Introduction
Thromboxane A2 receptor (TXA2R), a member of the G
protein-coupled receptor family (1), is broadly distributed in platelets
(2), as well as epithelial
(3), smooth muscle (4), glial and nerve cells in the brain
(5). TXA2R is regarded as a
traditional coagulation and inflammation-associated receptor, which
is also closely associated with neurological disorders. A previous
study of our research group indicated that mutation on TXA2R
influenced the morbidity of cerebral infarctions in humans, which
may be associated with the changes of coagulation functions
(6). Also, it was observed that
SQ29548, an antagonist of TXA2R, was able to attenuate the cerebral
ischemia-reperfusion damage in mice and improve their behavioral
scores (7), thus revealing the
potential therapeutic effect of TXA2R antagonists. However, the
underlying mechanisms involved remain unclear. A number of studies
revealed a new role for TXA2R in regulating the cellular oxidative
stress and the consequent apoptotic process (8–10).
TXA2R antagonists are reported to ameliorate organic oxidative
stress and protect the function of organs (9), which provided a new clue for the
treatment of oxidative stress-associated nervous system
diseases.
Oxidative stress is an important contributor in the
pathogenesis and pathological processes of several neurological
disorders (11,12). In general, the cellular reactive
oxygen species (ROS) production and the antioxidative defense were
observed to be in a dynamic balance. For instance, antioxidative
enzymes (13), including
superoxide dismutase (SOD) and catalase, efficiently eliminated the
ROS produced by cellular respiration. Cerebral infarction (CI) or
other neurological disorders lead to an excess of ROS production,
which distorts the balance between ROS production and antioxidant
defenses. The excessive ROS production results in cellular damage
and apoptosis via attacking the lipid, protein and DNA of cells,
and activating signaling pathways, such as the mitogen-activated
protein kinase (MAPK) pathways (14,15). Numerous studies have proven that
the elevation of cellular antioxidative stress capacity was
beneficial for the limitation of ROS generation, acceleration of
ROS elimination, alleviation of ROS damage and inhibition of MAPK
signaling pathways to reduce cell death (13,16). Compared with other organs
(17), brain tissue is sensitive
to oxidative stress damage due to its characteristics of high
energy consumption and weak antioxidative stress capacity.
Therefore, the identification of a novel effective neuroprotective
agent is essential in the prevention and treatment of oxidative
stress-associated neurological disorders.
In view of the association between TXA2R and
oxidative stress, increasing attention has concentrated on the role
of TXA2R and its antagonists in the treatment of oxidative
stress-associated neurological disorders and the underlying
molecular mechanisms. In the present study, an in vitro
oxidative stress model was established in order to investigate
whether the TXA2R antagonist SQ29548 regulated the cellular
oxidative stress response in nerve cells and determined the
mechanisms involved.
Materials and methods
Reagents
Hydrogen peroxide (H2O2; cat.
no. 323381; used for cell experiments), dimethyl sulfoxide (DMSO;
cat. no. D2650), poly-D-lysine hydrobromide (cat. no. P6407),
nicotinamide adenine dinucleotide (NADH; cat. no. N4505), sodium
pyruvate (cat. no. P4562) and Triton X-100 (cat. no. T8787) were
purchased from Sigma-Aldrich (Merck KGaA, Darmstadt, Germany).
SQ29548 (cat. no. 19025) was obtained from Cayman Chemical Company
(Ann Arbor, MI, USA). Bovine serum albumin (BSA; cat. no. 36102ES)
was from Yeasen Biotechnology Co., Ltd. (Shanghai, China).
Water-soluble tetrazolium salt (WST-1; cat. no. C0035),
2′,7′-dichlorofluorescin diacetate (DCFH-DA; cat. no. S0033) and
DAPI (cat. no. C1002) were purchased from Beyotime Institute of
Biotechnology (Shanghai, China). ApoScreen Annexin V
Apoptosis-phycoerythrin (PE) kit (cat. no. 10010-09) was obtained
from Southern Biotech (Birmingham, AL, USA). Terminal
deoxynucleotidyl transferase dUTP nick end labeling (TUNEL; cat.
no. TUN002) staining reagents were purchased from Mai bio
(Shanghai, China). An antibody against TXA2R was from Santa Cruz
Biotechnology, Inc. (Dallas, TX, USA; cat. no. sc-30036).
Antibodies against c-Jun N-terminal kinases (JNK; cat. no. 9926),
extracellular signal-regulated kinase (ERK; cat. no. 9926), p38
(cat. no. 9926), phosphorylated (p)-JNK (cat. no. 9910), p-ERK
(cat. no. 9910), p-p38 (cat. no. 9910), cleaved caspase3 (cat. no.
9661), caspase3 (cat. no. 9662), cleaved caspase9 (cat. no. 9501),
caspase9 (cat. no. 9502) and β-tubulin (cat. no. 15115), as well as
the anti-rabbit horseradish peroxidase-conjugated secondary
antibody (cat. no. 7074), were purchased from Cell Signaling
Technology, Inc. (Danvers, MA, USA). Antibodies for SOD2 (cat. no.
ab13534) and catalase (cat. no. ab16731) were from Abcam
(Cambridge, MA, USA). An antibody for NeuN (cat. no. MAB377) was
from EMD Millipore (Billerica, MA, USA). The Alexa-594 antibody
(cat. no. A21203; Invitrogen), TRIzol reagent (cat. no. 15596026)
and BCA protein detection kit (cat. no. 23225) were purchased from
Thermo Fisher Scientific, Inc. (Waltham, MA, USA). Paraformaldehyde
(PFA; cat. no. 80096618), potassium phosphate (cat. no. 10017518),
30% hydrogen peroxide (cat. no. 10011208; used for TUNEL staining)
from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China). The
primers used in this study were synthesised by Sangon Biotech Co.,
Ltd. (Shanghai, China).
Cell culture and oxidative stress
model
SH-SY5Y cells were acquired from the Cell Bank of
Shanghai Institute of Biochemistry and Cell Biology, Chinese
Academy of Sciences (Shanghai, China). SH-SY5Y cells were incubated
in the Dulbecco's modified Eagle's medium (DMEM; HyClone; GE
Healthcare, Logan, UT, USA) supplemented with 10% fetal bovine
serum (FBS; HyClone; GE Healthcare) and 1% penicillin-streptomycin
(Invitrogen; Thermo Fisher Scientific, Inc.) at 37°C in a 5%
CO2 incubator. When the cell confluence reached 80%,
cells were collected and transferred into 96-well plates at a cell
density of 2×104 cells/well for WST-1 assay, while
24-well plates (1×105 cells/well) were used for DCFH-DA,
TUNEL and lactate dehydrogenase (LDH) assays, and 6-well plates
(3×105 cells/well) were used for western blot analysis,
reverse transcription-quantitative polymerase chain reaction
(RT-qPCR) and flow cytometry. Subsequently, the cells were
incubated in DMEM without sodium pyruvate with 10% FBS and 1%
penicillin-streptomycin for 24 h. All the plates used were
pretreated with poly-D-lysine hydrobromide solution (0.1 mg/ml) for
24 h and then washed three times with phosphate-buffered saline
(PBS; HyClone; GE Healthcare). A total of 24 h after the cells
transferred to the plates, DMSO was added to the control group and
SQ29548 (dissolved in DMSO) in different concentrations were added
to the treatment groups. After 2-h treatment, the cells were washed
three times with PBS. Next, the cells were treated with or without
H2O2 in different concentrations in the
medium for 1 h, followed by washing three times with PBS. Finally,
DMEM (without sodium pyruvate) with 10% FBS and 1%
penicillin-streptomycin was used for subsequent culture and
experiments.
WST-1 assay
A WST-1 assay was utilized to detect the cell
viability, according to the protocol provided by the manufacturer.
After transferring the cells to the plates for 24 h, the cells were
treated with SQ29548 (0, 0.1, 0.5, 1.0 and 10 μM) for 2 h or
treated with H2O2 (0, 0.05, 0.1, 0.2, 0.5 and
1.0 mM) for 1 h or treated with H2O2 (0, 0.1
mM) for 1 h following pretreatment with SQ29548 (0, 0.1, 0.5, 1.0
and 10 μM) for 2 h. Subsequent to the incubation for 24 h,
10 μl WST-1 solution was added to the SH-SY5Y cells. After 1
h of incubation at 37°C, a microplate reader (Bio-Rad Laboratories,
Inc., Hercules, CA, USA) was used to measure the absorbance of each
well at 450 nm.
LDH assay
An LDH assay was conducted as reported previously
(18). The cells were treated
with H2O2 (0, 0.1 mM) for 1 h after
pretreated with SQ29548 (0, 0.1, 1.0 μM) for 2 h. After 24 h
of incubation, 150 μl culture medium of post-treated SH-SY5Y
cells was collected and added into 96-well plates. Next, 150
μl reaction buffer containing 500 mM potassium phosphate,
7.5 mM sodium pyruvate and 1.5 mM NADH was added successively and
the absorbance of each well at 340 nm was measured over 30 sec with
the microplate reader.
Intracellular ROS level assay
The intracellular ROS level was detected with
DCFH-DA reagents. The cells were treated with
H2O2 (0 or 0.1 mM) for 1 h following
pretreatment with SQ29548 (0, 0.1, 1.0 μM) for 2 h. Treated
SH-SY5Y cells were washed three times with PBS after 1-h
incubation. Next, 200 μl DMEM (without sodium pyruvate,
serum and antibiotics) with 10 μM DCFH-DA was added into
each well. After 20 min of incubation at 37°C, DMEM (without sodium
pyruvate, serum and antibiotics) was used to wash the cells for
three times. The plates were then observed with an inverted
fluorescence microscope (Leica Microsystems GmbH, Wetzlar, Germany)
at an excitation wavelength of 488 nm and an emission wavelength of
525 nm. All the data were subjected to fluorescence intensity
analysis with ImageJ software, version 1.14 (National Institutes of
Health, Bethesda, MA, USA).
Annexin V-PE and 7-aminoactinomycin D
(7-AAD) double-staining assay
Flow cytometry was performed as reported previously
(19). The cells were treated
with H2O2 (0, 0.1 mM) for 1 h following
pretreatment with SQ29548 (0, 0.1, 1.0 μM) for 2 h. Treated
SH-SY5Y cells were digested after 1-h incubation, and 10 μl
of which was then used for cell counting with Fuchs-Rosenthal
counting chambers. Subsequently, the cells were centrifuged at 74 ×
g for 4 min at 4°C. Centrifuged cells were resuspended with cold 1X
Annexin binding buffer to obtain a density of a 1×106
cells/ml. A total of 10 μl Annexin V-PE was added to 100
μl cell suspension and the cell suspension was then
incubated in the dark for 15 m at 4°C. After the incubation, 380
μl cold 1X Annexin binding buffer and 10 μl 7-AAD
were added to the cell suspension at 4°C, and the cell suspension
was immediately subjected to apoptosis assay by flow cytometry
(Bio-Rad Laboratories, Inc.).
TUNEL staining
The cells were treated with
H2O2 (0, 0.1 mM) for 1 h following
pretreatment with SQ29548 (0, 0.1, 1.0 μM) for 2 h. After 1
h of incubation, the treated SH-SY5Y cells were fixed with 4% PFA
for 10 m at 4°C and sealed with methanol containing 3%
H2O2, and 0.3% Triton X-100 was then used for
membrane rupture. Next, 50 μl In Situ Cell Death
reagent was added to each slide, and then cells were incubated
without light exposure for 60 min. Subsequent to 150 μl DAPI
staining (1:300) for 3 min at room temperature, slides were sealed
and observed with a confocal fluorescence microscope (Leica
Microsystems GmbH).
RT-qPCR
The cells were treated with
H2O2 (0, 0.1 mM) for 1 h following
pretreatment with SQ29548 (0, 0.1 μM) for 2 h. After 12 h of
incubation, total RNA was extracted from the treated SH-SY5Y cells
with TRIzol reagent and was reverse transcribed into cDNA with
PrimeScript RT reagent kit (Takara Bio, Inc., Otsu, Japan), as
previously described (20). qPCR
was subsequently implemented on an Applied Biosystems 7500
Real-Time PCR system (Thermo Fisher Scientific, Inc.) using a SYBR
Green kit (Takara Bio, Inc.) under the following conditions:
Denaturation at 95°C for 10 sec, followed by 40 cycles at 95°C for
5 sec and at 60°C for 30 sec. The primes used in qPCR were as
follows: SOD2, 5′-GGTGGTCATATCAATCATAG-3′ (forward) and
5′-AGTGGAATAAGGTTTGTTGT-3′ (reverse); catalase,
5′-TTAATCCATTCGATCTCACC-3′ (forward) and 5′-GGCGGTGAGTGTCAGGATAG-3′
(reverse); GAPDH, 5′-TCTTCTTTTGCGTCGCCAG-3′ (forward) and
5′-GGGGGCAGAGATGATGACC-3′ (reverse). GAPDH served as the loading
control. All the data were analyzed with the comparative threshold
cycle (Cq) method (21).
Western blot analysis
The cells were treated with
H2O2 (0, 0.1 mM) for 1 h following
pretreatment with SQ29548 (0, 0.1 μM) for 2 h. Protein was
extracted with standard lysis buffer (Cell Signaling Technology,
Inc.). Following purification, a BCA protein detection kit was used
for protein concentration measurement. Equal amounts of extracted
protein (30 μg) were separated with 10% SDS-PAGE.
Subsequently the proteins were transferred to nitrocellulose
membranes at 300 mA for 90 min. After 1 h of incubation in 5% BSA
for blocking at room temperature, membranes were incubated at 4°C
overnight with primary antibodies diluted at 1:1,000. Subsequent to
washing three times with TBST, anti-rabbit horseradish
peroxidase-conjugated secondary antibody diluted at 1:5,000 was
added for 1-h incubation at room temperature. Following further
washing with TBST for three times, the specific bands were
visualized with an enhanced chemiluminescence reagent (Thermo
Fisher Scientific, Inc.) in a gel imaging system (Tanon Science
& Technology Co., Ltd., Shanghai, China). ImageJ software was
applied for gray analysis of the data.
Statistical analysis
All the data were analyzed with GraphPad Prism
software, version 5.0 (GraphPad Software, Inc., La Jolla, CA, USA).
Comparison between two groups was performed with an unpaired
t-test, while comparison among several groups was performed with
one-way analysis of variance, followed by a Student-Newman-Keuls
test. All the data are expressed as the mean ± standard error of
the mean. P<0.05 was regarded as an indicator of a statistically
significant difference.
Results
SQ29548 attenuates the decrease in the
viability of SH-SY5Y cells induced by
H2O2
Previous studies have reported (5,22)
the expression of TXA2R on the membrane of nerve cells. In the
present study, the expression of TXA2R on the surface of SH-SY5Y
cells was confirmed by staining with TXA2R antibody and DAPI
(Fig. 1A). Subsequently, to
evaluate the function of SQ29548 and H2O2 on
SH-SY5Y cell viability, a WST-1 assay was applied after cells were
treated with different SQ29548 and H2O2
concentrations. It was observed that, compared with the untreated
control group, treatment with DMSO or with 0.1, 0.5, 1.0 and 10
μM SQ29548 resulted in cell viability of 98.86±4.55,
99.15±2.90, 95.97±2.55, 100.20±1.86 and 95.33±4.03%, respectively
(Fig. 1B). No statistically
significant difference was observed among these groups (P>0.05),
indicating that SQ29548 did not evidently affected the SH-SY5Y cell
viability. However, treatment with 0.05, 0.1, 0.2, 0.5 and 1.0 mM
H2O2 reduced the SH-SY5Y cell viability to
83.01±1.52, 31.91±1.85, 28.16±1.44, 25.32±1.44 and 23.30±1.65%,
respectively, compared with the untreated control group (Fig. 1C). Considering other relevant
findings (17,23), the concentration of 0.1 mM
H2O2 was selected for subsequent
experiments.
An oxidative stress model was then established in
SH-SY5Y cells by H2O2 treatment (0.1 mM).
H2O2-induced cells that were also pretreated
with 0.1, 0.5, 1.0 and 10 μM SQ29548 for 2 h exhibited
improved viability of 44.29±1.40, 43.28±1.07, 42.32±2.03 and
40.27±1.29%, respectively. By contrast, the cell viability of the
H2O2-treated only group was 32.27±1.54%,
compared with that of the control group (Fig. 1D). The results indicated the
protective effect of SQ29548 on cell viability. Furthermore, this
protective effect was independent of the concentration of
SQ29548.
The LDH assay demonstrated that, in the
H2O2-treated only group, the LDH release was
3.30±0.06-fold that of the control group; by contrast, 0.1 and 1.0
μM SQ29548 significantly decreased the LDH release to
2.57±0.09-fold and 2.74±0.14-fold that of the control, respectively
(Fig. 1E). Therefore, the
protective effect of SQ29548 on cell viability was further
confirmed.
SQ29548 reduces the intracellular ROS
level in SH-SY5Y cells induced by H2O2
A DCFH-DA assay was performed to evaluate the effect
of SQ29548 on the intracellular ROS level of SH-SY5Y cells
(Fig. 2A).
H2O2 increased the SH-SY5Y intracellular ROS
level to 4.95±0.97-fold that of the control group (Fig. 2B). However, pretreatment with 0.1
and 1.0 μM SQ29548 decreased the intracellular ROS level to
2.01±0.10-fold and 1.69±0.16-fold, respectively, of the control
group level (Fig. 2B). Thus,
SQ29548 reduced the ROS generation in SH-SY5Y cells induced by
H2O2.
SQ29548 ameliorates the decrease in SOD2
and catalase levels in SH-SY5Y cells induced by
H2O2
To determine whether the change of ROS level was
mediated by the regulatory effect of SQ29548 on intracellular
antioxidant proteins, the intracellular SOD2 and catalase protein
and mRNA levels were evaluated. Upon treatment with
H2O2, the protein levels of SOD2 (Fig. 3A and B) and catalase (Fig. 3A and C) were significantly
inhibited, while their mRNA levels was also decreased to
56.05±7.95% (Fig. 3D) and
45.83±9.70% (Fig. 3E) of the
control group levels, respectively. By contrast, pretreatment with
SQ29548 significantly ameliorated the
H2O2-induced decrease in the protein levels
of SOD2 and catalase (Fig. 3B and
C), while the mRNA levels were rescued to 85.70±5.75% (Fig. 3D) and 80.17±7.02% (Fig. 3E) of the control group levels,
respectively. Therefore, SQ29548 appeared to ameliorate the
H2O2-induced decrease in the expression
levels of SOD2 and catalase in SH-SY5Y cells, and to improve the
antioxidative stress capacities of SH-SY5Y cells.
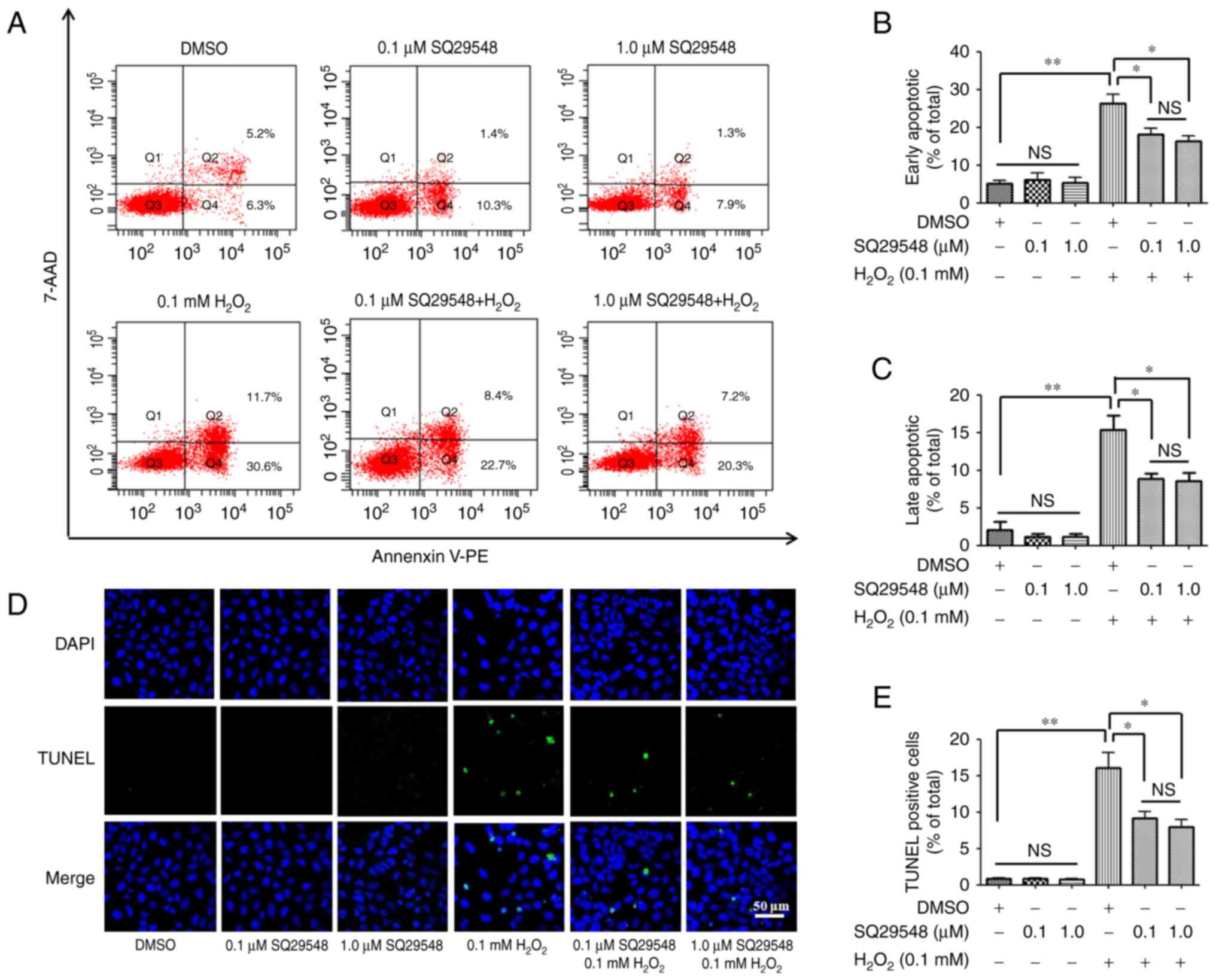
SQ29548 reduces the apoptosis of SH-SY5Y
cells induced by H2O2
Oxidative stress may result in cell damage and
consequently induce cell apoptosis. To investigate the impact of
SQ29548 on SH-SY5Y cell apoptosis, Annexin V-PE/7-AAD
double-staining assay (Fig. 4A–C)
and TUNEL staining (Fig. 4D and
E) were selected to estimate the cell apoptosis. The Annexin
V-PE/7-AAD double-staining assay revealed that
H2O2 increased the ratio of early-stage and
late-stage apoptosis in SH-SY5Y cells to 26.23±2.46 and 15.4±1.89%,
as compared with the ratios of 5.82±1.31 and 2.07±1.07% in the
control group, respectively (Fig. 4B
and C). However, pretreatment with 0.1 and 1.0 μM
SQ29548 significantly abated the early-stage apoptosis cell ratio
to 18.18±1.56 and 16.33±1.50%, respectively (Fig. 4B), while the late-stage apoptosis
cell ratio was at 8.85±0.57 and 8.58±1.05%, respectively (Fig. 4C). No significant difference was
observed between the 0.1 and 1.0 μM SQ29548 groups
(P>0.05).
TUNEL staining was also applied as a detector of
cell apoptosis. Compared with the control group,
H2O2 markedly raised the ratio of
TUNEL-positive cells to 15.21±2.71% from 0.90±0.1%. Pretreatment
with 0.1 and 1.0 μM SQ29548 significantly reduced the ratio
of TUNEL-positive cells to 9.19±0.82 and 8.00±1.00%, respectively
(Fig. 4D and E), indicating that
SQ29548 reduced the cell apoptosis induced by
H2O2.
SQ29548 inhibits cleaved caspase
expression in SH-SY5Y cells induced by
H2O2
Caspases are typical apoptosis-associated proteins,
whose cleaved products promote cell apoptosis. The present study
attempted to determine how SQ29548 affected the expression of
caspases and their cleaved products in SH-SY5Y cells induced by
H2O2. The results demonstrated that no
significant differences existed in the expression levels of
caspase3 and caspase9 among all the groups (P>0.05), while
H2O2 markedly elevated the expression levels
of cleaved caspase9 and cleaved caspase3 (Fig. 5A). However, SQ29548-pretreated
cells exhibited significantly inhibited expression levels of
cleaved caspase9 (Fig. 5B) and
cleaved caspase3 (Fig. 5C). Thus,
SQ29548 possibly inhibited the expression of cleaved caspases.
SQ29548 inhibits ERK and p38 signaling
pathway activation in SH-SY5Y cells induced by
H2O2
MAPK pathways participate in a number of cellular
physiological processes and regulate cell apoptosis induced by
oxidative stress. In order to examine how SQ29548 interfered in
MAPK pathways in SH-SY5Y cells induced by
H2O2, the activation of JNK, ERK and p38
signaling pathways was examined (Fig.
6A). It was observed that H2O2 evidently
activated JNK (Fig. 6B), ERK
(Fig. 6C) and p38 (Fig. 6D) signaling pathways in SH-SY5Y
cells, while SQ29548 pretreatment inhibited the activation of ERK
(Fig. 6C) and p38 (Fig. 6D) signaling pathways. Therefore,
SQ29548 may protect SH-SY5Y cells against oxidative stress via the
inhibition of ERK and p38 signaling pathway activation.
 | Figure 6SQ29548 inhibited the activation of
ERK and p38 signaling pathways in SH-SY5Y cells induced by
H2O2. (A) Western blot analysis results on
the expression levels of p-JNK, JNK, p-ERK, ERK, p-p38 and p38. (B)
Gray value quantification of (B) p-JNK/total JNK, (C) p-ERK/total
ERK and (D) p-p38/total p38. No significant difference was observed
in the expression levels of total JNK, ERK and p38 among the
groups. Results are expressed as the mean ± standard error of the
mean (n=3). *P<0.05, **P<0.01 and
***P<0.001. JNK, c-Jun N-terminal kinases; ERK,
extracellular signal-regulated kinase; p-, phosphorylated;
H2O2, hydrogen peroxide; DMSO, dimethyl
sulfoxide; n.s., no significant difference. |
Discussion
The results of the present study suggested that
SQ29548, one of the TXA2R antagonists, was able to improve the
antioxidative stress capacities of cells, reduce intracellular ROS
levels and attenuate nerve cell apoptosis via the inhibition of ERK
and p38 signaling pathway activation.
TXA2R, which is mainly distributed in platelets, has
been suggested to be an important membrane receptor in the
coagulation and inflammation processes in humans (6). In recent years, it has been reported
that TXA2R also exists in nerve cells (5), and participates in the regulation of
a series of physiological processes in peripheral and central
nervous cells (5,24,25). Our previous study observed that
the mutation of TXA2R affected the coagulation system function and
further affected the onset of CI (6). Upon the application of SQ29548, an
antagonist of TXA2R, ischemia-reperfusion damage of nerve cells and
neurological function defects were attenuated in mice, although the
underlying mechanism remains unclear (7). Furthermore, recent studies revealed
that TXA2R is also involved in other physiological processes. For
instance, activated TXA2R was found to inhibit growth factor
expression levels in vascular endothelial cells, which further
inhibited angiogenesis and vascular development (26). TXA2R was also closely associated
with the cellular oxidative stress response. It has been observed
that TXA2R participated in the regulation of cellular oxidative
stress (8) and induced cell
apoptosis (10), while TXA2R
antagonists helped to reduce oxidative stress (9). These aforementioned studies
indicated that, in certain neurological disorders, TXA2R may
participate in the regulation of oxidative stress and the
subsequent apoptotic process of nervous cells. Therefore, it is
inferred that TXA2R antagonists may protect nervous cells through
the regulation of cellular oxidative stress.
SH-SY5Y cells were selected in the present study as
the in vitro human nerve cell model, while SQ29548 was used
as a TXA2R antagonist and H2O2 was used to
simulate oxidative stress in vitro. SH-SY5Y, a neuron cell
line, is widely used in studies investigating neurological
disorders (27,28) and oxidative stress in nerve cells
(17,28). SQ29548 has been proposed to be a
specific, reliable and safe TXA2R antagonist (29), and is broadly utilized in studies
on TXA2R (7,25,30,31). H2O2, as one
of main ROS products in cellular oxidative stress (32), is typically used to establish an
in vitro oxidative stress cell model (23,33). In the current study, SH-SY5Y cells
were initially cultured in DMEM; however, due to the well-known
role of sodium pyruvate as a H2O2 scavenger
and neuroprotective agent (34),
the medium was altered to sodium pyruvate-negative DMEM following
the cell planking, in order to minimize the confounder effects. The
results revealed that different doses of SQ29548 had no significant
effect on the viability of SH-SY5Y cells. However, different
concentrations of H2O2 significantly
influence the cell viability, with even a low concentration of
H2O2 exhibiting a strong cytotoxic function.
In addition, the present study observed that different doses of
SQ29548 effectively protected against the
H2O2-induced inhibition of SH-SY5Y cell
viability, and this protective effect was independent of the dose
used.
The DCFH-DA experiment conducted in the current
study verified that SQ29548 reduced intracellular ROS levels in
SH-SY5Y cells treated with H2O2, which may be
associated with the regulation of intracellular antioxidant protein
production following TXA2R inhibition. Intracellular antioxidant
proteins, including SOD2 and catalase, serve to eliminate excessive
ROS in cells (13). Generally,
intracellular superoxide anion would be dismutated to
H2O2 by SODs, particularly SOD2, in
mitochondria; subsequently, catalase catalyzes
H2O2 to H2O and O2
molecules. However, excessive oxidative stress response not only
exceeds the elimination capacity of the antioxidant system, but
also impairs the antioxidant system and inhibits the expression of
antioxidant proteins (35,36).
The present study demonstrated decreased protein and mRNA levels of
SOD2 and catalase in SH-SY5Y cells treated with
H2O2. However, SQ29548 treatment rescued
these decreased levels, protected the antioxidant system and
improved the antioxidative stress capacities of SH-SY5Y cells.
SQ29548 also significantly reduced the
H2O2-induced cell apoptosis and inhibited the
expression levels of cleaved caspase9 and cleaved caspase3.
Caspases are apoptosis-associated proteins (37,38), whose cleaved products promote cell
apoptosis (38). In addition, the
MAPK signaling pathways, which possess numerous roles in cell
physiological processes, also participate in the cell apoptosis
process induced by oxidative stress (39,40). For instance, it has been reported
that, as a member of the MAPK family, the activation of the ERK
signaling pathway induced by H2O2 caused
SH-SY5Y cell apoptosis, and the inhibition of this pathway improved
the cell survival (41).
Furthermore, the inhibition of p38 pathway, another MAPK pathway,
was reported to be beneficial against cell apoptosis induced by
H2O2 (42).
The two aforementioned phenomena may be associated with the ERK
(43) and p38 (44) signaling pathways, which
participated in the cell apoptosis process and regulated the
expression of apoptosis-associated proteins, such as caspases.
Similarly, the present study indicated that SQ92548 regulated the
ERK and p38 signaling pathway activation in SH-SY5Y cells treated
with H2O2, suggesting that SQ29548 possibly
inhibited SH-SY5Y cell apoptosis and the expression of
intracellular apoptosis-associated proteins through interfering
with the ERK and p38 signaling pathways.
Oxidative stress is a vital pathological process in
CI and other neurological disorders (45). Excessive cerebral oxidative stress
severely impairs the neurons, thus, it is important to investigate
how to protect nerve cells affected by oxidative stress response.
In fact, TXA2R is a known therapeutic target in various diseases,
and SQ29548 has been reported to protect against
ischemia-reperfusion-induced intestinal impairment (46). There are also certain studies
indicating that SQ92548 was able to treat the microvascular
blood-brain barrier disruption caused by hyperglycemia and TXA2
(47). However, investigation of
the association between TXA2R and oxidative stress in nerve cells
has not been previously conducted. The present study further
verified the potential of TXA2R as a novel therapeutic target in
oxidative stress-associated neurological disorders, such as CI.
Among oxidative stress studies, drugs such as edaravone (48) and esculetin (49) were demonstrated to be able to
protect cells under oxidative stress. Nevertheless, the current
study found that the TXA2R antagonist SQ29548 functioned similarly
to these drugs.
However, certain issues remain to be solved. For
instance, it is unclear whether extending SQ29548 treatment time
amplifies the protective effect, whether the long-term inhibition
of TXA2R affected the growth and development of neurons, and
whether the nerve cell protection function of SQ29548 existed in
other disease models. In future studies, we will investigate other
in vivo and in vitro models, extend the SQ92548
treatment time and discuss the function of TXA2R antagonists in
other neurological disease models. Such investigations are
essential in clarifying the therapeutic mechanism of CI and other
neurological disorders.
In conclusion, the present study demonstrated that
SQ29548, an antagonist of TXA2R, was able to improve the
antioxidative stress capacities of SH-SY5Y cells, and reduce cell
apoptosis via the inhibition of ERK and p38 signaling pathways.
This provided a new basis for the therapy of oxidative
stress-associated neurological disorders.
Acknowledgments
Not applicable.
References
|
1
|
Miggin SM and Kinsella BT: Expression and
tissue distribution of the mRNAs encoding the human thromboxane A2
receptor (TP) alpha and beta isoforms. Biochim Biophys Acta.
1425:543–559. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kassouf N, Ambily A, Watson S, Hassock S,
Authi HS, Srivastava S, Watson SP and Authi KS:
Phosphatidylinositol-3,4,5-trisphosphate stimulates Ca2+
elevation and Akt phosphorylation to constitute a major mechanism
of thromboxane A2 formation in human platelets. Cell
Signal. 27:1488–1498. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bode M and Mackman N: Regulation of tissue
factor gene expression in monocytes and endothelial cells:
Thromboxane A2 as a new player. Vascul Pharmacol. 62:57–62. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Dagher OK, Jaffa MA, Habib A, Ziyadeh FN,
Luttrell LM and Jaffa AA: Abstract 133: Mechanistic insights into
bradykinin and thromboxane receptors heterodimerization in vascular
smooth muscle cells. Arterioscl Thromb Vascul Biol.
36:A1332016.
|
|
5
|
Sumimoto S, Muramatsu R and Yamashita T:
Thromboxane A2 stimulates neurite outgrowth in cerebral cortical
neurons via mitogen activated protein kinase signaling. Brain Res.
1594:46–51. 2015. View Article : Google Scholar
|
|
6
|
Shao J, Fu Y, Yang W, Yan J, Zhao J, Chen
S and Xia W: Thromboxane A2 receptor polymorphism in association
with cerebral infarction and its regulation on platelet function.
Curr Neurovasc Res. 12:15–24. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yan A, Zhang T, Yang X, Shao J, Fu N, Shen
F, Fu Y and Xia W: Thromboxane A2 receptor antagonist SQ29548
reduces ischemic stroke-induced microglia/macrophages activation
and enrichment, and ameliorates brain injury. Sci Rep. 6:358852016.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhang M, Dong Y, Xu J, Xie Z, Wu Y, Song
P, Guzman M, Wu J and Zou MH: Thromboxane receptor activates the
AMP-activated protein kinase in vascular smooth muscle cells via
hydrogen peroxide. Circ Res. 102:328–337. 2008. View Article : Google Scholar
|
|
9
|
Xu S, Jiang B, Maitland KA, Bayat H, Gu J,
Nadler JL, Corda S, Lavielle G, Verbeuren TJ, Zuccollo A and Cohen
RA: The thromboxane receptor antagonist S18886 attenuates renal
oxidant stress and proteinuria in diabetic apolipoprotein
E-deficient mice. Diabetes. 55:110–119. 2006. View Article : Google Scholar
|
|
10
|
Touchberry CD, Elmore CJ, Srinivas S and
Wacker MJ: Thromboxane A2 mediates apoptosis in cardiomyocytes via
IP3. FASEB J. 27:1122–1128. 2013.
|
|
11
|
Rezzani R, Rodella LF and Bonomini F:
Metabolic syndrome, aging and involvement of oxidative stress.
Aging Dis. 6:109–120. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Salim S: Oxidative stress and the central
nervous system. J Pharmacol Exp Ther. 360:201–205. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Birben E, Sahiner UM, Sackesen C, Erzurum
S and Kalayci O: Oxidative stress and antioxidant defense. World
Allergy Organ J. 5:9–19. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Manzanero S, Santro T and Arumugam TV:
Neuronal oxidative stress in acute ischemic stroke: Sources and
contribution to cell injury. Neurochem Int. 62:712–718. 2013.
View Article : Google Scholar
|
|
15
|
Schieber M and Chandel NS: Ros function in
redox signaling and oxidative stress. Curr Biol. 24:R453–R462.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Pisoschi AM and Pop A: The role of
antioxidants in the chemistry of oxidative stress: A review. Eur J
Med Chem. 97:55–74. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Jiang XW, Bai JP, Zhang Q, Hu XL, Tian X,
Zhu J, Liu J, Meng WH and Zhao QC: Caffeoylquinic acid derivatives
protect SH-SY5Y neuroblastoma cells from hydrogen peroxide-induced
injury through modulating oxidative status. Cell Mol Neurobiol.
37:499–509. 2017. View Article : Google Scholar
|
|
18
|
Shao J, Liu T, Xie QR, Zhang T, Yu H, Wang
B, Ying W, Mruk DD, Silvestrini B, Cheng CY and Xia W: Adjudin
attenuates lipopolysaccharide (LPS)- and ischemia-induced
microglial activation. J Neuroimmunol. 254:83–90. 2013. View Article : Google Scholar
|
|
19
|
Xu C, Hu Y, Hou L, Ju J, Li X, Du N, Guan
X, Liu Z, Zhang T, Qin W, et al: β-Blocker carvedilol protects
cardiomyocytes against oxidative stress-induced apoptosis by
up-regulating miR-133 expression. J Mol Cell Cardiol. 75:111–121.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yan A, Cai G, Xia W and Fu Y: Thromboxane
A2 receptor antagonist SQ29548 suppresses the LPS-induced release
of inflammatory cytokines in BV2 microglia cells via suppressing
MAPK and NF-κB signaling pathways. Mol Med Rep. 16:2491–2496. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2−ΔΔCT method. Methods.
25:402–408. 2001. View Article : Google Scholar
|
|
22
|
Wacker MJ, Tevis O, Hanke J, Howard T,
Gilbert W and Orr JA: Characterization of thromboxane A2 receptor
and TRPV1 mRNA expression in cultured sensory neurons. Neurosci
Lett. 515:12–17. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Qian D, Li Z, Zhang Y, Huang Y, Wu Q, Ru
G, Chen M and Wang B: Response of mouse zygotes treated with mild
hydrogen peroxide as a model to reveal novel mechanisms of
oxidative stress-induced injury in early embryos. Oxid Med Cell
Longev. 2016:15214282016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Cudd TA and Wood CE: Thromboxane A2
receptor antagonism prevents hormonal and cardiovascular responses
to mineral acid infusion. Am J Physiol. 267:R1235–R1240.
1994.PubMed/NCBI
|
|
25
|
Rebel AA, Urquhart SA, Puig KL, Ghatak A,
Brose SA, Golovko MY and Combs CK: Brain changes associated with
thromboxane receptor antagonist SQ 29,548 treatment in a mouse
model. J Neurosci Res. 93:1279–1292. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Benndorf RA, Schwedhelm E, Gnann A, Taheri
R, Kom G, Didié M, Steenpass A, Ergun S and Böger RH: Isoprostanes
inhibit vascular endothelial growth factor-induced endothelial cell
migration, tube formation, and cardiac vessel sprouting in vitro,
as well as angiogenesis in vivo via activation of the thromboxane
A(2) receptor: A potential link between oxidative stress and
impaired angiogenesis. Circ Res. 103:1037–1046. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lopes FM, Schröder R, da Frota ML Jr,
Zanotto-Filho A, Müller CB, Pires AS, Meurer RT, Colpo GD, Gelain
DP, Kapczinski F, et al: Comparison between proliferative and
neuron-like SH-SY5Y cells as an in vitro model for Parkinson
disease studies. Brain Res. 1337:85–94. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Cho GW, Koh SH, Kim MH, Yoo AR, Noh MY, Oh
S and Kim SH: The neuroprotective effect of
erythropoietin-transduced human mesenchymal stromal cells in an
animal model of ischemic stroke. Brain Res. 1353:1–13. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Chakraborty R, Bhullar RP, Dakshinamurti
S, Hwa J and Chelikani P: Inverse agonism of SQ 29,548 and
ramatroban on thromboxane A2 receptor. PLoS One. 9:e859372014.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yang W, Yan A, Zhang T, Shao J, Liu T,
Yang X, Xia W and Fu Y: Thromboxane A2 receptor stimulation
enhances microglial interleukin-1β and NO biosynthesis mediated by
the activation of ERK pathway. Front Aging Neurosci. 8:82016.
View Article : Google Scholar
|
|
31
|
Yun DH, Song HY, Lee MJ, Kim MR, Kim MY,
Lee JS and Kim JH: Thromboxane A2modulates migration,
proliferation, and differentiation of adipose tissue-derived
mesenchymal stem cells. Exp Mol Med. 41:17–24. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kumar KH and Khanum F: Hydroalcoholic
extract of cyperus rotundus ameliorates H2O2-induced human neuronal
cell damage via its anti-oxidative and anti-apoptotic machinery.
Cell Mol Neurobiol. 33:5–17. 2013. View Article : Google Scholar
|
|
33
|
Huang YY, Nagata K, Tedford CE, McCarthy T
and Hamblin MR: Low-level laser therapy (LLLT) reduces oxidative
stress in primary cortical neurons in vitro. J Biophotonics.
6:829–838. 2013.PubMed/NCBI
|
|
34
|
Jagtap JC, Chandele A, Chopde BA and
Shastry P: Sodium pyruvate protects against
H2O2 mediated apoptosis in human
neuroblastoma cell line-SK-N-MC. J Chem Neuroanat. 26:109–118.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Chen H, Yoshioka H, Kim GS, Jung JE, Okami
N, Sakata H, Maier CM, Narasimhan P, Goeders CE and Chan PH:
Oxidative stress in ischemic brain damage: Mechanisms of cell death
and potential molecular targets for neuroprotection. Antioxid Redox
Signal. 14:1505–1517. 2011. View Article : Google Scholar :
|
|
36
|
Miguel F, Augusto AC and Gurgueira SA:
Effect of acute vs chronic H2O2-induced
oxidative stress on antioxidant enzyme activities. Free Radic Res.
43:340–347. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
D'Amelio M, Cavallucci V and Cecconi F:
Neuronal caspase-3 signaling: Not only cell death. Cell Death
Differ. 17:1104–1114. 2010. View Article : Google Scholar
|
|
38
|
Brentnall M, Rodriguez-Menocal L, De
Guevara RL, Cepero E and Boise LH: Caspase-9, caspase-3 and
caspase-7 have distinct roles during intrinsic apoptosis. BMC Cell
Biol. 14:322013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Hsieh CJ, Kuo PL, Hsu YC, Huang YF, Tsai
EM and Hsu YL: Arctigenin, a dietary phytoestrogen, induces
apoptosis of estrogen receptor-negative breast cancer cells through
the ROS/p38 MAPK pathway and epigenetic regulation. Free Radic Biol
Med. 67:159–170. 2014. View Article : Google Scholar
|
|
40
|
Sui X, Kong N, Ye L, Han W, Zhou J, Zhang
Q, He C and Pan H: p38 and JNK MAPK pathways control the balance of
apoptosis and autophagy in response to chemotherapeutic agents.
Cancer Lett. 344:174–179. 2014. View Article : Google Scholar
|
|
41
|
Ruffels J, Griffin M and Dickenson JM:
Activation of ERK1/2, JNK and PKB by hydrogen peroxide in human
SH-SY5Y neuroblastoma cells: Role of ERK1/2 in H2O2-induced cell
death. Eur J Pharmacol. 483:163–173. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Filomeni G, Piccirillo S, Rotilio G and
Ciriolo MR: p38MAPK and ERK1/2 dictate cell
death/survival response to different pro-oxidant stimuli via p53
and Nrf2 in neuroblastoma cells SH-SY5Y. Biochem Pharmacol.
83:1349–1357. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Zhuang S, Yan Y, Daubert RA, Han J and
Schnellmann RG: ERK promotes hydrogen peroxide-induced apoptosis
through caspase-3 activation and inhibition of Akt in renal
epithelial cells. Am J Physiol Renal Physiol. 292:F440–F447. 2007.
View Article : Google Scholar
|
|
44
|
Zhuang S, Demirs JT and Kochevar IE: p38
mitogen-activated protein kinase mediates bid cleavage,
mitochondrial dysfunction, and caspase-3 activation during
apoptosis induced by singlet oxygen but not by hydrogen peroxide. J
Biol Chem. 275:25939–25948. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Liu J, Yang L, Tian H and Ma Q: Cathepsin
D is involved in the oxygen and glucose
deprivation/reperfusion-induced apoptosis of astrocytes. Int J Mol
Med. 38:1257–1263. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Wen SH, Ling YH, Liu WF, Qiu YX, Li YS, Wu
Y, Shen JT, Xia ZY and Liu KX: Role of 15-F2t-isoprostane in
intestinal injury induced by intestinal ischemia/reperfusion in
rats. Free Radic Res. 48:907–918. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Zhao Z, Hu J, Gao X, Liang H, Yu H, Liu S
and Liu Z: Hyperglycemia via activation of thromboxane A2 receptor
impairs the integrity and function of blood-brain barrier in
microvascular endothelial cells. Oncotarget. 8:30030–30038.
2017.PubMed/NCBI
|
|
48
|
Zhao ZY, Luan P, Huang SX, Xiao SH, Zhao
J, Zhang B, Gu BB, Pi RB and Liu J: Edaravone protects HT22 neurons
from H2O2-induced apoptosis by inhibiting the
MAPK signaling pathway. CNS Neurosci Ther. 19:163–169. 2013.
View Article : Google Scholar
|
|
49
|
Han MH, Park C, Lee DS, Hong SH, Choi IW,
Kim GY, Choi SH, Shim JH, Chae JI, Yoo YH and Choi YH:
Cytoprotective effects of esculetin against oxidative stress are
associated with the upregulation of Nrf2-mediated NQO1 expression
via the activation of the ERK pathway. Int J Mol Med. 39:380–386.
2017. View Article : Google Scholar
|