Introduction
Transmissible spongiform encephalopathies (TSEs) are
fatal neurodegenerative diseases in humans and animals (1) that share a pathognomonic triad as a
common histopathological trait: the spongiform vacuolation of grey
matter, neuronal cell death, glial cell proliferation and,
occasionally, amyloid deposition (2). One of the fundamental events related
to TSE pathogenesis is the refolding of a host-encoded
glycoprotein, the cellular prion protein (PrPC), into a
protease-insensitive isoform (PrPSc) that aggregates into deposits
of misfolded protein (3).
Conversion into PrPSc is driven by the transition of a large
N-terminal region of PrPC from a random coil to a β-sheet
structure, which becomes predominant over the α-helix content (43
vs. 30%) (3). The synthetic human
prion protein peptide (PrP) (106–126) corresponds to amino acid
residues 106–126 of the prion protein placed in the flexibly
disordered N-terminal domain. PrP (106–126) maintains some of the
pathogenic and physiological properties of PrPSc, including the
induction of the apoptosis of hippocampal neurons and the
proliferation of astrocytes (4).
Several biochemical and biological properties of PrPSc
(β-sheet-rich structure, amyloidogenesis, and neurotoxic and
gliotrophic effects) have been discovered (4–7)
from an analysis of the biological activity of different amino acid
segments, a peptide encompassing amino acids 106–126 of the PrP
sequence [PrP (106–126)] and its ability to reproduce in
vitro. The relevance of the 106–126 sequence in PrP biological
activity was further demonstrated by the observation that this
fragment contains the amyloidogenic palindrome AGAAAAGA sequence
(amino acids 113–120), which is the most conserved region within
PrP molecules of different species (8).
The treatment of neurotoxic and neurodegenerative
diseases involves limiting reactive oxygen species (ROS) production
and oxidative stress. A prevoius study addressed neurodegeneration
from a similar mechanistic standpoint based on ROS and oxidative
stress (9). Low levels of ROS,
such as superoxide anion (O2⨪), hydroxyl
radical (HO•), and H2O2, serve as
signaling molecules in various cellular pathways that lead to
growth and survival. However, high levels of ROS induce cellular
damage and death through the oxidation of lipids, proteins and DNA
(10,11). Mitochondrial failure caused by the
aggregation of misfolded proteins is a hallmark of
neurodegenerative disorders, including Alzheimer’s disease,
Parkinson’s disease and prion diseases (12–14). PrP (106–126), a neurotoxic prion
protein, induces mitochondrial dysfunction that results in
neurotoxicity (15).
The dried root of Scutellaria baicalensis
(S. baicalensis) Georgi has been used traditionally as a
Chinese herbal medicine (Huang-qin). Flavonoids extracted from
S. baicalensis are effective in treating the hypoxic brain,
chemical neuronal cell damage and cognitive impairment (16,17). One of the flavanoids, baicalein,
has been shown to have antioxidant activity (18), exert protective effects against
the cytotoxicity induced by oxidative stress (19), as well as some beneficial effects
on the cardiovascular system (20,21), among other biological activities
(22–24). A recent study demonstrated that
baicalein exerts neuroprotective effects in patients with
Parkinson’s disease (25).
In the present study, investigated the effects of
baicalein on human prion protein-induced neuroblastoma cell
apoptosis, as well as the cellular responses to prion
protein-related apoptotic signaling, ROS and mitochondrial
dysfunction. We also examined whether the inhibition of c-Jun
N-terminal kinase (JNK) reduces the neuronal apoptosis caused by
the prion peptide. Our results suggest that baicalein protects
against PrP (106–126)-induced apoptosis, the production of ROS and
mitochondrial dysfunction. Regulating the JNK pathway prevented
neurotoxicity in neuroblastoma cells caused by the prion
peptide.
Materials and methods
Cell culture
The human neuroblastoma cells (SH-SY5Y; SK-N-SH)
were obtained from the American Type Culture Collection (ATCC,
Rockville, MD, USA). The cells were cultured in minimum essential
medium (MEM; Invitrogen-Gibco, Carlsbad, CA, USA) supplemented with
10% fetal bovine serum (FBS; Invitrogen-Gibco), 100 U/ml penicillin
and 0.1 mg/ml gentamycin in a humidified incubator maintained at
37°C and 5% CO2. The cells were treated for 1 h with
baicalein (Sigma-Aldrich, St. Louis, MO, USA) and then exposed to
50 mM PrP (106–126) for 24 h with or without the JNK inhibitor,
SP600125 (Sigma-Aldrich). Untreated cells were used as
controls.
PrP (106–126) treatment
PrP (106–126) was synthesized as previously
described (8). The synthetic PrP
(106–126) peptide (sequence,
Lys-Thr-Asn-Met-Lys-His-Met-Ala-Gly-Ala-Ala-Ala-Ala-Gly-Ala-Val-Val-Gly-Gly-Leu-Gly)
was synthesized by Peptron (Seoul, Korea). The peptide was
dissolved in sterile dimethyl sulfoxide at a stock concentration of
10 mM and stored at −4°C.
Terminal deoxynucleotidyl transferase
dUTP nick end labeling (TUNEL) assay
TUNEL assay was carried out to measure the stage of
cellular apoptosis by a TUNEL-based assay kit (BioVision, Mountain
View, CA, USA). TUNEL analysis was performed according to the
manufacturer’s instructions. Propidium iodide (PI) was employed to
show the cell nuclei.
Annexin V assay
Apoptosis in the detached cells (SH-SY5Y and
SK-N-SH) was assessed using an Annexin V assay kit (Santa Cruz
Biotechnology, Santa Cruz, CA, USA) according to the manufacturer’s
instructions. Annexin V levels were determined by measuring
fluorescence at an excitation wavelength of 488 nm and an emission
wavelength of 525/30 nm using a Guava easyCyte HT System
(Millipore, Bedford, MA, USA).
DCFH-DA assay and visual detection
The SH-SY5Y cells were incubated in MEM containing
10 μM 2′,7′-dichlorodi-hydrofluorescein diacetate
(H2-DCFDA) at 37°C for 30 min. The cells were washed
with phosphate-buffer saline (PBS) and lysed in lysis buffer (25 mM
HEPES; pH 7.4, 100 mM NaCl, 1 mM EDTA, 5 mM MgCl2, 0.1
mM DTT and protease inhibitor mixture). The cells were transferred
to a clear 96-well plate, and fluorescence emissions were measured
at 515 nm (bottom read mode) with an excitation wavelength of 488
nm, using a SpectraMax® M2 (Molecular Devices,
Sunnyvale, CA, USA). The SH-SY5Y cells were cultured on coverslips
in a 24-well plate in MEM containing 10 μM
H2-DCFDA at 37°C for 30 min. The cells were washed with
PBS, mounted with DakoCytomation fluorescent medium (Dako,
Carpinteria, CA, USA) and visualized using a fluorescence
microscope (Nikon Eclipse 80i; Nikon Corporation, Tokyo,
Japan).
Mitochondrial transmembrane potential
(MTP) assay
The change in MTP was evaluated using the cationic
fluorescent indicator, JC-1 (Molecular Probes, Eugene, OR, USA), in
which J-aggregates in intact mitochondria become fluorescent red
with an emission at 583 nm, indicating high or normal MTP and
fluorescent green with an emission at 525 nm, indicating low MTP
when it remains in the monomeric form in the cytoplasm. The SH-SY5Y
cells were incubated in MEM containing 10 μM JC-1 at 37°C
for 15 min, washed with PBS, and then transferred to a clear
96-well plate. JC-1 aggregate fluorescence emissions were measured
at 583 nm with an excitation wavelength of 488 nm, and JC-1 monomer
fluorescence intensity was measured with excitation and emission
wavelengths of 488 and 525 nm, respectively, using a Guava easyCyte
HT System (Millipore). The SH-SY5Y cells were cultured on
coverslips in a 24-well plate, incubated in MEM containing 10
μM JC-1 at 37°C for 15 min, and washed with PBS. Finally,
the cells were mounted with DakoCytomation fluorescent medium
(Dako, Carpinteria, CA, USA) and visualized under a fluorescence
microscope (Nikon Eclipse 80i; Nikon Corporation).
Western blot analysis
The SH-SY5Y and SK-N-SH cells were lysed in lysis
buffer {25 mM HEPES [4-(2-hydroxyethyl)-1-piperazineethanesulfonic
acid], pH 7.4, 100 mM NaCl, 1 mM EDTA, 5 mM MgCl2, 0.1
mM dithiothreitol and a protease inhibitor mixture}, whole cell
proteins were electrophoretically resolved by 10–15% sodium dodecyl
sulfate polyacrylamide gel electrophoresis and transferred onto a
nitrocellulose membrane. Immunoreactivity was detected through
sequential incubation with primary antibodies, horseradish
peroxidase-conjugated secondary antibodies and enhanced
chemiluminescence reagents provided with the West Save Gold
Detection kit (AbFrontier Co., Ltd., Seoul, South Korea). The
primary antibodies used for immunoblotting were
anti-phospho-SAPK/JNK (Cat. no. 9255; Cell Signaling Technology,
Danvers, MA, USA), anti-phospho-Akt (Cat. no. 2118-1; Epitomics,
Burlingame, CA, USA) and anti-β-actin (A5441; Sigma-Aldrich).
Images were examined using a Fusion FX7 imaging system (Vilber
Lourmat, Torcy Z.I. Sud, France). The densitometry of the signal
bands was analyzed using Bio-1D software (Vilber Lourmat).
Statistical analysis
The unpaired t-test or Welch’s correction was used
for comparison between the two groups. For multiple comparison, the
one-way ANOVA followed by the Tukey-Kramer test was used. All
statistical analyses were performed using GraphPad Prism software.
Results were considered significant for values P<0.05, P<0.01
or P<0.001.
Results
Baicalein inhibits PrP (106–126)-induced
neuronal apoptosis
Baicalein has been shown to exert neuroprotective
effects against neuronal impairment (26). In this study, we examined whether
baicalein protects neuronal cells against prion-mediated
neurotoxicity. Initially, we investigated the effects of baicalein
on PrP (106–126)-induced neurotoxicity in SH-SY5Y and SK-N-SH cells
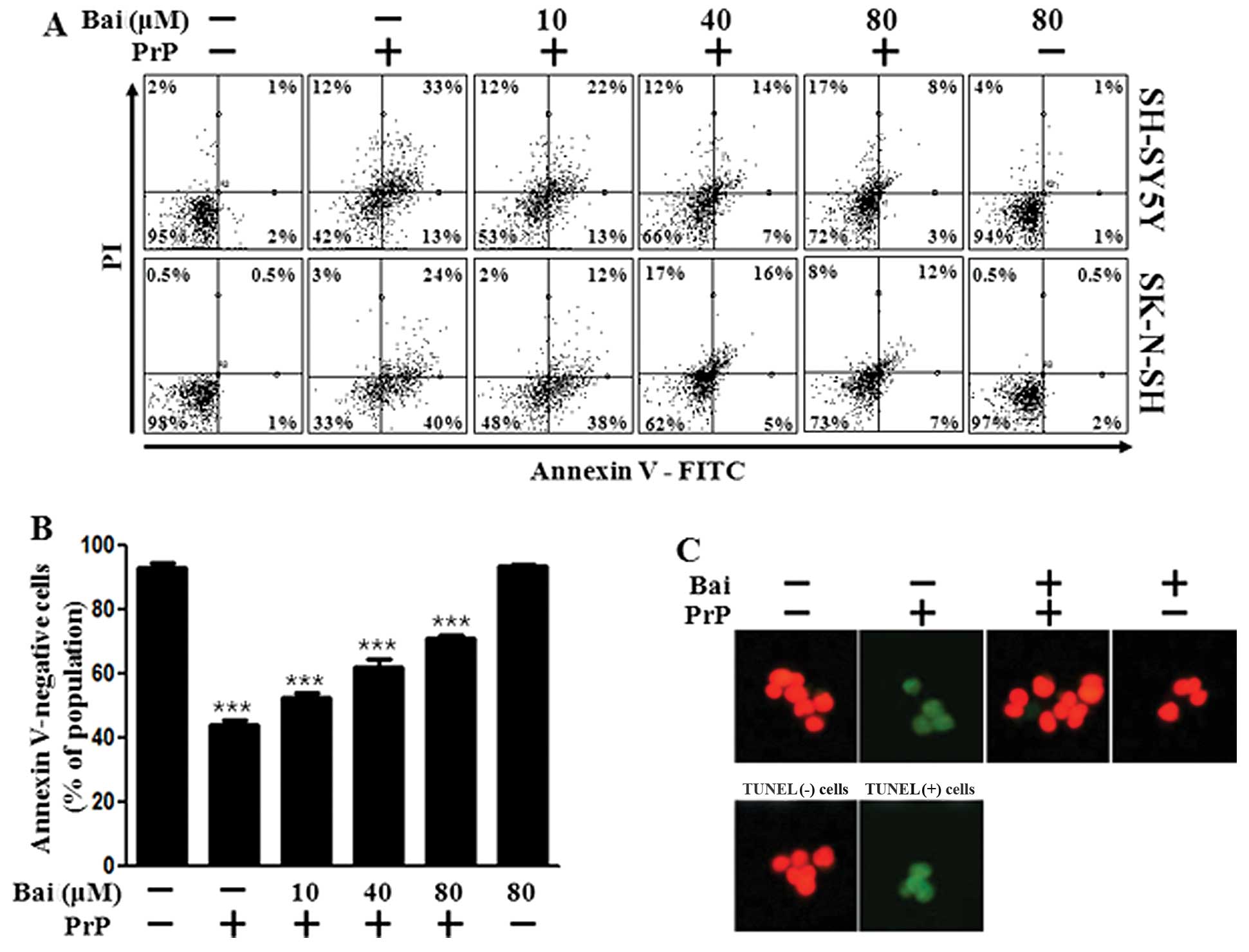
using an Annexin V assay. The SH-SY5Y and SK-N-SH cells were
exposed to baicalein with or without PrP (106–126). The cell
viability of the PrP (106–126)-treated cells decreased by
approximately 42% compared with that of the control SH-SY5Y cells
and by 33% compared with that of the control SK-N-SH cells. The
viability of the cells treated with baicalein only was similar to
that of the untreated controls. Baicalein treatment inhibited the
PrP (106–126)-induced neurotoxicity of the SH-SY5Y and SK-N-SH
cells (Fig. 1A and B). We used a
TUNEL assay (Fig. 1C) and
microscopic imaging (Fig. 1D) and
demonstrated that the apoptotic process in the PrP
(106–126)-treated cells led to the emission of green fluorescence,
indicating DNA strand breakage. Baicalein treatment recovered the
cell death induced by PrP (106–126) treatment as measured by cell
density (Fig. 1D). These results
indicate that baicalein is effective in preventing PrP
(106–126)-induced apoptosis in SH-SY5Y and SK-N-SH cells.
Treatment with baicalein exerts
inhibitory effects on PrP (106–126)-mediated ROS production and
mitochondrial dysfunction
Oxidative stress is responsible for the neuronal
cell death associated with prion diseases. Therefore, in this
study, we investigated the mechanisms through which baicalein
induces resistance to PrP (106–126) by assessing the antioxidant
properties and generation of ROS in neuronal cells following
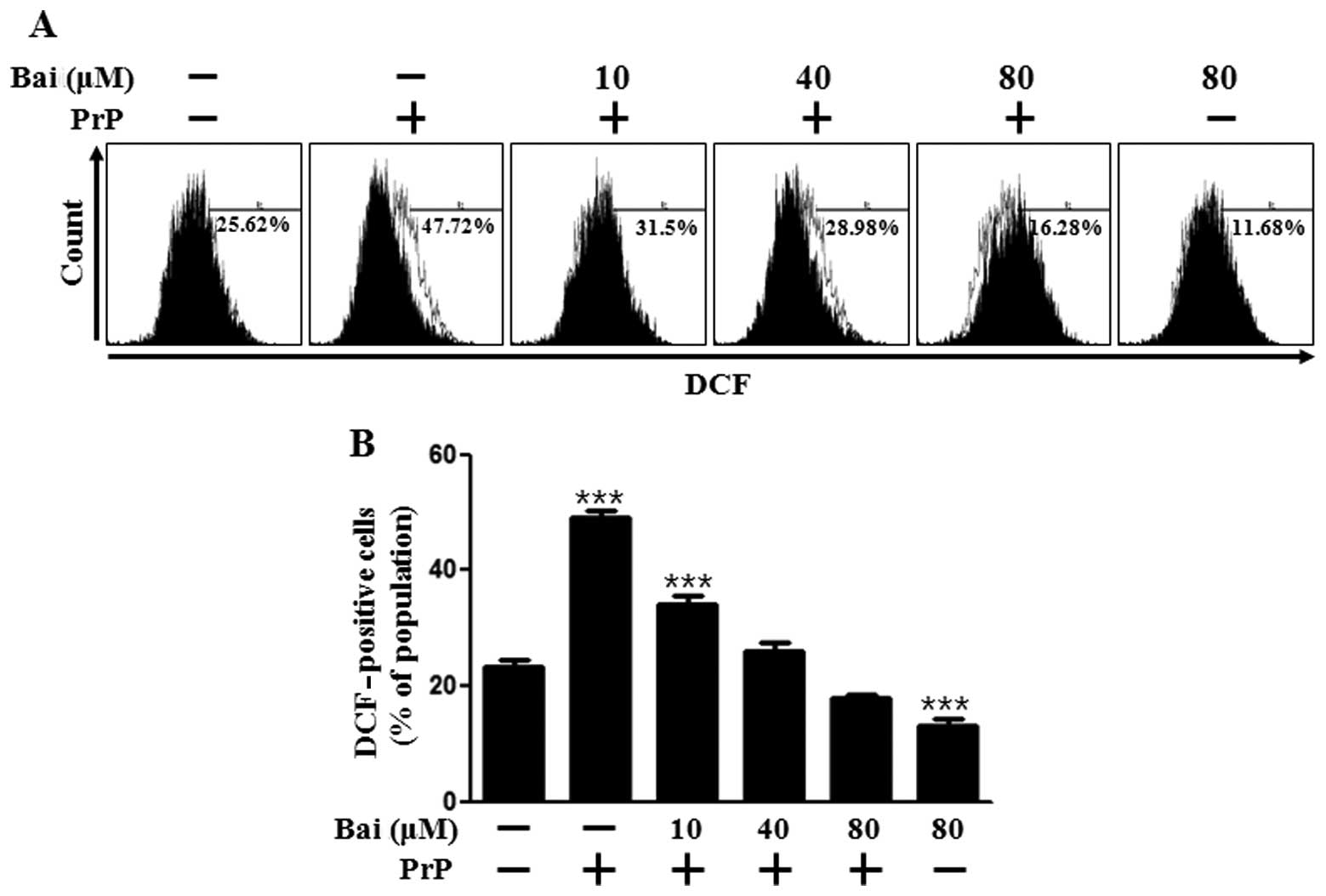
tretment with baicalein. As shown in Fig. 2A and B, DCFH-DA fluorescence
increased following treatment with PrP (106–126), whereas it was
inhibited by baicalein treatment in a dose-dependent manner. We
also assessed whether the protective effects of baicalein on PrP
(106–126)-induced neurotoxicity are related to the prevention of
mitochondrial dysfunction. The SH-SY5Y cells were pre-incubated
with 10 μm baicalein for 12 h and then exposed to 50
μm PrP (106–126). The PrP (106–126)-treated cells displayed
increased JC-1 monomers (58.2%), indicating low MTP values, whereas
baicalein treatment reduced PrP (106–126)-induced JC-1 monomers
(24.3%) in a dose-dependent manner, indicating high MTP values
(Fig. 2C). In accordance with
these results, the fluorescence microscopy images (Fig. 2D) showed cells with green
fluorescence (JC-1 monomer form) following treatment with PrP
(106–126), indicating lower MTP, whereas the control cells and
baicalein-treated cells displayed red fluorescence (JC-1 aggregate
form), indicating high MTP values. These data demonstrate that
baicalein reduces ROS production and attenuates mitochondrial
dysfunction induced by PrP.
Baicalein mediates survival and death
signals
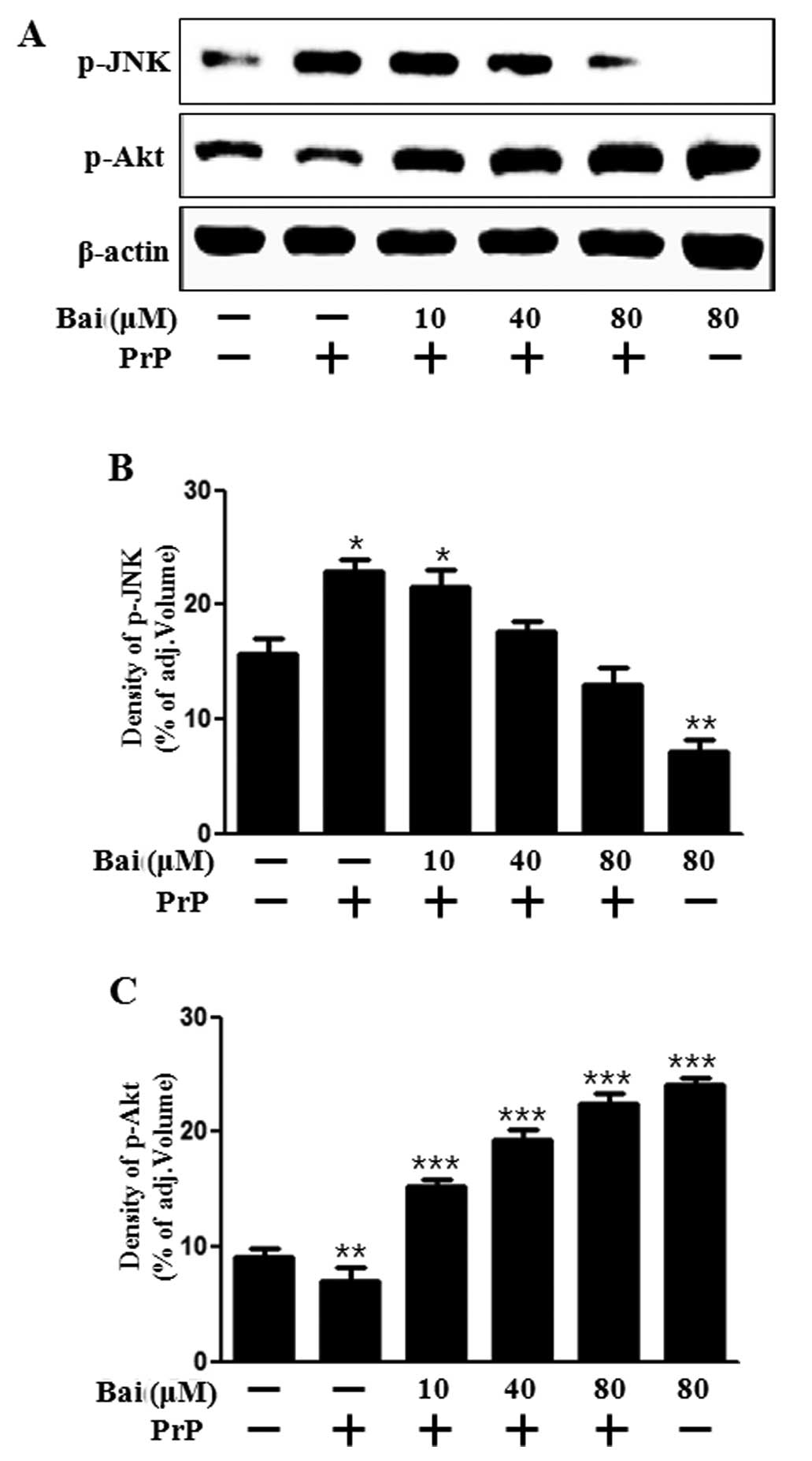
We then investigated whether baicalein exerts an
effect on cell growth and death signals. Akt promotes growth
factor-mediated cell survival both directly and indirectly. JNK is
activated by PrP (106–126)-induced apoptosis (27). In addition, baicalein attenuates
astroglial activation by downregulating the activation of JNK
(28). Therefore, in this study,
we examined Akt and JNK activation in neuronal cells following
treatment with baicalein and PrP (106–126). Our results
demonstrated that baicalein reversed the inhibition of Akt
activation induced by treatment with PrP (106–126) and blocked JNK
activation which was induced by PrP (106–126) (Fig. 3). These results suggest that
baicalein exerts a protective intracellular signal. It can thus be
hypothesized that the protective effects of baicalein may be
inhibited by an Akt inhibitor. This suggests that baicalein plays a
role similar to that of a JNK inhibitor under apoptotic conditions.
Taken together, our results suggest that treatment with baicalein
inhibits the PrP (106–126)-induced apoptosis of neuronal cells by
regulating JNK. Therefore, this was examined in the following
experiment.
Baicalein inhibits PrP (106–126)-induced
apoptosis by inactivating JNK
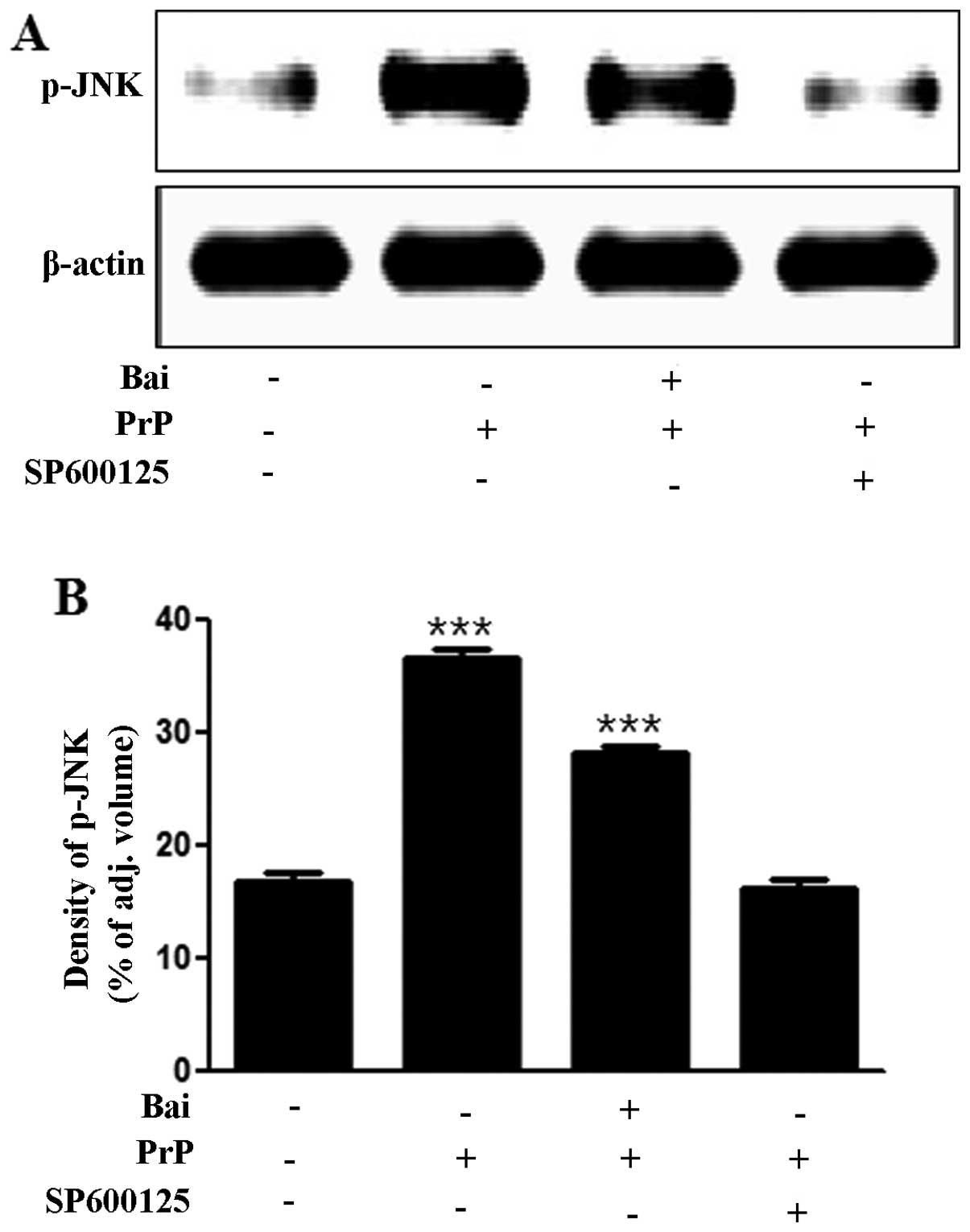
To investigate whether the inhibition of JNK exerts
neuroprotective effects against PrP (106–126)-induced
neurotoxicity, we analyzed the levels of JNK and cell viability
using a JNK inhibitor (SP600125). We found that the PrP
(106–126)-induced increase in JNK levels was inhibited by the JNK
inhibitor, SP600125, as shown by western blot and densitometric
analyses (Fig. 4A and B). We also
found that the JNK inhibitor (SP600125) blocked the PrP
(106–126)-induced neuronal apoptosis by inactivating JNK (Fig. 4C and D). The cells in the first
quadrant of the graph in Fig. 4C
signify the late apoptotic condition and those in the fourth
quadrant signify the early apoptotic condition. These results
strongly suggest that treatment with baicalein exerts
neuroprotective effects against neuronal cell apoptosis by
inactivating JNK.
Discussion
Baicalein, a flavonoid extracted from the
traditional Chinese herbal medicine, S. baicalensis Georgi
(Huang-qin), has been studied as a chemopreventive agent in
neuronal cell and brain models (28–30). In the present study, we
demonstrated that baicalein protected neuronal cells against PrP
(106–126)-induced neurotoxicity.
Certain studies have suggested that high levels of
ROS induce tissue injury, increase the formation of superoxide and
induce neurotoxicity (31,32).
As shown in the present study, treatment with baicalein attenuated
the PrP (106–126)-induced production of ROS and neurotoxicity in
dose-dependent manner (Figs. 1
and 2). These results indicate
that ROS are associated with neurodegeneration and that baicalein
protects against neurotoxicity induced by ROS. However, we were
unable to demonstrate whether the inhibition of ROS production
protects against PrP (106–126)-induced neurotoxicity. Further
research on ROS and neurodegeneration is clearly required.
The mitochondria are the main source of cellular
reactive oxygen, whose production further increases if the
mitochondria are damaged (33,34). The contribution of ROS-induced
mitochondrial dysfunction to the neurodegenerative process may
depend on the cell type involved (35). As the mitochondria are both the
main producers and targets of ROS, targeting mitochondrial
oxidative damage may be clinically useful in neurodegeneration. Our
data demonstrated that PrP (106–126) induced mitochondrial
dysfunction and that baicalein protected the neuronal cells against
mitochondrial damage (Fig. 2C and
D). Our results are fragmentary, but suggest that baicalein
protects neuronal cells against mitochondrial dysfunction and
neurotoxicity from ROS overproduction induced by PrP.
In addition, we determined whether baicalein affects
the mechanisms related to Akt and JNK signaling. Liu et al
(30) suggested that the
neuroprotective effects of baicalein involve the Akt pathway. Choi
et al (36) proposed that
baicalein protects hippocampal neuronal cells against apoptosis by
inhibiting JNK activation. As expected, the JNK inhibitor,
SP600125, protected the SK-N-SH cells against apoptotic death
(Fig. 4C). However, Akt
inhibitors (LY294002 and wortmannin) did not prevent the
neuroprotective effects of baicalein (data not shown). Akt and JNK
signals have been associated with baicalein, and our results
indicate that JNK plays a key role regulating PrP (106–126)-induced
neuronal apoptotic signals.
Several studies have suggested that JNK is
associated with the generation of ROS. Shen et al (37) demonstrated that the JNK signal is
a key modulator in cell death induced by ROS. It has also been
suggested that the ROS-mediated JNK activation is a critical
component deciding the fate of cells in response to various
stressful stimuli, which can be inhibited by pre-treatment with
antioxidants (38,39). Thus, further research on the
association between JNK and ROS is required.
In conclusion, our results indicate that baicalein
protects neuronal cells against PrP (106–126)-induced neurotoxicity
by inhibiting JNK activation. In addition, baicalein reduces PrP
(106–126)-induced mitochondrial dysfunction and ROS production.
These data may prove useful to future studies and suggest the
potential use of baicalein in the treatment of neurodegenerative
disorders.
Acknowledgments
The present study was supported by a grant from the
National Research Foundation of Korea (NRF), funded by the Korean
government (2013R1A2A2A01009614).
References
|
1
|
Peretz D, Williamson RA, Kaneko K, et al:
Antibodies inhibit prion propagation and clear cell cultures of
prion infectivity. Nature. 412:739–743. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Aguzzi A: Prion diseases of humans and
farm animals: epidemiology, genetics, and pathogenesis. J
Neurochem. 97:1726–1739. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Caughey B, Raymond GJ, Ernst D and Race
RE: N-terminal truncation of the scrapie-associated form of PrP by
lysosomal protease(s): implications regarding the site of
conversion of PrP to the protease-resistant state. J Virol.
65:6597–6603. 1991.PubMed/NCBI
|
|
4
|
Selvaggini C, De Gioia L, Cantu L, et al:
Molecular characteristics of a protease-resistant, amyloidogenic
and neurotoxic peptide homologous to residues 106–126 of the prion
protein. Biochem Biophys Res Commun. 194:1380–1386. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Forloni G, Angeretti N, Chiesa R, et al:
Neurotoxicity of a prion protein fragment. Nature. 362:543–546.
1993. View
Article : Google Scholar : PubMed/NCBI
|
|
6
|
Florio T, Grimaldi M, Scorziello A, et al:
Intracellular calcium rise through L-type calcium channels, as
molecular mechanism for prion protein fragment 106–126-induced
astroglial proliferation. Biochem Biophys Res Commun. 228:397–405.
1996. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
De Gioia L, Selvaggini C, Ghibaudi E, et
al: Conformational polymorphism of the amyloidogenic and neurotoxic
peptide homologous to residues 106–126 of the prion protein. J Biol
Chem. 269:7859–7862. 1994.PubMed/NCBI
|
|
8
|
Corsaro A, Thellung S, Villa V, et al:
Prion protein fragment 106–126 induces a p38 MAP kinase-dependent
apoptosis in SH-SY5Y neuroblastoma cells independently from the
amyloid fibril formation. Ann N Y Acad Sci. 1010:610–622. 2003.
View Article : Google Scholar
|
|
9
|
Halliwell B: Oxidative stress and
neurodegeneration: where are we now? J Neurochem. 97:1634–1658.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Jabs T: Reactive oxygen intermediates as
mediators of programmed cell death in plants and animals. Biochem
Pharmacol. 57:231–245. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Scherz-Shouval R, Shvets E, Fass E, Shorer
H, Gil L and Elazar Z: Reactive oxygen species are essential for
autophagy and specifically regulate the activity of Atg4. EMBO J.
26:1749–1760. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Borger E, Aitken L, Muirhead KE, et al:
Mitochondrial β-amyloid in Alzheimer’s disease. Biochem Soc Trans.
39:868–873. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Coskun P, Wyrembak J, Schriner SE, et al:
A mitochondrial etiology of Alzheimer and Parkinson disease.
Biochim Biophys Acta. 1820:553–564. 2012. View Article : Google Scholar :
|
|
14
|
Siskova Z, Mahad DJ, Pudney C, et al:
Morphological and functional abnormalities in mitochondria
associated with synaptic degeneration in prion disease. Am J
Pathol. 177:1411–1421. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
O’Donovan CN, Tobin D and Cotter TG: Prion
protein fragment PrP-(106–126) induces apoptosis via mitochondrial
disruption in human neuronal SH-SY5Y cells. J Biol Chem.
276:43516–43523. 2001. View Article : Google Scholar
|
|
16
|
Shang YZ, Gong MY, Zhou XX, Li ST and Wang
BY: Improving effects of SSF on memory deficits and pathological
changes of neural and immunological systems in senescent mice. Acta
Pharmacol Sin. 22:1078–1083. 2001.PubMed/NCBI
|
|
17
|
Shang Y, Cheng J, Qi J and Miao H:
Scutellaria flavonoid reduced memory dysfunction and neuronal
injury caused by permanent global ischemia in rats. Pharmacol
Biochem Behav. 82:67–73. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bochorakova H, Paulova H, Slanina J, Musil
P and Taborska E: Main flavonoids in the root of Scutellaria
baicalensis cultivated in Europe and their comparative antiradical
properties. Phytother Res. 17:640–644. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Heo HJ, Kim DO, Choi SJ, Shin DH and Lee
CY: Potent inhibitory effect of flavonoids in Scutellaria
baicalensis on amyloid beta protein-induced neurotoxicity. J Agric
Food Chem. 52:4128–4132. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Huang Y, Wong CM, Lau CW, et al:
Inhibition of nitric oxide/cyclic GMP-mediated relaxation by
purified flavonoids, baicalin and baicalein, in rat aortic rings.
Biochem Pharmacol. 67:787–794. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chen YC, Shen SC, Chen LG, Lee TJ and Yang
LL: Wogonin, baicalin, and baicalein inhibition of inducible nitric
oxide synthase and cyclooxygenase-2 gene expressions induced by
nitric oxide synthase inhibitors and lipopolysaccharide. Biochem
Pharmacol. 61:1417–1427. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chang WS, Lee YJ, Lu FJ and Chiang HC:
Inhibitory effects of flavonoids on xanthine oxidase. Anticancer
Res. 13:2165–2170. 1993.PubMed/NCBI
|
|
23
|
Kyo R, Nakahata N, Sakakibara I, Kubo M
and Ohizumi Y: Baicalin and baicalein, constituents of an important
medicinal plant, inhibit intracellular Ca2+ elevation by
reducing phospholipase C activity in C6 rat glioma cells. J Pharm
Pharmacol. 50:1179–1182. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Liao JF, Hung WY and Chen CF:
Anxiolytic-like effects of baicalein and baicalin in the Vogel
conflict test in mice. Eur J Pharmacol. 464:141–146. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yu X, He G and Du G: Neuroprotective
effect of baicalein in patients with Parkinson’s disease. Zhongguo
Zhong Yao Za Zhi. 37:421–425. 2012.in Chinese. PubMed/NCBI
|
|
26
|
Mu X, He GR, Yuan X, Li XX and Du GH:
Baicalein protects the brain against neuron impairments induced by
MPTP in C57BL/6 mice. Pharmacol Biochem Behav. 98:286–291. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Carimalo J, Cronier S, Petit G, et al:
Activation of the JNK-c-Jun pathway during the early phase of
neuronal apoptosis induced by PrP106–126 and prion infection. Eur J
Neurosci. 21:2311–2319. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lee E, Park HR, Ji ST, Lee Y and Lee J:
Baicalein attenuates astroglial activation in the
1-methyl-4-phenyl-1,2,3,4-tetrahydro-pyridine-induced Parkinson’s
disease model by downregulating the activations of nuclear
factor-kappaB, ERK, and JNK. J Neurosci Res. 92:130–139. 2014.
View Article : Google Scholar
|
|
29
|
Lee JH and Lee SR: The effect of baicalein
on hippocampal neuronal damage and metalloproteinase activity
following transient global cerebral ischaemia. Phytother Res.
26:1614–1619. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
30
|
Liu C, Wu J, Xu K, et al: Neuroprotection
by baicalein in ischemic brain injury involves PTEN/AKT pathway. J
Neurochem. 112:1500–1512. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kovacic P and Somanathan R: Redox
processes in neurode-generative disease involving reactive oxygen
species. Curr Neuropharmacol. 10:289–302. 2012. View Article : Google Scholar :
|
|
32
|
Chakraborty S, Bornhorst J, Nguyen TT and
Aschner M: Oxidative stress mechanisms underlying Parkinson’s
disease-associated neurodegeneration in C. elegans. Int J Mol Sci.
14:23103–23128. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Beal MF: Mitochondria take center stage in
aging and neurode-generation. Ann Neurol. 58:495–505. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Radi R, Cassina A and Hodara R: Nitric
oxide and peroxynitrite interactions with mitochondria. Biol Chem.
383:401–409. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Bolanos JP, Moro MA, Lizasoain I and
Almeida A: Mitochondria and reactive oxygen and nitrogen species in
neurological disorders and stroke: therapeutic implications. Adv
Drug Deliv Rev. 61:1299–1315. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Choi JH, Choi AY, Yoon H, et al: Baicalein
protects HT22 murine hippocampal neuronal cells against endoplasmic
reticulum stress-induced apoptosis through inhibition of reactive
oxygen species production and CHOP induction. Exp Mol Med.
42:811–822. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Shen HM and Liu ZG: JNK signaling pathway
is a key modulator in cell death mediated by reactive oxygen and
nitrogen species. Free Radic Biol Med. 40:928–939. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Sato M, Bagchi D, Tosaki A and Das DK:
Grape seed proanthocyanidin reduces cardiomyocyte apoptosis by
inhibiting ischemia/reperfusion-induced activation of JNK-1 and
C-JUN. Free Radic Biol Med. 31:729–737. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Yoshizumi M, Kogame T, Suzaki Y, et al:
Ebselen attenuates oxidative stress-induced apoptosis via the
inhibition of the c-Jun N-terminal kinase and activator protein-1
signalling pathway in PC12 cells. Br J Pharmacol. 136:1023–1032.
2002. View Article : Google Scholar : PubMed/NCBI
|