Introduction
Diabetes is a pathological condition whose
importance is reflected by the negative impact it has on patient
quality of life, life expectancy and the high costs it involves.
Current data highlight the impressive dynamics in the increasing
prevalence of diabetes worldwide: In 2021 there were ~537 million
individuals with diabetes, aged between 20 and 79 years, with a
global prevalence of 8.7%, and it is estimated that by the year
2045 this number will increase to 783 million (1). Type 2 diabetes is a chronic disease
with a progressive character that represents ~90% of all the
detected cases of diabetes and is an independent risk factor for
the development of both microvascular complications (retinopathy,
neuropathy and nephropathy) and macrovascular complications
(cerebrovascular disease, coronary artery disease and peripheral
vascular disease). The most frequent signs and symptoms of
hyperglycaemia include an increased thirst (polydipsia), frequent
urination (polyuria), increased hunger (polyphagia), unintentional
weight loss, blurred vision and fatigue (2,3). The
management of patients with diabetes includes some essential
elements, including early identification of chronic complications
and adopting an appropriate therapeutic attitude dependent on the
stage (4,5).
Diabetic neuropathy is considered the most frequent
of the chronic complications and is defined according to the
American Diabetes Association by ‘the presence of symptoms and/or
signs of peripheral nerve dysfunction in people with diabetes,
after excluding other causes’; it is a risk factor for injuries
occurring to the feet, with severe evolutionary potential. The
progressive damage, both functional and morphological, affects
various components in the peripheral nervous system (4,6).
This results in a series of changes, such as atrophy of the leg
muscles, decrease in muscle strength with an imbalance between
flexor and extensor muscles action and collapse of the plantar
arch, meaning that a large part of the plantar area is exposed to
mechanical stress or exaggerated pressure points may appear, so the
leg becomes vulnerable to tissue injuries (4,6). In
addition, autonomic diabetic neuropathy includes a series of
consequences involved in the decrease of tissue resistance to
various aggressive actions (such as reduction of sweat secretion.
resulting in skin dehydration with reduced resistance and tendency
to fissure, altered microcirculation vasomotility and opening of
arterio-venous shunts with depleted nutritious blood flow). The
progressive decrease of various types of sensitivity (superficial,
thermal, pain and proprioceptive) in combination with these
deficiencies represents important risk factors for neuropathic
ulcer occurrence (2).
Vasculitis is a group of conditions causing
inflammatory processes that affect the vascular walls; this is
produced by immuno-allergic mechanisms and has a polymorphic
clinical expression, with an aetiology that is established in only
40-60% of cases. The inflammation can affect any type of vessel
(artery, capillary and vein), with any calibre and any topography.
The inflammatory process within the vascular wall can be both
general (signs and symptoms) and local (hemodynamic impairment).
The histological lesions identified in the case of vasculitis
suggest that the production mechanism is an immunological one,
against the background of inflammatory reactions induced by immune
complexes or by cellular mechanisms (7). The reason why various vascular
segments are affected depends on the size and chemical properties
of the immune complexes, but also on physical factors, such as the
turbulence of the blood flow and/or the existence of previous
vascular lesions, conditions associated with additional aggression
of the endothelium (8).
According to the revised Chapel
Hill-Consensus-Conference Nomenclature of Vasculitis (2012)
(7), vasculitis are classified
into primary and secondary types. Primary vasculitis are
autonomous, while secondary vasculitis occur in association or as a
result of another pathology, or due to the consumption of a drug.
Primary and secondary vasculitis are subclassified into small,
medium and large vessel vasculitis according to the size of the
affected vessels (9,10). Systemic vasculitis is characterized
by localized inflammation in the blood vessel wall, which affects
multiple vascular territories and organs. Less frequently,
vasculitis presents in a localized form, reflecting either the
limited expression of a systemic vasculitis or vascular
inflammation affecting a single organ. To differentiate the two
forms of localized vasculitis the 2012 Revised International Chapel
Hill Consensus Conference Nomenclature of Vasculitis proposed the
term ‘single-organ vasculitis (SOV)’ which defines ‘vasculitis in
arteries or veins of any size in a single organ and has no features
that indicate that it is a limited expression of a systemic
vasculitis’ (11,12). SOV must be defined according to the
affected organ, as well as according to the observed inflammatory
pattern. The inflammatory process can have a multifocal or diffuse
distribution, affecting the skin, central nervous system and
kidneys, or a unifocal distribution, affecting the aorta, breast
and structures from the urogenital or gastrointestinal systems
(13).
The following types of vasculitis are currently
included in the vasculitis classification: Cutaneous arteritis,
hypersensitivity vasculitis [cutaneous leukocytoclastic angiitis
(CLA)], isolated aortitis and primary central nervous system
vasculitis. The Chapel Hill Consensus Conference (7) proposed changing the name of
hypersensitivity vasculitis to CLA due to the cutaneous
manifestations that dominate the clinical picture, in which
neutrophils have a special implication. CLA occurs as part of type
III immune reactions, induced by the presence of immune complexes
at the tissue level, and histologically involves polymorphonuclear
and mononuclear inflammatory infiltrates and necrosis (14). Cutaneous vasculitides varies in
terms of severity, ranging from self-limited skin eruptions to
life-threatening conditions with multiple organ failure. In most
cases of cutaneous vasculitides, neutrophilic small vessel
vasculitis is present; this is usually known as CLA and is
differentiated from cutaneous arteritis by its ability to affect
all the small blood vessels (arteries, veins and capillaries), not
just the arteries. CLA is the most frequently encountered cutaneous
vasculitis in clinical practice, with a higher predominance among
adults than children (15,16). In general, the size of vessel
involvement is associated with the clinical morphology on the
histopathological examination. Small, mainly superficial vessel
involvement prompts palpable or non-palpable purpura, erythema,
vesiculo-bullous and pustular lesions (17).
Infections are the basis of a wide number of
vasculitis and it is assumed that they act as a trigger factor,
with the skin being considered the most frequently affected organ
(18,19). Here, the present study aimed to
present the case of a male patient with long standing, poorly
controlled type 2 diabetes mellitus in whom the poor evolution of a
surgical wound after a toe amputation resulted in vasculitis,
although there was no sign of bacteraemia. It is important to
present such cases, as although rare, they can be associated with a
poor prognosis and could be avoided by proper care of the surgical
wounds and a good glycaemic control.
Case report
The present study reports the case of a male
patient, 60 years old, who had a hereditary history of diabetes
(mother and brother), who was diagnosed with type 2 diabetes 21
years previously due to signs and symptoms of hyperglycaemia. The
patient was recorded as a current smoker (smoking >20 cigarettes
a day for ~20 years) who consumed alcohol and was non-compliant
with regard to prophylactic therapeutic education and dietary and
medicinal recommendations.
The patient's medical history noted the following:
Type 2 diabetes discovered at the age of 39 years, with
unfavourable evolution towards the stage of multiple chronic
complications (preproliferative diabetic retinopathy, diabetic
peripheral sensory-motor neuropathy and diabetic arteriopathy of
the lower limbs), multiple post-traumatic wounds at the plantar
level with progression to ulceration, gangrene, superinfection and
amputation of the affected segments. In the last 2 years, two
amputations had been performed, namely, the left third finger and
the left fifth finger (in the last month prior to hospital
admission). At admission in the Department of Diabetes, Nutrition
and Metabolic Diseases of the County Clinical Emergency Hospital of
Craiova (Craiova, Romania), in August 2022, the patient was
undergoing antidiabetic therapy with 2 g metformin per day and 120
mg gliclazide per day. The patient stated that his diabetologist
had explained the importance of insulin therapy initiation, but he
always refused the treatment.
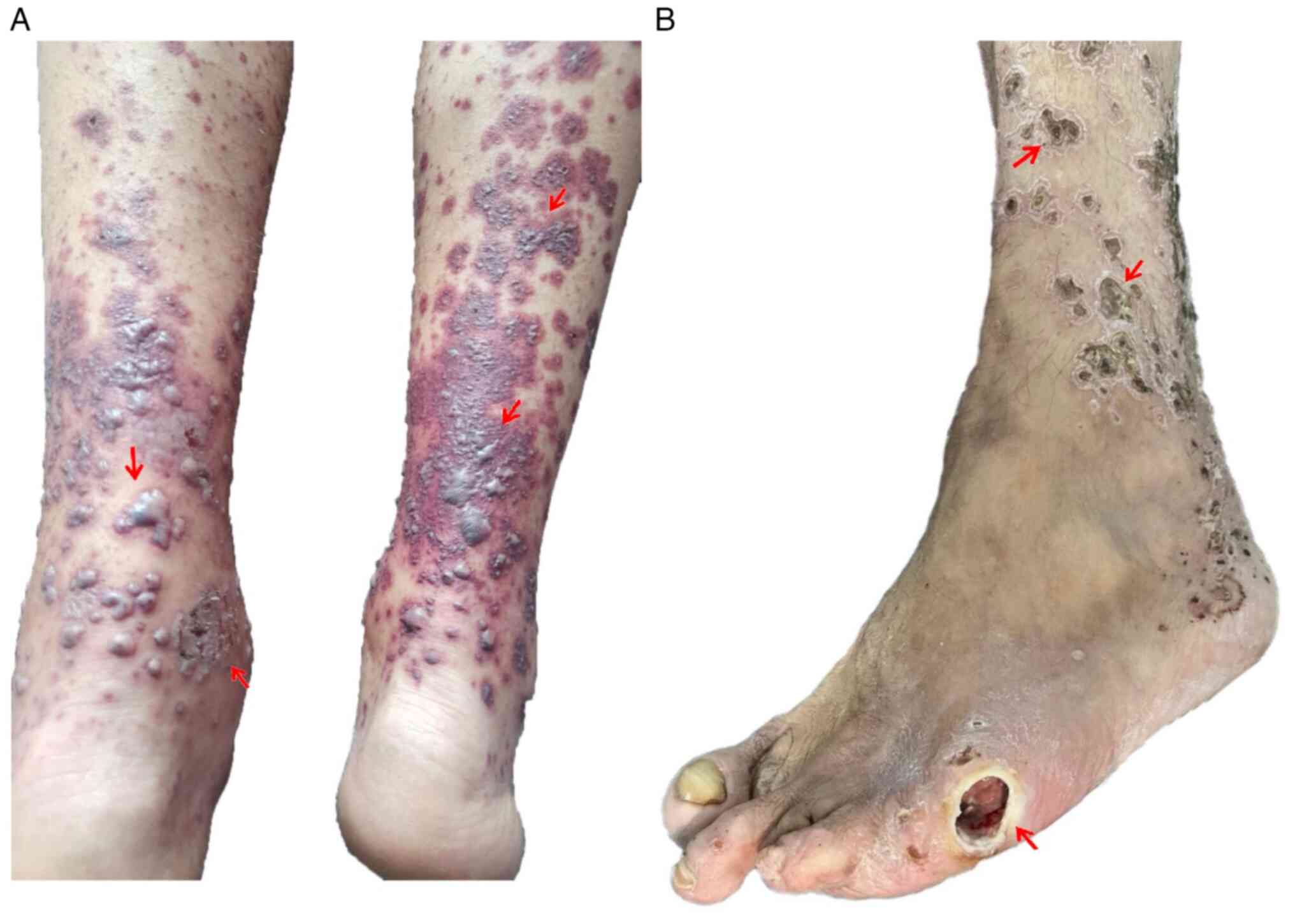
During this hospitalization, the patient presented
with a polymorphic eruption consisting of lesions with a purpuric
appearance, alternating with vesicles, bubbles and
erythemato-desquamative lesions covered by thick hematic crusts,
which were adherent, well defined and ranged in size between
0.5-6.0 cm in diameter located on the upper and lower limbs. This
was accompanied by itching and severe pain at night (Fig. 1A). An atonic wound was present,
with deep necrosis and osteitis, at the left fifth metatarsus
(Fig. 1B).
In July 2022, the amputation of the left fifth
finger was performed, and the resulting wound remained despite the
wound toilet and the daily local bandaging. This was followed by
the occurrence of a polymorphic eruption, which had an evolution of
~10 days prior to the current hospitalization. The patient denied
having any similar episodes previously. During the anamnesis and
physical examination, the patient reported ~10 days prior to
hospitalization the appearance of a rash consisting of raised
red-purple itchy lesions with a tendency to merge, and the
development of purplish plaques that persisted on digital pressure,
along with haemorrhagic vesicles and bubbles.
Laboratory tests revealed low haemoglobin levels
(12.09 g/dl; normal range: 13.10-17.20 g/dl), a normal platelet
count (446/ml; normal range: 150-450/ml), neutrophilia (18.85/ml;
normal range: 2-8/ml), leukocytosis (22.37/ml; normal range:
4-10/ml) and increased inflammatory markers compared with normal
reference values (fibrinogen, 575 mg/dl, normal range: 238-498
mg/dl; C reactive protein, 12 mg/l, normal range: 0-6 mg/dl).
Hepatorenal function tests were normal. The patient presented with
poorly controlled diabetes, with a haemoglobin A1c level of 86.5
mmol/mol (the individualized target for this patient was considered
53-58 mmol/mol). A complete evaluation was performed, including
assessment of markers for rheumatic diseases and vasculitis, serum
protein electrophoresis and extended antinuclear antibody blot
profile. The results of these markers were negative. Also, the
tests performed to detect hepatitis C virus (HCV) and hepatitis B
virus (HBV) infection (HB surface antigen and anti-HCV antibodies)
had negative results (Table I).
The patient also benefited from an arterial echo Doppler
examination of the lower limbs, which revealed atheromatous walls
of the circumflex femoral arteries, superficial femoral arteries
and popliteal arteries, while the posterior tibial arteries
presented very low flow on the right side, while the left side was
difficult to measure (Table
II).
 | Table ILaboratory tests results highlighting
the presence of the inflammatory syndrome. |
Table I
Laboratory tests results highlighting
the presence of the inflammatory syndrome.
| Parameters | At admission | 5th day after
treatment | 10th day after
treatment | Normal range |
|---|
| Hemoglobin, g/dl | 12.09 | 10.44 | 13.50 | 13.10-17.20 |
| White blood cell
count (x103/l) | 22.37 | 19.41 | 10.13 | 4-10 |
| Neutrophils, n
(x103/µl) | 18.85 | 15.38 | 9.45 | 2-8 |
| Platelet count
(x103/µl) | 446 | 435 | 412 | 150-450 |
| Serum creatinine,
mg/dl | 1.19 | - | 1.17 | 0.72-1.25 |
| Urea, mg/dl | 45 | - | 43 | 18-55 |
| Aspartate
transaminase, U/l | 14 | - | 16 | 5-34 |
| Alanine
transaminase, U/l | 25 | | 23 | 3-55 |
| C reactive protein,
mg/dl | 12 | <6 | <6 | 0-6 |
| Fibrinogen,
mg/dl | 575 | 512 | 451 | 238-498 |
| Erythrocyte
sedimentation rate, mm/h | 30 | 19 | 8 | 1-10 |
| Serum complement
C3, mg/dl | 94.53 | - | - | 90.00-180.00 |
| Serum complement
C4, mg/dl | 24.57 | - | - | 10.00-40.00 |
| Rheumatoid factor,
IU/ml | <8 | - | - | 0-8 |
| pANCA, U/ml | <2 | - | - | <2 |
| cANCA, U/ml | <1/10 | - | - | <1/10 |
| Antinuclear
antibodies | <1:100 | - | - | <1:100 |
| HbA1c,
mmol/mol | 86.5 | - | - | Individualized
target, 53-58 |
 | Table IIResults of arterial echo-Doppler
examination of the lower limbs. |
Table II
Results of arterial echo-Doppler
examination of the lower limbs.
| Region | Findings |
|---|
| Right CFA | 7.4 mm,
atheromatous walls, three-phase flow |
| Right SFA | 6.7 mm,
atheromatous walls, three-phase flow |
| Right popliteal
artery | 4.9 mm,
atheromatous walls, biphasic flow |
| Right PTA | 1.7 mm,
unreasonable flow |
| Right ATA | 1.4 mm, three-phase
flow |
| Left CFA | 7.1 mm,
atheromatous walls, three-phase flow |
| Left SFA | 5.9 mm,
atheromatous walls, biphasic flow |
| Left popliteal
artery | 4.9 mm,
atheromatous walls, three-phase flow |
| Left PTA | Difficult to
sonorize |
| Left ATA | 1.8 mm, biphasic
flow |
Following the clinical evaluation, a rheumatological
consultation was requested, and according to the recommendations,
treatment with methylprednisolone was initiated. Oral tablets at
24, 16, 8 and 4 mg concentrations were administered for 7 days
each, thus lowering the dose gradually to avoid possible adverse
effects, such as increased blood pressure, fluid retention and
changes in glucose tolerance. Regarding anti-diabetic therapy,
given the high value of the HbA1c and the glycaemic profile of the
patient, insulin therapy was initiated, using a basal bolus insulin
regimen, in doses that were adjusted daily according to the
capillary blood glucose levels of the patient determined 4-7
times/day. Considering the treatment with methylprednisolone and
the impact on the carbohydrate metabolism, the patient required
increased doses of insulin. The surgical consultation established
the need for excisional debridement and amputation of the distal
extremity in the left fifth metatarsal, with a tissue sample saved
for the antibiogram. The culture revealed methicillin-resistant
Staphylococcus aureus (MRSA), and according to the
antibiogram (Table III), the
patient received antibiotic treatment with a ciprofloxacin
intravenous infusion (10 mg/ml, 100 ml) twice a day for 14 days and
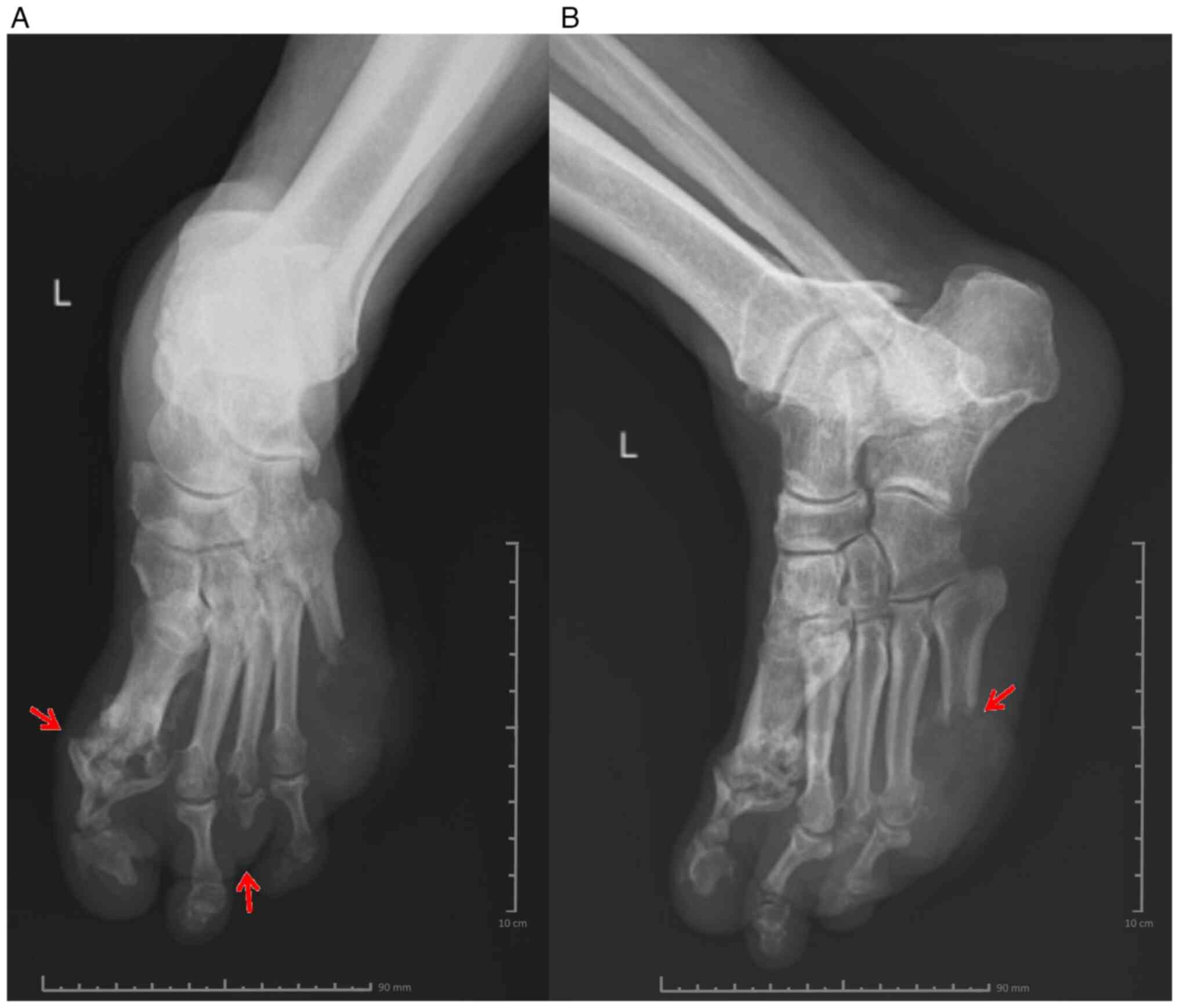
then ciprofloxacin tablets (500 mg) twice a day for 5 days. X-ray
of the left leg (frontal and lateral view) showed changes in the
bone structure at the level of the fifth metatarsal, and
circumscribed osteolysis at the level of the first
metatarsophalangeal joint and the distal extremity of the third
metatarsal (Fig. 2A and B).
 | Table IIIAntibiogram results from the tissue
samples highlighting the presence of MRSA sensitive to the
antibiotic ciprofloxacin. |
Table III
Antibiogram results from the tissue
samples highlighting the presence of MRSA sensitive to the
antibiotic ciprofloxacin.
| Antibiotic | MRSA
sensitivity |
|---|
| Ciprofloxacin | Sensitive |
| Clindamycin | Resistant |
| Cefazolin | Resistant |
| Clarithromycin | Resistant |
| Doxycycline | Resistant |
| Oxacilin | Resistant |
| Penicilin | Resistant |
|
Chloramphenicol | Resistant |
| Erythromycin | Resistant |
| Rifampicin | Resistant |
| Tigecycline | Resistant |
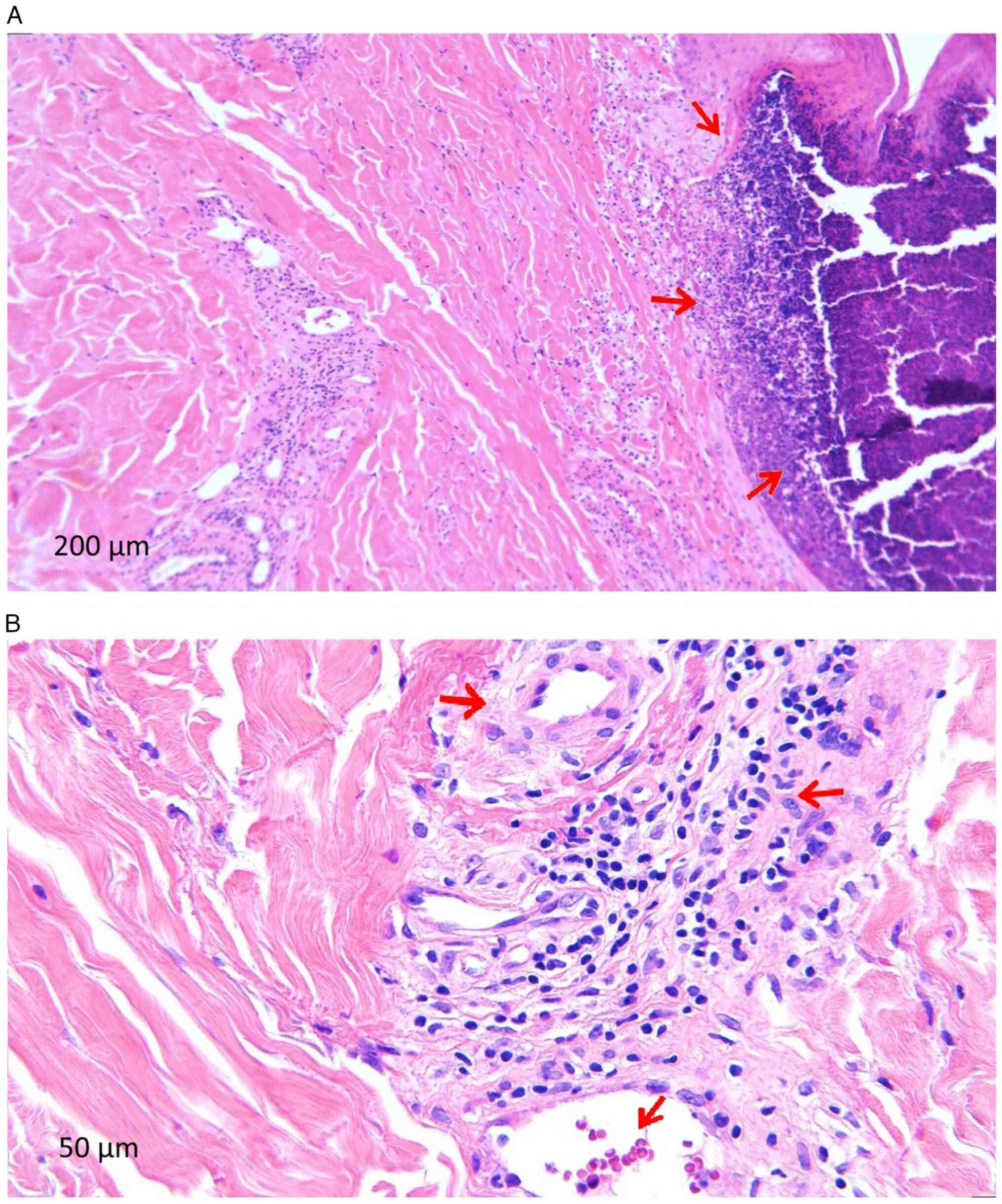
Following the dermatological examination, excisional
debridement was performed with the sampling of a lesional fragment
from the right calf. The histopathological examination was
performed in the Anatomical Pathology Department of the County
Clinical Emergency Hospital of Craiova following standard procedure
and the microscopic examination of a haematoxylin and eosin-stained
specimen performed highlighted the following features suggestive of
CLA (Fig. 3): i) Epidermis with
extensive ulcerated area (Fig.
3A); ii) numerous polymorphonuclear (PMN) capillaries with
thickened long walls; iii) turgescent endothelium, infiltrated with
lymphoid cells, arranged perivascularly but also in the vessel
walls; iv) presence of PMN in the capillary walls with fragmented
neutrophil nuclei; and vi) extravasated erythrocytes and
siderophages.
The rash improved significantly under the treatment
with methylprednisolone and ciprofloxacin, therefore it was assumed
that the rash was due to MRSA infection.
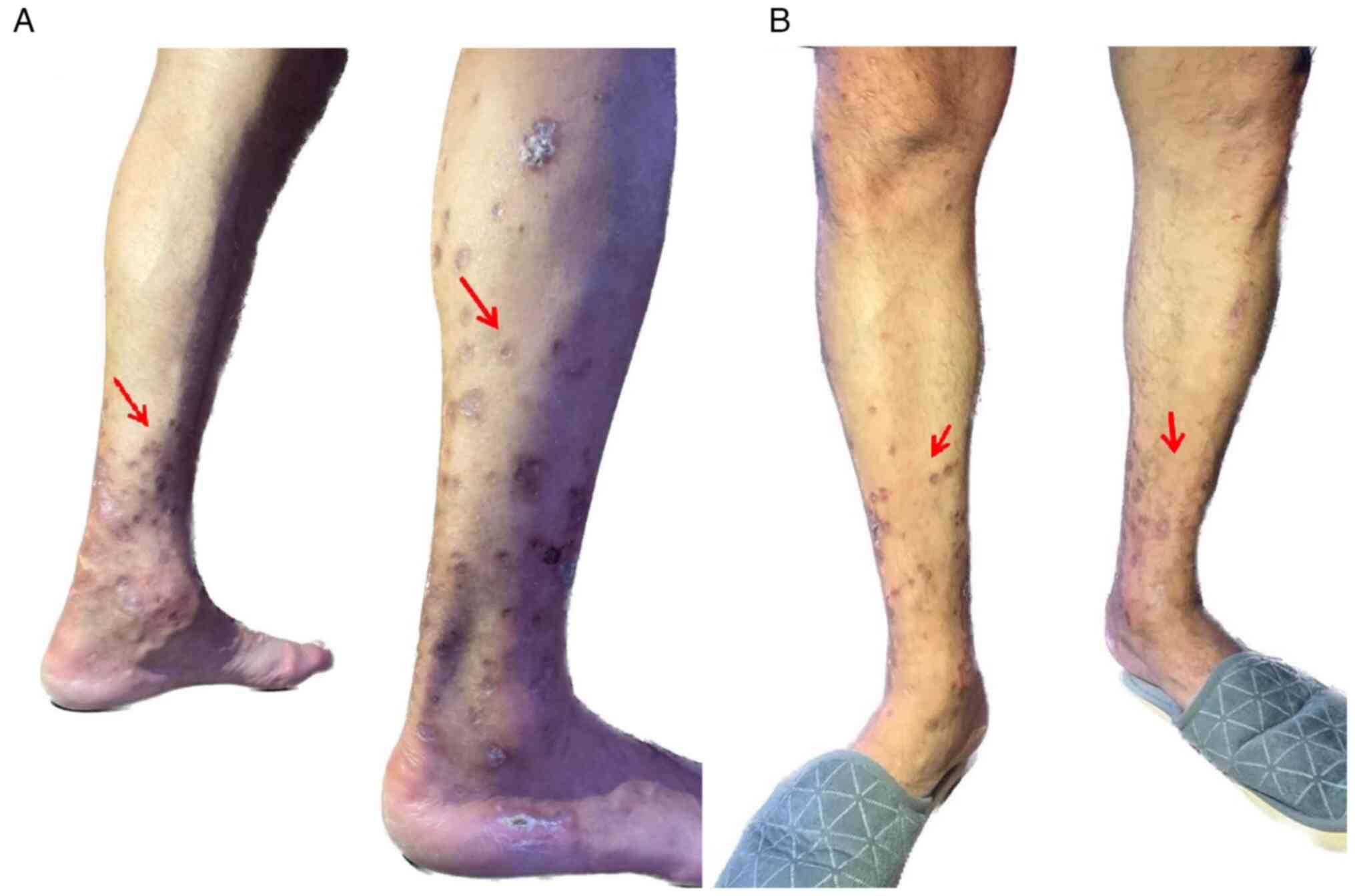
After 3 months, the patient presented for medical
examination, with repeated blood tests that showed remission of the
inflammatory syndrome (Table IV).
During the clinical evaluation, it was noted that the appearance of
the skin lesions on the lower limbs had improved (Fig. 4A and B). The patient continued the treatment
with insulin following the basal bolus regimen, with a small
improvement in the HbA1c level (72 mmol/mol compared with the
individualized target for the present patient which was considered
53-58 mmol/mol). Regarding the treatment of diabetes chronic
complications, during hospitalization the patient also received
oral atorvastatin (40 mg/day) and enoxaprine subcutaneously (0.5
mg/kg body weight, twice daily) for the presence of peripheral
arterial disease, at the indication of the surgeon and with the
approval of the ophthalmologist due to the presence of
pre-proliferative diabetic retinopathy and the high risk of
intraocular haemorrhage. Regarding diabetic peripheral neuropathy
the patient received intravenous 600 mg alpha lipoic acid daily.
After hospitalisation, the patient adhered to the prescribed
medication for diabetes (basal bolus insulin therapy) and its
associated complications (for peripheral arterial disease:
atorvastatin 40 mg per day orally and acetylsalicylic acid 75 mg
per day orally, as cilostazol therapy was contraindicated by the
ophthalmologist and for the presence of diabetic neuropathy alpha
lipoic acid 600 mg per day orally), the long-term prognosis remains
reserved due to the presence of advanced chronic diabetes
complications and the history of toe amputation, which are both
associated with an increased risk of cardiovascular disease
(20,21).
 | Table IVRepeated blood tests after 3 months
showing remission of the inflammatory syndrome. |
Table IV
Repeated blood tests after 3 months
showing remission of the inflammatory syndrome.
| Parameters | Result | Normal range |
|---|
| Hemoglobin,
g/dl | 14.13 | 13.10-17.20 |
| White blood cell
count (x103/l) | 8.45 | 4.00-10.00 |
| Neutrophils, n
(x103/µl) | 4.74 | 2.00-8.00 |
| Platelet count
(x103/µl) | 432 | 150-450 |
| Serum creatinine,
mg/dl | 0.78 | 0.72-1.25 |
| Aspartate
transaminase, U/l | 20 | 5-34 |
| Alanine
transaminase, U/l | 24 | 3-55 |
| C reactive protein,
mg/dl | 5 | 0-6 |
| Fibrinogen,
mg/dl | 276 | 238-498 |
| Erythrocyte
sedimentation rate, mm/h | 4 | 1-10 |
| HbA1c,
mmol/mol | 72 | Individualized
target, 53-58 |
Discussion
CLA is characterized by small vessel involvement and
the presence of an inflammatory infiltrate consisting of PMN
neutrophils and mononuclear cells. From a clinical point of view,
erythematous macules and papules are initially described, with
evolution towards a petechial eruption occurrence that does not
disappear by applying pressure and later palpable purpura. These
lesions can merge and ulcerate, and blisters, pustules and
haemorrhagic bubbles can develop. Most often, the lesions are
arranged symmetrically, in gravity-dependent areas, and can be
accompanied by symptoms such as itching, pain, tingling and
burning. This condition can be idiopathic or associated with an
infection, autoimmune disease, neoplasia or drug use; it is the
result of deposition of the immune complex in the vascular wall,
often in the context of a triggering event, such as an infectious
process. When the trigger factor is a drug or an infectious agent,
most patients develop symptoms after 7-9 days of exposure to the
antigenic elements (18,22). CLA manifests itself through an
isolated episode or through recurrent episodes, generally with
resolution in 20-30 days, while in cases of exposure to the sun or
extreme temperatures, the lesions may worsen. The skin is the most
frequently affected organ (15,19).
The evaluation of a patient with suspected CLA
should be guided towards confirming the diagnosis, identifying the
underlying aetiology and excluding major organ involvement. It is
necessary to identify suggestive symptoms for infection or systemic
disease. A detailed assessment is extremely important to identify a
possible trigger, as removal of the underlying cause may result in
the resolution of the CLA. The condition usually resolves with the
removal of triggering agents and treatment of the infection, but if
the skin rash is severe, additional therapies can be helpful
(17).
In 1990, the American College of Rheumatology
established the characteristic features of each form of vasculitis
and suggested the classification criteria for hypersensitivity
vasculitis (CLA) as follows: An age at disease onset of >16
years, possible medication in relation to symptoms, a palpable rash
that cannot be blanched with pressure and is not associated with
thrombocytopenia, a maculopapular rash defined by flat and raised
lesions of varying sizes covering one or more skin areas, and PMN
neutrophils in the wall of venules or arterioles upon biopsy
(18,23).
In the present case, the patient did not report a
similar episode in the past or the administration of drugs that
could be incriminated in triggering the rash. Also, based on the
clinical examination and the paraclinical findings, no systemic
cause was identified. The mainstay in establishing the diagnosis of
CLA was represented by the histopathological examination, where the
defining histological criteria were present in the form of vessel
wall infiltration by neutrophils, fragmented neutrophil nuclei and
extravasated erythrocytes.
The case was submitted to a multidisciplinary team
discussion for the differential diagnosis. The differential
diagnosis for CLA is extensive, but the main differentials
evaluated in this case were infection and small-vessel vasculitis.
Granulomatosis with polyangiitis is characterized by sinusitis,
oral and nasal ulcerations, antineutrophil cytoplasmic antibody
positivity in the majority of cases and involvement of the renal
system. Immunoglobulin A vasculitis may be similar in appearance
but occurs more often in children aged <11 years and affects the
gastrointestinal tract and the kidneys. Urticarial vasculitis
presents with low levels of total serum complement when there is
systemic involvement and would be recurrent. The syndrome of
essential cryoglobulinemia is also characterized by vasculitis of
small vessels, leukocytoclasia and palpable purpura, but is not
associated with a known antigenic stimulus and is generally a
chronic illness, whereas hypersensitivity vasculitis usually runs a
more limited course. Causes of infection, such as viruses and
fungi, were also considered in the differential diagnosis in the
present patient, but these types of infection would not be
associated with palpable purpura and such an extensive distribution
of lesions (7,18). The differential diagnosis for this
condition is difficult, particularly when comparing between small
vessel vasculitides. A biopsy is therefore essential to achieve a
definitive diagnosis (18).
In the present case, in the month prior to
hospitalization, a left fifth finger amputation was performed. The
resulting wound persisted, with the development of osteitis at the
level of the left fifth metatarsal against the background of the
unhealed ulcer and superinfection with MRSA, which triggered the
deposition of immune complexes in the wall of the small blood
vessels. MRSA infection can play multiple roles. The released S.
aureus phosphatase adheres to the endothelial cells, antibodies
bind to these elements, resulting in immune complexes, and
neutrophils are attracted and activated by these complexes
releasing reactive oxygen species, proteinase-3, myeloperoxidase
and tumour necrosis factor-α (22,24).
Cell wall components of S. aureus are able to stimulate
auto-reactive B cells given the mitogenic effect, and studies have
hypothesized that this bacteria functions as a ‘planted antigen’
and initiates vasculitis (18,22,25).
According to the literature, it is known that MRSA infection is
involved in vasculitis; however, the patient in the present study
was investigated to exclude another source such as systemic
autoimmune disease, HBV or HBC, which may be the base event
triggering the inflammatory process (26). Being a single episode, the presence
of MRSA foot infection followed by the onset of skin rash in the
present case, helped in establishing a cause-effect association,
which was confirmed by the histopathological examination. The
diagnosis was decided upon based on the cutaneous biopsy
results.
A previous study reported the case of a 34-year-old
man with diabetes mellitus, elbow site infection and a positive
result for methicillin-sensitive S. aureus (MSSA) in two of
four blood cultures. The patient presented with a leukocytoclastic
vasculitis rash on the extremities, which was successfully resolved
by the treatment of the infection with a third-generation
cephalosporin. In this case, the patient did not require
corticosteroid therapy (27).
Leukocytoclastic vasculitis was also mentioned in a case of MSSA
infection without bacteraemia in a 61-year-old male with peripheral
vascular disease and unbalanced diabetes mellitus (12). Multiple immune mechanisms are
implicated in infection-associated vasculitides pathogenesis. The
majority are a result of direct invasion and proliferation in the
vascular wall of pathogens, causing inflammation. Diabetes mellitus
is a common metabolic disorder, and chronic hyperglycaemia is
considered the central problem in the occurrence of macro- and
microvascular complications (3,28).
Glucose overload leads to the lack of antioxidant effect, oxidative
stress and the release of reactive oxygen species, and as a result,
the installation of an inflammatory state that contributes to the
occurrence and maintenance of an infectious process (3,19).
The infected plantar ulcer in a patient with diabetes and
neuropathy is considered one of the most feared complications, and
one of the isolated pathogens associated with these cases is S.
aureus (12,29-31).
In the interpretation of the data presented in the
present case report, the limitations of the present study must also
be taken into account. In this regard, there are only a few cases
described in the literature of patients with diabetes and
leukocytoclastic vasculitis. Furthermore, some of the laboratory
tests, such as those for ACPA antibodies, circulating immune
complexes and cryoglobulins were not available. The present case is
of note, as the metabolic imbalance led to the installation of
chronic complications and ultimately the worst consequences,
namely, amputation, ulcerations and superinfection with MRSA
followed by development of a consecutive rash. However, after 3
months of follow-up, although the patient received the
recommendation to undergo periodic evaluations to follow the
clinical and paraclinical evolution, contact with this patient was
lost.
Overall, in the present case described, a typical
situation for a patient with unbalanced type 2 diabetes was found,
where the onset of chronic complications in association with the
non-compliance with protection and care measures ultimately led to
ulceration, gangrene and amputation. By carrying out the
amputations, initially the pathological process was limited and a
reasonable functionality at the level of the lower limb was
achieved. At the same time, in the context of an unstable territory
with important metabolic imbalance and response to infections
affected on this background, the benefit of surgical resection was
limited, with ulcer persistence, superinfection with MRSA and an
episode of vasculitis occurrence.
Investigations in the present case revealed a
biological inflammatory syndrome and leukocytosis, metabolic
imbalance, MRSA infection and a histopathological appearance
suggestive of vasculitis. When evaluating a patient with suspected
CLA, it is important to identify the necessary elements to confirm
the diagnosis, detect the underlying aetiology and exclude major
organ involvement. If no specific cause has been identified, it is
essential to include the infection in the differential diagnosis.
Even in the absence of bacteraemia, CLA can be considered a
complication of a local infection. In the current case, the
patient's rash improved with antibiotics and corticosteroids.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study are included
in the figures and/or tables of this article.
Authors' contributions
RMM, MMR, VP, DC, AM, MAG, ERM, DR and IMV
contributed equally to the acquisition, analysis and
systematization of data, manuscript writing and critical revision
for important intellectual content. All authors read and approved
the final version of the manuscript. RMM and DC confirm the
authenticity of all the raw data.
Ethics approval and consent to
participate
Ethical approval was obtained from the ethical
committee of the University of Medicine and Pharmacy of Craiova
(approval no. 162/19.08.2022; Craiova, Romania).
Patient consent for publication
The patient provided written informed consent
regarding the publication of the present study.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Morita TCAB, Trés GFS, Criado RFJ, Sotto
MN and Criado PR: Update on vasculitis: An overview and
dermatological clues for clinical and histopathological
diagnosis-part I. An Bras Dermatol. 95:355–371. 2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Boulton AJ: Diabetic neuropathy and foot
complications. Handb Clin Neurol. 126:97–107. 2014.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Gupta S, Koirala J, Khardori R and
Khardori N: Infections in diabetes mellitus and hyperglycemia.
Infect Dis Clin North Am. 21:617–638. 2007.PubMed/NCBI View Article : Google Scholar
|
|
4
|
International Diabetes Federation: IDF
Diabetes Atlas - 10th Edition. Brussels, Belgium: International
Diabetes Federation, 2021. https://diabetesatlas.org/atlas/tenth-edition/.
Accesed June 15, 2022.
|
|
5
|
Jeon BJ, Choi HJ, Kang JS, Tak MS and Park
ES: Comparison of five systems of classification of diabetic foot
ulcers and predictive factors for amputation. Int Wound J.
14:537–545. 2017.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Bandyk DF: The diabetic foot:
Pathophysiology, evaluation, and treatment. Semin Vasc Surg.
31:43–48. 2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Jennette JC, Falk RJ, Bacon PA, Basu N,
Cid MC, Ferrario F, Flores-Suarez LF, Gross WL, Guillevin L, Hagen
EC, et al: 2012 revised international Chapel Hill consensus
conference nomenclature of vasculitides. Arthritis Rheum. 65:1–11.
2013.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Moldovan HR, Ionovici N, Nechita F,
Horváth E, Ianoşi ES, Papp EG, Popoviciu HV, Jimborean G, Moldovan
G, Vlasiu MA and Szasz S: A rare association of cutaneous
leukocytoclastic angiitis (hypersensitivity vasculitis) and
hypersensitivity pneumonia (extrinsic allergic alveolitis) in a
pigeon breeder-case report and literature review. Rom J Morphol
Embryol. 60:325–331. 2019.PubMed/NCBI
|
|
9
|
Katsuyama T, Sada KE and Makino H: Current
concept and epidemiology of systemic vasculitides. Allergol Int.
63:505–513. 2014.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Mohan N and Kerr G: Infectious etiology of
vasculitis: Diagnosis and management. Curr Rheumatol Rep.
5:136–141. 2003.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Martins-Martinho J, Dourado E, Khmelinskii
N, Espinosa P and Ponte C: Localized forms of vasculitis. Curr
Rheumatol Rep. 23(49)2021.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Lokineni S, Mohamed A, Gandhi RG and
Barrett M: Leukocytoclastic vasculitis as a rare manifestation of
staphylococcal osteomyelitis. Cureus. 13(e15685)2021.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Ferranti M, Cama E, Cacciavillani M,
Schiavon F, Felicetti M, Briani C and Alaibac M: Leukocytoclastic
vasculitis associated with multifocal sensory neuropathy responsive
to intravenous immunoglobulins: A case report. Sarcoidosis Vasc
Diffuse Lung Dis. 38(e2021022)2021.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Belizna CC, Hamidou MA, Levesque H,
Guillevin L and Shoenfeld Y: Infection and vasculitis. Rheumatology
(Oxford). 48:475–482. 2009.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Chen KR and Carlson JA: Clinical approach
to cutaneous vasculitis. Am J Clin Dermatol. 9:71–92.
2008.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Ishiguro N: Skin manifestations of
vasculitis. Brain Nerve. 71:339–344. 2019.PubMed/NCBI View Article : Google Scholar : (In Japanese).
|
|
17
|
Volmer-Thole M and Lobmann R: Neuropathy
and diabetic foot syndrome. Int J Mol Sci. 10(917)2016.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Frumholtz L, Laurent-Roussel S, Lipsker D
and Terrier B: Cutaneous vasculitis: Review on diagnosis and
clinicopathologic correlations. Clin Rev Allergy Immunol.
61:181–193. 2021.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Polk C, Sampson MM, Roshdy D and Davidson
LE: Skin and soft tissue infections in patients with diabetes
mellitus. Infect Dis Clin North Am. 35:183–197. 2021.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Vitale M, Orsi E, Solini A, Garofolo M,
Resi V, Bonora E, Fondelli C, Trevisan R, Vedovato M, Penno G and
Pugliese G: Independent association of history of diabetic foot
with all-cause mortality in patients with type 2 diabetes: The
renal insufficiency and cardiovascular events (RIACE) Italian
multicenter study. Cardiovasc Diabetol. 23(34)2024.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Ionac S, Rogers SK, Bondor CI, Bowling FL,
Dragoi II and Ionac M: Lower extremity amputation and peripheral
revascularisation rates in Romania and their relationship with
comorbidities and vascular care. J Clin Med. 13(52)2023.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Alberti-Violetti S, Berti E and Marzano
AV: Cutaneous and systemic vasculitides in dermatology: A
histological perspective. G Ital Dermatol Venereol. 153:185–193.
2018.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Calabrese LH, Michel BA, Bloch DA, Arend
WP, Edworthy SM, Fauci AS, Fries JF, Hunder GG, Leavitt RY and Lie
JT: The American college of rheumatology 1990 criteria for the
classification of hypersensitivity vasculitis. Arthritis Rheum.
33:1108–1113. 1990.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Furuya K and Itoh N: Hospital-onset IgA
vasculitis triggered by infectious endocarditis. IDCases.
33(e01865)2023.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Zito A, De Pascalis A, Montinaro V, Ria P,
Carbonara MC, Ferramosca E and Napoli M: Successful treatment of
infectious endocarditis-associated glomerulonephritis during active
hepatitis C infection: A case report. BMC Nephrol.
23(390)2022.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Richard JL, Sotto A, Jourdan N, Combescure
C, Vannereau D, Rodier M and Lavigne JP: Nîmes University Hospital
Working Group on the Diabetic Foot (GP30). Risk factors and healing
impact of multidrug-resistant bacteria in diabetic foot ulcers.
Diabetes Metab. 34:363–369. 2008.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Mosher CA, Owen JL and Barker BR:
Staphylococcus aureus Bacteremia Masquerading as Leukocytoclastic
Vasculitis. Am J Med. 129:e5–e7. 2016.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Lima AL, Illing T, Schliemann S and Elsner
P: Cutaneous manifestations of diabetes mellitus: A review. Am J
Clin Dermatol. 18:541–553. 2017.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Sharma VK, Khadka PB, Joshi A and Sharma
R: Common pathogens isolated in diabetic foot infection in Bir
Hospital. Kathmandu Univ Med J (KUMJ). 4:295–301. 2006.PubMed/NCBI
|
|
30
|
Macdonald KE, Boeckh S, Stacey HJ and
Jones JD: The microbiology of diabetic foot infections: A
meta-analysis. BMC Infect Dis. 21(770)2021.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Knapp S: Diabetes and infection: Is there
a link?-A mini-review. Gerontology. 9:99–104. 2013.PubMed/NCBI View Article : Google Scholar
|