Introduction
Taoism is a religion native to China that has
existed for ~2,000 years, since the Han Dynasty. The core value of
Taoist philosophy is to promote longevity and immortality by oral
administration of certain Chinese herbs; thus, Taoist medicine has
been well developed and may be considered the origin of Traditional
Chinese Medicine (1). As the leading
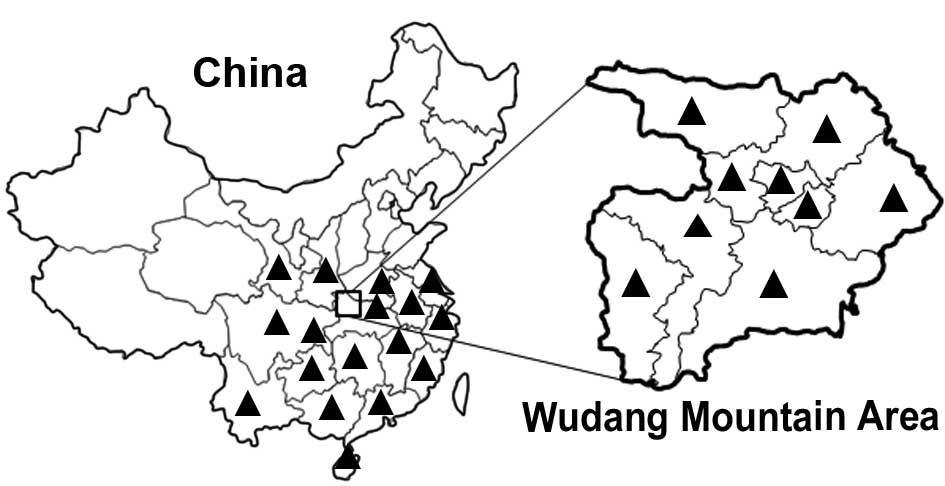
center of Taoism, the Wudang mountain area in the Hubei province of
central China has emerged as the center of Taoist training and
Taoist medicine development since the Ming Dynasty ~600 years ago
(2). At present, 59.1% of patients
with chronic diseases in the Wudang mountain area prefer
traditional medicine or integrated medicine compared with Western
medicine (3). Therefore,
investigation of the role of Taoist medicine in the Chinese
medicine system is crucial.
Radix Polygoni multiflori, which is known as
Heshouwu in China, is the dried root form of Polygonum
multiflorum Thunb. The key places of Heshouwu production in
China are the central and southern provinces, including the
Shaanxi, Gansu, Jiangsu, Zhejiang, Anhui, Fujian, Jiangxi, Henan,
Hunan, Guangdong, Guangxi, Hainan, Guizhou, Chongqing, Sichuan and
Yunnan provinces (4). However,
Heshouwu is distributed across the entire Wudang mountain area
(5), which is localized to the
north-west district of Hubei province (Fig. 1).
Heshouwu is one of the major components of Taoist
formulations used in tonics, for dyeing hair, to slow aging
(6) and for the treatment of
gynecological diseases such as metrorrhagia (7). Although Heshouwu is commercially
available and has been documented in the Chinese Pharmacopoeia
(4), Taoists and the residents of
the Wudang mountain area collect fresh Heshouwu for preparation of
the formulations themselves. However, at present, there has been no
data assessing the quality of wild Heshouwu in the Wudang mountain
area. The main compounds in Heshouwu are phospholipids,
anthraquinones (chrysophanol, chrysophanol anthrone, emodin,
physcion and rhein) and glucosides, among which the main bioactive
components are combined anthraquinones (CAQ) and
2,3,5,4′-tetrahydroxystilbene-2-O-β-D-glucoside (TSG) (8). As CAQ and TSG content are indicators of
the quality of Heshouwu, according to the criteria of the Chinese
Pharmacopeia (2010 version) (4), the
present study collected wild Heshouwu roots from the Wudang
mountain area and analyzed their predominant components, in
addition to the presence of any organic pesticide residues.
Materials and methods
Reagents
TSG (batch no. 110844-201109), emodin (batch no.
110756-200110) and physcion (batch no. 110758-200006) were
purchased from the National Institute for the Control of
Pharmaceutical and Biological Products (Beijing, China). The
following compounds were purchased from the Agro-Environmental
Protection Institute, Ministry of Agriculture (Tianjin, China) for
use as standards in high performance liquid chromatography (HPLC)
and gas chromatography (GC): Organochlorines, including α-benzene
hexachloride (α-BHC; batch no. GSB05-2276-2008), β-BHC (batch no.
GSB05-2277-2008), γ-BHC (batch no. GSB05-2278-2008), δ-BHC (batch
no. GSB05-2279-2008), p,p'-dichlorodiphenyltrichloroethane
(p,p'-DDT; batch no. GSB05-2283-2008),
p,p'-dichlorodiphenyldichloroethylene (DDE; batch no.
GSB05-2280-2008), p,p'-dichlorodiphenyldichloroethane (p,p'-DDDE;
batch no. GSB05-2282-2008), o,p'-DDT (batch no. GSB05-2281-2008)
and pentachloronitrobenzene (PCNB; batch no. GSB05-1845-2008);
pyrethroids, including cypermethrin (batch no. GSB05-2308-2008),
fenvalerate (batch no. GSB05-2307-2008) and deltamethrin (batch no.
GSB05-2310-2008); and organophosphorus compounds, including
malathion (batch no. GSB05-2293-2008), parathion (batch no.
GSB05-2284-2008), ethion (batch no. GSB05-2292-2008), methidathion
E.C. (batch no. GSB05-2295-2008), omethoate (batch no.
GSB05-2288-2008), parathion-methyl (batch no. GSB05-2285-2008),
diazinon (batch no. GSB05-2291-2008), dimethoate (batch no.
GSB05-2286-2008), acephate (batch no. GSB05-2322-2008) and
o,o-dimethyl-O-2,2-dichlorovinyl phosphate (batch no.
GSB05-2398-2008). The water used was double distilled and all other
solvents were HPLC-grade.
Sample collection
The fresh roots of wild Heshouwu were collected from
across the Wudang mountain area (Shiyan, Hubei, China) between June
2013 and July 2014, and were identified by Dr Xuanbin Wang. The
plant specimens were nos. 20130614-20140719 (Fig. 2; Table
I), according to a YY/MM/DD system used in our lab. Commercial
Heshouwu was provided by Hubei Shennong Chinese Materia Medica,
Co., Ltd. (Shiyan, China), Wuhan Liyuan Pharmaceutical, Co., Ltd.
(Wuhan, China) and Bozhou Hongyu Chinese Materia Medica, Co., Ltd.
(Bozhou, China). All samples were ground down into powder, passed
through a 40-mesh filter and stored at 4°C until use.
 | Table I.Information of the collected HSW
samples. |
Table I.
Information of the collected HSW
samples.
| Plant specimen | Sample | Source | Date of
collection |
|---|
| 20130614 | HSW-1 | Dachuan, Shiyan | June 14, 2013 |
| 20130914 | HSW-2 | Liuliping,
Danjiangkouy | September 14,
2013 |
| 20140705 | HSW-3 | Longtanhe, Yunxi | July 5, 2014 |
| 20140329 | HSW-4 | Yanhu Park,
Shiyan | March 29, 2014 |
| 20140719 | HSW-5 | Shuitianfan,
Zhushan | July 19, 2014 |
Reversed phase (RP)-HPLC analysis
TSG and CAQ in the Heshouwu samples were analyzed
using a LC-20A HPLC system (Shimadzu Corporation, Kyoto, Japan),
equipped with a diode array detector. The methodology of HPLC was
consistent with the criteria of the Chinese Pharmacopoeia (2010
edition) (4). Briefly, for TSG, HPLC
was performed using acetonitrile water (25:75 v/v) as the mobile
phase on a SinoChrom ODS BP C18 RP column (250×4.6 mm, 5 µm), with
a flow rate of 0.9 ml/min. The detection wavelength was set at 320
nm with the column temperature at 30°C. For CAQ, HPLC was performed
using methanol in 0.1% phosphoric acid water (80:20 v/v) as the
mobile phase, with a flow rate of 1.0 ml/min. The detection
wavelength was set at 254 nm with the column temperature at
30°C.”
GC analysis
Organochlorine, pyrethroid and organophosphorus
residues in the Heshouwu samples were analyzed using an Agilent
7890A GC system (Agilent Technologies, Inc., Santa Clara, CA, USA),
equipped with a micro-electron capture detector (µECD). The
methodology of GC was consistent with the criteria of the Chinese
Pharmacopoeia (2010 edition) (9).
Briefly, for detection of organochlorine residues, GC was performed
using nitrogen (purity, 99.9999%) as the mobile phase on an Agilent
HP-5 elastic quartz capillary GC column (30×0.32 mm, 0.25 µm;
Agilent Technologies, Inc.), with a column flow rate of 0.9 ml/min.
The inlet gas flow rate was 24 ml/min at 225°C. The initial
temperature of the column was 100°C, after which the column was
programmed to increase by 6°C/min up to 220°C, followed by 7°C/min
up to 250°C, and finally maintained at 250°C for 6 min. The
temperature of the µECD was 300°C, and 1 µl samples were injected
using the split injection technique (9). The flow rate of nitrogen was 25
ml/min.
For detection of pyrethroid residues, GC was
performed using nitrogen as the mobile phase (99.9999%) on an
Agilent HP-5 elastic quartz capillary GC column (30×0.32 mm, 0.25
µm), with a column flow rate of 1.0 ml/min. The inlet gas flow rate
was 24 ml/min at 200°C. The temperature of the column was
programmed as follows: Holding at 150°C for 2 min, increases of
6°C/min from 150–270°C, holding at 270°C for 23 min, increases of
7°C/min from 220–250°C, followed by holding at 250°C for 6 min. The
temperature of the µECD was 320°C and 1 µl samples were injected
using the split injection technique (9). The flow rate of nitrogen was 25
ml/min.
For organophosphorus residue detection, GC was
performed using nitrogen as the mobile phase (99.9999%) on an
Agilent HP-5 elastic quartz capillary GC column (30mx0.25 mm, 0.25
µm), with a column flow rate of 1.0 ml/min. The inlet gas flow rate
was 24 ml/min at 230°C. The temperature of the column was
programmed as follows: Increases of 10°C/min from 120–220°C,
followed by holding at 220°C for 2 min, increases of 20°C/min from
240–270°C, and finally holding at 270°C for 30 sec. The temperature
of the µECD was 300°C and 1 µl samples were injected without the
split.
Results
RP-HPLC to quantify TSG in Heshouwu
from the Wudang mountain area
There was a reliable linear relationship at a range
of 1.8–144 µg/l. The calibration curve was
Y=5.7645649×104−1.63377×105
(r2=0.998). The limit of detection (LOD) and limit of
quantification (LOQ) were 0.0092 and 0.0279 µg/ml, respectively.
The average percentage of TSG in Heshouwu sourced from the Wudang
mountain area and in the commercial product were 2.39 and 1.10%,
respectively (Fig. 3; Table II). The criterion of TSG in the
Chinese Pharmacopoeia (2010 edition) is ≥1.0% (4); thus the content of TSG in the wild
Heshouwu from the Wudang Mountain Area exceeded the criterion of
the Chinese Pharmacopoeia, in addition to that of Heshouwu
available commercially.
 | Table II.TSG, emodin, physcion and combined
anthraquinone content determination in the wild roots of HSW. |
Table II.
TSG, emodin, physcion and combined
anthraquinone content determination in the wild roots of HSW.
| Sample | TSG (%) | Emodin (%) | Physcion (%) | Combined
anthraquinone (%) |
|---|
| HSW-1 | 2.43 | 2.32 | 0.245 | 2.57 |
| HSW-2 | 2.70 | 0.67 | 0.10 | 0.78 |
| HSW-3 | 2.44 | 1.52 | 0.13 | 1.63 |
| HSW-4 | 2.02 | 1.17 | 0.14 | 1.32 |
| HSW-5 | 2.33 | 0.66 | 0.08 | 0.74 |
| HSW average content
in WMA | 2.39 | 1.27 | 0.14 | 1.41 |
| HSW average content
in commercial product | 1.10 | 3.25 | 0.21 | 3.46 |
RP-HPLC to quantify CAQ in Heshouwu
from the Wudang mountain area
The data for emodin and physcion showed a reliable
linear relationship at a range of 0.17–170 and 0.23–46 µg/ml,
respectively. The calibration curves were Y=2626.071X+205.3
(r2=0.999) and Y=8554.358X+2629.933
(r2=0.998), respectively. The LOD and LOQ for emodin
were 0.086 and 0.260 µg/ml, respectively, whereas LOD and LOQ for
physcion were 0.023 and 0.070 µg/ml, respectively. The percentage
of total CAQ (calculated as the sum of emodin and physcion) in the
Heshouwu from the Wudang Mountain Area and in the commercial
product was 1.41 and 3.46%, respectively (Fig. 4; Table
II). According to the Chinese Pharmacopoeia (2010 edition), the
content of CAQ should be >0.05%; thus, the content of CAQ in the
wild Heshouwu sourced from the Wudang mountain area was consistent
with the criterion of the Chinese Pharmacopoeia (9).
GC to detect organochlorine pesticides
in Heshouwu samples from the Wudang mountain area
As organochlorine pesticide residues have previously
been reported in agriculture in other parts of China (10), the present study also determined
whether the wild Heshouwu from the in Wudang mountain area was
contaminated with pesticides. GC was performed to quantify
organochlorine pesticide residues in these samples. The calibration
curves, LOD and LOQ for α-BHC, PCNB, γ-BHC, β-BHC, p,p'-DDE, δ-BHC,
p,p'-DDDE, p,p'-DDT and o,p'-DDT are presented in Table III. Organochlorine residues were
not detected in the samples (Fig.
5), suggesting that Heshouwu from the Wudang Mountain Area and
the commercial products are not contaminated with organochlorine
pesticide residues.
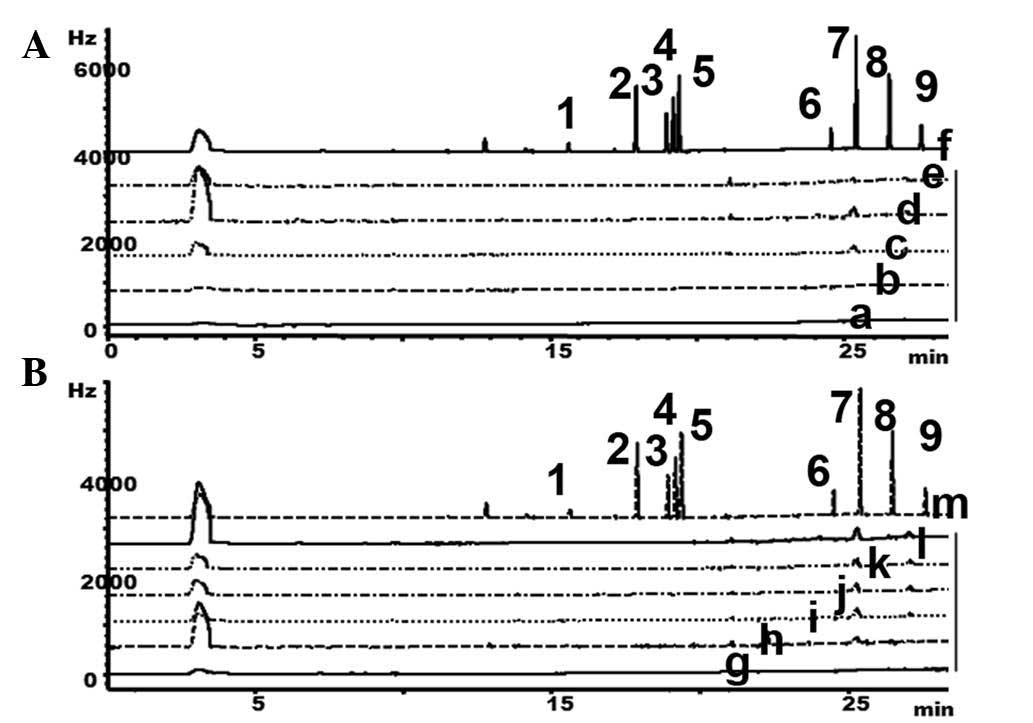 | Figure 5.GC was performed to detect
organochlorine residues in HSW sourced from (A) the Wudang mountain
area and (B) the commercial products. 1, α-benzenehexachloride
(α-BHC); 2, pentachloronitrobenzene; 3, γ-BHC; 4, β-BHC; 5,
p,p'-dichlorodiphenyldichloroethylene; 6, δ-BHC; 7,
p,p'-dichlorodiphenyldichloroethane; 8,
p,p'-dichlorodiphenyltrichloroethane (p,p'-DDT); 9, o,p'-DDT; a,
HSW1; b, HSW2; c, HSW3; d, HSW4; e, HSW5; f, standard; g, Sichuan;
h, Yunan; i, Hubei; j, Hunan; k, Henan; l, Guangdong; m, standard;
Hz, hertz; GC, gas chromatography; HSW, Heshouwu. |
 | Table III.Calibration curve, LOD and LOQ of
organochlorine pesticides in Heshouwu derived from the Wudang
Mountain Area, as determined by gas chromatography. |
Table III.
Calibration curve, LOD and LOQ of
organochlorine pesticides in Heshouwu derived from the Wudang
Mountain Area, as determined by gas chromatography.
|
Compoundsa | Regression
equation | Correlation
coefficient | LOD (ng/ml) | LOQ (ng/ml) |
|---|
| α-BHC | Y=2.725X+3.482 | 0.9998 | 1.05 | 3.50 |
| PCNB | Y=1.963X+4.186 | 0.9996 | 0.95 | 3.16 |
| γ-BHC |
Y=2.083X+10.650 | 0.998 | 0.84 | 2.80 |
| β-BHC |
Y=2.805X+16.196 | 0.996 | 0.75 | 2.50 |
| p,p'-DDE | Y=1.049X+4.436 | 0.997 | 0.61 | 2.03 |
| δ-BHC | Y=1.751X+1.212 | 0.99995 | 0.97 | 3.23 |
| p,p'-DDDE | Y=1.796X-0.197 | 0.99990 | 1.03 | 3.43 |
| p,p'-DDT | Y=1.650X+1.975 | 0.9997 | 1.08 | 3.60 |
| o,p'-DDT | Y=1.965X+1.535 | 0.9998 | 0.98 | 3.27 |
GC to detect pyrethroids in Heshouwu
samples from the Wudang mountain area
GC was used to detect pyrethroid residues, including
cypermethrin, fenvalerate and deltamethrin, in the collected
Heshouwu (Table IV). The results
suggested that there were no pyrethroids detected in the samples
(Fig. 6).
 | Figure 6.GC was performed to detect pyrethroid
residues in HSW derived from (A) the Wudang mountain area and (B)
the commercial products. 1, cypermethrin; 2, envalerate; 3,
deltamethrin; a, HSW1; b, HSW2; c, HSW3; d, HSW4; e, HSW5; f,
standard; g, Sichuan; h, Yunan; i, Hubei; j, Hunan; k, Henan; l,
Guangdong; m, standard; Hz, hertz; GC, gas chromatography; HSW,
Heshouwu. |
 | Table IV.Calibration curve, LOD and LOQ of
pyrethroids in Heshouwu sourced from the Wudang mountain area, as
determined by gas chromatography. |
Table IV.
Calibration curve, LOD and LOQ of
pyrethroids in Heshouwu sourced from the Wudang mountain area, as
determined by gas chromatography.
|
Compoundsa | Regression
equation | Correlation
coefficient | LOD (ng/ml) | LOQ (ng/ml) |
|---|
| Cypermethrin |
Y=6.142X-65.528 | 0.97 | 0.99 | 3.30 |
| Fenvalerate |
Y=10.990X-116.270 | 0.97 | 1.08 | 3.50 |
| Deltamethrin |
Y=15.717X-165.620 | 0.97 | 1.05 | 3.30 |
GC to detect organophosphorus
pesticides in Heshouwu samples from the Wudang mountain area
As organophosphorus pesticides have been the most
widely used pesticides in China, and have previously been
demonstrated to contaminate water and food (11–17), the
present study aimed to detect the presence of 10 organophosphorus
pesticides using GC, according to the criteria of the Chinese
Pharmacopoeia (9). The results
suggested that there were no organophosphorus pesticide residues in
the samples (Table V; Fig. 7).
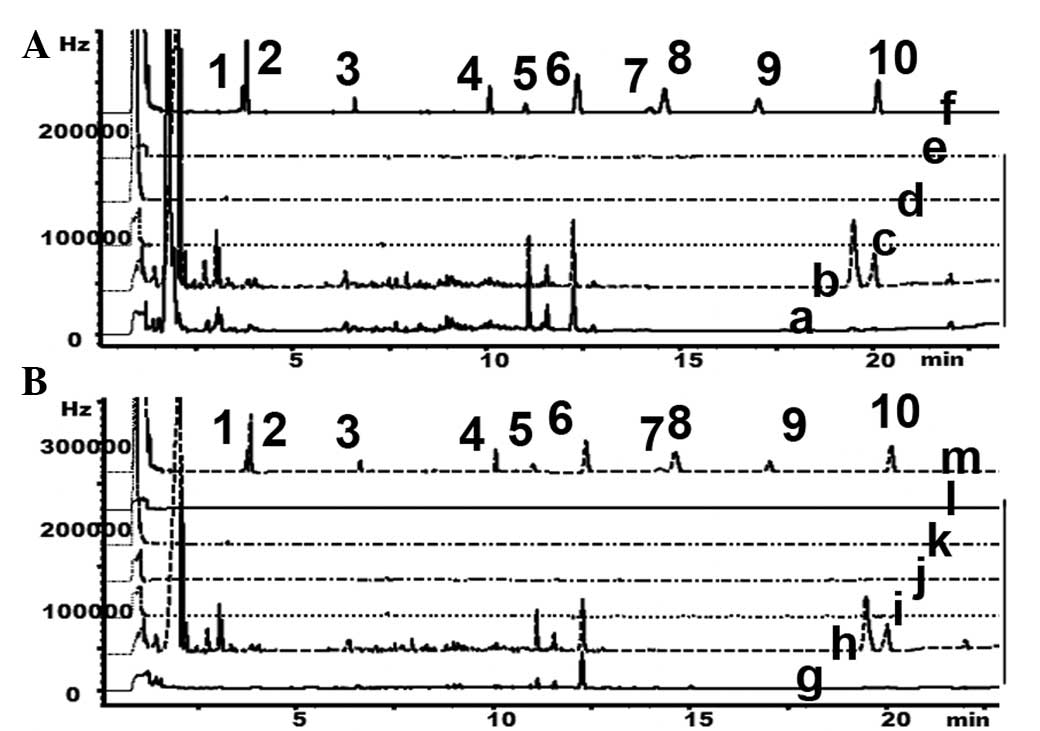 | Figure 7.GC was performed to detect
organophosphorus residues in HSW derived from (A) the Wudang
mountain area and (B) the commercial products. 1,
o,o-dimethyl-O-2,2-dichlorovinylphosphate; 2, omethoate; 3,
acephate; 4, dimethoate; 5, diazinon; 6, parathion-methyl; 7,
malathion; 8, parathion; 9, methidathion E.C; 10, ethion; a, HSW1;
b, HSW2; c, HSW3; d, HSW4; e, HSW5; f, standard; g, Sichuan; h,
Yunan; i, Hubei; j, Hunan; k, Henan; l, Guangdong; m, standard; Hz,
hertz; GC, gas chromatography; HSW, Heshouwu. |
 | Table V.Calibration curve, LOD and LOQ of
organophosphorus pesticides in Heshouwu sourced from the Wudang
mountain area, as determined by gas chromatography. |
Table V.
Calibration curve, LOD and LOQ of
organophosphorus pesticides in Heshouwu sourced from the Wudang
mountain area, as determined by gas chromatography.
| Compounds | Regression
equation | Correlation
coefficient | LOD (ng/ml) | LOQ (ng/ml) |
|---|
| Malathion |
Y=9763.1X-5009.5 | 0.92 | 0.75 | 2.50 |
| Parathion |
Y=9763.1X-5009.5 | 0.97 | 0.64 | 2.13 |
| Ethiofos |
Y=43629.0X-22022.0 | 0.96 | 0.45 | 1.50 |
| Methidathion |
Y=20295.0X-10986.0 | 0.95 | 0.60 | 2.00 |
| Omethoate |
Y=42666.0X-19621.0 | 0.97 | 0.92 | 3.07 |
| Methyl
parathion |
Y=48727.0X-24117.0 | 0.96 | 1.20 | 4.03 |
| Diazinon |
Y=10560.0X-4395.5 | 0.97 | 0.98 | 3.27 |
| Dimethoate |
Y=21569.0X-12247.0 | 0.95 | 1.09 | 3.63 |
| Acephate |
Y=11168.0X-6745.0 | 0.93 | 0.35 | 1.17 |
| Dichlorvos |
Y=7423.1X+869.0 | 0.992 | 0.56 | 1.86 |
Discussion
Taoism is a religion native to China, which has
pursued longevity and immortalization since the Han Dynasty
(18). According to Taoism, aging
may be slowed by performing religious activities, Tai Chi Kung Fu
and Ba Duan Jin (a Taoist physical exercise) and applying Taoist
medical theories using medicines listed in the Materia
medica (1,19,20).
Heshouwu has been described as a dietary herbal medicine, rather
than a therapeutic drug in the Wudang Taoist medicine system
(6,7). Previous studies have demonstrated
various biological effects for Heshouwu, including anti-aging,
immunomodulatory, antihyperlipidemic, hepatoprotective, anticancer
and anti-inflammatory effects, in addition to an ability to improve
learning and memory (21–28). At present, residents of the Wudang
Mountain Area prefer to collect wild Heshouwu for their dietary and
medicinal requirements, instead of purchasing it commercially
(3,6,7).
Therefore, it was important to determine the quality of the wild
Heshouwu in the Wudang mountain area, as its quality is currently
unclear.
In the present study, fresh Heshouwu roots from
across the Wudang mountain area and the commercial products were
evaluated, in order to assess the concentrations of CAQ and TSG.
Furthermore, due to concerns of environmental pollution, organic
pesticide residues were also quantified. In the present study, the
concentration of TSG in the Wudang mountain area exceeded the
criterion of the Chinese Pharmacopoeia (4), in addition to that of commercially
available Heshouwu, and that concentrations of CAQ were consistent
with the criteria of the Chinese Pharmacopoeia (4). These results suggested that the wild
Heshouwu from the Wudang mountain area met the criteria of the
Chinese Pharmacopoeia, and exceeded those in the commercial product
with regard to TSG content. Furthermore, GC data demonstrated that
there was no organic pesticide contamination in wild Heshouwu,
indicating its safety for consumption.
In conclusion, the present study demonstrated that
the wild Heshouwu from the Wudang mountain area met the criteria of
the Chinese Pharmacopeia (2010 edition), and that there were no
pesticide residues in the wild Heshouwu. These results indicated
that wild Heshouwu from the Wudang mountain area may safely be used
as a medicine.
Acknowledgements
The present study was supported by the Young
Scientist Innovation Team Project of Hubei Colleges (grant no.
T201510); the 2013 Innovation & Investment Projects for
Undergraduates of State (grant no. 201310929008); the Scientific
Research Project of Health and Family Planning Commission of Hubei
Province (grant no. WJ2015Z113); the 2011 Project of Hubei Province
(no. 4); the Natural Science Foundation of Hubei Province of China
(grant no. 2010CDZ066, 2014CFB642); the Foundation for Innovative
Research Team of Hubei University of Medicine and the Key
Discipline Project of Hubei University of Medicine (grant no.
2014CXG03); and the Open Project of Hubei Key Laboratory of Wudang
Local Chinese Medicine Research (Hubei University of Medicine;
grant no. WDCM001).
References
|
1
|
Wen MX: The formation and development of
complementary medicine in Wudang Taoism. Hu Nan Zhong Yi Xue Yuan
Xue Bao. 26:14–15. 2006.(In Chinese).
|
|
2
|
Xu D, Wang D, Hu J, Xiong T and Zhu Y:
Study on development history of Taoist medicine in Wudang. Zhonghua
Yi Shi Za Zhi. 30:225–227. 2000.(In Chinese). PubMed/NCBI
|
|
3
|
Chai Y, Xu Z, Wang B and Mao Z: Studies on
rural residents' utilization and tendency of traditional Chinese
drugs in Wudang Town. Shaanxi Zhong Yi Xue Yuan Xue Bao. 28:67–68.
2005.(In Chinese).
|
|
4
|
Commission of Chinese Pharmacopoeia:
Heshouwu, . Pharmacopoeia of the People's Republic of China. 1.
1st. China Medico-Pharmaceutical Science & Technology
Publishing House; Beijing: pp. 164–165. 2010, (In Chinese).
|
|
5
|
Chen JY, Tu HJ, Tu ZL and Li GF:
HeshouwuMateria Medica in Wudang, China. Hubei Ke Xue Chu Ban She;
Wuhan, China: pp. 200–207. 2009, (In Chinese).
|
|
6
|
Shang R: Kan Gua formula in Taoist
medicine. Wu Dang. 55–56. 2004.(In Chinese).
|
|
7
|
Shang R: Introduction of plasters of
Wudang Taoist medicine for gynaecology. Wu Dang. 52–53. 2006.(In
Chinese).
|
|
8
|
Hou JY and Fang TH: Pharmacology of
Chinese Materia Medica. 2nd. Zhong Guo Zhong Yi Yao Chu Ban She;
Beijing, China: pp. 230–232. 2007, (In Chinese).
|
|
9
|
Commission of Chinese Pharmacopoeia.
Appendum, Methods of residues of pesticidesPharmacopoeia of the
People's Republic of China. 1. 1st. China Medico-Pharmaceutical
Science & Technology Publishing House; Beijing: appendum. 2010,
(In Chinese).
|
|
10
|
Da C, Liu G, Tang Q and Liu J:
Distribution, sources, and ecological risks of organochlorine
pesticides in surface sediments from the Yellow River Estuary,
China. Environ Sci Process Impacts. 15:2288–2296. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yang Y, Asiri AM, Du D and Lin Y:
Acetylcholinesterase biosensor based on a gold
nanoparticle-polypyrrole-reduced graphene oxide nanocomposite
modified electrode for the amperometric detection of
organophosphorus pesticides. Analyst (Lond). 139:3055–3060. 2014.
View Article : Google Scholar
|
|
12
|
Hu X, Shi W, Yu N, Jiang X, Wang S, Giesy
JP, Zhang X, Wei S and Yu H: Bioassay-directed identification of
organic toxicants in water and sediment of Tai Lake, China. Water
Res. 73:231–241. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tang X, Liang B, Yi T, Manco G, Palchetti
I and Liu A: Cell surface display of organophosphorus hydrolase for
sensitive spectrophotometric detection of p-nitrophenol substituted
organophosphates. Enzyme Microb Technol. 55:107–112. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wu L, Yao J, Trebse P, Zhang N and Richnow
HH: Compound specific isotope analysis of organophosphorus
pesticides. Chemosphere. 111:458–463. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yu G, Wu W, Zhao Q, Wei X and Lu Q:
Efficient immobilization of acetylcholinesterase onto amino
functionalized carbon nanotubes for the fabrication of high
sensitive organophosphorus pesticides biosensors. Biosens
Bioelectron. 68:288–294. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liu X, Li D, Li J, Rose G and Marriott PJ:
Organophosphorus pesticide and ester analysis by using
comprehensive two-dimensional gas chromatography with flame
photometric detection. J Hazard Mater. 263:761–767. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yuan Z, Yao J, Liu H, Han J and Trebše P:
Photodegradation of organophosphorus pesticides in honey medium.
Ecotoxicol Environ Saf. 108:84–88. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wen MX: History and Characters of Wudang
Taoist Medicine. Nei Meng Gu Zhong Yi Yao. 32:118–119. 2013.(In
Chinese).
|
|
19
|
Shang R: Properties of Taoist medicine in
Wudang. Wudang. 37–39. 2011.(In Chinese).
|
|
20
|
Xu D: Nature of Wudang Taoist Medicine. Wu
Dang. 49–50. 2006.
|
|
21
|
Lin L, Ni B, Lin H, Zhang M, Li X, Yin X,
Qu C and Ni J: Traditional usages, botany, phytochemistry,
pharmacology and toxicology of Polygonum multiflorum Thunb.: A
review. J Ethnopharmacol. 159:158–183. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhou XX, Yang Q, Xie YH, Sun JY, Qiu PC,
Cao W and Wang SW: Protective effect of tetrahydroxystilbene
glucoside against D-galactose induced aging process in mice.
Phytochem Lett. 6:372–378. 2013. View Article : Google Scholar
|
|
23
|
Chen Q, Zhang SZ, Ying HZ, Dai XY, Li XX
and Yu CH and Yu CH: Chemical characterization and
immunostimulatory effects of a polysaccharide from Polygoni
Multiflori Radix Praeparata in cyclophosphamide-induced anemic
mice. Carbohyd Polym. 88:1476–1482. 2012. View Article : Google Scholar
|
|
24
|
Wang W, He Y, Lin P, Li Y, Sun R, Gu W, Yu
J and Zhao R: In vitro effects of active components of Polygonum
Multiflorum Radix on enzymes involved in the lipid metabolism. J
Ethnopharmacol. 153:763–770. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang M, Zhao R, Wang W, Mao X and Yu J:
Lipid regulation effects of Polygoni Multiflori Radix, its
processed products and its major substances on steatosis human
liver cell line L02. J Ethnopharmacol. 139:287–293. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lin P, He YR, Lu JM, Li N, Wang WG, Gu W,
Yu J and Zhao RH: In vivo lipid regulation mechanism of polygoni
multiflori radix in high-fat diet fed rats. Evid Based Complement
Alternat Med. 2014:6420582014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lee BH, Huang YY, Duh PD and Wu SC:
Hepatoprotection of emodin and Polygonum multiflorum against
CCl(4)-induced liver injury. Pharm Biol. 50:351–359. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Jin W: Studies of Zhiheshouwu on
CCl4-induced liver fibrosis in rats. Masters ThesisDalian Medical
University Dalian: 2004, (In Chinese).
|