Introduction
Gastroparesis is a common symptom in patients with a
long history of diabetes. Between 30 and 50% of patients that have
had diabetes for >10 years exhibit such symptoms of gastric
neuromuscular dysfunction as satiety, weight loss, abdominal pain,
distension and discomfort, nausea and vomiting. Neuropathological
changes, a reduction in the number of gastrointestinal interstitial
cells of Cajal (ICCs), morphological changes, hyperglycemia,
microvascular diseases, microcirculatory disorders,
gastrointestinal hormone changes and Helicobacter pylori are
all believed to be involved in the pathogenesis of diabetic
gastroparesis (DGP) (1–7). The inherent gastric nerves have
recently been subject to significant focus, and it has been
suggested that the most important mechanism of DGP is the
deficiency of neuronal nitric oxide synthase expression and the
reduction of ICCs (1–7).
Electroacupuncture (EA) is a method which stimulates
the acupoints via electric currents using acupuncture needles.
Compared with manual acupuncture, EA is more readily controllable
and easier to standardize, and it allows stronger and more
continuous stimulation with less pain and tissue damage (8). In addition, its effects are more rapid
and longer lasting (8). Studies have
indicated that, EA can effectively reduce the symptoms of
gastroparesis, including nausea, vomiting and abdominal distension,
and promote gastric emptying in patients with diabetes (9,10).
Furthermore, EA has been shown to increase the number of ICCs in
the colons of slow-transit constipated rats (11); EA stimulation towards ‘Zusanli’, one
of the most frequently used acupuncture points, may increase the
electrical activity of ICCs, thus improving the gastric electrical
activity and increasing the gastric motility (12,13). It
has yet to be fully elucidated, however, whether the alleviation of
DGP symptoms in rats by EA is associated with improvements in the
gastric neuromuscular function and alterations in the numbers and
morphology of gastric ICCs. The present study investigated the
effect of EA on the numbers and morphology of gastric ICCs, with
the aim of exploring the mechanism of EA and facilitating its
potential use in alleviating the symptoms of patients with DGP.
Materials and methods
Animal models
Male Sprague Dawley rats, aged 8 weeks and each
weighing 180–220 g, were provided by the Experimental Animal Center
of Guangzhou University of Chinese Medicine, (Guangzhou, China;
certificate no. 0055684). The rats were bred in a
specific-pathogen-free laboratory and left to adapt for 3 days
under a 12-h light/dark cycle, with the temperature at 20–28°C and
the humidity at 75–90%. The rats were fed a normal diet, with free
access to drinking water. Following adaptation, 25 of the rats were
randomly allocated to the blank group and fed the basal diet, while
the remaining rats (n=50) were fasted for 12 h. Streptozotocin
(STZ; Sigma-Aldrich, St. Louis, MO, USA) was dissolved in 0.l
mmol/l sodium citrate buffer (pH 4.5; Guangzhou Chemical Reagent
Factory, Guangzhou, China; batch no. 20081202-2) to prepare a 1%
solution. The solution was then intraperitoneally injected in one
step into the 50 rats (50 mg/kg). Two days later, the fasting blood
glucose (FBG) of the rats was measured using an
Accu-Chek® Advantage blood glucose meter and test strip
(Roche, Indianapolis, IN, USA). If the 72-h FBG was >200 mg/dl
and persisted at this level for 2 weeks, it indicated that the
diabetes model was successfully established (14,15). A
high-fat, high-sugar diet was then fed to the model rats in
accordance with the irregular diet method, in which the rats were
fed the high-fat diet on the morning of an odd date and the
afternoon of an even date, for 10 weeks.
There were significant differences in
gastrointestinal propulsive indicators between the model and blank
groups; the gastrointestinal propulsive indicators were calculated
using the following formula: Gastrointestinal propulsive rate =
[distance from C-powder front to pyloric sphincter (cm)/distance
from pyloric sphincter to end of small intestine (cm)] ×100%
(statistical method: One-way analysis of variance). These
differences, taken together with the changes in the stool quantity
and characteristics, indicated that the DGP model was successfully
established. Following the successful establishment of the DGP
model, the rats were then randomly divided into the model group
(without any treatment) and the EA group (EA treatment once/day for
5 continuous days, followed by 2 days rest, for a total of 19
days). Each group contained 25 rats.
Treatment method
Each EA rat was placed in the homemade rat fixer,
and the acupoints of Housanli, Zhongwan and Weiyu were disinfected
with 75% alcohol. The disinfection was performed on the left and
right sides in turn, and the acupoint selection method was based on
the ‘Experimental Animal Acupoint Map’ developed by Hua et
al (16). Sterile acupuncture
needles were selected (Qiu model 30; needling depth, 0.76–1.0 cm;
Suzhou Huatuo Medical Supplies Company, Suzhou, China). EA was
performed with a Han's Acupoint Nerve Stimulator (LH202H; Shanghai
TCM Co., Ltd., Shanghai, China); the stimulating waveforms used
15-Hz waves, and the intensity was set to make the acupoint skin
jitter slightly. The needle was maintained for 15 min. Following
the treatment, the rats could eat normally.
Sample collection and handling
Following the EA treatment, the rats underwent
spinal dislocation, followed by a laparotomy to obtain a
rectangular strip of antrum (2.0×0.5 cm2). The antrum
was placed in 10% neutral formalin for the fixation, and then
dehydrated, paraffin-embedded and sliced into sections (a total of
four consecutive slices, each with a thickness of 4 µm). The slices
were dried at 60°C for 30 min, and two samples from each rat were
randomly selected for hematoxylin and eosin (HE) staining. The 4-µm
sections were dewaxed using xylene, dehydrated with graded alcohol,
stained with 1% HE and then rinsed with twice-distilled water,
dehydrated, hyalinized, dried and mounted using neutral gum.
Immunohistochemical staining
In order to assess the number and distribution of
the ICCs, the samples were subjected to anti-c-kit monoclonal
antibody immunohistochemical staining (15) and observed using light microscopy.
The immunohistochemical staining steps were performed at room
temperature. The 4-µm thick paraffin samples were attached onto
pre-coated poly-lysine slides, baked at 60°C for l h and at then at
37°C for 2 days. The slices were subsequently dewaxed, dehydrated
and rinsed twice (for 3 min each time) with 0.0l mol/l pH 7.4
phosphate-buffered saline (PBS). The same rinsing method was
performed following the steps to block the endogenous peroxidase,
expose and retrieve the antigens and overnight storage at 4°C.
Endogenous peroxidase activity was blocked via incubation with
hydrogen peroxide (3%) for 15 min. For antigen exposure, proteinase
K was applied at 37°C for 20 min, and the slices were then placed
into the antigen retrieval box, which was filled with pH 6.0 citric
acid buffer. Antigen retrieval was performed by microwaving the
samples at 5,883.99 watts for 9 min. The samples were then removed
and allowed to cool down to room temperature. Normal goat serum
(10%) was used to block the slices for 15 min, prior to the serum
being removed and the liquid surrounding the slices being dried
under suction. The slices were not washed. Rabbit anti-c-Kit
polyclonal primary antibody (cat. no. bs-10005R; Beijing Bioss
Biosynthesis Biotechnology Co. Ltd., Beijing, China) was diluted to
1:200 with 0.01 M PBS, and pipetted onto the slice. The simple
addition of 0.01 M PBS was used as the negative control. Each slice
was then placed into the wet box and stored overnight at 4°C. Goat
anti-rabbit IgG secondary antibody (cat. no. bs-0295G-SA; dilution,
1:200; Beijing Donglin Changsheng Biotechnology Co. Ltd., Beijing,
China) was then pipetted onto the slice, placed into the wet box
and stored overnight at 4°C. 3,3′-Diaminobenzidine was then added
to the slice and observed under the light microscope for 3–10 min
until positive staining could be observed. The slices were then
washed with distilled water, stained, dehydrated and mounted.
Microscopy
The number and distribution of the ICCs was assessed
under using light microscopy. A micrometer-scale eyepiece was
installed on a light microscope (LSM 510 META laser scanning
confocal microscope; Carl Zeiss AG, Oberkochen, Germany) and used
for the observation of the myenteric nerve plexus and smooth
muscles at low (x10) and high (x40) magnification, as well as the
number and intensity of c-kit-positive cells. The 8502 image
analyzer (Tracor Northern, Middleton, WI, USA) was used: Brownish
yellow-stained regions were set as the positive, and the percentage
of positive regions in each field was calculated to express the ICC
content.
An electron microscope (H-7500 transmission electron
microscope; Hitachi, Tokyo, Japan) was used for the tissue
observations. Full-layer gastric tissues (~1 cm2; 0.5 cm
to the pylorus) were cut into 1×1 mm2 tissue blocks, and
stored in ice-cold 3% glutaraldehyde for 2–4 h. The tissue samples
were then rinsed with 0.1 mol/l PBS for 10 min twice, fixed with 1%
osmic acid for 1.5–2 h and rinsed with 0.1 mol/l PBS for 5 min
twice. Ethanol at 50 and 70% was used for the gradient dehydration,
with 10 min at each level. The samples were then dehydrated with
80, 90 and 100% acetone, for 10 min at each level, twice. Epoxy
resin and Epon 812 embedding agent were mixed in a ratio of 1:1 and
replaced 100% acetone for 40 min; the Epon 812 was then used to
soak the tissues at 37°C overnight. The tissue blocks were
subsequently embedded in the Epon 812 embedding agent and
polymerized for 48 h at 60°C. AO ultramicrotome was used to prepare
1-µm ultra thin slices (American Optical, Southbridge, MA, USA),
flattened on water. The flattening procedure was performed as
follows: The slices were transferred onto a drop of water added to
a glass slide, heated to make the slices flat. Finally, the slices
were subjected to HE staining. The slices were positioned under the
dissecting and ordinary microscopes. The AO ultramicrotome was used
to prepare the 40- to 60-nm ultra thin slices, and the slices were
collected with a copper mesh, followed by double-staining with 70%
ethanol-prepared saturated uranyl acetate and lead solution. Once
the slices had dried, the slices were observed and the images
recorded by electron microscopy.
Statistical analysis
All statistical analysis was carried out using SPSS
17.0 software (SPSS, Inc., Chicago, IL, USA). Data are presented as
the mean ± standard deviation. The comparisons among different
groups were performed using the t-test. P<0.05 was considered to
indicate a statistically significant difference.
Results
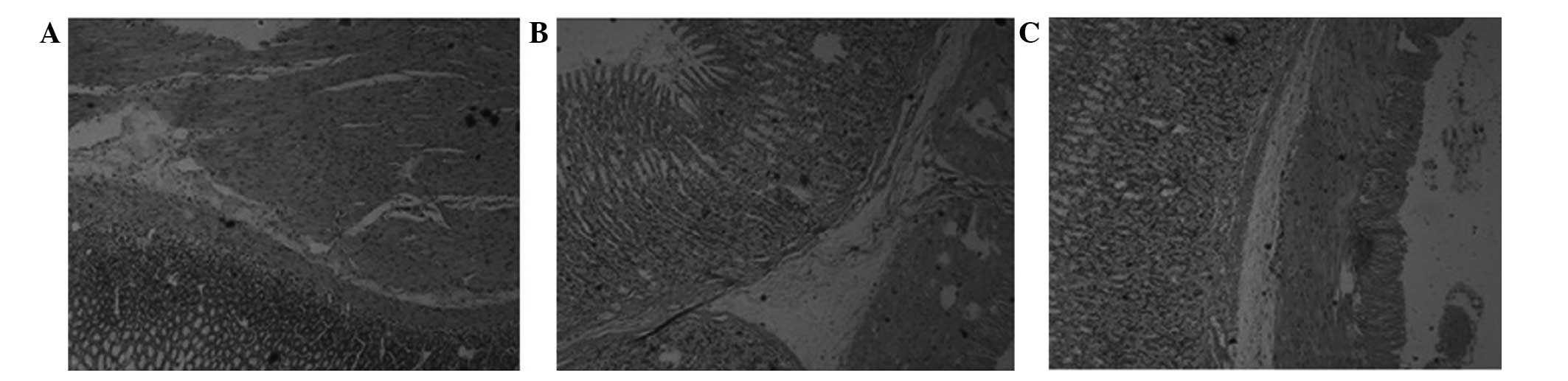
HE staining of gastric tissues
The HE staining results are shown in Fig. 1. The gastric glandular cells and
smooth muscle cells (SMCs) of the blank group were arranged
regularly and tightly, with moderate intercellular gaps and no
vacuolar degeneration or vascular dilation (Fig. 1A). In comparison, the glands of the
model group were misaligned, the intracellular gaps were larger and
the glandular cytoplasm was reduced in amount and lightly stained
(Fig. 1B). Vacuolar degeneration
appeared, the mucosa and submucosa were congested, the blood
vessels were dilated, the cytoplasm of the muscular gastric wall
was lightly stained and translucent, and vacuolar changes were
exhibited compared with the control group. In the EA group,
compared with the model group, the vascular congestion in the
gastric mucosa and submucosa was mitigated, the mucosal gland cells
and SMCs were arranged more neatly, and with fewer vacuoles, the
intercellular gaps were marginally larger and only minor vacuolar
degeneration was observed in the gastric wall (Fig. 1).
c-kit immunohistochemical
staining
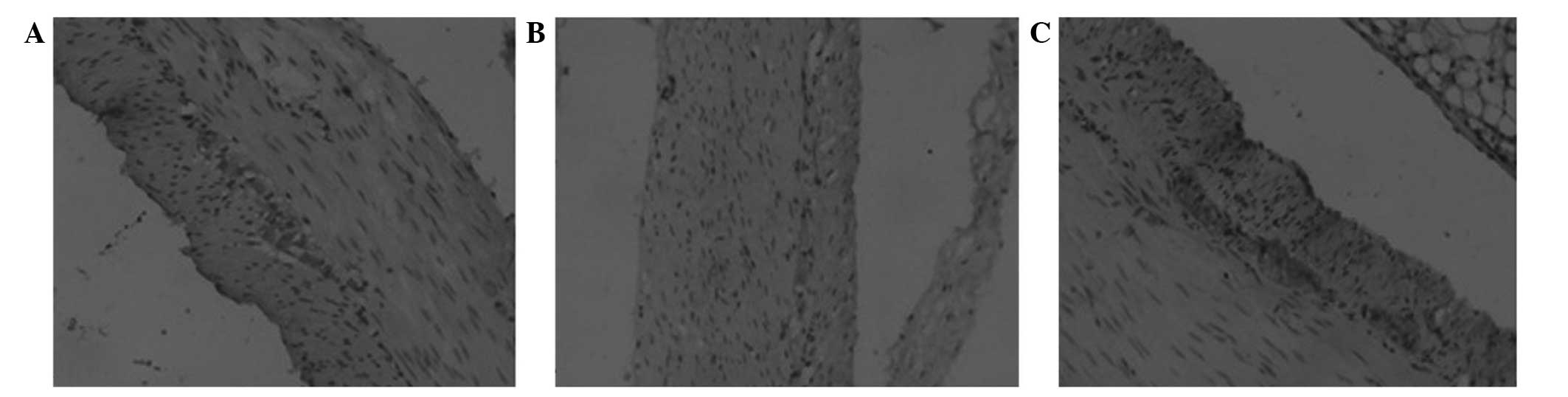
Distribution and morphology
Following the c-kit immunohistochemical staining,
observation under the light microscope revealed that the cell
membrane and cytoplasm of the ICCs were stained brown (Fig. 2). The positively stained ICCs were
most dense in the gastric antrum of the blank group. The cells were
located between the circular and longitudinal muscle layers, and
between and on the inner surface of the circular muscle layers, in
particular between the ring and longitudinal muscle layers. The
cells were mostly accompanied by nerve fiber endings and nerve
bundles, and connected together tightly by gap junctions,
exhibiting an aggregated distribution. The cells were generally
fusiform, elongated or irregular, and exhibited numerous cell
processes, little cytoplasm and large nuclei, which were mostly
oval and deeply stained. The ICCs of the model group were atrophic
and significantly reduced in number; the cell processes had
disappeared and the cell contrasts were poor. The tight junctions
between the ICCs, nerve cells and SMCs were significantly reduced.
The ICCs of the EA group were marginally decreased in number
compared with those of the blank group, although the cell
morphology was basically normal. The cells exhibited shortened
processes, and the tight junctions between the ICCs and the other
cells remained.
Comparison of quantity and intensity of
c-kit-positive cells in each group
Image analysis showed that the largest number of
positive ICCs was found in the blank group. Statistically
significant differences existed among the model, EA and blank
groups: Blank group > EA group > model group (P<0.01).
These findings indicated that EA regulated the number of
c-kit-positive ICCs. The positive area percentage of the normal
blank group was the highest (Table
I).
 | Table I.Comparison of the quantity and
intensity of gastric c-kit-positive cells among the three
groups. |
Table I.
Comparison of the quantity and
intensity of gastric c-kit-positive cells among the three
groups.
| Group | Number of positive
cells | Positive area
percentage |
|---|
| Blank | 32.33±5.51 | 0.358±0.153 |
| Model |
18.67±6.11a |
0.127±0.074c |
| EA |
31.33±4.73b |
0.290±0.123d |
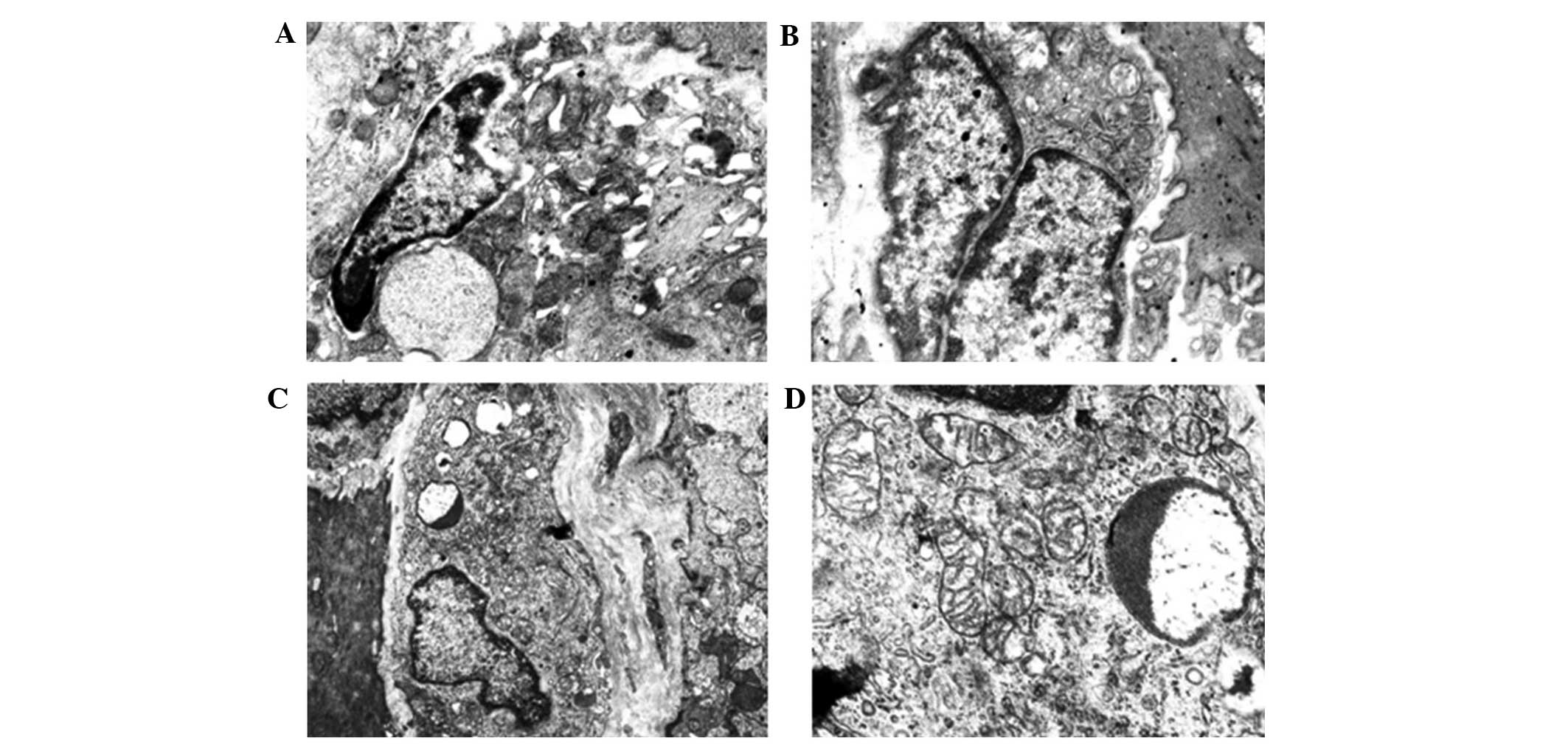
Ultrastructural features of gastric ICCs
The gastric ICCs of the blank-group rats were mainly
distributed in the myenteric nerve plexus of the circular and
longitudinal muscle layers, as well as among the circular muscle
cells, and they were closely linked by gap junctions. Many foveolae
were distributed along the cell membrane, and the basal cell
membrane was complete. In the majority of cells, large, irregular
nuclei could be observed; heterochromatin was common and was
distributed along and under the nuclear membrane. The structure was
well-defined, with little cytoplasm. One or more processes were
present, and the processes were rich in cytoplasm and organelles,
particularly mitochondria, which were closely arranged inside the
cytoplasm. Fully developed rough endoplasmic reticulum, smooth
endoplasmic reticulum and Golgi apparatus could be observed,
accompanied by numerous intermediate filaments but no rough
filaments (Fig. 3).
In the model group, the gap junctions among the
ICCs, SMCs and nerve endings were significantly reduced, and the
structure of the surviving cells was destroyed. The cells exhibited
an unclear and loosely connected structure, with an incomplete
basal cell membrane that lacked continuity. Regions of the basal
membrane were observed to have separated from the cell membrane and
formed cavities. The numbers of intracellular organelles, e.g.
ribosomes and mitochondria, were significantly reduced compared
with the normal ICCs; furthermore, the mitochondria showed
significant swelling, vacuolar degeneration and myelin-like
degeneration or even dissolution. The endoplasmic reticulum was
expended, the rough endoplasmic reticulum was degranulated and the
intracellular cytoplasm had extensively dissolved, leading to
considerable vacuolization inside the cytoplasm, which was mostly
distributed along the vacuolar membrane. In addition, the
perinuclear gaps had widened (Fig.
3).
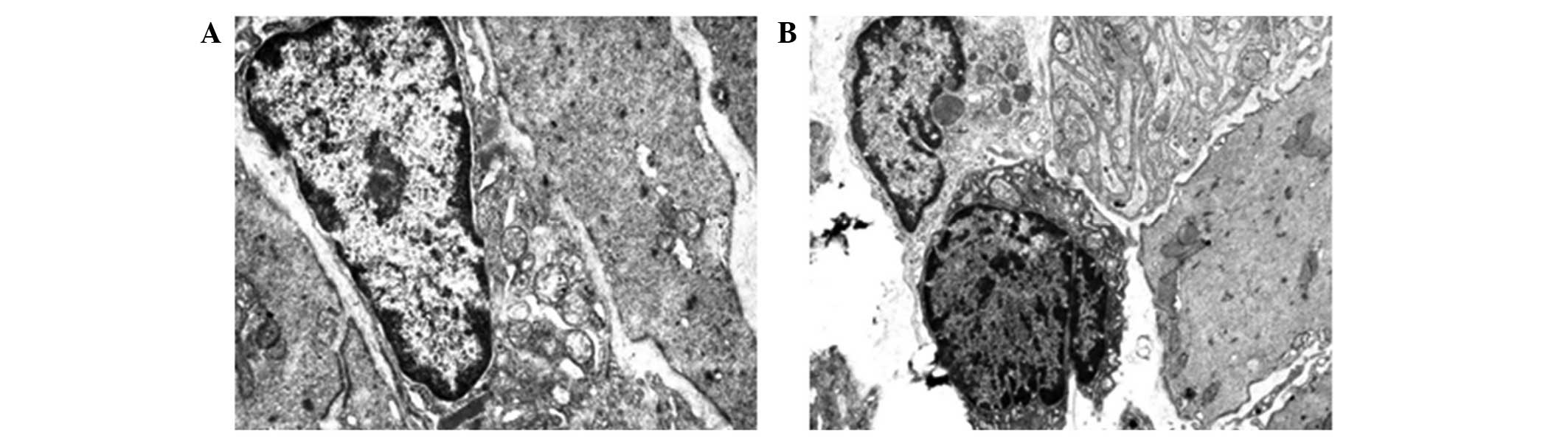
The number of ICCs in the EA group was more than
that in the model group, while slightly less than that in the
control group. The morphological structure of the ICCs was
basically normal, although the mitochondria were reduced in number
compared with the blank group and exhibited mild swelling, and the
gaps among the cells remained. The pathological damage to the ICCs,
nerve endings and SMCs was less than that in the model group, and
the structures were more distinct and ordered. The pathological
damage to the nerves within the wall and the cell processes was
also reduced compared with that in the model group, and the ICC
distribution was improved. The gastric capillary walls were not
smooth, while the endothelial cell swelling, as well as the luminal
congestion and bending, were reduced, and the vacuolar degeneration
was less (Fig. 4).
Discussion
In a diabetic state in rats, the gastric gland
cells, SMCs in the gastromuscular layers, circular muscle nerve
cells and ICCs exhibit varying degrees of damage. The observations
in the present study revealed that, compared with normal rats, the
glands were arranged in a disorderly manner, the cellular gaps were
larger, the glandular cytoplasm was reduced and lightly stained,
and vacuolar degeneration was apparent. The mucosa and submucosa
were congested and the vascular lumens were expended. The SMCs
exhibited a disordered arrangement, and several dissolved vacuoles
were observed within the muscular cytoplasm. The mitochondria were
swollen and exhibited vacuolar degeneration or had dissolved. The
neuronal nuclei inside the myenteric plexus were dissolved and
necrotic. The cytoplasm was partially dissolved, there were fewer
organelles and a number of autophagic vacuoles could be observed
within the cytoplasm. The reduction in the number of organelles
within the ICCs was consistent with the literature (3,4,7,17). In
addition, the peripheral axons and dendrites were partially swollen
or dissolved, the electron density was not uniform, and vacuoles
had formed. The synaptic vesicle content inside the nerve
varicosity was significantly reduced, and the number of gastric
ICCs was significantly decreased compared with the blank group.
Furthermore, the connections among the ICCs, SMCs and nerve endings
were significantly reduced, the connections among the surviving
ICCs were loose, the gaps were widened and the perinuclear
cytoplasm of the ICCs was significantly reduced. The changes in the
number of ICCs may have been associated with the chronic high blood
sugar or insulin resistance in diabetes (18,19).
Due to the arrangement of the muscle cells in the
DGP model, the cells lacked coordination, which would have strongly
affected their contraction and transmission functions; the severe
damage to the structure of the muscle cells would also have been
associated with a decline in function. At the same time, the energy
supply of the muscle cells would have been affected due to lesions
within the mitochondria. The reduction in cell function due to the
above factors would have led to the inevitable result of a series
of gastrointestinal motility disorder reactions, such as stomach
dysrhythmia, increased gastric relaxation, easy expansion,
weakness, propagation delay, wall thickening and luminal expansion
(20,21). Delayed gastric emptying is a
complication that can lead to the difficulty of blood control in
diabetes (22).
Compared with the model group, the mucosal and
submucosal vascular congestion of the EA group were alleviated. In
addition, the mucosal gland cells were arranged in an orderly
fashion with fewer vacuoles; the cell gaps were marginally larger,
and there only existed a small amount of vacuolar degeneration on
the gastric wall. The gastric SMCs of the EA-group rats were more
orderly arranged than those of the model group; the large, soluble
vacuoles within the cytoplasm of the muscle cells were rare and the
mitochondrial swelling was not obvious. The electron densities in
the gastric neurite and dendrite endings remained uniform. The
myenteric plexus neurons were clear and the organelles, including
the mitochondria and rough endoplasmic reticulum, remained
abundant. It was also found that the number of gastric ICCs in the
EA group was greater than that in the model group. It has been
suggested that the acceleration of intestinal transition may
increase the numbers of ICCs (23).
The pathological damage to the ICCs, as well as the nerve endings
between ICCs and SMCs, was less than that in the model group, and
the structures were more distinct and tightly connected. The
distribution of intramural nerves and ICCs, as well as the
pathological damage to the process connections, was reduced
compared with that in the model group. The gastric capillary wall
of the rats was uneven; the endothelial cell swelling and luminal
congestion and bending were reduced; and there was less vacuolar
degeneration.
The changes of gastric ICCs in the model group
observed in the present study are consistent with the results of
previous studies (24,25). EA can reduce the pathological damage
to the gastric antrum in DGP rats, and has been shown to have a
positive effect on the number and morphology of ICCs. A limitation
of the present study, however, was that the findings did not
clarify whether this effect is achieved through the acupuncture,
local action of electric stimulation, or both; therefore, the
precise mechanism of the effects of EA on ICCs, requires further
investigation.
The experimental results of the present study showed
that EA could adjust the ultrastructural changes of the gastric
ICCs of the DGP model group, thus accelerating gastric emptying and
improving the gastric motility disorders of the DGP rats. In
conclusion, EA could reverse the cellular pathological damage to
gastric tissue layers in the STZ diabetic rat model.
References
|
1
|
Forster J, Damjanov I, Lin Z, Sarosiek I,
Wetzel P and McCallum RW: Absence of the interstitial cells of
Cajal in patients with gastroparesis and correlation with clinical
findings. J Gastrointest Surg. 9:102–108. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Grover M, Farrugia G, Lurken MS, Bernard
CE, Faussone-Pellegrini MS, Smyrk TC, Parkman HP, Abell TL, Snape
WJ, Hasler WL, et al: NIDDK Gastroparesis Clinical Research
Consortium: Cellular changes in diabetic and idiopathic
gastroparesis. Gastroenterology. 140:1575–1585. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Oh JH and Pasricha PJ: Recent advances in
the pathophysiology and treatment of gastroparesis. J
Neurogastroenterol Motil. 19:18–24. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ordög T, Takayama I, Cheung WK, Ward SM
and Sanders KM: Remodeling of networks of interstitial cells of
Cajal in a murine model of diabetic gastroparesis. Diabetes.
49:1731–1739. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gangula PR, Sekhar KR and Mukhopadhyay S:
Gender bias in gastroparesis: Is nitric oxide the answer? Dig Dis
Sci. 56:2520–2527. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Thazhath SS, Jones KL, Horowitz M and
Rayner CK: Diabetic gastroparesis: Recent insights into
pathophysiology and implications for management. Expert Rev
Gastroenterol Hepatol. 7:127–139. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Iwasaki H, Kajimura M, Osawa S, Kanaoka S,
Furuta T, Ikuma M and Hishida A: A deficiency of gastric
interstitial cells of Cajal accompanied by decreased expression of
neuronal nitric oxide synthase and substance P in patients with
type 2 diabetes mellitus. J Gastroenterol. 41:1076–1087. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang C P, Kao C H, Chen W K, et al: A
single-blinded, randomized pilot study evaluating effects of
electroacupuncture in diabetic patients with symptoms suggestive of
gastroparesis. J Altern Complement Med. 14:833–839. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang CP, Kao CH, Chen WK, Lo WY and Hsieh
CL: A single-blinded, randomized pilot study evaluating effects of
electroacupuncture in diabetic patients with symptoms suggestive of
gastroparesis. J Altern Complement Med. 14:833–839. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yang M, Li X, Liu S, Li Z, Xue M, Gao D,
Li X and Yang S: Meta-analysis of acupuncture for relieving
non-organic dyspeptic symptoms suggestive of diabetic
gastroparesis. BMC Complement Altern Med. 13:3112013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lyford GL, He CL, Soffer E, Hull TL,
Strong SA, Senagore AJ, Burgart LJ, Young-Fadok T, Szurszewski JH
and Farrugia G: Pan-colonic decrease in interstitial cells of Cajal
in patients with slow transit constipation. Gut. 51:496–501. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sun JH, Guo H, Chen L, Wu XL, Li H, Pei
LX, Peng YJ and Lu B: Effect of electroacupuncture at ‘Tianshu’ (ST
25) on colonic smooth muscle structure and interstitial cells of
Cajal in slow transit constipation rats. Zhen Ci Yan Jiu.
36:171–175. 2011.(In Chinese). PubMed/NCBI
|
|
13
|
Yang Q, Chen HQ, Huang YX, Wang W and Wang
JJ: Effect of electroacupuncture-regulated gastric motility on
interstitial cells of Cajal. Progr Mod Biomed. 9:1676–1678.
2009.
|
|
14
|
Jin QH, Shen HX, Wang H, Shou QY and Liu
Q: Curcumin improves expression of SCF/c-kit through attenuating
oxidative stress and NF-κB activation in gastric tissues of
diabetic gastroparesis rats. Diabetol Metab Syndr. 5:122013.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lee J, Cummings BP, Martin E, Sharp JW,
Graham JL, Stanhope KL, Havel PJ and Raybould HE: Glucose sensing
by gut endocrine cells and activation of the vagal afferent pathway
is impaired in a rodent model of type 2 diabetes mellitus. Am J
Physiol Regul Integr Comp Physiol. 302:657–666. 2012. View Article : Google Scholar
|
|
16
|
Hua XB, Li CR, Zhou HL, et al:
Experimental animal acupoint map. Shi Yan Dong Wu Yu Dong Wu Shi
Yan. 1:1–5. 1991.(In Chinese).
|
|
17
|
He C, Soffer EE, Ferris CD, Walsh RM,
Szurszewski JH and Farrugia G: Loss of interstitial cells of Cajal
and inhibitory innervation in insulin-dependent diabetes.
Gastroenterology. 121:427–434. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Horváth VJ, Vittal H and Ördög T: Reduced
insulin and IGF-I signaling, not hyperglycemia, underlies the
diabetes-associated depletion of interstitial cells of Cajal in the
murine stomach. Diabetes. 54:1528–1533. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Horváth VJ, Vittal H, Lörincz A, Chen H,
Almeida-Porada G, Redelman D and Ordög T: Reduced stem cell factor
links smooth myopathy and loss of interstitial cells of cajal in
murine diabetic gastroparesis. Gastroenterology. 130:759–770. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ordög T: Interstitial cells of Cajal in
diabetic gastroenteropathy. Neurogastroenterol Motil. 20:8–18.
2008. View Article : Google Scholar
|
|
21
|
Long QL, Fang DC, Shi HT and Luo YH:
Gastro-electric dysrhythm and lack of gastric interstitial cells of
Cajal. World J Gastroenterol. 10:1227–1230. 2004.PubMed/NCBI
|
|
22
|
Choi KM, Gibbons SJ, Nguyen TV, Stoltz GJ,
Lurken MS, Ordog T, Szurszewski JH and Farrugia G: Heme oxygenase-1
protects interstitial cells of Cajal from oxidative stress and
reverses diabetic gastroparesis. Gastroenterology. 135:2055–2064.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Bellier S, Da Silva NR, Aubin-Houzelstein
G, Elbaz C, Vanderwinden JM and Panthier JJ: Accelerated intestinal
transit in inbred mice with an increased number of interstitial
cells of Cajal. Am J Physiol Gastrointest Liver Physiol.
288:151–158. 2005. View Article : Google Scholar
|
|
24
|
Wu QX, Zhao M, Tan ZR, Qin LR, Huang X and
Zhang HJ: Changes of interstitial cells of Cajal and connexin 43
expression in the gastric antrum of rats with diabetic
gastroparesis: Implications for interventional effect of insulin.
World J Gastroentero. 22:4399–4405. 2014.
|
|
25
|
Li N, Liu JM, Zhou HJ, et al: Effects of
‘Shuang Gu Yi Tong’ acupuncture on electrogastrogram and expression
of gastric interstitial cells in rats with diabetic gastroparesis.
Zhong Hua Zhong Yi Yao Xue Kan. 32:1855–1857. 2014.(In
Chinese).
|