environment in tumor
progression: the role of tumor-associated macrophages. Crit Rev
Oncol Hematol. 66:1–9. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ricci-Vitiani L, Pallini R, Biffoni M, et
al: Tumour vascularization via endothelial differentiation of
glioblastoma stem-like cells. Nature. 468:824–828. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Erreni M, Mantovani A and Allavena P:
Tumor-associated macrophages (TAM) and inflammation in colorectal
cancer. Cancer Microenviron. 4:141–154. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Hoshino A, Ishii G, Ito T, et al:
Podoplanin-positive fibroblasts enhance lung adenocarcinoma tumor
formation: podoplanin in fibroblast functions for tumor
progression. Cancer Res. 71:4769–4779. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Nagano H, Ichikawa W, Simizu M, et al:
Thymidylate synthase and dihydropyrimidine dehydrogenase gene
expression in colorectal cancer using the Danenberg tumor profile
method. Gan To Kagaku Ryoho. 31:889–892. 2004.(In Japanese).
|