Introduction
The incidence of hepatocellular cancer (HCC) ranks
fifth among all malignant tumors worldwide. Moreover, HCC is the
third most common cause of cancer-related mortality worldwide
(1–4), with more than 600,000 individuals
succumbing to the disease each year. Similar to other solid tumors,
surgical resection is the main treatment option for HCC (5), yet the high frequency of local
recurrence and distant metastasis remain the largest obstacles to
the survival of post-operative HCC patients (6). Due to the difficulty in the early
diagnosis of HCC, most patients are treated during advanced or
late-stage disease, and only a small percentage of patients have
the chance of radical treatment. Therefore, the development of new
approaches for clinical therapy of liver cancer is of utmost
importance.
Embelin is a small-molecule inhibitor extracted from
plants of the Myrsinaceae family. It is a polyphenolic compound
that inhibits the X-linked inhibitor of apoptosis protein (XIAP)
through bonding with the Smac bonding site in the BIR3 domain in
the XIAP protein molecule (7,8).
Previous studies have demonstrated that embelin has
anti-inflammatory and anti-oxidative effects (9–11).
Embelin has been found to suppress the growth of various types of
tumor cells including breast, colon, prostate and pancreatic cancer
cells. However, there have been no reports on whether embelin
inhibits the growth of human hepatocellular carcinoma cells.
Moreover, the detailed mechanism of embelin against tumors remains
unknown.
Studies have demonstrated that apoptosis of liver
cancer cells is closely associated with the mitochondrial pathway
(16–18). Our previous study found that
embelin promoted changes in the expression of the Bax and Bcl-2
proteins. The migration of Bax and Bcl-2 proteins resulted in
changes in mitochondrial membrane potential, which in turn released
cytochrome c and activated the caspase family, resulting in
apoptosis (19). A previous study
also demonstrated that the Bax and Bcl-2 proteins are two important
targets of anticancer drugs (20).
The present study was designed to investigate the
impacts of embelin in vitro on the apoptosis of HepG2 human
hepatocellular carcinoma cells and the cell cycle, and to explore
the embelin-induced apoptosis signaling pathway. We found that via
the regulation of the Bax and Bcl-2 proteins, embelin releases
cytochrome c and activates the caspase family to induce the
apoptosis of HepG2 cells.
Materials and methods
Materials and chemicals
Embelin (≥99% purity), propidium iodide (PI), JC-1
and MTT were purchased from Sigma Inc. (St. Louis, MO, USA). The
Annexin V-FITC/PI kit was purchased from BD Biosciences (Franklin
Lakes, NJ, USA). All antibodies were from Santa Cruz (Santa Cruz,
CA, USA).
Cell culture
HepG2 human hepatocellular carcinoma cells were from
American Type Culture Collection (ATCC). Cells after cell passage
were inoculated in RPMI-1640 culture medium (Gibco-BRL, NY, USA)
containing 10% fetal calf serum (Hyclone Laboratories, UT, USA),
100 U/ml penicillin and 100 U/ml streptomycin. The cells were then
cultured in an incubator containing 5% CO2 and 95%
oxygen at 37°C.
Cell viability
HepG2 cells in logarithmic growth phase were
inoculated in a 96-well culture plate with a density of
1×105 cells/ml. After the cells grew to adherence,
different doses of embelin were administered to various groups with
6 duplicate wells for each concentration. TAa negative control
group without embelin treatment was established. All the cells were
placed in a 5% CO2 incubator for further culture for 24,
48 and 72 h prior to color reaction. Each well was supplemented
with 20 μl of MTT (5 mg/ml) and cultured in a CO2
incubator for 4 h before the culture solution was disposed of. DMSO
(150 μl)was added to each well at room temperature and oscillation
was carried out for 10 min and the OD values (A570nm) of each well
were measured with a microplate reader.
Evaluation of apoptosis
During the early stage of apoptosis,
phosphatidylserine was exposed outside the membrane from the
interior of the cell membrane due to the lack of symmetry of the
cell membrane. Trypsin (0.25%) was digested to collect the cells of
all groups, and the cell density was adjusted to 1×106
cells/ml. Annexin V-FITC (5 μl), and 5 μl of PI were added,
respectively, for dyeing (30 min at 4°C) before analysis with flow
cytometry.
Analysis of the cell cycle by flow
cytometry
The cells of all groups were collected and the
trypsin method was used. The cells were washed with PBS 3 times and
fixed at 4°C with 75% cold ethanol overnight. The ethanol was
removed and the cells were washed in PBS 3 times. The cell density
was adjusted to 1×106 cells/ml with a final volume of
100 μl. DNAStain comprehensive dye liquor (500 μl) with RNase at a
final concentration of 50 mg/l, PI at a final concentration of 100
mg/l and Triton X-100 at a final concentration of 1 ml/l was added
and cells were stored at room temperature in a dark place for 30
min before analysis with flow cytometry.
Flow cytometric analysis of mitochondrial
membrane potential
The cell density was adjusted to 1×106
cells/ml, JC-1 dye liquor was added with a final concentration of
10 μg/ml and even mixing was carried out; the cells were cultured
for 30 min at 37°C in a dark place. Analysis with flow cytometry
was performed (BD Biosciences). JC-1 monomer and polymer
fluorescent signals from FL1 and FL2 probes, respectively, were
obtained. FL1-H indicated green fluorescence intensity and FL2-H
indicated red fluorescence intensity. Quantitative analysis was
carried out with Cellquest analysis software.
Western blot assay
The cells of all groups were prepared using the
trypsin method and 2 ml of lysis solution (50 mM of Tris-HCl, 137
mM of Nacl, 10% glycerin, 100 mM of sodium vanadate, 1 mM of PMSF,
10 mg/ml of apotinin, 10 mg/ml of eupeptin, 1% NP-40 and 5 mM of
cocktail; pH 7.4) was added for cell lysis to retrieve the
proteins. Proteins were loaded following the BCA method of
rationing. The proteins were separated with SDS-PAGE. The proteins
were shifted to PVDF membranes using a semidrying method and sealed
with 5% skim mild powder at 4°C overnight. The membranes were
washed with TBST and the first antibody was added at 37°C for
hybrid for 1 h before bleaching with TBST. The second antibody was
added at 37°C for hybridization for 1 h before bleaching with TBST
and completing color reaction for 5 min with autoradiography.
Quantity One was used for optical density value analysis and
measurement. The results are presented as optical density
value/actin optical density value of the samples.
Statistical analysis
SPSS 16.0 statistical software was used for
statistical analysis. Values are shown as the mean ± SD.
Statistical analysis was carried out using the Student’s t-test.
Differences between groups were considered statistically
significant at p<0.05.
Results
Embelin-induced inhibition of HepG2 human
hepatocellular carcinoma cell growth
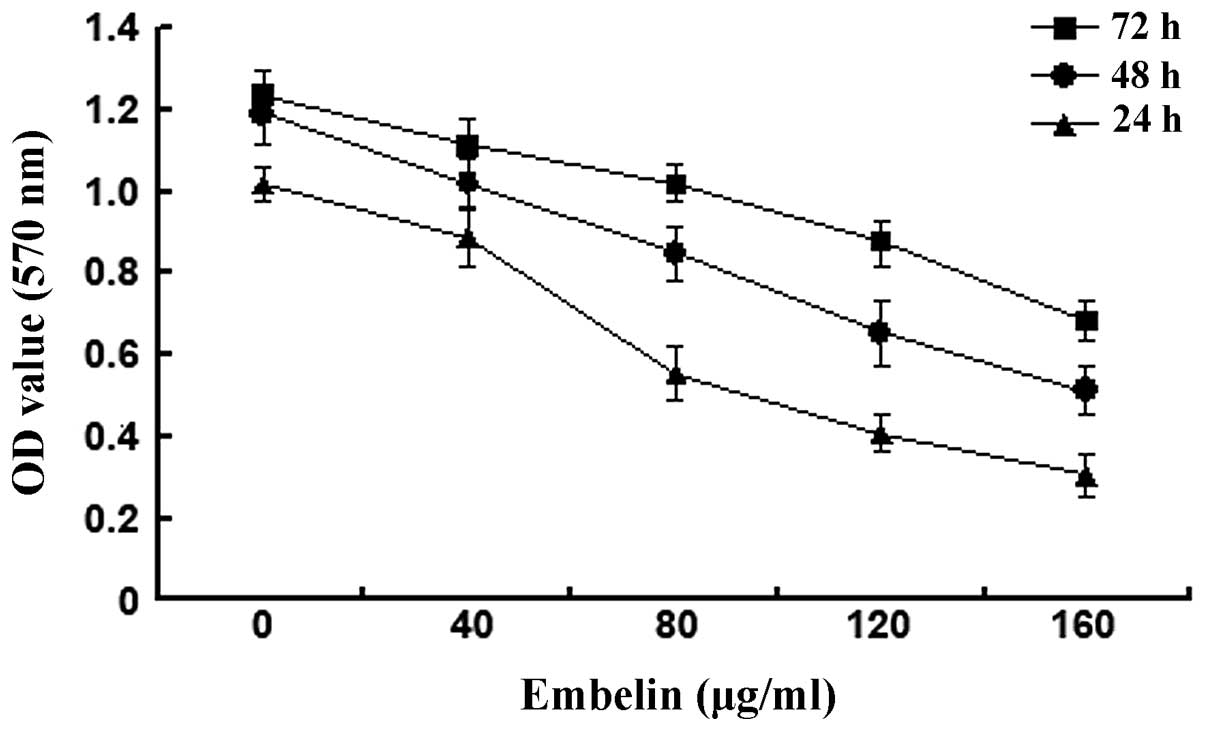
Embelin of different concentrations (0, 40, 80, 120
and 160 μg/ml) was administered to HepG2 cells for 24, 48 and 72 h
before the MTT method was used to determine the cell viability
(Fig. 1). The results showed that
A570 values of HepG2 cells gradually decreased when the
concentration of embelin increased from 40 to 160 μg/ml and the
A570 value decreased the most when the concentration of embelin was
at 120 μg/ml. This suggests that embelin has potent inhibitory
effects on the growth of HepG2 cells.
Embelin-induced apoptosis of HepG2 human
hepatocellular carcinoma cells
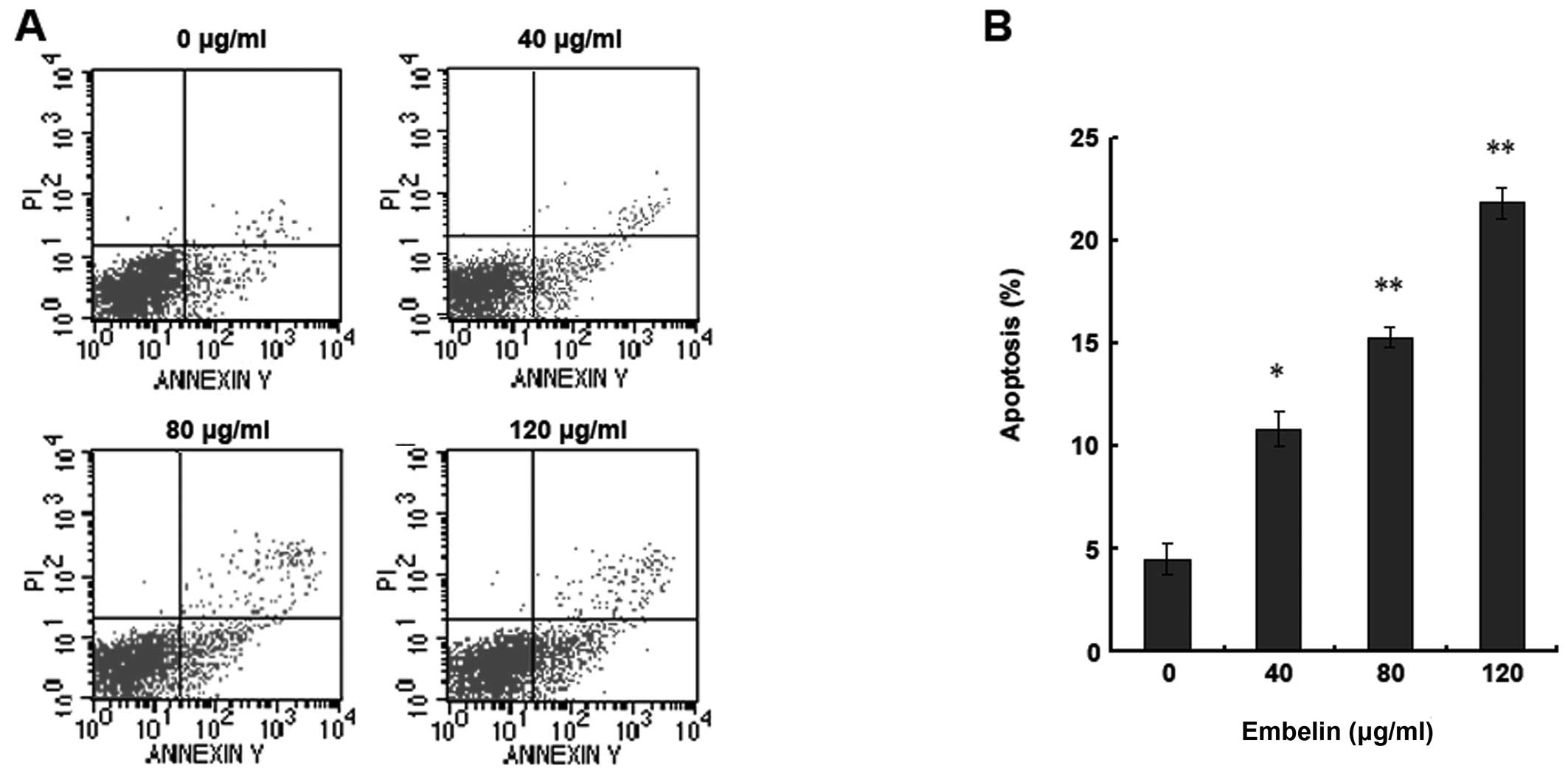
HepG2 human hepatocellular carcinoma cells were
treated with different doses of embelin (0, 40, 80 and 120 μg/ml)
for 48 h and flow cytometry was used to assess the rate of
apoptosis (Fig. 2). Our results
indicate that with the increase of the embelin dose, the quantity
of HepG2 apoptosis rose significantly (Fig. 2A and B). Therefore, embelin induces
apoptosis of HepG2 human hepatocellular carcinoma cells in a
dose-dependent manner.
Embelin induces apoptosis of HepG2 human
hepatocellular carcinoma cells through the mitochondrial
pathway
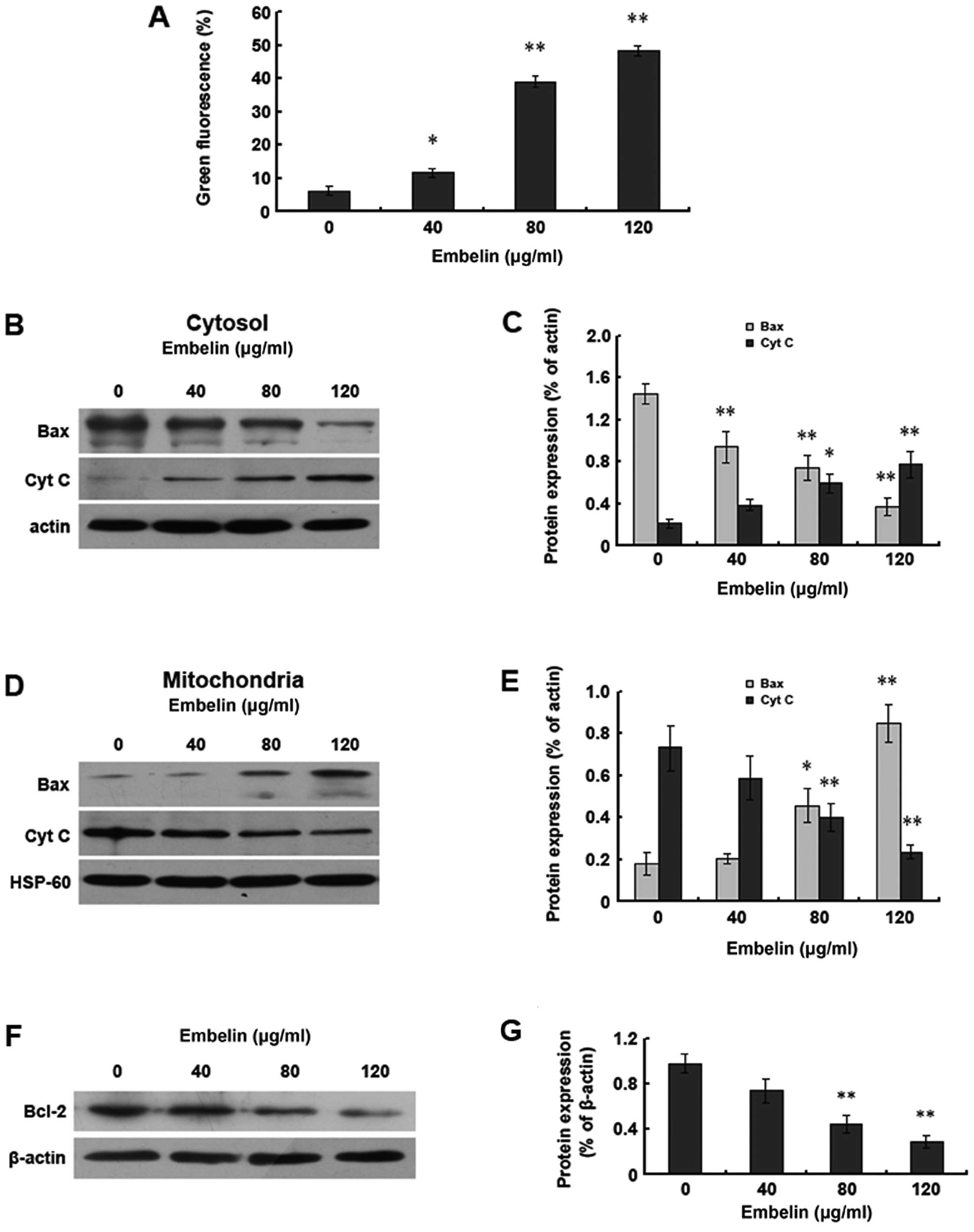
In order to prove the relationship between the
induction of HepG2 cell apoptosis by embelin and the mitochondrial
pathway, JC-1 coloration was used to assess the changes in the
mitochondrial membrane potential. The results demonstrated the
gradual decrease of the mitochondrial membrane potential along with
the increase of the dosage of embelin, with the change of the
mitochondrial membrane potential occurring before apoptosis
(Fig. 3A). Bax gene shift and the
release of cytochrome c are another sign of apoptosis. We
observed the levels of Bax and cytochrome c inside the
cytoplasm and mitochondria using western blot analysis and found
that with the increase of the embelin dosage, the Bax protein
levels inside the cytoplasm decreased whereas the cytochrome
c levels increased. However, the levels of Bax and
cytochrome c inside the mitochondria were just opposite to
those inside the cytoplasm (Fig.
3B–E). We also found that the levels of the Bcl-2 protein
decreased with the increase of the embelin dosage (Fig. 3F and G). Our data demonstrate that
embelin is able to change the mitochondrial membrane potential to
promote the shift of Bax and Bcl-2 as well as the release of
cytochrome c, which results in the apoptosis of HepG2 human
hepatocellular carcinoma cells.
Effect of embelin on the expression
levels of human hepatocellular carcinoma HepG2 apoptosis-related
proteins
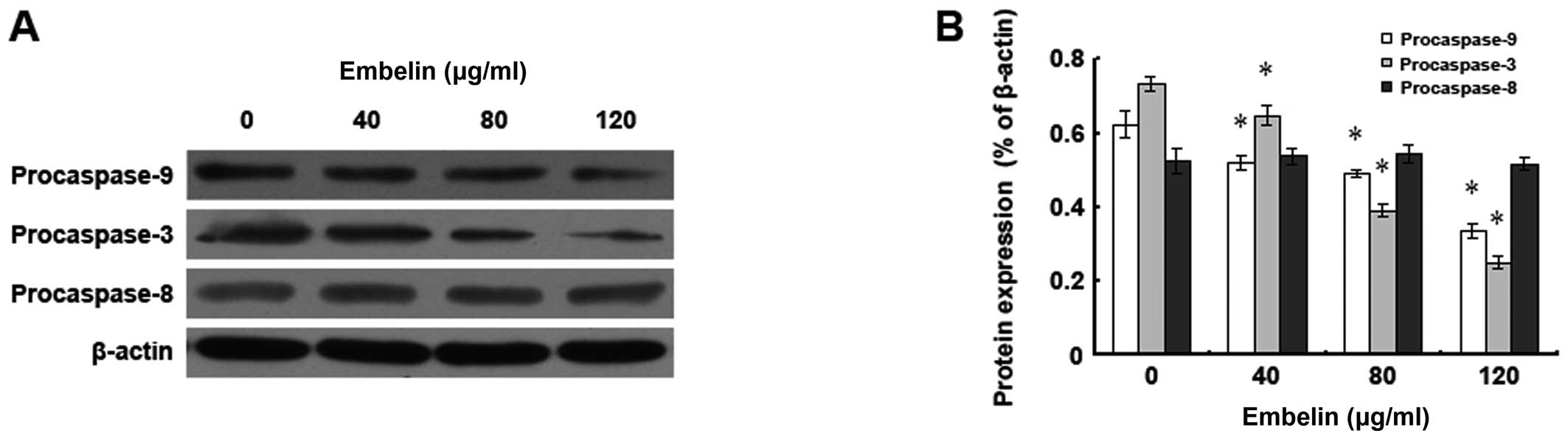
In order to study the impact of embelin on the
expression levels of HepG2 human hepatocellular carcinoma cell
apoptosis-related proteins, different doses of embelin were
administered to HepG2 cells for 48 h, and western blot analysis was
used to examine the expression of the procaspase 3, 8, 9 proteins
(Fig. 4). It was found that after
the treatment with 120 μg/ml of embelin, the expression levels of
both the HepG2 cell procaspase 3 and 9 proteins decreased
significantly, while there was no significant change in the
expression level of the procaspase 8 protein. Our data demonstrate
that the HepG2 embelin-induced apoptosis may be through the
mitochondria-mediated caspase 3 and 9 pathways.
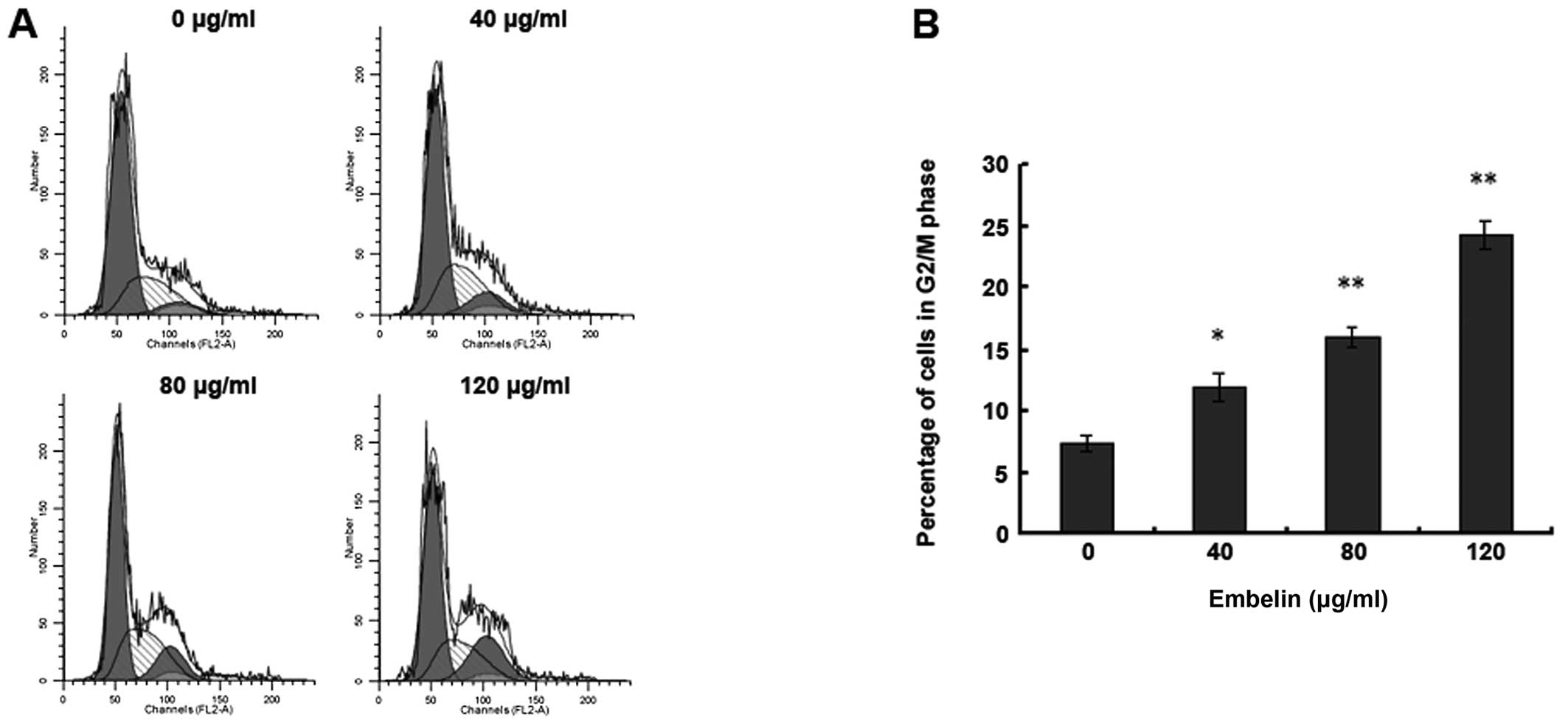
Embelin-induced HepG2 human
hepatocellular carcinoma cell cycle blockade in the G2/M phase
In order to investigate whether embelin effects the
HepG2 human hepatocellular carcinoma cell cycle, we used flow
cytometry for the analysis. The results showed that 48 h after
administering different doses of embelin to HepG2 cells, the HepG2
cells were blocked in the G2/M phase, which was apparently higher
than that of the control group (Fig.
5). This indicates that embelin is able to inhibit the G2/M
phase percentage of HepG2 cells to restrain the hepatocellular
carcinoma cell proliferation.
Discussion
Kerr et al (21) were the first to report on the
concept of apoptosis. Apoptosis is ubiquitous in most of the tumor
cells and plays an important role in the genesis and progression of
tumors (22,23). Previous studies have demonstrated
that antitumor drugs generally inhibit tumors through the induction
of apoptosis of sensitive tumor cells, and their antitumor effects
relate to the interior activation of the apoptosis of the tumor
cells induced by the drugs. Therefore, the induction of apoptosis
to treat tumors has become a new target for the development of
antitumor drugs and constitutes a new direction in the current
research in tumor pharmacology.
Embelin is a small-molecule inhibitor exhibiting
specific inhibition of XIAP that affects the proliferation and
apoptosis of various tumor cells. Embelin has gained much worldwide
attention for its antitumor effects. As shown in previous studies,
embelin inhibits the proliferation of various tumor cells, with
particularly significant effects on breast and pancreatic cancer as
well as on other solid tumor cells (24,15).
Our results support the hypothesis that embelin induces the
apoptosis of human hepatocellular carcinoma cells. Flow cytometry
revealed that after human hepatocellular carcinoma cells were
treated with different doses of emblin for 48 h, the Annexin
V-positive rate of the cells increased in a dose-dependent manner,
indicating that embelin can induce human hepatocellular carcinoma
apoptosis instead of cell death. Embelin is also capable of
restraining the cell cycle alterations of hepatocellular carcinoma
cells to cause blockade of the cell cycle in the G2/M phase so as
to change the cell cycle progress to induce apoptosis.
There are two main signaling pathways to trigger
apoptosis: the endogenous mitochondrial pathway and the exogenous
death receptor pathway (25). Our
research found that after human hepatocellular carcinoma cells were
treated with different doses of emblin for 48 h, Bax and Bcl-2
migrated, the mitochondrial membrane potential decreased, and
cytochrome c was released. These findings indicate that the
induction of human hepatocellular carcinoma cell apoptosis by
embelin is mediated by the mitochondrial pathway. The Bcl-2/Bax
family is the key factor in regulating the endogenous mitochondrial
apoptosis pathway (26,27). With the pro-apoptosis effect, the
Bax gene migrates from the cytoplasm to the mitochondrial outer
membrane, changing the permeability of the mitochondrial outer
membrane to promote the release of cytochrome c from the
mitochondria (28). Moreover, the
Bcl-2 protein can regulate the opening and closing of mPTP. mPTP, a
non-selective pathway spanning over the inner and outer
mitochondrial membrane and the mitochondrial irritation receptor,
is considered to trigger the life or death of the cells (29). Our study found that with embelin
treatment, the Bcl-2 protein level inside the cytoplasm decreased
gradually while the release of cytochrome c increased step
by step. This indicates that embelin induces the activity of Bcl-2,
makes mPTP open irreversibly, causes further changes in the
permeability of the mitochondrial membrane and promotes the release
of cytochrome c. The combination of these two factors
results in apoptosis.
It is well known that the caspase family activates
apoptosis-related protease when apoptosis occurs (30), so the activation of the caspase
family is an important prerequisite for apoptosis. We analyzed the
changes in the procaspase 9, 8, 3 proteins using western blot
analysis after the treatment with embelin and found that the
expression levels of procaspase 9 and 3 decreased significantly
when hepatocellular carcinoma apoptosis occurred, but no apparent
changes in procaspase 8 expression were observed. The release of
cytochrome c from mitochondria to activate caspase 9, 3 is a
key step of the apoptosis pathway (31). However, no apparent changes in
procaspase 8 expression were found. These results indicate that the
induction of hepatocellular carcinoma apoptosis by embelin is
realized via the endogenous mitochondrial pathway instead of the
exogenous death receptor pathway.
In summary, our study demonstrated that embelin
releases cytochrome c and activates the caspase family,
resulting in the induction of hepatocellular carcinoma apoptosis by
regulating the action of the Bcl-2/Bax family on the mitochondrial
pathway. Embelin may offer important contributions in the
development of a new drug to prevent and cure HCC in the
future.
References
|
1
|
Wörns MA and Galle PR: Future perspectives
in hepatocellular carcinoma. Dig Liver Dis. 3:S302–S309. 2010.
|
|
2
|
Rampone B, Schiavone B, Martino A, Viviano
C and Confuorto G: Current management strategy of hepatocellular
carcinoma. World J Gastroenterol. 15:3210–3216. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
El-Serag HB and Rudolph KL: Hepatocellular
carcinoma: epidemiology and molecular carcinogenesis.
Gastroenterology. 132:2557–2576. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rahbari NN, Mehrabi A, Mollberg NM, Müller
SA, Koch M, Büchler MW and Weitz J: Hepatocellular carcinoma:
current management and perspectives for the future. Ann Surg.
253:453–469. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Parkin DM, Bray F, Ferlay J and Pisani P:
Global cancer statistics, 2002. CA Cancer J Clin. 55:74–108. 2005.
View Article : Google Scholar
|
|
6
|
Llovet JM, Burroughs A and Bruix J:
Hepatocellular carcinoma. Lancet. 362:1907–1917. 2003. View Article : Google Scholar
|
|
7
|
Ahn KS, Sethi G and Aggarwal BB: Embelin,
an inhibitor of X chromosome-linked inhibitor-of-apoptosis protein,
blocks nuclear factor-kappaB (NF-kappaB) signaling pathway leading
to suppression of NF-kappaB-regulated antiapoptotic and metastatic
gene products. Mol Pharmacol. 71:209–219. 2007. View Article : Google Scholar
|
|
8
|
Nikolovska-Coleska Z, Xu L, Hu Z, Tomita
Y, Li P, Roller PP, Wang R, Fang X, Guo R, Zhang M, Lippman ME,
Yang D and Wang S: Discovery of embelin as a cell-permeable,
small-molecular weight inhibitor of XIAP through structure-based
computational screening of a traditional herbal medicine
three-dimensional structure database. J Med Chem. 47:2430–2440.
2004. View Article : Google Scholar
|
|
9
|
Joshi R, Kamat JP and Mukherjee T: Free
radical scavenging reactions and antioxidant activity of embelin:
biochemical and pulse radiolytic studies. Chem Biol Interact.
167:125–134. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sreepriya M and Bali G: Effects of
administration of embelin and curcumin on lipid peroxidation,
hepatic glutathione antioxidant defense and hematopoietic system
during N-nitrosodiethylamine/ phenobarbital-induced
hepatocarcinogenesis in Wistar rats. Mol Cell Biochem. 284:49–55.
2006. View Article : Google Scholar
|
|
11
|
Singh D, Singh R, Singh P and Gupta RS:
Effects of embelin on lipid peroxidation and free radical
scavenging activity against liver damage in rats. Basic Clin
Pharmacol Toxicol. May 26–2009.(Epub ahead of print).
|
|
12
|
Aird KM, Ding X, Baras A, Wei J, Morse MA,
Clay T, Lyerly HK and Devi GR: Trastuzumab signaling in
ErbB2-overexpressing inflammatory breast cancer correlates with
X-linked inhibitor of apoptosis protein expression. Mol Cancer
Ther. 7:38–47. 2008. View Article : Google Scholar
|
|
13
|
Dai Y, Qiao L, Chan KW, Yang M, Ye J, Ma
J, Zou B, Gu Q, Wang J, Pang R, Lan HY and Wong BC: Peroxisome
proliferator-activated receptor-gamma contributes to the inhibitory
effects of Embelin on colon carcinogenesis. Cancer Res.
69:4776–4783. 2009. View Article : Google Scholar
|
|
14
|
Danquah M, Li F, Duke CB III, Miller DD
and Mahato RI: Micellar delivery of bicalutamide and embelin for
treating prostate cancer. Pharm Res. 26:2081–2092. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Mori T, Doi R, Kida A, Nagai K, Kami K,
Ito D, Toyoda E, Kawaguchi Y and Uemoto S: Effect of the XIAP
inhibitor Embelin on TRAIL-induced apoptosis of pancreatic cancer
cells. J Surg Res. 142:281–286. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Xu YZ, Zheng RL, Zhou Y, Peng F, Lin HJ,
Bu Q, Mao YQ, Yu LT, Yang L, Yang SY and Zhao YL: Small molecular
anticancer agent SKLB703 induces apoptosis in human hepatocellular
carcinoma cells via the mitochondrial apoptotic pathway invitro and
inhibits tumor growth invivo. Cancer Lett. 313:44–53. 2011.
View Article : Google Scholar
|
|
17
|
Yang L, Ling Y, Zhang Z, Zhao Q, Tang J,
Ji H and Zhang Y: ZL11n is a novel nitric oxide-releasing
derivative of farnesylthiosalicylic acid that induces apoptosis in
human hepatoma HepG2 cells via MAPK/mitochondrial pathways. Biochem
Biophys Res Commun. 409:752–757. 2011. View Article : Google Scholar
|
|
18
|
Qian H, Yang Y and Wang X: Curcumin
enhanced adriamycin-induced human liver-derived hepatoma G2 cell
death through activation of mitochondria-mediated apoptosis and
autophagy. Eur J Pharm Sci. 43:125–131. 2011. View Article : Google Scholar
|
|
19
|
Zhang ZF, Guo Y, Zhang JB and Wei XH:
Induction of apoptosis by chelerythrine chloride through
mitochondrial pathway and Bcl-2 family proteins in human hepatoma
SMMC-7721 cell. Arch Pharm Res. 34:791–800. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Handayani T, Sakinah S, Nallappan M and
Pihie AH: Regulation of p53-, Bcl-2- and caspase-dependent
signaling pathway in xanthorrhizol-induced apoptosis of HepG2
hepatoma cells. Anticancer Res. 27:965–971. 2007.PubMed/NCBI
|
|
21
|
Kerr JF, Wyllie AH and Currie AR:
Apoptosis: a basic biological phenomenon with wide-ranging
implications in tissue kinetics. Br J Cancer. 26:239–257. 1972.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chiarugi P and Giannoni E: Anoikis: a
necessary death program for anchorage-dependent cells. Biochem
Pharmacol. 76:1352–1364. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yang C, Jo SH, Csernus B, Hyjek E, Liu Y,
Chadburn A and Wang YL: Activation of peroxisome
proliferator-activated receptor gamma contributes to the survival
of T lymphoma cells by affecting cellular metabolism. Am J Pathol.
170:722–732. 2007. View Article : Google Scholar
|
|
24
|
Allensworth JL, Aird KM, Aldrich AJ,
Batinic-Haberle I and Devi GR: XIAP inhibition and generation of
reactive oxygen species enhances TRAIL sensitivity in inflammatory
breast cancer cells. Mol Cancer Ther. June 6–2012.(Epub ahead of
print).
|
|
25
|
Yang CL, Ma YG, Xue YX, Liu YY, Xie H and
Qiu GR: Curcumin induces small cell lung cancer NCI-H446 cell
apoptosis via the reactive oxygen species-mediated mitochondrial
pathway and not the cell death receptor pathway. DNA Cell Biol.
31:139–150. 2012. View Article : Google Scholar
|
|
26
|
Mattson MP and Kroemer G: Mitochondria in
cell death: novel targets for neuroprotection and cardioprotection.
Trends Mol Med. 9:196–205. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Burlacu A: Regulation of apoptosis by
Bcl-2 family proteins. J Cell Mol Med. 7:249–257. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Saito M, Korsmeyer SJ and Schlesinger PH:
BAX-dependent transport of cytochrome c reconstituted in pure
liposomes. Nat Cell Biol. 2:553–555. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Haeberlein SL: Mitochondrial function in
apoptotic neuronal cell death. Neurochem Res. 29:521–530. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Nicholson DW, Ali A, Thornberry NA,
Vaillancourt JP, Ding CK, Gallant M, Gareau Y, Griffin PR, Labelle
M and Lazebnik YA: Identification and inhibition of the ICE/CED-3
protease necessary for mammalian apoptosis. Nature. 376:37–43.
1995. View
Article : Google Scholar : PubMed/NCBI
|
|
31
|
Riedl SJ and Shi Y: Molecular mechanisms
of caspase regulation during apoptosis. Nat Rev Mol Cell Biol.
5:897–907. 2004. View
Article : Google Scholar : PubMed/NCBI
|