Introduction
Rheumatoid arthritis (RA) is an inflammatory
autoimmune disease characterized by synovitis and pannus formation,
which causes severe pain and seriously affects patient quality of
life (1). Synovial tissue is
principally composed of fibroblast-like synovial cells, which are
closely associated with the pathogenesis and development of RA,
particularly joint damage. Fibroblast-like synovial cells may
secrete pro-inflammatory cytokines, chemokines and matrix
protein-degrading enzymes (2,3). These
factors may cause an imbalance in the proliferation and apoptosis
of synovial cells, and induce abnormalities in signal transduction
in synovial tissue (4), resulting in
the inflammation and destruction of joints.
It has been reported that RA-related joints are
associated with a hypoxic microenvironment (5), which regulates angiogenesis, induces
inflammatory cell infiltration and elevates pro-inflammatory factor
production (6,7). Hypoxia-inducible factor-1α (HIF-1α) is
an important regulatory factor for the hypoxic response. Under
hypoxic conditions, HIF-1α may activate and upregulate the
expression of hypoxic adaptation-related genes, which are involved
in energy metabolism, intracellular signal transduction and
angiogenesis processes in RA fibroblasts (8). Furthermore, vascular endothelial growth
factor (VEGF) is a potent angiogenesis-stimulating factor, which
serves key roles in angiogenesis and pathogenesis of RA (9). Toll-like receptors (TLRs) are
regulators of adaptive immune responses (10). Activated TLRs may induce the
antimicrobial defense system to produce interleukin (IL)-6, IL-1β,
tumor necrosis factor-α (TNF-α) and other chemokines, thought to be
involved in the pathogenesis of RA (11). TNF-α is an inflammatory mediator with
multiple biological roles. It is primarily produced by mononuclear
macrophages and serves a critical function in the pathogenesis and
development of RA. Furthermore, it may stimulate the proliferation
of synovial fibroblasts, as well as the secretion of IL-6,
granulocyte-macrophage colony stimulating factor, chemokines,
matrix metalloproteinases and prostaglandin (12). At present, there are a number of
commercially available TNF-α antagonists that may be used to treat
RA (13,14); however, long-term usage of TNF-α
antagonists may induce drug resistance and cause infection
(15).
Aconitum leucostomum Worosch. is a perennial
herb belonging to the family Ranunculaceae and is primarily
found in Gansu, Xinjiang and northeastern areas of China (16). The root of A. leucostomum
Worosch. is commonly used in Kazak medicine for the treatment of
indigestion and pain (17,18). In particular, A. leucostomum
Worosch. root has been reported to be an effective treatment of
rheumatic diseases (19,20). A previous study by the current
authors demonstrated that A. leucostomum Worosch. could
alleviate the inflammatory response in the joints of rats with
adjuvant arthritis (21). However,
the detailed mechanism of this effect has not yet been fully
elucidated. In the current study, the effects of A.
leucostomum Worosch. crude drug, processed products and monomer
components on in vitro human rheumatoid fibroblast-like
synoviocyte RA (HFLS-RA) cells were investigated. The levels of
HIF-1α, VEGF and TLR4, as well as the related pro-inflammatory
cytokines, were analyzed and discussed.
Materials and methods
Cell line and cell culture
Human fibroblast-like synoviocyte rheumatoid
arthritis (HFLS-RA) cells were purchased from the European
Collection of Authenticated Cell Cultures (Porton Down, UK). These
cells were cultured in DMEM medium (Gibco; Thermo Fisher
Scientific, Inc., Waltham, MA, USA) containing 10% fetal bovine
serum (Gibco; Thermo Fisher Scientific, Inc.) at 37°C in an
incubator containing 25% CO2.
Drug preparation and
administration
A. leucostomum Worosch. (~30 kg) was obtained
from Nilka County (Yili, Xinjiang, China) and identified by
pharmacist Professor Yonghe Li at the Traditional Chinese Medicine
Hospital Affiliated to Xinjiang Medical University (Urumqi, China).
Monomer components were isolated and purified according to our
previously published procedures, and identified to be delvestidine
(WF-1) and anthranoyllycoctonine (WF-4), according to spectral
analysis (22). Three kinds of
processed products were prepared using water-boiling, high-pressure
steaming and excipient co-boiling methods, respectively (23).
Decoction solution of the crude drug and processed
products was prepared as previously reported (24). Briefly, 200 g crude drug or processed
product was immersed in water (w/v=1/8) for 1 h, then boiled for 30
min. Following filtration with 4 layers of bandage, the solution
was collected and subjected to another decoction. Three decoctions
were performed in total. Then, 40% (v/v) ethanol (95%) was added to
the decoction solution, which was kept at 4°C for 2 days. Following
centrifugation 700 × g at room temperature for 10 min, the
supernatant was concentrated under reduced pressure in a 65°C water
bath and dry extract was obtained.
For drug administration, HFLS-RA cells were divided
into the following groups: i) Control group, in which the cells
received no treatment; five treatment groups, in which the cells
were treated with ii) 2 mg/ml crude drug, iii) 1.8 mg/ml
water-boiled processed products, iv) 1.5 mg/ml high-pressure
steamed processed products, v) 1.2 mg/ml excipient co-boiled
processed products, vi) 40 µg/ml monomer component WF-1 or vii) 100
µg/ml monomer component WF-4; and viii) a positive control group,
in which cells were treated with 150 µg/ml leflunomide, which is
used for the treatment of RA (25)
(Sigma-Aldrich; Merck KGaA, Darmstadt, Germany). For the drug
treatment, WF-1, WF-4, and leflunomide were dissolved in DMSO, and
dry drug extracts were dissolved in the DMEM medium, which were
then used to incubate the cell cultures, respectively. The
inhibition rates were calculated based on the data from the control
group.
Cell Counting Kit-8 (CCK-8) assay
HFLS-RA cells were plated onto a 96-well plate at a
density of 1×104 cells/well. Following drug treatment,
20 µl CCK-8 solution (FC101-03; BestBio, Shanghai, China) was added
to each well and the cells were incubated at 37°C for 4 h. The
absorbance at 450 nm was read using a spectrophotometer (xMark;
Bio-Rad Laboratories, Inc., Hercules, CA, USA).
Flow cytometry
Cellular apoptotic process and cell cycle stages
were evaluated using flow cytometry. For the apoptosis assessment,
cells were washed with PBS, and then digested with 1 ml 0.25%
trypsin. Following centrifugation at 350 × g for 5 min, cells were
resuspended in pre-cooled PBS. Apoptosis detection was performed
with the Annexin V-FITC kit (cat. no. BB-4101-3; BestBio) using the
MACSQuant flow cytometer (Miltenyi Biotec GmbH, Bergisch Gladbach,
Germany). For cell cycle detection, following drug treatment, cells
were collected and centrifuged at 37°C at 350 × g for 5 min.
Following PBS washing and centrifugation, cells were treated with
pre-cooled 70% ethanol and stored at 4°C overnight. The cells were
then centrifuged at 37°C at 350 × g for 5 min and stained with 0.5
ml propidium iodide solution at 37°C in the dark for 30 min.
Fluorescence was detected using the MACSQuant flow cytometer with
MACSQuantify software (Miltenyi Biotec, Auburn, CA, USA).
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Levels of HIF-1α, VEGF and TLR4 mRNA were evaluated
using RT-qPCR. Total RNA was extracted from cells using TRIzol
(Sigma-Aldrich; Merck KGaA). cDNA was obtained using a FastQuant RT
kit with gDNase (cat. no. KR106-02; Tiangen Biotech Co., Ltd.,
Beijing, China). Quantitative PCR was performed with the SYBR
Select Master mix (cat. no. 4472920; Applied Biosystems; Thermo
Fisher Scientific, Inc.) on a 7500 qPCR instrument (Applied
Biosystems; Thermo Fisher Scientific, Inc.). The primer sequences
were as follows: HIF-1α, forward, 5′-TTTGGCAGCAACGACACAGA-3′ and
reverse, 5′-TTTCAGCGGTGGGTAATGGA-3′; VEGF, forward,
5′-GGCCTCCGAAACCATGAACT-3′ and reverse,
5′-TCCATGAACTTCACCACTTCGT-3′; TLR4, forward,
5′-ACAACCTCCCCTTCTCAACC-3′ and reverse, 5′-TTGTCTGGATTTCACACCTGG3′;
and GAPDH, forward, 5′-TGTTGCCATCAATGACCCCTT-3′ and reverse,
5′-CTCCACGACGTACTCAGCG3′. Amplification conditions were as follows:
Denaturation at 95°C for 2 min, followed by 40 cycles of 95°C for
15 sec and 60°C for 1 min, followed by extension at 50°C for 2 min.
The relative expression levels of target genes were calculated
using the 2−∆∆Cq method (26).
Western blot analysis
The expression of HIF-1α, VEGF and TLR4 proteins
were evaluated using western blot analysis. Cells were collected
and lysed with radioimmunoprecipitation lysis buffer (Tiangen
Biotech Co., Ltd., Beijing, China). After vortexing for 20 sec, the
lysis was incubated at 4°C for 30 min, followed by centrifugation
at 20,000 × g at 4°C for 15 min, and then the supernatant was
harvested. Protein concentrations were determined using the BCA
protein assay kit (Tiangen Biotech Co., Ltd.). Protein samples were
subjected to 10% SDS-PAGE (15 µg in each lane), then electronically
transferred onto a polyvinylidene difluoride membrane. Following
blocking with 5% bovine serum albumin (Tiangen Biotech Co., Ltd.)
at room temperature for 1 h, the membrane was incubated with mouse
anti-human anti-HIF-1α primary antibody (1:400 dilution; cat. no.
ab463; Abcam, Cambridge, MA, USA), mouse anti-human anti-VEGF
primary antibody (1:500 dilution; cat. no. ab1316; Abcam) rabbit
anti-human anti-TLR4 primary antibody (1:250 dilution; cat. no.
ab13867; Abcam) or rabbit anti-human anti-β-actin primary antibody
(1:500 dilution; cat. no. BA2305; BOSTER, Wuhan, Hubei, China), at
4°C overnight. The membrane was then incubated with goat
anti-rabbit immunoglobulin G (Pierce; Thermo Fisher Scientific,
Inc.) at room temperature for 1 h. Following treatment with 1 ml
SuperSignal West Pico Chemiluminescent Substrate (Thermo Scientific
Inc., Inc.), protein bands were detected and analyzed with the
ChemiScope mini chemiluminescent analyzer (Chemiscope 3000; CLINX,
Shanghai, China). β-actin was used as an internal reference.
Enzyme-linked immunosorbent assay
(ELISA)
Levels of pro-inflammatory cytokines IL-6, IL-1β and
TNF-α were evaluated using ELISA kits (EH004-96, EH001-96 and
EH009-96, respectively; Shanghai ExCell Biology, Inc., Shanghai,
China), according to the manufacturer's instructions.
Statistical analysis
Data are expressed as the mean ± standard deviation.
SPSS 19.0 software (IBM SPSS, Armonk, NY, USA) was used for
statistical analysis. Student's t-test was performed for group
comparisons. P<0.05 was considered to indicate a statistically
significant result.
Results
Effect of A. leucostomum Worosch. on
the proliferation of HFLS-RA cells
HFLS-RA cells were treated with the crude drug,
processed products (water-boiling, high-pressure steaming and
excipient co-boiling processed products) and monomer components
(WF-1 and WF-4), for 24 h, then cell proliferation was assessed
with a CCK-8 assay. The IC50 values for these treatments
are presented in Table I.
Considering that the amount of the extracted monomer was limited
and high intervention concentrations (e.g., IC50) may be
toxic, the concentrations of 1/2 IC50 were used.
Subsequently, the cells were treated with the crude drug, processed
products and monomer components at the concentrations of 1/2
IC50 for 24, 48 and 72 h. According to the results in
Table I, the optimal inhibition
effect was observed at 48 h. Thus, in the following experiments,
the drug treatment concentrations were set as 1/2 IC50
values, and the treatment duration was set as 48 h.
 | Table I.Effect of Aconitum leucostomum
Worosch. on the proliferation of human fibroblast-like synoviocyte
rheumatoid arthritis cells. |
Table I.
Effect of Aconitum leucostomum
Worosch. on the proliferation of human fibroblast-like synoviocyte
rheumatoid arthritis cells.
|
|
|
|
|
| Inhibition rate at
1/2 IC50 (%) |
|---|
|
|
|
|
|
|
|
|---|
| Treatment | Concentration
(mg/ml) | Inhibition rate
(%) | IC50 (mg/ml) | 1/2 IC50
(mg/ml) | 24 h | 48 h | 72 h |
|---|
| Leflunomide (positive
control) | 0.4 | 52.11 | 0.33 | 0.15 | 23.99 | 35.12 | 53.33 |
| Crude drug | 6.0 | 51.74 | 5.32 | 2.00 | 17.01 | 41.89 | 48.62 |
| Water-boiling
processed product | 3.0 | 51.83 | 2.50 | 1.50 | 22.42 | 37.24 | 48.55 |
| High-pressure
steaming processed product | 3.0 | 64.82 | 2.52 | 1.80 | 21.50 | 41.26 | 48.82 |
| Excipient
co-boiling processed product | 2.5 | 62.30 | 1.85 | 1.20 | 21.67 | 34.52 | 40.91 |
| WF-1 monomer
component | 0.2 | 52.88 | 0.12 | 0.04 | 18.23 | 27.70 | 35.68 |
| WF-4 monomer
component | 0.2 | 62.55 | 0.17 | 0.10 | 19.80 | 24.38 | 32.00 |
Analysis of cell proliferation inhibition rates
indicated that, crude drug and processed products notably inhibited
the cell proliferation, and the highest inhibition rate was
observed in the high-pressure steaming processed product group
(Table I). These results suggested
that the crude drug, processed products and monomer components of
A. leucostomum Worosch. may inhibit the proliferation of
HFLS-RA cells, with more potent inhibition effects observed in the
crude drug and processed products.
Effect of A. leucostomum Worosch. on
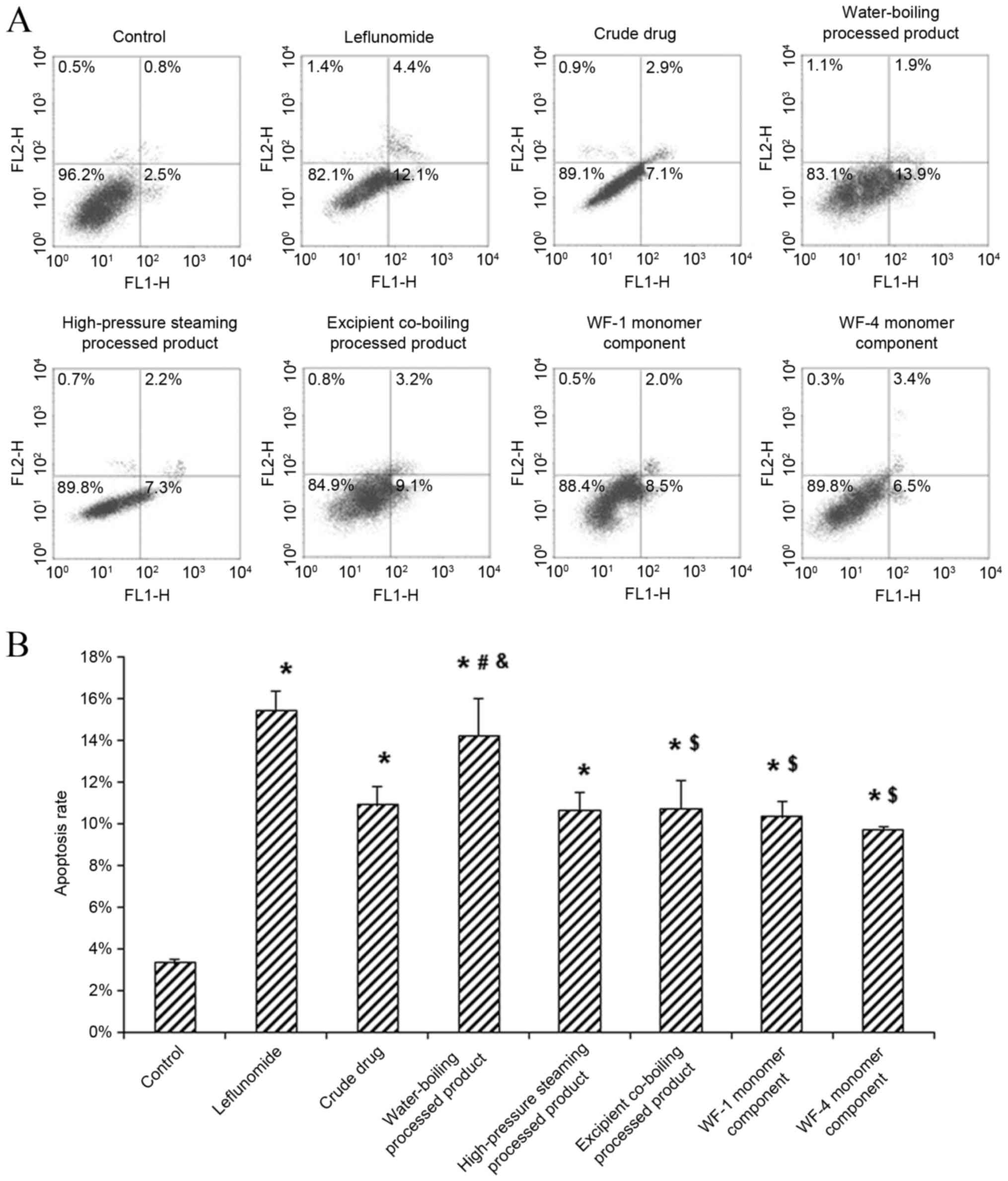
the apoptosis rate of HFLS-RA cells
The effects of A. leucostomum Worosch. crude
drug, processed products and monomer components on the apoptosis of
HFLS-RA cells were evaluated using flow cytometry. The results
indicated that, compared with the control group, apoptosis rates
were significantly elevated in the positive control and all
treatment groups (all P<0.05; Fig.
1). Among the treatment groups, the highest apoptosis rate was
observed for the water-boiling processed product group, which was
significantly higher than the other treatment groups (all
P<0.05) and comparable to that of the positive control group.
These results suggest that the crude drug, processed products and
monomer components of A. leucostomum Worosch. may
significantly promote the apoptosis of HFLS-RA cells, and the most
potent effect was observed for the water-boiling processed
product.
Effect of A. leucostomum worosch. on
HFLS-RA cell cycle
The effects of A. leucostomum Worosch. crude
drug, processed products and monomer components on the HFLS-RA cell
cycle were also evaluated using flow cytometry. These results
demonstrated that, compared with the control group, the proportion
of cells in the G0/G1 phase was significantly decreased in the
positive control group and all treatment groups (all P<0.05;
Table II). Among the treatment
groups, the lowest G0/G1 phase percentage was observed in the
high-pressure steaming processed product group, which was
significantly lower than the other treatment groups (P<0.05;
Table II). These results suggest
that the crude drug, processed products and monomer components of
A. leucostomum Worosch. may induce cell cycle arrest in
HFLS-RA cells; the most potent effect was observed for the
high-pressure steaming processed product.
 | Table II.Effects of Aconitum
leucostomum Worosch. on the human fibroblast-like synoviocyte
rheumatoid arthritis cell cycle. |
Table II.
Effects of Aconitum
leucostomum Worosch. on the human fibroblast-like synoviocyte
rheumatoid arthritis cell cycle.
| Treatment | G0/G1 phase
(%) | S-phase (%) | G2/M phase (%) |
|---|
| Control | 72.03±1.10 | 14.50±0.92 | 13.47±0.46 |
| Leflunomide |
56.83±0.67a |
24.73±1.25a |
18.43±1.76a |
| Crude drug |
55.17±3.98a |
27.77±3.86a | 17.7±2.83 |
| Water-boiling
processed product |
54.60±2.47a |
25.13±2.58a |
20.27±4.24a |
| High-pressure
steaming processed product |
52.57±2.14a–d |
25.57±3.81a |
21.87±2.71a |
| Excipient
co-boiling processed product |
56.77±1.30a,e |
25.73±0.87a |
17.5±0.98a,e |
| WF-1 monomer
component |
59.33±1.78a,b,c,e |
25.50±1.76a |
15.17±0.90c,e |
| WF-4 monomer
component |
59.10±2.11a,b,c,e |
26.27±1.66a |
14.63±.55c,e |
Effect of A. leucostomum Worosch. on
HIF-1α, VEGF and TLR4 expression in HFLS-RA cells
To investigate the effects of A. leucostomum
Worosch. crude drug, processed products and monomer components on
the expression of HIF-1α, VEGF and TLR4 in HFLS-RA cells, mRNA and
protein levels were measured using RT-qPCR and western blot
analysis, respectively. Results from RT-qPCR indicated that,
compared with the control group, levels of HIF-1α and TLR4 mRNA
were significantly downregulated in the positive control group and
all treatment groups (all P<0.05; Fig. 2A and C). Compared with the control
group, only slight changes in VEGF mRNA levels were observed in the
water boiling and excipient co-boiling processed product groups, as
well as in the monomer component (WF-1 and WF-4) groups (P>0.05;
Fig. 2B). Results from western blot
analysis indicated that, compared with the control group, there
were decreases in the expression of HIF-1α, VEGF and TLR4 proteins
in the crude drug, processed product and monomer component groups
(Fig. 3). These results suggest that
the crude drug, processed products and monomer components of A.
leucostomum Worosch. may downregulate the expression of HIF-1α,
VEGF and TLR4 in HFLS-RA cells.
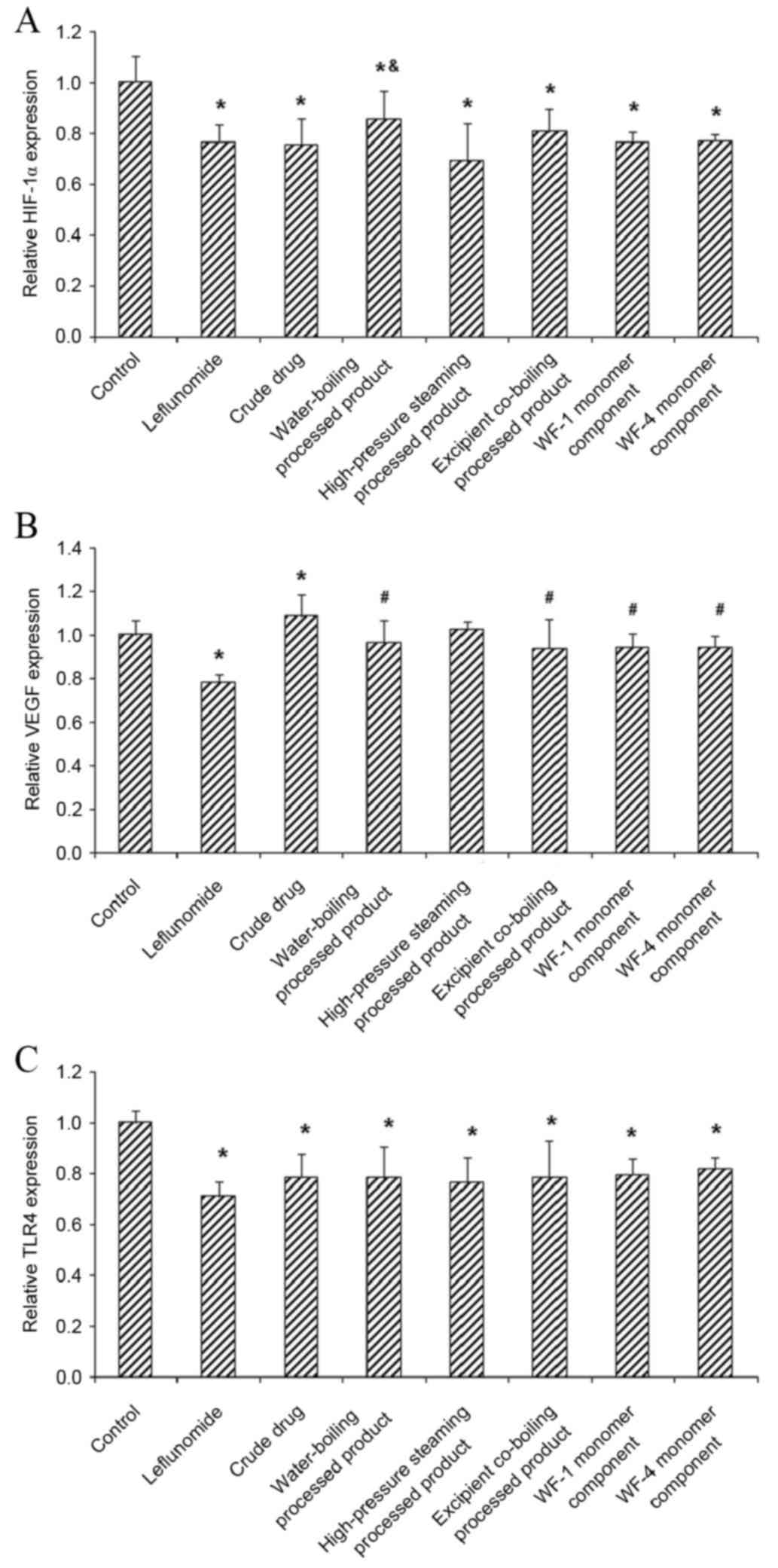 | Figure 2.Effect of Aconitum leucostomum
Worosch. on the expression of HIF-1α, VEGF and TLR4 mRNA in HFLS-RA
cells. HFLS-RA cells were treated with the crude drug, processed
products (water-boiling, high-pressure steaming and excipient
co-boiling processed products) or monomer components (WF-1 and
WF-4) for 48 h. Leflunomide was used as a positive control. The
expression of (A) HIF-1α, (B) VEGF and (C) TLR4 mRNA in HFLS-RA
cells were evaluated using reverse transcription-quantitative
polymerase chain reaction. Data are presented as mean ± standard
deviation. *P<0.05 vs. control group; #P<0.05 vs.
crude drug group; &P<0.05 vs. high-pressure
steaming processed product group. HFLS-RA, human fibroblast-like
synoviocyte rheumatoid arthritis; WF-1, delvestidine; WF-4,
anthranoyllycoctonine; HIF-1α, hypoxia-inducible factor-1α; VEGF,
vascular endothelial growth factor; TLF4, Toll-like receptor 4. |
 | Figure 3.Effect of Aconitum leucostomum
Worosch. on the expression of HIF-1α, VEGF and TLR4 protein in
HFLS-RA cells. HFLS-RA cells were treated with the crude drug,
processed products (water-boiling, high-pressure steaming and
excipient co-boiling processed products) or monomer components
(WF-1 and WF-4) for 48 h. Leflunomide was used as a positive
control. The expression of HIF-1α, VEGF and TLR4 protein in HFLS-RA
cells were evaluated using western blot analysis. HFLS-RA, human
fibroblast-like synoviocyte rheumatoid arthritis; WF-1,
delvestidine; WF-4, anthranoyllycoctonine; HIF-1α,
hypoxia-inducible factor-1α; VEGF, vascular endothelial growth
factor; TLF4, Toll-like receptor 4. |
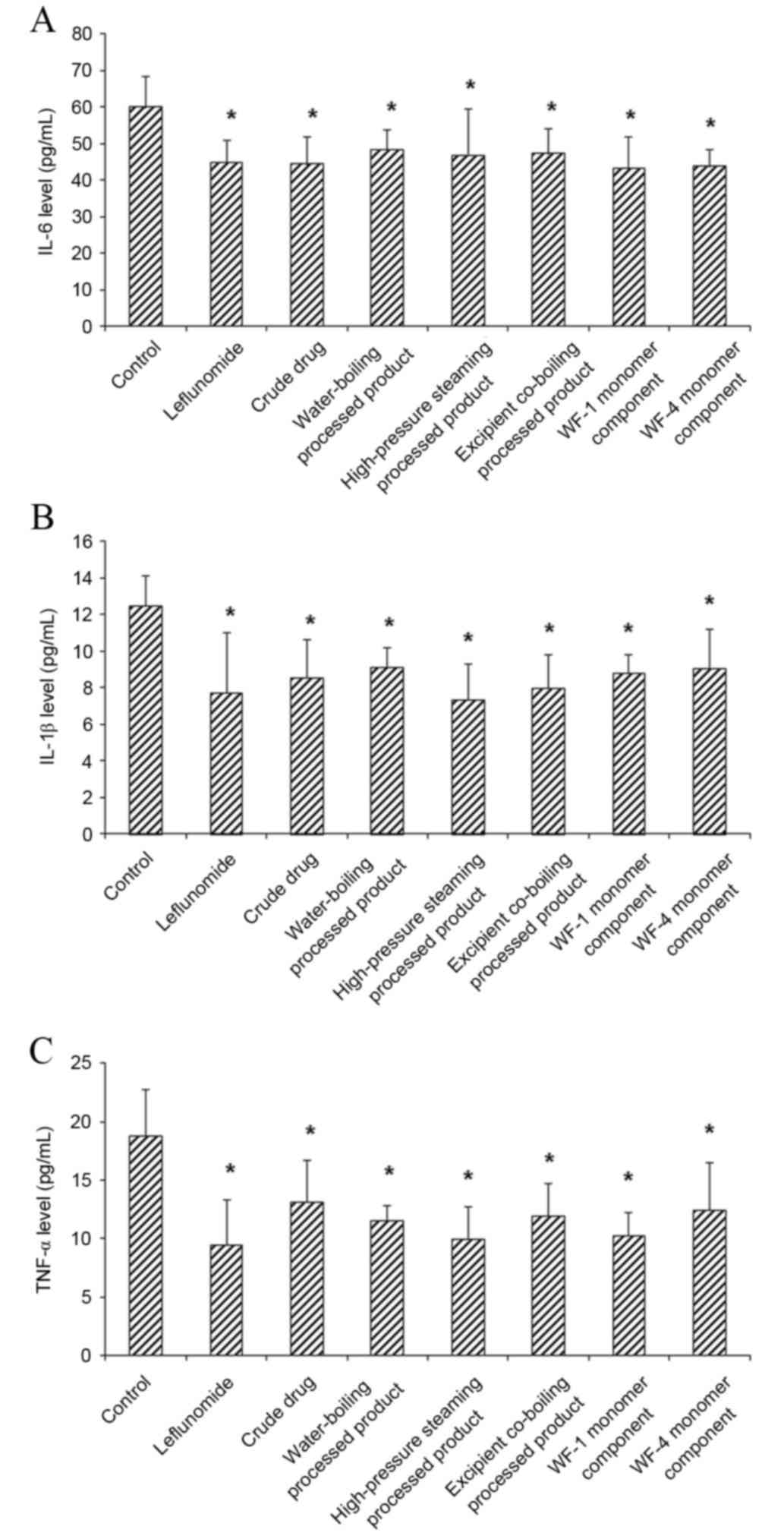
Effect of A. leucostomum Worosch. on
pro-inflammatory cytokine levels in cell culture supernatant
The effects of A. leucostomum Worosch. crude
drug, processed products and monomer components on the
pro-inflammatory cytokine levels in the culture supernatant of
HFLS-RA cells were detected with ELISA. The results indicated that,
compared with the control group, levels of IL-6, IL-1β and TNF-α in
the cell culture supernatant were all significantly decreased in
the treatment groups (all P<0.05; Fig. 4). These results suggest that the
crude drug, processed products and monomer components of A.
leucostomum Worosch. may decrease pro-inflammatory cytokine
levels in HFLS-RA cells.
Discussion
RA is pathologically characterized by synovitis, in
which the pathologically altered synovial membrane enhances
inflammatory cell infiltration and increased pro-inflammatory
cytokine release causes hypoxia in the joint microenvironment and
leads to angiogenesis and pannus formation (27). Pannus has been associated with
erosion, which may eventually result in joint cartilage and bone
destruction (28). Pannus formation
serves a key role in the occurrence and development of RA, and
HIF-1α is a key regulator of the hypoxia response in the body.
Brouwer et al (29)
demonstrated that HIF-1α expression is upregulated in the synovial
tissue in patients with RA and that the number of
HIF-1α+ cells in the RA synovial tissue is positively
correlated with the blood vessel number and inflammation cell
infiltration. Another study indicated that synovial hyperblastosis
and vascular density was increased in RA patients (30). Furthermore, it has been reported that
the serum level of HIF-1α is significantly elevated in patients
with early-stage RA and is even higher than in patients with
advanced-stage or stable RA (7). In
addition, it has been determined that the serum level of VEGF is
increased in patients with RA (31).
TLRs are involved in the regulation of adaptive
immune responses in the body, and they serve key roles in the
pathogenesis and development of various autoimmune diseases,
particularly RA (32). Most
activated TLRs can induce the antimicrobial defense system to
produce IL-6, IL-1β and TNF-α. Therefore, the innate immune
response may be involved in the pathogenesis of RA.
In the present study, HFLS-RA cells were treated
with A. leucostomum Worosch. crude drug, processed products
and monomer components. Cell proliferation was assessed using a
CCK-8 assay, and apoptosis and the cell cycle were evaluated with
flow cytometry. The results demonstrated that drug treatments could
markedly inhibit the proliferation of HFLS-RA cells. Furthermore,
the drug treatments significantly elevated the apoptosis rates of
these cells, and decreased the proportion of cells in G0/G1 phase.
These results suggested that A. leucostomum Worosch. may
induce apoptosis and inhibit proliferation of HFLS-RA cells. The
expression of HIF-1α, VEGF and TLR4 mRNA and protein in the HFLS-RA
subjected to drug treatments were evaluated using RT-qPCR and
western blot analysis, and levels of IL-6, IL-1β and TNF-α in the
cell culture supernatant were detected with ELISA. A previous study
demonstrated that hypoxic conditions are closely associated with
upregulated levels of HIF-1α and VEGF (33). However, the results of the present
study demonstrated that, compared with the control group, the drug
treatments significantly decreased the mRNA and protein levels of
HIF-1α and TLR4 in HFLS-RA cells, but not VEGF. Furthermore, it was
demonstrated that, compared with the control group, levels of IL-6,
IL-1β and TNF-α in the cell culture supernatant were significantly
decreased, which was in accordance with previous results (4).
In the present study, our results showed that,
persistent hypoxia may induce the upregulated expression of HIF-1α
and TLR4, which, together with the enhanced release of
pro-inflammatory cytokines, may contribute to enhanced synovial
inflammation. Ca2+/calmodulin-dependent protein kinase
II (CaMKII) is a member of the CaMK family and is expressed in the
fibroblast-like synovial cells in RA. It has been determined that
CaMKII may regulate the transcription and activation of HIF-1α and
other factors, and inhibiting CaMKII may downregulate the
expression of HIF-1α and VEGF in RA synovial cells, potentially by
suppressing the pI3K/Akt signaling pathway (34). The pI3K/Akt and mitogen-activated
protein kinase signaling pathways mediate the activation of HIF-1α
in RA synovial cells under hypoxic conditions (35). It has been demonstrated that
treatment with artesunate may inhibit the pI3 K/Akt signaling
pathway and downregulate the expression of HIF-1α and VEGF in RA
fibroblast-like synovial cells (36). Another study indicated that
inhibition of the pI3 K/Akt signaling pathway reduced HIF-1α
expression in rat models of collagen-induced arthritis, which
significantly alleviated arthritis clinical symptoms, imaging
alterations, synovial hyperplasia and inflammatory cell
infiltration (37). Therefore,
HIF-1α may be activated via various pathways by receptors in the RA
fibroblast-like synovial cells in a hypoxic microenvironment
(38).
In conclusion, the current results indicated that
the crude drug, processed products and monomer components of A.
leucostomum Worosch. significantly enhanced the apoptosis of
HFLS-RA cells, with the most potent effect observed for
water-boiling processed products. The A. leucostomum
treatments induced cell cycle arrest in the HFLS-RA cells, with the
most potent effect observed for high-pressure steaming processed
products. Furthermore, the A. leucostomum treatments
downregulated the expression levels of HIF-1α and TLR4 in HFLS-RA
cells, and decreased the pro-inflammatory cytokine levels in the
culture supernatant. These findings may contribute to understanding
of the pathogenesis and development of RA and the development of
novel therapeutic strategies to clinically treat the disease.
Acknowledgements
The present study was supported by the National
Natural Science Foundation of China (grant no. 81160498).
References
|
1
|
Seyler TM, Park YW, Takemura S, Bram RJ,
Kurtin PJ, Goronzy JJ and Weyand CM: BLyS and APRIL in rheumatoid
arthritis. J Clin Invest. 115:3083–3092. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Yamaannishi Y and Firestein GS:
Pathogenesis of rheumatoid arthritis: The role of synoviocytes.
Rheum Dis Clin North Am. 27:355–371. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wang JB and Pan L: Etiology and
pathogenesis of rheumatoid arthritis. Shandong Yi Yao Za Zhi.
42:69–70. 2002.
|
|
4
|
Perlman H, Pagliari LJ, Liu H, Koch AE,
Haines GK III and Pope RM: Rheumatoid arthritis synovial
macrophages express the Fas-associated death domain-like
interleukin-1beta-converting enzyme-inhibitory protein and are
refractory to Fas-mediated apoptosis. Arthritis Rheum. 44:21–30.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sivakumar B, Akhavani MA, Winlove CP,
Taylor PC, Paleolog EM and Kang N: Synovial hypoxia as a cause of
tendon rupture in rheumatoid arthritis. J Hand Surg Am. 33:49–58.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Konisti S, Kiriakidis S and Paleolog EM:
Hypoxia-a key regulator of angiogenesis and inflammation in
rheumatoid arthritis. Nat Rev Rheumatol. 8:153–162. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Huang X, Chen Y and Gong L: Measurement of
serum hypoxia inducible factor-1α in patients with rheumatoid
arthritis and the correlation with ultrasonographic assessment of
synovial lesions. Zhejiang Med J. 33:1420–1422. 2011.
|
|
8
|
Del Rey MJ, Izquierdo E, Usategui A,
Gonzalo E, Blanco FJ, Acquadro F and Pablos JL: The transcriptional
response of normal and rheumatoid arthritis synovial fibroblasts to
hypoxia. Arthritis Rheum. 62:3584–3594. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
You P, Lin M, Li K, Ye X and Zheng J:
Normobaric oxygen therapy inhibits HIF-1α and VEGF expression in
perihematoma and reduces neurological function defects.
Neuroreport. 27:329–336. 2016.PubMed/NCBI
|
|
10
|
Cheng S, He C, Zhou H, Kong X, Xie H, Xia
L and Yan J: The effect of Toll-like receptor 4 on β2-glycoprotein
I-induced B cell activation in mouse model. Mol Immunol. 71:78–86.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Huang B, Wang QT, Liu KK, Jiang L and Wei
W: The relationship between TNF-α signaling pathway and CD4 + T
cells in the development of rheumatoid arthritis. Zhong Guo Yao Li
Xue Tong Bao. 29:900–903. 2013.
|
|
12
|
Xiao JY, Wang BS and Wang SJ: Expression
of IL-1 and TNF-α in adjuvant arthritis rat models. J Emer Trad
Chin Med. 20:607–608. 2011.
|
|
13
|
Manfredi AA, Baldini M, Camera M,
Baldissera E, Brambilla M, Peretti G, Maseri A, Rovere-Querini P,
Tremoli E, Sabbadini MG and Maugeri N: Anti-TNFα agents curb
platelet activation in patients with rheumatoid arthritis. Ann
Rheum Dis. 75:1511–1520. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cacciapaglia F, Anelli MG, Rizzo D,
Morelli E, Scioscia C, Mazzotta D, Iannone F and Lapadula G:
Influence of TNF-α inhibition on oxidative stress of rheumatoid
arthritis patients. Reumatismo. 67:97–102. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang XK, Li B, Ren Y, Liang ZC and Yang
ZB: Effects of Alcohol Extract of toddalia asiatica on the
Inflammation-associated cytokines of model rats with Adjuvant
arthritis. Zhong Guo Yao Fang. 27:3524–3527. 2016.
|
|
16
|
Editorial Committee of Flora of China,
China Academy of Sciences: Flora of China. 27. Chinese Science
Publishing and Media Ltd.; Beijing: pp. 113–326. 1979
|
|
17
|
Xin Xu: Bahaerguli Huangerhan. Kazakh
Pharmacopeia. 1. Ethnic Publishing House; Beijing: pp. 45–47.
2009
|
|
18
|
Meng Q, Liang J and Wu G: Advances in
pharmacological effects of alkaloids. Lishizhen Med Materia Med
Res. 14:700–702. 2003.
|
|
19
|
Xinjiang Institute of Biological Soil
Desert, . Xinjiang Medicinal Flora. 1. Xinjiang People's Publishing
House; Urumqi: 1977
|
|
20
|
Editorial Committee of Flora of Xinjiang,
. Flora of Xinjiang. 1. Xinjiang Science and Technology Health
Press; Urumqi: 1999
|
|
21
|
Yang JL, Lu J, Liu J and Nie JH: Effects
of Diphtheria Aconitum and processed products on adjuvant-induced
arthritis rat model. Chinese Medicine. 37:2495–2501. 2015.
|
|
22
|
Wang F, Zhao J, Zhao F and Nie J: Study on
the chemical constituents of Aconitum leucostomum Worosch. China
Pharmacy. 26:1233–1235. 2015.
|
|
23
|
Wu M and Wei Y: Ethnobotany of Aconitum in
Xinjiang. Chinese Wild Plant Resources. 23:29–30. 2004.(In
Chinese).
|
|
24
|
Editorial Committee of Chinese
Pharmacopoeia, . Chinese Pharmacopoeia. 1. Chinese Medical Science
and Technology Press; Beijing: pp. 33–34. 2010
|
|
25
|
Wiacek R, Kolossa K, Jankowski T, Jeka S,
Karmowski A, Karmowski M and Gworys B: The efficacy and safety of
leflunomide in patients with active rheumatoid arthritis. Adv Clin
Exp Med. 21:337–342. 2012.PubMed/NCBI
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) Method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chen L, Wang WS, Wang YM and Shi CH: The
expression of VEGF and TGF-β1 in synovial membrane of the patients
of Knee joint pannus. Shi He Zi Da Xue Xue Bao (Zi Ran Ke Xue Ban).
28:72–75. 2010.
|
|
28
|
Shiozawa S, Tsumiyama K, Yoshida K and
Hashiramoto A: Pathogenesis of joint destruction in rheumatoid
arthritis. Arch Immunol Ther Exp (Warsz). 59:89–95. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Brouwer E, Gouw AS, Posthumus MD, van
Leeuwen MA, Boerboom AL, Bijzet J, Bos R, Limburg PC, Kallenberg CG
and Westra J: Hypoxia inducible factor-1-alpha (HIF-1alpha) is
related to both angiogenesis and inflammation in rheumatoid
arthritis. Clin Exp Rheumatol. 27:945–951. 2009.PubMed/NCBI
|
|
30
|
Shankar J, Thippegowda PB and Kanum SA:
Inhibition of HIF-1alpha activity by BP-1 ameliorates adjuvant
induced arthritis in rats. Biochem Biophys Res Commun. 387:223–228.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Huang XQ, Chen Y, Chen LB, Gong LM, Xie
BH, Peng Y, Huang H, Xin XF, Wu XD and Zhang Z: Measurement of
serum vascular endothelial growth factor, angiopoietin-1 and
angiopoietin-2 in patients with rheumatoid arthritis and the
correlation with synovial lesions. Chin J Rheumatol. 16:679–683.
2012.(In Chinese).
|
|
32
|
Migita K, Miyashita T, Maeda Y, Nakamura
M, Yatsuhashi H, Kimura H, Ishibashi H and Eguchi K: Toll-like
receptor expression in lupus peripheral blood mononuclear cells. J
Rheumatol. 34:493–500. 2007.PubMed/NCBI
|
|
33
|
Ning X and Lu ZJ: Expression of
Fibrosis-related Cytokine in Budd-Chiari Syndrome. Chin Gen Prac.
18:2026–2029. 2015.
|
|
34
|
Chou LW, Wang J, Chang PL and Hsieh YL:
Hyaluronan modulates accumulation of hypoxiainducible factor-1
alpha, inducible nitric oxide synthase, and matrix
metalloproteinase-3 in the synovium of rat adjuvant-induced
arthritis model. Arthritis Res Ther. 13:R902011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Westra J, Brouwer E, van Roosmalen IA,
Doornbos-van der Meer B, van Leeuwen MA, Posthumus MD and
Kallenberg CG: Expression and regulation of HIF-1alpha in
macrophages under inflammatory conditions; significant reduction of
VEGF by CaMKII inhibitor. BMC Musculoskelet Disord. 11:612010.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Aletaha D, Neogi T, Silman AJ, Funovits J,
Felson DT, Bingham CO III, Birnbaum NS, Burmester GR, Bykerk VP,
Cohen MD, et al: 2010 Rheumatoid arthritis classification criteria:
An American college of rheumatology/european league against
rheumatism collaborative initiative. Arthritis Rheum. 62:2569–2581.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hochberg MC, Altman RD, Brandt KD, Clark
BM, Dieppe PA, Griffin MR, Moskowitz RW and Schnitzer TJ:
Guidelines for the medical management of osteoarthritis. Part II.
Osteoarthritis of the knee. American College of Rheumatology.
Arthritis Rheum. 38:1541–1546. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Cramer T, Yamanishi Y, Clausen BE, Förster
I, Pawlinski R, Mackman N, Haase VH, Jaenisch R, Corr M, Nizet V,
et al: HIF-1alpha is essential for myeloid cell-mediated
inflammation. Cell. 112:645–657. 2003. View Article : Google Scholar : PubMed/NCBI
|