Introduction
Lung cancer is one of the most common malignant
tumors in humans and has high morbidity and mortality. Worldwide,
over 1.3 million people are diagnosed each year with lung cancer,
with over 1.1 million deaths (1,2).
Non-small cell lung cancer (NSCLC) is the most common type of lung
cancer, accounting for 85% of cases. The prognosis of patients with
NSCLC is extremely poor because most patients are diagnosed in an
advanced stage with local or distant metastases. Surgical treatment
is reserved only for patients with early stage disease.
Radiotherapy and chemotherapy are not specific to cancer cells and
produce severe side effects, including bone marrow suppression,
renal impairment and second cancer occurrence (3,4).
Therefore, development of new therapeutic strategies is an urgent
need for the treatment of NSCLC.
Angiogenesis, the outgrowth of new blood vessels
from pre-existing ones, plays an essential role in tumor growth and
metastasis (5). A positive
association between tumor angiogenesis and tumor aggressiveness in
carcinoma of the breast, lung, prostate, malignant melanoma and
other solid tumors has been confirmed by many clinical studies
(6). Thus, depriving a tumor of
its vascular supply with anti-angiogenic agents is a promising
approach against tumors (7).
Pigment epithelium-derived factor (PEDF), a 50 kDa secreted
glycoprotein that belongs to the non-inhibitory serpin group, is a
potent endogenous inhibitor of angiogenesis (8,9).
PEDF exerts its antitumor effects via antiangiogenesis, tumor
differentiation, and direct tumor suppression by apoptosis
(8). Much focus on PEDF as a
promising therapeutic target in cancer has stemmed from its potent
antiangiogenic activity, which has shown to be more effective than
any other known endogenous angiogenic inhibitor, including
angiostatin, thrombospondin-1, and endostatin (10). It has been demonstrated that PEDF
can inhibit the proliferation and migration of endothelial cells
and can induce apoptosis (8). A
number of studies showed inverse correlation between PEDF levels,
grade and metastatic potential of prostate cancer (11), pancreatic cancer (12), glioma (13), cervical carcinoma (14) as well as lung carcinoma (15).
Marrow-derived stromal cells (MSCs), also known as
mesenchymal stem cells or nonhematopoietic progenitor cells, are
precursors that have the potential to differentiate along
osteogenic, adipogenic and chondrogenic lineages when placed in the
appropriate environments. This differentiation plasticity makes
MSCs the ultimate candidates for future utilization in cell therapy
and tissue regeneration (16,17). Furthermore, MSCs expressing
transgenes maintained long-term expression (up to 6 months) in
vivo because of their hypoimmunogenic properties and production
of immunosuppressive molecules (18,19). The tumor microenvironment is known
to provide a preferential niche for MSCs homing and survival. As
MSCs have the capacity to home to the tumor microenvironment, they
may be promising tools for the selective delivery of antineoplastic
drugs to tumors (20,21).
In this study, we developed a novel strategy of
tumor gene therapy in which MSCs loaded with recombinant
adenoviruses expressing PEDF could effectively suppress tumor
growth and prolong the lifespan through inhibiting angiogenesis in
a mouse model, indicating that MSCs may be employed as an effective
carrier for tumor gene therapy.
Materials and methods
Cell lines and culture
Lewis lung carcinoma (LL/2) and human embryonic
kidney 293 cell lines (purchased from American Type Culture
Collection, ATCC, Rockville, MD) were maintained in Dulbecco’s
modified Eagle’s medium (Gibco-BRL, Grand Island, NY) supplemented
with 10% heat-inactivated fetal bovine serum (Gibco, Auckland, NZ),
2 mM L-glutamine and 100 μg/ml of Amikacin. Human umbilical vein
endothelial cells (HUVECs) were isolated from human umbilical cord
veins as previously described (22), and grown in EBM-2 medium with
SingleQuots (Lonza) containing VEGF and other growth factors.
HUVECs at passages 2 to 6 were used for all experiments.
Isolation and culture of marrow MSCs
Female C57BL/6 mice 4- to 6-week-old were
sacrificed, femurs and tibias were isolated, and whole bone marrow
was retrieved by flushing these bones with low-glucose DMEM
(Gibco), supplemented with 10% FBS and 50 U/ml penicillin, 50 μg/ml
streptomycin. All of these bone marrow cells were then cultured at
37°C in a 5% CO2 and a 95% humidified atmosphere. Two
days later, the nonadherent hematopoietic cells were discarded and
the adherent MSCs were preserved for further expansion. The medium
was changed twice/week and the MSCs were cultured for 14-days
before first passage. Cells of 4–5 passages were used in the
experiments (23). MSCs have been
proved to express several cell lineage-specific antigens (24). Therefore, the plate-adhering cells
were further sorted by flow cytometry using CD34, CD44, CD45, CD73,
CD90 and CD105 (BD Biosciences) for phenotype characteristics of
MSCs. Isotypic control analyses were conducted in parallel. Flow
cytometric analysis was performed on a BD Biosciences FACSCalibur
flow cytometer, and data were analyzed with the CellQuest Pro
software (BD Immunocytometry Systems).
Adenoviral transduction of MSCs
The adenoviruses were created using the AdEasy
system. The viruses were amplified in HEK293 cells and purified on
CsCl gradients according to standard methods (25). When the confluence of
proliferating MSCs reached or surpassed ∼90%, the cells were
transduced with recombinant adenovirus at a multiplicity of
infection (MOI) of 1,500 for 4 h. MSCs were also infected with
adenovirus-LacZ (Ad-LacZ) at an MOI of 1,500 as a control.
Verification of expressed PEDF from
MSCs-PEDF in vitro
Western blot analysis was performed as described
previously (26). Briefly, MSCs
were infected with adenoviruses for 4 h and then the
virus-containing medium was removed and additionally incubated in
low-serum medium (low-glucose DMEM containing 2% FBS). After a
further 48 h of incubation, the conditioned media was collected.
The secreted proteins in the supernatants of the culture were
precipitated by TCA-DOC/acetone and the western blotting assay was
performed using a standard method with a mouse anti-human PEDF
monoclonal antibody (R&D Systems, Boston, MA, USA) and a
biotinylated secondary antibody, and the bands were detected using
an enhanced chemiluminescence detection system (Pierce, Rockford,
IL, USA). The concentration of the PEDF secreted in the culture
supernatants was detected using a sandwich enzyme-linked
immunosorbent assay (ELISA) kit for the human PEDF protein
(Groundwork Biotechnology Diagnosticate Ltd., San Diego, CA, USA)
following the manufacturer’s protocol.
Tube formation assay
The tube formation assay was conducted as described
previously (27) to evaluate the
antiangiogenic activity of PEDF produced by MSCs-PEDF. Each well of
a 96-well plate was coated with 50 μl Matrigel (BD Biosciences).
After polymerization at 37°C for 30 min, HUVECs
(2×104/well) were suspended in 200 μl of the conditioned
media derived from MSCs, MSCs-LacZ, MSCs-PEDF respectively and
seeded onto the Matrigel. Six hours later, cells were viewed and
photographed under an inverted microscope with a digital camera.
The tubes of five fields were counted and the data were
averaged.
Transwell invasion assay in vitro
To determine whether PEDF secreted by MSCs-PEDF can
inhibit HUVEC invasion, a Transwell invasion assay was performed as
previously described (28).
Briefly, the filter of the Transwell chamber (Millipore) was coated
with 50 μl Matrigel (BD Biosciences). After Matrigel
polymerization, HUVEC (2×104/well) were suspended in 200
μl of the conditioned media derived from MSCs, MSCs-LacZ, MSCs-PEDF
respectively and loaded in the upper chamber. The lower well of the
Transwell plate was filled with 600 μl of EBM-2 medium containing
various growth factors. Cells were allowed to migrate for 24 h at
37°C. Non-migrated cells were scraped with a cotton swab, and
migrated cells were fixed with 100% methanol and stained with 0.05%
crystal violet. The number of cells that had migrated to the lower
side of the filter was quantified by manual counting under a light
microscope with five fields (x100). All assays were conducted in
triplicate.
Alginate encapsulation assay
An alginate-encapsulated tumor cell assay was
conducted as previously described (26). Alginate beads were formed with
about 1×105 LLC cells per bead and implanted s.c. into
both dorsal sides of C57BL/6 mice. Then the mice were injected with
PBS, MSCs-LacZ, MSCs-PEDF or free Ad-PEDF via the tail vein on Day
2 for once. Twelve days after the beads were implanted, the mice
were injected intravenously with a dose at 100 mg/kg FITC-dextran
solution (Sigma) in 100 μl. Alginate beads were removed and
photographed with a digital camera within 20 min after FITC-dextran
administration. The uptake of FITC-dextran was quantified against a
calibration curve of FITC-dextran.
PEDF-expressing MSCs reduce subcutaneous
tumor growth in vivo
Female syngeneic C57BL/6 mice 6–8 weeks of age were
purchased from the West China Experimental Animal Center of Sichuan
University (Sichuan, China) and maintained in a dedicated
pathogen-free environment. All animal procedures were approved by
the Institute’s Animal Care and Use Committee. Mice were implanted
s.c. in the right flank with LLC cells at 5×105/mouse.
When tumor diameters reached 3 mm, mice were randomly divided into
four groups and i.v. injected with PBS, 108 plaque
forming units (PFU) of Ad-PEDF, 5×105 MSCs-LacZ or
5×105 MSCs-PEDF at 4 days intervals for two times. Tumor
growth and survival rate were monitored every 3 days by caliper.
Tumor volumes were determined at various time points using the
formula: width2 × length × 0.52. Cohorts of mice (n=8)
from each group were sacrificed at Day-30 to determine the effect
of therapy and remaining mice (n=10) were monitored for long-term
survival. The duration of survival was recorded when the mouse died
or had to be sacrificed secondary to tumor diameter >20 mm,
tumor ulceration or bleeding. The difference in survival was
determined by a log-rank test.
Immunofluorescence staining of tumor
vessels
To determine the degree of tumor-induced
angiogenesis, frozen sections were prepared from the tumor tissue
30 days after inoculation. The procedure of immunofluorescence
staining was done as described previously (29). Briefly, sections were probed with
a monoclonal anti-CD31 antibody (BD Biosciences) and nuclei were
stained with DAPI. The sections were viewed at low magnification
(x40) to identify the most vascular-rich area (hot spot) and 5
non-overlapping fields were selected. Microvessel counting was
performed at a high-power field (×200) under fluorescence
microscopy and data were expressed as the mean number of
CD31-positive vessels per field from three sections in each
tumor.
TUNEL assay for in situ apoptosis
To determine the degree of apoptosis, terminal dUTP
nick-end labeling (TUNEL) staining was performed using an in
situ cell death detection kit (Promega) following the
manufacturer’s protocol. Tumor species were prepared as described
above. Three tumors from each group were analyzed. The number of
apoptotic cells was quantified in 5 randomly selected fields at
×200 magnifications using a fluorescence microscope.
ELISA for expressed PEDF in serum and
tumor tissue
Serum samples and fresh tumor tissues were harvested
and detected using ELISA (Groundwork Biotechnology Diagnosticate,
Ltd.) in triplicate for the tendencies of PEDF levels at 7, 14 and
21 days after i.v. administration of treatment. Non-necrotic fresh
tumor tissues were cut into small pieces, lysed in RIPA buffer on
ice, passed through a sieve (BellCo Glass), and evaluated for the
local production of PEDF as ng/mg.
Statistical analysis
All values are presented as means ± SEM (standard
error of the mean). SPSS 17.0 was used for statistical analysis.
Significance was evaluated using one-way ANOVA, and P<0.05 was
considered significant differences. The survival rates were
compared by means of the log-rank test.
Results
MSCs isolation, genetic modification and
confirmation of PEDF expression in vitro
The adherent cells have a typical spindle-like
morphology (Fig. 1B). Analysis of
immunophenotype showed that isolated BM-derived cells shared
classical immunophenotype of MSCs, including positivity for CD44,
CD73, CD90, CD105, but negativity for CD34, CD45 (Fig. 1A). Thus, as the data showed, we
successfully obtained abundant MSCs from mouse bone marrow.
After BMSCs were cultured to reach 90% confluence
and incubated with adenoviruses at a MOI of 1,500 for 4 h, the
secreted PEDF in the supernatants of conditioned media was verified
by western blotting. As shown in Fig.
1C, a specific 50 kDa band was observed only in the
supernatants from Ad-PEDF-transduced MSCs, which indicated that
PEDF was appropriately synthesized and processed. PEDF levels in
the collected media (n=3) were determined utilizing a sandwich
enzyme-linked immunosorbent assay (ELISA) kit for the human PEDF
protein (Groundwork Biotechnology Diagnosticate Ltd.). It showed
that PEDF was produced in vitro in great amounts for 48 h,
with a concentration as high as 78.8±4.8 ng/ml (Fig. 1D).
Bioactivity of PEDF produced by
PEDF-transduced MSCs in vitro
HUVECs when seeded on Matrigel become elongated and
form capillary-like structures mimicking the in vivo
neoangiogenesis process (30). We
used this assay to examine the antiangiogenic activity of PEDF
expressed by MSCs-PEDF. As shown in Fig. 2A, HUVECs plated on the surface of
Matrigel formed capillary-like structures in the MSCs and MSCs-LacZ
groups after 6 h, however, treatment with the conditioned media
(CM) from MSCs-PEDF dramatically blocked the tube formation
(P<0.01). We also conducted Transwell invasion assays to
evaluate the ability of HUVECs to pass through the Matrigel and
membrane barrier of the Transwell in the presence of the CM derived
from MSCs, MSCs-LacZ and MSCs-PEDF. Data showed that the CM from
the MSCs-PEDF significantly inhibited the invasion properties of
endothelial cells (Fig. 2B)
(P<0.01).
Therapeutic effect of MSCs-PEDF on LLC
cell growth in vivo
To learn if the PEDF secreted in vivo by PEDF
gene-modified MSCs can engender a beneficial effect against LLC
cells in vivo, we implanted isogenic C57BL/6 mice with LLC
cells s.c. and treated with PBS, 108 PFU of Ad-PEDF,
5×105 MSCs-LacZ or 5×105 MSCs-PEDF at 4 days
intervals for two times i.v. Cohorts of mice (n=8) from each group
were sacrificed at Day-30 to determine the effect of therapy and
remaining mice (n=10) were monitored for long-term survival. As
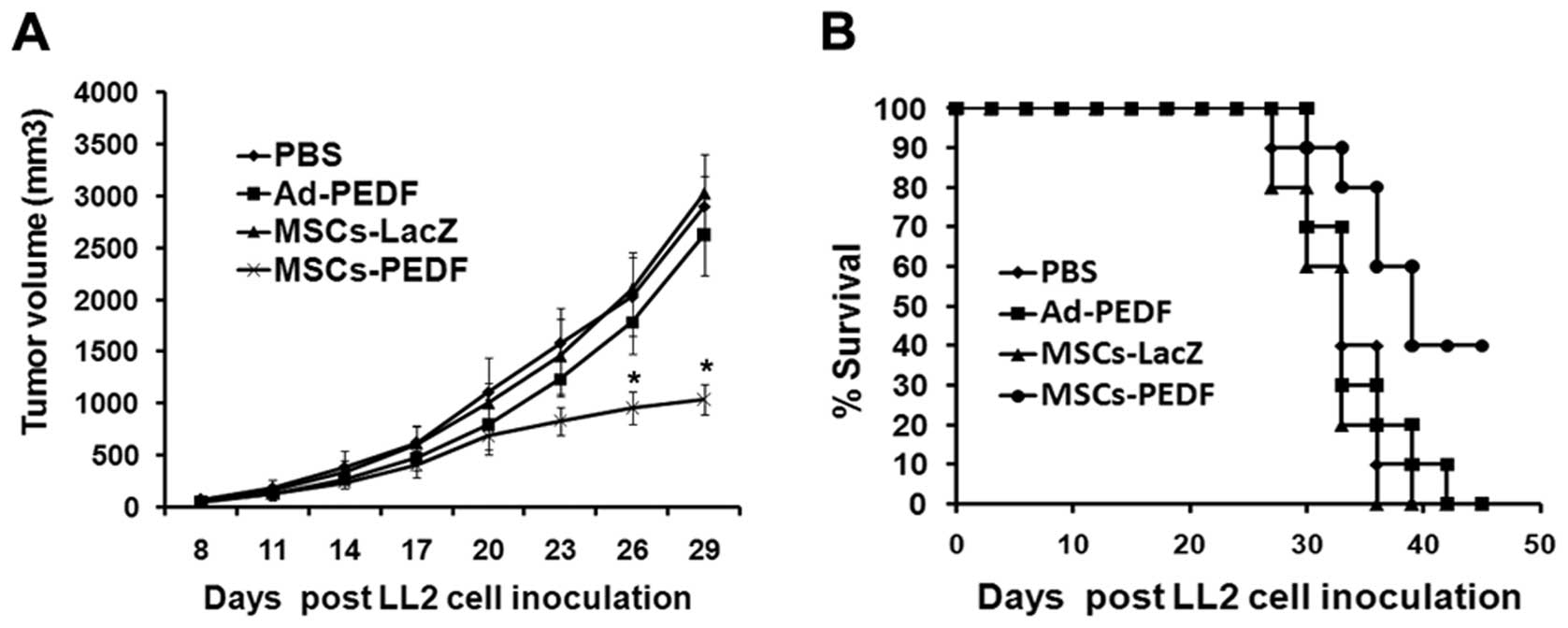
shown in Fig. 3A, the tumor
volume in the MSCs-PEDF group was dramatically smaller compared
with that of the PBS group (1038.8±139.0 vs. 2897.7±274.9
mm3, ANOVA; P<0.01). By contrast, the growth rate of
tumors was not affected by administration of free Ad-PEDF
(2622.8±359.1 mm3) and MSCs transduced with Ad-LacZ
(3019.2±360.9 mm3). These data suggest that intravenous
administration of MSC-PEDF cells inhibits the growth of tumors,
whereas systemically injected free Ad-PEDF or MSC-LacZ cells does
not.
Moreover, MSCs producing PEDF was found to
significantly increase the survival of mice. All animals in the
control groups died by Day 42, whereas 40% of mice treated with
MSC-PEDF survived beyond that time (P<0.01). There was no
significant increase in survival of mice treated with MSC
transduced with Ad-LacZ or free Ad-PEDF (Fig. 3B).
Inhibition of angiogenesis in vivo
Immunofluorescence anti-CD31 staining was performed
to evaluate the consequence of anti-angiogenesis therapy. The most
highly vascularized area of each tumor section was identified on
low power and five high-powered fields were counted in this area of
greatest vessel density. The tumor tissue from MSCs-PEDF-treated
mice showed apparently decreased microvessel density compared with
the other three control groups (Fig.
4A). The inhibition of angiogenesis was confirmed in the
alginate-encapsulated tumor cell assay. The treated tumor exhibited
relatively little vascularity (Fig.
4B). Vascularization of beads over 12 days can then be measured
by uptake of FITC-dextran into beads. Vascularization of alginate
beads was apparently reduced, and FITC-dextran uptake was decreased
in MSCs-PEDF-treated mice compared with that in controls (ANOVA;
P<0.01).
Induction of apoptosis in vivo
The TUNEL assay was performed to detect tumor cell
apoptosis to further investigate the role of MSCs-PEDF treatment in
tumors in vivo. As shown in Fig. 5, MSCs-PEDF-treated tumors showed
significantly more apoptotic cells (with green nuclei) than tumors
from the PBS, Ad-PEDF or MSCs-LacZ-treated groups. The apoptosis
index was also significantly higher in the MSCs-PEDF-treated group
compared with the controls (ANOVA; P<0.01). The inhibition of
angiogenesis and the consequent induction of apoptosis underlie the
mechanism of tumor volume decrease and the lifespan prolongation in
tumor-bearing mice.
ELISA for expressed PEDF in serum and
tumor tissue
The systemic and local (serum and intratumoral)
expression tendencies of PEDF at indicated points by ELISA are
shown in Fig. 6. In the MSCs-PEDF
group, the serum levels of PEDF were ∼38 ng/ml on Day 7, and
declined to ∼26 ng/ml after that; meanwhile, the intratumoral
levels of PEDF rose from ∼168 ng/mg (7 days) to ∼253 ng/mg (14
days), and then slightly declined to ∼238 ng/mg in the 21 days,
∼7-fold vs. the intratumoral level in the free Ad-PEDF group (32
ng/mg). In free Ad-PEDF group, the levels of PEDF, diminished
quickly from ∼60 to 25 ng/ml in serum and from ∼50 to ∼32 ng/mg
intratumorally. Other controls including PBS, MSCs-LacZ hardly led
to any elevation of serous or intratumoral levels of PEDF.
Discussion
In this study, we demonstrated that systemically
delivered MSCs loaded with the PEDF gene could preferentially home
to tumor loci and induce antiangiogenesis as well as apoptosis of
cancer cells, resulting in the inhibition of tumor growth and
prolongation of lifespan in the mouse Lewis lung carcinoma (LLC)
model.
The choice of PEDF over other cytokines for
antitumor effects on LLC in our studies is based on its potent
antiangiogenic activity, which has shown to be more effective than
any other known endogenous angiogenic inhibitor, including
angiostatin, thrombospondin-1, and endostatin. Moreover, PEDF can
also exert its antitumor activity via tumor differentiation and
direct tumor suppression by apoptosis. It has been reported that
PEDF can inhibit several types of tumors including lung carcinomas
(8). Purified recombinant PEDF
and PEDF-based viral vectors have been investigated in a lot of
studies. Nevertheless, a suboptimal half-life in plasma reduces
their possible therapeutic effects (14,25). To overcome this shortcoming, a
novel and more effective vehicle is needed. In this study, we
demonstrated that the engineered MSCs can secret high-level PEDF
and the conditioned media derived from these engineered MSCs was
capable of inhibiting tube formation and migration of human
umbilical vein endothelial cells in vitro.
MSCs are attractive as a cellular vehicle for
delivery of antitumoral agents in tumor gene therapy, since they
possess several unique biological features. First, the poorer
immunogenicity of MSCs enables them to survive for hundreds of days
in the host, which can offer a relatively long effect for chronic
disease (31). Second, the
capacity of MSCs homing to malignant tissues can offer a higher
concentration of antineoplastic drugs in the local tumor sites and
lower side effect on other healthy organs (21,31). Last, MSCs are easy to harvest,
isolate, expand and to gene modify in vitro, which means the
offer convenience and cost effectiveness in future preclinical or
clinical applications.
It has been shown that the migratory ability of MSCs
was not affected by viral transduction in vitro and in
vivo (21,32,33). As a relatively perfect candidate
of cellular vehicles used in our study, MSCs could protect PEDF to
escape from being destroyed due to entering the circulatory system
and being accompanied by potential adverse effects owing to
interfering spontaneously with other systems and thereby improving
its pharmacokinetics. MSCs were capable of migrating to the tumor
sites after intravenous infusion; by contrast, free Ad-PEDF lacks
this attribute (32,33). In our study, consecutive
administration of free Ad-PEDF could only partly retard the tumor
growth to some extent in the midway stage; yet, with potential
systemic side effects to the hosts. This also has no therapeutic
effect in the ultimate stage. This phenomenon could be partly
attributed to the temporary high serum yet low intratumor levels of
PEDF as a result of nonselective expression and subsequent
elimination of free Ad-PEDF.
Our results suggest that the antitumor activity of
MSC-PEDF cells is associated not with the serum concentration of
PEDF but rather with the engraftment of MSCs in tumors and the
local effects of the PEDF they produce on malignant cells.
Therefore, selective engraftment of MSC in tumors is an important
issue for further clinical development of this method. Despite the
antitumor effects of MSCs-PEDF observed, there was an absence of
higher systemic levels of the transgenic protein, suggesting the
local effects of MSCs-produced PEDF in the tumor microenvironment.
These findings originally suggest that i.v. infused MSCs can
circulate over the lung vascular bed and migrate into s.c. growing
tumors, supporting the concept that MSCs have the ability of homing
into tumor sites. Therefore, therapy with MSCs-PEDF cells could be
used to increase PEDF expression in tumors and surrounding tissues
and to control the growth of malignant cells. In fact, the
potential clinical application of MSCs-PEDF cells could be broad,
because many types of malignancies are sensitive to the
antiproliferative or proapoptotic effects of PEDF in vitro.
The data indicate that systemic administration of MSCs-PEDF led to
a pronounced growth inhibition of established subcutaneous LLC and
prolonged survival of mice.
In conclusion, our investigation reveals the
potential of genetically modified MSC expressing PEDF for Lewis
lung cancer. This may prefigure an integrated cellular and
molecular therapeutic weapon targeted against advanced human
malignancies. However, the same approach could be explored in the
delivery of other agents with antitumor activities.
Acknowledgements
The authors thank members of the State
Key Laboratory of Biotherapy for helpful discussions. This study
was supported by the National Natural Science Foundation (30973507)
and the National 973 Basic Research Program of China (2010CB529900
and 2010CB529906).
References
|
1.
|
J FerlayP AutierM BoniolM HeanueM
ColombetP BoyleEstimates of the cancer incidence and mortality in
Europe in 2006Ann
Oncol18581592200710.1093/annonc/mdl49817287242
|
|
2.
|
DM ParkinF BrayJ FerlayP PisaniGlobal
cancer statistics, 2002CA Cancer J
Clin5574108200510.3322/canjclin.55.2.74
|
|
3.
|
DS EttingerW AkerleyG BeplerNon-small cell
lung cancerJ Natl Compr Cancer Netw8740801201020679538
|
|
4.
|
A JemalR SiegelE WardY HaoJ XuMJ
ThunCancer statistics, 2009CA Cancer J
Clin59225249200910.3322/caac.20006
|
|
5.
|
J FolkmanY ShingAngiogenesisJ Biol
Chem26710931109341992
|
|
6.
|
VW van HinsberghA CollenP
KoolwijkAngiogenesis and anti-angiogenesis: perspectives for the
treatment of solid tumorsAnn Oncol10Suppl 4S60S63199910436787
|
|
7.
|
CR DassTM TranPF ChoongAngiogenesis
inhibitors and the need for anti-angiogenic therapeuticsJ Dent
Res86927936200710.1177/15440591070860100517890668
|
|
8.
|
ET EkCR DassPF ChoongPigment
epithelium-derived factor: a multimodal tumor inhibitorMol Cancer
Ther516411646200610.1158/1535-7163.MCT-06-010716891449
|
|
9.
|
FR SteeleGJ ChaderLV JohnsonJ
Tombran-TinkPigment epithelium-derived factor: neurotrophic
activity and identification as a member of the serine protease
inhibitor gene familyProc Natl Acad Sci
USA9015261530199310.1073/pnas.90.4.15268434014
|
|
10.
|
DW DawsonOV VolpertP GillisPigment
epithelium-derived factor: a potent inhibitor of
angiogenesisScience285245248199910.1126/science.285.5425.24510398599
|
|
11.
|
JA DollVM StellmachNP BouckPigment
epithelium-derived factor regulates the vasculature and mass of the
prostate and pancreasNat Med9774780200310.1038/nm87012740569
|
|
12.
|
R HaseM MiyamotoH UeharaPigment
epithelium-derived factor gene therapy inhibits human pancreatic
cancer in miceClin Cancer
Res1187378744200510.1158/1078-0432.CCR-05-132316361561
|
|
13.
|
M GuanHF YamB SuLoss of pigment epithelium
derived factor expression in glioma progressionJ Clin
Pathol56277282200310.1136/jcp.56.4.27712663639
|
|
14.
|
J YangS ChenX HuangGrowth suppression of
cervical carcinoma by pigment epithelium-derived factor via
anti-angiogenesisCancer Biol
Ther9967974201010.4161/cbt.9.12.1163520364117
|
|
15.
|
L WangV SchmitzA Perez-MediavillaI IzalJ
PrietoC QianSuppression of angiogenesis and tumor growth by
adenoviral-mediated gene transfer of pigment epithelium-derived
factorMol Ther87279200310.1016/S1525-0016(03)00128-X12842430
|
|
16.
|
BD RoordaA ter ElstWA KampsES de BontBone
marrow-derived cells and tumor growth: contribution of bone
marrow-derived cells to tumor micro-environments with special focus
on mesenchymal stem cellsCrit Rev Oncol
Hematol69187198200910.1016/j.critrevonc.2008.06.00418675551
|
|
17.
|
MF PittengerAM MackaySC BeckMultilineage
potential of adult human mesenchymal stem
cellsScience284143147199910.1126/science.284.5411.14310102814
|
|
18.
|
N EliopoulosA Al-KhaldiM CrosatoK
LachapelleJ GalipeauA neovascularized organoid derived from
retrovirally engineered bone marrow stroma leads to prolonged in
vivo systemic delivery of erythropoietin in nonmyeloablated,
immunocompetent miceGene Ther10478489200310.1038/sj.gt.3301919
|
|
19.
|
SH SeoKS KimSH ParkThe effects of
mesenchymal stem cells injected via different routes on modified
IL-12-mediated antitumor activityGene
Ther18488495201110.1038/gt.2010.17021228885
|
|
20.
|
U GalderisiA GiordanoMG PaggiThe bad and
the good of mesenchymal stem cells in cancer: Boosters of tumor
growth and vehicles for targeted delivery of anticancer agentsWorld
J Stem Cells2512201010.4252/wjsc.v2.i1.521607110
|
|
21.
|
M StudenyFC MariniRE ChamplinC ZompettaIJ
FidlerM AndreeffBone marrow-derived mesenchymal stem cells as
vehicles for interferon-beta delivery into tumorsCancer
Res6236033608200212097260
|
|
22.
|
EA JaffeRL NachmanCG BeckerCR
MinickCulture of human endothelial cells derived from umbilical
veins. Identification by morphologic and immunologic criteriaJ Clin
Invest5227452756197310.1172/JCI1074704355998
|
|
23.
|
N EliopoulosM FrancoisMN BoivinD
MartineauJ GalipeauNeo-organoid of marrow mesenchymal stromal cells
secreting interleukin-12 for breast cancer therapyCancer
Res6848104818200810.1158/0008-5472.CAN-08-016018559528
|
|
24.
|
S BobisD JarochaM MajkaMesenchymal stem
cells: characteristics and clinical applicationsFolia Histochem
Cytobiol44215230200617219716
|
|
25.
|
LP YangP ChengXC PengAntitumor effect of
adenovirus-mediated gene transfer of pigment epithelium-derived
factor on mouse B16–F10 melanomaJ Exp Clin Cancer
Res2875200919500366
|
|
26.
|
JY LiuYQ WeiL YangImmunotherapy of tumors
with vaccine based on quail homologous vascular endothelial growth
factor
receptor-2Blood10218151823200310.1182/blood-2002-12-377212750177
|
|
27.
|
X PangZ YiX
ZhangAcetyl-11-keto-beta-boswellic acid inhibits prostate tumor
growth by suppressing vascular endothelial growth factor receptor
2-mediated angiogenesisCancer
Res6958935900200910.1158/0008-5472.CAN-09-075519567671
|
|
28.
|
S ZhangZ CaoH TianSKLB1002, a novel potent
inhibitor of VEGF receptor 2 signaling, inhibits angiogenesis and
tumor growth in vivoClin Cancer
Res1744394450201110.1158/1078-0432.CCR-10-310921622720
|
|
29.
|
KA MohamedaliD KedarP SweeneyThe
vascular-targeting fusion toxin VEGF121/rGel inhibits the growth of
orthotopic human bladder carcinoma
tumorsNeoplasia7912920200510.1593/neo.0529216242074
|
|
30.
|
G TarabolettiR GiavazziModelling
approaches for angiogenesisEur J
Cancer40881889200410.1016/j.ejca.2004.01.00215120043
|
|
31.
|
V FritzC JorgensenMesenchymal stem cells:
an emerging tool for cancer targeting and therapyCurr Stem Cell Res
Ther33242200810.2174/15748880878348946218220921
|
|
32.
|
X ChenX LinJ ZhaoA tumor-selective
biotherapy with prolonged impact on established metastases based on
cytokine gene-engineered MSCsMol
Ther16749756200810.1038/mt.2008.318362930
|
|
33.
|
Y GaoA YaoW ZhangHuman mesenchymal stem
cells overexpressing pigment epithelium-derived factor inhibit
hepatocellular carcinoma in nude
miceOncogene2927842794201010.1038/onc.2010.38
|