Introduction
Atherosclerosis is a chronic inflammatory disease of
the medium-and large-sized arteries, which is associated with the
interactions between endothelial cells, vascular smooth muscle
cells, macrophages, platelets and cytokines (1). Endothelial dysfunction and
inflammation are early markers of atherosclerosis (2). Normally, endothelial cells prevent
interaction between vascular muscle cells and circulating monocytes
and lymphocytes (3). Oxidative
stress and inflammation may injure endothelial cells and promote
the development of atherosclerosis (4). The damaged endothelial cells
synthesize and release pro-inflammatory factors, including
intercellular adhesion molecule-1 (ICAM-1) and vascular cell
adhesion molecule-1 (VCAM-1), which stimulate the inflammatory
cells to attach at the dysfunctional endothelial cells (5,6).
Therefore, protecting endothelial function is an important
therapeutic strategy for the prevention of atherosclerosis.
MicroRNAs (miRNA/miR) are small, noncoding RNAs,
that negatively regulate target gene expression at the
post-transcriptional level by directly binding at 3′-untranslated
regions (UTRs) (7). It was
demonstrated previously that miRNAs may participate in the
development of atherosclerosis (8,9).
For example, the miR-17-92 cluster was significantly downregulated
among patients with atherosclerosis (10). miR-181b and miR-18a were
demonstrated to inhibit endothelial inflammatory responses by
targeting nuclear factor kappa-light-chain-enhancer of activated B
cells signaling in acute and chronic vascular disease states
(8,11,12). miR-21 suppressed the apoptosis and
death of vascular smooth muscle cell induced by hydrogen peroxide
(H2O2) via regulating programmed cell death 4
(13). miR-429 promoted
endothelial cell apoptosis in high-fat diet mice through
suppressing B-cell lymphoma 2 (Bcl-2) expression (14).
miR-291b-3p is a member of the miR-290 cluster. It
was demonstrated that miR-291b-3p promoted hepatocyte apoptosis via
the downregulation of RNA-binding protein Hu antigen R (HuR)
(15). Additionally, miR-291b-3p
may also regulate the metabolism of lipids and glucose in the liver
via targeting adenosine monophosphate-activated kinase α1 and
transcription factor p65 (16,17). However, it remains unclear whether
the miR-291b-3p is associated with endothelial dysfunction.
Therefore, the present study explored the role of miR-291b-3p in
endothelial dysfunction. The present study demonstrated that the
treatment of H2O2 induced the apoptosis and
increased the mRNA levels of miR-291b-3p, ICAM-1 and VCAM-1 in EOMA
cells. And overexpression of miR-291b-3p promoted EOMA cell
apoptosis and dysfunction. In addition, HuR was identified as a
target gene of miR-291b-3p in EOMA cells. The overexpression of HuR
reversed the endothelial dysfunction induced by an miR-291b-3p
mimic. It was hypothesized that miR-291b-3p may be involved in the
endothelial cell dysfunction induced by H2O2
via targeting HuR.
Materials and methods
Cell culture
The EOMA mouse endothelial cell line (American Type
Culture Collection, Manassas, VA, USA) was cultured in high-glucose
Dulbecco's modified Eagle's medium (H-DMEM; Invitrogen; Thermo
Fisher Scientific, Inc., Waltham, MA, USA) supplemented with 10%
fetal calf serum (HyClone; GE Healthcare Life Sciences, Logan, UT,
USA), 100 units/ml penicillin (Invitrogen; Thermo Fisher
Scientific, Inc.) and 0.1 mg/ml streptomycin (HyClone; GE
Healthcare Life Sciences) at 37°C with humidified air and 5%
CO2. EOMA cells were treated with 100 µM
H2O2 (Sigma-Aldrich; Merck KGaA, Darmstadt,
Germany) at 37°C for 24 h.
Transfection of miR-291b-3p mimics and
inhibitors in EOMA cells
The sequences of negative control (NC), microRNA
inhibitor negative control (NCI), miR-291b-3p mimic (291m) and
inhibitor (291i) were as follows (5′-3′): NC sense, UUC UCC GAA CGU
GUC ACG UTT; NC antisense, ACG UGA CAC GUU CGG AGA ATT; NCI, CAG
UAC UUU UGU GUA GUA CAA; 291m sense, AAA GUG CAU CCA UUU UGU UUG U;
291m antisense, AAA CAA AAU GGA UGC ACU UUU U; and 291i, ACA AAC
AAA AUG GAU GCA CUU U. All the siRNA oligos were purchased from
Shanghai GenePharma Co., Ltd, Shanghai, China). According to the
manufacturer's protocol of HiperFect transfection reagent (Qiagen,
GmbH, Hilden, Germany), EOMA cells were seeded in 6-well plates
with 1×105 cell/well with 2 ml H-DMEM medium containing
serum and antibiotics prior to transfection. 5 µl 20 µM NC, NCI,
291m or 291i sequences were pre-incubated with HiperFect
transfection reagent at room temperature for 10 min. Then, the
solutions were added into the EOMA cells at a final concentration
of 50 nM. The cells were then cultured at 37°C for an additional 48
h.
Adenovirus construction
Recombinant adenovirus vectors expressing mouse HuR
(AD-HUR) and control adenovirus vectors containing green
fluorescent protein (GFP) (AD-CON) were purchased from Shanghai
GeneChem Co., Ltd. (Shanghai, China). A total of 15 µl AD-HuR
(1×1010 pfu/ml) was transfected into EOMA cells at a
multiplicity of infection of 100 for 48 h at 37°C. A total of 15 µl
AD-CON (1×1010 pfu/ml) was added to the control groups
at a multiplicity of infection of 100 to maintain a consistent
viral load.
RNA isolation and reverse transcription
quantitative polymerase chain reaction (RT-qPCR)
Total RNA was harvested from EOMA cells using
TRIzol® reagent (Life Technologies; Thermo Fisher
Scientific, Inc.) according to the manufacturer's protocols. A
stem-loop reverse transcription primer and avian myeloblastosis
virus transcriptase (Takara Biotechnology Co., Ltd., Dalian, China)
were used to quantify mature miRNAs. A total of 1 µg RNA was
reversed transcribed into first-strand cDNA using random primers
(Takara Biotechnology Co., Ltd.). qPCR was performed to determine
the mRNA levels of miR-291b-3p, VCAM-1, ICAM-1 and HuR. The
relative gene expression was normalized to U6 or 18s. Each reaction
was performed in triplicate, and analysis was performed by the
2−ΔΔCq method (18).
The relative level of miR-291b-3p was normalized by U6 small
nucleolar RNA, which was used as the housekeeping gene. The
sequences of RT primers were as follows (5′-3′): miR-291b-3p, GTC
GTA TCC AGT GCA GGG TCC GAG GTA TTC GCA CTG GAT ACG ACA CAA AC; U6,
GTC GTA TCC AGT GCA GGG TCC GAG GTA TTC GCA CTG GAT ACG ACA AAT
ATG. The primers used for the qPCR were as follows (5′-3′):
miR-291b-3p forward, GCA AAG TGC ATC CAT TTT GTT TGT; U6 forward,
GCG CGT CGT GAA GCG TTC; Universal reverse primer, GTG CAG GGT CCG
AGG T; VCAM-1 forward, CTC TTA CCT GTG CGC TGT GA; VCAM-1 reverse,
GAC AGG TCT CCC ATG CAC AA; ICAM-1 forward, TTT TCA GCT CCG GTC CTG
AC; ICAM-1 reverse, CCG CTC AGA AGA ACC ACC TT; 18s forward, GGA
AGG GCA CCA CCA GGA GT; and 18s reverse, TGC AGC CCC GGA CAT CTA
AG.
Western blot analysis
Western blot analysis was performed as previously
described (17). The cells were
lysed with radioimmunoprecipitation assay lysis buffer (Beijing
Solarbio Science and Technology Co., Ltd., Beijing, China)
containing a protease inhibitor cocktail (Sigma-Aldrich; Merck
KGaA) and phosphatase inhibitor cocktail (Sigma-Aldrich; Merck
KGaA). Following centrifugation at 10,000 × g for 15 min at 4°C,
the supernatants were collected. The proteins were quantified using
a bicinchoninic acid kit (Thermo Fisher Scientific, Inc.). Cell
lysates containing 15 µg protein were separated by 12% SDS-PAGE and
transferred to polyvinylidene fluoride membranes (EMD Millipore,
Billerica, MA, USA). The membranes were blocked with 5% non-fat dry
milk in TBS with Tween-20 (pH 8.0) (Beijing Solarbio Science and
Technology, Co., Ltd.) and probed with the primary antibodies
(1:1,000) at 4°C overnight. Then, the blots were incubated with
horseradish peroxidase-conjugated anti-rabbit IgG secondary
antibody (cat. no. ABCA2103366; Ab-mart, Inc., Berkeley Heights,
NJ, USA) for 2 h at room temperature, followed by detection with a
Immobilon Western chemiluminescence kit (EMD Millipore). The
antibodies against HuR (cat. no. 12582), Bcl-2 (cat. no. 3498,
Bcl-2-associated X protein (Bax; cat. no. 5023), GAPDH (cat. no.
5174), phosphorylated extracellular signal-regulated kinase
(p-ERK)1/2 (p42/44; cat. no. 4370), ERK1/2 (cat. no. 4695) and
GAPDH were purchased from Cell Signaling Technology, Inc.,
(Danvers, MA, USA). Image J version 1.42 (National Institutes of
Health, Bethesda, MD, USA) was used to calculated the band
intensity.
Luciferase assay
To determine the target gene of miR-291b-3p, the
3′-UTR and coding region of HuR containing miR-291b-3p binding
sites were amplified from NCTC 1469 cells (America Type Culture
Collection, Manassas, VA, USA) by the following primers
(restriction sites are underlined): HuR-coding
region-F-XhoI: 5′-CCC TCG AGC TGG CTC TGG AAT CAT TGC T-3′;
HuR-coding region-R-XhoI: 5′-CCC TCG AGA GGC AGT CTT CGG TTC
TTG A-3′; HuR-UTR -F-XhoI: 5′-CCC TCG AGC CTA TAT GGG GTT
GCT TCC A-3′; HuR-UTR-R-XhoI: 5′-CCC TCG AGC CAA CCA GCC TTC
TTT TCT G-3′.
PCR was performed with genomic DNA isolated from
EOMA cells. The thermocycler conditions for PCR were as follows:
95°C for 10 min, followed by 40 cycles of 95°C for 15 sec, 55°C for
45 sec and 72°C for 30 sec. A total of 2 µg PCR products were
digested with 1 µl XhoI (New England Biolabs. Inc., Ipswich,
MA, USA) and inserted into XhoI-linearlized luciferase
reporter vector pmirGLO (Promega Corporation, Madison, WI, USA). To
perform the luciferase reporter assay, EOMA cells were cultured in
96-well plates at 5,000 cells/well in 100 µl H-DMEM culture medium.
The luciferase reporter plasmid was pmirGLO from Promega
Corporation. Subsequently, the recombinant luciferase vector (0.1
µg) and miR-291b-3p mimic (5 ng) were transfected into EOMA cells
with Effectene reagent (Qiagen GmbH) for 48 h. A dual-luciferase
reporter assay system (Promega Corporation) was subsequently used
to detect the luciferase activity of cells. Luciferase activity was
normalized to Renilla luciferase activity. A total of 6
samples were measured for each group. The experiment was repeated
three times.
Terminal
deoxynucleotidyl-transferase-mediated dUTP nick end labelling
(TUNEL) staining
TUNEL staining was used to detect DNA fragmentation
of individual cells using a TUNEL fluorescence fluorescent
isothiocyanate kit (Roche Diagnostics GmbH, Mannheim, Germany).
EOMA cells were fixed with 4% paraformaldehyde (Beijing Solarbio
Science and Technology, Co., Ltd.) for 20 min at 37°C followed by
permeabilization with 0.1% Triton X-100 (Sigma-Aldrich; Merck
KGaA). Then, cells were incubated with TUNEL reaction mixture at
37°C for 1 h. The nuclei were counterstained by DAPI (1 µg/ml) at
room temperature for 10 min. And the slide was mounted by using
ProLong Diamond Antifade Mountant (Invitrogen; Thermo Fisher
Scientific, Inc.). Cells in 10 randomly chosen fields from each
cultured cell slide were counted to determine the percentage of
apoptotic nuclei. The experiment was repeated for 4 times. The
stained cells were examined using a fluorescence microscope
(magnification, x200; Olympus Corporation, Tokyo, Japan).
Statistical analysis
Data were expressed as the mean ± standard error of
the mean. The two-tailed unpaired Student's t-test was used for
comparisons of two groups. And one-way analysis of variance tests
followed by Turkey post hoc test were performed for comparison of
two more groups by using SPSS 3.0 (SPSS, Inc., Chicago, USA).
P<0.05 were considered to indicate a statistically significant
difference.
Results
H2O2 promotes
miR-291b-3p expression and apoptosis in EOMA endothelial cells
It has been confirmed that
H2O2 induces endothelial cell apoptosis
(19). To investigate the effects
of miR-291b-3p on endothelial cell apoptosis, the level of
miR-291b-3p was determined in the EOMA cells treated with 100 µM
H2O2 for 24 h. TUNEL staining confirmed that
H2O2 treatment led to induced apoptosis in
EOMA cells (Fig. 1A). Compared
with the control group, the mRNA levels of miR-291b-3p, ICAM-1 and
VCAM-1 were increased in EOMA cells treated with
H2O2 (Fig. 1B
and C). Additionally, H2O2 treatment
induced the phosphorylation of ERK and upregulated Bax expression,
accompanied by decreased Bcl-2 protein expression (Fig. 1D). These results suggested that
miR-291b-3p may be involved in the process of endothelial cell
injury.
 | Figure 1H2O2 promotes
miR-291b-3p expression and apoptosis in EOMA endothelial cells. (A)
The levels of apoptosis in EOMA cells treated with
H2O2 was measured by TUNEL staining. (B) The
mRNA levels of miR-291b-3p and (C) ICAM-1 and VCAM-1 were measured
by quantitative polymerase chain reaction. (D) The phosphorylation
of ERK and Bax and Bcl-2 expression were analyzed by western blot
analysis. Data are presented as the mean ± standard error of the
mean (n=5). **P<0.01 and ***P<0.001 vs.
control. CON, control; TUNEL, terminal
deoxynucleotidyl-transferase-mediated dUTP nick end labelling; miR,
microRNA; NCI, microRNA inhibitor negative control; 291m,
miR-291b-3p mimic; 291i, miR-291b-3p inhibitor; ICAM-1,
intercellular adhesion molecule-1; VCAM-1, vascular cell adhesion
molecule-1; ERK, extracellular signal-regulated kinase; p-ERK,
phosphorylated ERK; Bcl-2, B-cell lymphoma 2; Bax, Bcl-2-associated
X protein; H2O2, hydrogen peroxide. |
miR-291b-3p modulates endothelial cell
dysfunction
Next, the effects of miR-291b-3p on EOMA cell
dysfunction were observed. 291m and 291i were transfected into EOMA
cells for 48 h. The results of the qPCR assay indicated that the
level of miR-291b-3p was increased significantly in EOMA cells
transfected with 291m compared with those transfected with miRNA
mimic controls (Fig. 2A).
Overexpression of miR-291b-3p induced apoptosis and upregulated
ICAM-1 and VCAM-1 mRNA expression levels in EOMA cells (Fig. 2B and C). In EOMA cells transfected
with miR-291b-3p mimics, the levels of p-ERK and Bax proteins were
increased, whilst Bcl-2 protein expression was decreased (Fig. 2D). In contrast, the level of
miR-291b-3p was decreased to 40-50% in EOMA cells transfected with
the miR-291b-3p inhibitor compared with those transfected with the
miRNA inhibitor control (Fig.
2E). Transfection with miR-291b-3p inhibitors decreased the
apoptosis rate and downregulated mRNA expression levels of ICAM-1
and VCAM-1 in EOMA cells (Fig. 2F
and 2G). In addition,
transfection with miR-291b-3p inhibitors in EOMA cells led to
decreased p-ERK and BAX protein levels, accompanied by increased
Bcl-2 protein expression (Fig.
2H). The results demonstrated that miR-291b-3p may modulate
apoptosis and the expression of ICAM-1 and VCAM-1 in EOMA
cells.
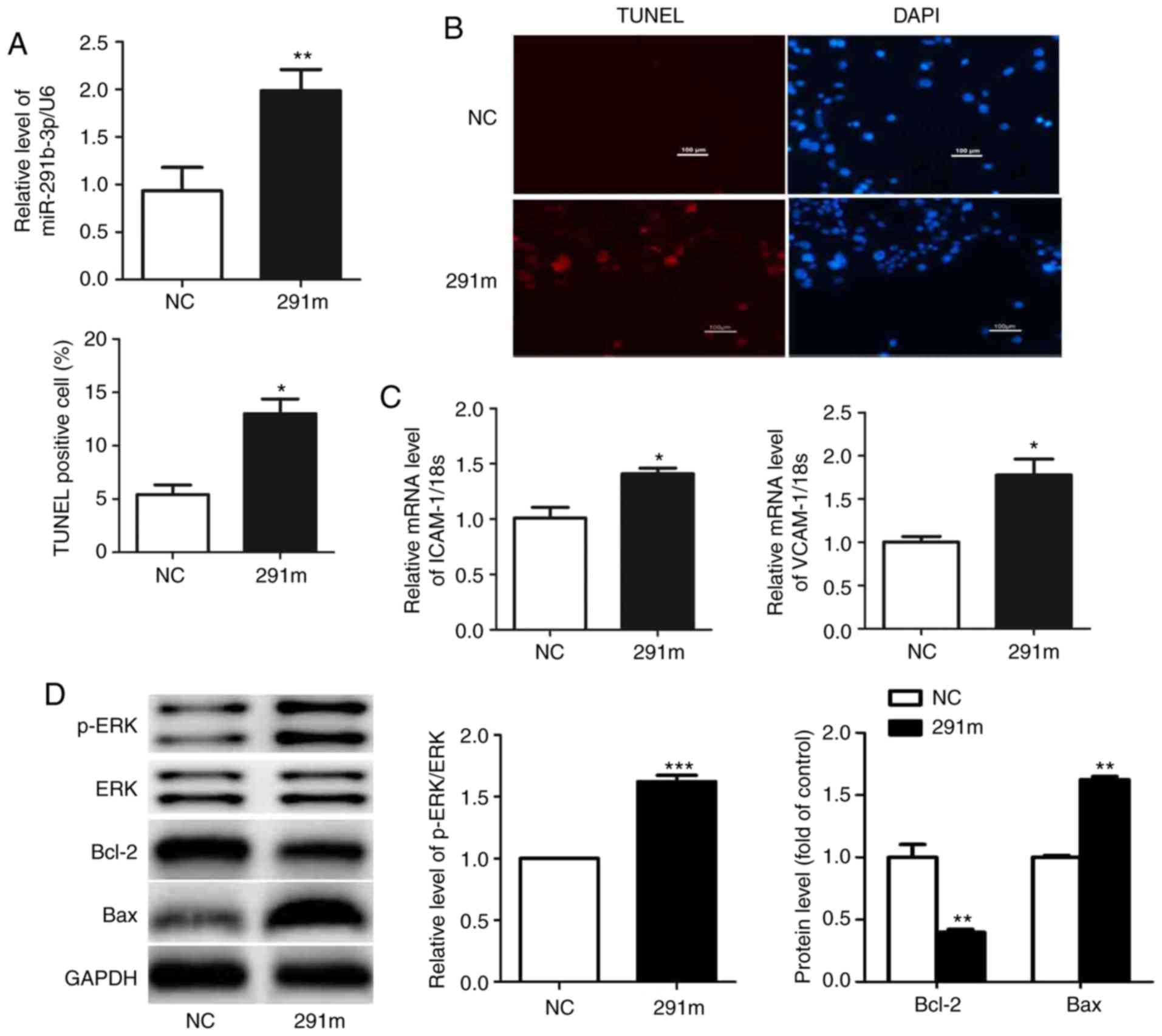 | Figure 2miR-291b-3p modulates EOMA cell
dysfunction. (A) In EOMA cells, transfection with miR-291b-3p mimic
increased miR-291b-3p levels and (B) cell apoptosis rate,
accompanied by elevated (C) mRNA expression of ICAM-1 and VCAM-1.
(D) The levels of p-ERK/ERK and Bax were raised and the protein
level of Bcl2 was reduced in EOMA cells transfected with the
miR-291b-3p mimic. (E) In EOMA cells, transfection with miR-291b-3p
inhibitors decreased miR-291b-3p levels and (F) cell apoptosis
rate, accompanied by reduced mRNA expression of (G) ICAM-1 and
VCAM-1. (H) The levels of p-ERK/ERK and Bax were decreased and the
protein level of Bcl2 was increased in EOMA cells transfected with
miR-291b-3p mimic. Data are presented as the mean ± standard error
of the mean (n=5). *P<0.05, **P<0.01
and ***P<0.001 vs. the control. miR, microRNA; NC,
negative control; ICAM-1, intercellular adhesion molecule-1;
VCAM-1, vascular cell adhesion molecule-1; ERK, extracellular
signal-regulated kinase; p-ERK, phosphorylated ERK; Bcl-2, B-cell
lymphoma 2; Bax, Bcl-2-associated X protein; NCI, miRNA inhibitor
negative control; 291m, miR-291b-3p mimic; 291i, miR-291b-3p
inhibitor. |
Downregulation of miR-291b-3p rescues
H2O2-induced dysfunction in EOMA cells
To additionally assess the role of miR-291b-3p in
H2O2-induced EOMA cell dysfunction,
miR-291b-3p inhibitors were transfected into EOMA cells for 48 h
followed by treatment with H2O2 for 24 h.
Downregulation of miR-291b-3p inhibited
H2O2-induced apoptosis in EOMA cells
(Fig. 3A). Transfection with
miR-291b-3p inhibitors rescued the effect of
H2O2 on mRNA expression of ICAM-1 and VCAM-1
(Fig. 3B). In addition,
H2O2-induced the activation of ERK and
upregulation of Bax expression was also inhibited by transfection
with miR-291b-3p inhibitors (Fig.
3C). These observations indicated that miR-291b-3p may be
involved in the endothelial dysfunction induced by
H2O2.
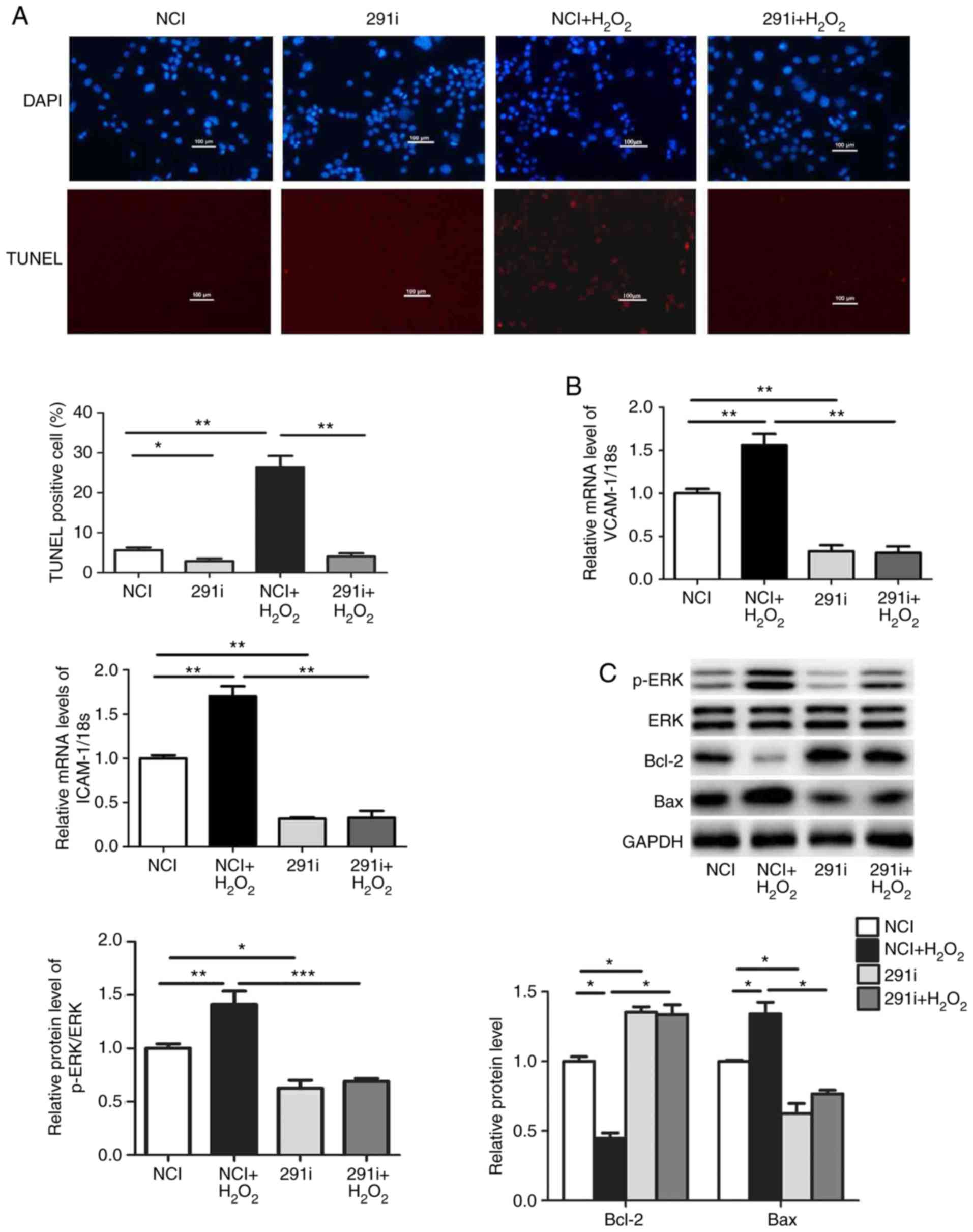 | Figure 3Downregulation of miR-291b-3p rescues
H2O2-induced dysfunction in EOMA cells. (A)
The level of apoptosis in EOMA cells transfected with miR-291b-3p
inhibitor followed by treatment with H2O2 was
measured TUNEL staining. (B) The mRNA levels of ICAM-1 and VCAM-1
were measured by quantitative polymerase chain reaction. (C) The
levels of p-ERK, Bax and Bcl-2 were analyzed by western blot
analysis. Data are presented as the mean ± standard error of the
mean (n=5). *P<0.05, **P<0.01 and
***P<0.001 vs. control. TUNEL, terminal
deoxynucleotidyl-transferase-mediated dUTP nick end labelling;
miRNA, microRNA; NCI, miRNA inhibitor negative control; 291m,
miR-291b-3p mimics; 291i, miR-291b-3p inhibitor; ICAM-1,
intercellular adhesion molecule-1; VCAM-1, vascular cell adhesion
molecule-1; ERK, extracellular signal-regulated kinase; p-ERK,
phosphorylated ERK; Bcl-2, B-cell lymphoma 2; Bax, Bcl-2-associated
X protein; H2O2, hydrogen peroxide. |
HuR, as a target gene of miR-291b-3p,
modulates endothelial apoptosis and dysfunction
It was demonstrated previously that HuR was a target
gene of miR-291b-3p in mouse hepatocytes (15). To confirm the effect of HuR on
H2O2-induced endothelial dysfunction, the HuR
protein and mRNA levels in EOMA cells treated with
H2O2 were analyzed. The results indicated
that H2O2 treatment only decreased HuR
protein levels but did not affect HuR mRNA levels (Fig. 4A). A previous study had suggested
that miR-291b-3p contained 2 binding sites in the coding region
from 902-923 bp and in 3′-UTR from 4,289-4,312 bp (15). The results of the luciferase assay
indicated that the overexpression of miR-291b-3p significantly
decreased the luciferase activity in EOMA cells transfected with
the luciferase reporter vector containing the HuR coding region.
However, the luciferase activity was only slightly decreased when
the EOMA cells were co-transfected with the luciferase reporter
vector containing HuR-3′-UTR and miR-291b-3p mimic (Fig. 4B). Therefore, miR-291b-3p may
directly bind at the coding region of HuR from 902-923 bp. The
overexpression of miR-291b-3p decreased HuR levels (Fig. 4C), whilst the downregulation of
miR-291b-3p led to increased HuR levels (Fig. 4D). However, miR-291b-3p did not
alter the mRNA levels of HuR (Fig.
4E). In addition, the overexpression of HuR decreased the rate
of apoptosis and the mRNA expression of ICAM-1 and VCAM-1 in EOMA
cells (Fig. 4F and G). In EOMA
cells transfected with AD-HUR, the p-ERK and BAX levels were
decreased, while the Bcl-2 expression levels were increased
(Fig. 4H).
 | Figure 4HuR modulated endothelial apoptosis
and dysfunction. (A) The levels of HuR protein and mRNA were
measured in EOMA cells treated with H2O2. (B)
The luciferase activity was analyzed in EOMA cells transfected with
luciferase reporter vector containing the HuR coding region or
3′-UTR. The protein level of HUR was analyzed in EOMA cells
transfected with (C) 291m or (D) 291i. (E) The mRNA level of HuR
was analyzed by qPCR in EOMA cells transfected with 291m or 291i.
(F) The levels of apoptosis in EOMA cells transfected AD-HUR or
control AD-CON vectors was measured by TUNEL staining. (G) The mRNA
levels of ICAM-1 and VCAM-1 in EOMA cells transfected with AD-HUR
or AD-CON were measured by qPCR. (H) The levels of p-ERK, Bax and
Bcl-2 were analyzed by western blot analysis. Data are presented as
the mean ± standard error of the mean (n=5). *P<0.05,
**P<0.01 and ***P<0.001 vs. control.
Con/NC, negative control; miRNA, microRNA; NCI, miRNA inhibitor
negative control; qPCR, quantitative polymerase chain reaction;
TUNEL, terminal deoxynucleotidyl-transferase-mediated dUTP nick end
labelling; ICAM-1, intercellular adhesion molecule-1; VCAM-1,
vascular cell adhesion molecule-1; ERK, extracellular
signal-regulated kinase; p-ERK, phosphorylated ERK; Bcl-2, B-cell
lymphoma 2; Bax, Bcl-2-associated X protein; HuR, Hu antigen R;
AD-CON, recombinant adenovirus vector expressing mouse HuR; AD-HUR,
adenovirus vector containing green fluorescent protein;
H2O2, hydrogen peroxide; 291m, miR-291b-3p
mimics; 291i, miR-291b-3p inhibitor; UTR, untranslated region. |
miR-291b-3p regulates apoptosis and
dysfunction of EOMA cells via targeting HuR
In order to additionally assess whether miR-291b-3p
regulated EOMA cell apoptosis via targeting HuR, miR-291b-3p mimics
and AD-HUR were co-transfected into EOMA cells for 48 h. The
results indicated that transfection with AD-HUR rescued the
miR-291b-3p mimic-induced apoptosis and the increased mRNA
expression of ICAM-1 and VCAM-1 (Fig.
5A and B). However, overexpression of miR-291b-3p did not
affect the activation of ERK and the expression levels of Bax and
Bcl-2 in EOMA cells transfected with AD-HUR (Fig. 5C). Taken together, these results
suggested that miR-291b-3p participated in endothelial dysfunction
via regulating HuR protein expression.
 | Figure 5miR-291b-3p regulates apoptosis and
dysfunction of EOMA cells via targeting HuR. (A) The levels of
apoptosis in EOMA cells co-transfected with miR-291b-3p mimics and
AD-HUR was measured by TUNEL staining. (B) The mRNA levels of
ICAM-1 and VCAM-1 were measured by quantitative polymerase chain
reaction. (C) The levels of p-ERK, Bax and Bcl-2 were analyzed by
western blot analysis. Data are presented as the mean ± standard
error of the mean (n=5). *P<0.05,
**P<0.01 and ***P<0.001 vs. control.
miR, microRNA; NC, negative control; TUNEL, terminal
deoxynucleotidyl-transferase-mediated dUTP nick end labelling;
ICAM-1, intercellular adhesion molecule-1; VCAM-1, vascular cell
adhesion molecule-1; ERK, extracellular signal-regulated kinase;
p-ERK, phosphorylated ERK; Bcl-2, B-cell lymphoma 2; Bax,
Bcl-2-associated X protein; NCI, miRNA inhibitor negative control;
291m, miR-291b-3p mimic; 291i, miR-291b-3p inhibitor; HuR, Hu
antigen R; AD-CON, recombinant adenovirus vector expressing mouse
HuR; AD-HUR, adenovirus vector containing green fluorescent
protein. |
Discussion
In the present study, it was demonstrated that
miR-291b-3p participated in endothelial dysfunction via targeting
HuR. In particular, the results indicated that: i)
H2O2 treatment increased miR-291b-3p
expression; ii) miR-291b-3p may serve an important role in
endothelial dysfunction, which is involved in the
H2O2-induced endothelial dysfunction; and iv)
miR-291b-3p regulated endothelial function via targeting HuR.
Endothelial dysfunction is a major cause of
atherosclerosis. It was demonstrated that
H2O2 damaged endothelial function by
promoting cell apoptosis and inflammation (20). In the present study, EOMA cells
were treated with H2O2 to establish cell
models of endothelial dysfunction. In this cell model, the levels
of miR-291b-3p and apoptosis were increased, accompanied by
increased mRNA levels of ICAM-1 and VCAM-1.
The association between H2O2
and endothelial dysfunction remains incompletely characterized.
Accumulating evidence has suggested that miRNAs are involved in
endothelial dysfunction (21).
miR-291b-3p belongs to the miR-290 cluster, which contains
miR-290-3p, miR-291a-3p, miR-291b-3p, miR-292-3p, miR-294 and
miR-295 (22). It was reported
that mR-291b-3p may serve important roles in differentiation of
embryonic stem cells, and the metabolism of lipids and glucose in
the liver (15-17,23). In the present study, miR-291b-3p
mimics and inhibitors were transfected into EOMA cells to
additionally investigate the role of miR-291b-3p in
H2O2-induced endothelial dysfunction. The
results suggested that miR-291b-3p served as an effector molecule
of H2O2-associated endothelial dysfunction.
miR-291b-3p may modulate the protein levels of p-ERK, Bax, Bcl-2
and mRNA expression of ICAM-1 and VCAM-1 in EOMA cells. VCAM-1 and
ICAM-1 are secreted by dysfunctional endothelial cells, leading to
attachment of inflammatory cells to the damaged endothelial cells.
The activated ERK pathway induces endothelial cells to generate
excessive levels of ICAM-1 and VCAM-1, which are major factors
responsible for the infiltration of inflammatory cells to the
atheroma-prone sites (5,24).
Next, the present study additionally identified that
miR-291b-3p regulated endothelial function via targeting HuR. It
was demonstrated previously that miR-291b contributed to hepatocyte
apoptosis by regulating the expression of HuR, which in turn
increased Bcl-2 mRNA stability (25). In the present study, it was
identified that miR-291b-3p may negatively modulate HuR protein
levels, and that the overexpression of HuR inhibited the effects of
miR-291b-3p mimics on the endothelial functions. HuR is an RNA
binding protein widely expressed in mammalian cells. AU-rich
elements (AREs)-mediated transcript degradation is considered to be
an important gene regulation mechanism at the post-transcriptional
level (26). HuR may specifically
recognize and bind to AREs to adjust mRNA stability and
translation. HuR may also be transported between the nucleus and
cytoplasm. This translocation allows HuR to efficiently modulate
the mRNA stability (27). HuR may
modulate gene expression in two distinctive mechanisms: Through one
mechanism, HuR may positively regulate gene expression by
stabilizing target mRNA, including cyclooxygenase-2, cyclin D1 and
cyclin-dependent kinase inhibitor 1 (28-30). Through the other mechanism, HuR
may also negatively modulate gene expression by decreasing the
translation efficiency of mRNA, including tumor necrosis factor-α,
myc proto-oncogene protein and cyclin-dependent kinase inhibitor 1B
(31-33). In a previous study, it was
suggested that HuR upregulated Bcl-2 expression by stabilizing its
mRNA (15). In the present study,
it was identified that the levels of p-ERK and Bax were also
decreased in EOMA cells transfected with AD-HUR. However, the
mechanism through which HuR regulates ERK phosphorylation, and Bax
and Bcl-2 expression, requires additional study.
In conclusion, the present study provides novel data
that miR-291b-3p contributes to H2O2-induced
endothelial dysfunction via targeting HUR. The present study may
provide a novel therapeutic strategy for the prevention of
atherosclerosis.
Funding
The present study was funded by Basic Research
Project of Heilongjiang Provincial Department of Education Basic
Research Project Fee (grand no. 2016-KYYWF-0593), the General
Program of Heilongjiang Province Natural Science Foundation of
China (grant no. H2015076), the Graduate Science and Technology
Innovation Projects in Jiamusi University (grant no. YZ2016_020),
the National Natural Science Foundation of China (grant nos.
81570789 and 81600618) and the Beijing Natural Science Foundation
(grant no. 7182144).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XS, SY and LD planed the experiments, XC and XL
performed the cellular experiments, JY, YS and SW analyzed the
data. FW and JL were involved in the study conception and design,
analysis and interpretation of data, drafting and critical revision
of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgments
Not applicable.
References
|
1
|
Mannarino E and Pirro M: Molecular biology
of atherosclerosis. Clin Cases Miner Bone Metab. 5:57–62.
2008.PubMed/NCBI
|
|
2
|
Ghiadoni L, Taddei S and Virdis A:
Hypertension and endothelial dysfunction: Therapeutic approach.
Curr Vasc Pharmacol. 10:42–60. 2012. View Article : Google Scholar
|
|
3
|
Yung LM, Leung FP, Yao X, Chen ZY and
Huang Y: Reactive oxygen species in vascular wall. Cardiovasc
Hematol Disord Drug Targets. 6:1–19. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Baumgartner R, Forteza MJ and Ketelhuth
DFJ: The interplay between cytokines and the Kynurenine pathway in
inflammation and atherosclerosis. Cytokine. Sep 9–2017.(Epub ahead
of print). pii: S1043-4666(17)30259-4. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Marzolla V, Armani A, Mammi C, Moss ME,
Pagliarini V, Pontecorvo L, Antelmi A, Fabbri A, Rosano G, Jaffe IZ
and Caprio M: Essential role of ICAM-1 in aldosterone-induced
atherosclerosis. Int J Cardiol. 232:233–242. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jang YJ, Park B, Lee HW, Park HJ, Koo HJ,
Kim BO, Sohn EH, Um SH and Pyo S: Sinigrin attenuates the
progression of atherosclerosis in ApoE- mice fed a high-cholesterol
diet potentially by inhibiting VCAM-1 expression. Chem Biol
Interact. 272:28–36. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Menghini R, Stohr R and Federici M:
MicroRNAs in vascular aging and atherosclerosis. Ageing Res Rev.
17:68–78. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Feinberg MW and Moore KJ: MicroRNA
regulation of atherosclerosis. Circ Res. 118:703–720. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Karakas M, Schulte C, Appelbaum S, Ojeda
F, Lackner KJ, Münzel T, Schnabel RB, Blankenberg S and Zeller T:
Circulating microRNAs strongly predict cardiovascular death in
patients with coronary artery disease-results from the large
AtheroGene study. Eur Heart J. 38:516–523. 2017.
|
|
10
|
Fichtlscherer S, De Rosa S, Fox H,
Schwietz T, Fischer A, Liebetrau C, Weber M, Hamm CW, Röxe T,
Müller-Ardogan M, et al: Circulating microRNAs in patients with
coronary artery disease. Circ Res. 107:677–684. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sun X, Icli B, Wara AK, Belkin N, He S,
Kobzik L, Hunninghake GM, Vera MP, MICU Registry, Blackwell TS,
Baron RM and Feinberg MW: MicroRNA-181b regulates NF-κB-mediated
vascular inflammation. J Clin Invest. 122:1973–1990.
2012.PubMed/NCBI
|
|
12
|
Sun X, He S, Wara AKM, Icli B, Shvartz E,
Tesmenitsky Y, Belkin N, Li D, Blackwell TS, Sukhova GK, et al:
Systemic delivery of microRNA-181b inhibits nuclear factor-κB
activation, vascular inflammation, and atherosclerosis in
apolipoprotein E-deficient mice. Circ Res. 114:32–40. 2014.
View Article : Google Scholar
|
|
13
|
Lin Y, Liu X, Cheng Y, Yang J, Huo Y and
Zhang C: Involvement of MicroRNAs in hydrogen peroxide-mediated
gene regulation and cellular injury response in vascular smooth
muscle cells. J Biol Chem. 284:7903–7913. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhang T, Tian F, Wang J, Jing J, Zhou SS
and Chen YD: Atherosclerosis-associated endothelial cell apoptosis
by miR-429-mediated down regulation of Bcl-2. Cell Physiol Biochem.
37:1421–1430. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Guo J, Li M, Meng X, Sui J, Dou L, Tang W,
Huang X, Man Y, Wang S and Li J: miR-291b-3p induces apoptosis in
liver cell line NCTC1469 by reducing the level of RNA-binding
protein HuR. Cell Physiol Biochem. 33:810–822. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Meng X, Guo J, Fang W, Dou L, Li M, Huang
X, Zhou S, Man Y, Tang W, Yu L and Li J: Liver MicroRNA-291b-3p
promotes hepatic lipogenesis through negative Regulation of
Adenosine 5′-Monophosphate (AMP)-activated protein kinase α1. J
Biol Chem. 291:10625–10634. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Guo J, Dou L, Meng X, Chen Z, Yang W, Fang
W, Yang C, Huang X, Tang W, Yang J and Li J: Hepatic miR-291b-3p
mediated glucose metabolism by directly targeting p65 to upregulate
PTEN expression. Sci Rep. 7:398992017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR ad
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
19
|
Xu MC, Gao XF, Ruan C, Ge ZR, Lu JD, Zhang
JJ, Zhang Y, Wang L and Shi HM: miR-103 regulates oxidative stress
by targeting the BCL2/Adenovirus E1B 19 kDa interacting protein 3
in HUVECs. Oxid Med Cell Longev. 2015:4896472015. View Article : Google Scholar
|
|
20
|
Yang B, Oo TN and Rizzo V: Lipid rafts
mediate H2O2 prosurvival effects in cultured endothelial cells.
FASEB J. 20:1501–1503. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Watkin RL, Fitzpatrick GG and Kerrigan SW:
The evolving role of microRNAs in endothelial cell dysfunction in
response to infection. Semin Thromb Hemost. 44:216–223. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Marson A, Levine SS, Cole MF, Frampton GM,
Brambrink T, Johnstone S, Guenther MG, Johnston WK, Wernig M,
Newman J, et al: Connecting microRNA genes to the core
transcriptional regulatory circuitry of embryonic stem cells. Cell.
134:521–533. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zheng GX, Ravi A, Calabrese JM, Medeiros
LA, Kirak O, Dennis LM, Jaenisch R, Burge CB and Sharp PA: A latent
pro-survival function for the mir-290-295 cluster in mouse
embryonic stem cells. PLoS Genet. 7:e10020542011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hoefen RJ and Berk BC: The role of MAP
kinases in endothelial activation. Vasc Pharmacol. 38:271–273.
2002. View Article : Google Scholar
|
|
25
|
Yaman I, Fernandez J, Sarkar B, Schneider
RJ, Snider MD, Nagy LE and Hatzoglou M: Nutritional control of mRNA
stability is mediated by a conserved AU-rich element that binds the
cytoplasmic shuttling protein HuR. J Biol Chem. 277:41539–41546.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sharma S, Verma S, Vasudevan M, Samanta S,
Thakur JK and Kulshreshtha R: The interplay of HuR and miR-3134 in
regulation of AU rich transcriptome. RNA Biol. 10:1283–1290. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chen CY, Xu N and Shyu AB: Highly
selective actions of HuR in antagonizing AU-rich element-mediated
mRNA destabilization. Mol Cell Biol. 22:7268–7278. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang W, Furneaux H, Cheng H, Caldwell MC,
Hutter D, Liu Y, Holbrook N and Gorospe M: HuR regulates p21 mRNA
stabilization by UV light. Mol Cell Biol. 20:760–769. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kim GY, Lim SJ and Kim YW: Expression of
HuR, COX-2, and survivin in lung cancers; cytoplasmic HuR
stabilizes cyclooxygenase-2 in squamous cell carcinomas. Mod
Pathol. 24:1336–1347. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yuan Z, Sanders AJ, Ye L, Wang Y and Jiang
WG: Knockdown of human antigen R reduces the growth and invasion of
breast cancer cells in vitro and affects expression of cyclin D1
and MMP-9. Oncol Rep. 26:237–245. 2011.PubMed/NCBI
|
|
31
|
Nabors LB, Suswam E, Huang Y, Yang X,
Johnson MJ and King PH: Tumor necrosis factor alpha induces
angiogenic factor up-regulation in malignant glioma cells: A role
for RNA stabilization and HuR. Cancer Res. 63:4181–4187.
2003.PubMed/NCBI
|
|
32
|
Talwar S, Jin J, Carroll B, Liu A,
Gillespie MB and Palanisamy V: Caspase-mediated cleavage of
RNA-binding protein HuR regulates c-Myc protein expression after
hypoxic stress. J Biol Chem. 286:32333–32343. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kullmann M, Göpfert U, Siewe B and Hengst
L: ELAV/Hu proteins inhibit p27 translation via an IRES element in
the p27 5′UTR. Genes Dev. 16:3087–3099. 2002. View Article : Google Scholar : PubMed/NCBI
|



















