Introduction
Heart failure (HF) is a cardiac disease induced by
multiple factors, such as a high fat diet, lack of exercise and
aging, that can be recognized by dysfunction in cardiac systolic
and diastolic processes. The number of individuals affected by HF
has increased from 5.7 million to 6.2 million in the US between
2013 and 2016, and HF-related deaths contributed to 13.4% of all
deaths that occurred in the US in 2018(1). Previous studies reported that
proinflammatory cytokines serve a critical role in promoting the
development of HF (2) and that an
increased concentration of cytokines and biomarkers are exhibited
in patients with HF (3).
Inflammation has been regarded as a possible therapeutic target for
patients with HF (4). Thus,
pathways regulating inflammation and oxidative responses may act as
a treatment method for reducing cardiovascular disease risk.
Dehydrocostus lactone (DHE) is a natural sesquiterpene lactone and
the major compound in the roots of a well-known traditional Chinese
herbal medicine, Saussurea lappa. DHE has presented
anti-inflammatory and immunomodulatory effects in previous studies
(5,6). A previous study also indicated that
DHE protects HepG2 cells against oxidative stress via the induction
of heme oxygenase-1 (HO-1) expression (7), and that this effect may be mediated
by the inhibition of the NF-κB signaling pathway (8). Moreover, DHE has numerous biological
functions, including anti-inflammatory properties, immunomodulatory
capabilities and antitumor action (9). Previous studies found that DHE
significantly inhibits the proliferation of cancer cells without
affecting normal cells in breast and prostate cancer (10,11).
Another study indicated that DHE reduces lipopolysaccharide-induced
acute lung injury in a rat model via the inhibition of the
inflammatory response (12).
Thioredoxin-interacting protein (TXNIP) is a protein
that serves a critical role in multiple cellular processes,
including metabolism, growth and antioxidative responses, by
inhibiting the ability of cells to take up glucose (13). Previous research has indicated that
activation of the inflammatory response is mediated by the
activation of TXNIP (14), thus,
we hypothesized that TXNIP inhibition could enhance the therapeutic
effect of DHE in cardiomyocytes.
In the present study, a cell model of TXNIP
overexpression and inhibition was established to study the
proliferation of H9c2 cells and the expression of pro and
anti-inflammatory genes. The involvement of the nuclear factor
erythroid 2-related factor 2 (Nrf2)/HO-1/NF-κB signaling pathway
was also investigated.
Materials and methods
Vector constructions
The TXNIP cDNA fragment was obtained using PCR with
the following primers: Forward, 5'-AGTGATTGGCAGCAGGTC-3' and
reverse, 5'-GGTGTCTGGGATGTTTAGG-3'. A blank pcDNA3.1 vector (cat.
no. HG-VPH0683; HonorGene) and the product obtained from PCR were
digested with KpnI (cat. no. R3142S; New England BioLabs,
Inc.) and XhoI (cat. no. R0146S; New England BioLabs, Inc.)
enzymes and the TXNIP fragment was linked with the digested vector
using T4 DNA Ligase (cat. no. M0202S; New England BioLabs, Inc.).
The pcDNA3.1-TXNIP overexpression vector (1.5 µg) was subsequently
transfected into H9c2 cells using Lipofectamine™ 3000 Transfection
Reagent (cat. no. L3000008; Thermo Fisher Scientific, Inc.) for 48
h at 37°C according to the manufacturer's protocol.
Stable TXNIP-expressing H9c2 cells were screened using 1,000 µg/ml
G418. After four weeks of screening, stable TXNIP overexpression
cells were obtained and cells were used to perform western blotting
and ELISA experiments.
The TXNIP knockdown vector was constructed as
previously described (15). The
knockout of the TXNIP gene was performed using the CRISPR-Cas9
system. A blank lentiCRISPR v2 vector (cat. no. 52961; Addgene) was
digested using the BsmBI (cat. no. R0580S; New England
BioLabs, Inc.) enzyme as recommended according to previous study
(15) and oligos were constructed
using the following gRNAs: Forward, 5'-CACCGGAGACAGACACCCGCCCATC-3'
and reverse, 5'-CGATGGGCGGGTGTCTGTCTCCAAA-3', designed using an
online CRISPR design tool (CRISPRdirect; https://crispr.dbcls.jp). The digested vector and
oligos were ligated using quick ligase (cat. no. M0201S; New
England BioLabs, Inc.) to construct the TXNIP knockdown vector
[target gene sequence: Forward, 5'-GATGGGCGGGTGTCTGTCTC-3' and
reverse, 5'-GAGACAGACACCCGCCCATC-3'; target exon, TXNIP-202
(Transcript ID: ENSRNOT00000028793.7; domain affected, IPR011022
(InterPro database)]. The vector was first transfected into 293T
cells to assemble lentiviruses using Lipofiter transfect reagent
(cat. no. KS-TRLF-200; Hanbio Biotechnology Co., Ltd.) at
37°C. The virus was collected at 72 h, after which it
was subsequently transduced into H9c2 cells for 48 h at
37°C. Stable TXNIP-knockout cells were screened using 2
µg/ml puromycin. The blank pcDNA3.1 vector was used as a blank
control for TXNIP overexpression detection and the blank
lentiCRISPR v2 vector was used as a blank control for TXNIP
knockout detection.
Cell culture, grouping and MTT
assay
The rat cardiomyoblast H9c2 (cat. no. CRL-1446) and
the human kidney epithelial 293T (cat. no. CRL-11268) cell lines
were purchased from the American Type Culture Collection and
cultured in high glucose DMEM (cat. no. 11965092; Thermo Fisher
Scientific, Inc.) supplemented with 10% FBS (cat. no. 10091; Thermo
Fisher Scientific, Inc.) in a 5% CO2 humid atmosphere at
37°C. Subsequently, the H9c2 cells were divided into
four groups: i) The control group (NC); ii) the DHE treatment group
(OT); iii) the DHE treatment combined with TXNIP inhibition group
(OI); and iv) the DHE treatment combined with TXNIP overexpression
group (OH). In all groups, H9c2 cells were first treated with 5 µM
doxorubicin and incubated for 24 h at 37˚C (16) with or without 30 µM DHE for 24 h
(17). The viability of cells was
measured using an MTT assay. H9c2 cells were treated as described
above, after which they were incubated with 5 mg/ml MTT reagent for
4 h at 37˚C. Cells were then dissolved in DMSO and absorbance was
detected at 490 nm using a microplate reader (cat. no. PT-3502C;
Potenov).
Ethics statement
This study was carried out in strict accordance with
the recommendations of the Guide for the Care and Use of Laboratory
Animals of the National Institutes of Health (18). The experimental protocols were
approved by the Ethics Committee of Animal Experiments of Anqiu
People's Hospital (Anqiu, China). All experiments were conducted in
accordance with the Declaration of Helsinki.
Mouse model and grouping
A total of 32 C57BL/6 mice (age, 8-10 weeks; weight,
24-27 g) were purchased from the Xiamen University Laboratory
Animal Center. Mice were kept in a 22-24˚C and 50-60% humidity
atmosphere under a 12-h light/dark cycle, where food and water was
freely available. Mice were randomly divided into the following
four groups: i) The control group (NC); ii) the DHE treatment group
(OT); iii) the DHE treatment combined with TXNIP inhibition group
(OI); and iv) the DHE treatment combined with TXNIP overexpression
group (OH). In order to construct the TXNIP overexpression and
knockout mouse model, 200 µl of the TXNIP overexpression, knockout
lentivirus vector or blank vector were injected in mice via the
tail vein. The vector used for animal experiments was purchased
from Hanbio Biotechnology Co., Ltd. A total of 32 mice were divided
into two groups: The DHE-treated group (n=20) and the TXNIP
expression detection group (n=12). Mice were treated with 15 mg/kg
doxorubicin for 21 days to construct the HF model (19). In the DHE treatment groups, mice
were treated with 5 mg/kg DHE for 21 days (12). For blood sample collection, 100 µl
blood was collected through ophthalmic vein into a 1.5 ml tube, and
incubated at room temperature for 1 h. After centrifugation at
1,500 x g at 4˚C for 10 min, serum was collected and subjected to
ELISA. Then, mice in each group were anesthetized using sodium
pentobarbital (100 mg/kg) via intraperitoneal injection and then
sacrificed by cervical dislocation. For heart tissue assessment,
the expression of TXNIP in full heart tissue of mice without DHE
treatment was detected using western blotting analysis. The full
heart tissue of mice treated with DHE was collected after mice were
sacrificed. Heart tissues were stored at -80°C until
further experimentation.
RNA extraction
RNA extraction was performed according to the
protocol of the RNApure Tissue and Cell kit (cat. no. CW0560; CoWin
Biosciences). Heart tissues and cells were first lysed with lysis
buffer. After incubation at room temperature for 5 min, the mixture
was centrifuged at 13,000 x g, 4°C for 5 min.
Subsequently, the RNA samples were transferred into spin columns
and centrifuged at 13,000 x g, 4°C for 1 min. After
washing with washing buffer, RNA samples were eluted from the spin
column using elution buffer after centrifugation at 13,000 x g,
4°C for 1 min. RNA sample concentration was detected
using a spectrophotometer (UV5Nano; Mettler-Toledo International,
Inc.). RNA samples were used for subsequent reverse
transcription-quantitative PCR (RT-qPCR) experiments.
RT-qPCR
RT-qPCR experiments were performed using an
UltraSYBR One Step RT-qPCR kit according to the manufacturer
protocol (cat. no. CW2623; CoWin Biosciences). The reaction mixture
was prepared as recommended by the manufacturer protocol and the
reaction was performed with the following steps: Reverse
transcription at 45˚C for 10 min; initial denaturation at 95˚C for
5 min; and 40 cycles of denaturation at 95˚C for 10 sec, annealing
and elongation at 58˚C for 10 sec and final extension at 72˚C for
30 sec. The primers used for qPCR were as follows: Inducible nitric
oxide synthase (iNOS) forward, 5'-CCCTTCAATGGTTGGTACATGG-3' and
reverse, 5'-ACATTGATCTCCGTGACAGCC-3'; TNF-α forward,
5'-TTCTCATTCCTGCTTGTGG-3' and reverse, 5'-ACTTGGTGGTTTGCTACG-3';
IL-6 forward, 5'-CCACCAAGAACGATAGTCAA-3' and reverse,
5'-TTTCCACGATTTCCCAGA-3'; IL-1 forward,
5'-CCAGCTTCAAATCTCACAGCAG-3' and reverse,
5'-CTTCTTTGGGTATTGCTTGGGATC-3'; IL-10 forward,
5'-GGCAGATCTATGCTTGGCTCAGCACTG-3' and reverse,
5'-GCGATATCCCTGCAGTCCAGTAGACG-3'; cyclooxygenase-2 (COX-2) forward,
5'-TCACAGGCTTCCATTGACCAG-3' and reverse, 5'-CCGAGGCTTTTCTACCAGA-3';
NF-E2-Related Factor 2 (Nrf2) forward, 5'-GTCTTCACTGCCCCTCATC-3'
and reverse, 5'-TCGGGAATGGAAAATAGCTCC-3'; HO-1 forward,
5'-AGAGTCCCTCACAGACAGAGTTT-3' and reverse,
5'-CCTGCAGAGAGAAGGCTACATGA-3'; NF-κB forward,
5'-ACGGGAGGGGAAGAAATCTATC-3' and reverse,
5'-AATGGCAAACTGTCTGTGAACA-3'; GAPDH forward,
5'-GATGCTGGTGCTGAGTATGTCG-3', reverse,
5'-GTGGTGCAGGATGCATTGCTCTGA-3'. GAPDH was used as an internal
control. The expression of each target gene was calculated using
2-ΔΔCq method (20).
Western blotting
Heart tissues and cells from each group were lysed
using RIPA lysis buffer (cat. no. CW2333; CoWin Biosciences)
supplemented with a protease inhibitor cocktail (cat. no. CW2200;
CWbio Biosciences). The concentration of proteins was determined
using a BCA assay (cat. no. CW0014; CoWin Biosciences).
Subsequently, 60 µg protein/lane was separated by SDS-PAGE on a 10%
gel. After electrophoresis, the proteins were transferred onto PVDF
membranes through Trans-Blot (cat. no. 1703940; Bio-Rad
Laboratories, Inc.) transfer blotter. The membranes were first
incubated with 5% skimmed milk for 1 h at room temperature,
followed by incubation with the following primary antibodies at 4˚C
overnight (all, Abcam; all, 1:1,000): Kelch like ECH associated
protein 1 (Keap1; cat. no. ab226997; 1:1,000), Nrf2 (cat. no.
ab31163), HO-1 (cat. no. ab68477), inhibitor of nuclear factor κB
kinase (IKK; cat. no. ab178870), NF-κB (cat. no. ab32360), high
mobility group box 1 (HMGB1; cat. no. ab18256), toll-like receptor
(TLR)2 (cat. no. ab209217), TLR4 (cat. no. ab22048), IL-1β (cat.
no. ab9722) and NLR family pyrin domain containing 3 (NLRP3; cat.
no. ab263899), antibodies were purchased from Abcam). After washing
with TBST (0.5% Tween-20) for three times, the membranes were
subsequently incubated with goat-anti rabbit and goat-anti mouse
HRP-labeled secondary antibodies at room temperature (1:1,000; cat.
nos. ab6721 and ab6789; Abcam) for 1 h. The protein expression
levels were detected using Immobilon Western HRP Substrate (cat.
no. WBKLS0100; EMD Millpore) and were semi-quantified using Image
Pro Plus 6.0 software (Media Cybernetics, Inc.). GAPDH was used as
an internal control.
ELISA
ELISA was performed according to the protocol of
each respective kit. The following kits were used and purchased
from Abcam: iNOS (cat. no. ab285316), COX-2 (cat. no. ab210574),
CCL9 (cat. no. ab240689), CXCL1 (cat. no. ab219044), CXCL9 (cat.
no. ab203364), CXCL11 (cat. no. ab204519). Briefly, cultured medium
and serum samples obtained from cells and mice were added to each
well of a 96-well plate and incubated at room temperature for 3 h.
Then, after washing four times with washing buffer, the samples
were incubated with antibodies included in each ELISA kit according
to the manufacturer's protocol at room temperature for 1 h,
followed by incubation with HRP-streptavidin solution for 45 min at
room temperature. After four incubations in washing buffer, the
samples were incubated with one-step substrate reagent for 30 min
at room temperature in the dark, then finally incubated with stop
solution. The absorbance value was detected at 450 nm.
Statistical analysis
Data are presented as the mean ± SD. Experiments
were repeated three times independently. The differences among
groups were evaluated with one-way ANOVA and followed by Tukey's
post hoc test. P<0.05 was considered to indicate a statistically
significant difference.
Results
Detection of H9c2 cell viability and
expression of TXNIP in cell and mice model of HF
As presented in Fig.
1, the protein expression levels of TXNIP in the control (blank
pcDNA3.1 vector), overexpression and control (blank lentiCRISPR v2)
knockdown groups as well as the expression of TXNIP in the mice
model of control, TXNIP overexpression and TXNIP knockout group,
were detected using western blotting. The protein expression levels
of TXNIP were significantly increased in the overexpression group
and significantly decreased in the knockdown group compared with
the controls (P<0.05; Fig. 1A).
And the viability rates of H9c2 cells in the NC, OT, OI and OH
groups were 100.0±8.8, 115.6±9.3, 142.2±13.3 and 96.2±9.1,
respectively (Fig. 1B). The
results indicated that the viability rate was significantly
increased in the OI group but markedly reduced in the OH group
compared with the NC group, which indicated that inhibition of
TXNIP may serve a protective role in H9c2 cells. The mRNA
expression levels of inflammatory-related genes and Nrf2/HO-1
signaling pathway genes, such as IL-6, IL-10, COX-2, Nrf2, HO-1 and
NF-κB, in each group without DHE treatment was detected via
RT-qPCR. Although the mRNA expression levels of these genes were
slightly changed, no significant difference was observed (Fig. 1C and D).
 | Figure 1Evaluation of model construction
using reverse transcription-quantitative PCR and western blotting,
and cell viability rate assessment using an MTT assay. (A) TXNIP
expression in overexpression and knockout groups without DHE
treatment in H9c2 cells and mice. H9c2 cells and mice transfected
with blank pcDNA3.1 vector were set as the negative control for the
TXNIP overexpression group, and cells transfected with blank
lantiCRISPR v2 vector were set as the negative control for the
TXNIP knockout group. (B) DHE effect on H9c2 cell viability rate
(%) compared with the control group. (C) Expression of inflammatory
related genes without DHE treatment. (D) Expression of Nrf2/HO-1
signaling pathway genes without DHE treatment.
*P<0.05 vs. control group. Data are presented as the
mean ± SD. Each experiment was repeated three times independently.
DHE, dehydrocostus lactone; TXNIP, thioredoxin-interacting protein;
Nrf2, nuclear factor erythroid 2-related factor 2; HO-1, heme
oxygenase-1; NC, negative control; OT, DHE treatment group; OI, DHE
treatment combined with the TXNIP inhibition group; OH, DHE
treatment combined with the TXNIP overexpression group; COX-2,
cyclooxygenase 2. |
mRNA expression levels of
inflammation-related genes in mouse heart tissues and H9c2
cells
As presented in Fig.
2, the expression levels of iNOS, TNF-α, IL-1β, IL-6, IL-10 and
COX-2 mRNA in H9c2 cells of the NC, OT, OI and OH groups were
detected using RT-qPCR. Briefly, the expression of iNOS mRNA was
significantly increased in OI group compared with the NC and OT
groups (P<0.05). The expression of TNF-α in these groups was
significantly decreased in OT and OI group compared with NC group
(P<0.05), and was significantly decreased in the OI group
compared with the OT group (P<0.05). The expression of IL-1β in
these groups was significantly decreased in the OI group compared
with the NC and OT groups (P<0.05). IL-10 expression levels were
significantly increased in the OT and OI groups compared with the
NC group (P<0.05), and were significantly increased in the OI
group compared with the OT group (P<0.05). The expression of
COX-2 in these groups was also significantly decreased in the OI
group compared with NC and OT groups (P<0.05).
 | Figure 2Expression levels of inflammation and
oxidative-reduction related mRNA in H9c2 cells using reverse
transcription-quantitative PCR. (A) iNOS, (B) TNF-α, (C) IL-1, (D)
IL-6, (E) IL-10 and (F) COX-2 in H9c2 cells. Data are presented as
the mean ± SD. Each experiment was repeated three times
independently. *P<0.05 vs. NC group;
#P<0.05 vs. OT group. DHE, dehydrocostus lactone;
TXNIP, thioredoxin-interacting protein; NC, negative control; OT,
DHE treatment group; OI, DHE treatment combined with the TXNIP
inhibition group; OH, DHE treatment combined the with TXNIP
overexpression group; iNOS, inducible nitric oxide synthase; COX-2,
cyclooxygenase-2. |
As presented in Fig.
3, the expression of iNOS, TNF-α, IL-1β, IL-6, IL-10 and COX-2
mRNA in mice heart tissue of NC, OT, OI and OH group was detected
using RT-qPCR. The expression of iNOS in these groups was
significantly increased in the OI group compared with the NC and OT
group (P<0.05). The expression of TNF-α in these groups was
significantly decreased in the OT and OI groups compared with the
NC group (P<0.05), and was significantly decreased in the OI
group compared with the OT group (P<0.05). IL-1β expression
levels were significantly decreased in the OI group compared with
the NC and OT groups (P<0.05). Additionally, the expression of
IL-6 in these groups was significantly decreased in the OT and OI
groups compared with the NC group (P<0.05), and was
significantly decreased in the OI group compared with the OT group
(P<0.05). The expression of IL-10 in these groups was
significantly increased in OT and OI group compared with the NC
group (P<0.05), and was significantly increased in the OI group
compared with the OT group (P<0.05). The expression of COX-2 in
these groups was significantly decreased in OI group compared with
the NC and OT groups (P<0.05). iNOS and IL-10 mRNA expression
levels were significantly increased after TXNIP knockout compared
with the NC and OT groups, whereas the expression of
proinflammatory factors, such as TNF-α, IL-1β, IL-6 and COX-2, was
decreased following TXNIP knockout compared with the NC and OT
groups. The results indicated that inhibition of TXNIP reduced the
inflammatory response in H9c2 cells and in mouse heart tissues.
 | Figure 3Expression levels of inflammation and
oxidative-reduction related mRNA in heart tissue of mice using
reverse transcription-quantitative PCR. (A) iNOS, (B) TNF-α, (C)
IL-1, (D) IL-6, (E) IL-10 and (F) COX-2 in mouse heart tissues.
Data are presented as the mean ± SD. Each experiment was repeated
three times independently. *P<0.05 vs. NC group;
#P<0.05 vs. OT group. DHE, dehydrocostus lactone;
TXNIP, thioredoxin-interacting protein; NC, negative control; OT,
DHE treatment group; OI, DHE treatment combined with TXNIP
inhibition group; OH, DHE treatment combined with TXNIP
overexpression group; iNOS, inducible nitric oxide synthase; COX-2,
cyclooxygenase-2. |
Protein expression levels of the
HO-1/NF-κB signaling pathway in mice heart tissues and H9c2
cells
As presented in Fig.
4, the expression of the HO-1/NF-κB signaling pathway in NC,
OT, OI and OH groups of H9c2 cells was detected using western
blotting. The expression of Keap1 in these groups was significantly
decreased in OT and OI groups, while significantly increased in the
OH group compared with the NC group (P<0.05). Levels were also
significantly decreased in the OI group and significantly increased
in the OH group compared with the OT group (P<0.05). The
expression of Nrf2 was significantly increased in OT and OI groups
compared with the NC group (P<0.05), and significantly increased
in the OI group compared with OT group (P<0.05). Additionally,
the expression of HO-1 was significantly increased in OT and OI
groups compared with the NC group (P<0.05), and significantly
increased in the OI group and significantly decreased in the OH
group compared with the OT group (P<0.05). The expression of IKK
in these groups was significantly increased in all treatment groups
compared with NC group (P<0.05), and was significantly increased
in OI group compared with OT group (P<0.05). The expression of
NF-κB in these groups was significantly decreased in OI group
compared with NC and OT group (P<0.05), and was significantly
increased in OH group compared with NC and OT group
(P<0.05).
 | Figure 4Protein expression levels of HO-1
regulator proteins in H9c2 cells using western blot method. (A)
Western blotting analysis and quantitative analysis of (B) Keap1,
(C) Nrf2, (D) HO-1, (E) IKK and (F) NF-κB in H9c2 cells. Data are
presented as the mean ± SD. Each experiment was repeated three
times independently. *P<0.05 vs. NC group.
#P<0.05 vs. OT group. DHE, dehydrocostus lactone;
TXNIP, thioredoxin-interacting protein; NC, negative control; OT,
DHE treatment group; OI, DHE treatment combined with TXNIP
inhibition group; OH, DHE treatment combined with TXNIP
overexpression group; Nrf2, nuclear factor erythroid 2-related
factor 2; HO-1, heme oxygenase-1; Keap1, Kelch-like ECH-associated
protein 1; MW, molecular weight. |
As presented in Fig.
5, the expression of HO-1/NF-κB signaling pathway in NC, OT, OI
and OH groups of heart tissues was detected using western blotting.
The expression of Keap1 in these groups was significantly decreased
in the OI group compared with NC and OT groups, and significantly
decreased in the OH group compared with the NC group. The
expression of Nrf2 in these groups was significantly increased in
the OI group compared with NC and OT groups, while significantly
decreased in OH group compared with the NC and OT groups. The
expression of HO-1 in these groups was significantly increased in
the OI group compared with the NC and the OT group. The expression
of IKK in these groups was significantly increased in OT and OI
group compared with the NC group, and was significantly increased
in the OI group and significantly decreased in the OH group
compared with the OT group. The expression of NF-κB in these groups
was significantly decreased in the OI group compared with NC and OT
groups.
 | Figure 5Protein expression levels of
proinflammation proteins in heart tissue of mice using western blot
method. (A) Western blotting analysis and quantitative analysis of
(B) Keap1, (C) Nrf2, (D) HO-1, (E) IKK and (F) NF-κB. Data are
presented as the mean ± SD. Each experiment was repeated three
times independently. *P<0.05 vs. NC group.
#P<0.05 vs. OT group. DHE, dehydrocostus lactone;
TXNIP, thioredoxin-interacting protein; NC, negative control; OT,
DHE treatment group; OI, DHE treatment combined with TXNIP
inhibition group; OH, DHE treatment combined with TXNIP
overexpression group; TLR, toll-like receptor; HMGB1, high mobility
group protein B1; NLRP3, NOD-, LRR- and pyrin domain-containing
protein 3; MW, molecular weight. |
These results indicated that, in both cells and
mice, the HO-1/NF-κB signaling pathway was activated after TXNIP
knockout, as the expression of HO-1 was significantly increased. It
was therefore hypothesized that TXNIP knockout may have a
protective role in H9c2 cells and mouse heart tissues.
Protein expression levels of
inflammation response-related molecules in mice heart tissues and
H9c2 cells
As presented in Fig.
6, the expression of HMGB1, TLR2, TLR4, IL-1β and NLRP3 in NC,
OT, OI and OH group of H9c2 cells were detected using western
blotting. The expression of HMGB1 in these groups was significantly
decreased in OT and OI group compared with NC group (P<0.05),
and was significantly decreased in OI group and significantly
increased in OH group compared with OT group (P<0.05). The
expression of TLR2 in these groups was significantly decreased in
OI group compared with NC and OT group (P<0.05), and was
significantly increased in OH group compared with OT group
(P<0.05). The expression of TLR4 in these groups was
significantly decreased in OI group compared with NC and OT group
(P<0.05), and was significantly increased in OH group compared
with NC and OT group (P<0.05). The expression of IL-1β in these
groups was significantly increased in OH group compared with NC and
OT group (P<0.05). The expression of NLRP3 in these groups was
significantly decreased in OI group compared with NC and OT group
(P<0.05), and significantly increased in OH group compared with
OT group (P<0.05).
 | Figure 6Protein expression levels of HO-1
regulator proteins in H9c2 cells using western blot method. (A)
Western blotting analysis and quantitative analysis of (B) HMGB1,
(C) TLR2, (D) TLR4, (E) IL-1β and (F) NLRP3 in H9c2 cells. Data are
presented as the mean ± SD. Each experiment was repeated three
times independently. *P<0.05 vs. NC group.
#P<0.05 vs. OT group. DHE, dehydrocostus lactone;
TXNIP, thioredoxin-interacting protein; NC, negative control; OT,
DHE treatment group; OI, DHE treatment combined with TXNIP
inhibition group; OH, DHE treatment combined with TXNIP
overexpression group; Nrf2, nuclear factor erythroid 2-related
factor 2; HO-1, heme oxygenase-1; Keap1, Kelch-like ECH-associated
protein 1; MW, molecular weight. |
As for mouse heart tissues (Fig. 7), the expression of HMGB1, TLR2,
TLR4, IL-1β and NLRP3 in NC, OT, OI and OH group of heart tissues
were detected using western blotting. The expression of HMGB1 in
these groups was significantly deceased in OI group compared with
NC and OT group (P<0.05), and significantly increased in OH
group compared with NC and OT group (P<0.05). The expression of
TLR2 in these groups was significantly decreased in OI group
compared with NC and OT group (P<0.05). The expression of TLR4
in these groups was significantly decreased in OT and OI group
compared with NC group (P<0.05), and was significantly decreased
in OI group compared with OT group and significantly increased in
OH group compared with OT group (P<0.05). The expression of
IL-1β in these groups was significantly decreased in OT and OI
group compared with NC group (P<0.05), and was significantly
decreased in OI group and significantly increased in OH group
compared with OT group (P<0.05). The expression of NLRP3 in
these groups was significantly decreased in OI group compared with
NC and OT group (P<0.05).
 | Figure 7Expression of pro-inflammation
proteins in mouse heart tissues using western blot method. (A)
Western blotting analysis and quantitative analysis of (B) HMGB1,
(C) TLR2, (D) TLR4, (E) IL-1β and (F) NLRP3 in H9c2 cells. Data are
presented as the mean ± SD. Each experiment was repeated three
times independently. *P<0.05 vs. NC group.
#P<0.05 vs. OT group. DHE, dehydrocostus lactone;
TXNIP, thioredoxin-interacting protein; NC, negative control; OT,
DHE treatment group; OI, DHE treatment combined with TXNIP
inhibition group; OH, DHE treatment combined with TXNIP
overexpression group; TLR, toll-like receptor; HMGB1, high mobility
group protein B1; NLRP3, NOD-, LRR- and pyrin domain-containing
protein 3; MW, molecular weight. |
These results indicated that TXNIP inhibition
reduced the protein expression levels of inflammation-related
molecules.
Concentration of inflammation-related
and oxidation-reduction related factors in serum samples of mice
and cultured medium of H9c2 cells
The concentrations of iNOS, which is related to the
oxidative-reduction process, and COX-2, CCL9, CXCL1, CXCL9 and
CXCL11 which were critical for the activation of inflammation
process, in culture medium of H9c2 cells of NC, OT, OI and OH group
was quantified using ELISA method and the results are presented in
Fig. 8. The concentration of iNOS
in NC, OT, OI and OH group of H9c2 cells was significantly
increased in OI and OT group compared with NC group (P<0.05),
and was significantly increased in OI group and significantly
decreased in OH group compared with OT group (P<0.05). The
concentration of COX-2 in these groups was significantly decreased
in OT and OI group compared with NC group (P<0.05), and was
significantly decreased in OI group compared with OT group
(P<0.05). The concentration of CCL9 in these groups was
significantly decreased in OT and OI group compared with NC group
(P<0.05), and was significantly decreased in OI group compared
with OT group (P<0.05). The changing in concentration of CXCL1
and CXCL9 in these groups presented a similar trend with CCL9
(P<0.05). And the concentration of CXCL11 in these groups was
significantly decreased in OI group compared with NC and OT group
(P<0.05).
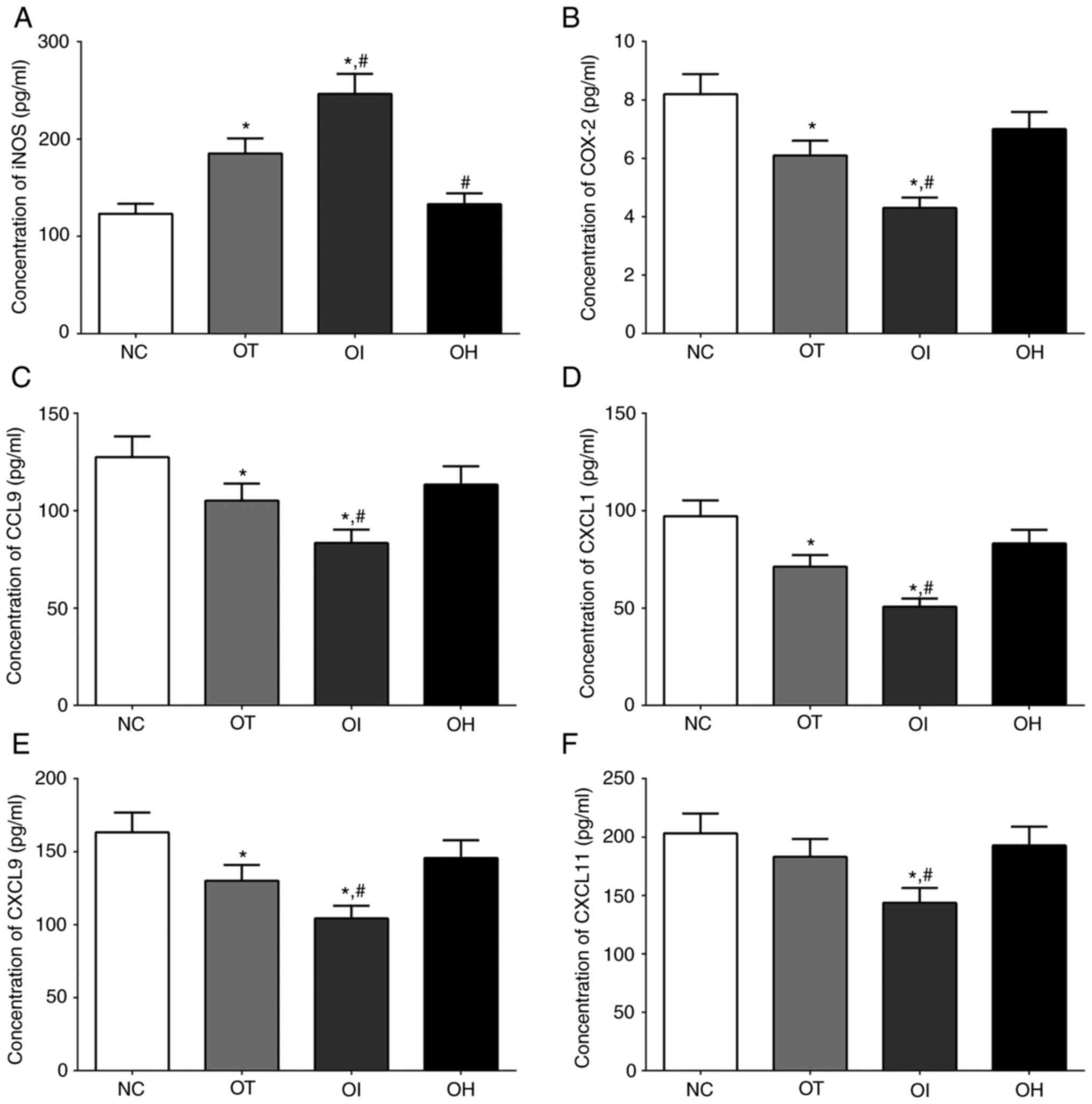 | Figure 8Detection of inflammation and
oxidative-reduction related factors in H9c2 cells using ELISA
method. (A) iNOS, (B) COX-2, (C) CCL9, (D) CXCL1, (E) CXCL9 and (F)
CXCL11 in the culture medium of H9c2 cells. Data are presented as
the mean ± SD. Each experiment was repeated three times
independently. *P<0.05 vs. NC group.
#P<0.05 vs. OT group. DHE, dehydrocostus lactone;
TXNIP, thioredoxin-interacting protein; NC, negative control; OT,
DHE treatment group; OI, DHE treatment combined with TXNIP
inhibition group; OH, DHE treatment combined with TXNIP
overexpression group; iNOS, inducible nitric oxide synthase; COX-2,
cyclooxygenase-2; CCL9, C-C motif ligand 9; CXCL, chemokine C-X-C
motif ligand. |
As reported in Fig.
9, The concentration of iNOS in NC, OT, OI and OH group of
heart tissues was significantly increased in OI group compared with
NC and OT group (P<0.05). The concentration of COX-2 in these
groups was significantly decreased in OT and OI group compared with
NC group (P<0.05), and was significantly decreased in OI group
compared with OT group (P<0.05). The concentration of CCL9 and
CXCL1 in these groups presented a similar trend with COX-2
(P<0.05). The concentration of CXCL9 and CXCL11 in these groups
was significantly decreased in OI group compared with NC and OT
group (P<0.05).
 | Figure 9Detection of inflammation and
oxidative-reduction related factors in heart tissue of mice using
ELISA method (A) iNOS, (B) COX-2, (C) CCL9, (D) CXCL1, (E) CXCL9
and (F) CXCL11 in mouse heart tissues. Data are presented as the
mean ± SD. Each experiment was repeated three times independently.
*P<0.05 vs. NC group. #P<0.05 vs. OT
group. DHE, dehydrocostus lactone; TXNIP, thioredoxin-interacting
protein; NC, negative control; OT, DHE treatment group; OI, DHE
treatment combined with TXNIP inhibition group; OH, DHE treatment
combined with TXNIP overexpression group; iNOS, inducible nitric
oxide synthase; COX-2, cyclooxygenase-2; CCL9, C-C motif ligand 9;
CXCL, chemokine C-X-C motif ligand. |
These results indicated that DHE treatment decreased
the secretion of inflammatory response-related factors and the
knockout of TXNIP significantly enhanced this trend compared with
control group.
Discussion
HF affects more than 5.7 million people in the US,
according to previous data (21).
In 2013, nearly 500,000 new cases occurred in people >55 years
(22). DHE is obtained from
Chinese herbs and can be used in the treatment of various diseases,
such as liver cancer, colorectal cancer and leukemia (23,24).
TXNIP, an endogenous inhibitor of thioredoxin, serves an important
role in the resistance to oxidative stress induced by extracellular
stimulations, including reactive oxygen species and hypoxia
(25).
Under inflammatory conditions, TNF-α and the
expression of other inflammation-related cytokines are induced by
coagulation factors, such as factor VII and XI, further inducing
the formation of complexes with factor VII and resulting in the
activation of thrombin (26).
Moreover, factor Xa, thrombin and the tissue factor-factor VII
complex can increase the expression of TNF-α and other
proinflammatory cytokines (27),
promoting the blood coagulation process. Furthermore, factor Xa and
thrombin directly affect the function of vascular cells (27) via protease-activated receptors and
the nitric oxide (NO)/cGMP signaling pathway (28). The NO/cGMP signaling pathway has
numerous effects on the vasculature and dysfunction of this
signaling pathway leads to a reduction in cGMP production, which
results in the occurrence of cardiovascular diseases (29). A previous study also indicated that
the synthesis of NO inhibited the activity of TF (30) and prevented the formation of
thrombi in circulation via its vasodilator and anti-aggregatory
ability (31). The present study
demonstrated that DHE significantly inhibited the expression of
inflammation-related factors, including IL-1, IL-6, IL-10, COX-2
and TNF-α and had an inhibitory effect on the activation of the
inflammatory response. Furthermore, the present study reported that
DHE increased the expression levels of iNOS, which contributes to
the protective effect of DHE.
TLRs are divided into two groups according to their
cellular distribution: The cell membrane group (TLR1, 2, 4, 5 and
6) and the endosome membrane within the cell group (TLR3, 7, 8 and
9) (32). The transduction of
cellular signals via TLRs to the nucleus leads to the activation of
innate and adaptive immune responses via the myeloid
differentiation primary response 88-dependent pathway and the
Toll/IL-1 receptor domain-containing adapter-inducing
IFN-β-dependent pathway, leading to the activation of the NF-κB
signaling pathway (33). Under
extracellular stimulation, the NF-κB signaling pathway is activated
via TLRs, nod-like receptors or other cytokine receptors.
Activation of the NF-κB signaling pathway further leads to the
activation of NLRP3, initiating inflammasome activation (34). The NLRP3 inflammasome can recognize
numerous stimuli and responds to endogenous factors that can lead
to cellular damage, including uric acid crystals, drusen and
extracellular ATP (35,36). TLR activation also induces the
expression of pro-IL-1β (37). In
the present study, the activation of these factors upon HF was
inhibited after DHE treatment and was even lower after TNXIP
inhibition, indicating that the activation of the inflammatory
response in H9c2 cardiomyocytes and heart tissues was inhibited,
and presented a protective role.
HO-1 is an enzyme presenting cytoprotective and
antioxidant effects (38). The
expression of HO-1 is regulated at the transcriptional level via
the regulation of Nrf2(39). Under
normal conditions, Nrf2 binds to Keap1(40) and when oxidated Nrf2 is released
from Keap1 and translocated into the nucleus, further binding with
antioxidant response element sequences of the HO-1 promoter region
(41). The activation of HO-1
further leads to the activation of NF-κB, which inhibits the
expression of IL-6, TNF-α and IL-1β and subsequently the
inflammatory response process (42). In the present study, the protein
expression levels of HO-1 were significantly increased after
treatment with DHE combined with TXNIP konckout, presenting a
protective effect (Fig. 10).
Although the results indicated that TXNIP inhibition
contributed to the therapeutic effect of DHE, the present study has
numerous limitations. The detailed mechanism of how TXNIP
inhibition promoted the effect of DHE is not fully understood and
it should be explored in future experiments. The Nrf2/HO-1
signaling pathway expression in TXNIP overexpression and knockout
groups without DHE treatment was detected using RT-qPCR only; in
further experiments, the protein levels should also be detected.
Other methods could contribute to explore the aforementioned
mechanisms, such as mass spectrometry, RNA sequencing and chromatin
immunoprecipitation sequencing. Clinical samples would also help
understand the association between TXNIP and DHE, and the mechanism
of TXNIP knockout promoting the function of DHE.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XZ and CC performed the cell, mice, protein and
nucleic acid experiments, and wrote the manuscript. YH designed the
experiment and revised the manuscript. XZ and YH confirm the
authenticity of all the raw data. All authors have read and
approved the final manuscript.
Ethics approval and consent to
participate
The present study was carried out in strict
accordance with the recommendations in the Guide for the Care and
Use of Laboratory Animals of the National Institutes of Health
(17). The present protocol and
experiments were approved by the Ethics Committee of Animal
Experiments of Anqiu People's Hospital (Anqiu, China; approval no.
IACUC-20200920).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Virani SS, Alonso A, Benjamin EJ,
Bittencourt MS, Callaway CW, Carson AP, Chamberlain AM, Chang AR,
Cheng S, Delling FN, et al: Heart disease and stroke
statistics-2020 Update: A report from the American Heart
association. Circulation. 141:e139–e596. 2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Hofmann U and Frantz S: How can we cure a
heart ‘in flame’? A translational view on inflammation in heart
failure. Basic Res Cardiol. 108(356)2013.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Ammirati E, Cannistraci CV, Cristell NA,
Vecchio V, Palini AG, Tornvall P, Paganoni AM, Miendlarzewska EA,
Sangalli LM, Monello A, et al: Identification and predictive value
of interleukin-61 interleukin-101 and interleukin-62
interleukin-101 cytokine patterns in ST-elevation acute myocardial
infarction. Circ Res. 111:1336–1348. 2012.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Bozkurt B, Torre-Amione G, Warren MS,
Whitmore J, Soran OZ, Feldman AM and Mann DL: Results of targeted
anti-tumor necrosis factor therapy with etanercept (ENBREL) in
patients with advanced heart failure. Circulation. 103:1044–1047.
2001.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Taniguchi M, Kataoka T, Suzuki H, Uramoto
M, Ando M, Arao K, Magae J, Nishimura T, Otake N and Nagai K:
Costunolide and dehydrocostus lactone as inhibitors of killing
function of cytotoxic T lymphocytes. Biosci Biotechnol Biochem.
59:2064–2067. 1995.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Yuuya S, Hagiwara H, Suzuki T, Ando M,
Yamada A, Suda K, Kataoka T and Nagai K: Guaianolides as
immunomodulators. Synthesis and biological activities of
dehydrocostus lactone, mokko lactone, eremanthin, and their
derivatives. J Nat Prod. 62:22–30. 1999.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Jeong GS, Pae HO, Jeong SO, Kim YC, Kwon
TO, Lee HS, Kim NS, Park SD and Chung HT: The
alpha-methylenegamma-butyrolactone moiety in dehydrocostus lactone
is responsible for cytoprotective heme oxygenase-1 expression
through activation of the nuclear factor E2-related factor 2 in
HepG2 cells. Eur J Pharmacol. 565:37–44. 2007.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Zhang HW, Liu YM, Fang X, Gu L, Luo C,
Chen L and Wang Q: Vitamin D3 protects mice from
Diquat-induced oxidative stress through the NF-κB/Nrf2/HO-1
signaling pathway. Oxid Med Cell Longev.
2021(6776956)2021.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Wu YX, Jiang FJ, Liu G, Wang YY, Gao ZQ,
Jin SH, Nie YJ, Chen D, Chen JL and Pang QF: Dehydrocostus lactone
attenuates methicillin-resistant staphylococcus Aureus-induced
inflammation and acute lung injury via modulating macrophage
polarization. Int J Mol Sci. 22(9754)2021.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Kim HR, Kim JM, Kim MS, Hwang JK, Park YJ,
Yang SH, Kim HJ, Ryu DG, Lee DS, Oh H, et al: Saussurea
lappa extract suppresses TPA-induced cell invasion via
inhibition of NF-κB-dependent MMP-9 expression in MCF-7 breast
cancer cells. BMC Complement Altern Med. 14(170)2014.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Kim EJ, Lim SS, Park SY, Shin HK, Kim JS
and Park JH: Apoptosis of DU145 human prostate cancer cells induced
by dehydrocostus lactone isolated from the root of Saussurea
lappa. Food Chem Toxicol. 46:3651–3658. 2008.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Nie Y, Wang Z, Chai G, Xiong Y, Li B,
Zhang H, Xin R, Qian X, Tang Z, Wu J and Zhao P: Dehydrocostus
lactone suppresses LPS-induced acute lung injury and macrophage
activation through NF-κB signaling pathway mediated by p38 MAPK and
Akt. Molecules. 24(1510)2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Masutani H, Yoshihara E, Masaki S, Chen Z
and Yodoi J: Thioredoxin binding protein (TBP)-2/Txnip and
alpha-arrestin proteins in cancer and diabetes mellitus. J Clin
Biochem Nutr. 50:23–34. 2012.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Dai X, Liao R, Liu C, Liu S, Huang H, Liu
J, Jin T, Guo H, Zheng Z, Xia M, et al: Epigenetic regulation of
TXNIP-mediated oxidative stress and NLRP3 inflammasome activation
contributes to SAHH inhibition-aggravated diabetic nephropathy.
Redox Biol. 45(102033)2021.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Neville ES, Ophir S and Zhang F: Improved
vectors and genome-wide libraries for CRISPR screening. Nat
Methods. 11:783–784. 2014.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Wen JX, Zhang L, Liu H, Wang J, Li J, Yang
Y, Wang Y, Cai H, Li R and Zhao Y: Salsolinol attenuates
doxorubicin-induced chronic heart failure in rats and improves
mitochondrial function in H9c2 cardiomyocytes. Front Pharmacol.
10(1135)2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Wang K, Zhou A, Ruan M, Jin Z, Lu J, Wang
Q and Lu C: Dehydrocostus lactone suppresses ox-LDL-induced
attachment of monocytes to endothelial cells. Am J Transl Res.
11:6159–6169. 2019.PubMed/NCBI
|
|
18
|
National Research Council (US) Institute
for Laboratory Animal Research. Guide for the Care and Use of
Laboratory Animals. Washington (DC), National Academies Press (US),
1996.
|
|
19
|
Wang X, Gao Y, Tian Y, Liu X, Zhang G and
Wang Q, Xie W, Liu K, Qian Q and Wang Q: Integrative serum
metabolomics and network analysis on mechanisms exploration of
Ling-Gui-Zhu-Gan Decoction on doxorubicin-induced heart failure
mice. J Ethnopharmacol. 250(112397)2020.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods 25: 402-408.
|
|
21
|
Freddy AS and David G: Cardiac
contractility modulation: A novel approach for the treatment of
heart failure. Heart Fail Rev. 21:645–660. 2016.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Benjamin EJ, Blaha MJ, Chiuve SE, Cushman
M, Das SR, Deo R, de Ferranti SD, Floyd J, Fornage M, Gillespie C,
et al: Heart disease and stroke statistics-2017 update: A report
from the American Heart Association. Circulation. 135:e146–e603.
2017.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Peng Z, Wang Y, Fan J, Lin X, Liu C, Xu Y,
Ji W, Yan C and Su C: Costunolide and dehydrocostuslactone
combination treatment inhibit breast cancer by inducing cell cycle
arrest and apoptosis through c-Myc/p53 and AKT/14-3-3 pathway. Sci
Rep. 7(41254)2017.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Sun X, Kang H, Yao Y, Chen H, Sun L, An W,
Jiang E, Wang S and Hu X: Dehydrocostus lactone suppressed the
proliferation, migration, and invasion of colorectal carcinoma
through the downregulation of eIF4E expression. Anticancer Drugs.
26:641–648. 2015.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Han X, Wu YC, Meng M, Sun QS, Gao SM and
Sun H: Linarin prevents LPS-induced acute lung injury by
suppressing oxidative stress and inflammation via inhibition of
TXNIP/NLRP3 and NF-κB pathways. Int J Mol Med. 42:1460–1472.
2018.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Iannucci J, Renehan W and Grammas P:
Thrombin, a mediator of coagulation, inflammation, and
neurotoxicity at the neurovascular interface: Implications for
Alzheimer's disease. Front Neurosci. 14(762)2020.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Pawlinski R, Pedersen B, Kehrle B, Aird
WC, Frank RD, Guha M and Mackman N: Regulation of tissue factor and
inflammatory mediators by Egr-1 in a mouse endotoxemia model.
Blood. 10:3940–3947. 2003.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Scotton CJ, Krupiczojc MA, Königshoff M,
Mercer PF, Lee YC, Kaminski N, Morser J, Post JM, Maher TM,
Nicholson AG, et al: Increased local expression of coagulation
factor X contributes to the fibrotic response in human and murine
lung injury. J Clin Invest. 1199:2550–2563. 2009.PubMed/NCBI View
Article : Google Scholar
|
|
29
|
Motley ED, Eguchi K, Patterson MM, Palmer
PD, Suzuki H and Eguchi S: Mechanism of endothelial nitric oxide
synthase phosphorylation and activation by thrombin. Hypertension.
49:577–583. 2007.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Stasch JP and Hobbs AJ: NO-independent,
haem-dependent soluble guanylate cyclase stimlators. Handb Exp
Pharmacol: 277-308, 2009 doi: 10.1007/978-3-540-68964-5_13.
|
|
31
|
Yang Y and Loscalzo J: Regulation of
tissue factor expression in human microvascular endothelial cells
by nitric oxide. Circulation. 101:2144–2148. 2000.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Tschudi MR and Luscher TF: Nitric oxide:
The endogenous nitrate in the cardiovascular system. Herz.
21:50–60. 1996.PubMed/NCBI
|
|
33
|
Rakoff-Nahoum S and Medzhitov R: Toll-like
receptors and cancer. Nat Rev Cancer. 9:57–63. 2009.PubMed/NCBI View
Article : Google Scholar
|
|
34
|
Piras V and Selvarajoo K: Beyond MyD88 and
TRIF pathways in Toll-like receptor signaling. Front Immunol.
5(70)2014.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Bauernfeind FG, Horvath G, Stutz A,
Alnemri ES, MacDonald K, Speert D, Fernandes-Alnemri T, Wu J, Monks
BG, Fitzgerald KA, et al: Cutting edge: NF-kappaB activating
pattern recognition and cytokine receptors license NLRP3
inflammasome activation by regulating NLRP3 expression. J Immunol.
183:787–791. 2009.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Franchi L, Muñoz-Planillo R and Núñez G:
Sensing and reacting to microbes through the inflammasomes. Nat
Immunol. 13:325–332. 2012.PubMed/NCBI View
Article : Google Scholar
|
|
37
|
Mariathasan S, Weiss DS, Newton K, McBride
J, O'Rourke K, Roose-Girma M, Lee WP, Weinrauch Y, Monack DM and
Dixit VM: Cryopyrin activates the inflammasome in response to
toxins and ATP. Nature. 440:228–232. 2006.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Franchi L, Eigenbrod T and Núñez G:
Cutting edge: TNF-alpha mediates sensitization to ATP and silica
via the NLRP3 inflammasome in the absence of microbial stimulation.
J Immunol. 183:792–796. 2009.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Kapitulnik J: Bilirubin: An endogenous
product of heme degradation with both cytotoxic and cytoprotective
properties. Mol Pharmacol. 66:773–779. 2004.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Srisook K, Kim C and Cha YN: Molecular
mechanisms involved in enhancing HO-1 expression: De-repression by
heme and activation by Nrf2, the ‘one-two’ punch. Antioxid Redox
Signa. l7:1674–1687. 2005.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Farombi EO and Surh YJ: Heme oxygenase-1
as a potential therapeutic target for hepatoprotection. J Biochem
Mol Biol. 39:479–491. 2006.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Owuor ED and Kong AN: Antioxidants and
oxidants regulated signal transduction pathways. Biochem Pharmacol.
64:765–770. 2002.PubMed/NCBI View Article : Google Scholar
|
























