Introduction
Kuding tea (Ilex kudingcha C.J. Tseng) is a
beverage consumed in China as an alternative to the more common,
green tea (1). Kuding tea has a
reputation for preventing deterioration of the heart and brain
function and maintaining proper body weight. The main active
components are triterpene glycosides (saponins), which have been
dubbed kudinosides and kudinlactones; Kuding tea also contains
polyphenols and flavonoids, similar to those in ordinary tea
(2,3). Caffeoyl quinic acid (CQA) derivatives
have also been identified as major phenolic compounds in Kuding
tea. CQA derivatives have been isolated from the natural functional
compounds of a variety of plants and have demonstrated
pharmacological properties in numerous diseases, including as
antioxidants, hepatoprotectants, antibacterial agents,
antihistamines and as anticancer and neuroprotective agents
(4,5).
Oral squamous cell carcinoma is a type of cancer
that usually develops in the squamous or epithelial cells that
cover the lips and the oral cavity. The malignant or cancerous
cells are usually located in the floor of the mouth or on the
surface of the tongue (6).
Squamous carcinoma cells are located in the epidermis of the skin,
and this type of cancer is one of the major forms of skin cancer.
Squamous cell carcinoma is the second most common skin cancer
(7). The U14 mouse tumor is a
squamous cell carcinoma. It is an ectopically-induced
carcinoma, induced by treating the uterine cervix with
20-methylcholanthrene (8). U14
cells are widely used in studies of tumor invasion, metastasis,
recurrence and drug screening. The establishment of a cultured
tumor cell line, capable of forming a tumor in vivo, is
likely to be helpful for the study of tumor biology on a cellular
and molecular level (9).
Apoptosis is characterized by a series of typical
morphological features, including cell shrinkage, membrane
blebbing, chromatin condensation and nuclear fragmentation
(10). The induction of apoptosis
is a defense against cancer. Apoptosis leads to cell death with
cell shrinkage, pyknosis and karyorrhexis. The regulation of cell
death may be a significant component in cancer as aberrantly
regulated apoptotic cell death causes apoptotic diseases, including
cancer. Elucidating the critical events associated with
carcinogenesis provides the opportunity to prevent cancer
development via dietary intervention by inducing apoptosis,
particularly with bioactive agents or functional foods. Drink is a
significant environmental factor in the overall cancer process, and
is capable of exacerbating or interfering with disease progression.
In addition to dietary effects on protein expression and function,
there is evidence that a large number of food components may exert
effects on the human genome, by directly or indirectly modulating
gene expression (11). Balkwill
and Mantovani demonstrated that inflammation may predispose an
individual to cancer (12).
Hallmarks of inflammation-related cancers include the presence of
inflammatory cells and mediators in tumor cells. Inflammation may
occur in the early stages of cancer, but inflammatory mediators and
cells are also involved in the migration, invasion and metastasis
of malignant cells (13).
Metastasis is the leading cause of mortality among cancer patients
and involves the cancer spreading from a primary site and forming
new tumors in distant organs. Matrix metalloproteases (MMPs) have
an important role in numerous physiological or pathological
processes including embryonic development, morphogenesis,
reproduction, tissue remodeling, arthritis, cardiovascular disease
and metastasis (14). MMP activity
is inhibited by specific endogenous tissue inhibitors of
metalloproteinases (TIMPs). In order to prevent the majority of
cancer types, improved metastasis treatments are required (15).
In the present study, the anticancer and
antimetastatic effects of Kuding tea were examined. Kuding tea was
administered to TCA8113 human tongue carcinoma cells and the
molecular mechanisms underlying the anticancer effects of the tea
were studied. The variations in the activities of Kuding tea at
different concentrations were evaluated and the antimetastatic
effects were assessed in mice with tumors propagated by U14
squamous cell carcinoma cells. An animal model was established in
which the mice were injected with U14 cells for the in vivo
evaluation of buccal mucosa cancer.
Materials and methods
Kuding tea (Ilex kudingcha C.J.
Tseng) extracts
Kuding tea was provided by the Henglv Tea Co.
(Chongqing, China). The Kuding tea was stored at −80ºC and
freeze-dried to produce a powder. The powder was extracted for 24 h
with a 20-fold volume of methanol, twice. The methanol extract was
evaporated using a rotary evaporator (N-1100; Eyela, Tokyo, Japan),
concentrated and subsequently dissolved in DMSO (Amresco; Solon,
OH, USA) in order to create the stock concentration (20%, w/v).
Cancer cells
TCA8113 cells were obtained from the Shanghai
Institute of Biochemistry and Cell Biology (SIBCB, Shanghai, China)
and the U14 squamous cell carcinoma cells were obtained from the
Chinese Academy of Medical Sciences (Beijing, China). The cancer
cells were cultured in RPMI-1640 medium (Welgene Inc., Daegu,
Korea) supplemented with 10% fetal bovine serum (FBS) and 1%
penicillin-streptomycin (Gibco Co., Grand Island, NY, USA) at 37ºC
in a humidified atmosphere containing 5% CO2 (incubator
model 311 S/N29035; Forma, Waltham, MA, USA). The medium was
changed two or three times each week (16).
In vitro cultured U14 cells
(5×106/mouse) were injected into the abdominal cavity of
7-week-old female imprinting control region (ICR) mice. After one
week, the carcinoma ascites were obtained and diluted in sterile
saline to achieve a concentration of 1×107/ml.
3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyltetrazolium bromide (MTT)
assay
The anticancer effects of Kuding tea were assessed
with an MTT assay. The TCA8113 cells were seeded in a 96-well plate
(2×104 cells/ml per well) in a volume of 180 μl.
Subsequently, 20 μl of the 50, 100 or 200 μg/ml Kuding tea samples
were added. The cells were subsequently incubated with the Kuding
tea solutions for 48 h at 37ºC in an incubator (model 311 S/N29035)
in a humidified atmosphere containing 5% CO2. MTT
(Amresco) solution (200 μl; 5 mg/ml) was added to each well and the
cells were cultured for a further 4 h under the same conditions.
Following the removal of the supernatant, 150 μl DMSO was added to
each well and mixed for 30 min. Finally, the absorbance of each
well was measured using an ELISA plate reader (model 680; Bio-Rad,
Hercules, CA, USA) at 540 nm (17).
Reverse transcription-polymerase chain
reaction (RT-PCR) to measure mRNA expression
Total RNA was isolated from TCA8113 cells using
TRIzol reagent (Invitrogen, Carlsbad, CA, USA) according to the
manufacturer’s instructions. The RNA was digested with RNase
(Roche, Basel, Switzerland) for 15 min at 37ºC and purified using
an RNeasy kit (Qiagen, Hilden, Germany) according to the
manufacturer’s instructions. cDNA was synthesized from 2 μg total
RNA by incubating at 37ºC for l h with avian myeloblastosis virus
reverse transcriptase (GE Healthcare, Little Chalfont, UK) with
random hexanucleotides according to the manufacturer’s
instructions. Primer sequences used to specifically amplify the
genes of interest are provided in Table I. Amplification was performed in a
thermal cycler (T100; Bio-Rad), with cycles at 94ºC for 30 sec,
60ºC for 30 sec, then 72ºC for 30 sec performed 50 times for
denaturation. The amplified PCR products were run on 1.0% agarose
gels and visualized by ethidium bromide (EtBr) staining (18).
 | Table ISequences of RT-PCR primers used in
this study. |
Table I
Sequences of RT-PCR primers used in
this study.
| Gene name | Sequence |
|---|
| Bax | Forward: 5′-AAG CTG
AGC GAG TGT CTC CGG CG-3
Reverse: 5′-CAG ATG CCG GTT CAG GTA CTC AGT C-3′ |
| Bcl-2 | Forward: 5′-CTC GTC
GCT ACC GTC GTG ACT TGG-3′
Reverse: 5′-CAG ATG CCG GTT CAG GTA CTC AGT C-3′ |
| Caspase-9 | Forward: 5′-GGC CCT
TCC TCG CTT CAT CTC-3′
Reverse: 5′-GGT CCT TGG GCC TTC CTG GTA T-3′ |
| Caspase-3 | Forward: 5′-CAA ACT
TTT TCA GAG GGG ATC G-3′
Reverse: 5′-GCA TAC TGT TTC AGC ATG GCA-3′ |
| NF-κB | Forward: 5′-CAC TTA
TGG ACA ACT ATG AGG TCT CTG G-3′
Reverse: 5′-CTG TCT TGT GGA CAA CGC AGT GGA ATT TTA GG-3′ |
| IκB-α | Forward: 5′-GCT GAA
GAA GGA GCG GCT ACT-3′
Reverse: 5′-TCG TAC TCC TCG TCT TTC ATG GA-3′ |
| iNOS | Forward: 5′-AGA GAG
ATC GGG TTC ACA-3′
Reverse: 5′-CAC AGA ACT GAG GGT ACA-3′ |
| COX-2 | Forward: 5′-TTA AAA
TGA GAT TGT CCG AA-3′
Reverse: 5′-AGA TCA CCT CTG CCT GAG TA-3′ |
| MMP-2 | Forward: 5′-CTT CTT
CAA GGA CCG GTT CA-3′
Reverse: 5′-GCT GGC TGA GTA CCA GTA-3′ |
| MMP-9 | Forward: 5′-TGG GCT
ACG TGA CCT ATG AC-3′
Reverse: 5′-GCC CAG CCC ACC TCC ACT CC-3′ |
| TIMP-1 | Forward: 5′-GTC AGT
GAG AAG CAA GTC GA-3′
Reverse: 5′-ATG TTC TTC TCT GTG ACC CA-3′ |
| TIMP-2 | Forward: 5′-TGG GGA
CAC CAG AAG TCA AC-3′
Reverse: 5′-TTT TCA GAG CCT TGG AGG AG-3′ |
| GAPDH | Forward: 5′-CGG AGT
CAA CGG ATT TGG TC-3′
Reverse: 5′-AGC CTT CTC CAT GGT CGT GA-3′ |
Protein extraction and western blot
analysis
Total cell lysates were obtained with an extraction
buffer as described previously (18). Protein concentrations were
determined using a protein assay kit (Bio-Rad). For western blot
analysis, aliquots of the lysate containing 30–50 μg protein were
separated by SDS-PAGE and subsequently electrotransferred to a
nitrocellulose membrane (Schleicher and Schuell, Keene, NH, USA).
The membranes were subjected to immunoblot analysis and the
proteins were visualized using an enhanced chemiluminescence (ECL)
assay kit (GE Healthcare). The cell lysates were separated by 12%
SDS-PAGE, transferred to a polyvinylidene fluoride membrane (GE
Healthcare), blocked with 5% skimmed milk, and incubated with the
primary antibodies (1:1,000 dilution). Antibodies against Bax,
Bcl-2, caspases, iNOS, COX-2, NF-κB, IκB-α, MMPs and TIMPs were
obtained from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA, USA).
Following incubation with the horseradish peroxidase-conjugated
goat anti-mouse secondary antibodies (Abbkine, Redlands, CA, USA)
at room temperature, immunoreactive proteins were detected using a
chemiluminescent ECL assay kit (GE Healthcare) according to the
manufacturer’s instructions. Bands in the blot were visualized
using a LAS3000 luminescent image analyzer (Fujifilm Life Science,
Tokyo, Japan).
Induction of buccal mucosa cancer
Female ICR mice (n=50, 6-weeks-old) were purchased
from the Experimental Animal Center of Chongqing Medical University
(Chongqing, China). They were maintained in a
temperature-controlled (temperature 25±2ºC, relative humidity
50±5%) facility with a 12-hour light/dark cycle and had unlimited
access to a standard mouse chow diet and water.
To investigate the preventive effect of the Kuding
tea against buccal mucosa cancer induced by injecting U14 cells
into the mice, the mice were divided into five groups with ten mice
in each group. The experiment design was as follows: Kuding tea
solutions (Group A: 400 mg/kg, Group B: 800 mg/kg, Group C: 1,600
mg/kg) were administered to the three groups, respectively, by
gavage, and Kuding tea solutions (Group A: 100 mg/ml, Group B: 200
mg/ml, Group C: 400 mg/ml) were topically applied to the buccal
mucosa of the mice in the three Kuding tea groups once every 12 h
for 14 days. The control and Kuding tea groups were subsequently
inoculated with the U14 cancer cell suspension
(1×107/ml; 0.05 ml per mouse) on the buccal mucosa. The
normal group mice received no treatment during the experiment. The
mice were sacrificed 14 days later and their tumor volumes and
lymph node metastasis rates were determined (16).
Histological grading of buccal mucosa
cancer
Buccal mucosa and lymph node tissues were removed
and embedded in paraffin for histological analysis with hematoxylin
and eosin staining. The buccal mucosa cancers were graded as
follows: I, well-differentiated carcinoma, cells appear much like
the adjacent benign squamous epithelium; II, moderately
differentiated carcinoma, cells form large anastomosing areas in
which keratin pearls are formed; they are not numerous and the main
component consists of cells with pronounced cytonuclear atypia;
III, poorly differentiated carcinoma: cells have lost the majority
of their squamous epithelial characteristics and architecture
(16).
Statistical analysis
Analysis of variance (ANOVA) was performed and the
results are presented as the means ± SD. Differences between mean
values of the individual groups were assessed with a one-way ANOVA
and Duncan’s multiple range tests. P<0.05 was considered to
indicate a statistically significant difference. The SAS v9.1
statistical software package (SAS Institute Inc., Cary, NC, USA)
was used to perform all statistical analyses.
Results
Kuding tea induced the inhibition of
TCA8113 cell growth
The inhibitory growth effect of Kuding tea on
TCA8113 cells was evaluated using an MTT assay. When the Kuding tea
methanol extracts were added to the TCA8113 cells, the growth
inhibition rates associated with the 50, 100 and 200 μg/ml extracts
were 10, 41 and 75%, respectively (P<0.05, Table II). These results demonstrated
that Kuding tea has an antiproliferative effect on TCA8113
cells.
 | Table IIGrowth inhibition of TCA8113 human
tongue carcinoma cells by various concentrations of Kuding tea as
evaluated by an MTT assay. |
Table II
Growth inhibition of TCA8113 human
tongue carcinoma cells by various concentrations of Kuding tea as
evaluated by an MTT assay.
|
OD540 |
|---|
|
|
|---|
| Treatment | 50 μg/ml | 100 μg/ml | 200 μg/ml |
|---|
| Control | | 0.497±0.004a | |
| Kuding tea | 0.447±0.010b (10) | 0.293±0.008c (41) | 0.124±0.008d (75) |
Apoptosis-related expression of Bax,
Bcl-2 and caspases
The expression levels of Bax, Bcl-2, and caspase-3
and caspase-9 mRNA and proteins in TCA8113 cells were analyzed by
RT-PCR and western blotting, respectively, following 48 h
incubation with 50, 100 and 200 μg/ml solutions of Kuding tea. As
shown in Fig. 1, treatment with
200 μg/ml Kuding tea markedly altered the levels of pro-apoptotic
Bax and anti-apoptotic Bcl-2. Bax mRNA and protein expression
levels were upregulated, while those of Bcl-2 were decreased
significantly. These results suggest that a high concentration of
Kuding tea markedly induced apoptosis in TCA8113 cells via a
Bcl-2-dependent pathway compared with a low concentration. Modest
mRNA and protein expression levels of caspase-9 and -3 were
detected in the untreated control TCA8113 cells, but higher
expression levels were detected once the cells had been treated
with the Kuding tea samples. Notably, the mRNA expression levels of
caspase-3 and -9 significantly increased in the presence of the
Kuding tea with increasing extract concentrations.
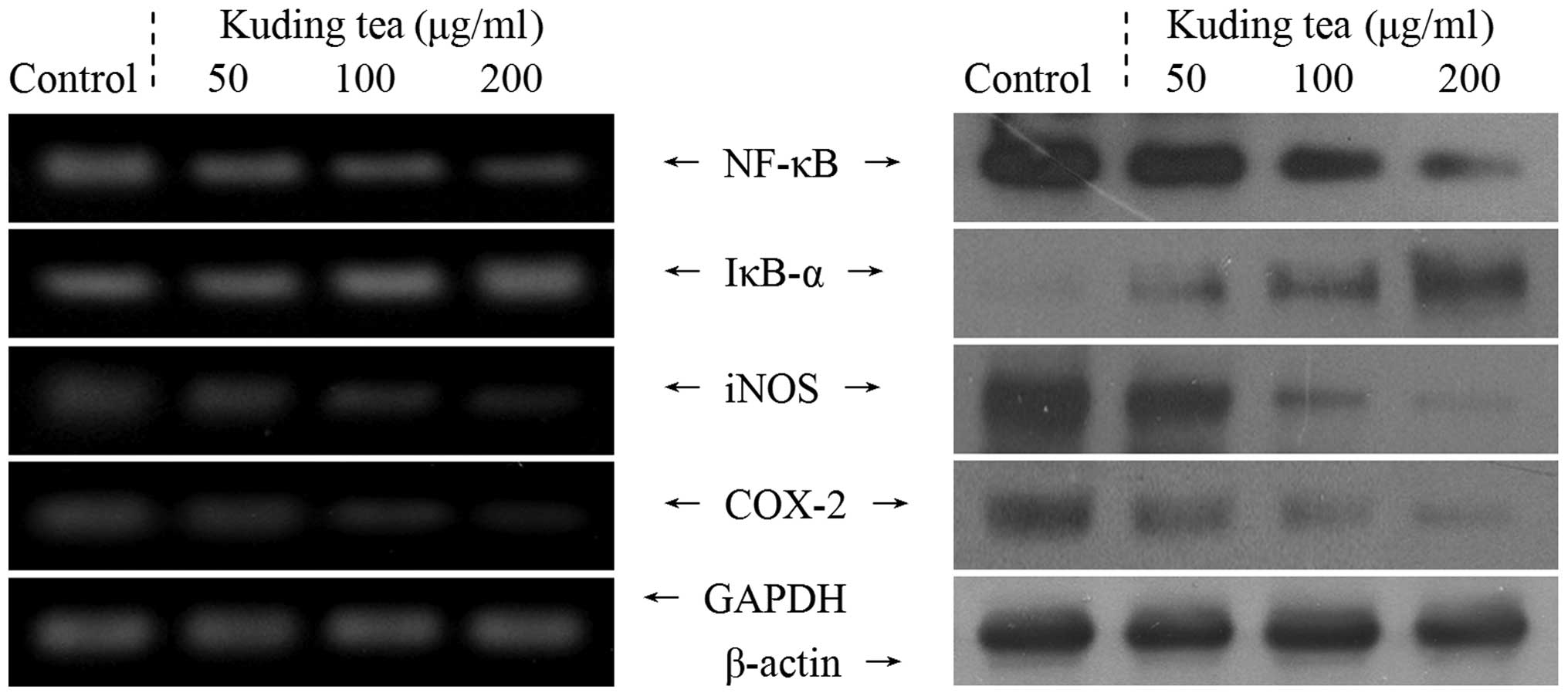
Inflammation-related expression of NF-κB,
IκB-α, iNOS and COX-2
Subsequently, the correlation of the anticancer
characteristics of Kuding tea with the inhibition of the expression
of the inflammation-related genes NF-κB, IκB-α, iNOS, and COX-2 was
investigated. At a concentration of 200 μg/ml, Kuding tea
demonstrated anti-inflammatory activity in the TCA8113 cells, with
decreased mRNA and protein expression of NF-κB along with increased
IκB-α expression compared with that in cells treated with the other
concentrations of Kuding tea (Fig.
2). As shown in Fig. 2, the
levels of mRNA and protein expression of COX-2 and iNOS were high
in the untreated control TCA8113 cells; however, these indicators
were almost undetectable in the presence of 200 μg/ml Kuding tea.
Following incubation with 200 μg/ml Kuding tea, the mRNA and
protein levels of COX-2 and iNOS were significantly decreased.
Metastasis-related MMP and TIMP
expression
RT-PCR and western blot analyses were performed in
order to determine whether the antimetastatic effect of the Kuding
tea was due to the genetic regulation of metastatic mediators,
specifically MMPs (MMP-2 and MMP-9) and TIMPs (TIMP-1 and TIMP-2),
in TCA8113 cells. As shown in Fig.
3, Kuding tea significantly decreased the mRNA and protein
expression levels of MMP-2 and MMP-9 and increased the expression
levels of TIMP-1 and TIMP-2. These changes in TIMP and MMP
expression resulting from Kuding tea treatment may lead to
metastatic inhibition in vitro. The results also
demonstrated that Kuding tea had strong antimetastatic
activity.
In vivo preventive effects on buccal
mucosa cancer of Kuding tea
Buccal mucosa cancer was induced by injecting U14
cells into the mice. After 14 days, the mice in all of the groups
exhibited carcinogenesis. The tumor volumes of buccal mucosa
tissues were measured. The tumor volumes for the control and A, B
and C Kuding tea groups were 12.4, 11.8, 9.6 and 6.4
mm3, respectively (Table
III). Five mice in the control group, five in Group A, four in
Group B and two in Group C exhibited lymph node metastasis.
Consequently, the lymph node metastasis rates were 50, 50, 40 and
20%, respectively. These results demonstrated that Kuding tea was
effective in impeding carcinogenesis, proliferation and
metastasis.
 | Table IIITumor volumes and lymph node
metastasis rates of mice topically treated with various
concentrations of Kuding tea. |
Table III
Tumor volumes and lymph node
metastasis rates of mice topically treated with various
concentrations of Kuding tea.
| Group | Normal | Control | Group A | Group B | Group C |
|---|
| Tumor volume
(mm3) | 0 | 12.4±0.6a | 11.8±0.8a,b | 9.6±0.5b | 6.4±0.5c |
| Lymph node
metastasis rate | 0 | 5/10 (50%) | 5/10 (50%) | 4/10 (40%) | 2/10 (20%) |
Histological changes in the buccal mucosa of the
mice injected with U14 cells were examined by hematoxylin and eosin
staining. The histological tissue sections of mice in the normal
group exhibited the normal histological morphology of squamous
epithelium tissue. Histopathological evaluation revealed
indications of buccal mucosa cancer in all groups receiving U14
cells (Fig. 4). The sections from
the mice in Group A and the control group demonstrated that all
tissue had lost its squamous epithelial characteristics and
architecture, but the tissues from Group A demonstrated
intracellular bridging between the normal squamous cells. The
histopathological sections indicated that mice in Group A and the
control group developed poorly differentiated carcinoma (grade
III), and the control group experienced more serious
carcinogenesis. The tumor cells of the mice in Group B were in
nests, and there were certain larger, eosinophilic, polygonal cells
that were attempting to layer themselves in a squamous-like
fashion. However, for Group B, the overall resemblance to a normal
squamous epithelium was less clear (grade II). The tissue sections
of Group C appeared less like normal squamous epithelium. The cells
appeared similar to those of the adjacent benign squamous
epithelium (grade I). From these sections, Kuding tea demonstrated
a preventive effect on buccal mucosa cancer.
Discussion
Although Kuding tea is a traditional drink, little
scientific data on its effects are available. Ilex kudingcha
C.J. Tseng, also known as Kuding tea, has high levels of ursolic
acid, β-amyrin, lupeol and taraxerol (19). Kuding tea has been reported to have
various therapeutic effects, including hepatoprotective,
antibacterial, antihistamine and anticancer activities, on numerous
pathologic conditions (4). Kuding
tea has also been shown to exhibit in vitro anticancer
effects on human nasopharyngeal carcinoma cells according to the
MTT assay (20).
The induction of apoptosis in cancer cells is a
promising approach for cancer therapy (21). In the normal cell, the
anti-apoptotic Bcl-2 gene is expressed on the outer mitochondrial
membrane surface (22). As the Bax
and Bcl-2 genes are mostly expressed during apoptosis, it has been
determined that these genes regulate apoptotic activity. Apoptosis
results from the activation of caspases that act as
aspartate-specific proteases (23). The results of the present study
demonstrated that changes in Bax and Bcl-2 expression correlated
with apoptosis promoted by the Kuding tea. Apoptosis results from
the activation of caspase family members that act as
aspartate-specific proteases. Caspases form a proteolytic network
within the cell whereby upstream initiator caspases are activated
early in the apoptotic process (caspase-9) and in turn activate
other downstream caspases (caspase-3). Downstream of the region in
which apoptosis is initiated in the mitochondrial pathway,
caspase-9 and caspase-3 play major roles. Caspase-3 appears to
amplify caspase-9 initiation signals to induce a fully fledged
commitment to nuclear disassembly (24,25).
NF-κB is present in the cytosol where it is bound to the inhibitory
protein, IκB. Following its induction by a variety of agents, NF-κB
is released from IκB and translocates to the nucleus where it binds
to the κB binding sites in the promoter regions of the target genes
(27). NF-κB is involved in the
inhibition of apoptosis, stimulation of cell proliferation,
inflammation, immune response and tumorigenesis. The activation of
NF-κB normally prevents apoptosis. iNOS and COX-2, two genes
regulated by NF-κB, which are induced by inflammation, are
frequently overexpressed in cancer cells. Increased NF-κB activity
localized in the nucleus is particularly identified within cells
where there is abundant expression of iNOS and COX-2 (28). It has been suggested that COX-2 has
an important role in colon carcinogenesis, and NOS, in addition to
iNOS, may be a successful target for cancer chemoprevention
(26). These mechanisms may be
involved in the anticancer effects of Kuding tea on cancer cells.
Based on the results of the MTT assay and the expression patterns
of pro-apoptotic genes observed in the present study, it was
concluded that cancer cells treated with Kuding tea underwent
apoptosis.
MMPs, a family of zinc-dependent endopeptidases,
play an important role in tumorigenesis and metastasis. MMPs are
capable of cleaving almost all extracellular matrix (ECM)
substrates. Degradation of the ECM is a key event in tumor
progression, invasion and metastasis (29). Among the MMP family members, MMP-2
and MMP-9 are molecules crucial for cancer invasion (30), and are highly expressed in breast
and colon cancer cells (31).
Notably, the inhibition of MMP activity is useful for controlling
tumorigenesis and metastasis (32). TIMPs are naturally occurring
inhibitors of MMPs that prevent catalytic activity by binding to
activated MMPs, thereby blocking ECM breakdown (33). Disturbances in the ratio between
MMPs and TIMPs have been observed during tumorigenesis (34). Maintaining the balance between MMPs
and TIMPs or increasing TIMP activity are useful methods for
controlling tumor metastasis (35). Experimental evidence demonstrating
the role of MMPs in metastasis has been obtained by in vitro
invasion assays (36).
MMP-2 and MMP-9 are key factors in cancer cell
invasion and metastasis in vitro (37). Spontaneous and experimental
metastasis to the liver decreased in mice that overexpressed TIMP1,
and increased in mice that expressed antisense TIMP-1 mRNA. The
ectopic overexpression of TIMP-1 in the brains of transgenic mice
also reduced experimental metastasis to the brain (38). Notably, MMP-2 and MMP-9 are
important for tumor invasion and angiogenesis. Thus, tumor
metastasis may be inhibited by blocking MMP synthesis and activity
(39).
Histopathology is an important tool in anatomical
pathology, as the accurate diagnosis of cancer usually requires the
histopathological examination of samples. Histopathology is an
important clinical standard for diagnosing oral cancer (40). As buccal mucosa cancer is the most
common cancer of the oral cavity (41), the cancer-preventive effect of
Kuding tea was evaluated and confirmed using the buccal mucosa
cancer mouse model. Accordingly, Kuding tea may be expected to
contribute to the prevention of buccal mucosa cancer.
In conclusion, various in vitro experimental
methods, including MTT, RT-PCR, and western blotting assays, were
used to evaluate the anticancer effects of Kuding tea. The in
vivo anticancer effects of Kuding tea were confirmed in mice
injected with U14 squamous cell carcinoma cells. The results from
the present study demonstrated that the in vivo
antimetastatic and anticancer effects of high concentrations of
Kuding tea were stronger than those of low concentrations. Overall,
Kuding tea demonstrated potent in vitro and in vivo
anticancer activities. The active compounds present in Kuding tea
require further identification and evaluation.
References
|
1
|
Liu LX, Sun Y, Laura TG, Liang XF, Ye H
and Zeng XX: Determination of polyphenolic content and antioxidant
activity of kudingcha made from Ilex kudingcha C.J. Tseng.
Food Chem. 112:35–41. 2009. View Article : Google Scholar
|
|
2
|
Sun Y, Xu WQ, Zhang WQ, Hu QH and Zeng XX:
Optimizing the extraction of phenolic antioxidants from kudingcha
made from Ilex kudingcha C.J. Tseng by using response
surface methodology. Sep Purif Technol. 78:311–320. 2011.
View Article : Google Scholar
|
|
3
|
Bravo L, Goya L and Lecumberri E: LC/MS
characterization of phenolic constituents of mate (Ilex
paraguariensis, St. Hil) and its antioxidant activity compared
to commonly consumed beverages. Food Res Int. 40:393–405. 2007.
|
|
4
|
Nakajima Y, Shimazawa M, Mishima S and
Hara H: Water extract of propolis and its main constituents,
caffeoylquinic acid derivatives, exert neuroprotective effects via
antioxidant actions. Life Sci. 80:370–377. 2007. View Article : Google Scholar
|
|
5
|
Han J, Miyamae Y, Shigemori H and Isoda H:
Neuroprotective effect of 3,5-di-O-caffeoylquinic acid on SH-SY5Y
cells and senescence-accelerated-prone mice 8 through the
up-regulation of phosphoglycerate kinase-1. Neuroscience.
169:1039–1045. 2010. View Article : Google Scholar
|
|
6
|
Chaturvedi AK, Engels EA, Anderson WF and
Gillison ML: Incidence trends for human papillomavirus-related and
-unrelated oral squamous cell carcinomas in the United States. J
Clin Oncol. 26:612–619. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Alam M and Ratner D: Cutaneous
squamous-cell carcinoma. New Engl J Med. 344:975–983. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gu B, Feng HL, Dong JH, Zhang H, Bian XC
and Liu YQ: The establishment and characterization of a continuous
cell line of mouse cervical carcinoma. Chinese J Clin Oncol.
5:44–48. 2008. View Article : Google Scholar
|
|
9
|
Su GY, Liu XF, Ren LR, Gu P, Zhang JB and
Liu YQ: Establishment and characterization of a continuous cell
line of mouse breast cancer. Chinese J Clin Oncol. 33:1177–1179.
2006.
|
|
10
|
Lowe SW and Lin AW: Apoptosis in cancer.
Carcinogenesis. 21:485–495. 1999. View Article : Google Scholar
|
|
11
|
Kwon JI, Kim GY, Park KY, Ryu CH and Choi
YH: Induction of apoptosis by linoleic acid is associated with the
modulation of Bcl-2 family and Fas/FasL system and activation of
caspases in AGS human gastric adenocarcinoma cells. J Med Food.
11:1–8. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Balkwill F and Mantovani A: Inflammation
and cancer: back to Virchow? Lancet. 357:539–545. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Mantovani A, Allavena P, Sica A and
Balkwill F: Cancer-related inflammation. Nature. 454:436–444. 2008.
View Article : Google Scholar
|
|
14
|
Itoh Y and Nagase H: Matrix
metalloproteinases in cancer. Essays Biochem. 38:21–36.
2002.PubMed/NCBI
|
|
15
|
Chambers AF, Groom AC and MacDonald IC:
Dissemination and growth of cancer cells in metastatic sites. Nat
Rev Cancer. 2:563–572. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhao X, Pang L, Qian Y, et al: An animal
model of buccal mucosa cancer and cervical lymph node metastasis
induced by U14 squamous cell carcinoma cells. Exp Ther Med.
5:1083–1088. 2013.PubMed/NCBI
|
|
17
|
Zhao X, Kim SY and Park KY: Bamboo salt
has in vitro anticancer activity in HCT-116 cells and exerts
anti-metastatic effects in vivo. J Med Food. 16:9–19.
2013.
|
|
18
|
Zhao X, Deng XX, Park KY, Qiu LH and Pang
L: Purple bamboo salt has anticancer activity in TCA8113 cells
in vitro and preventive effects on buccal mucosa cancer in
mice in vivo. Exp Ther Med. 5:549–554. 2013.
|
|
19
|
Ma HY, Xu FL and Liu YM: Analysis of
microelements in Kudingcha and common tea. J Sichuan Univ (Nat Sci
Edit). 39:770–772. 2002.(In Chinese).
|
|
20
|
Nong CZ, Li SS, Mao DW, Huang ZH, Guo LX,
Wei SY and Nong SY: The proliferation inhibition effect of ursolic
acid from Kuding tea on NCE carcinoma cell. Lishizhen Med Mater Med
Res. 22:2687–2689. 2011.
|
|
21
|
Milanezi F, Leitão D, Ricardo S, Augusto I
and Schmitt F: Evaluation of HER2 in breast cancer: reality and
expectations. Expert Opin Med Diagn. 3:607–620. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chao DT and Korsmeyer SJ: Bcl-2 family:
regulators of cell death. Annu Rev Immunol. 16:395–419. 1998.
View Article : Google Scholar
|
|
23
|
Kidd VJ: Proteolytic activities that
mediate apoptosis. Annu Rev Physiol. 60:533–573. 1998. View Article : Google Scholar
|
|
24
|
Blanc C, Deveraux QL, Krajewski S, Jänicke
RU, Porter AG, Reed JC, Jaggi R and Marti A: Caspase-3 is essential
for procaspase-9 processing and cisplatin-induced apoptosis of
MCF-7 breast cancer cells. Cancer Res. 60:4386–4390.
2000.PubMed/NCBI
|
|
25
|
Cryns V and Yuan JY: Proteases to die for.
Genes Dev. 12:1551–1570. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Delić R and Štefanović M: Optimal
laboratory panel for predicting preeclampsia. J Matern Fetal
Neonatal Med. 23:96–102. 2010.PubMed/NCBI
|
|
27
|
Baeuerle PA: IkappaB-NF-kappaB structures:
at the interface of inflammation control. Cell. 95:729–731. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Tak PP and Firestein GS: NF-kappaB: a key
role in inflammatory diseases. J Clin Invest. 107:7–11. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Vihinen P, Ala-aho R and Kähäri VM: Matrix
metalloproteinase as therapeutic targets in cancer. Curr Cancer
Drug Targets. 5:203–220. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Bogerieder T and Herlyn M: Axis of evil:
molecular mechanism of cancer metastasis. Oncogene. 22:6524–6536.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zucker S and Vacirca J: Role of matrix
metalloproteinases (MMPs) in colorectal cancer. Cancer Metastasis
Rev. 23:101–117. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Chen PN, Chu SC, Chiou HL, Kuo WH, Chiang
CL and Hsieh YS: Mulberry anthocyanins, cyanidin 3-rutinoside and
cyanidin 3-glucoside, exhibited an inhibitory effect on the
migration and invasion of a human lung cancer cell line. Cancer
Lett. 235:248–259. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Uzui H, Harpf A, Liu M, et al: Increased
expression of memebrane type 3-matrix metalloproteinase in human
atherosclerotic plaque: role of activated macrophage and
inflammatory cytokines. Circulation. 106:3024–3030. 2002.
View Article : Google Scholar
|
|
34
|
Lambert E, Dassé E, Haye B and Petitfrére
E: TIMPs as multifacial proteins. Crit Rev Oncol Hematol.
49:187–198. 2004. View Article : Google Scholar
|
|
35
|
Mysliwiec AG and Ornstein DL: Matrix
metalloproteinases in colorectal cancer. Clin Colorect Cancer.
1:208–219. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Chambers AF and Matrisian LM: Changing
views of the role of matrix metalloproteinases in metastasis. J
Natl Cancer Inst. 89:1260–1270
|
|
37
|
Deryugina EI and Quigley JP: Matrix
metalloproteinases and tumor metastasis. Cancer Metastasis Rev.
25:9–34. 2006. View Article : Google Scholar
|
|
38
|
Krüger A, Fata JE and Khokha R: Altered
tumor growth and metastasis of a T-cell lymphoma in Timp-1
transgenic mice. Blood. 90:1993–2000. 1997.PubMed/NCBI
|
|
39
|
Shin DY, Kim GY, Kim JI, et al:
Anti-invasive activity of diallyl disulfide through tightening of
tight junctions and inhibition of matrix metalloproteinase
activities in LNCaP prostate cancer cells. Toxicol In Vitro.
24:1569–1576. 2010. View Article : Google Scholar
|
|
40
|
Sankaranarayanan R, Ramadas K, Thomas G,
et al: Effect of screening on oral cancer mortality in Kerala,
India: a cluster-randomised controlled trial. Lancet.
365:1927–1933. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kolanjiappan K, Ramachandran CR and
Manoharan S: Biochemical changes in tumor tissues of oral cancer
patients. Clin Biochem. 36:61–65. 2003. View Article : Google Scholar : PubMed/NCBI
|