Introduction
The temporomandibular joint (TMJ) is the only
bilateral linkage joint in the human body, and it is characterized
by stability and mobility. Activities such as mastication,
swallowing, talking, and facial expressions are controlled by the
TMJ and the associated muscles. The condylar process is an
important developmental zone of the mandible, and the development
of the TMJ is closely related to functional stimulation and plays
an important role in oral-maxillary facial development. The growth
and development of chondrocytes within the TMJ are closely related
to functional mechanical stimulation. The TMJ disc is located
between the mandible and the skull and plays a critical role in
mandibular movements, functioning as a stress absorber (1). During various functional movements,
such as chewing and talking, or some dysfunctional movements such
as bruxism, the disc is continuously subjected to biomechanical
loading in the form of tensile, compressive, and shear stress from
different directions (2).
Moreover, during orthodontic treatment, patients are often
subjected to intermaxillary traction. The cartilage matrix as well
as chondrocytes are responsible for responding to mechanical stress
and maintaining the function of articular joints. During clinical
treatment, intermaxillary traction is highly effective for
achieving tooth movement, but may also cause a series of undesired
results related to the force-induced response, such as
temporomandibular disorders (3).
One study reported that endochondral bone formation is reduced,
with restriction of mandibular movements, during the chondylar
cartilage development process (4). Mandibular condylar chondrocytes
(MCCs) are the main type of chondylar chondrocytes, and they play
an important role in maintaining cartilage homeostasis. MCC
proliferation, differentiation, and apoptosis directly influence
the mechanical characteristics and functional status of the joint.
However, despite advances in our understanding of the molecular
mechanisms of cartilage development and repair, the mechanical
signals that regulate cartilage repair remain poorly understood
(5–7).
A previous study by Kronenberg (8) found that several signaling pathways
are activated during the development and growth of chondrocytes,
including the Sox 9, transforming growth factor (TGF)-β, and Wnt
signaling pathways. Moreover, a study by Maruyama et al
(9) on systemic skeletal diseases
and metabolic diseases focused on the Wnt signaling pathway, and it
has been confirmed that the Wnt signaling pathway is associated
with biological force stimulation (10). The Wnt signaling pathway plays an
important role in the regulation of chondrogenic differentiation
and hypertrophy (11,12). Three distinct intracellular Wnt
signaling cascades are well known at present: the Wnt/β-catenin
pathway (the canonical Wnt signaling pathway), the c-Jun N-terminal
kinase (JNK) pathway, and the Wnt/Ca2+ pathway. The
Wnt/β-catenin pathway is mediated by β-catenin, which accumulates
in the cytoplasm in the presence of Wnt and is then transferred
into the nucleus. Glycogen synthase kinase-3β (GSK-3β) acts as a
critical negative control factor in the Wnt signaling pathway. The
phosphorylation of GSK-3β results in GSK-3β inactivation and blocks
the ability of GSK-3β to phosphorylate β-catenin, thereby
activating the Wnt/β-catenin signaling pathway. Therefore, we
hypothesized that after mechanical force is applied to
chondrocytes, GSK-3β is phosphorylated and the Wnt/β-catenin
signaling pathway is activated, triggering chondrogenesis among
chondrocytes.
In the present study, the ATDC5 cell line was
selected and cultured based on its chondrogenic potential in the
presence of insulin. ATDC5 cells are cells isolated from the
feeder-independent teratocarcinoma stem cell line AT805 (13). Compared with primary MCCs, the
advantages of the ATDC5 cell line are greater stability, ease of
culture and expansion, and greater humanization. The cells were
subjected to mechanical stretching using a pressure-operated
instrument. Subsequently, immunofluorescence, western blot
analysis, quantitative PCR (qPCR), and other experimental methods
were used to observe activation of the Wnt/β-catenin signaling
pathway in order to determine the effect of mechanical loading on
β-catenin signaling in chondrocytes. In order to observe cartilage
reconstruction after different durations of orthodontic traction,
cells were subjected to mechanical loading for different amounts of
time.
Materials and methods
Cell culture
ATDC5 cells (Riken Cell Bank, Tsukuba, Japan) were
maintained in a log growth phase in hybrid Dulbecco's modified
Eagle's medium (DMEM; Thermo Scientific Hyclone, Waltham, MA, USA)
containing 10% fetal bovine serum (FBS) and 10 µg/ml
insulin-transferrin-selenium (ITS) (both from Gibco, Grand Island,
NY, USA). Cells were cultured at 37°C in a humidified atmosphere of
5% CO2 in air for 21 days. Cells were initially seeded
at a density of 6×104 cells/well in 6-well plates. The
medium was replaced every 2 days. Alcian blue staining and qPCR
analysis were used to confirm successful induction of
chondrogenesis. The sequences of primers used in the present study
for β-actin, collagen II, and aggrecan were as follows: β-actin
sense, 5′-ATGTACGTAGCCATCCAG-3′ and antisense,
5′-GAGGTAGTCTGTCAGGTC-3′; collagen II sense,
5′-CCAGAACATCACCTACCACTGTAA-3′ and anti-sense,
5′-GCCCTCATCTCTACATCATTGGA-3′; and aggrecan sense,
5′-CCATGTGTGGGTGACAAAGACAG-3′ and antisense,
5′-TCCACGTAGCAGTAGACATCATAGG-3′. These sequences were designed by
Takara Bio Co., Ltd. (Kyoto, Japan).
Mechanical loading
Cells were subjected to mechanical stretching using
a pressure-operated instrument (Flexercell Strain Instrument;
Flexcell Corp., Hillsborough, NC, USA). Chondrocytic cells were
plated in 6-well elastic-bottomed culture plates (Flex II culture
plates; Flexcell Corp.), in which the bottomed surfaces of the
plates were coated with collagen II. When the cells reached
confluence, 4 plates were fixed on a Flexercell strain unit
(Flexercell Corp.) and cyclic mechanical tension was applied.
Culture plate bottoms were deformed to a known percentage of
elongation, which was maximal at the edge and decreased toward the
center. The strain was transferred to the cultured cells. In our
experiments, the culture plate bottoms were deformed to 12%
elongation in a 0.05 Hz cyclic (5 sec on, 5 sec off) manner. The
cells were stimulated for 1, 2, 4, or 8 h, and control cells were
not exposed to mechanical loading.
Protein extraction and western blot
analysis
After cyclic stretch loading, cells were washed once
with ice-cold phosphate-buffered saline (PBS) immediately and lysed
in 500 µl sodium dodecyl sulfate (SDS) buffer [125 mM
Tris-HCL (pH 6.8), 1% w/v SDS, 8.5% glycerol) per well. Samples
were heated to 95°C for 5 min. Aliquots of 10 µl protein
were resolved by SDS-PAGE (10% resolving gel), transferred to
nitrocellulose membranes (Bio-Rad Laboratories, Hercules, CA, USA),
and blocked with blocking buffer [10 mM Tris-HCl (pH 7.6), 150 mM
NaCl, 3% w/v BSA, 0.1% Tween-20] for 1 h at room temperature. After
washing three times for 5 min each with Tris-buffered saline/Tween
[10 mM Tris-HCl (pH 7.6), 150 mM NaCl, 0.1% Tween-20], cell
membranes were incubated with primary antibodies overnight at 4°C
with gentle shaking. The membranes were washed three times for 5
min with Tris-buffered saline/Tween 20 and incubated with
horseradish peroxidase-conjugated goat anti-rabbit secondary
antibodies (1:1,000; Perbio Science, Erembodegem, Belgium) for 1 h
at room temperature. The primary antibodies were against active
β-catenin (clone 8E7, 1:1,000; Millipore, Billerica, MA, USA),
phospho-GSK-3β (Ser9, 1:1,000; 9331S) and GSK-3β (1:1,000; 9237S)
(both from Cell Signaling Technology, Danvers, MA, USA). To
demonstrate equal protein loading, specific β-actin antibodies
(1:1,000; 4967; Cell Signaling Technology) were used.
RNA extraction and qPCR
After the cells were exposed to cyclic stretch
loading for 1, 2, 4, or 8 h, total RNA was extracted using TRIzol
(Gibco) according to the manufacturer's instructions, and the
concentration and purity were subsequently assayed by
spectrophotometry. First-strand cDNA synthesis was performed using
the High-Capacity cDNA Archive kit (Applied Biosystems, Foster
City, CA, USA) according to the manufacturer's instructions. The
sequences of primers for β-actin and collagen II were the same as
those which were listed above. The sequences of primers for Wnt3a,
disheveled homolog 1 (Dvl-1), and T cell-specific transcription
factor (Tcf)-1 used in the present study were as follows: Wnt3a
sense, 5′-CAGCTGTGAAGTGAAGAC-3′ and antisense,
5′-CTCGTAGTAGACCAGGTC-3′; Dvl-1 sense, 5′-ATC AACCAATGTCTTCAG-3′
and antisense, 5′-AATGTCAA GCCAATATCC-3′; and Tcf-1 sense,
5′-CTACGAACATTTCAGCAG-3′ and antisense, 5′-GTCTATATCCGCAGGAAG-3′.
The sequences for all primers were designed by Takara Bio Co., Ltd.
PCR was performed with the ABI Prism 7000 Sequence Detection system
(Applied Biosystems). Relative quantification was carried out using
the ΔΔCt method by taking the difference (ΔCt) between the ΔCt of
β-actin and the ΔCt of each transcript and computing the ΔΔCt.
Triplicate reactions were carried out in three separate
experiments.
Immunofluorescence staining
After culture with or without cyclic stretch
loading, cells which adhered to the surface of elastic-bottomed
plates were rinsed twice with PBS, fixed with 4% paraformaldehyde
for 30 min, and rinsed twice with PBS again. The elastic bottoms of
the plates were cut into 8–10 pieces each, and then cells were
incubated with primary antibody (mouse anti-active β-catenin, clone
8E7, 1:100; Millipore) for 1 h. The secondary antibody was
fluorescein isothiocyanate-conjugated rabbit anti-mouse
immunoglobulin G (1:200; 7074S; Cell Signaling Technology), and the
stained cells were viewed through a laser scanning confocal
microscope (FV1200; Olympus, Tokyo, Japan). We randomly selected
six visual fields for each point and counted both the number of
cells exhibiting nuclear translocation of β-catenin and the total
number of cells in each field of view.
Treatment of the cells with Wnt/β-catenin
signaling pathway inhibitory protein Dickkopf-related protein 1
(DKK-1)
Single-cell suspensions of the chondrocytes at
logarithmic growth phase were prepared, and the cells were
transferred to 6-well plates. Cells were incubated with 100 ng/ml
mouse recombinant Dickkopf-related protein 1 (R&D Systems,
Minneapolis, MN, USA) in DMEM containing 5% fetal bovine serum for
24 h to inhibit the Wnt/β-catenin pathway. MTT,
[3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide]
assay was carried out on days 1, 3, 5, and 7 in cultures to
evaluate the efficiency of DKK-1. Cells were plated at a density of
2×103 cells/well in 96-well plates for 4 h at 37°C. The
supernatant was then removed and 150 µl dimethyl sulfoxide
(DMSO) was added, followed by 10 min of oscillation. The optical
density (OD) values for each well were measured
spectrophotometrically at 490 nm, and the tests were repeated 5
times. Control cells were not exposed to DKK-1.
To examine the effect of DKK-1 treatment on
chondrocytes exposed to mechanical loading, the culture plate
bottoms were deformed to 12% elongation in a 0.05 Hz cyclic (5 sec
on, 5 sec off) manner, as described above, for 1, 2, 4, or 8 h. The
control group was not exposed to loading. Immunofluorescence
staining was used to track β-catenin localization in chondro-cytes,
as also described above, after treatment with DKK-1, and qPCR
analysis was used to detect the corresponding mRNA expression of
Wnt3a, Dvl-1, Tcf-1 and collagen II, as described above. The
results for cells treated with DKK-1 were compared to those for
cells not treated with DKK-1.
Statistical analysis
All the experiments were repeated three times, and
all statistical analyses were performed using SPSS version 16.0
software (SPSS Inc., Chicago, IL, USA). The student's t-test was
employed to evaluate the significance of differences among groups,
and a P-value <0.05 was considered to indicate a statistically
significant difference.
Results
Chondrogenic differentiation and
cartilage nodule formation in ATDC5 cells
ATDC5 cells can be induced to differentiate into
chondrocytes in the presence of insulin (13), and a previous study has
demonstrated that in the presence of 10 µg/ml insulin, ATDC5
cells form cartilage nodules through a cellular condensation
process that gives rise to proliferating chondrocytes (14). In the present study, the size and
number of cartilage nodules within ATDC5 cultures increased each
day over 3 weeks in culture after treatment with ITS. Increasing
intensity of Alcian blue staining of sulfated glycosaminoglycan,
which is secreted by chrondrocytes, over 3 weeks in culture
confirmed chondrogenic differentiation of the ATDC5 cells (Fig. 1A).
Collagen type II and aggrecan are the main
components of the extracellular matrix in cartilage. To further
confirm that ATDC5 cells differentiated into chondrocytes in the
present study, we analyzed the expression of chondrogenic marker
genes including collagen II and aggrecan by qPCR (Fig. 1B) upon culture of the cells in
medium containing insulin. Collagen II and aggrecan expression
increased from day 1 to 21, indicating that the undifferentiated
ATDC5 cells had differentiated into proliferative chondrocytes.
Mechanical loading induces nuclear
translocation of β-catenin in the differentiated chondrocytes
It has previously been noted that the Wnt/β-catenin
signaling pathway is mediated by β-catenin, which accumulates in
the cytoplasm and translocates into the nucleus to activate Wnt
signaling (15). To determine
whether mechanical loading activates Wnt/β-catenin signaling,
immunofluorescence staining was used to identify dephosphorylated
β-catenin in cells at 1, 2, 4, and 8 h after stretch loading.
Compared to control cells which did not receive mechanical loading,
the nuclei of cells subjected to force loading for 1 h showed
significantly increased staining for dephosphorylated β-catenin.
Nuclear translocation of β-catenin occurred after 1 h, and the
amount of nuclear staining in cells subjected to stretch loading
for 8 h was comparable to that in the control group (Fig. 2B). These results indicate that
translocation of β-catenin is a rapid process.
Mechanical loading causes the
phosphorylation of GSK-3β and the accumulation of β-catenin
GSK-3β is a key component of the Wnt/β-catenin
signaling pathway, and its activity is inhibited by phosphorylation
at serine (16). GSK-3β is
normally inhibited in the Wnt pathway, where its primary target is
β-catenin. Phosphorylation of GSK-3β inhibits its ability to
phosphorylate β-catenin, which marks β-catenin for degradation
(17). Therefore, we hypothesized
that upon mechanical loading, GSK-3β in the chondrocytes would be
phosphorylated, thereby activating the Wnt/β-catenin signaling
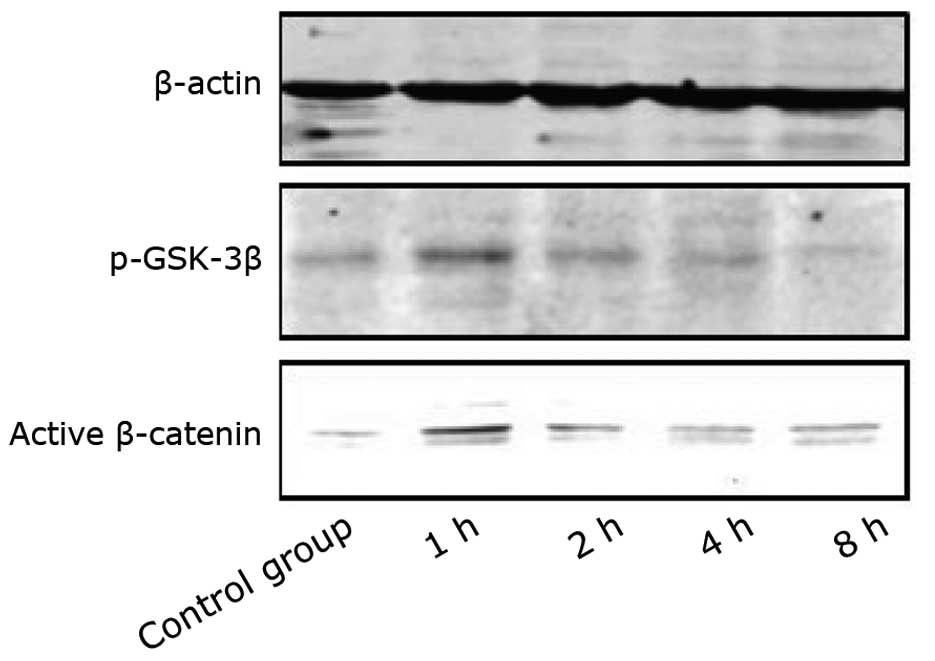
pathway. Western blot analysis showed that the level of p-GSK-3β
increased with the application of stretch loading, and this
increase was accompanied by an increase in the level of
dephosphorylated β-catenin at 1 and 2 h after application of
mechanical loading (Fig. 3).
Wnt3a, Dvl-1, Tcf-1, and collagen II mRNA
expression in chondrocytes subjected to mechanical loading
To investigate the role of the Wnt/β-catenin pathway
in chondrogenic differentiation, we simultaneously examined the
expression of key proteins in the Wnt/β-catenin pathway (Wnt3a,
Dvl-1, and Tcf-1) as well as collagen II, as a marker of
differentiation. After 2 h of mechanical loading, the expression of
Wnt3a mRNA was significantly higher than that in the control group,
whereas after 4 h of mechanical loading, Wnt3a mRNA expression was
almost equal to that in the 8 h group. Wnt3a expression increased
initially and then decreased. Dvl mRNA expression showed the same
trend. However, the expression of Tcf-1 mRNA increased and remained
at a high level almost equivalent to that after 1 h over the entire
8 h of mechanical loading. Collagen II expression followed a
similar trend as Tcf-1, increasing and then remaining at a high
level, but decreased at 8 h (Fig.
4).
Effects of the inhibitor DKK-1 on
Wnt/β-catenin pathway activation in chondrocytes subjected to
mechanical loading
Wnt signaling is tightly regulated by members of
several families of secreted antagonists (18). DKK-1 is a natural regulator of the
Wnt/β-catenin signaling pathway that has the ability to
specifically block the pathway (19,20). In the present study, we applied
DKK-1 to differentiated ATDC5 cells to further examine the role of
the Wnt/β-catenin signaling pathway in these cells when subjected
to mechanical loading. According to the results of the MTT assay,
chondrocytes showed restricted growth after treatment with DKK-1
(Fig. 5). Immunofluorescence
staining demonstrated an obvious decrease in the amount of
dephosphorylated β-catenin in the nuclei of cells subjected to
stretch loading and exposed to DKK-1 compared to cells subjected to
stretch loading only (Fig. 6). In
accordance with these results, western blot analysis also
demonstrated that exposure to DKK-1 generally decreased the level
of dephosphorylated β-catenin in whole cell lysate. The levels of
p-GSK-3β were increased slightly after mechanical loading and also
DKK-1 treatment (Figure 7),
compared with levels in cells not exposed to DKK-1 (Figure 5). These changes in p-GSK-3β
levels correlated with those in levels of dephosphorylated
β-catenin at 1 h (Fig. 7). The
expression of Wnt3a, Dvl-1, Tcf-1, and collagen II mRNA were also
suppressed by DKK-1 (Fig. 8).
Discussion
Mechanical loading is an important regulator of
chondrocyte function, but the mechanisms by which mechanical
stimulation influences intracellular signaling are still not fully
understood. In orthodontics, more attention is paid to the TMJ
cartilage, and therapeutic corrections are applied to stimulate
cell and tissue remodeling by manipulation using mechanical forces
(21). In the present study, we
found that the Wnt/β-catenin signaling pathway is activated in
chondrocytes upon application of mechanical loading.
GSK-3β is an important negative control factor in
the Wnt signaling pathway, and phosphorylation of GSK-3β
inactivates GSK-3β and activates the Wnt/β-catenin signaling
pathway (18). Thus, we
hypothesized that after mechanical force acts upon chondrocytes,
GSK-3β is phosphorylated to activate the Wnt/β-catenin signaling
pathway and trigger chondrogenesis. In our study, chondrocytes were
exposed to a 12% cycle mechanical stretching for 1, 2, 4, and 8 h,
which simulated orthodontic traction. Via immunofluorescence
staining, we observed that β-catenin translocated from the
cytoplasm to the nucleus after 1 h of mechanical loading,
indicating activation of the Wnt/β-catenin signaling pathway.
However, by 8 h of mechanical loading, the proportion of cells
demonstrating nuclear translocation of β-catenin had declined,
indicating rapid changes in the activity of this protein. More
detailed analyses of these protein events are necessary to improve
our understanding of cartilage physiology and pathology.
GSK-3β constitutively phosphorylates the N-terminus
of β-catenin cyclin-dependent kinase at Ser, which marks β-catenin
for degradation. When GSK-3β is phosphorylated at Ser, β-catenin
accumulates in the cytoplasm before trans-locating to the nucleus
to activate the Wnt/β-catenin signaling pathway (18). According to western blot analysis,
active dephosphorylated β-catenin and p-GSK-3β protein expression
increased in chondrocytes in response to mechanical strain,
indicating activation of the Wnt/β-catenin signaling pathway as
well as simultaneous inactivation of GSK-3β by mechanical
loading.
The Wnt family proteins play important roles in
patterning and cell fate determination; Wnt4 and Wnt9a are
expressed in the developing joints (22). Wnt3a promotes chondrogenic
differentiation but delays hypertrophy (11,23). The Wnt family binds to a complex
consisting of the Frizzled family of transmembrane receptors and
the lipoprotein receptor-related protein (LRP) family of
co-receptors (24). Wnt3a is
thought to be a 'canonical' Wnt protein, signaling through a
β-catenin-dependent signaling pathway. In the absence of the Wnt
signal, Tcf acts as a repressor of Wnt target genes by forming a
complex with Groucho (25). When
β-catenin translocates to the nucleus, β-catenin interacts with
transcription factors such as Tcf to affect the transcription of
target genes (26). It has
previously been noted that the physiological dynamic load induces
the synthesis of collagen II, and that the absence of this load
leads to a reduction in collagen II synthesis (27). In the present study, qPCR analysis
was used to evaluate the mRNA expression of Wnt3a, Dvl-1, Tcf-1,
and collagen II in chondrocytes exposed to mechanical strain. The
increased expression of these key proteins in the Wnt/β-catenin
signaling pathway suggests that mechanical loading induced
activation of β-catenin in chondrocytes.
DKK-1 has been noted to specifically affect the
canonical Wnt signaling pathway by binding and modulating Wnt
coreceptors of LRP5/6 (28).
Therefore, we used DKK-1 to inhibit mechanical loading-induced
dephosphorylation of β-catenin, and the results showed that
treatment with DKK-1 generally decreased the levels of
dephosphorylated β-catenin and p-GSK-3β in whole cell lysates. The
expression levels of Wnt3a, Dvl-1, Tcf-1, and collagen II mRNA were
also suppressed with addition of DKK-1 but were still increased and
then declined by exposure to mechanical loading. Thus, the
mechanical loading-induced activation of β-catenin in chondrocytes
is not only mediated by Wnt/β-catenin receptors. This finding is in
agreement with those of previous studies on periodontal ligament
cells and osteoblasts (17,29), but the mechanism underlying
phosphorylation of GSK-3β requires further research.
It is well known that physiological dynamic loads
induce the synthesis of collagen II, whereas a static load has the
opposite effect (10,30). The increase in collagen II
expression upon the application of mechanical strain in the present
study further confirms the close association between mechanical
stain and collagen II production by chondrocytes. In addition, the
simultaneous increases in GSK-3β phosphorylation and Wnt/β-catenin
signaling pathway activation indicate that the Wnt/β-catenin
signaling pathway plays an important role in the process of
collagen II upregulation induced by mechanical loading. Therefore,
we suggest that the Wnt/β-catenin signaling pathway represents a
new therapeutic target for inducing cartilage formation in the
field of regenerative medicine. In the field of oral medicine,
specifically, these findings should be applied to the development
of novel strategies for TMJ reconstruction and regeneration, which
are necessary for the treatment of TMJ disorders and to guide
orthodontic treatments.
In conclusion, the present research first
demonstrated that mechanical loading induced the translocation of
active β-catenin from the cytoplasm to the nucleus in chondrocytes.
Thus, we suggest that β-catenin plays an important role in the
general response of chondrocytes to mechanical strain. Overall, our
results indicate that the Wnt/β-catenin signaling pathway is
functional and activated by mechanical strain in chondrocytes.
Abbreviations:
|
DKK-1
|
Dickkopf-related protein 1
|
|
DMSO
|
dimethyl sulfoxide
|
|
GSK-3β
|
glycogen synthase kinase-3β
|
|
LRP5/6
|
lipoprotein receptor-related protein
5/6
|
|
TMJ
|
temporomandibular joint
|
|
MCC
|
mandibular condylar chondrocyte
|
|
MTT
|
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
|
References
|
1
|
Kinzinger G, Kober C and Diedrich P:
Topography and morphology of the mandibular condyle during fixed
functional orthopedic treatment - a magnetic resonance imaging
study. J Orofac Orthop. 68:124–147. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bewyer DC: Biomechanical and physiologic
processes leading to internal derangement with adhesion. J
Craniomandib Disord. 3:44–49. 1989.PubMed/NCBI
|
|
3
|
Meikle MC: Remodeling the dentofacial
skeleton: the biological basis for orthodontics and dentofacial
orthopedics. J Dent Res. 86:12–24. 2007. View Article : Google Scholar
|
|
4
|
Habib H, Hatta T, Udagawa J, Zhang L,
Yoshimura Y and Otani H: Fetal jaw movement affects condylar
cartilage development. J Dent Res. 84:474–479. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Madhavan S, Anghelina M, Rath-Deschner B,
Wypasek E, John A, Deschner J, Piesco N and Agarwal S:
Biomechanical signals exert sustained attenuation of
proinflammatory gene induction in articular chondrocytes.
Osteoarthritis Cartilage. 14:1023–1032. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ker RF, Wang XT and Pike AV: Fatigue
quality of mammalian tendons. J Exp Biol. 203:1317–1327.
2000.PubMed/NCBI
|
|
7
|
Kuboki T, Shinoda M, Orsini MG and
Yamashita A: Viscoelastic properties of the pig temporomandibular
joint articular soft tissues of the condyle and disc. J Dent Res.
76:1760–1769. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kronenberg HM: Developmental regulation of
the growth plate. Nature. 423:332–336. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Maruyama T, Mirando AJ, Deng CX and Hsu W:
The balance of WNT and FGF signaling influences mesenchymal stem
cell fate during skeletal development. Sci Signal.
3:ra402010.PubMed/NCBI
|
|
10
|
Larsson T, Aspden RM and Heinegård D:
Effects of mechanical load on cartilage matrix biosynthesis in
vitro. Matrix. 11:388–394. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Church V, Nohno T, Linker C, Marcelle C
and Francis-West P: Wnt regulation of chondrocyte differentiation.
J Cell Sci. 115:4809–4818. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chimal-Monroy J, Montero JA, Gañan Y,
Macias D, Garcia-Porrero JA and Hurle JM: Comparative analysis of
the expression and regulation of Wnt5a, Fz4, and Frzb1 during digit
formation and in micromass cultures. Dev Dyn. 224:314–320. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Atsumi T, Miwa Y, Kimata K and Ikawa Y: A
chondrogenic cell line derived from a differentiating culture of
AT805 teratocarcinoma cells. Cell Differ Dev. 30:109–116. 1990.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Shukunami C, Shigeno C, Atsumi T, Ishizeki
K, Suzuki F and Hiraki Y: Chondrogenic differentiation of clonal
mouse embryonic cell line ATDC5 in vitro: differentiation-dependent
gene expression of parathyroid hormone (PTH)/PTH-related peptide
receptor. J Cell Biol. 133:457–468. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Saito-Diaz K, Chen TW, Wang X, Thorne CA,
Wallace HA, Page-McCaw A and Lee E: The way Wnt works: components
and mechanism. Growth Factors. 31:1–31. 2013. View Article : Google Scholar :
|
|
16
|
Gould TD and Manji HK: Glycogen synthase
kinase-3: a putative molecular target for lithium mimetic drugs.
Neuropsychopharmacology. 30:1223–1237. 2005.PubMed/NCBI
|
|
17
|
Case N, Ma M, Sen B, Xie Z, Gross TS and
Rubin J: Beta-catenin levels influence rapid mechanical responses
in osteoblasts. J Biol Chem. 283:29196–29205. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Liu F, Kohlmeier S and Wang CY: Wnt
signaling and skeletal development. Cell Signal. 20:999–1009. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Aguilera O, Fraga MF, Ballestar E, Paz MF,
Herranz M, Espada J, García JM, Muñoz A, Esteller M and
González-Sancho JM: Epigenetic inactivation of the Wnt antagonist
DICKKOPF-1 (DKK-1) gene in human colorectal cancer. Oncogene.
25:4116–4121. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
González-Sancho JM, Aguilera O, García JM,
Pendás-Franco N, Peña C, Cal S, García de Herreros A, Bonilla F and
Muñoz A: The Wnt antagonist DICKKOPF-1 gene is a downstream target
of beta-catenin/TCF and is downregulated in human colon cancer.
Oncogene. 24:1098–1103. 2005. View Article : Google Scholar
|
|
21
|
Alanen P: Occlusion and temporomandibular
disorders (TMD): still unsolved question? J Dent Res. 81:518–519.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hartmann C and Tabin CJ: Dual roles of Wnt
signaling during chondrogenesis in the chicken limb. Development.
127:3141–3159. 2000.PubMed/NCBI
|
|
23
|
Rudnicki JA and Brown AM: Inhibition of
chondrogenesis by Wnt gene expression in vivo and in vitro. Dev
Biol. 185:104–118. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yano F, Kugimiya F, Ohba S, Ikeda T,
Chikuda H, Ogasawara T, Ogata N, Takato T, Nakamura K, Kawaguchi H
and Chung UI: The canonical Wnt signaling pathway promotes
chondrocyte differentiation in a Sox9-dependent manner. Biochem
Biophys Res Commun. 333:1300–1308. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Brannon M, Gomperts M, Sumoy L, Moon RT
and Kimelman D: A beta-catenin/XTcf-3 complex binds to the siamois
promoter to regulate dorsal axis specification in Xenopus. Genes
Dev. 11:2359–2370. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
van de Wetering M, Cavallo R, Dooijes D,
van Beest M, van Es J, Loureiro J, Ypma A, Hursh D, Jones T,
Bejsovec A, et al: Armadillo coactivates transcription driven by
the product of the Drosophila segment polarity gene dTCF. Cell.
88:789–799. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ratcliffe A, Billingham ME, Saed-Nejad F,
Muir H and Hardingham TE: Increased release of matrix components
from articular cartilage in experimental canine osteoarthritis. J
Orthop Res. 10:350–358. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Krupnik VE, Sharp JD, Jiang C, Robison K,
Chickering TW, Amaravadi L, Brown DE, Guyot D, Mays G, Leiby K, et
al: Functional and structural diversity of the human Dickkopf gene
family. Gene. 238:301–313. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Premaraj S, Souza I and Premaraj T:
Mechanical loading activates β-catenin signaling in periodontal
ligament cells. Angle Orthod. 81:592–599. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Rizkalla G, Reiner A, Bogoch E and Poole
AR: Studies of the articular cartilage proteoglycan aggrecan in
health and osteoarthritis. Evidence for molecular heterogeneity and
extensive molecular changes in disease. J Clin Invest.
90:2268–2277. 1992. View Article : Google Scholar : PubMed/NCBI
|