Introduction
Preeclampsia (PE) is a pregnancy complication
characterized by new-onset hypertension in pregnant patients
following 20 weeks of gestation (1,2). PE
occurs in 2-8% of all pregnancies worldwide, making it one of the
leading causes of high maternal and neonatal morbidity and
mortality (2). Despite extensive
research (3-6),
the etiology and pathogenesis of PE are incompletely understood.
Placental dysfunction, particularly abnormal invasion and
remodeling of maternal uterine spiral arteries by invasive
extravillous trophoblast cells, is widely accepted to serve a
critical role in the early stages of PE (7,8).
Shallow invasion of trophoblasts into the endometrium and placental
vascular recasting barriers leads to an insufficient oxygen supply,
further leading to placental tissue ischemia, hypoxia and
endothelial cell damage (7).
Additionally, other potential molecular mechanisms such as
oxidative stress, endoplasmic stress, inflammation and genetic
factors have also been suggested (8). However, the exact molecular
mechanisms underlying the role of trophoblast cells in PE
progression are unclear.
Long non-coding RNAs (lncRNAs) are a class of RNA
transcripts with no protein-coding capacity and >200 nucleotides
in length (9,10). Studies have reported that lncRNAs
can regulate gene expression and function at the epigenetic,
transcriptional and post-transcriptional levels, while
participating in numerous biological processes, such as cell
differentiation, proliferation, apoptosis and invasion (11-14).
lncRNAs have been implicated in numerous diseases such as cancer,
diabetes, and neurodegenerative and cardiovascular diseases
(13,15-18).
Aberrantly expressed lncRNAs such as MALAT1, SNX17, SNHG14 and
pseudogene HK2P1 have been identified in PE, and are involved in
the pathogenesis of PE, potentially via regulation of angiogenesis,
decidualization, inflammation and immune regulation, proliferation,
migration, and invasion of trophoblast cells (19-23).
However, to the best of our knowledge, there is a lack of detailed
studies on lncRNAs in PE and the functions and mechanisms of these
lncRNAs.
MIR193BHG is located on human chromosome 16p13.12. A
previous study reported that MIR193BHG expression is upregulated in
placental tissue from patients with PE (24). Furthermore, hypoxia induces the
expression of MIR193BHG in breast cancer cell lines (25). Overexpression of MIR193BHG inhibits
breast cancer cell invasion and metastasis under both hypoxic and
normoxic conditions; conversely, inhibition of MIR193BHG promotes
cancer cell invasion and metastasis (25). Nevertheless, the effect of
MIR193BHG on trophoblast cells in PE remains unclear. Therefore,
the aim of the present study was to investigate the potential
effects and underlying mechanisms of MIR193BHG on HTR-8/SVneo
cells.
Materials and methods
Cell culture
The human first trimester extravillous trophoblast
cell line (HTR-8/SVneo) was purchased from Wuhan Boster Biological
Technology, Ltd. Cells were cultured in RPMI-1640 medium (HyClone;
Cytiva) supplemented with 10% FBS (Zhejiang Tianhang Biotechnology
Co., Ltd.). Cells were cultured at 37˚C in an atmosphere containing
5% CO2 and used in further experiments during the
logarithmic growth phase.
Cell transfection
MIR193BHG Smart Silencer and overexpression plasmids
used to knock down (MIR193BHG-KD) and overexpress (MIR193BHG-OE)
MIR193BHG as well as their negative controls (NCs) were synthesized
by Guangzhou RiboBio Co., Ltd. The plasmid backbone was
pEXP-RB-Mam-EGFP. The empty pEXP-RB-Mam-EGFP vector was used as the
NC for overexpression (NC-OE). Smart Silencer is a mixture of three
small interfering RNAs (siRNAs) and three antisense
oligonucleotides (ASOs). The target sequences of the siRNAs were as
follows: 5'-AAACCTGCCAGTAATTTCAG-3', 5'-ATAAAAGGAGGCCTGCTTGG-3' and
5'-GCAAAGATGTTTCCAGAGA-3'. The target sequences of the ASOs were as
follows: 5'-GAGCGTGTATAAAACCAAA-3', 5'-GCAACATGTTATTCTGAGTG-3' and
5'-GTGCTTCTGAACATTTGTT-3'. The Smart Silencer NC (NC-KD) did not
contain domains homologous to humans, mice or rats (cat. no.
lnc3N0000001-1-5). Three siRNAs specific for p53, as well as a
non-targeting universal NC (NC-siRNA; cat. no. siN0000001-1-5),
were synthesized by Guangzhou RiboBio Co., Ltd. The corresponding
target sequences of p53 siRNA were as follows: p53-siRNA-1,
5'-GTACCACCATCCACTACAA-3'; p53-siRNA-2, 5'-AGAGAATCTCCGCAAGAAA-3'
and p53-siRNA-3, 5'-GGAGTATTTGGATGACAG-3'. Overexpression plasmids
(2.5 µg), Smart Silencer (50 nM), siRNA (50 nM) or their respective
NCs (2.5 µg, 50 and 50 nM, respectively) were transfected into
HTR-8/SVneo cells using Lipofectamine® 3000 reagent
(Invitrogen; Thermo Fisher Scientific, Inc.) in Opti-MEM Reduced
Serum (Gibco; Thermo Fisher Scientific, Inc.) at 37˚C with 5%
CO2 for 48 h according to the manufacturer's
instructions. At 48 h after transfection, the cells were collected
for subsequent experiments.
RNA extraction and reverse
transcription-quantitative PCR (RT-qPCR)
Total RNA was extracted from HTR-8/SVneo cells using
TRIzol® (Invitrogen; Thermo Fisher Scientific, Inc.)
according to the manufacturer's protocol. The RNA was
reverse-transcribed to cDNA using the ReverTra Ace qPCR RT kit
(Toyobo Life Science) under the following conditions: 37˚C for 15
min, 50˚C for 5 min and 98˚C for 5 min. qPCR was performed using
the SYBR Green Realtime PCR Master Mix (Toyobo Life Science) and an
Applied Biosystems StepOne Real Time PCR system (Applied
Biosystems; Thermo Fisher Scientific, Inc.). The following
thermocycling conditions were used: Initial denaturation at 95˚C
for 60 sec, followed by 40 cycles of 95˚C for 15 sec, 60˚C for 15
sec and 72˚C for 45 sec. The primers are listed in Table I. mRNA levels were quantified for
each target gene using the 2-ΔΔCq method (26), and normalized to GAPDH as an
internal control.
 | Table IPrimer sequences for reverse
transcription-quantitative PCR. |
Table I
Primer sequences for reverse
transcription-quantitative PCR.
| Gene | Sequence
(5'-3') |
|---|
| MIR193BHG | F:
AGGGGCTGATGAATTGAGGG |
| | R:
TCAATGGCAGCAGGAGGTTA |
| p53 | F:
GAGGTTGGCTCTGACTGTACC |
| | R:
TCCGTCCCAGTAGATTACCAC |
| U6 | F:
CTCGCTTCGGCAGCACA |
| | R:
AACGCTTCACGAATTTGCGT |
| GAPDH | F:
AGAACGGGAAGCTTGTCATC |
| | R:
CATCGCCCCACTTGATTTTG |
Fluorescent in situ hybridization
(FISH)
FITC-labeled RNA probes for MIR193BHG were designed
and synthesized by Shanghai QuiCell Biotechnology Co., Ltd. The
MIR193BHG probe sequence was 5'-TCCAGCCGCAGCTCAATAAA-3'-FITC. FISH
assays were performed using a Fluorescence In Situ
Hybridization Kit for RNA (cat. no. R0306S; Beyotime Institute of
Biotechnology). HTR-8/SVneo cells (2.5x104 cells/well)
were seeded into a 24-well plate (Corning, Inc.) with sterile
slides overnight. Cells were washed using 1X PBS for 5 min and
fixed in 4% formaldehyde at room temperature for 10 min. Next,
cells were washed twice with 1X PBS for 5 min each, after which
cells were permeabilized in 1X PBS containing 0.5% Triton X-100
(Beyotime Institute of Biotechnology) for 5 min at 4˚C. After
washing the cells with 1X PBS three times, they were blocked in
pre-hybridization solution (hybridization solution containing 1%
yeast RNA; cat. no. R0306S; Beyotime Institute of Biotechnology) at
37˚C for 30 min. Pre-hybridization solution was then discarded and
cells were incubated with a hybridized mixture containing probe (1
µg/ml) at 37˚C overnight. Cells were sequentially washed in the
dark at 42˚C for 5 min each with Washing Buffer I, II and III (cat.
no. R0306S; Beyotime Institute of Biotechnology). After washing
cells in 1X PBS for 5 min, they were stained with DAPI at room
temperature for 5 min and washed with 1X PBS. The slides taken from
the 24-well plate were then observed using a laser confocal
microscope (magnification, x200). The entire process was protected
against light to prevent quenching.
Nuclear and cytoplasmic RNA
fractionation
A Cytoplasmic and Nuclear RNA Purification kit
(Norgen Biotek Corp.) was used to isolate and purify both
cytoplasmic and nuclear RNA from HTR-8/SVneo cells according to the
manufacturer's instructions. RNA was subjected to RT-qPCR analysis
as aforementioned. U6 is a type of small nuclear RNA and is stably
expressed in the nucleus (27). U6
RNA was used as the nuclear control and GAPDH mRNA as the
cytoplasmic control. The relative expression (%) of nuclear
RNA=2^-nuclear Ct/(2^-cytoplasmic Ct + 2^-nuclear Ct). The relative
expression (%) of cytoplasmic RNA=1-nuclear RNA (%).
Cell Counting Kit-8 (CCK-8)
assays
Cell proliferation was detected using a CCK-8 assay
kit (Dojindo Laboratories, Inc.) according to the manufacturer's
instructions. Briefly, transfected HTR-8/SVneo cells were seeded
into 96-well plates (5x103/well) with three replicates
per group. At 0, 24, 48 and 72 h post-transfection, 10 µl CCK-8
working solution was dripped into each well and the cells were
cultured for another 1 h at 37˚C. A microplate reader (Thermo
Fisher Scientific, Inc.) was used to measure the optical density
values at 450 nm.
Wound healing assay
At 24 h after transfection, HTR-8/SVneo cells were
inoculated into a six-well plate and grown to 90-100% confluence. A
200-µl pipette tip was used to vertically scratch the bottom of the
6-well plate. The scratched cells were washed with PBS and
incubated at 37˚C for 24 and 48 h in RPMI-1640 medium containing 2%
FBS. Images were captured under an inverted light microscope.
ImageJ software (version 1.51; National Institutes of Health) was
used to quantify the wound closure. The percentage of wound closure
was calculated as follows: Migration area (%)=(initial wound
area-remaining wound area)/initial wound area x100.
Transwell assay
At 24 h post-transfection, cells were resuspended
and adjusted to 2.5x104 cells/ml using serum-free
RPMI-1640 medium. Subsequently, 200 µl cell suspension was added
into the upper chamber (8 µm; cat. no. 3422; Corning, Inc.), which
was pre-coated with Matrigel (Corning, Inc.) at 37˚C for 1 h, and
500 µl RPMI-1640 medium containing 20% FBS was added into the lower
chamber. Cells were incubated at 37˚C for 24 h. The invaded cells
were fixed using 4% paraformaldehyde for 20 min at room temperature
and stained with 0.1% crystal violet for 15 min at room
temperature. The invaded cells were observed and counted manually
in five random fields under a light microscope (Olympus
Corporation).
Flow cytometry
Cell apoptosis was assessed using an Annexin
V-FITC/PI kit (BD Biosciences). At 48 h post-transfection,
HTR-8/SVneo cells were collected and resuspended with 500 µl
precooled 1X binding buffer (including in the Annexin V-FITC/PI
Apoptosis Detection Kit; BD Biosciences). Next, cells were
double-stained in the dark with 5 µl Annexin V-FITC and PI (BD
Biosciences) for 15 min at room temperature. Apoptosis including
early apoptotic cells and late apoptotic cells, was determined
within 1 h of staining using the DxFLEX flow cytometer (Beckman
Coulter, Inc.) and analyzed using FlowJo software version 10.4
(FlowJo LLC).
Western blotting
Total proteins were extracted from HTR-8/SVneo cells
using RIPA buffer (CoWin Biosciences) supplemented with 1%
phenylmethanesulfonyl fluoride. The lysate was centrifuged at
10,000 x g for 10 min at 4˚C and the supernatant was collected. The
total protein concentration was measured using the BCA protein
assay kit (Thermo Fisher Scientific, Inc.). Equal amounts of
protein (50 µg/lane) were separated by 10% SDS-PAGE and transferred
to PVDF membranes. Following blocking with 5% non-fat milk in 1X
Tris-buffered saline containing 0.1% Tween-20 (TBST) for 2 h at
room temperature, membranes were incubated overnight with primary
antibodies against p53 (1:2,000; cat. no. ab179477; Abcam) and
GAPDH (1:4,000; cat. no. ab9485; Abcam) at 4˚C. Next, membranes
were washed three times with TBST and incubated with horseradish
peroxidase-conjugated goat anti-rabbit secondary antibody
(1:10,000; cat. no. IH-0012; Beijing Dingguo Changsheng
Biotechnology Co., Ltd.) for 1 h at room temperature. The bands
were visualized using an ECL Plus Chemiluminescence Reagent kit
(Beyotime Institute of Biotechnology) on an AI600 imaging system
(Cytiva) to quantify their intensity. ImageJ software (version
1.51; National Institutes of Health) was used to quantify the
relative protein expression. GAPDH was used as the loading
control.
RNA-sequencing (RNA-seq) and
computational analysis
HTR-8/SVneo cells transfected with MIR193BHG
overexpression and empty plasmids were collected and lysed with
TRIzol (CoWin Biosciences). The RNA concentration and quality were
determined using a Qubit® 3.0 Fluorometer (Thermo Fisher
Scientific, Inc.) and the Nanodrop One spectrophotometer (Thermo
Fisher Scientific, Inc.). The integrity of total RNA was assessed
using the Agilent 2100 Bioanalyzer (Agilent Technologies, Inc.),
and samples with RNA integrity number values >7.0 were used for
sequencing. Paired-end libraries were synthesized using the
Stranded mRNA-seq Lib Prep Kit for Illumina (ABclonal Biotech Co.,
Ltd.) in accordance with the manufacturer's protocols. Briefly, the
poly-A containing mRNA molecules were purified using poly-T
oligo-attached magnetic beads. Following purification, the mRNA was
fragmented into small pieces using divalent cations at 94˚C for 10
min. The cleaved RNA fragments were copied into first strand cDNA
using reverse transcriptase and random primers. This was followed
by second strand cDNA synthesis using DNA Polymerase I and RNase H.
These cDNA fragments then underwent an end repair process, the
addition of a single ‘A’ base and ligation of the adapters. The
products were amplified by PCR and purified with magnetic beads
(cat. no. RK20257; ABclonal Biotech Co., Ltd.) to create the final
cDNA library according to the manufacturer's protocols of the
mRNA-seq Lib Prep Kit for Illumina. Purified libraries were
quantified using a Qubit® 3.0 Fluorometer (Thermo Fisher
Scientific, Inc.) and validated using an Agilent 2100 bioanalyzer
(Agilent Technologies, Inc.). The cluster was generated by cBot
with the library diluted to 10 pM and then paired-end 150-bp
sequencing was performed on the Illumina NovaSeq 6000 sequencing
system (Illumina, Inc.) using the NovaSeq 6000 S4 Reagent kit v1.5
(cat. no. 20028312; Illumina, Inc.). cDNA library construction and
RNA sequencing were performed by Sinotech Genomics. The sequenced
reads were then aligned to the human reference genome (GRCh38.91)
using HISAT2 (version 2.1.0; https://daehwankimlab.github.io/hisat2/). Differential
gene expression analysis was performed using EdgeR (version 3.30.3;
Bioconductor). The threshold used to screen upregulated or
downregulated mRNAs was |log2(fold change)|>0.5 and
P<0.05.
Kyoto Encyclopedia of Genes and
Genomes (KEGG) enrichment analysis
After obtaining the differentially expressed genes,
clusterProfiler (version 4.0.5; https://bioconductor.org/packages/clusterProfiler/)
was used to perform the KEGG enrichment analysis in R software
(version 4.1.0; https://www.r-project.org/). KEGG pathway enrichment
(https://www.kegg.jp/kegg/kegg1.html)
was used to identify the critical pathways that were closely
related to MIR193BHG overexpression. P<0.05 was used to identify
significant KEGG pathways. There was no gene count threshold.
Construction of the protein-protein
interaction (PPI) network of differentially expressed genes
A PPI network of differentially expressed genes was
constructed using the Search Tool for the Retrieval of Interacting
Genes database (version 11.5; https://string-db.org/). Cytoscape (version 3.8.2;
https://cytoscape.org/) was used to display the
PPI network. Interactions with a combined score >0.4 were
considered significant. The gene with the highest degree of
connection to others was considered to be the hub gene, the
associations of which were illustrated in Cytoscape.
Rescue assay
Rescue assays were performed to assess whether
MIR193BHG affected viability, apoptosis, migration and invasion of
HTR-8/SVneo cells by upregulating the expression of p53.
HTR-8/SVneo cells were divided into three groups: MIR193BHG
overexpression plasmid and p53-siRNA group, MIR193BHG
overexpression plasmid and siRNA NC group, and empty plasmid and
siRNA NC group.
Statistical analysis
Data from three independent replicates are presented
as the mean ± standard deviation. SPSS 21.0 (IBM Corp.) was used to
perform statistical analysis. Two group comparisons were conducted
using the unpaired two-tailed Student's t-test and multiple group
comparisons were performed using one-way ANOVA followed by
Bonferroni's post hoc test. P<0.05 was considered to indicate a
statistically significant difference.
Results
MIR193BHG inhibits proliferation and
promotes apoptosis of trophoblast cells
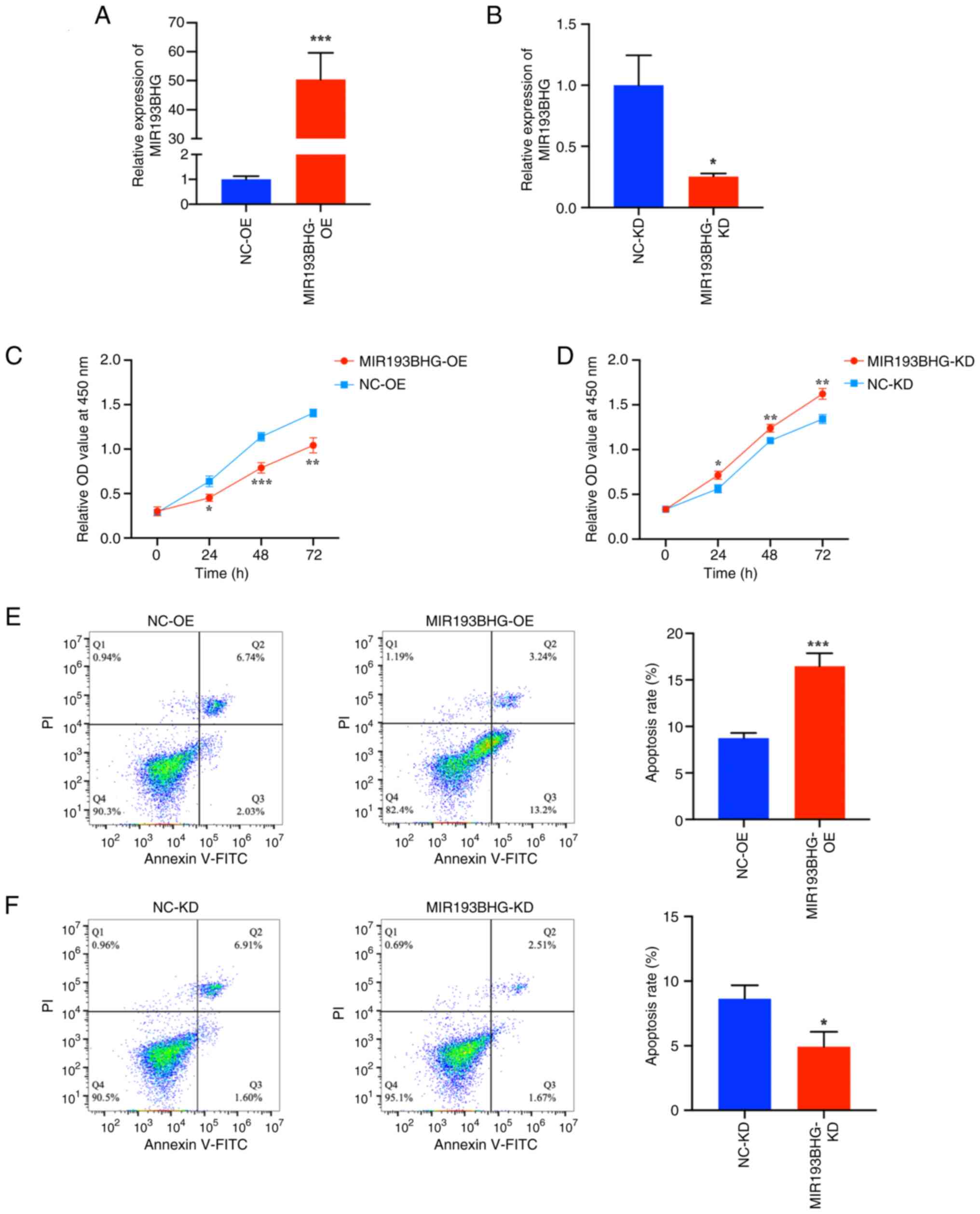
HTR-8/SVneo cells were transfected with MIR193BHG
overexpression or empty plasmids and the transfection efficiency
was confirmed by RT-qPCR. The results indicated that the MIR193BHG
overexpression vector significantly increased the levels of
MIR193BHG compared with the NC (Fig.
1A). The levels of MIR193BHG were significantly decreased in
HTR-8/SVneo cells following transfection with the MIR193BHG Smart
Silencer compared with the control (Fig. 1B).
To assess the role of MIR193BHG in HTR-8/SVneo
cells, a CCK-8 assay was performed. There was a significant
decrease in cell proliferation at 24, 48 and 72 h in the
MIR193BHG-OE group compared with the NC-OE group (Fig. 1C). Conversely, knockdown of
MIR193BHG resulted in a significant increase in cell proliferation
at 24, 48 and 72 h compared with the NC-KD group (Fig. 1D).
Flow cytometry demonstrated that the apoptosis rate
of HTR-8/SVneo cells was significantly higher in the MIR193BHG-OE
group compared with the NC-OE group (Fig. 1E). The apoptosis rate was
significantly lower in the MIR193BHG-KD group compared with the
NC-KD group (Fig. 1F).
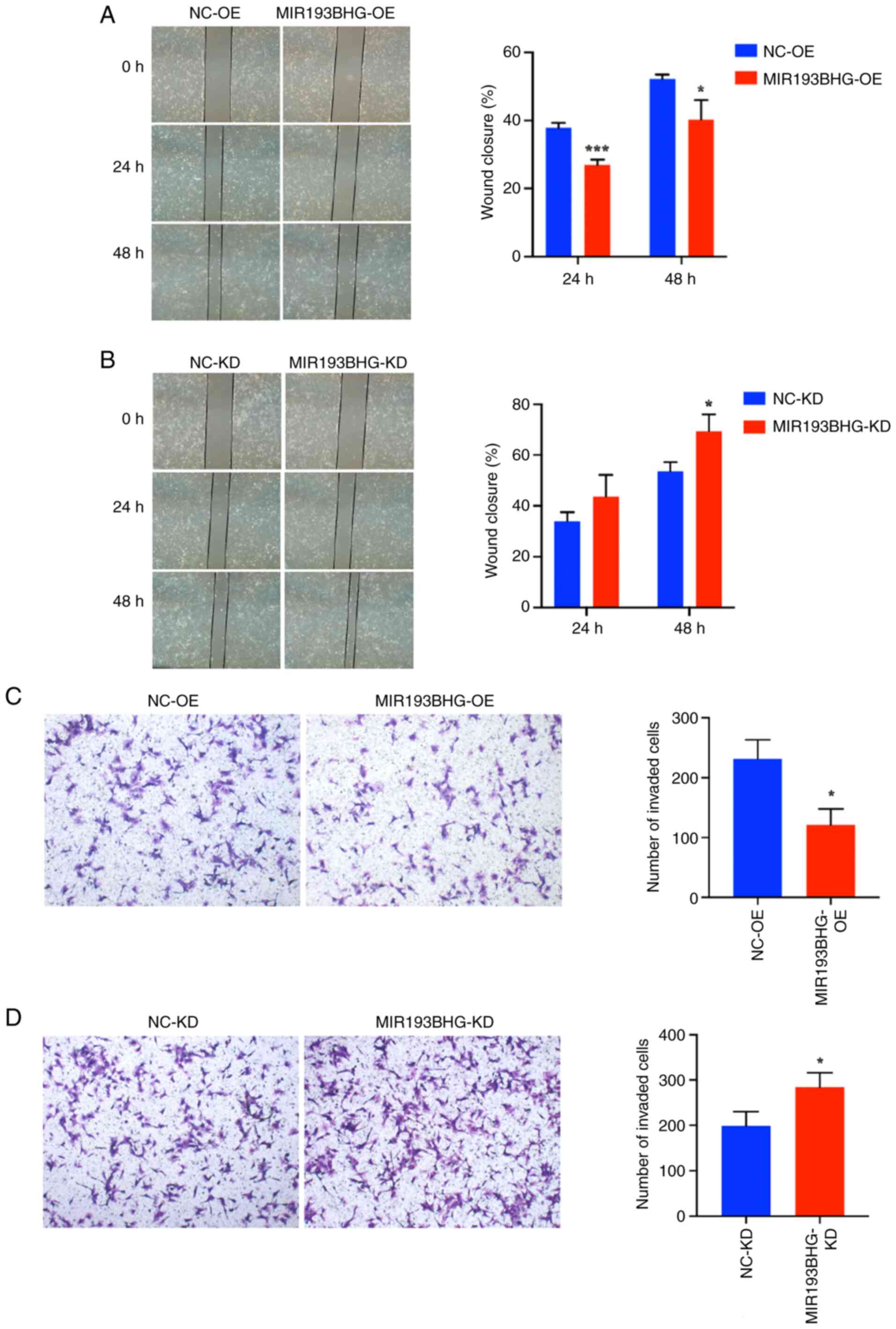
MIR193BHG inhibits trophoblast cell
migration and invasion
The invasion and migration of trophoblasts are key
for placentation, with dysregulation serving a critical role in the
pathogenesis of PE (28,29). Therefore, wound healing and
Transwell assays were performed to assess the migration and
invasion of HTR-8/SVneo cells in response to MIR193BHG
overexpression and knockdown. The percentage of wound closure was
significantly decreased in the MIR193BHG-OE group compared with the
NC-OE group at 24 and 48 h (Fig.
2A), as was the number of invaded cells at 24 h (Fig. 2C). Conversely, the percentage of
wound closure was significantly increased at 48 h in the
MIR193BHG-KD group compared with the NC-KD group (Fig. 2B), as was the number of invaded
cells at 24 h (Fig. 2D).
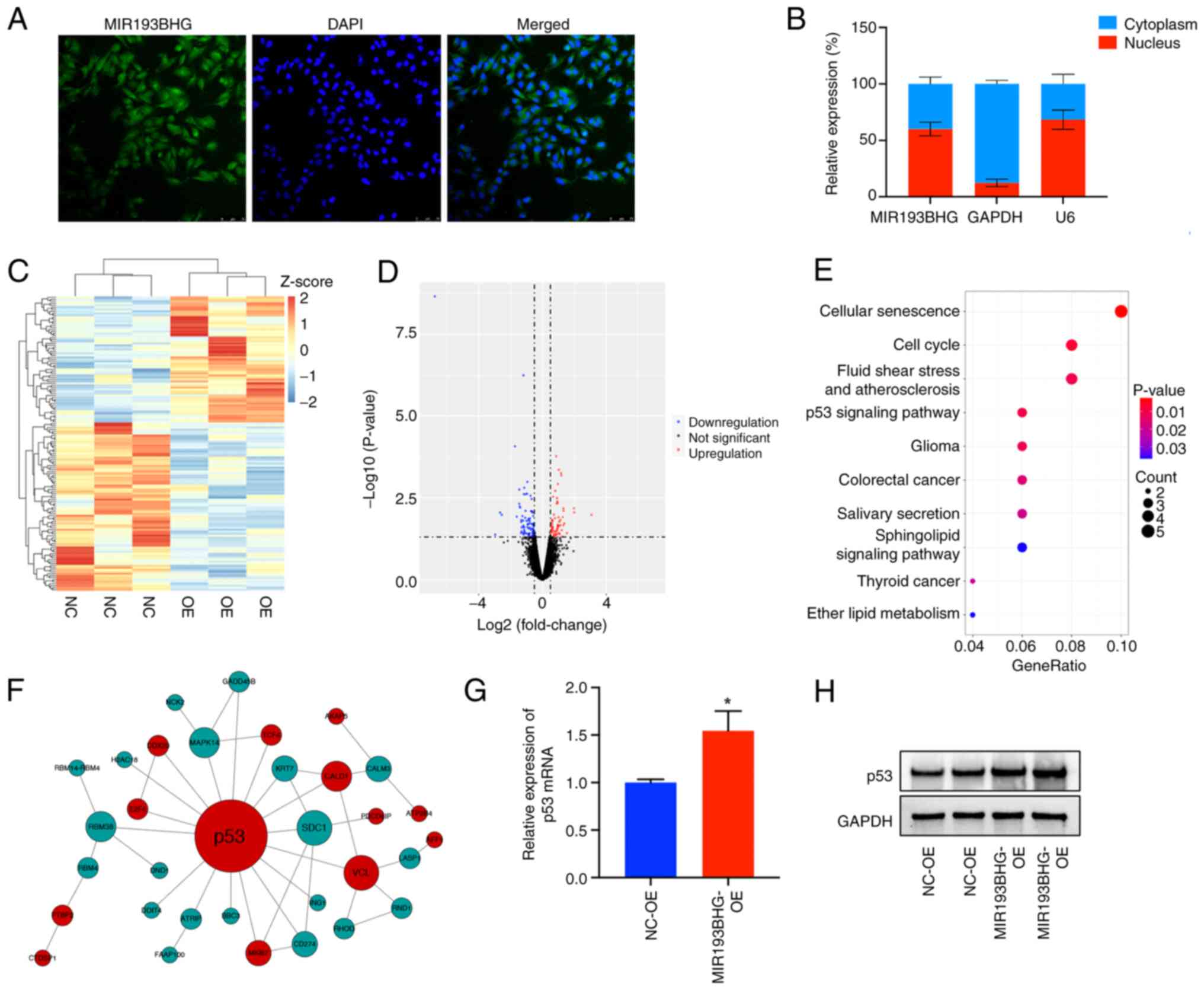
MIR193BHG upregulates p53 expression
in HTR-8/SVneo cells
Using FISH and cell fractionation assays, it was
demonstrated that MIR193BHG was predominantly localized in the
nucleus (~60%) (Fig. 3A and
B), suggesting it may participate
in the transcriptional regulation processes. To assess downstream
molecules and signaling pathways regulated by MIR193BHG, RNA-seq
was conducted in the MIR193BHG overexpression and control groups.
This demonstrated that 63 mRNAs were upregulated and 84 mRNAs were
downregulated in the MIR193BHG overexpression group compared with
the control group (Fig. 3C and
D). KEGG pathway analysis
demonstrated that the differentially expressed genes were primarily
enriched in ‘cellular senescence’, ‘cell cycle’, ‘fluid shear
stress and atherosclerosis’ and ‘p53 signaling pathway’ (Fig. 3E). Notably, p53 emerged as a hub
gene within the PPI network constructed based on differentially
expressed mRNAs (Fig. 3F),
suggesting its potential role as a downstream molecule regulated by
MIR193BHG.
RT-qPCR demonstrated a significant increase in the
levels of p53 mRNA in the MIR193BHG overexpression group compared
with the control group (Fig. 3G).
Western blotting also demonstrated increased protein expression of
p53 in the MIR193BHG overexpression group, which corroborated the
findings of RNA-seq analysis (Fig.
3H).
Silencing of p53 attenuates the
effects of MIR193BHG on HTR-8/SVneo cells
To assess whether MIR193BHG affects viability,
apoptosis, migration and invasion of HTR-8/SVneo cells by
upregulating the expression of p53, rescue assays were performed.
RT-qPCR demonstrated that the mRNA expression levels of p53 in
HTR-8/SVneo cells were significantly downregulated following
transfection with p53-siRNA-1, p53-siRNA-2 and p53-siRNA-3 compared
with NC-siRNA. A decrease in p53 protein levels was also
demonstrated by western blotting following transfection with
p53-siRNA-1, p53-siRNA-2 and p53-siRNA-3. Furthermore, the
knockdown efficiency of p53-siRNA-3 was relatively high, and thus,
p53-siRNA-3 was used for subsequent experiments (Fig. 4A and B).
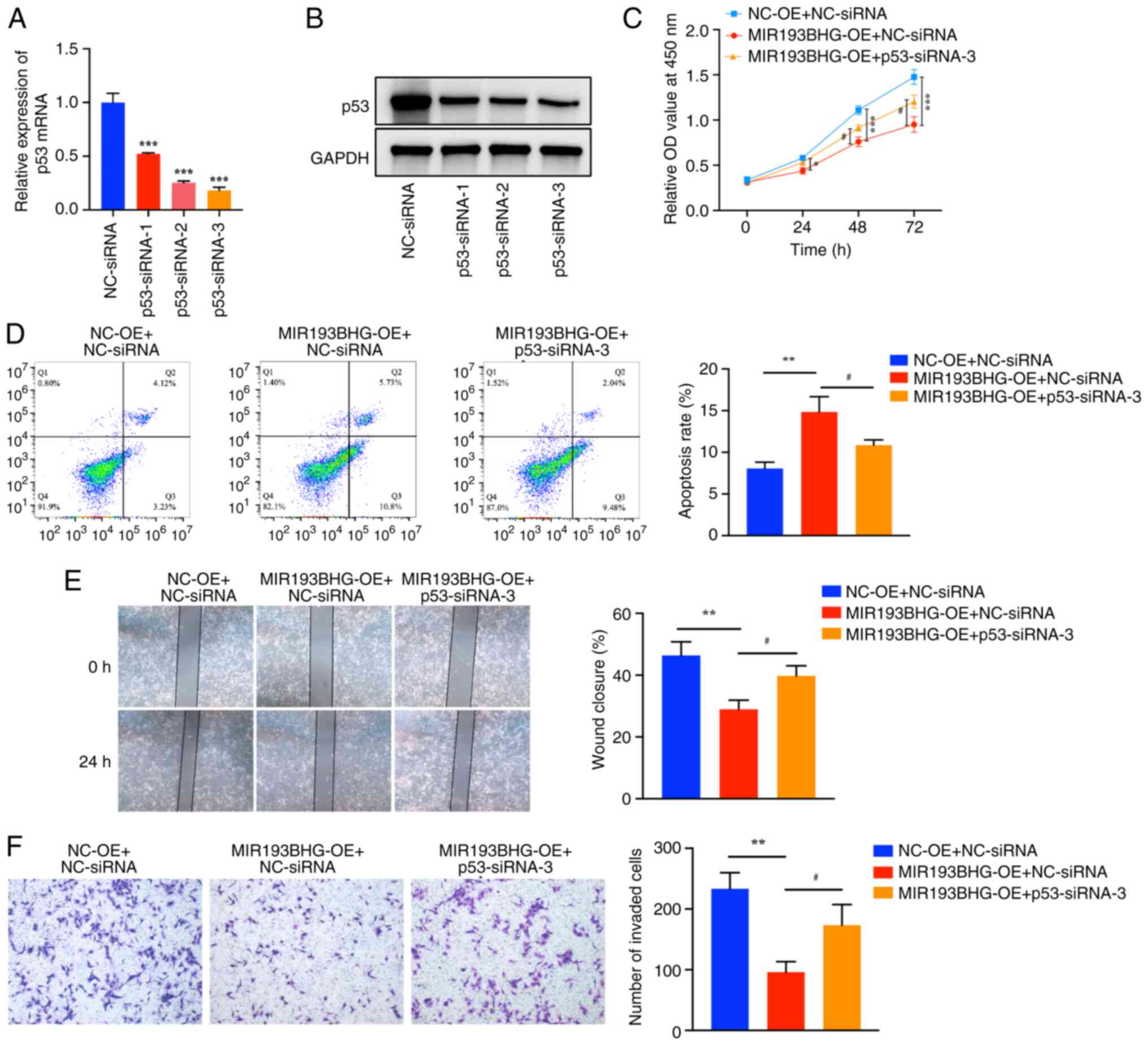 | Figure 4Silencing of p53 attenuates the
effects of MIR193BHG on HTR-8/SVneo cells. HTR-8/SVneo cells were
divided into three groups (MIR193BHG-OE + p53-siRNA-3, MIR193BHG-OE
+ NC-siRNA and NC-OE + NC-siRNA), after which rescue experiments
were performed. (A) Reverse transcription-quantitative PCR and (B)
western blotting were performed to examine mRNA and protein
expression levels of p53, respectively, following transfection with
p53-siRNA-1, p53-siRNA-2 and p53-siRNA-3. ***P<0.001
vs. NC-siRNA. (C) Cell proliferation was measured using a Cell
Counting Kit-8 assay and (D) flow cytometry was used to detect
apoptosis. (E) Wound healing assays were used to examine cell
migration (magnification, x40). (F) A Transwell invasion assay was
used to examine cell invasion (magnification, x100).
*P<0.05, **P<0.01 and
***P<0.001 vs. NC-OE + NC-siRNA;
#P<0.05 vs. MIR193BHG-OE + NC-siRNA. OE,
overexpression; NC, negative control; siRNA, small interfering RNA;
OD, optical density; NC-OE, empty vector; NC-siRNA, non-targeting
siRNA negative control. |
The CCK-8 assay demonstrated that the viability of
HTR-8/SVneo cells was decreased at 24, 48 and 72 h in the MIR193BHG
overexpression group compared with the control group, whereas
co-transfection of MIR193BHG and p53-siRNA-3 partially reduced this
effect (Fig. 4C). Flow cytometry
demonstrated that the apoptosis rate of HTR-8/SVneo cells was
significantly higher in the MIR193BHG overexpression group compared
with the control group, whereas co-transfection of MIR193BHG and
p53-siRNA-3 partially counteracted the apoptotic effects induced by
MIR193BHG in HTR-8/SVneo cells (Fig.
4D). The percentage of wound closure (Fig. 4E) and the number of invaded cells
(Fig. 4F) were significantly
decreased in the MIR193BHG overexpression group compared with the
control group at 24 h, and this repression was partially reversed
by co-transfection of MIR193BHG and p53-siRNA-3.
Discussion
PE is a leading cause of high maternal and neonatal
morbidity and mortality worldwide (2). However, its etiology and pathogenesis
remain unclear, with no effective preventive and therapeutic
measures currently available. lncRNAs are aberrantly expressed in
numerous types of disease, including cancer (30), cardiovascular disease (31), neurodegenerative diseases (32) and diabetic nephropathy (33). Previous studies have highlighted
the association between lncRNAs and trophoblast cell function in PE
(16,34,35).
A previous study reported that MIR193BHG was highly expressed in
the placenta in PE and exhibited robust performance in
differentiating patients with preeclampsia from healthy controls
(24). Furthermore, upregulation
of MIR193BHG has been reported in the serum of patients with PE,
with its expression levels associated with disease severity
(36). Therefore, MIR193BHG is
suggested to serve a role in the development, early diagnosis and
treatment of PE.
In the present study, overexpression of MIR193BHG
significantly inhibited proliferation, invasion and migration,
while increasing the apoptosis rate of HTR-8/SVneo cells.
Conversely, knockdown of MIR193BHG had the opposite effects. Wu
et al (25) reported that
knockdown of MIR193BHG in MDA-MB-231 cells promoted invasion and
viability; however, overexpression of MIR193BHG inhibited invasion.
This finding was consistent with the present study, suggesting that
highly expressed MIR193BHG may be involved in the development of PE
via inhibition of the biological function of trophoblast cells.
The function of lncRNAs primarily relies on their
subcellular localization. Studies have reported that most lncRNAs
are located in the nucleus and participate in gene regulation at
the epigenetic and transcriptional levels, such as chromatin
remodeling, histone modification and DNA methylation, primarily via
interaction with DNA, RNA or proteins (37,38).
Cytoplasmic lncRNAs regulate gene expression mostly at the
post-transcriptional level by regulating mRNA stability, altering
mRNA translation efficiency, serving as microRNA (miRNA) precursors
or competing with miRNA-mediated repression to regulate gene
expression (39,40). The findings of the present study
indicated that MIR193BHG was primarily localized in the nucleus,
suggesting its potential involvement in transcriptional
regulation.
RNA-seq was performed to assess the downstream
targets of MIR193BHG. In vitro experiments demonstrated
upregulation of both mRNA and protein levels of p53 following
overexpression of MIR193BHG. The p53 gene, located on
chromosome 17q13.1, encodes the p53 protein and serves as a key
tumor suppressor gene involved in tumorigenesis (41). Additionally, p53 can regulate a
large number of genes associated with cellular senescence, cell
cycle progression, apoptosis and DNA repair (42). p53 expression is increased in
placental tissues, HUVECs and maternal serum in PE (43,44).
Elevated levels of p53 in trophoblasts have been reported to
inhibit cell viability, proliferation, migration and invasion,
while increasing the levels of apoptosis (45). This leads to cell cycle arrest and
contributes to the development of PE (46). Gao et al (44) reported that p53 expression in
HUVECs isolated and cultured from PE pregnancies was increased,
along with suppressed cell proliferation characterized by increased
G1 arrest and apoptosis. Furthermore, decreased
expression of protein disulfide isomerase family A member 3 (PDIA3)
induces trophoblast apoptosis and suppresses trophoblast
proliferation by regulating the mouse double minute 2
(MDM2)/p53/p21 signaling pathway in PE (47). Additionally, downregulation of
lumican in the placental tissues of PE may also be involved in PE
via inhibition of Bcl-2 expression as well as promotion of p53
expression (48).
Furthermore, lncRNAs serve roles in the p53
regulatory network, and thus, participate in the occurrence and
development of certain types of diseases (42,49).
In colon cancer, the overexpression of lncRNA RNA component of
mitochondrial RNA-processing endoribonuclease inhibits p53 activity
by promoting MDM2-induced p53 ubiquitination and degradation,
thereby promoting proliferation of colorectal cancer cells in a
p53-dependent manner (50). In
renal cell carcinoma, the lncRNA activated by TGF-β inhibits the
expression of p53 by binding to DNA methyltransferase 1, which
leads to an increase in proliferation and migration of renal
carcinoma cells as well as a decrease in apoptosis (51). Additionally, lncRNA growth arrest
specific 5 acts as a negative regulator of vascular smooth muscle
survival in vascular remodeling by directly binding to p53 and
p300, stabilizing p53-p300 interaction and regulating vascular
smooth muscle cell survival by inducing p53 downstream target genes
(52). Knockdown of lncRNA
maternally expressed gene 3 protects the heart against apoptosis
induced by endoplasmic reticulum stress by targeting p53(53).
To the best of our knowledge, no studies have
elucidated the involvement of lncRNAs in the pathogenesis of PE by
regulating p53. Furthermore, to the best of our knowledge, no
research has been conducted on regulation of p53 expression
mediated by MIR193BHG. By constructing a PPI network of
differentially expressed mRNAs identified using RNA-seq, the
present study identified p53 as the hub gene. The KEGG pathway
enrichment analysis also demonstrated that the significantly
enriched pathways of the differentially expressed genes included
‘cellular senescence’, ‘cell cycle’, ‘fluid shear stress and
atherosclerosis’ and the ‘p53 signaling pathway’. These pathways
have all been confirmed to be related to p53 (54,55).
Therefore, it was suggested that p53 may serve as a downstream
molecule of MIR193BHG. Subsequently, expression levels of p53 mRNA
and protein were verified by RT-qPCR and western blotting following
overexpression of MIR193BHG, which yielded consistent results with
those obtained by RNA-seq. Notably, overexpression of MIR193BHG led
to an increase in p53 mRNA and protein expression. Given that p53
is hypothesized to serve a role in the pathogenesis of PE,
evaluating whether MIR193BHG can affect behavior of trophoblasts by
regulating the expression of p53 could enhance the understanding of
the underlying mechanisms involved in PE. Consequently,
co-transfection experiments involving both p53-siRNA and MIR193BHG
overexpression plasmids were conducted using HTR-8/SVneo cells to
evaluate whether knockdown of p53 could reverse changes in
trophoblast behavior caused by MIR193BHG overexpression. Knockdown
of p53 partially restored the function of trophoblasts via
modulation of p53 expression, thus highlighting the involvement of
p53 in the pathogenic mechanism underlying PE.
The present study preliminarily assessed the
pathogenesis of MIR193BHG in PE and provided an experimental basis
for subsequent research. However, certain limitations exist within
the present study. Firstly, it is necessary to confirm expression
patterns and localization of p53 in vivo through
immunohistochemistry using clinical specimens to validate these
conclusions. Additionally, the precise mechanism by which MIR193BHG
regulates the expression of p53 remains unknown. Subsequent
experiments should use chromatin immunoprecipitation assays, RNA
pulldown, RNA immunoprecipitation assays and other techniques to
assess whether MIR193BHG directly binds to p53 or indirectly
regulates p53 expression through alternative mechanisms.
Finally, the present study primarily focused on
evaluating the impact of MIR193BHG on the biological functions of
extravillous trophoblastic cells. However, as the placenta contains
stromal components such as fibroblasts and vascular endothelial
cells, as well as blood components, including inflammatory cells
such as lymphocytes and macrophages, the effects of MIR193BHG on
other types of placental cells should be explored in future
studies. These investigations may provide additional insights into
the involvement of MIR193BHG in the pathogenesis of PE.
To the best of our knowledge, the present study was
the first to demonstrate that lncRNA MIR193BHG upregulated p53
expression and influenced the biological behaviors of HTR-8/SVneo
cells in PE. The findings of the present study may provide a novel
diagnostic and therapeutic target for PE.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by the Medical Science
and Technology Research Plan of Henan Province in 2019 (grant no.
LHGJ20190356).
Availability of data and materials
The data generated in the present study may be found
in the Gene Expression Omnibus database under accession number
GSE245279 or at the following URL: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE245279.
The other data generated in the present study may be requested from
the corresponding author.
Authors' contributions
HL and ZZ designed the study. PW, YC and SY
performed experiments and conducted the data analysis. JG performed
bioinformatics analysis. PW wrote the manuscript and JG revised the
manuscript critically for important intellectual content. PW, HL,
ZZ and JG confirm the authenticity of all the raw data. All authors
have read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Chappell LC, Cluver CA, Kingdom J and Tong
S: Pre-eclampsia. Lancet. 398:341–354. 2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
No authors listed. Gestational
hypertension and preeclampsia: ACOG practice bulletin summary,
number 222. Obstet Gynecol. 135:1492–1495. 2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Jung E, Romero R, Yeo L, Gomez-Lopez N,
Chaemsaithong P, Jaovisidha A, Gotsch F and Erez O: The etiology of
preeclampsia. Am J Obstet Gynecol. 226 (2S):S844–S866.
2022.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Cooke WR, Jiang P, Ji L, Bai J, Jones GD,
Lo YMD, Redman C and Vatish M: Differential 5'-tRNA fragment
expression in circulating preeclampsia syncytiotrophoblast vesicles
drives macrophage inflammation. Hypertension. 81:876–886.
2024.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Wang J, Gao Y, Ren S, Li J, Chen S, Feng
J, He B, Zhou Y and Xuan R: Gut microbiota-derived trimethylamine
N-Oxide: A novel target for the treatment of preeclampsia. Gut
Microbes. 16(2311888)2024.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Li Y, Sang Y, Chang Y, Xu C, Lin Y, Zhang
Y, Chiu PCN, Yeung WSB, Zhou H, Dong N, et al: A galectin-9-driven
CD11chigh decidual macrophage subset suppresses uterine
vascular remodeling in preeclampsia. Circulation. 149:1670–1688.
2024.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Jim B and Karumanchi SA: Preeclampsia:
Pathogenesis, prevention, and long-term complications. Semin
Nephrol. 37:386–397. 2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Hod T, Cerdeira AS and Karumanchi SA:
Molecular mechanisms of preeclampsia. Cold Spring Harb Perspect
Med. 5(a023473)2015.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Stein LD: Human genome: End of the
beginning. Nature. 431:915–916. 2004.PubMed/NCBI View
Article : Google Scholar
|
|
10
|
Djebali S, Davis CA, Merkel A, Dobin A,
Lassmann T, Mortazavi A, Tanzer A, Lagarde J, Lin W, Schlesinger F,
et al: Landscape of transcription in human cells. Nature.
489:101–108. 2012.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Dhaka B, Zimmerli M, Hanhart D, Moser MB,
Guillen-Ramirez H, Mishra S, Esposito R, Polidori T, Widmer M,
García-Pérez R, et al: Functional identification of cis-regulatory
long noncoding RNAs at controlled false discovery rates. Nucleic
Acids Res. 52:2821–2835. 2024.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Sun N, Qin S, Zhang L and Liu S: Roles of
noncoding RNAs in preeclampsia. Reprod Biol Endocrinol.
19(100)2021.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Liu SJ, Dang HX, Lim DA, Feng FY and Maher
CA: Long noncoding RNAs in cancer metastasis. Nat Rev Cancer.
21:446–460. 2021.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Zhang Y, He XY, Qin S, Mo HQ, Li X, Wu F,
Zhang J, Li X, Mao L, Peng YQ, et al: Upregulation of PUM1
expression in preeclampsia impairs trophoblast invasion by
negatively regulating the expression of the lncRNA HOTAIR. Mol
Ther. 28:631–641. 2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Ponting CP, Oliver PL and Reik W:
Evolution and functions of long noncoding RNAs. Cell. 136:629–641.
2009.PubMed/NCBI View Article : Google Scholar
|
|
16
|
McAninch D, Roberts CT and Bianco-Miotto
T: Mechanistic insight into long noncoding RNAs and the placenta.
Int J Mol Sci. 18(1371)2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Statello L, Guo CJ, Chen LL and Huarte M:
Gene regulation by long non-coding RNAs and its biological
functions. Nat Rev Mol Cell Biol. 22:96–118. 2021.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Mirzadeh Azad F, Polignano IL, Proserpio V
and Oliviero S: Long noncoding RNAs in human stemness and
differentiation. Trends Cell Biol. 31:542–555. 2021.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Wu HY, Wang XH, Liu K and Zhang JL: LncRNA
MALAT1 regulates trophoblast cells migration and invasion via
miR-206/IGF-1 axis. Cell Cycle. 19:39–52. 2020.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Guiyu S, Quan N, Ruochen W, Dan W, Bingnan
C, Yuanyua L, Yue B, Feng J, Chong Q and Leilei W: LncRNA-SNX17
promotes HTR-8/SVneo proliferation and invasion through
miR-517a/IGF-1 in the placenta of diabetic macrosomia. Reprod Sci.
29:596–605. 2022.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Zhang Y and Zhang M: lncRNA SNHG14
involved in trophoblast cell proliferation, migration, invasion and
epithelial-mesenchymal transition by targeting miR-330-5p in
preeclampsia. Zygote. 29:108–117. 2021.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Lv H, Tong J, Yang J, Lv S, Li WP, Zhang C
and Chen ZJ: Dysregulated pseudogene HK2P1 may contribute to
preeclampsia as a competing endogenous RNA for hexokinase 2 by
impairing decidualization. Hypertension. 71:648–658.
2018.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Li X, Song Y, Liu F, Liu D, Miao H, Ren J,
Xu J, Ding L, Hu Y, Wang Z, et al: Long non-coding RNA MALAT1
promotes proliferation, angiogenesis, and immunosuppressive
properties of mesenchymal stem cells by inducing VEGF and IDO. J
Cell Biochem. 118:2780–2791. 2017.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Zhang Z, Wang P, Zhang L, Huang C, Gao J,
Li Y and Yang B: Identification of key genes and long noncoding
RNA-associated competing endogenous RNA (ceRNA) networks in
early-onset preeclampsia. Biomed Res Int.
2020(1673486)2020.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Wu X, Niculite CM, Preda MB, Rossi A,
Tebaldi T, Butoi E, White MK, Tudoran OM, Petrusca DN, Jannasch AS,
et al: Regulation of cellular sterol homeostasis by the oxygen
responsive noncoding RNA lincNORS. Nat Commun.
11(4755)2020.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Didychuk AL, Butcher SE and Brow DA: The
life of U6 small nuclear RNA, from cradle to grave. RNA.
24:437–460. 2018.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Sun M, Gao J, Meng T, Liu S, Chen H, Liu
Q, Xing X, Zhao C and Luo Y: Cyclin G2 upregulation impairs
migration, invasion, and network formation through RNF123/Dvl2/JNK
signaling in the trophoblast cell line HTR8/SVneo, a possible role
in preeclampsia. FASEB J. 35(e21169)2021.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Chen Q, Jiang S, Liu H, Gao Y, Yang X, Ren
Z, Gao Y, Xiao L, Hu H, Yu Y, et al: Association of lncRNA
SH3PXD2A-AS1 with preeclampsia and its function in invasion and
migration of placental trophoblast cells. Cell Death Dis.
11(583)2020.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Peng WX, Koirala P and Mo YY:
LncRNA-mediated regulation of cell signaling in cancer. Oncogene.
36:5661–5667. 2017.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Yaghoobi A, Rezaee M, Behnoush AH, Khalaji
A, Mafi A, Houjaghan AK, Masoudkabir F and Pahlavan S: Role of long
noncoding RNAs in pathological cardiac remodeling after myocardial
infarction: An emerging insight into molecular mechanisms and
therapeutic potential. Biomed Pharmacother.
172(116248)2024.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Balusu S, Horré K, Thrupp N, Craessaerts
K, Snellinx A, Serneels L, T'Syen D, Chrysidou I, Arranz AM,
Sierksma A, et al: MEG3 activates necroptosis in human neuron
xenografts modeling Alzheimer's disease. Science. 381:1176–1182.
2023.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Guo J, Zheng W, Liu Y, Zhou M, Shi Y, Lei
M, Zhang C and Liu Z: Long non-coding RNA DLX6-AS1 is the key
mediator of glomerular podocyte injury and albuminuria in diabetic
nephropathy by targeting the miR-346/GSK-3β signaling pathway. Cell
Death Dis. 14(172)2023.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Wang L, Shi L, Zhou B, Hong L, Gong H and
Wu D: METTL3-mediated lncRNA HOXD-AS1 stability regulates
inflammation, and the migration and invasion of trophoblast cells
via the miR-135a/β-TRCP axis. Noncoding RNA Res. 9:12–23.
2023.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Tang X, Cao Y, Wu D, Sun L and Xu Y:
Downregulated DUXAP8 lncRNA impedes trophoblast cell proliferation
and migration by epigenetically upregulating TFPI2 expression.
Reprod Biol Endocrinol. 21(58)2023.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Dong N, Li D, Cai H, Shi L and Huang L:
Expression of lncRNA MIR193BHG in serum of preeclampsia patients
and its clinical significance. J Gynecol Obstet Hum Reprod.
51(102357)2022.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Postepska-Igielska A, Giwojna A,
Gasri-Plotnitsky L, Schmitt N, Dold A, Ginsberg D and Grummt I:
LncRNA Khps1 regulates expression of the proto-oncogene SPHK1 via
triplex-mediated changes in chromatin structure. Mol Cell.
60:626–636. 2015.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Xia K, Yu LY, Huang XQ, Zhao ZH and Liu J:
Epigenetic regulation by long noncoding RNAs in osteo-/adipogenic
differentiation of mesenchymal stromal cells and degenerative bone
diseases. World J Stem Cells. 14:92–103. 2022.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Zhao S, Heng N, Weldegebriall Sahlu B,
Wang H and Zhu H: Long noncoding RNAs: Recent insights into their
role in male infertility and their potential as biomarkers and
therapeutic targets. Int J Mol Sci. 22(13579)2021.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Graf J and Kretz M: From structure to
function: Route to understanding lncRNA mechanism. Bioessays.
42(e2000027)2020.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Niyaz M, Ainiwaer J, Abudureheman A, Zhang
L, Sheyhidin I, Turhong A, Cai R, Hou Z and Awut E: Association
between TP53 gene deletion and protein expression in esophageal
squamous cell carcinoma and its prognostic significance. Oncol
Lett. 20:1855–1865. 2020.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Zhang A, Xu M and Mo YY: Role of the
lncRNA-p53 regulatory network in cancer. J Mol Cell Biol.
6:181–191. 2014.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Sharp AN, Heazell AE, Baczyk D, Dunk CE,
Lacey HA, Jones CJ, Perkins JE, Kingdom JC, Baker PN and Crocker
IP: Preeclampsia is associated with alterations in the p53-pathway
in villous trophoblast. PLoS One. 9(e87621)2014.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Gao Q, Zhu X, Chen J, Mao C, Zhang L and
Xu Z: Upregulation of P53 promoted G1 arrest and apoptosis in human
umbilical cord vein endothelial cells from preeclampsia. J
Hypertens. 34:1380–1388. 2016.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Fang Y, Zhang J, Zhu D, Mei Q, Liao T,
Cheng H, He Y, Cao Y and Wei Z: MANF promotes unexplained recurrent
miscarriages by interacting with NPM1 and downregulating
trophoblast cell migration and invasion. Int J Biol Sci.
20:296–311. 2024.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Bao D, Zhuang C, Jiao Y and Yang L: The
possible involvement of circRNA DMNT1/p53/JAK/STAT in gestational
diabetes mellitus and preeclampsia. Cell Death Discov.
8(121)2022.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Mo HQ, Tian FJ, Ma XL, Zhang YC, Zhang CX,
Zeng WH, Zhang Y and Lin Y: PDIA3 regulates trophoblast apoptosis
and proliferation in preeclampsia via the MDM2/p53 pathway.
Reproduction. 160:293–305. 2020.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Liu C, Hu Y, Wang Z, Pan H, Ren Y, Li X,
Liu Z and Gao H: The downregulation of placental lumican promotes
the progression of preeclampsia. Reprod Sci. 28:3147–3154.
2021.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Qin G, Tu X, Li H, Cao P, Chen X, Song J,
Han H, Li Y, Guo B, Yang L, et al: Long noncoding RNA
p53-stabilizing and activating RNA promotes p53 signaling by
inhibiting heterogeneous nuclear ribonucleoprotein K deSUMOylation
and suppresses hepatocellular carcinoma. Hepatology. 71:112–129.
2020.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Chen Y, Hao Q, Wang S, Cao M, Huang Y,
Weng X, Wang J, Zhang Z, He X, Lu H and Zhou X: Inactivation of the
tumor suppressor p53 by long noncoding RNA RMRP. Proc Natl Acad Sci
USA. 118(e2026813118)2021.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Song C, Xiong Y, Liao W, Meng L and Yang
S: Long noncoding RNA ATB participates in the development of renal
cell carcinoma by downregulating p53 via binding to DNMT1. J Cell
Physiol. 234:12910–12917. 2019.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Tang R, Mei X, Wang YC, Cui XB, Zhang G,
Li W and Chen SY: LncRNA GAS5 regulates vascular smooth muscle cell
cycle arrest and apoptosis via p53 pathway. Biochim Biophys Acta
Mol Basis Dis. 1865:2516–2525. 2019.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Li X, Zhao J, Geng J, Chen F, Wei Z, Liu
C, Zhang X, Li Q, Zhang J, Gao L, et al: Long non-coding RNA MEG3
knockdown attenuates endoplasmic reticulum stress-mediated
apoptosis by targeting p53 following myocardial infarction. J Cell
Mol Med. 23:8369–8380. 2019.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Stein Y, Rotter V and Aloni-Grinstein R:
Gain-of-function mutant p53: All the roads lead to tumorigenesis.
Int J Mol Sci. 20(6197)2019.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Vousden KH and Prives C: Blinded by the
light: The growing complexity of p53. Cell. 137:413–431.
2009.PubMed/NCBI View Article : Google Scholar
|