Introduction
The repair of bone defects resulting from tumors,
congenital malformations and surgical resections remains a
challenge that is being addressed by advanced bone tissue
engineering approaches (1,2). A
combined approach for treating large bone defects including the use
of cell seeding techniques, growth factors and biomaterials further
enhances the success of cell therapy (3,4).
The interactions between various growth factors involved in bone
healing and remodeling has been extensively studied in recent
years. The role of transforming growth factor-β1 (TGF-β1) in normal
human fracture healing has been demonstrated in a previous study
(5). TGF-β1 is known to induce
the differentiation and proliferation of osteoblasts and bone
marrow-derived mesenchymal stem cells (BMSCs), and also to
stimulate the appearance of extracellular matrix (ECM) proteins
in vitro (6,7). TGF-β1 has been widely used to
promote the osteogenesis of BMSCs in a variety of in vitro
culture systems by continuously supplying it in the medium for
>4 weeks (8–10). Thus, the controlled administration
of TGF-β1 may represent an emerging tissue engineering technology
that modulates cellular responses to encourage bone regeneration at
the site of a skeletal defect (11,12).
BMSCs are multipotent progenitor cells that have the
ability to differentiate into mesenchymal lineages, including
osteoblasts, chondrocytes and adipocytes (13). The differentiation and function of
BMSCs is regulated by TGF-β1. For example, TGF-β1 stimulates the
proliferation of BMSCs and promotes their differentiation into
chondrocytes (14). TGF-β1
promotes chondrogenesis as well as the early phase of osteogenesis;
however, in the absence of bone morphogenetic proteins (BMPs),
TGF-β1 is unable to promote the differentiation of BMSCs into
osteoblasts (15). Thus, in the
present study, BMSCs were induced into osteoblast-like cells
together with osteogenic medium prior to transplantation.
Osteoblast-like cells derived from BMSCs share similar
characteristics with osteoblasts. The present study evaluated the
effects of BMSCs and of osteoblast-like cells differentiated from
BMSCs, on TGF-β1-silk fibroin-chitosan (TGF-β1-SF-CS) scaffolds and
silk fibroin-chitosan (SF-CS) scaffolds, as well as analyzing the
effects of TGF-β1 in SF-CS scaffolds and in vitro
biocompatibility for the first time, to the best of our
knowledge.
Both silk fibroin (SF) and chitosan (CS) are natural
biomaterials with good biocompatibility which are available in
abundance. It is, however, difficult to prepare a pure SF scaffold
with a porous structure, and SF has poor anticoagulant properties.
In addition, a pure CS scaffold degrades relatively quickly, and is
unstable in aqueous solution (16–18). In our previous study (19), we blended SF and CS into a
three-dimensional (3D) scaffold with unique chemical, structural
and mechanical properties, which has potential applications in bone
tissue engineering and regenerative medicine. Previously, it was
found that the the most appropriate proportion between CS and SF is
5:5 for bone tissue engineering (20). Our current study at our laboratory
is examining soluble growth factor supplementation schemes in order
to enhance the bioactivity of bone cells for bone tissue
engineering applications.
The optimal strategy for delivering growth factors
to cells for the purposes of bone tissue engineering and bone
repair remains an unmet challenge. For this purpose, we added
TGF-β1 into a SF-CS scaffold to construct a 3D scaffold for the
first time to the best of our knowledge. In previous studies,
growth factors were added to the medium, and then the growth
factors in the solution were incorporated into the resulting
polymer scaffolds and the sustained release of growth factors
facilitated bone regeneration in vitro and in vivo
(21–26). This approach was limited by the
fact that the dose and spatial distribution of growth factors in
these scaffold systems was uncontrolled. In another study, rapid
drug delivery occurred in the early stages as the drug that was
close to the surface was released or the drug easily diffused out
of the bulk of the material until supplies were exhausted resulting
in lower release rates (27). To
address this issue, we aimed to explore the novel strategy of
directly adding TGF-β1 onto an SF-CS scaffold during SF-CS scaffold
fabrication, rather than after scaffold fabrication. In the future,
this novel strategy may be applied to pattern and immobilize other
growth factors to the SF-CS 3D scaffolds developed herein. We
discussed the application and biocompatibility of TGF-β1-SF-CS
scaffolds which meet the requirements of bone tissue engineering.
One of the aims of our study was to ensure that the release of
TGF-β1 from the SF-CS scaffolds occurred at therapeutic levels and
enhanced matrix production by BMSCs. The effect of TGF-β1-SF-CS
scaffolds on the morphology, differentiation and function of BMSCs
has not yet been studied and no study of this type has been
performed previously on BMSCs, to the best of our knowledge. To
examine the in vivo biocompatibility and osteogenesis of the
TGF-β1-SF-CS scaffolds, both the TGF-β1-SF-CS scaffolds and the
SF-CS scaffolds were implanted in rabbit mandibles and studied
histologically and microradiographically. The effect of all the
materials on morphogenesis was analyzed from the in vivo
response in order to identify the clinical relevance of the SF-CS
scaffold. We examined the ability of SF-CS scaffold-immobilized
TGF-β1 to drive TGF-β1 bioactivity in the TGF-β1-SF-CS scaffolds.
The sustained delivery of TGF-β1 combined with the attractive
mechanical properties of a biodegradable SF-CS scaffold may prove
more advantageous for bone repair and regeneration. The present
study enabled us to evaluate this technique as a potential method
of constructing an ideal TGF-β1-SF-CS scaffold for bone tissue
engineering. To the best of our knowledge, this is the first study
to use TGF-β1-SF-CS composite scaffolds for mandibular repair.
Materials and methods
Animals and ethics statement
In the present study, we established an animal model
to examine the repair of mandible defects using tissue engineering
techniques. Eighteen healthy New Zealand white rabbits of either
gender (3 months old, weighing approximately 3.0 kg) were obtained
from the Center for Experimental Animals at China Medical
University (Shenyang, China) with a National Animal Use License
number of SCXK-LN2011-0009. Animal use was approved by the Animal
Use and Care Committee at China Medical University with a protocol
number of CMU62043006. All experiments and surgical procedures were
approved by the Animal Care and Use Committee at China Medical
University, and complied with the National Institutes of Health
Guide for the Care and Use of Laboratory Animals. All efforts were
made to minimize the number of animals used and their
suffering.
Materials
Raw silk was purchased from Nanchong Silk Co., Ltd.
(Nanchong, China). Chitosan was purchased from Nantong Xing Cheng
Biological Products Factory (Jiangsu, China). TGF-β1 (100
ng/bottle) was purchased from ProSpec-Tany TechnoGene, Ltd.
(Rehovot, Israel). Chemicals including NaHCO3,
CaCl2 and ethanol were purchased from Jiancheng
Biotechnology Institute (Nanjing, China). All other chemicals and
reagents were of analytical grade unless specified otherwise.
Preparation of TGF-β1-SF-CS and SF-CS
scaffolds
Bombyx mori silk fibers (Nanchong Silk Co.,
Ltd.) were treated with 0.5% (w/w) NaHCO3 solution twice
at 100°C for 30 min and then rinsed with 70°C distilled water for
30 min to remove sericin. Degummed silk was dissolved in a mixed
solvent system of
CaCl2/CH3CH2OH/H2O
(mole ratio of 1:2:8) at 70°C for 6 h and filtered to obtain a SF
solution. The solution was dialyzed in cellulose dialysis tubing
(MWCO=50,000) against distilled water for 3 days and the water was
replaced every 12 h to remove all impurities. CS solutions were
prepared by dissolving high molecular weight CS (82.7%
deacetylation; Tongxing Co.) at 3.66% (w/v) in 2% acetic acid. The
final concentration of CS used was 2%.
TGF-β1 solution was prepared by dissolving 100 ng
TGF-β1 in 1 ml deionised sterile water (100 ng/ml). The selected
concentrations were based on the mid-range concentrations used in
previous studies (28,29).
SF/CS blend solutions (100 ml) with a SF/CS weight
ratio 5:5 were prepared in the same solvent system at 10 wt%
(combined weight of CS and SF). Following dialysis in cellulose
dialysis tubing (MWCO=50,000) against distilled water for 3 days
with a water change every 12 h, CS and SF solutions were obtained.
TGF-β1 solution (1 ml) was then added and stirred with a magnetic
stirrer for 50 min. The CS, SF and TGF-β1 solution was poured into
a 24-well Teflon culture plate. The CS, SF and TGF-β1 solution was
lyophilized (VFD-2000; Boyikang Laboratory Instruments Co., Ltd.,
Beijing, China) for a total time of 48 h to obtain TGF-β1-SF-CS
scaffolds. We then prepared SF-CS scaffolds using the same
method.
To improve water stability, the dry TGF-β1-SF-CS and
SF-CS scaffolds were treated with methanol solution (concentration
<10%) for 2 h to crystallize the SF content and neutralize the
CS content. The TGF-β1-SF-CS and SF-CS scaffolds were lyophilized
to remove excess methanol.
The dry TGF-β1-SF-CS and SF-CS scaffolds were
fractured in liquid nitrogen using a razor blade. They were cut
into square pieces of 0.5×0.5 cm in size, placed on aluminum stubs
with conductive adhesive tape composed of carbon, coated with gold
using a JFC-110E Ion Sputter (Jeol USA, Inc., Peabody, MA, USA) for
60 sec at 20 mA prior to observation under a scanning electron
microscope (JSM-TM3000; Hitachi, Tokyo, Japan). Scanning electron
microscopy (SEM) images and pore size were analyzed using ImageJ
software. For each type of scaffold, a minimum of 30 pores were
examined.
Determination of scaffold porosity and
water absorption capacity
The porosity of the TGF-β1-SF-CS and SF-CS scaffolds
was evaluated using hexane displacement (30). The scaffolds were immersed in a
known volume (V1) of hexane. The total volume of
hexane and hexane-impregnated scaffold was recorded as
V2. The volume difference (V2 −
V1) was the volume of the polymer scaffold.
Hexane-impregnated scaffold was removed, and the residual hexane
volume was recorded as V3. The porosity of the
SF-CS scaffold was calculated as [(V1 −
V3)/(V2 −
V3)] ×100.
The water absorption capacity of the TGF-β1-SF-CS
and SF-CS scaffolds was determined by immersion in phosphate
buffered saline (PBS; pH 7.4) at 37°C for 24 h. At predetermined
time intervals, three scaffold samples of each scaffold were
carefully blotted to remove excess water and weighed. Wet weight
(W1, swollen weight) and dry weight
(W2, dried at 37°C overnight) were measured. The
water absorption capacity of the scaffold was calculated using the
following formula: [(W1 −
W2)/W2] ×100.
The swelling ability of SF-CS scaffolds, using three
samples/scaffold, was determined by immersion in PBS (pH 7.4) at
37°C for 24 h. Wet volume (S1, swollen volume)
and dry volume (S2, dried at 37°C overnight) were
measured. The swelling index of the scaffold was calculated using
the following formula: [(S1 −
S2)/S2] ×100.
Isolation, culture and differentiation of
BMSCs
Prior to the surgical procedures, the rabbits were
anesthetized with 10% chloral hydrate (0.35 ml/100 g)
intraperitoneally. Bone marrow (3–5 ml) was extracted from rabbit
femurs using a heparinized syringe, and then centrifuged and washed
with PBS. The cell suspension was centrifuged with Percoll
lymphocyte separation medium, and the ivory white layer in the
middle was collected, washed with PBS and cultured in complete
low-glucose Dulbecco's modified Eagle's medium (DMEM). The cells
were seeded in culture flasks at a density of 1.0×104
cells/cm2 and incubated at 37°C in a humidified
atmosphere with 5% CO2. Half of the culture medium was
replaced after 24 h, and completely replaced after 48 h, followed
by medium changes every 2–3 days. The unattached cells were
discarded by refreshing the medium. The cells at 85% confluency
were trypsinized, centrifuged and passaged at a 1:2 ratio.
Third-passage cells were induced to differentiate with osteogenesis
induction liquid (0.1 µmol/l dexamethasone, 50
µmol/ml vitamin C, 10 mmol/l β-glycerophosphate and 10%
fetal bovine serum in glucose DMEM), followed by medium changes
every 2–3 days. BMSC morphology at different time points was
examined under a light-inverted microscope (CKX41; Olympus, Tokyo,
Japan).
Alkaline phosphatase (ALP) staining and
the von Kossa assay
ALP activity was evaluated in BMSCs 14 days after
osteogenesis induction and in those without osteogenesis induction
(control group). The cells were fixed for 10 min at 4°C and
incubated with 2% cobalt nitrate and 1% ammonium sulfide (ALP kit;
Hongqiao, Shanghai, China). Areas stained tan were designated as
positive.
BMSCs plated in 6-well plates were fixed in 70%
ethanol 18 days after osteogenesis induction and those without
osteogenesis induction (control group). The cells were stained with
von Kossa silver (Jiancheng Biotechnology Institute) and placed
under ultraviolet (UV) light for 10 min. The cells were then
treated with 5% NaS2O3 for 2 min, and washed
with distilled water. Calcium nodules with a diameter >1 mm were
visualized under a light-inverted microscope.
BMSC seeding onto scaffolds and
morphological analysis
The scaffolds were sterilized with 75% alcohol under
UV light overnight and then rinsed extensively three times with
sterile PBS. Prior to cell culturing, the scaffolds were pre-wetted
by immersion in DMEM for 12 h in an incubator at 37°C.
The BMSCs were cultured onto TGF-β1-SF-CS scaffolds,
SF-CS scaffolds and 24-well tissue culture plate wells (control
group; n=3; diameter 15 mm; height 13 mm) for 3 and 6 days after
osteogenesis induction at 37°C under an atmosphere of 5%
CO2 and 95% air, with the original cell culture density
of 4,000 cells/well.
BMSC morphology following osteogenesis induction on
the TGF-β1-SF-CS scaffold and the SF-CS scaffold was observed on
days 1, 3 and 7 by SEM. The samples were washed with PBS and fixed
with 3.0% glutaraldehyde at 4°C for 4 h. Subsequently, they were
dehydrated through a graded ethanol series (30, 50, 70, 80, 90, 95
and 100%), air-dried overnight, and sputtered with gold for SEM
observation. Adherent cell morphology, patterns of cellular
adherence with respect to structural features of the TGF-β1-SF-CS
scaffold and the SF-CS scaffold, cell-cell interactions, and
adherent cell density were assessed.
Adhesion of BMSCs to scaffolds
Following osteogenesis induction on the TGF-β1-SF-CS
scaffold, the SF-CS scaffold and the control group, the BMSCs were
harvested and the concentration was adjusted to
1×104/ml. The cells were placed in cell culture plates
pre-coated with TGF-β1-SF-CS scaffold and SF-CS scaffold at 1
ml/well. The cells cultured in culture plates with no scaffold
served as the controls. Three parallel wells were used for each
group. The cells were cultured in an incubator at 37°C with 5%
CO2. The number of non-adherent cells was quantified at
1, 3 and 6 h. The cell adhesion rate (%) was calculated as (number
of seeded cells − non-adherent cells)/(number of seeded cells)
×100.
Cell Counting Kit-8 (CCK-8) assay and ALP
activity
To examine grafted cell survival, cell viability and
proliferation was determined using a CCK-8 assay. After the samples
were cultured for 1, 3, 5 and 7 days, the culture medium was
replaced with a serum-free culture medium containing CCK-8 (0.5
mg/ml). After 4 h of culturing, the samples were transferred to a
96-well plate. Absorbance was measured at 450 nm using an ELISA
reader (Bio-Rad Model 550; Bio-Rad, Berkeley, CA, USA). Optical
density (OD) at 450 nm was proportional to the number of cells.
Intracellular ALP activity in the scaffolds and the
control group was measured using an ALP assay kit (Nanjing
Jiancheng Bioengineering Institute, Nanjing, China). The samples
were cultured for 1, 4, 7, and 10 days, and then washed with PBS
three times. The samples and the control group with cells were
immersed overnight in 500 ml cell lysis solution containing 0.1%
Triton X-100 and 5 mM MgCl2. The solution was
transferred to a tube and centrifuged at 28,340 × g for 10 min at
4°C. ALP activity was measured by mixing 50 µl supernatant
with 50 ml p-nitrophenyl phosphate (5 mM) in 150 mM
2-amino-2-methyl-1-propanol buffer solution at room temperature for
30 min in the dark. Following the incubation period, the reaction
was stopped by adding 50 µl 0.2 N NaOH to denature ALP and
the OD was measured at 520 nm using an ELISA reader (Bio-Rad Model
550; Bio-Rad).
Surgical procedure and implantation
The bone-forming ability of the TGF-β1-SF-CS
scaffolds and the SF-CS scaffolds was assessed in the rabbit model
of a submaxillary defect. The rabbits were randomly divided into
the following 3 groups: i) the TGF-β1-SF-CS scaffolds group (n=6);
ii) the pure SF-CS scaffolds group (n=6); iii) the non-scaffold as
a control group (n=6). The surgical procedures were performed under
aseptic conditions. The rabbits were anesthetized with 10% chloral
hydrate (0.35 ml/100 g) intraperitoneally, and the jaw was exposed
and disinfected. Bilateral 3-cm parallel incisions were made on the
jaw of the rabbit. The periosteum was retracted and the submaxilla
was exposed. A critical round full-thickness bone defect, 8×8
mm2, was made in a buccal-lingual direction using a
slow-speed dental drill on one side of the mandible that was cooled
continuously. To avoid tissue damage due to overheating, 0.9%
saline was dripped onto the contact point between the bur and bone.
A 8×8-mm2 sized pure SF-CS scaffold or TGF-β1-SF-CS
scaffold construct was inserted into the defect. To ensure that the
bony defects were critically sized, rabbits in the control group
had nothing inserted into the defect. The region of the defect was
washed with normal saline, and the incision was sutured layer by
layer. Penicillin (400,000 units) was intramuscularly injected over
3 days to prevent infection.
General observations, dual energy X-ray
absorptiometry (DXA) analysis, and radiographic analysis
Following surgery, all rabbits were housed and fed
routinely, and monitored for changes in their appearance,
submaxilla movements, and healing of the surgical wounds.
Additionally, the local bone mineral densities (BMDs) were measured
on a Discovery DXA system (Hologic Inc., Bedford, MA, USA). 3D
images of the mandibles were captured with a multi-slice spiral
computed tomography (CT) scanner (GE Lightspeed Ultra 16; GE
Healthcare, Milwaukee, WI, USA) at 12 weeks after implantation.
Biomechanical evaluation
Flexural modulus and peak flexural strength were
measured. The specimens were tested to flexural failure in a
three-point bending configuration with a 10-mm spacing between the
loading supports and a 20-mm spacing between the base supports. The
specimens were loaded in buccal-lingual orientation at a constant
strain rate of 0.5 mm/min on an Insight 5 uniaxial test frame (MTS
Systems Corp., Eden Prairie, MN, USA).
Histomorphometric analysis of bone
regeneration
Rabbits from each group were sacrificed at 4, 8 and
12 weeks after implantation by acute blood loss under anesthesia.
To quantitatively determine the amount of newly formed bone, we
statistically analyzed histological sections from different
implantation periods. At every implantation time (4, 8 and 12
weeks), three histological sections were randomly chosen from the
pure SF-CS scaffold, the TGF-β1-SF-CS scaffold and the control
groups. These specimens were fixed in 4% paraformaldehyde, embedded
in paraffin and serial buccal and lingual sagittal cross sections
were made, which was followed by staining with hematoxylin and
eosin (H&E; Jiancheng Biotechnology Institute). Each section
was then observed under a light microscope (CKX41; Olympus) at ×100
magnification, and at least 5 images were randomly obtained from
one section. Using image analysis software Image-Pro Plus (Media
Cybernetics, Inc., Rockville, MD, USA), new bone volume (NBV) was
expressed as the percentage of newly formed bone area in the
available pore space (bone area/pore area ×100).
Statistical analysis
All quantitative data are expressed as the means ±
standard deviation. Statistical analyses were performed using the
statistic program SPSS 17.0. The results were analyzed by one-way
analysis of variance (ANOVA) with a Student's t-test. A P-value
<0.05 was considered to indicate a statistically significant
difference. Before the results were analyzed by one-way ANOVA, all
quantitative data obeyed the normality distribution.
Results
Evaluation of porosity and water
absorption capacity of scaffolds
With the addition of TGF-β1, the pore size and
porosity of SF-CS scaffolds increased, although the difference was
not significant (P>0.05). The hexane displacement experiment
demonstrated that the porosity of the TGF-β1-SF-CS scaffolds
(90.2±4.6%), as well as the pore size (236±73 µm) (Table I), may facilitate nutrient and
waste transport into and out of the scaffolds.
 | Table IPorosity, swelling and water
absorption of TGF-β1-SF-CS and SF-CS scaffolds. |
Table I
Porosity, swelling and water
absorption of TGF-β1-SF-CS and SF-CS scaffolds.
| Porosity (%) | Pore size
(µm) | Water absorption
(%) | Swelling index
(%) |
|---|
| TGF-β1-SF-CS | 90.2±4.6 | 236±73 | 271±12 | 25.6±0.4 |
| SF-CS | 89.5±7.9 | 220±91 | 282±4 | 24.5±0.5 |
Water absorption capacity, swelling ability and
retain ability are other important factors in determining the
usefulness of the biomaterials. The absorption capacity of the
TGF-β1-SF-CS and SF-CS scaffolds was measured in terms of the
degree of swelling at equilibrium. It was found that the degree of
swelling of the scaffolds was in range of 24–26% of their dry
weight and the difference was not significant (P>0.05) following
the addition of TGF-β1 (Table I).
The TGF-β1-SF-CS and SF-CS scaffolds, which exhibited a high degree
of swelling and water absorption, also retained their structure
after immersion in PBS (pH 7.4) for 24 h.
Cell morphology and osteogenic
differentiation of BMSCs
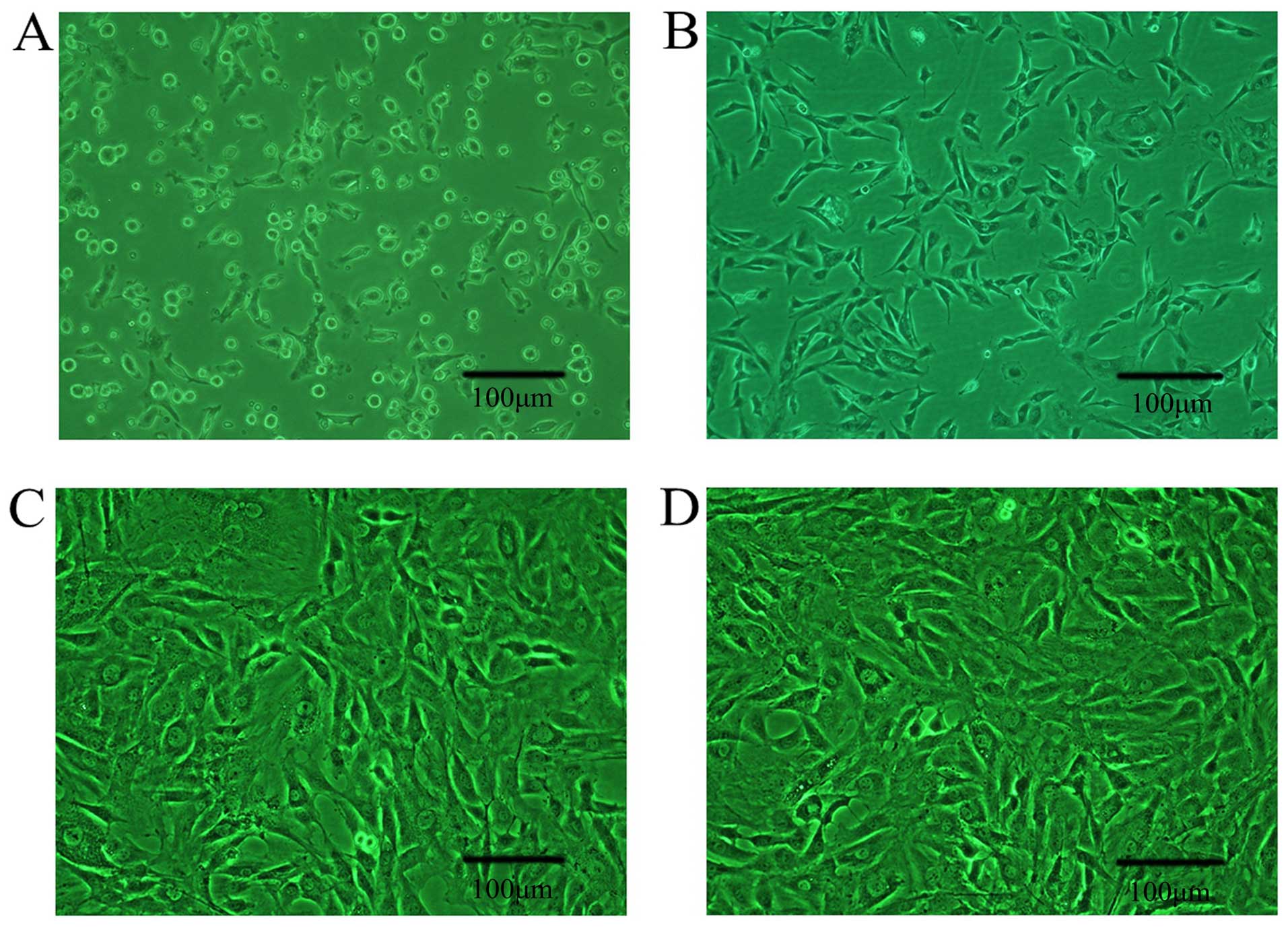
Cell morphology was studied using a light-inverted
microscope (Fig. 1). The BMSCs
demonstrated good survival and rapid proliferation.
In primary cells, BMSCs exhibited a round
morphology. After 12 h, the BMSCs were attached to the culture
flasks sparsely and displayed a fibroblast-like, spindle-shaped
morphology (Fig. 1A). After 3–4
days of incubation, cell numbers increased, the cells continued to
grow well and actively proliferate and the cells gradually grew
into small colonies, and they assumed a more spindle-shaped,
fibroblastic morphology (Fig.
1B). After 10–12 days, the majority of the cells were attached
and had acquired a large, flattened or multilayer growth morphology
(Fig. 1C) which remained
unchanged throughout subsequent passages (Fig. 1D).
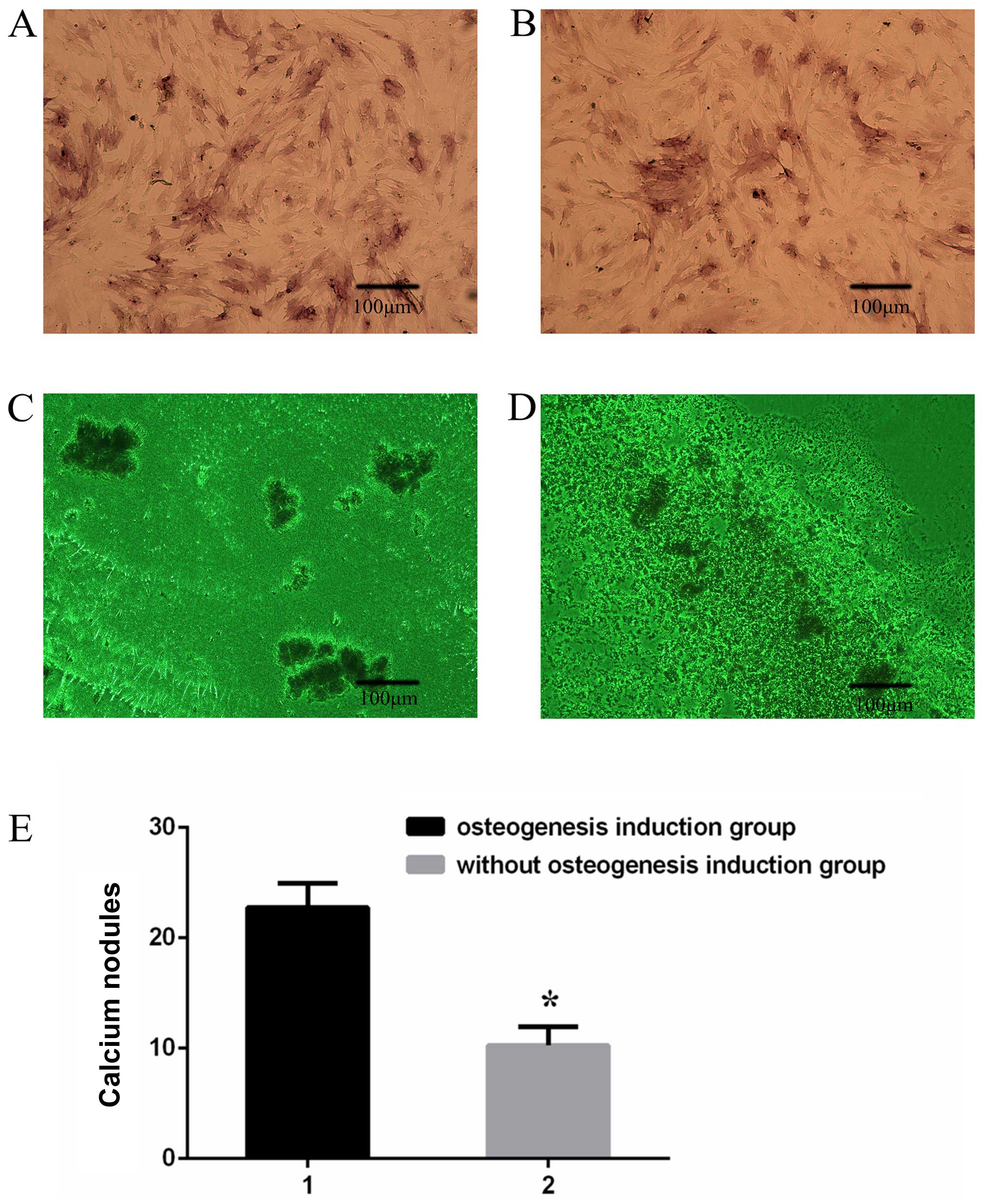
Two weeks later, ALP staining was greater in the
BMSCs after osteogenesis induction (Fig. 2A) than in those without
osteogenesis induction (Fig. 2B).
In addition, Von Kossa staining 2 weeks after osteogenesis
induction (Fig. 2C) revealed a
significant increase in calcium nodules compared with that in the
BMSCs without osteogenesis induction (Fig. 2D).
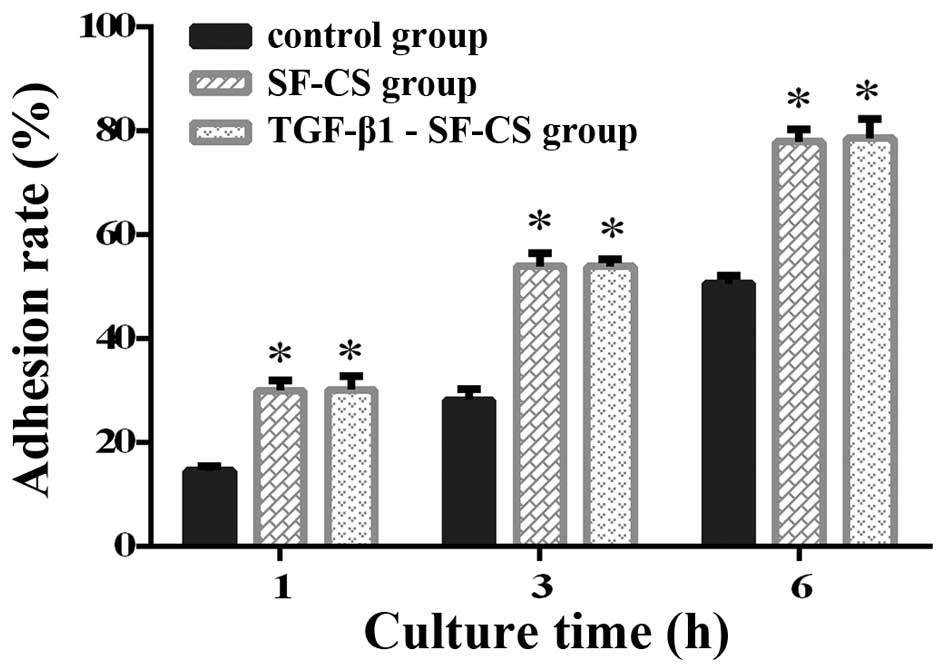
Adhesion rate of BMSCs
After 1, 3 and 6 h, the cell adhesion rate increased
in all three groups, in a time-dependent manner (Fig. 3). The adhesion rates in the
TGF-β1-SF-CS group and the SF-CS group were higher than those in
the control group (P<0.01). At 1 h, the adhesion rate in the
SF-CS scaffold group was nearly the same as that in the
TGF-β1-SF-CS scaffold group; there were no significant differences
between the scaffold groups (P>0.05). After 3 h, there was a
significant increase in the adhesion rate compared with the groups
at 1 h. The adhesion rate in the TGF-β1-SF-CS group and the SF-CS
group was significantly higher than that in the control group
(P<0.05); however, there was still no significant difference
between the scaffold groups (P>0.05). These results have–proved
that the SF-CS scaffolds increased BMSC adhesion–and that TGF-β1
was unable to promote BMSC adhesion in the SF-CS scaffolds.
SEM images of TGF-β1-SF-CS scaffolds and
BMSCs cultured with the TGF-β1-SF-CS scaffolds
Following the addition of TGF-β1, the morphology of
the scaffolds did not change markedly. SEM revealed that all of the
TGF-β1-SF-CS scaffolds and the SF-CS scaffolds exhibit porous
structures (Fig. 4). The
composite of TGF-β1-SF-CS scaffolds was mixed well by a homogeneous
method. A smooth surface is observable in the SEM images. The
TGF-β1-SF-CS scaffolds exhibit well-oriented, porous structures
from the surface to the inside which may be beneficial for cell
seeding and distribution (Fig.
4A). The TGF-β1-SF-CS scaffold with fully porous structures may
facilitate uniform cell seeding and nutrient delivery, which are
important for cell growth in a 3D porous scaffold. To measure the
pores in the TGF-β1-SF-CS scaffolds, the scaffold was cut
transversely to expose the internal porous structures. The
TGF-β1-SF-CS scaffolds exhibited a homogeneous pore structure. The
inner pore size of the TGF-β1-SF-CS scaffolds was approximately
10–300 μm. Fig. 4B shows
that the pore wall of the TGF-β1-SF-CS scaffold has a smooth
surface morphology and the width of the pore wall is approximately
1–2 µm. The homogeneous pore wall section morphology shows
that TGF-β1, SF and CS are compatible, even at nanoscale. Fig. 4C shows that the SF-CS scaffold has
a similar morphology to the TGF-β1-SF-CS scaffold. This result
suggests that TGF-β1, SF and CS may be combined to generate a
composite scaffold, possessing the advantages of the growth factor
and two naturally-derived biomaterials.
The growth of osteogenically-induced BMSCs on
scaffolds, cellular attachment and interactions within the
TGF-β1-SF-CS scaffold were evaluated by electron microscopy. The
SEM images reveal that BMSCs are present on the surfaces and the
pores of the scaffolds. The SEM images also show that on the
TGF-β1-SF-CS scaffold, there are a number of BMSCs present that
extend towards other cells in the culture.
On day 1, BMSCs seeded onto the scaffold had a
distinct, round morphology and a large number of particles were
observed on the cell surface, characterized by numerous plasma
membrane microstructural extensions. Adherent BMSCs were observed
preferentially localized to the fold regions of the TGF-β1-SF-CS
sheets, with BMSCs noted among fibrils projecting outwards from the
scaffold edges (Fig. 4D).
After 3 days of culture, a large number of cells are
adhered to the scaffold. The cell aggregates are enlarged and have
actively proliferated. The SEM images reveal that the BMSCs have
spread to the scaffold surface and the pores, and they exhibit a
flat and elongated morphology. Granular and filiform substances are
observable around the cells, with microvilli and larger
lamellipodia extensions extending from the BMSCs localized to the
scaffold. The cells initially exhibit a stereo spindle and
dendritic morphology at day 3 (Fig.
4E).
At day 7, the BMSCs have spread wider and deeper
into the scaffold, an increased number of cells are adhered to the
surface and the pores of the scaffold compared with that at day 3,
and the cells have grown and proliferated well. They exhibit
typical osteoblast cell morphology and adhere tightly to the
surface of the scaffold or inside, by forming lamellipodia and
filopodia. Cell-cell interactions are also observable on the
surface of the TGF-β1-SF-CS scaffold as evidenced by connection of
the fibrous extensions between two BMSCs. Similar cellular
spreading and cell-cell interactions are apparent in more cells
spread throughout the scaffold, which is crucial for the viability
and function of BMSCs in vitro (Fig. 4F).
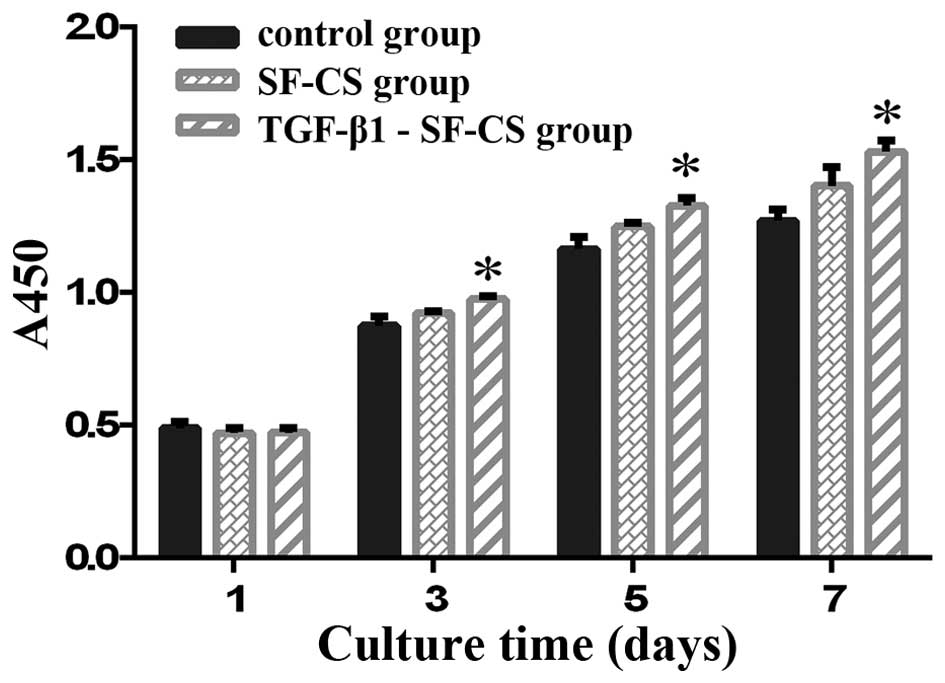
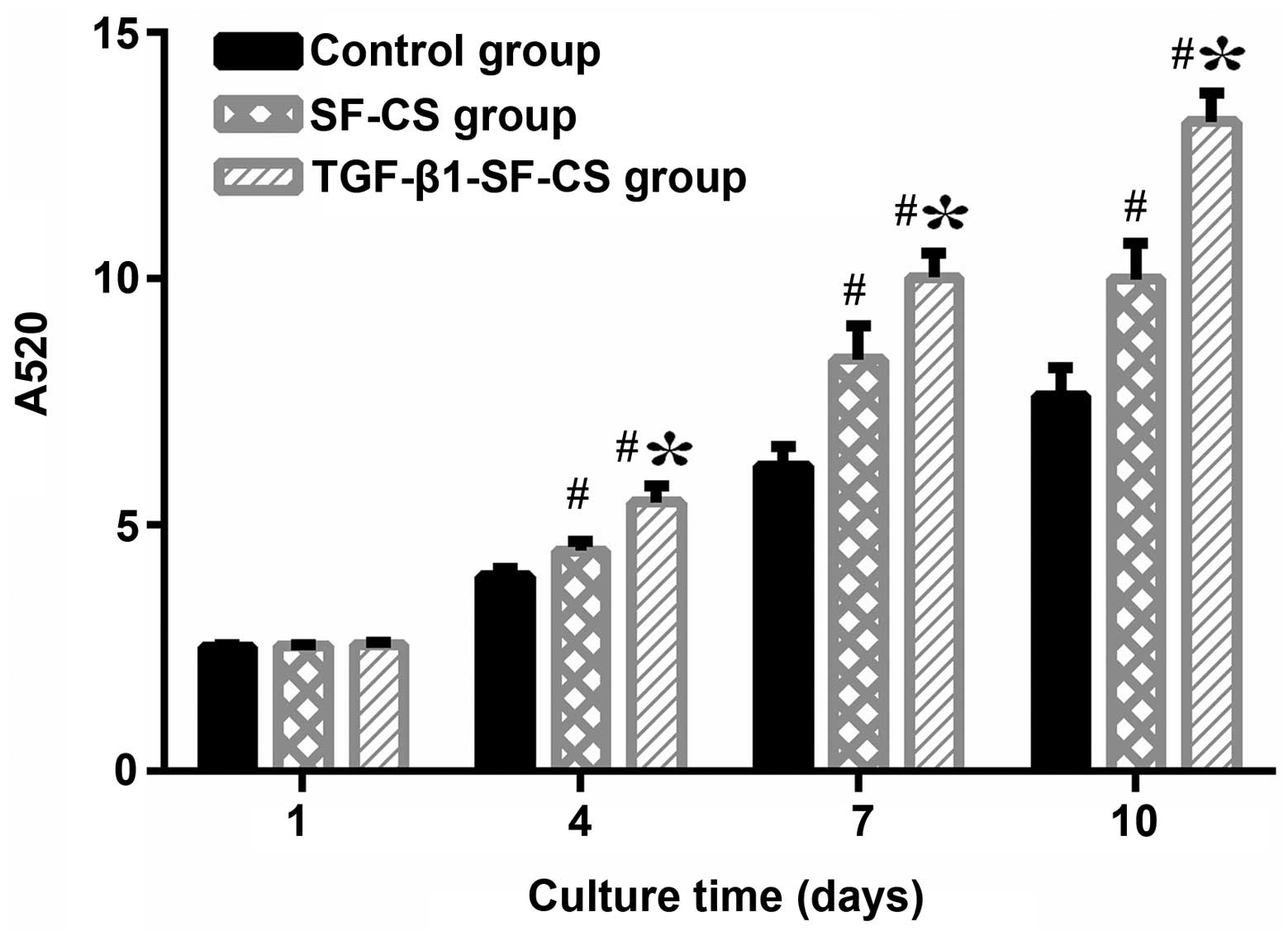
CCK-8 assay and ALP activity
The proliferation of BMSCs on the TGF-β1-SF-CS
scaffolds, the SF-CS scaffolds and in the control group cultured
for 1, 3, 5 and 7 days were compared using a CCK-8 assay (Fig. 5). The absorbance index of the
three groups increased with the increase in culture time, which
indicated that significant cell growth had occurred within both
scaffolds. The data regarding BMSCs cultured without scaffolds is
also present. The BMSCs proliferated well in the TGF-β1-SF-CS
scaffolds and the SF-CS scaffolds, and there were no significant
differences with the cells cultured in the control group at day 1
(P>0.05). However, after 3 days, more proliferation was observed
in the BMSCs cultured on the TGF-β1-SF-CS scaffolds than in the
cells cultured on the SF-CS scaffolds, and the proliferation of
BMSCs cultured on both scaffolds was markedly increased compared
with those cultured without scaffolds (P<0.05). These results
indicate that the TGF-β1-SF-CS scaffolds significantly enhanced the
attachment and proliferation of BMSCs under these conditions
compared with the SF-CS scaffolds.
To evaluate the osteoblastic differentiation of the
mesenchymal stem cells on the TGF-β1-SF-CS scaffolds and the SF-CS
scaffolds, we measured ALP activity using spectrophotometry. The
ALP activity of the BMSCs cultured on the TGF-β1-SF-CS scaffolds,
the SF-CS scaffolds and the control group is shown in Fig. 6. For total ALP activity
measurement on day 1, there was no significant difference in OD
values between the TGF-β1-SF-CS, SF-CS and control groups
(P>0.05). On days 4 to 10, a significant difference in OD values
was found between the scaffolds groups and the control group
(P<0.01). The OD values in the TGF-β1-SF-CS group were the
highest. This suggests that TGF-β1 exerts an effect on the activity
of BMSCs. As the culture time increased from days 4 to 10, the
growth trend of the OD values in the three groups increased
rapidly. The OD values reached a peak at day 10. This suggests that
the TGF-β1-SF-CS scaffolds are a stable, prolonged culture system
and stabilized TGF-β1 greatly enhanced ALP activity.
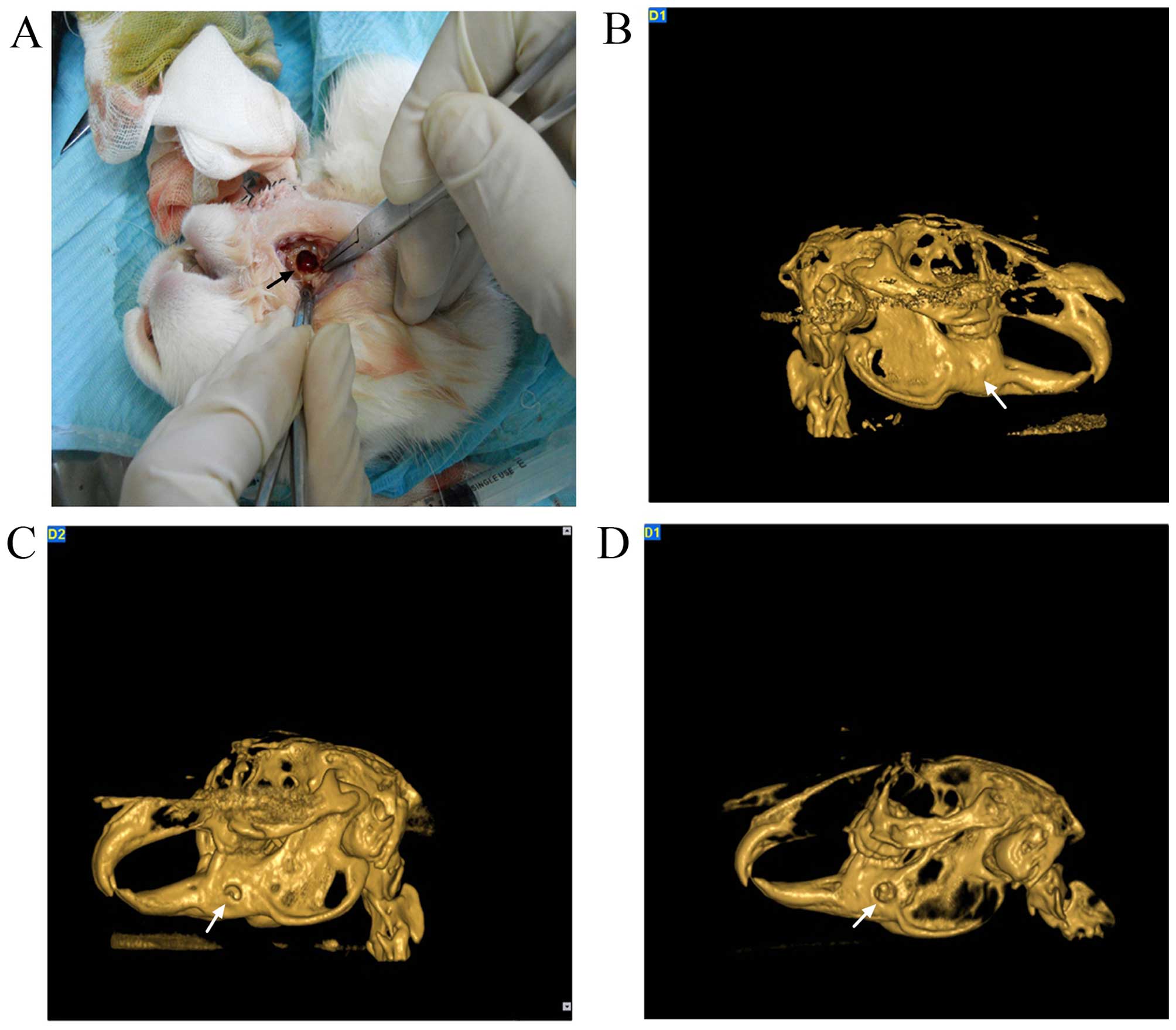
Animals and clinical observations
A total of eighteen rabbits underwent the successful
creation of a mandibular defect (Fig.
7A). All animals developed some degree of postoperative
swelling at the incision site that resolved within one week of the
operation with no functional consequences. The animals were allowed
to heal for 4–12 weeks and then sacrificed for analysis. No rabbit
developed an abscess or was sacrificed and excluded from
analysis.
Analysis of 3D-CT images
To follow new bone formation and the development of
bone unions within the defects, 3D images were captured 3 months
postoperatively. Representative images from each group are shown in
Fig. 7.
Analysis of the 3D-CT images confirmed the findings
that the pure SF-CS scaffolds and the TGF-β1-SF-CS scaffold
substitutes are bioactive and biocompatible with the host bone. In
the TGF-β1-SF-CS scaffold group, the disappearance of the defect at
12 weeks after implantation indicated that the density of the newly
formed bone was as high as that of the host bone, and that complete
osteointegration of the defect area had been achieved (Fig. 7B). In comparison, in the pure
SF-CS scaffold group, the volume and radiopacity of the newly
formed bone are highly increased; however, the new bone formation
failed to cover the defect completely, and bone formation occurred
along the periphery of the defect area, which illustrated that the
new bone had increased in density and quantity and had grown into
the scaffold although incompletely (Fig. 7C). In the empty control group, the
untreated defect exhibited no evidence of healing by itself even at
12 weeks (Fig. 7D).
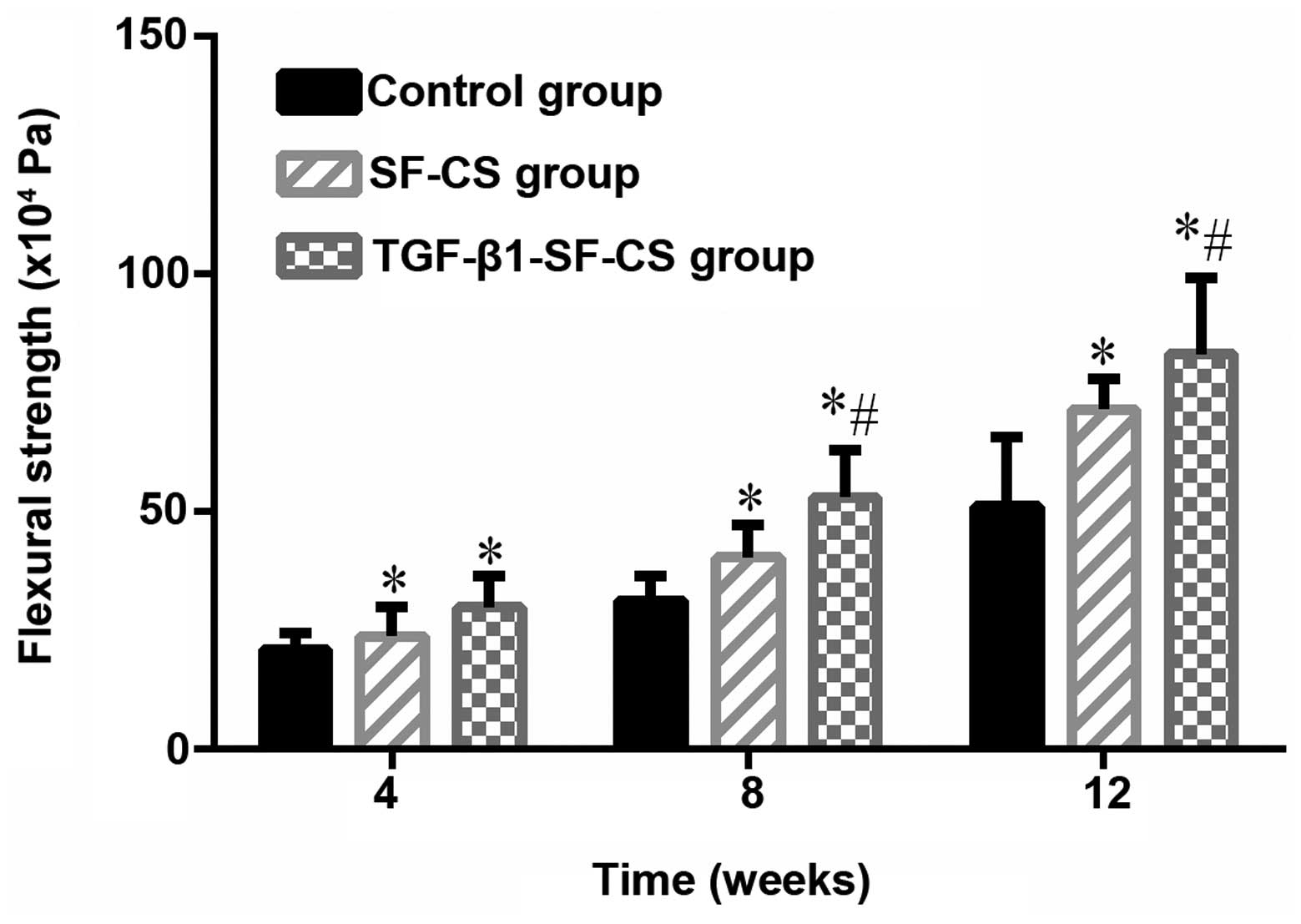
Biomechanical evaluation
As shown in Fig.
8, the flexural strength of the implanted scaffold groups and
the control group cultured for 4, 8 and 12 weeks were compared. The
flexural strength for the implanted scaffold groups gradually
increased, indicating the occurrence of significant bone growth
within both scaffolds. After 4 weeks, the flexural strength of the
implanted scaffold groups (23.9±6.223×104 Pa and
29.9±6.481×104 Pa) was significantly greater than the
flexural strength of the empty defects (20.9±3.610×104
Pa) (P<0.05). The increase in flexural strength was highest in
the TGF-β1-SF-CS group, and the flexural strength values reached a
peak at 12 weeks (87.2±11.766×104 Pa).
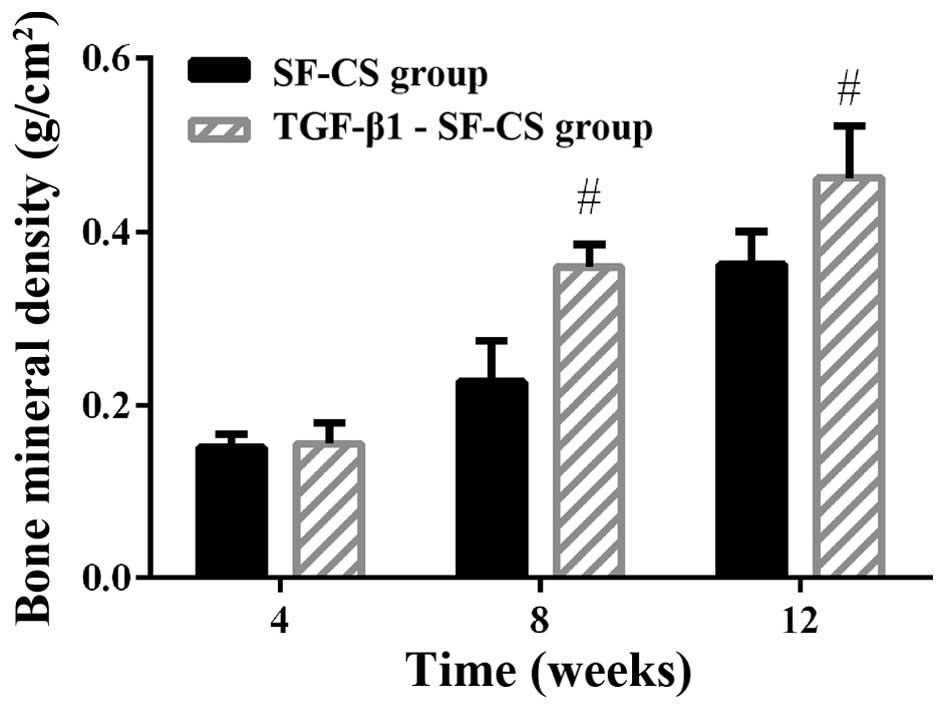
Evaluation of local BMD
To quantify the level of calcification in the
repaired mandibles, local BMD in all animals was measured on a DXA
system at 4, 8 and 12 weeks postoperatively (Fig. 9). The BMD for the implanted
scaffold groups gradually increased, indicating significant bone
growth within both scaffolds. The BMD of the TGF-β1-SF-CS scaffold
group was not significantly different from that in the SF-CS
scaffold group at 4 weeks (P>0.05). However, at 8 weeks, the
TGF-β1-SF-CS scaffold group showed a relatively higher BMD of
0.359±0.025 g/cm2. In the SF-CS scaffold group, the
local BMD was 0.227±0.047 g/cm2, which was significantly
lower than that in the TGF-β1-SF-CS scaffold group (P<0.05). At
12 weeks, the BMD of the TGF-β1-SF-CS scaffold group (0.462±0.060
g/cm2) was significantly higher than the BMD of the
SF-CS scaffold group (0.362±0.038 g/cm2) (P<0.05).
The control group with a defect the same size as those implanted
did not display any detectable BMD value in the DXA tests, thus,
the contribution to the total density was negligible (data not
shown).
Histomorphometric analysis of bone
regeneration
Based on the in vivo histological study, all
of the light microscope images showed the architecture of the
scaffold clearly, which revealed that the scaffolds were not
markedly deformed. In the TGF-β1-SF-CS scaffold group and the SF-CS
scaffold group, all the scaffolds implanted after 4 weeks are
encapsulated by fibrous collagen, and some newly formed bone
tissues are deposited at the interface of material and bone with
the presence of active osteoblasts (Fig. 10A1 and B1). Moreover, a large
proportion of osteoblasts have penetrated into the porous cavities,
which accelerate the mineralization and regeneration of bone, and a
large number of inflammatory cells are observable, as well as some
newly generated tissues. Additionally, the interface between the
SF-CS scaffold and natural bone is clearly visible, whereas in the
TGF-β1-SF-CS scaffold group the boundary between the material and
host bone is unclear. In the control group, repaired tissues are
not observable, and the defects are filled with few fiber-like
tissues and adipocytes (Fig.
10C1).
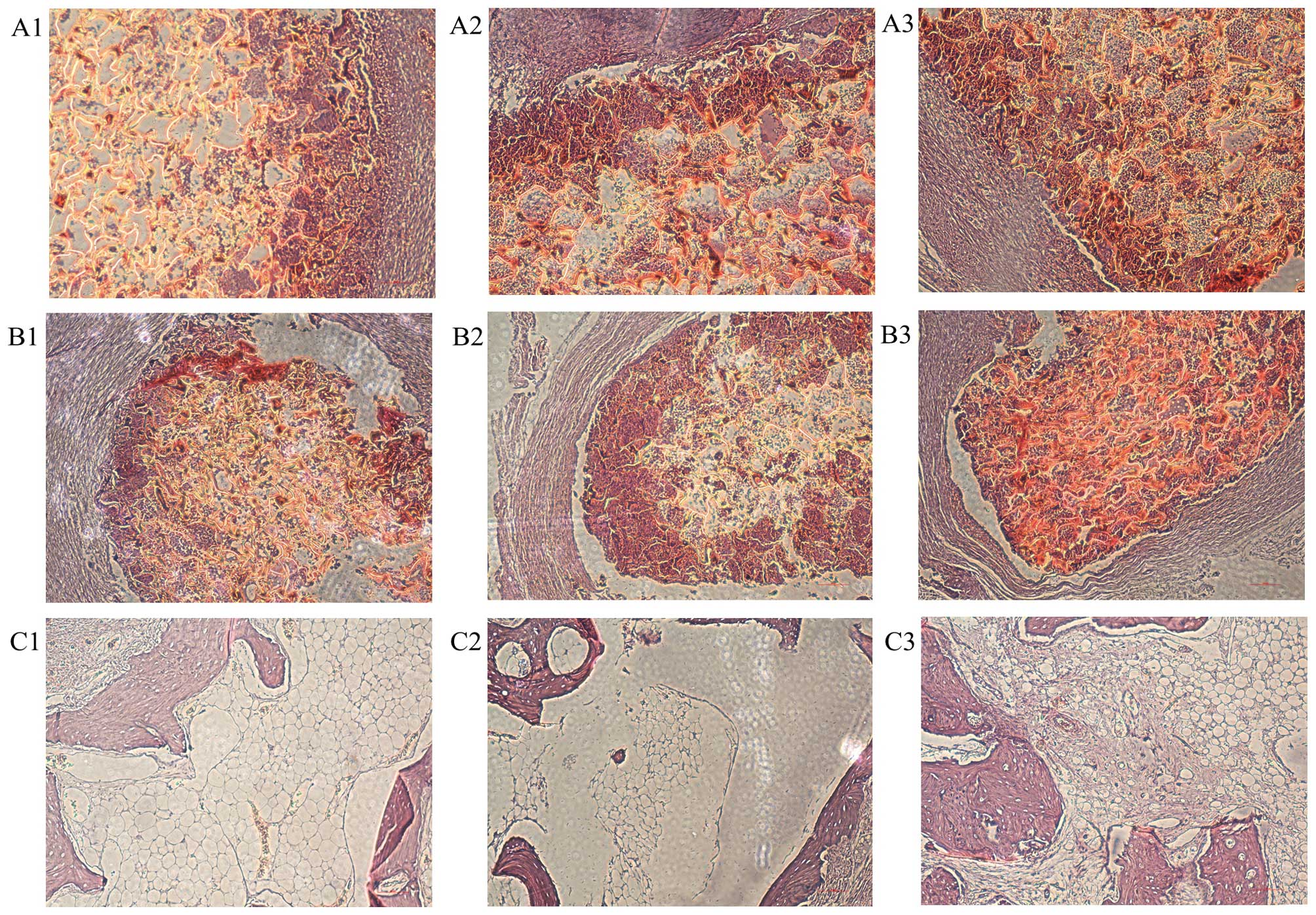 | Figure 10Sample histology of healed defects
for the TGF-β1-silk fibroin-chitosan (TGF-β1-SF-CS) scaffolds, the
silk fibroin-chitosan (SF-CS) scaffolds and the control group
(light microscope, ×100 magnification). At 4 weeks: (A1 and B1) All
the scaffolds are encapsulated by fibrous collagen, some newly
formed bone tissues are deposited at the interface between material
and host bone with the presence of active osteoblasts. (C1) In the
control group, repaired tissues are not present, and the defects
are filled with few fiber-like tissues and adipocytes. At 8 weeks:
(A2) In the TGF-β1-SF-CS scaffold group, the boundary between
material and host bone is unclear, new bone has regenerated and
penetrated through the interconnective pores to the center of the
scaffolds, increasing the quantity and density of the defect area.
(B2) In the SF-CS scaffold group, the interface between scaffold
and natural bone remains clearly visible, some newly formed bone
tissues are deposited at the interface of material and bone with
the presence of active osteoblasts. (C2) In the control group, the
defects are filled with some fiber-like tissues and adipocytes. At
12 weeks: (A3) In the TGF-β1-SF-CS scaffold group, the interface
between material and host bone is hardly detectable and forms a
close union without any gap. There is an ordered cell arrangement
and an increased number of cells. (B3) In the SF-CS scaffold group,
new bone formation is observable with more residual scaffold at the
center of the defect compared with the TGF-β1-SF-CS scaffold group.
(C3) In the empty control group, the mandible defects exhibit no
signs of recovery and there is limited encapsulation by fibrous
tissues. Bony defects remain present in the center with new bone
only formed at the periphery. |
At 8 weeks post-implantation, in the TGF-β1-SF-CS
scaffold group the boundary between material and host bone is more
unclear due to the sufficient formation of mature bone tissues
which have grown into the pores of the artificial scaffold and
bonded tightly with the material. With the implantation prolonged,
new bone has regenerated and penetrated through the interconnective
pores to the center of the scaffolds, increasing the quantity and
density of the defect area, and decreasing the number of
inflammatory cells (Fig. 10A2).
In the SF-CS scaffold group, the interface between scaffold and
natural bone was still clearly visible, some newly formed bone
tissues have been deposited at the interface of material and bone
with the presence of active osteoblasts (Fig. 10B2). In the control group, the
defects are filled with some fiber-like tissues and adipocytes
(Fig. 10C2).
The histological results of the TGF-β1-SF-CS
scaffold group at 12 weeks show that the interface between material
and host bone has formed a close union. There is an ordered cell
arrangement and an increased number of cells. Additionally,
inflammatory cells, including lymphocytes, are not observable.
Despite some bone ingrowth from the interface region, there is
insufficient bone formation in the central area (Fig. 10A3). In the SF-CS scaffold group,
new bone formation is apparent with more residual scaffold at the
center of the defect compared with that in the TGF-β1-SF-CS
scaffold group. The SF-CS scaffolds still hold their pore structure
and are unfilled by sufficient newly formed bone (Fig. 10B3). By contrast, in the empty
control group, the mandible defects exhibited no signs of recovery
with minimal encapsulation by fibrous tissues. Bony defects are
still present in the center, the defects were filled with some
fiber-like tissues and adipocytes, and new bone was only formed at
the periphery. Despite some bone ingrowth from the interface
region, there is insufficient bone formation in the central area
(Fig. 10C3).
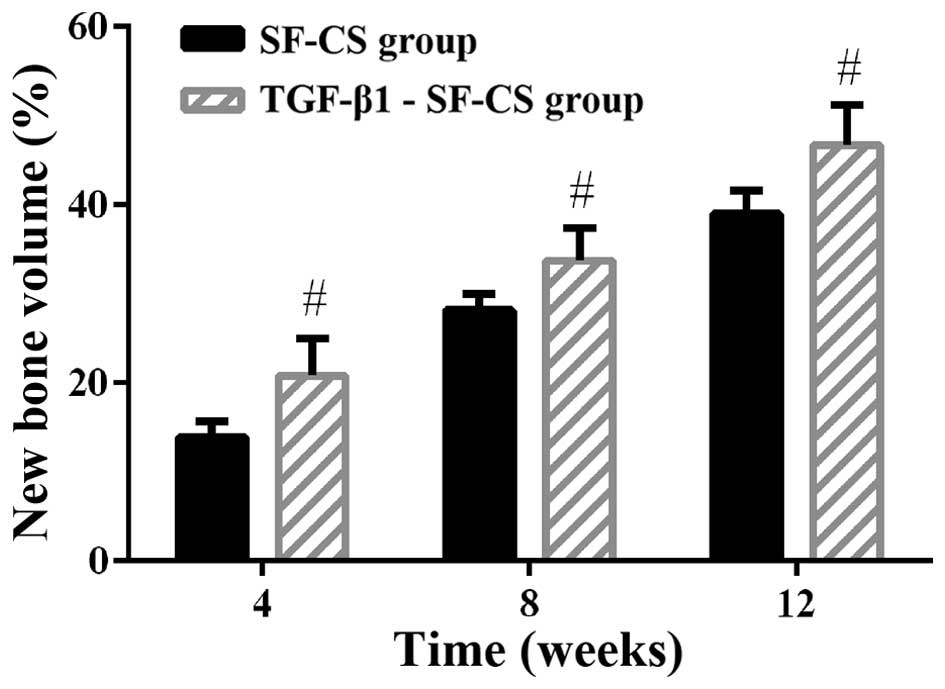
Quantitative determination of newly formed bone was
performed by statistical analysis of the histological sections.
Fig. 11 shows the NBV at each
implantation period. Clearly, after 4 weeks post-implantation, the
amount of newly formed bone in the TGF-β1-SF-CS scaffold group
increased markedly, much more than that in the pure SF-CS scaffold
group. However, after that period, bone formation in the pure SF-CS
scaffold slowed down, whereas in the TGF-β1-SF-CS scaffold group
the speed of new bone formation gradually increased. At 12 weeks,
NBV in the TGF-β1-SF-CS scaffold group was the highest. These
results confirmed that at the latter stage of implantation, the
TGF-β1-SF-CS scaffold achieved a higher efficiency of bone
formation than pure SF-CS scaffolds, and in the long-term, the
TGF-β1-SF-CS scaffolds showed far better biocompatibility and
osteogenesis. The TGF-β1-SF-CS scaffolds group showed a healing
percentage of 46.71±4.46%, which was higher than the SF-CS
scaffolds group (38.97±2.63%) (P<0.05). The control group did
not display marked bone formation in the NBV tests, so the
contribution to the total NBV was negligible (data not shown).
Discussion
Growth factors may be incorporated directly into a
polymeric scaffold to be used for tissue formation after scaffold
fabrication (21,22,31). Growth factor supplementation
strategies have shown significant functional value in the context
of bone tissue regeneration. Previous findings have 'immobilised'
or 'incorporated' growth factors such as TGF-β1 and fibroblast
growth factor into tissue scaffold systems with sustained release
properties (32). Active forms
were used and inevitably involved processing steps which may have
resulted in reduced bioactivity. In addition, the majority of
scaffold release systems are hydrogel based, and the release of
growth factors is by diffusion or degradation of the scaffold
matrix. This often results in a 'burst' release and a compromise in
the mechanical properties of the scaffold and in other properties
required for bone engineering (33). In the present study, a composite
scaffold design was constructed to increase the osteogenesis of
BMSCs through the incorporation of TGF-β1 during SF-CS scaffold
fabrication, rather than after scaffold fabrication. This method
was simple, convenient and had a high rate of success as well as
easily controlling the of growth factor content. It also improved
the loading content and loading efficiency of TGF-β1 on the SF-CS
scaffold. We analyzed the TGF-β1-SF-CS composite scaffolds for bone
tissue engineering applications. Such a scaffold may be
particularly suited for implantation into bone defects as it
promotes the proliferation, migration and differentiation of cells
as well as being osteoconductive. For this purpose, we added TGF-β1
into the SF-CS scaffold to construct a 3D scaffold for the first
time.
An optimum scaffold should meet certain criteria,
such as a suitable 3D structure for cell growth and nutrient
transport and optimal pore size to prevent cell loss from the
scaffolds. We found that the SF-CS scaffolds had random porous
microstructures, whether or not TGF-β1 was added. Porous structures
allow for the optimal interaction of the scaffold with the cells
(34). Pore size determines the
efficiency at which the cells seed into the scaffold (35). Small scaffold pores may result in
pore occlusion by the cells, preventing cellular penetration
whereas large pores prevent cell attachment due to a reduced area.
It is necessary for the scaffold to have an adequate porosity; this
includes the magnitude of the porosity, the pore size distribution
and its interconnectivity. A pore size >100 µm and
<400 µm is usually considered optimal for osteoconduction
(36). In the present study, the
TGF-β1-SF-CS scaffold had an open and interconnected pore network
and a high degree of porosity. The inner pore size of the scaffold
was approximately ten to three hundred microns, which led to
uniform cell distribution inside the scaffolds. It was an ideal
scaffold for interacting and integrating with the bone tissue. We
also found that despite adding TGF-β1 to the SF-CS scaffold, the
porosity and swelling index of the scaffold did not markedly alter
the shape and structure or morphology of the scaffolds.
CS is a polysaccharide found in the native ECM and
it is known to play a critical role in sequestering growth factors
within the matrix. Structural variations in the carbohydrate
backbone, allows CS to possess varying levels of negative charges
depending on the degree of sulfation (37,38), making it attractive option for the
development of growth factor-sequestering biomaterials. In addition
to the non-specific, electrostatic growth factor-CS interactions, a
similar study has also shown that TGF-β1 may be adsorbed onto
biomaterials such as SF composed of type I collagen (39). It is important to consider
strategies to immobilize and pattern proteins, such as growth
factors and other cytokines, to develop long-term tissue
engineering solutions (40–44). Factor immobilization may offer
numerous advantages, including improved protein stability and
reduced diffusion, as well as increased localization of the
therapeutic effects within the material. A previous study
demonstrated that TGF-β1 was immobilized in the SF-CS scaffold
through ionic interactions and covalent bonds (45). The effectiveness of adsorbing
TGF-β1 onto the SF-CS scaffold may be due to protection of the
molecule from proteolytic digestion, allowing a more sustained
release of TGF-β1 over time. This indicates that the growth factor
remains bioactive within the matrix for a long period of time
(46).
When constructing the TGF-β1-SF-CS scaffold for bone
tissue engineering, it is critical to maintain the bioactivity of
TGF-β1 as during this process TGF-β1 bioactivity is mostly like to
be destroyed by lyophilization and methanol crosslinking. During
the process of lyophilization, the use of a cryoprotectant may
reduce or even prevent the adverse effects of stress on protein
activity (47). Silk protein and
CS may be used as a cryoprotectant to maintain the activity of
TGF-β1 (48,49). Klibanov proved that the
conformation of protein treated with low concentrations of methanol
was basically unchanged and the covalent structure of protein
molecules was not destroyed (50). We implemented the following
measures in manufacturing the TGF-β1-SF-CS scaffold to prevent the
loss of TGF-β1 bioactivity: i) setting the lyophilization
temperature at −40°C and using SF and CS as cryoprotectants to
further maintain TGF-β1 activity; ii) selecting a methanol
concentration <10%; iii) designing the whole TGF-β1-SF-CS
manufacturing procedure as a physical process. The results of the
analysis of ALP activity and the CCK-8 assay revealed that there
was no clear degradation of TGF-β1 bioactivity. We immobilized
TGF-β1 such that the total amount immobilized on the SF-CS scaffold
was equivalent to the total soluble biomolecule content in the
TGF-β1 solution. This experiment revealed that, as expected, the
freely soluble TGF-β1 induced high bioactivity.
SEM analysis confirmed that subcellular microvillous
and lamellopodia extensions were employed by adherent cells to
establish anchorage fronts with the SF-CS substrate. Additionally,
a BMSC growth differentiation study was performed to further
evaluate the biocompatibility of these scaffolds. The seeding of
BMSCs onto TGF-β1-SF-CS scaffolds resulted in a completely flat and
elongated cell morphology, similar to what is typically observed in
standard cell culture flasks. These observations confirmed the
long-term cell viability and proliferation of BMSCs on TGF-β1-SF-CS
scaffolds, assuring its biocompatibility.
Nominal differences in cellular adhesion were
observed between BMSCs after osteogenesis induction. Additionally,
these results suggested that the SF-CS scaffolds were suitable for
the proposed in vitro studies as the initial attachment onto
the surface as well as the spreading and subsequent growth of BMSCs
was facilitated. The adhesion rate of BMSCs in the TGF-β1-SF-CS
group and in the SF-CS group significantly exceeded those in the
control groups (P<0.05). However, no statistically significant
differences (P>0.05) were observed between the SF-CS and the
TGF-β1-SF-CS scaffold groups. It proved that TGF-β1 was unable to
promote BMSC adhesion in the SF-CS scaffolds.
The number of viable cells at day 1 (after 24 h of
cell attachment) was taken as a reference point to compare the
growth and proliferation rate of BMSCs on the TGF-β1-SF-CS and
SF-CS scaffolds. It was observed that the absorbance index of the
tested groups increased in a time-dependent manner. However, there
was a TGF-β1-dependent effect of the scaffolds on cell
proliferation rate. At day 1, the viable cell number was not
significantly different among the three groups, indicating that
cell attachment to scaffolds was similar. With regard to cell
growth, from day 3, a significant difference (P<0.05) in cell
number was observed between the scaffolds groups and the control
group as well as between the TGF-β1-SF-CS and SF-CS scaffolds.
These results indicated that TGF-β1 significantly promoted the
growth and proliferation of BMSCs after osteogenesis induction
under these conditions. The in vitro experiments performed
in the present study also suggest that TGF-β1 may regulate bone
formation through a direct effect on BMSCs after osteogenesis
induction. Our results indicated a difference in the activity of
BMSCs in the different types of scaffold which may be due to: i)
the release of TGF-β1 from TGF-β1-SF-CS scaffolds in a sustained
manner thus, the released TGF-β1 was able to reach the cells more
quickly and homogeneously, and ii) TGF-β1 maintained its
bioactivity after being released from the TGF-β1-SF-CS scaffold.
Through the ALP and CCK-8 assay results, we basically determined
that in TGF-β1-SF-CS scaffolds, IGF-1 was evenly and continuously
released within a certain time period.
Due to the rapid immobilization of TGF-β1 in the
SF-CS scaffold and the sustained and effective local release of
growth factors after seeding BMSCs in the porous TGF-β1-SF-CS
scaffold, as expected, the level of proliferation and
differentiation of the BMSCs was controlled by the presence of
TGF-β1. The results obtained from the analysis of ALP activity and
the CCK-8 assay in vitro showed that the TGF-β1-SF-CS
scaffolds did not contain products toxic to cells and the
proliferation of BMSCs on the scaffolds increased regularly in a
time-dependent manner. The SEM images revealed the spindle-like
shape of the attached cells with filopodia-like extensions adhering
to the scaffold and connecting to adjacent BMSCs. Evidence of
cell-to-cell interactions and cell spreading may be considered as
signs of healthy BMSCs and indicative of a noncytotoxic response of
the cells on the supporting material (51,52). In the present study, TGF-β1
bioactivity was studied over the first 2 weeks of drug delivery. It
has been shown that TGF-β1 incorporated into the SF-CS scaffolds is
capable of maintaining bioactivity over at least 2 weeks,
suggesting that longer-term maintenance of bioactivity is
feasible.
To evaluate the mechanical recovery of bone grafts
in rabbit models of segmental radius defects, bending test
configurations have been reported (53–55). Our studies have shown that both
three-point bending resulted in similar outcomes when testing the
mechanical integrity of rabbit mandible (53). In agreement with this study, the
primary failure mode for bending was a short oblique fracture.
Additionally, the reported primary failure zone was constrained to
the location of the central contact in the three-point bending
whereas the four-point mode was reported to have a more uniform
stress distribution within the central loading span (56). Since the objective of this study
was to evaluate the interfacial integration of the scaffold to the
bone, three-point bending testing was selected, with the scaffold
lying completely within the loading span. Other confounding factors
such as loading to support span ratios, contact geometry and load
rates were matched to the suggestions in the literature (56).
In the present study, bony union was achieved as
early as 1 to 3 months after transplantation and an improved
mandibular shape was observed after sacrifice. We also found that
the BMD and NBV of the TGF-β1-SF-CS scaffold group was higher than
the SF-CS scaffold group at 4 weeks postoperatively. This indicated
that new bone formation and mineralization in the TGF-β1-SF-CS
scaffold group surpassed that occurring during the repair process
in the SF-CS scaffold group. Our data indicated that the SF-CS
scaffold was feasible and conferred that TGF-β1 possessses the
potential physiological benefit of accelerated reconstruction. The
results of CT scans, gross view and histological analyses
consistently showed that the TGF-β1-SF-CS scaffold group had the
strongest capacity for bone regeneration compared with the other
groups.
The capability of the TGF-β1-SF-CS scaffold to
enhance the viability and proliferation of BMSCs as a function of
time demonstrates its cytocompatibility and feasibility for bone
tissue engineering applications. TGF-β1 did not promote the
activity and proliferation of osteoblasts significantly at early
time-points; however, it had a marked effect when culturedfor
longer periods of time (3–10 days), demonstrating that the effect
of TGF-β1, which promotes the activity and proliferation of BMSCs,
is time-dependent. In the in vivo experiment, TGF-β1 did not
induce significantly more new bone formation at early time-points
(4 weeks) whereas it had a marked effect at later time-points
(after 8 weeks), demonstrating that the effect of TGF-β1 which
promotes the activity and proliferation of osteoblasts is
time-dependent. These results indicate that correct TGF-β1 delivery
strategies greatly affect osteogenic differentiation and are of
importance when designing a delivery system. The ideal duration of
TGF-β1 delivery is dependent on the drug delivery application. For
example, for slowly progressing bone healing, controlled-release
formulations leading to sustained improvements in patient symptoms
and/or disease conditions allow for more convenient dosage and
increased compliance with treatment.
Our data suggested that the anchorage of TGF-β1 onto
the SF-CS scaffold may achieve the intended natural configuration,
as TGF-β1 was protected and available for activation. The
TGF-β1-SF-CS scaffold exerts positive effects on matrix production
as well as the proliferation and differentiation of BMSCs. Our data
supports the potential use of SF-CS as a biocompatible scaffold.
Our findings demonstrated that TGF-β1-SF-CS scaffolds are suitable
substrates for bone tissue engineering and TGF-β1 confers the
potential physiological benefit of accelerated reconstruction. We
explored the immobilization of TGF-β1 within the SF-CS scaffold to
evaluate relative biomolecule bioactivity as a function of
presentation modality. Future bone tissue engineering studies are
warranted, focusing on how TGF-β1 combined with the scaffold, and
whether the sustained release of TGF-β1 is maintained and enhanced
in longer-term studies, or examining whether the culture of
different types of cells on the TGF-β1-SF-CS scaffolds yields
similar outcomes. Our future studies aim to examine the potential
repair reaction of the TGF-β1-SF-CS scaffolds in a bone defect
model in large animals. We also aim to explore additional soluble
factors and exploit immobilization techniques to identify
combinations of biomolecules capable of improving scaffolds. The
materials developed here are also likely to be useful as 3D
templates for bone tissue engineering.
In conclusion, we compared, for the first time to
the best of our knowledge, the physiochemistry and osteogenesis of
TGF-β1-SF-CS scaffolds and SF-CS scaffolds. Our results confirmed
that TGF-β1, a bioactive growth factor, has a significant capacity
to improve the bioactivity of SF-CS scaffolds. The results showed
that TGF-β1-SF-CS scaffolds exhibit good biocompatibility and
extensive osteoconductivity with the host bone. Moreover, the
introduction of TGF-β1 to the SF-CS scaffolds markedly enhanced the
efficiency of new bone formation, particularly 12 weeks after
implantation. The TGF-β1-SF-CS scaffolds possessed as good
biocompatibility and osteogenesis as the hybrid ones. All these
results indicated that the TGF-β1-SF-CS scaffolds fulfiled the
basic requirements of an ideal scaffold for use in bone tissue
engineering, and have the potential to be applied in orthopedic,
reconstructive and maxillofacial surgery.
Acknowledgments
The present study was supported by the Science and
Technology Plan Project of Liaoning Province (grant no.
2012-B-00002012225082).
References
|
1
|
Vacanti JP and Langer R: Tissue
engineering: the design and fabrication of living replacement
devices for surgical reconstruction and transplantation. Lancet.
354(Suppl 1): SI32–SI34. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Scheller EL, Krebsbach PH and Kohn DH:
Tissue engineering: state of the art in oral rehabilitation. J Oral
Rehabil. 36:368–389. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hankenson KD, Dishowitz M, Gray C and
Schenker M: Angiogenesis in bone regeneration. Injury. 42:556–561.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Marsell R and Einhorn TA: The biology of
fracture healing. Injury. 42:551–555. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Andrew JG, Hoyland J, Andrew SM, Freemont
AJ and Marsh D: Demonstration of TGF-beta 1 mRNA by in situ
hybridization in normal human fracture healing. Calcif Tissue Int.
52:74–78. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Baylink DJ, Finkelman RD and Mohan S:
Growth factors to stimulate bone formation. J Bone Miner Res.
8(Suppl 2): S565–S572. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bonewald LF and Mundy GR: Role of
transforming growth factor-beta in bone remodeling. Clin Orthop
Relat Res. 250:261–276. 1990.PubMed/NCBI
|
|
8
|
Connelly JT, Wilson CG and Levenston ME:
Characterization of proteoglycan production and processing by
chondrocytes and BMSCs in tissue engineered constructs.
Osteoarthritis Cartilage. 16:1092–1100. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Mouw JK, Connelly JT, Wilson CG, Michael
KE and Levenston ME: Dynamic compression regulates the expression
and synthesis of chondrocyte-specific matrix molecules in bone
marrow stromal cells. Stem Cells. 25:655–663. 2007. View Article : Google Scholar
|
|
10
|
Johnstone B, Hering TM, Caplan AI,
Goldberg VM and Yoo JU: In vitro chondrogenesis of bone
marrow-derived mesenchymal progenitor cells. Exp Cell Res.
238:265–272. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Murphy CM, O'Brien FJ, Little DG and
Schindeler A: Cell-scaffold interactions in the bone tissue
engineering triad. Eur Cell Mater. 26:120–132. 2013.PubMed/NCBI
|
|
12
|
Roberts AB and Sporn MB: Physiological
actions and clinical applications of transforming growth
factor-beta (TGF-beta). Growth Factors. 8:1–9. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Pittenger MF, Mackay AM, Beck SC, Jaiswal
RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S and
Marshak DR: Multilineage potential of adult human mesenchymal stem
cells. Science. 284:143–147. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cashman JD, Eaves AC, Raines EW, Ross R
and Eaves CJ: Mechanisms that regulate the cell cycle status of
very primitive hematopoietic cells in long-term human marrow
cultures. I. Stimulatory role of a variety of mesenchymal cell
activators and inhibitory role of TGF-beta. Blood. 75:96–101.
1990.PubMed/NCBI
|
|
15
|
Spinella-Jaegle S, Roman-Roman S, Faucheu
C, Dunn FW, Kawai S, Galléa S, Stiot V, Blanchet AM, Courtois B,
Baron R and Rawadi G: Opposite effects of bone morphogenetic
protein-2 and transforming growth factor-beta1 on osteoblast
differentiation. Bone. 29:323–330. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Park SJ, Lee KY, Ha WS and Park SY:
Structural changes and their effect on mechanical properties of
silk fibroin/chitosan blends. J Appl Polym Sci. 74:2571–2575. 1999.
View Article : Google Scholar
|
|
17
|
El Sayed K, Marzahn U, John T, Hoyer M,
Zreiqat H, Witthuhn A, Kohl B, Haisch A and Schulze-Tanzil G:
PGA-associated heterotopic chondrocyte cocultures: implications of
nasoseptal and auricular chondrocytes in articular cartilage
repair. J Tissue Eng Regen Med. 7:61–72. 2013. View Article : Google Scholar
|
|
18
|
De Santis R, Gloria A, Russo T, D'Amora U,
Zeppetelli S, Dionigi C, Sytcheva A, Herrmannsdörfer T, Dediu V and
Ambrosio L: A basic approach toward the development of
nanocomposite magnetic scaffolds for advanced bone tissue
engineering. J Appl Polym Sci. 122:3599–3605. 2011. View Article : Google Scholar
|
|
19
|
Tong S, Xu DP, Liu ZM and Wang XK:
Construction and in vitro characterization of three-dimensional
silk fibroinchitosan scaffolds. Dent Mater J. 34:475–484. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gobin AS, Froude VE and Mathur AB:
Structural and mechanical characteristics of silk fibroin and
chitosan blend scaffolds for tissue regeneration. J Biomed Mater
Res A. 74:465–473. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Whang K, Tsai DC, Nam EK, Aitken M,
Sprague SM, Patel PK and Healy KE: Ectopic bone formation via
rhBMP-2 delivery from porous bioabsorbable polymer scaffolds. J
Biomed Mater Res. 42:491–499. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Uebersax L, Merkle HP and Meinel L:
Insulin-like growth factor I releasing silk fibroin scaffolds
induce chondrogenic differentiation of human mesenchymal stem
cells. J Control Release. 127:12–21. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Li C, Vepari C, Jin HJ, Kim HJ and Kaplan
DL: Electrospun silk-BMP-2 scaffolds for bone tissue engineering.
Biomaterials. 27:3115–3124. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hosseinkhani H, Hosseinkhani M,
Khademhosseini A and Kobayashi H: Bone regeneration through
controlled release of bone morphogenetic protein-2 from 3-D tissue
engineered nano-scaffold. J Control Release. 117:380–386. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Luginbuehl V, Wenk E, Koch A, Gander B,
Merkle HP and Meinel L: Insulin-like growth factor I-releasing
alginate-tricalciumphosphate composites for bone regeneration.
Pharm Res. 22:940–950. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Chung YI, Ahn KM, Jeon SH, Lee SY, Lee JH
and Tae G: Enhanced bone regeneration with BMP-2 loaded functional
nanoparticle-hydrogel complex. J Control Release. 121:91–99. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Li B, Brown KV, Wenke JC and Guelcher SA:
Sustained release of vancomycin from polyurethane scaffolds
inhibits infection of bone wounds in a rat femoral segmental defect
model. J Control Release. 145:221–230. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Long JL and Tranquillo RT: Elastic fiber
production in cardiovascular tissue-equivalents. Matrix Biol.
22:339–350. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Shi J, Wang A, Sen S, Wang Y, Kim HJ,
Mitts TF and Hinek A: Insulin induces production of new elastin in
cultures of human aortic smooth muscle cells. Am J Pathol.
180:715–726. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kim UJ, Park J, Kim HJ, Wada M and Kaplan
DL: Three-dimensional aqueous-derived biomaterial scaffolds from
silk fibroin. Biomaterials. 26:2775–2785. 2005. View Article : Google Scholar
|
|
31
|
Li C, Vepari C, Jin HJ, Kim HJ and Kaplan
DL: Electrospun silk-BMP-2 scaffolds for bone tissue engineering.
Biomaterials. 27:3115–3124. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Chou CH, Cheng WT, Lin CC, Chang CH, Tsai
CC and Lin FH: TGF-beta1 immobilized tri-co-polymer for articular
cartilage tissue engineering. J Biomed Mater Res B Appl Biomater.
77:338–348. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Sohier J, Moroni L, van Blitterswijk C, de
Groot K and Bezemer JM: Critical factors in the design of growth
factor releasing scaffolds for cartilage tissue engineering. Expert
Opin Drug Deliv. 5:543–566. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Lyons F, Partap S and O'Brien FJ: Part 1:
scaffolds and surfaces. Technol Health Care. 16:305–317.
2008.PubMed/NCBI
|
|
35
|
Mooney DJ, Baldwin DF, Suh NP, Vacanti JP
and Langer R: Novel approach to fabricate porous sponges of poly
(D,L-lactic-co-glycolic acid) without the use of organic solvents.
Biomaterials. 17:1417–1422. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Chung HJ and Park TG: Surface engineered
and drug releasing pre-fabricated scaffolds for tissue engineering.
Adv Drug Deliv Rev. 59:249–262. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Raman R, Sasisekharan V and Sasisekharan
R: Structural insights into biological roles of
protein-glycosaminoglycan interactions. Chem Biol. 12:267–277.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Gama CI, Tully SE, Sotogaku N, Clark PM,
Rawat M, Vaidehi N, Goddard WA III, Nishi A and Hsieh-Wilson LC:
Sulfation patterns of glycosaminoglycans encode molecular
recognition and activity. Nat Chem Biol. 2:467–473. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Hempel U, Hintze V, Möller S,
Schnabelrauch M, Scharnweber D and Dieter P: Artificial
extracellular matrices composed of collagen I and sulfated
hyaluronan with adsorbed transforming growth factor β1 promote
collagen synthesis of human mesenchymal stromal cells. Acta
Biomater. 8:659–666. 2012. View Article : Google Scholar
|
|
40
|
Ker EDF, Chu B, Phillippi JA, Gharaibeh B,
Huard J, Weiss LE and Campbell PG: Engineering spatial control of
multiple differentiation fates within a stem cell population.
Biomaterials. 32:3413–3422. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Shen YH, Shoichet MS and Radisic M:
Vascular endothelial growth factor immobilized in collagen scaffold
promotes penetration and proliferation of endothelial cells. Acta
Biomater. 4:477–489. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Oh SH, Kim TH and Lee JH: Creating growth
factor gradients in three dimensional porous matrix by
centrifugation and surface immobilization. Biomaterials.
32:8254–8260. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Anderson SM, Chen TT, Iruela-Arispe ML and
Segura T: The phosphorylation of vascular endothelial growth factor
receptor-2 (VEGFR-2) by engineered surfaces with electrostatically
or covalently immobilized VEGF. Biomaterials. 30:4618–4628. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Martin TA, Caliari SR, Williford PD,
Harley BA and Bailey RC: The generation of biomolecular patterns in
highly porous collagen-GAG scaffolds using direct photolithography.
Biomaterials. 32:3949–3957. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Weadock K, Olson RM and Silver FH:
Evaluation of collagen crosslinking techniques. Biomater Med
Devices Artif Organs. 11:293–318. 1983–1984. View Article : Google Scholar
|
|
46
|
Hortensius RA and Harley BA: The use of
bioinspired alterations in the glycosaminoglycan content of
collagen-GAG scaffolds to regulate cell activity. Biomaterials.
34:7645–7652. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Haines AH: Non-equivalence of D- and
L-trehalose in stabilising alkaline phosphatase against
freeze-drying and thermal stress. Is chiral recognition involved?
Org Biomol Chem. 4:702–706. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Kreilgaard L, Jones LS, Randolph TW,
Frokjaer S, Flink JM, Manning MC and Carpenter JF: Effect of Tween
20 on freeze-thawing- and agitation-induced aggregation of
recombinant human factor XIII. J Pharm Sci. 87:1597–1603. 1998.
View Article : Google Scholar
|
|
49
|
Pikal-Cleland KA and Carpenter JF:
Lyophilization-induced protein denaturation in phosphate buffer
systems: monomeric and tetrameric beta-galactosidase. J Pharm Sci.
90:1255–1268. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Klibanov AM: Enzymatic catalysis in
anhydrous organic solvents. Trends Biochem Sci. 14:141–144. 1989.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Nwe N, Furuike T and Tamura H: The
mechanical and biological properties of chitosan scaffolds for
tissue regeneration templates are significantly enhanced by
chitosan from Gongronella butleri. Materials (Basel). 2. pp.
374–398. 2009, View Article : Google Scholar
|
|
52
|
Neamnark A, Sanchavanakit N, Pavasant P,
Bunaprasert T, Supaphol P and Rujiravanit R: In vitro
biocompatibility evaluations of hexanoyl chitosan film. Carbohydr
Polym. 68:166–172. 2007. View Article : Google Scholar
|
|
53
|
Kim J, Yang HJ, Cho TH, Lee SE, Park YD,
Kim HM, Kim IS, Seo YK, Hwang SJ and Kim SJ: Enhanced regeneration
of rabbit mandibular defects through a combined treatment of
electrical stimulation and rhBMP-2 application. Med Biol Eng
Comput. 51:1339–1348. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Cui X, Zhang B, Wang Y and Gao Y: Effects
of chitosan-coated pressed calcium sulfate pellet combined with
recombinant human bone morphogenetic protein 2 on restoration of
segmental bone defect. J Craniofac Surg. 19:459–465. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Shafiei Z, Bigham AS, Dehghani SN and
Nezhad ST: Fresh cortical autograft versus fresh cortical allograft
effects on experimental bone healing in rabbits: radiological,
histopathological and biomechanical evaluation. Cell Tissue Bank.
10:19–26. 2009. View Article : Google Scholar
|
|
56
|
Draper ER and Goodship AE: A novel
technique for four-point bending of small bone samples with
semi-automatic analysis. J Biomech. 36:1497–1502. 2003. View Article : Google Scholar : PubMed/NCBI
|