Introduction
The development of gene therapy and transgenic
technology has provided an effective means with which to treat a
variety of diseases (1). With
regard to non-union fractures and bone defects, bone regeneration
is a major challenge. Ex vivo gene therapy has been
demonstrated to strengthen bone regeneration by guiding osteogenic
differentiation (2).
Bone morphogenetic proteins (BMPs), a group of
secreted proteins that belong to the transforming growth factor-β
(TGF-β) superfamily, initiate cartilage and bone formation in a
sequential cascade (3). Among the
BMP family members, BMP2 has the highest osteoinductive activity
in vivo (4). Although
osteogenic factor BMP2 is important during the induction of bone
formation, bone regeneration is a highly complicated process that
involves a number of growth factors. Vascular endothelial growth
factor (VEGF), the most well-characterized angiogenic factor, is
crucial in bone healing and skeletal development (5). BMP-induced VEGF production in
osteoblast-like cells is important in the coupling of bone
formation and angiogenesis (6).
Since BMP2 and VEGF are involved in bone formation,
it has been proposed that the combined delivery of BMP2 and VEGF
may have a more effective impact on bone regeneration than the
delivery of a single gene alone. Furthermore, the specific roles of
BMP2 and VEGF in the process of bone formation require further
investigation. In order to elucidate the efficacy of the
combination of the two growth factors, a constructive gene therapy
model was created using Sprague Dawley (SD) rat bone marrow-derived
mesenchymal stem cells (MSCs) that were lentivirally cotransfected
with hBMP2 and hVEGF165. The expression of bone regeneration was
then investigated using this model.
Materials and methods
Isolation and expansion of rat MSCs
The bone marrow was obtained from purchased rats.
After the rats were sacrificed, the femur and tibia were separated,
both ends of each bone were snipped and bone marrow was washed with
10 ml Dulbecco’s modified Eagle’s medium (DMEM) culture medium
twice. The bone marrow cells were cultured in tissue culture dishes
(BD Falcon™; BD Biosciences, Franklin Lakes, NJ, USA) in DMEM
supplemented with 10% (v/v) fetal bovine serum (FBS) and 1% (w/v)
penicillin/streptomycin at 37°C in 5% CO2. Since the
MSCs were able to adhere to the surface of culture dishes (whereas
hemopoietic cells were not), the adherent cells were isolated from
the bone marrow through adherence-separation culturing, with the
medium changed twice a week. Upon reaching 80% confluency, the
cells were detached using 0.25% (w/v) trypsin/1 mM EDTA solution
(1:1, v/v), replated at a density of 1×104
cells/cm2 in tissue culture dishes and cultured as
first-passage cells (P1) until confluency (5–7 days). MSCs of
passage 3 (P3) were used for transfection.
Lentiviral vector construction and
production
The cDNAs for BMP2, VEGF165 and enhanced green
fluorescent protein (EGFP), obtained from Cyagen Biosciences, were
amplified using the polymerase chain reaction (PCR) with the
primers listed in Table I.
Following this, the BMP2-EGFP and BMP2-VEGF165-EGFP cDNAs were
subcloned into the pLV-EX2d-EF1A expression lentivector (Invitrogen
Life Technologies, Carlsbad, CA, USA), respectively, and 293FT
producer cells were cotransfected with pLV/helper packaging plasmid
mix (Invitrogen Life Technologies) and expression lentivector
(containing BMP2-EGFP or BMP2-VEGF165-EGFP) plasmid using
Lipofectamine™ 2000 (Invitrogen Life Technologies). Thus, the
lentivirus containing BMP2-EGFP cDNA (Lv-BMP2-EGFP-Neo) and the
lentivirus containing BMP2-VEGF165-EGFP cDNA
(Lv-BMP2-VEGF165-EGFP-Neo) were obtained.
 | Table IPrimers of VEGF165 and BMP2 genes from
Cyagen Biosciences for the amplification of VEGF165 and BMP2 by
PCR. |
Table I
Primers of VEGF165 and BMP2 genes from
Cyagen Biosciences for the amplification of VEGF165 and BMP2 by
PCR.
| Gene | Primer |
|---|
|
attB1-Kozak-BMP2-F |
GGGGACAAGTTTGTACAAAAAAGCAGGCTGCCACCATGGTGGCCGGGACCCGC |
| BMP2-T2A-R-1 |
AAGACTTCCCCTGCCCTCTCCGGAGCCGCGACACCCACAACCCTCC |
| T2A-VEGF165-F |
CGGGGACGTGGAGGAAAATCCCGGCCCCATGAACTTTCTGCTGTCTTGGGTG |
| VEGF165-P2A-R-1 |
ACAGAGAGAAGTTCGTGGCGCCGCTGCCCCGCCTCGGCTTGTCACAT |
| P2A-EGFP-F |
GCAAGCAGGAGATGTTGAAGAAAACCCCGGGCCTATGGTGAGCAAGGGCGAGGA |
| T2A-EGFP-F |
CGTGGAGGAAAATCCCGGCCCCATGGTGAGCAAGGGCGAGG |
| attB2-EGFP-R |
GGGGACCACTTTGTACAAGAAAGCTGGGTTTACTTGTACAGCTCGTCCATG |
Cultured rat MSCs of passage 5 were transfected with
Lv-BMP2-EGFP-Neo and Lv-BMP2-VEGF165-EGFP-Neo at a multiplicity of
infection (MOI) of 5, 10 and 20, respectively. The efficiency of
the lentiviral gene transfer in the MSCs was quantitatively
determined according to the fraction of fluorescent cells using
fluorescence microscopy, in accordance with the manufacturer’s
instructions (Invitrogen Life Technologies) at 2 days subsequent to
transfection. The fraction of cells that glowed green and reflected
the lentiviral gene transfer efficiency was dose-dependent in the
range of 5–20 MOI. The highest transfection efficiency, of <90%,
was obtained at an MOI of 20. There were three groups in this
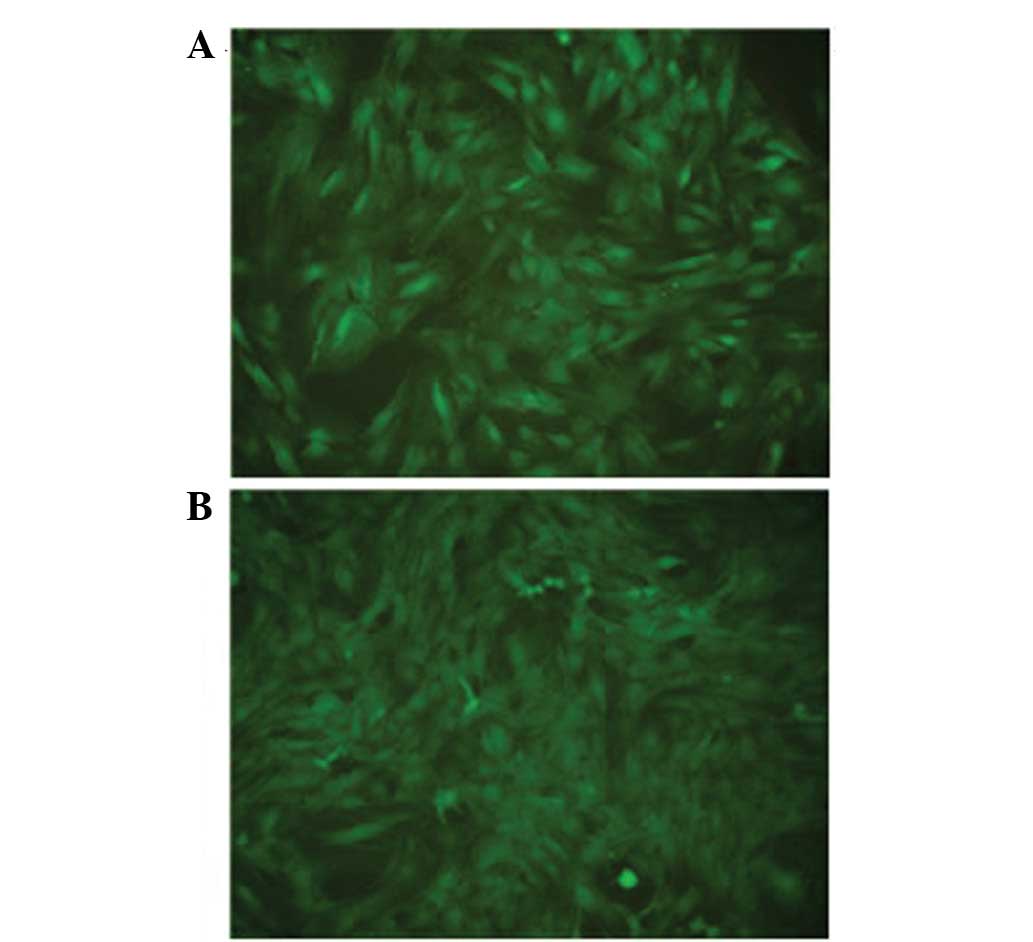
study: MSCs infected with Lv-BMP2-EGFP-Neo (BMP2 group) at 20 MOI
(Fig. 1A), MSCs infected with
Lv-BMP2-VEGF165-EGFP-Neo (BMP2 + VEGF165 group) at 20 MOI (Fig. 1B) and untransfected control.
Western blot analysis of BMP2 and
VEGF
SD rat bone marrow-derived MSCs were transfected
with Lv-BMP2-EGFP-Neo and Lv-BMP2-VEGF165-EGFP-Neo virus and
incubated for 48 h. The transfected and untransfected SD-MSCs were
lysed with radioimmunoprecipitation assay (RIPA) buffer with
phenylmethylsulfonyl fluoride (PMSF) on ice for 5 min, and the
total protein of the sample was assessed. A total of 100 μg protein
was suspended in 5X sodium dodecyl sulfate (SDS) buffer and
subjected to SDS-polyacrylamide gel electrophoresis (PAGE) using 8%
Tris-HCl gel. The separated proteins were subsequently transferred
to a polyvinylidene difluoride (PVDF) transfer membrane. The
membranes were blocked for 1 h at room temperature in Tris-buffered
saline with casein. Antibodies against BMP2 or VEGF monoclonal
immunoglobulin G (IgG; dilution, 1:250; Santa Cruz Biotechnology,
Inc., Santa Cruz, CA, USA) were incubated on the membrane overnight
at 4°C and detected using secondary horseradish
peroxidase-conjugated antibodies (dilution, 1:2,500; Santa Cruz
Biotechnology, Inc.) mixed at room temperature for 1 h. The PVDF
transfer membrane was then washed with Tris-buffered saline and
developed using an enhanced chemiluminescence detection system with
exposure to FluorChem™ HD2 (Cell Biosciences, Inc., Santa Clara,
CA, USA).
Quantitative reverse
transcription-polymerase chain reaction (qPCR) analysis
Gene-specific primers for VEGF165, BMP2 and
glyceraldehyde-3-phosphate dehydrogenase (GAPDH) were designed as
the primer set (Table II) to
detect the relative mRNA expression. The MSCs were collected
respectively from the BMP2 and BMP2 + VEGF165 groups described
previously at 5 days subsequent to transfection, with untransfected
cells as a control. Total RNA was extracted using the TRIzol method
(Ambion, Austin, TX, USA) from each cell sample and the cDNA was
synthesized from total RNA. qPCR was performed using
SYBR-Green® Realtime PCR Master mix (Toyobo, Osaka,
Japan). To correct for differences in the RNA quality and quantity
among the samples, the data were normalized to the data for
GAPDH.
 | Table IIGene-specific primers for VEGF165,
BMP2 and GAPDH were designed as the primer set for qPCR analysis to
detect the relative mRNA expression. |
Table II
Gene-specific primers for VEGF165,
BMP2 and GAPDH were designed as the primer set for qPCR analysis to
detect the relative mRNA expression.
| Gene | Primers |
|---|
| BMP2-F |
CGTCAAGCCAAACACAAACAGC |
| BMP2-R |
GAGCCACAATCCAGTCATTCCAC |
| VEGF165-F |
GCCTTGCTGCTCTACCTCCAC |
| VEGF165-R |
GCACACAGGATGGCTTGAAGATG |
| GAPDH Rat-F |
CCTTCCGTGTTCCTACCC |
| GAPDH Rat-R |
CAACCTGGTCCTCAGTGTAG |
Alkaline phosphatase (ALP) and alizarin
red staining
ALP (BCIP/NBT staining buffer) and alizarin red
staining were performed in each group (control, BMP2 and BMP2 +
VEGF165 groups) at 14 days subsequent to osteogenic differentiation
using osteogenic differentiation medium (R&D Systems,
Minneapolis, MN, USA). The samples were observed under a microscope
(Olympus IX5; Olympus Corp., Tokyo, Japan).
Analysis of ALP activity
The cells of each group were analyzed 3, 7 and 14
days subsequent to cell seeding for osteogenic differentiation. The
ALP activities in the cells were assessed using an ALP detection
kit (Nanjing Jiancheng Biotechnology Ltd., Nanjing, China). The
absorbance of the p-nitrophenol was quantified using an
enzyme-linked immunosorbent assay (ELISA) plate reader at a
wavelength of 495 nm, by correlating the fluorescence with
p-nitrophenol content using standards containing 0.1 mg/ml
p-nitrophenol.
Statistical analysis
The results are expressed as the mean ± standard
deviation (SD). The statistical analysis was conducted using
analysis of variance (ANOVA). P<0.05 was considered to indicate
a statistically significant difference.
Results
qPCR
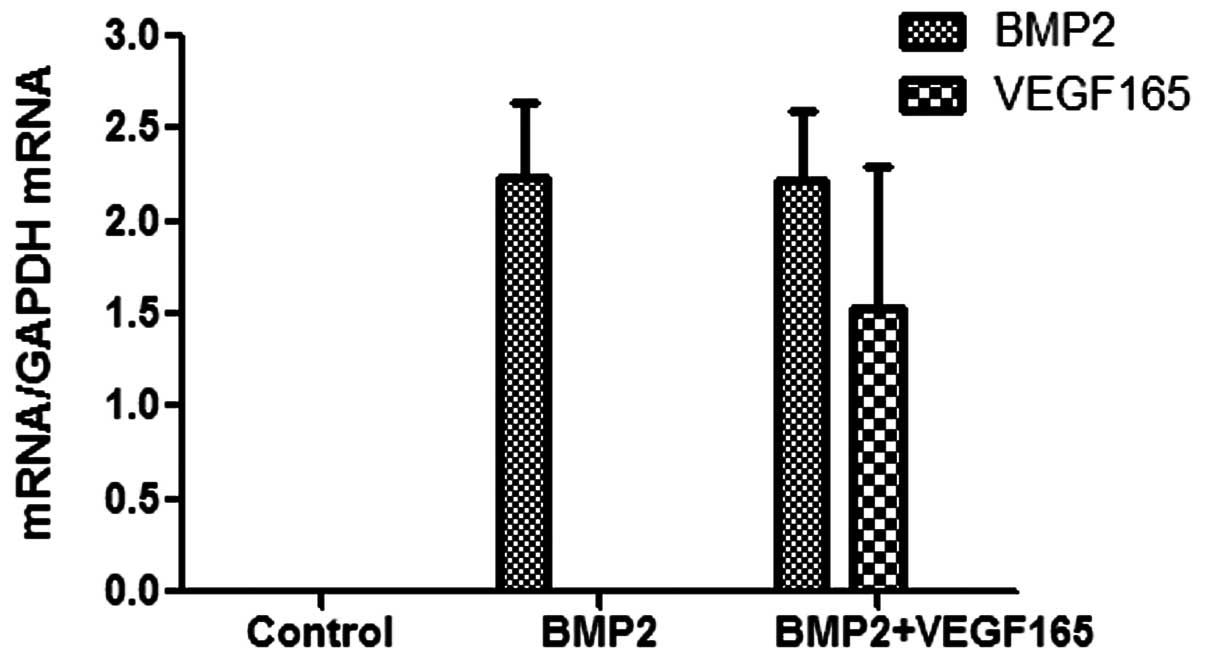
The qPCR results are shown in Fig. 2. The mRNA expression levels of the
BMP2 and VEGF165 genes in the control group were undetectable.
VEGF165 was immeasurable in the BMP2 group; however, VEGF165
expression was detected in the BMP2 + VEGF165 group. With regard to
BMP2 expression, BMP2 expression was detected in the BMP2 and the
BMP2 + VEGF165 group. Moreover, no significant difference in the
level of BMP2 expression was shown between the BMP2 + VEGF165 and
BMP2 groups (P>0.05).
Western blot analysis of BMP2 and VEGF165
secreted from the transfected MSCs
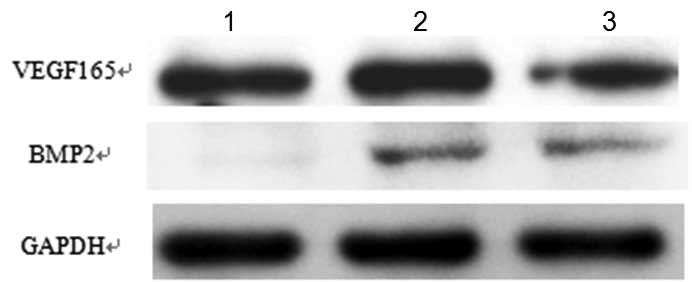
Western blot analysis indicated that the SD-MSCs
which were transfected with Lv-BMP2-VEGF165 or Lv-BMP2 secreted
large quantities of BMP2; however, the SD-MSCs which were not
transfected secreted low quantities, as shown in Fig. 3. By contrast, in all of the groups,
the SD-MSCs secreted large quantities of VEGF, irrespective of
whether they were transfected with Lv-BMP2-VEGF165 or Lv-BMP2, or
were not transfected.
ALP and alizarin red staining
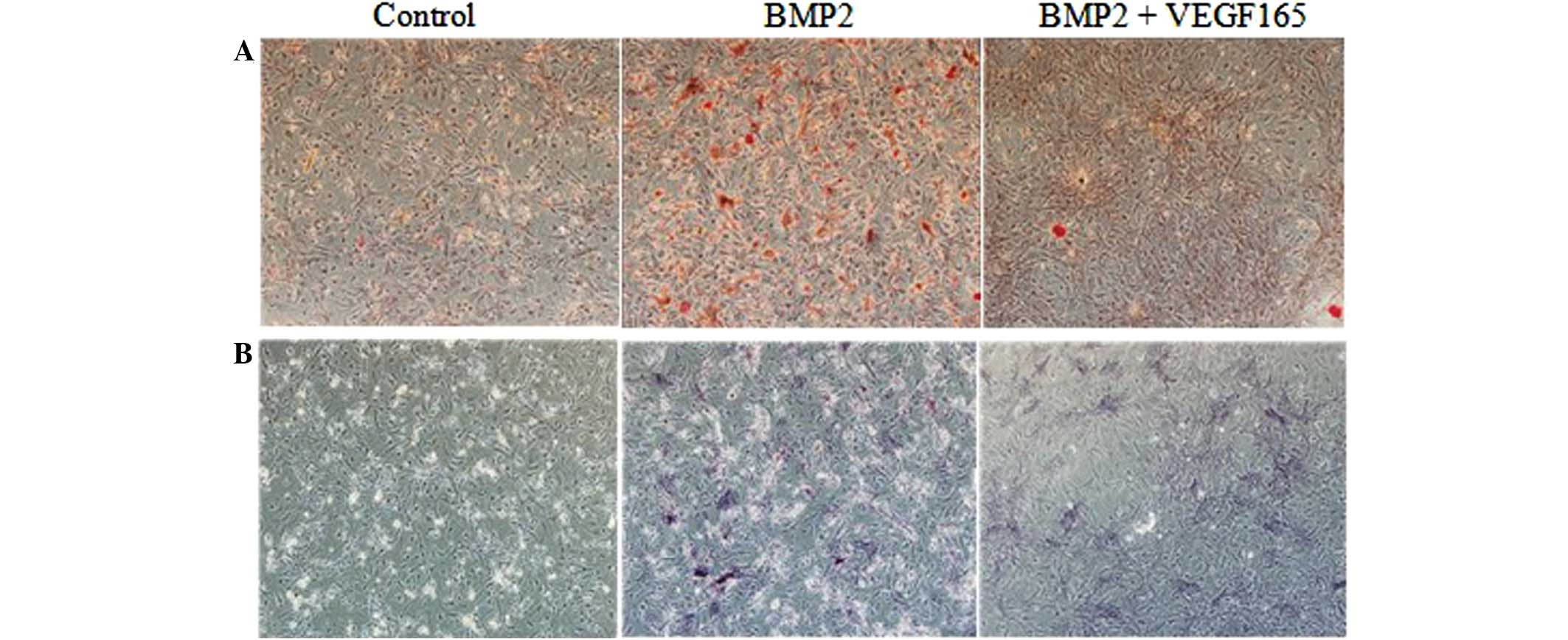
The results of alizarin red and ALP staining are
shown in Fig. 4. The data
indicated that the area exhibiting positive staining in the BMP2
group was significantly the largest among the three groups at 14
days subsequent to transfection. Furthermore, the area that
exhibited positive staining in the BMP2 + VEGF165 group was
significantly greater than that in the control group, although it
was not as large as that in the BMP2 group. The alizarin red and
ALP staining revealed negative results in the control group.
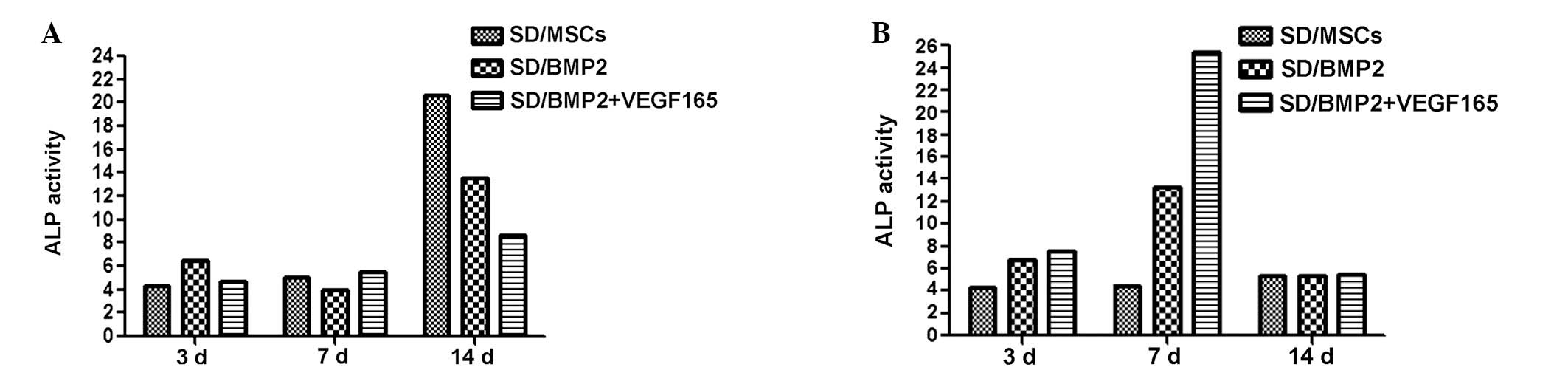
ALP activity assay
The ALP activity results at 3, 7 and 14 days
subsequent to culturing the cells with increasing of inducing
medium and normal medium are shown in Fig. 5A and B, respectively. With inducing
medium, the ALP activity was enhanced at 14 days in all groups
compared with that at 3 and 7 days (P<0.01). At 14 days, the ALP
activity of the BMP2 + VEGF165 group was notably suppressed
compared with that of the BMP2 group (P<0.01). However, there
were no differences between the groups at 3 and 7 days (Fig. 5A).
In the normal medium culture, the ALP activity was
significantly enhanced in the BMP2 + VEGF165 group following 7 days
incubation (P<0.01); however, this increase in activity was not
apparent at 3 and 14 days. In a comparison between the BMP2 and
BMP2 + VEGF165 groups, the ALP activity was significantly enhanced
in the BMP2 group at 7 days (P<0.01; Fig. 5B).
Discussion
Bone tissue engineering is a potential pathway for
bone regeneration, and bone marrow-derived MSCs are dominant seed
cell sources for bone engineering. MSCs possess the capability to
differentiate into bone, cartilage, muscle and fat when supplied
with nutrition and different types of growth factors (8). Establishing a stabilized surrounding
environment, which enables tissue formation, delays the ageing
process of seeding cells and promotes the activities of cell
proliferation and differentiation, is crucial, whether the process
occurs in vivo or in vitro. The development of gene
therapy and transgenic technology has provided a viable means with
which to tackle the previously mentioned problems (9).
BMPs regulate bone and cartilage differentiation by
the isolation, cloning and expression of genes (8). Lieberman et al (10) demonstrated that BMP2-producing
cells, via adenoviral gene transfer, produced sufficient protein to
heal segmental bone defects. The present study also indicates the
significance of using MSCs transduced with BMP2 for the repair of
segmental defects. Compared with the use of MSCs alone, bone
regeneration is accelerated by BMP2-expressing MSCs.
BMP2 has been shown to be unable to increase the
rate of bone healing, due to inadequate vascularization in certain
critical-sized bone defects (11).
Previous studies have shown that, following treatment with BMP2,
25% of non-union fractures required a secondary bone graft
procedure, due to the lack of adequately vascularized tissue
(12,13). Moreover, Furumatsu et al
(14) demonstrated that
vascularization is a critical problem in tissue engineering.
VEGF has been characterized as a heparin-binding
angiogenic factor with specific mitogenic actions on endothelial
cells (15). VEGF, which induces
endothelial cell proliferation, angiogenesis and capillary
permeability, is produced in a regulated manner by osteoblasts
(16). VEGF-A has four isomers,
which are composed of 121, 165, 189 and 206 amino acids,
respectively. Among these isomers, VEGF165 has the most potent
activity. A previous study only used the isomer of VEGF that
resulted the weakest induced activity (17). In the present study, the VEGF165
isomer was used, which has been shown to lead to the most
efficacious induction in activity. The process of bone metabolism
includes bone formation and resorption, which are regulated by
osteoblasts and osteoclasts, respectively. It has been demonstrated
that VEGF is an essential coordinator of extracellular matrix
remodeling, angiogenesis and bone formation in the growth plate
(18). However, the role of VEGF
production in osteoblasts has recently been widely discussed.
Schönmeyr et al (19)
revealed that VEGF was a potent inhibitor of BMP2 expression in
MSCs, and that supplementation with or overexpression of VEGF
inhibited BMP2 mRNA expression, protein production and MSC
differentiation. In the present study the most positive alizarin
red and ALP staining results were in the BMP2 group, which showed
that the effect of BMP2 on bone regeneration was stronger than that
of BMP2 and VEGF165 combined. The result suggested that the
overexpression of VEGF inhibited the action of BMP2 in osteogenesis
in vitro. Similarly, in the inducing culture medium, the ALP
activity of the BMP2 + VEGF165 group was notably suppressed
compared with that of the BMP2 group. However, the inhibition
occurred at 14 days, which was inconsistent with the results of a
previous study, in which the inhibition occurred at 21 days
(20). This inconsistency may be
due to a number of reasons, including cell line differences. We
propose that the primary reason was that VEGF required interactions
with various factors involved in bone formation to induce a greater
effect. Song et al (21)
indicated that the regulation of Id1 expression by VEGF and BMP2
may be critical to cell and gene-based approaches for bone
regeneration. However, the specific role of VEGF in bone healing
has yet to be elucidated. The ALP activity was higher at 14 days in
inducing medium compared with that in normal medium, which
indicated that there may be certain compositions that are able to
promote the induction of rat bone marrow MSC differentiation.
In conclusion, the present study, which constructed
BMP2 and VEGF-modified bone for tissue engineering through
lentiviral transfection, revealed important implications for novel
therapeutic strategies to enhance bone regeneration. BMP2 was shown
to be important in bone regeneration in the in vitro study.
The overexpression of VEGF inhibited BMP2-induced MSC
differentiation and osteogenesis in vitro. Whether or not
local VEGF gene therapy is likely to affect bone regeneration in
vivo has yet to be elucidated. A further study is ongoing.
Acknowledgements
This research was supported by the Guangzhou Science
and Technology Plan Projects (2011J5200002).
References
|
1
|
Wang Y, Zeng B and Li X: Expression of
human calcitonin by microencapsulated recombinant myoblasts.
Biotechnol Lett. 28:1453–1458. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Roldán JC, Detsch R, Schaefer S, et al:
Bone formation and degradation of a highly porous biphasic calcium
phosphate ceramic in presence of BMP-7, VEGF and mesenchymal stem
cells in an ectopic mouse model. J Craniomaxillofac Surg.
38:423–430. 2010.PubMed/NCBI
|
|
3
|
Peng H, Wright V, Usas A, et al:
Synergistic enhancement of bone formation and healing by stem
cell-expressed VEGF and bone morphogenetic protein-4. J Clin
Invest. 110:751–759. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kiyozuka Y, Miyazaki H, Yoshizawa K, et
al: An autopsy case of malignant mesothelioma with osseous and
cartilaginous differentiation: bone morphogenetic protein-2 in
mesothelial cells and its tumor. Dig Dis Sci. 44:1626–1631. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zelzer E, McLean W, Ng YS, et al: Skeletal
defects in VEGF(120/120) mice reveal multiple roles for VEGF in
skeletogenesis. Development. 129:1893–1904. 2002.PubMed/NCBI
|
|
6
|
Deckers MM, van Bezooijen RL, van der
Horst G, et al: Bone morphogenetic proteins stimulate angiogenesis
through osteoblast-derived vascular endothelial growth factor A.
Endocrinology. 143:1545–1553. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tsuda H, Wada T, Yamashita T and Hamada H:
Enhanced osteoinduction by mesenchymal stem cells transfected with
a fiber-mutant adenoviral BMP2 gene. J Gene Med. 7:1322–1334. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Reddi AH: Bone and cartilage
differentiation. Curr Opin Genet Dev. 32:737–744. 1994. View Article : Google Scholar
|
|
9
|
Ishihara A and Bertone A: Cell-mediated
and direct gene therapy for bone regeneration. Expert Opin Biol
Ther. 12:411–423. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lieberman JR, Daluiski A, Stevenson S, et
al: The effect of regional gene therapy with bone morphogenetic
protein-2-producing bone-marrow cells on the repair of segmental
femoral defects in rats. J Bone Joint Surg Am. 81:905–917.
1999.PubMed/NCBI
|
|
11
|
Xiao C, Zhou H, Liu G, et al: Bone marrow
stromal cells with a combined expression of BMP-2 and VEGF-165
enhanced bone regeneration. Biomed Mater. 6:0150132011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bostrom MP, Saleh KJ and Einhorn TA:
Osteoinductive growth factors in preclinical fracture and long bone
defects models. Orthop Clin North Am. 30:647–658. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kujala S, Raatikainen T, Ryhänen J, et al:
Composite implant of native bovine bone morphogenetic protein (BMP)
and biocoral in the treatment of scaphoid nonunions - a preliminary
study. Scand J Surg. 91:186–190. 2002.PubMed/NCBI
|
|
14
|
Furumatsu T, Shen ZN, Kawai A, et al:
Vascular endothelial growth factor principally acts as the main
angiogenic factor in the early stage of human osteoblastogenesis. J
Biochem. 33:633–639. 2003. View Article : Google Scholar
|
|
15
|
Murphy WL, Peters MC, Kohn DH and Mooney
DJ: Sustained release of vascular endothelial growth factor from
mineralized poly(lactide-co-glycolide) scaffolds for tissue
engineering. Biomaterials. 21:2521–2527. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Goad DL, Rubin J, Wang H, et al: Enhanced
expression of vascular endothelial growth factor in human SaOS-2
osteoblast-like cells and murine osteoblasts induced by
insulin-like growth factor I. Endocrinology. 137:2262–2268.
1996.PubMed/NCBI
|
|
17
|
Kempen DH, Lu L, Heijink A, et al: Effect
of local sequential VEGF and BMP-2 delivery on ectopic and
orthotopic bone regeneration. Biomaterials. 30:2816–2825. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wu GP, He XC, Yang ZH and Guo L: Influence
on the osteogenic activity of the human bone marrow mesenchymal
stem cells transfected by liposome-mediated recombinant plasmid
pIRES-hBMP2-hVEGF165 in vitro. Ann Plast Surg. 65:80–84. 2010.
View Article : Google Scholar
|
|
19
|
Schönmeyr BH, Soares M, Avraham T, et al:
Vascular endothelial growth factor inhibits bone morphogenetic
protein 2 expression in rat mesenchymal stem cells. Tissue Eng Part
A. 16:653–662. 2010.PubMed/NCBI
|
|
20
|
Kuroda S, Sumner DR and Virdi AS: Effects
of TGF-β1 and VEGF-A transgenes on the osteogenic potential of bone
marrow stromal cells in vitro and in vivo. J Tissue Eng.
3:20417314124597452012.
|
|
21
|
Song XB, Liu SH, Qu X, et al: BMP2 and
VEGF promote angiogenesis but retard terminal differentiation of
osteoblasts in bone regeneration by up-regulating Id1. Acta Biochim
Biophys Sin (Shanghai). 43:796–804. 2011. View Article : Google Scholar : PubMed/NCBI
|