Introduction
As a non-invasive wound therapy method, negative
pressure wound therapy (NPWT) has been extensively applied to
accelerate wound healing in chronic, acute and complex wounds
(1). The technique includes
negative pressure and an airtight wound, and the suction force
created by the NPWT equipment that helps to drain excess fluid,
leading to the alliviation of wound edema and bacterial count
reduction, thus promoting granulation tissue formation, as well as
affecting blood flow perfusion in the wound (2,3).
For accelerating the speed of wound healing, NPWT
has mainly been applied to wounds that are open and hard to
agglutinate. The relevant molecular mechanisms and biological
effects primarily include the following (4,5):
macrodeformation, microdeformation, removal of exudate and
maintaining a warm and moist microenvironment on the surface of the
wound. These molecular mechanisms and biological effects play vital
roles in inducing the deformation of wound tissue (1,6);
in addition, tissue deformation stimulates wound angiogenesis, the
overexpression of growth factors and fresh granulation tissue
formation (7,8). On the other hand, NPWT can promote
collagen deposition, a vital component of granulation tissue in the
wound site, which serves as a critical component of blood vessels
and affects wound healing (4).
Since the formation of fresh granulation tissue is a critical
process of the tissue proliferation stage during wound healing
(9), the quality of fresh
granulation tissue could determine the rate of wound healing
(9).
Although the positive therapeutic effects of NPWT
have been generally recognized, its side-effects are not yet fully
understood. There are numerous controversies as to the side-effects
and the identification of the related molecular mechanisms,
particularly in terms of blood flow perfusion following NPWT
treatment. Xia et al (10)
found that blood flow perfusion was significantly increased on the
surface of extremity wounds following NPWT treatment. However, a
recent study reported that blood flow perfusion was decreased in
the wound following NPWT treatment (11). It is well known that alterations
in blood flow can be affected by many factors, and previous studies
have mostly focused on the application of physical methods for
detecting alterations in blood flow in the wound (11,12). Therefore, in the present study,
and to the best of our knowledge, for the first time, we
investigate and explore alterations in wound blood flow coupled
with microvascular characteristics and relevant expression levels
on the molecular level using blood flow physical test methods
following NPWT treatment.
In the present study, a tyrosine kinase receptor-2
(Tie-2) inhibitor was successfully applied to block vessel
maturation and pericyte proliferation. Microvascular blood flow
perfusion was detected using a laser Doppler blood perfusion
imager, and the expression levels of angiogenin-1 (Ang-1), the
tyrosine phosphorylation of Tie-2 (p-Tie-2), α-smooth muscle actin
(α-SMA) and collagen type IV were detected and analyzed through
immunohistochemistry, immunofluorescence, RT-qPCR and western blot
analysis. We further discuss whether blood flow perfusion is
increased following microvessel maturation and the number of
pericytes, and we also further explore the underlying molecular
mechanisms. In addition, we investigate the association between
pericytes and collagen type IV.
Materials and methods
Animals
A total of 96 Sprague-Dawley rats with a male:female
at a ratio of 1:1, weighing approximately 210–250 g, were obtained
from the Experimental Animal Central Research Laboratory at Wuhan
University (Wuhan, China). All rats were individually caged under
specific pathogen-free conditions, housed under a controlled
temperature of approximately 24±1°C and a relative humidity of
45–55% and a 12-h light/12-h dark cycle. The rats were allowed
ad libitum to drink filtered water and eat standard animal
feed. All experiments were carried out between 09:00 and 17:00 h.
Treatment of the animals was carried out in strict accordance with
the recommendations described in the Guide for the Care and use of
Laboratory Animals of the National Institutes of Health. The
experimental protocol was approved by the Committee on the Ethics
of Animal Experiments of Wuhan University, Wuhan, China. All
surgical procedures were performed under chloral hydrate
anesthesia, and efforts were made to minimize animal suffering.
Induction and maintenance of diabetes in
rats
A total of 96 rats were administered a single
injection of freshly dissolved streptozotocin (STZ; 100 mg/kg;
Sigma, St. Louis, MO, USA) in a 0.1 mmol/l citrate buffer (pH 4.5)
intraperitoneally after overnight fasting, as previously described
(13). The rat diabetic model was
successfully established by detecting the fasting blood glucose
levels of the rats (measured using a glucometer; Johnson &
Johnson, Milpitas, CA, USA).
Three days after the STZ injection, the rats were
screened for serum glucose levels. The diabetes mellitus status was
determined, with continuation to the next step of the experiment if
the fasting blood glucose level remained at >300 mg/dl for 7
days. All the rat fasting blood glucose levels were detected 3
times a week. Minimal insulin doses of 0.1–0.2 units/mouse were
administered by subcutaneous injection if the fasting serum glucose
levels of the rats were >450 mg/dl. The fasting blood glucose
levels of the rats were sustained at a level >300 mg/dl during
the experimental period.
Wound protocol and animal grouping
The dorsa of all the animals were completely shaved
with an electrical clipper 24 h prior to the experiment. General
intraperitoneal anesthesia was conducted by injection of 350 mg/kg
chloral hydrate (7%) 5 min prior to the surgery. The dorsa of all
the rats were disinfected with povidone iodine solution and 75%
medical alcohol, and a 3.5×3.5 cm area of skin and panniculus
carnosus were removed to create a full-thickness diabetic wound,
the depth extending to the deep fascia on the dorsum. All animals
were randomly divided into the following 4 groups: NPWT, NPWT +
Tie-2 inhibitor (NPWT + Tie), gauze and gauze + Tie-2 inhibitor
(gauze + Tie). In the NPWT and NPWT + Tie groups, a total of 48
rats with diabetes mellitus wounds were covered with a 3.5×3.5 cm
area of black polyurethane (Pu) foam (VSD Medical Technology Co.,
Ltd., Hubei, China) and then with a vacuum-assisted closure device
(Wego, Shandong, China). The negative pressure value was set
constantly at continuous −125 mmHg. The VAC device affected neither
the ambulation nor the diet and lifestyle of the treated animals
(data not shown). In the gauze and gauze + Tie groups, a total of
48 rats with diabetes mellitus wounds received petrolatum gauze
treatment. In addition, in the NPWT + Tie and gauze + Tie groups, a
total of 48 rats with diabetes mellitus were selected to receive an
intraperitoneal injection of 50 mg/kg Tie-2 kinase inhibitor
(Selleck Chemicals, Houston, TX, USA) twice a week, which was
dissolved into a 1 ml of the vehicle (5% ethanol, 5% Cremophor and
90% distilled water), as previously described (14) and according to the supplier's
instructions.
In total, 24 rats were euthanized by cervical
dislocation on the 1st, 3rd, 7th and 10th day after the surgery [24
rats (6 from each group) were sacrificed on each selected day]. The
wound fresh granulation tissue samples were harvested aseptically
from the surface of the wounds. Of the samples that were bisected,
half were placed in 4% neutral formalin formaldehyde (Aspen, Wuhan,
China) and the other half were snap-frozen in liquid nitrogen and
stored at −80°C.
Wound surface blood flow detection
On the 1st, 3rd, 7th and 10th day after the surgery,
a laser Doppler blood perfusion imager (LDPI) and PeriScan PIM 3
system (both from Perimed, Stockholm, Sweden) was used to scan the
blood flow of the wound surface, as previously described (15). Subsequently, PIM software version
1.5 (Perimed) was applied to analyze blood flow changes on the
scanned images, as previously described (10).
Wound contraction analysis
The skin wound site in each rat was digitally
photographed at the prescribed time points, and the wound
contraction was measured using Image Pro-Plus version 6.0 (Media
Cybernetics, Bethesda, MA, USA) under double-blinded conditions.
The wound areas of the each rat were normalized to the initial
wound size, and the wound healing rate was expressed as a
percentage of wound contraction as follows: [(wound area on day 0 −
wound area on day n)/(wound area on day 0)] ×100% (n=1, 3, 7 and
10). All data were expressed as the means ± SD, and compared among
the different groups using one-way analysis of variance.
Immunohistochemistry
The samples were embedded in paraffin wax and
sectioned serially into 5-µm-thick slices. The slices were
stained with standard haematoxylin and eosin (H&E) (Boster
Biological Technology, Hubei, China) stain for observation of the
histological changes in the wounds.
Antibodies against rat Ang-1 (ab102015; 1:200;
Abcam, Cambridge, uK), Tie-2 (sc-9026; 1:200; Santa Cruz
Biotechnology, Inc., Dallas, TX, USA), α-SMA (ab32575; 1:200) and
collagen type IV (ab6586; 1:200) (both from Abcam) served as the
primary antibodies. The sections were dewaxed and hydrated for
immunohistochemical staining. First, the activity of endogenous
peroxidase was quenched with (3%) hydrogen peroxide (Aspen).
Second, citrate buffer was used for antigen retrieval. The sections
were then treated with microwave (Galanz, Guangdong, China) set at
500 W for 5 min. Third, the sections were incubated with primary
antibodies and set at 4°C overnight. Fourth, the sections were
washed 3 times with phosphate-buffered saline (PBS; Bioyear, Hubei,
China), and subsequently the sections were incubated with goat
anti-rabbit secondary antibody (1:500; Aspen) for 30 min. Fifth,
the sections were incubated with avidin-biotin complex (VECTASTAIN
Elite ABC kit; Vector Laboratories, Inc., Burlingame, CA, USA) for
30 min. Finally the reaction was visualised by using
3′3-diaminobenzidine (Dako, Glostrup, Denmark), and with
hematoxylin and eosin to stain the nuclei (Sigma-Aldrich). The
negative control groups staining experiments were performed using
1:1,000 normal goat serum substituting for primary antibody in each
section. The images were captured using a light microscope (BX51WI;
Olympus Corp., Tokyo, Japan).
Immunofluorescence staining
A double-labeling immunofluorescence technique was
applied to analyze the number of pericytes and the microvascular
pericyte coverage index (MPI). The pericyte coverage of the
microvessels was detected using anti-CD31 (ab9498; 1:400) and
anti-α-SMA (ab124964; 1:400) antibodies (both from Abcam). The
sections were blocked with BSA (5%) for 2 h and incubated with
primary antibody at 4°C overnight. After thorough washing, the
sections were incubated with secondary goat anti-rabbit secondary
antibodies (Ki67, AS1110; CD31, AS1111; α-SMA, AS1110; 1:400; all
from Aspen) for 1 h in the dark. Subsequently, the sections were
incubated in 4′6-diamidino-2 phenylindole (DAPI; Aspen) for nuclei
staining. All images were captured using a fluorescence microscope
(Eclipse TE2000-E; Nikon Corp., Tokyo, Japan), and images were
merged by using Image Pro-Plus version 6.0 software (Media
Cybernetics, Inc., Rockville, MD, USA).
Quantification of microvessel density
(MVD)
The MVD counting technique has been widely used to
assess blood vessel number (16).
MVD was quantified as the average number of microvessels per
viewing field, and red CD31 staining was used as an endothelial
cell marker. CD31-positive endothelial cells and neglected vessels
with or without lumen were counted under a power field of 20×10 in
5 randomly selected fields from 3 separate sections of each
sample.
Quantification of the MPI
MPI was used to assess the maturity of new blood
vessels, as previously described (17,18). MPI was correspondingly established
by quantifying the percentage of CD31-positive microvessels that
exhibited co-localization of endothelial cell staining (CD31) and
pericyte immunostaining (α-SMA) under a power field of 10×20, as
previously described (18). A
single endothelial cell was regarded as a unit of quantification
regardless of whether it formed a tube, and a pericyte was defined
as a single layer of α-SMA-positive cells co-localizing with
CD31-positive cells. For MPI quantification, at least 5
non-overlapping microscopic fields per section were independently
analyzed under double-blinded conditions. The MPI was expressed as
the α-SMA/CD31 ratio.
Quantitative RT-PCR (RT-qPCR)
Total RNA was extracted using an RNeasy mini kit
(Qiagen AB, Sollentuna, Sweden), and 5 µg RNA was reverse
transcribed into cDNA using the RevertAid First Strand cDNA
Synthesis kit (Fermentas; Thermo Fisher Scientific, Inc., Waltham,
MA, USA) to a final reaction volume of 20 µl and an S1000
Thermal Cycler (Bio-Rad Laboratories, Inc., Hercules, CA, USA) at
65°C for 5 min, cooling on ice and 42°C for 60 min, according to
the manufacturer's protocol. The reaction was terminated by heating
at 70°C for 5 min. The primer sequences are as follows: Ang-1
forward, 5′-TAA CCT CGC CCT GCA AAG AG-3′ and reverse primer,
5′-CTG TAT GCT TGC AGG TGG TGAT-3′; α-SMA forward, 5′-CAA CCC CTA
TAC AAC CAT CAC AC-3′ and reverse primer, 5′-CCC AAA CTG CTT GCG
TAA CC-3′; collagen IV forward, 5′-ACT GTG GAT TGG CTA TTC CTT
TG-3′ and reverse primer, 5′GCT TCT TGA ACA TCT CGC TTC TC-3′; and
glyceraldehyde 3-phosphate dehydrogenase (GAPDH) forward, 5′-CGC
TAA CAT CAA ATG GGG TG-3′ and reverse primer, 5′-TTG CTG ACA ATC
TTG AGG GAG-3′.
Following DNase treatment to remove genomic DNA,
RT-qPCR was performed to a final volume of 20 µl using 1
µl template cDNA, 10 µl SYBR qPCR mix (2X; Toyobo
Co., Ltd., Osaka, Japan), 6.6 µl
diethylpyrocarbonate-treated water, 1 µl forward primer (5
µm), 1 µl reverse primer (5 µm), 0.4 µl
ROX reference dye (50X) on a iQ5 Real-Time PCR detection system
(Bio-Rad Laboratories, Inc.). Respective negative (no cDNA) and RT
controls were used for each gene. The thermocycling profile for
SYBR-reen RT-qPCR was as follows: initially set at 95°C and
sustained for 1 min for the initial denaturation step; followed by
40 cycles of degeneration with the temperature set at 95°C, which
was held for 15 sec, then set at 60°C and sustained for 15 sec for
annealing; and, finally, the elongation step, involving a 60 sec
hold at 72°C. Each sample was run in triplicate and quantified
using the 2−ΔΔCt method to determine relative mRNA
expression levels.
Western blot analysis
The samples were homogenized and total proteins
extracted using a radioimmunoprecipitation assay (RIPA) buffer
(Beyotime Institute of Biotechnology, Haimen, China). A BCA kit
(Beyotime Institute of Biotechnology) was used to determine the
protein concentration. The proteins were loaded on sodium dodecyl
sulfate (SDS) polyacrylamide gels (10%) (Aspen), transferred onto
nitrocel-lulose membranes (Pall Corp., New York, NY, USA), and
blocked in non-fat dry milk (5%) at room temperature to set for 2
h. The membranes were incubated overnight at 4°C with primary
antibodies against Ang-1 (1:1,000; ab102015; Abcam), p-Tie-2
(1:1,500; sc-9026; Santa Cruz Biotechnology, Inc.), α-SMA (1:2,000;
ab32575), collagen type IV (1:1,500; ab6586) and GAPDH (1:5,000;
ab37168) (all from Abcam). Subsequently, the membranes were
incubated with horseradish peroxidase-conjugated secondary antibody
(Aspen) for 1 h, and finally the membranes were detected using an
enhanced chemiluminescence substrate (Beyotime Institute of
Biotechnology).
Statistical analysis
All data are presented as the means ± SD and
statistical significance was assessed by one-way analysis of
variance (ANOVA). SPSS 18.0 software (SPSS, Chicago, IL, USA) was
used for statistical analysis. Differences between groups were
considered statistically significant at P<0.05 or P<0.01.
Results
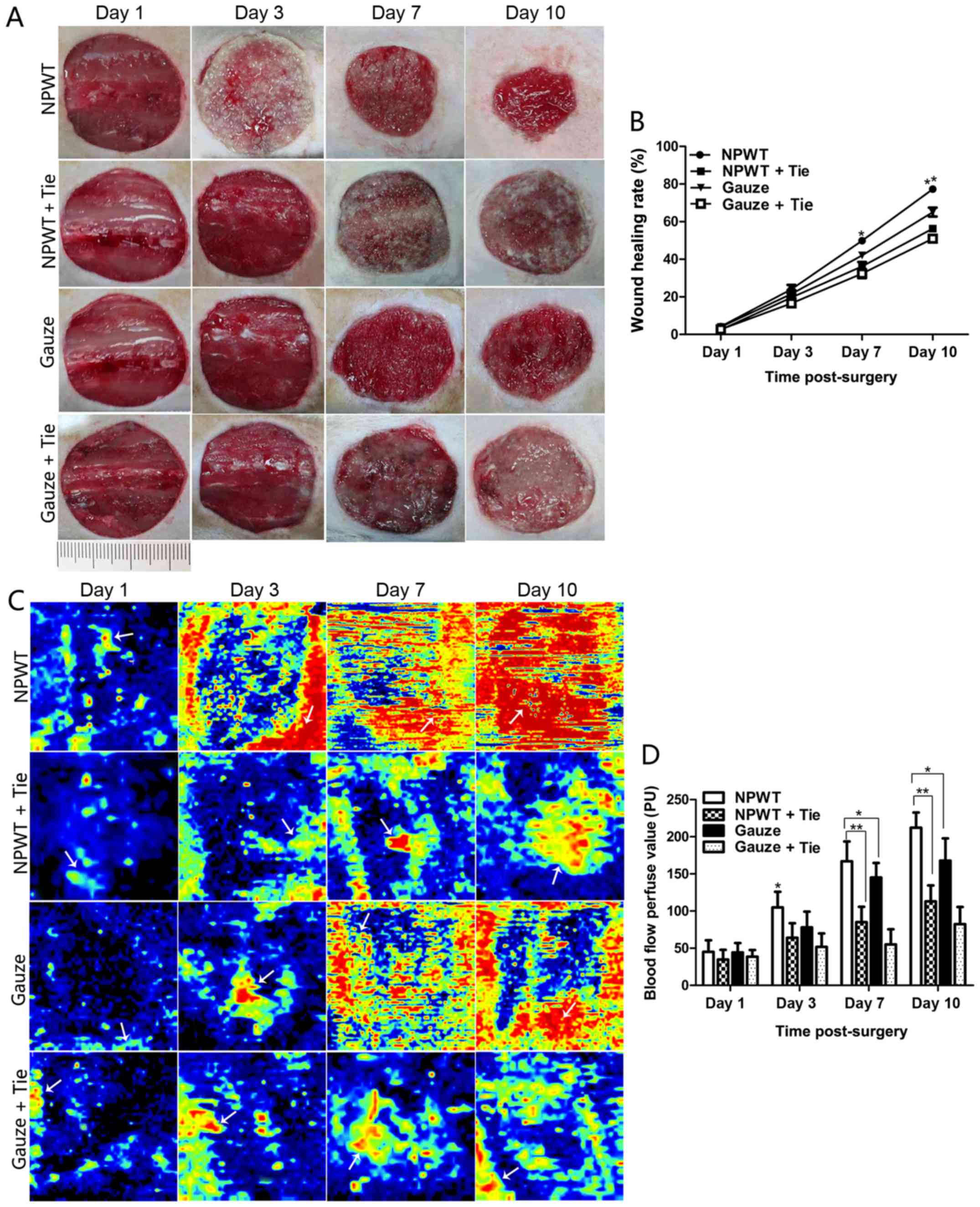
Wound contraction
The wound contraction was not markedly altered in
the early stages (1–3 days) of wound healing in the 4 groups,
although a small number of granulation tissue implants in the wound
was observed in the NPWT group on the 3rd day; however, there was
an abundance of fresh granulation tissues filled in the surface
area of the wound accompanied by marked wound contraction in the
NPWT group on the 7th day (Fig.
1A). In the other groups, less granulation tissue was found in
the wounds, particularly in the NPWT + Tie and gauze + Tie groups
on the 7th day. The percentage of wound contraction in the NPWT
group was significantly higher than those in the other groups at
day 7 and 10 (P<0.05) (Fig.
1B). On the 10th day, wound contraction was significantly
greater in the NPWT group than in the other groups (P<0.05 for
the gauze group, P<0.01 for the NPWT + Tie group or gauze + Tie
group) (Fig. 1B).
Changes in microvessel blood flow
perfusion
Wound blood flow perfusion is shown in Fig. 1C. The results revealed that the
areas of blood flow perfusion were gradually extended and enlarged
in the NPWT group from the 3rd to the 10th day. However, there were
fewer areas of blood perfusion in the gauze group during that time
period. The wound blood flow perfusion value gradually increased
from the 3rd to the 10th day in the NPWT group and gauze group, and
it was significantly higher in the NPWT group than in the gauze
group during that time period (P<0.05; Fig. 1D). In the NPWT + Tie and gauze +
Tie groups, the area of blood flow perfusion was the least among
all the groups, and the blood flow perfusion value was also the
lowest among all the groups, with the difference being
statistically significant compared to that in the NPWT group from
the 7th to the 10th day (P<0.01; Fig. 1D).
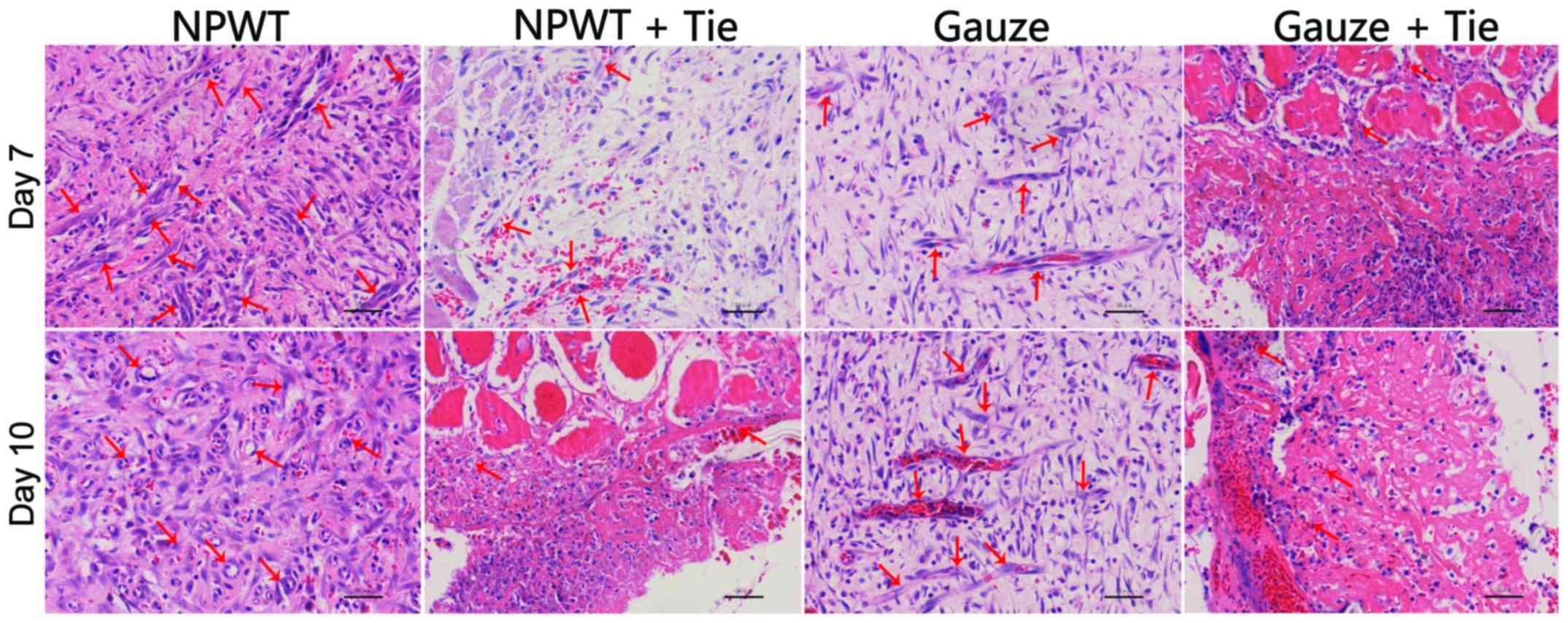
Histopathological analysis
In the early stages, in the NPWT group, the number
of new blood vessels was higher than that in the other groups, and
the degrees of inflammatory cell infiltration and epidermal
necrolysis were more slight in the NPWT group than in the other
groups. On the 7th and 10th day, in the NPWT group, the new
microvessels were abundant, and the collagen fibrils were
distributed compactly and regularly; by contrast, compared to the
NPWT group, the lower number of new blood vessels in the NPWT + Tie
and gauze + Tie groups, and the collagen fibrils were broken
(Fig. 2).
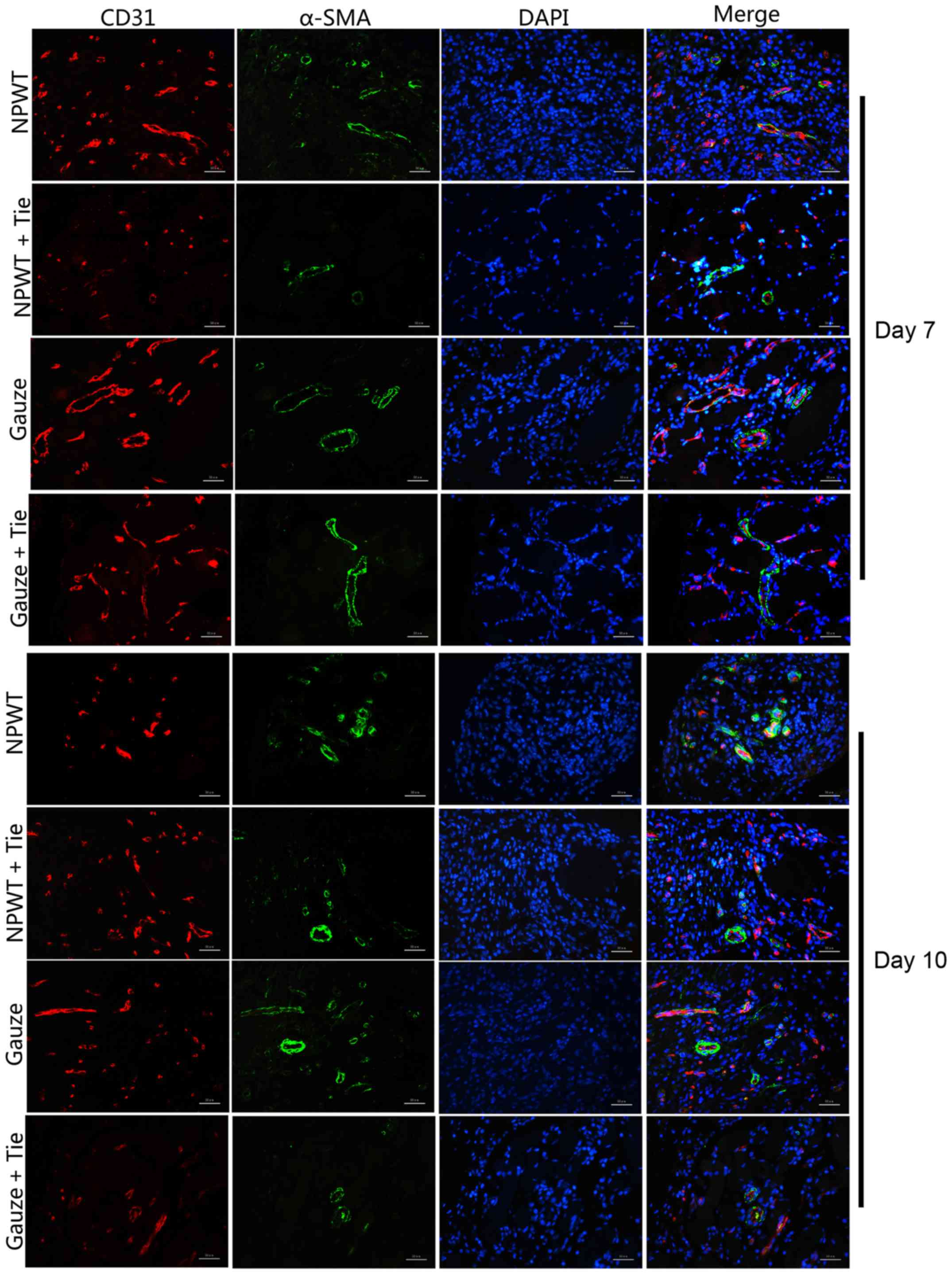
MVD
The double immunofluorescence marker, CD31, and
α-SMA were combined to mark vascular endothelial cells and
microvascular pericytes (Fig. 3).
On the 1st day, a few CD31-positive cells were observed in the NPWT
group, and a small amount of endothelial cells was detected with
red marker CD31-positive cells in the NPWT group at day 3 (data not
shown). On the 7th day, a large amount of endothelial cells was
detected; in addition, a small number of endothelial cells was
observed in the gauze group on the 3rd and 7th day, although it was
less than that in the NPWT group. the In NPWT + Tie and gauze + Tie
groups, the number of endothelial cells with red marked
CD31-positive cells was less than that in the gauze group. The MVD
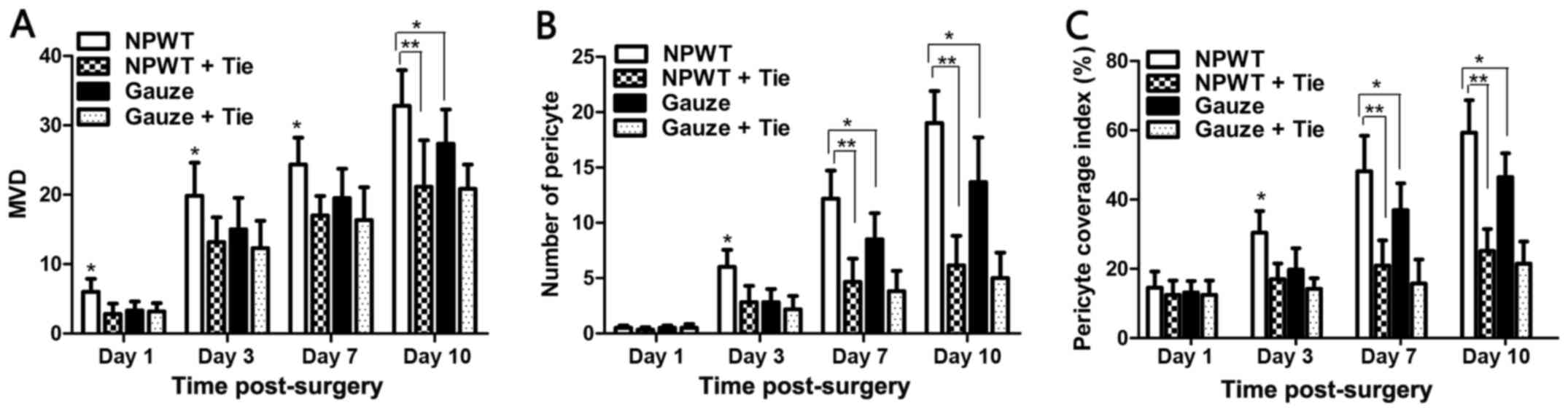
was used to quantify the number of new blood vessels, and the
results revealed that the MVD was higher in the NPWT group than the
other groups from day 1 to day 10 (P<0.05 for gauze group;
P<0.01 either NPWT + Tie group or gauze + Tie group) (Fig. 4A.)
The number of pericytes and MPI
We used a combination of the specific markers, CD31
and α-SMA, to simultaneously immunostain microvascular endothelial
cells and pericytes to assess the functional status of the
neovasculature in the wound (19). The results revealed that a small
number of green marker α-SMA-positive pericytes was detected on the
3rd day in the NPWT group and gauze group. By contrast, in the
other groups, few pericytes were detected on the 3rd day (data not
shown). On the 7th day, a great number of pericytes was detected,
and was distributed discontinuously around the endothelial cell
lumen in the NPWT group. An abundance of pericytes was detected in
the NPWT group on the 10th day, with the pericytes tightly
encircling and covering the microvessel endothelial tubes (Fig. 3). Subsequently, a small number of
pericytes was detected in the gauze group on the 3rd and 7th day,
and a number of pericytes was detected on the 10th day in the gauze
group; however, this number was significantly lower than that in
the NPWT group at the same time points (P<0.05), and in the NPWT
group it was significantly higher than that in the NPWT + Tie group
or gauze + Tie group (P<0.01; Fig.
4B).
Although the MVD values can be applied to evaluate
the presence of new blood vessels, these MVD values do not provide
the information of the functional status of the microvessels or the
maturity of the neovasculature (18). Therefore, the MPI values were
applied to reflect the percentage of microvessels covered with
pericytes and quantified to assess the maturity of the
neovasculature (20). The results
revealed that in the NPWT group, a significantly higher average MPI
was detected compared to the gauze group from the 3rd to the 10th
day (P<0.05), and the MPI was significantly higher than that in
the Tie-2 inhibitor groups (P<0.01; Fig. 4C).
Changes in the expression of Ang-1 and
p-Tie-2 following NPWT treatment
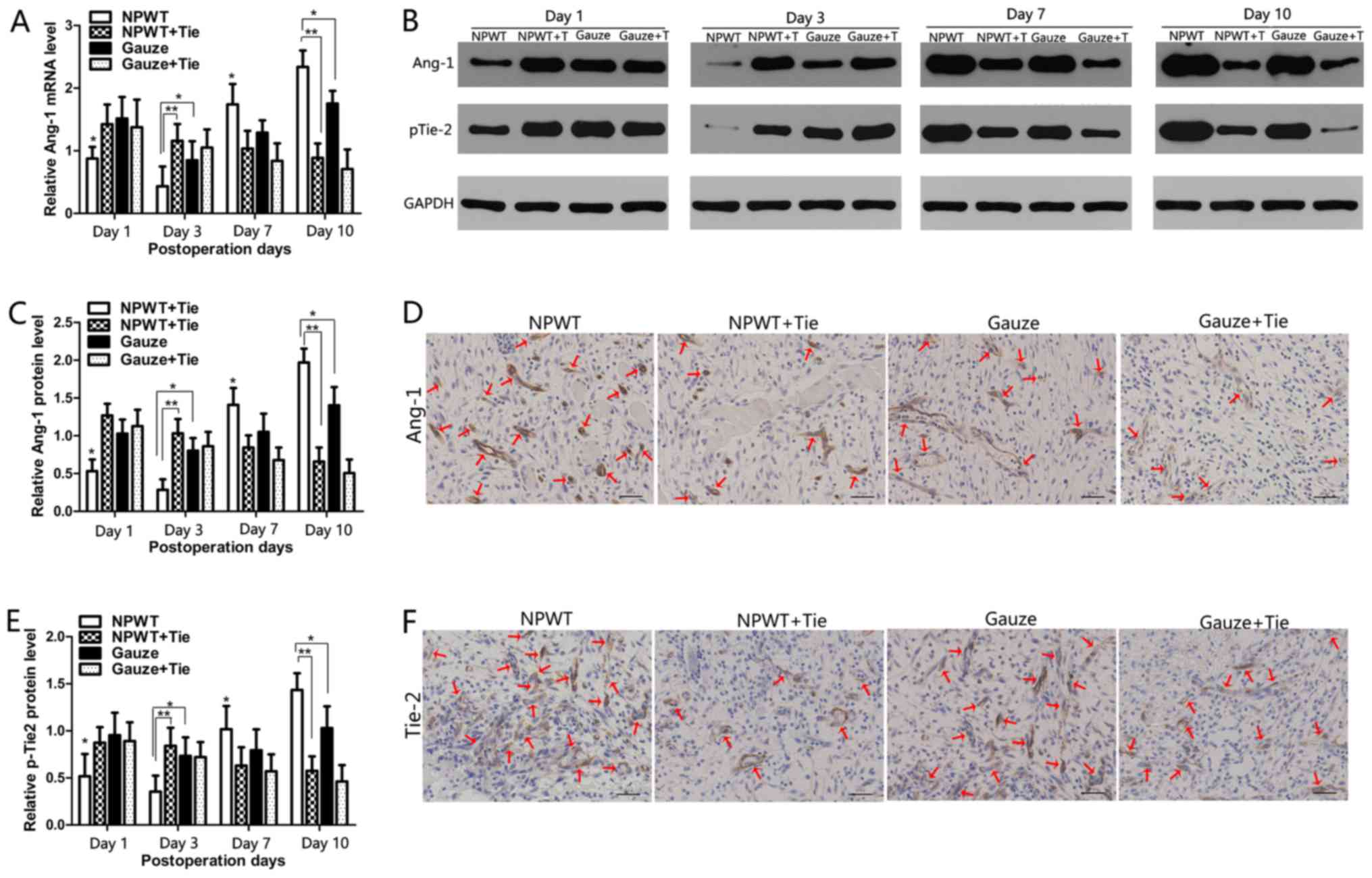
The changes in the mRNA expression levels of Ang-1
were quantitatively analyzed by RT-qPCR, as shown in Fig. 5A. In the NPWT group, the mRNA
expression of Ang-1 was significantly lower than that in the other
groups from the 1st to the 3rd day (P<0.05 vs. gauze group;
P<0.01 vs. either NPWT + Tie group or gauze + Tie group).
However, on the 7th and 10th day, the mRNA expression of Ang-1 was
significantly higher than that in the other groups (P<0.05 vs.
gauze group; P<0.01 vs. either NPWT + Tie group or gauze + Tie
group). In the NPWT + Tie and gauze + Tie groups, the mRNA
expression trend of Ang-1 was opposite to that in the NWPT group,
and the mRNA expression of Ang-1 was significantly lower than that
in the NPWT group on days 7 and 10 (P<0.01). Representative
western blots of Ang-1 protein expression are shown in Fig. 5B, which present the same trend as
Ang-1 mRNA expression. The results of statistical analysis of Ang-1
protein expression are shown in Fig.
5C. Immunohistochemical staining also revealed that Ang-1 was
primarily presented in pericytes, and in the NPWT group,
Ang-1-positive staining was much higher than that in the other
groups on the 7th day (Fig.
5D).
The tyrosine phosphorylation of Tie-2 was detected
by western blot analysis as shown in Fig. 5B. The results revealed that the
expression of p-Tie-2 was gradually decreased from days 1 to 3 in
the NPWT group, and it was significantly lower than that in the
other groups at the same time points (P<0.05 vs. gauze group;
P<0.01 vs. either NPWT + Tie group or gauze + Tie group);
subsequently, it increased rapidly from days 7 to 10 in the NPWT
group, and was significantly higher than that in the other groups
(P<0.05 vs. gauze group; P<0.01 vs. either NPWT + Tie group
or gauze + Tie group) (Fig. 5E).
Although the expression trend of p-Tie-2 in the gauze group was
similar to that in the NPWT group, the expression level of p-Tie-2
was significantly lower than that in the NPWT group on days 7 and
10 (P<0.05). The expression level of p-Tie-2 was gradually
decreased in the NPWT + Tie and gauze + Tie groups, and it was
significantly lower than that in the NPWT group from the 3rd to the
10th day (P<0.01). Immunohistochemical staining revealed that
Tie-2 was mainly expressed in endothelial cells, and in the NPWT
group, Tie-2 positive staining was much higher than that in the
other groups on the 7th day (Fig.
5F).
Changes in the expression levels of α-SMA
and collagen type IV following NPWT treatment
Changes in the mRNA expression levels of α-SMA are
shown in Fig. 6A. The mRNA
expression of α-SMA gradually increased from days 3 to 10 in the
NPWT group, and it was significantly higher than that in the gauze
group (P<0.05). In particular, in the NPWT + Tie and gauze + Tie
groups, the mRNA expression levels of α-SMA were significantly
lower than those in the NPWT group from the 7th to the 10th day
(P<0.01). The protein expression of α-SMA was consistent with
its mRNA expression profile (Fig. 6B
and C). Immunohistochemical staining revealed that α-SMA was
primarily present in pericytes, and in the NPWT group,
α-SMA-positive staining was much higher than that in the other
groups on the 7th day (Fig.
6D).
Changes in the mRNA expression of collagen type IV
are shown in Fig. 6E. The results
revealed that the mRNA expression of collagen type IV was gradually
increased from the 1st to the 10th day in the NPWT group, and was
significantly higher than that in the other groups (P<0.05 vs.
gauze group; P<0.01 vs. either NPWT + Tie group or gauze + Tie
group), particularly on the 7th and 10th day. The changes in the
protein expression levels of collagen type IV were consistent with
its mRNA expression profile. The results are shown in Fig. 6B and F. Immunohistochemical
staining revealed that the positive expression level of collagen
type IV was higher than that in the other groups on the 7th day
(Fig. 6G).
Discussion
It has been demonstrated that NPWT can promote the
overexpression of various cytokines and growth factors (3), as well as improve the
microenvironment of wounds, and thus accelerates wound healing
(21,22). In this study, we found that NPWT
not only increased microvascular density, but also promoted
micro-vascular stabilization and maturation in the wounds, as well
as effect blood flow perfusion. To further elucidate the
mechan-sisms through which NPWT mediates blood flow perfusion, we
examined and analyzed changes in blood flow perfusion from the
aspect of microvessel structure characteristics and vessel
maturation, simultaneously, with the addition of physical methods
for detecting changes in blood flow following NPWT treatment.
Ang-1 is primarily expressed in pericyte-like
perivascular mural cells (pericytes), suggesting a paracrine effect
on new vascular function (16,23), and it serves as a vascular
pro-maturation factor that facilitates vascular maturation.
Tyrosine kinase receptor Tie-2 can affect vascular maturation and
stabilization (24), and it plays
a direct role in affecting pericyte recruitment to endothelial cell
tubes (23). Ang-1 can bind to
Tie-2, and can thus promote microvascular integrity and maturation
(25,26). Blocking of Tie-2 leads to tissue
edema and hemorrhaging, and retards the vessel remodeling and
maturation process (23). Based
on these findings, in this study, we found that the levels of Ang-1
and p-Tie-2 were preferentially and coordinately increased or
decreased in the NPWT group; the results suggested that Ang-1
preferentially binds to Tie-2 and promotes phosphorylation only
following NPWT treatment, and thus coordinately helps to complete
microvascular maturation and integrity process. In addition, the
levels of Ang-1 and MPI were gradually increased in the NPWT group
at the later stages. The results suggested that the microvessels
became gradually more mature at the later stages of wound healing
following NPWT treatment.
Mature microvessels are usually characterized by the
endothelial lumen covered with abundant pericytes, separated by the
integral basement membrane (20).
In particular, the pericyte is an important component of the mature
blood vessel (24). Pericytes are
ubiquitously present and comprised of a heterogeneous cell
population in close contact with endothelial cells. unlike
endothelial cells, pericytes do not form the continuous sheath or
directly to the luminal surface of microvessle network. However,
pericytes serve as single cells distributed at the capillary
network and around blood capillaries, sharing the basement membrane
with endothelial cells (27), and
they are physically embedded in the basement membrane between the
extracellular environment and microvessel tube (28). Previous studies have demonstrated
that pericytes have contraction and dilation characteristics on
capillaries (3,24), and can regulate cerebral blood
flow perfusion (28,29). Studies have also suggested that
capillary constriction and dilation are regulated by the vasoactive
molecules and neurotransmitters in vitro (24,30,31). In vivo studies have
demonstrated that active loss of pericytes will promote capillaries
to dilate (29,32,33). Simultaneously, α-SMA can serve as
a general marker of pericytes (34,35). Therefore a mutual association
exists between pericytes and blood flow perfusion, and the pericyte
tone may reflect microvascular blood blow perfusion heterogeneity
differences (36). In this study,
the levels of Ang-1 and α-SMA, and MPI were significantly higher in
the NPWT group than in the other groups at the later stages. The
results suggested that microvessels were preferentially mature in
the NPWT group. Simultaneously, pericytes are an important
characteristic of mature blood vessels (16,20,23). As shown by our results, pericytes
were markedly evident in the NPWT group. Previous studies have
suggested that NPWT can promote angiogenesis and enhance wound
healing due to the microdeformation and macrodeforation of the
wounds (5,37). In this study, we also speculated
that microvessel maturation and pericyte proliferation induced by
NPWT were closely related to microdeformation and macrodeforation.
unlike the normal microvasculature, tumor blood vessels are
tortuous, leaky and show abnormal pericyte coverage, and thus
effect blood flow perfusion (38,39). We observed that the changes in
blood flow perfusion in the whole process of wound healing were
affected along with the expression levels of α-SMA and MPI in the 4
groups. These results demonstrated that NPWT preferentially
promoted the involvement of pericytes, leading to microvessels
gradual maturation, and thus changing blood flow perfusion due to
the contraction and dilation characteristics of pericytes.
The basement membrane and pericytes serve as two
important components of mature blood vessels to affect the
microvascular maturation process. Recent studies have suggested
that pericytes are recruited to the endothelial cell tube surface,
resulting in the deposition of a continuous basement membrane
matrix. The endothelial tube is not able to deposit basement
membrane in the absence of pericyte (40,41). Collagen type IV serves as a major
basement membrane component that can stabilize vascular structure
(42,43), regulating vessel morphogenesis and
vascular maturation (44). Vessel
basement membrane matrix assembly represents a fundamental step in
the maturation process of vessels (45). In this study, the expression of
collagen type IV gradually increased, and the basement membrane was
gradually integrated along with the increase of the number of
pericytes in the NPWT group, but in NPWT + Tie and gauze + Tie
groups the collagen type IV was markedly reduced and broken, along
with a decreased number of pericytes. These results suggested that
NPWT preferentially induced pericyte proliferation and basement
membrane assembly to form an enveloped endothelium tube, eventually
making the new vessels prone to integrity and maturation.
Therefore, we demonstrated that pericytes and collagen type IV were
complementary to each other for promoting new blood vessel
maturation. The results also suggested that the Ang/Tie-2 signaling
pathway not only regulates pericytes, but also regulates basement
membrane matrix deposition, assembly and formation. On the other
hand, the basement membrane matrix protein deposition and assembly
are regulated by the pericytes, even though the molecular
mechanisms involved are not yet clear. Tahajjodi et al
(43) considered that this
process was probably associated with the integrins. In this study,
the results demonstrated that pericytes and collagen type IV were
complementary to each other for promoting the vascular maturation
process jointly.
In conclusion, we demonstrate that NPWT can
preferentially promote vessel maturation, and can thus affect blood
flow perfusion on the surface of wounds. In particular, pericytes
plays a vital role that can effectively affect blood flow in the
later stage of wound healing following NPWT treatment. In addition,
pericytes and collagen type IV serve as important structural
components that are characteristic of vessel maturation, and they
complement each other, promoting the maturation of new blood
vessels, and thus leading to increased blood flow perfusion on the
surface of wounds.
Acknowledgments
This study was supported by the National Natural
Science Foundation of China (no. 81572163) and by the Hubei
National Natural Science Fund projects (no. 2014CFB751). We would
like to acknowledge the Wuhan VSD Medical Science and Technology,
Co., Ltd. (Wuhan, China) for supplying the vacuum material. We
would also like to thank the Medical Science Experimentation Center
of Wuhan University providing the experiment equipment. This study
has been satisfactorily edited for proper English by an agent of
the English Editing Service of Experimental Biology and
Medicine.
References
|
1
|
Morykwas MJ, Argenta LC, Shelton-Brown EI
and McGuirt W: Vacuum-assisted closure: a new method for wound
control and treatment: animal studies and basic foundation. Ann
Plast Surg. 38:553–562. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Timmers MS, Le Cessie S, Banwell P and
Jukema GN: The effects of varying degrees of pressure delivered by
negative-pressure wound therapy on skin perfusion. Ann Plast Surg.
55:665–671. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Li X, Liu J, Liu Y, Hu X, Dong M, Wang H
and Hu D: Negative pressure wound therapy accelerates rats diabetic
wound by promoting agenesis. Int J Clin Exp Med. 8:3506–3513.
2015.PubMed/NCBI
|
|
4
|
Erba P, Ogawa R, Ackermann M, Adini A,
Miele LF, Dastouri P, Helm D, Mentzer SJ, D'Amato RJ, Murphy GF, et
al: Angiogenesis in wounds treated by microdeformational wound
therapy. Ann Surg. 253:402–409. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Orgill DP and Bayer LR: Negative pressure
wound therapy: Past, present and future. Int Wound J. 10(Suppl 1):
15–19. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Morykwas MJ, Simpson J, Punger K, Argenta
A, Kremers L and Argenta J: Vacuum-assisted closure: State of basic
research and physiologic foundation. Plast Reconstr Surg.
117(Suppl): 121S–126S. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chen SZ, Li J, Li XY and Xu LS: Effects of
vacuum-assisted closure on wound microcirculation: an experimental
study. Asian J Surg. 28:211–217. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wackenfors A, Sjögren J, Gustafsson R,
Algotsson L, Ingemansson R and Malmsjö M: Effects of
vacuum-assisted closure therapy on inguinal wound edge
microvascular blood flow. Wound Repair Regen. 12:600–606. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Saxena V, Hwang CW, Huang S, Eichbaum Q,
Ingber D and Orgill DP: Vacuum-assisted closure: microdeformations
of wounds and cell proliferation. Plast Reconstr Surg.
114:1086–1098. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Xia CY, Yu AX, Qi B, Zhou M, Li ZH and
Wang WY: Analysis of blood flow and local expression of
angiogenesis associated growth factors in infected wounds treated
with negative pressure wound therapy. Mol Med Rep. 9:1749–1754.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kairinos N, McKune A, Solomons M, Hudson
DA and Kahn D: The flaws of laser Doppler in negative-pressure
wound therapy research. Wound Repair Regen. 22:424–429. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lindstedt S, Malmsjö M, Hlebowicz J and
Ingemansson R: Comparative study of the microvascular blood flow in
the intestinal wall, wound contraction and fluid evacuation during
negative pressure wound therapy in laparostomy using the V.A.C.
abdominal dressing and the ABThera open abdomen negative pressure
therapy system. Int Wound J. 12:83–88. 2015. View Article : Google Scholar
|
|
13
|
Ashokkumar N and Pari L: Effect of
N-benzoyl-D-phenylalanine and metformin on carbohydrate metabolic
enzymes in neonatal streptozotocin diabetic rats. Clin Chim Acta.
351:105–113. 2005. View Article : Google Scholar
|
|
14
|
Hasenstein JR, Kasmerchak K, Buehler D,
Hafez GR, Cleary K, Moody JS and Kozak KR: Efficacy of Tie2
receptor antagonism in angiosarcoma. Neoplasia. 14:131–140. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lindgren M, Malmqvist LA, Sjöberg F and Ek
AC: Altered skin blood perfusion in areas with non blanchable
erythema: An explorative study. Int Wound J. 3:215–223. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Weidner N: Tumoural vascularity as a
prognostic factor in cancer patients: The evidence continues to
grow. J Pathol. 184:119–122. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Uemura A, Ogawa M, Hirashima M, Fujiwara
T, Koyama S, Takagi H, Honda Y, Wiegand SJ, Yancopoulos GD and
Nishikawa S: Recombinant angiopoietin-1 restores higher-order
architecture of growing blood vessels in mice in the absence of
mural cells. J Clin Invest. 110:1619–1628. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Eberhard A, Kahlert S, Goede V, Hemmerlein
B, Plate KH and Augustin HG: Heterogeneity of angiogenesis and
blood vessel maturation in human tumors: Implications for
antiangiogenic tumor therapies. Cancer Res. 60:1388–1393.
2000.PubMed/NCBI
|
|
19
|
Goede V, Schmidt T, Kimmina S, Kozian D
and Augustin HG: Analysis of blood vessel maturation processes
during cyclic ovarian angiogenesis. Lab Invest. 78:1385–1394.
1998.PubMed/NCBI
|
|
20
|
Zhao J, Chen L, Shu B, Tang J, Zhang L,
Xie J, Qi S and Xu Y: Granulocyte/macrophage colony-stimulating
factor influences angiogenesis by regulating the coordinated
expression of VEGF and the Ang/Tie system. PLoS One. 9:e926912014.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Glass GE, Murphy GF, Esmaeili A, Lai LM
and Nanchahal J: Systematic review of molecular mechanism of action
of negative-pressure wound therapy. Br J Surg. 101:1627–1636. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Dumville JC, Hinchliffe RJ, Cullum N, Game
F, Stubbs N, Sweeting M and Peinemann F: Negative pressure wound
therapy for treating foot wounds in people with diabetes mellitus.
Cochrane Database Syst Rev. 10:CD0103182013.
|
|
23
|
Gaengel K, Genové G, Armulik A and
Betsholtz C: Endothelial-mural cell signaling in vascular
development and angiogenesis. Arterioscler Thromb Vasc Biol.
29:630–638. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Armulik A, Genové G and Betsholtz C:
Pericytes: Developmental, physiological, and pathological
perspectives, problems, and promises. Dev Cell. 21:193–215. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kim KE, Cho CH, Kim HZ, Baluk P, McDonald
DM and Koh GY: In vivo actions of angiopoietins on quiescent and
remodeling blood and lymphatic vessels in mouse airways and skin.
Arterioscler Thromb Vasc Biol. 27:564–570. 2007. View Article : Google Scholar
|
|
26
|
Holash J, Wiegand SJ and Yancopoulos GD:
New model of tumor angiogenesis: Dynamic balance between vessel
regression and growth mediated by angiopoietins and VEGF. Oncogene.
18:5356–5362. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chantrain CF, Henriet P, Jodele S, Emonard
H, Feron O, Courtoy PJ, DeClerck YA and Marbaix E: Mechanisms of
pericyte recruitment in tumour angiogenesis: A new role for
metalloproteinases. Eur J Cancer. 42:310–318. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kutcher ME and Herman IM: The pericyte:
Cellular regulator of microvascular blood flow. Microvasc Res.
77:235–246. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Hall CN, Reynell C, Gesslein B, Hamilton
NB, Mishra A, Sutherland BA, O'Farrell FM, Buchan AM, Lauritzen M
and Attwell D: Capillary pericytes regulate cerebral blood flow in
health and disease. Nature. 508:55–60. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Attwell D, Buchan AM, Charpak S, Lauritzen
M, Macvicar BA and Newman EA: Glial and neuronal control of brain
blood flow. Nature. 468:232–243. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hamilton NB, Attwell D and Hall CN:
Pericyte-mediated regulation of capillary diameter: A component of
neurovascular coupling in health and disease. Front
Neuroenergetics. 2:22010. View Article : Google Scholar
|
|
32
|
Peppiatt CM, Howarth C, Mobbs P and
Attwell D: Bidirectional control of CNS capillary diameter by
pericytes. Nature. 443:700–704. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Puro DG: Physiology and pathobiology of
the pericyte-containing retinal microvasculature: New developments.
Microcirculation. 14:1–10. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Hellström M, Kalén M, Lindahl P, Abramsson
A and Betsholtz C: Role of PDGF-B and PDGFR-beta in recruitment of
vascular smooth muscle cells and pericytes during embryonic blood
vessel formation in the mouse. Development. 126:3047–3055.
1999.PubMed/NCBI
|
|
35
|
Ohlsson R, Falck P, Hellström M, Lindahl
P, Boström H, Franklin G, Ahrlund-Richter L, Pollard J, Soriano P
and Betsholtz C: PDGFB regulates the development of the
labyrinthine layer of the mouse fetal placenta. Dev Biol.
212:124–136. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Jespersen SN and Østergaard L: The roles
of cerebral blood flow, capillary transit time heterogeneity, and
oxygen tension in brain oxygenation and metabolism. J Cereb Blood
Flow Metab. 32:264–277. 2012. View Article : Google Scholar :
|
|
37
|
Kim PJ, Attinger CE, Steinberg JS and
Evans KK: Negative pressure wound therapy with instillation: Past,
present, and future. Surg Technol Int. 26:51–56. 2015.PubMed/NCBI
|
|
38
|
Benjamin LE, Golijanin D, Itin A, Pode D
and Keshet E: Selective ablation of immature blood vessels in
established human tumors follows vascular endothelial growth factor
withdrawal. J Clin Invest. 103:159–165. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Rivera LB and Brekken RA: SPARC promotes
pericyte recruitment via inhibition of endoglin-dependent TGF-β1
activity. J Cell Biol. 193:1305–1319. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Stratman AN, Malotte KM, Mahan RD, Davis
MJ and Davis GE: Pericyte recruitment during vasculogenic tube
assembly stimulates endothelial basement membrane matrix formation.
Blood. 114:5091–5101. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Stratman AN, Schwindt AE, Malotte KM and
Davis GE: Endothelial-derived PDGF-BB and HB-EGF coordinately
regulate pericyte recruitment during vasculogenic tube assembly and
stabilization. Blood. 116:4720–4730. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Yousif LF, Di Russo J and Sorokin L:
Laminin isoforms in endothelial and perivascular basement
membranes. Cell Adhes Migr. 7:101–110. 2013. View Article : Google Scholar
|
|
43
|
Tahajjodi SS, Amerion M, Mahdavi Shahri N,
Jalali M and Nikravesh MR: The effect of maternal nicotine on
basement membrane collagen IV of brain microvessels changes in
neonatal Balb/C mice. Iran J Reprod Med. 12:275–280.
2014.PubMed/NCBI
|
|
44
|
Senger DR and Davis GE: Angiogenesis. Cold
Spring Harb Perspect Biol. 3:a0050902011. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Miner JH and Yurchenco PD: Laminin
functions in tissue morphogenesis. Annu Rev Cell Dev Biol.
20:255–284. 2004. View Article : Google Scholar : PubMed/NCBI
|