Introduction
Ultraviolet B (UVB) radiation (wavelength, 280-315
nm) is reported as the most damaging component of solar radiation
reaching the surface of the Earth, predominantly exerting its
action on the epidermal layer of the skin (1,2).
Extensive exposure of the skin to UVB results in various harmful
responses, including edema, sunburn, hyperplasia, inflammation,
erythema, as well as immunosuppression, which have been reported in
different types of skin disease, and which may eventually develop
into skin cancer (3,4). Various molecular mechanisms
associated with skin injury induced by UVB have been investigated,
including oxidative stress, inflammatory responses and apoptosis
(5-7). In the present study, female animal
models were used. Previous studies using an SKH-1 mouse model
demonstrated that acute UVB exposure induces higher levels of
inflammation and lower levels of DNA damage in female mice compared
with male mice (8). Furthermore,
male SKH-1 mice exhibited a reduced capacity to eliminate reactive
oxygen species (ROS) produced following UVB exposure (9), and a reduction of the
immunoprotective response following UVA exposure, as well as a
reduction of the immunoprotective response following UVA exposure
relative to females (10,11).
As previously described, ROS is crucial for skin
damage, accounting for 50% of skin injury due to UVB irradiation
(12). Exposure of skin to UVB
initiates a photo-oxidative reaction, accelerating cellular ROS
levels (13). The redox-sensitive
transcription factor, nuclear factor-E2-related factor 2 (Nrf2) is
important in modulating ROS production. Nrf2, thus, has been
considered as an essential molecular target for various
pharmacological prevention strategies in human and animal
pathologies due to exposure to environmental toxicants, which
involve UV light (14,15). According to a previous study, Nrf2
is proposed as a protective signal in gene expression mediation to
inhibit skin damage induced by UV irradiation (16). Therefore, pharmacological
modulation of Nrf2 may potentially protect skin from damage by UV
exposure. Additionally, skin inflammation is another major
mechanism that causes skin injury induced by UV (17). Nuclear factor (NF)-κB), a
redox-sensitive transcriptional factor, is critical in the
pathogenesis of UVB-triggered inflammation, which is linked to skin
carcinogenesis (18). NF-κB
activation modulates inflammatory responses by enhancing
pro-inflammatory cytokine secretion, including tumor necrosis
factor (TNF)-α, interleukin (IL)-1β and cyclic oxidase 2 (COX2)
(19,20). Hence, NF-κB inactivation blocks
inflammatory responses induced by UVB irradiation.
Juglanin (Fig.
1A), as a natural compound extracted from the crude
Polygonum aviculare, displayed inhibitory activity against
the inflammatory response, dependent on NF-κB de-phosphorylation,
as well as anticancer activity through regulating ROS production
with little cytotoxicity on normal cells (21-23). Considering its effect on
inflammation and ROS, in the current study, the effects of juglanin
on skin damage induced by UVB were investigated. The underlying
molecular mechanisms were also further explored in vitro and
in vivo.
Materials and methods
Animals and treatment
Sixty female, SKH-1 hairless mice (age, 6-8 weeks;
weight, 18-20 g) were purchased from the Shanghai Laboratory Animal
Research Center (Shanghai, China). The mice were acclimatized for 1
week prior to the experiments in specific pathogen-free (SPF)
conditions in static micro-isolator cages with access to tap water
ad libitum and maintained under standard conditions (12-h
light/dark cycle; 8:00 a.m. to 8:00 p.m.) at a temperature of
23±2°C and relative humidity of 50±5%. All animal experiments were
conducted according to the Guide for the Care and Use of Laboratory
Animals, issued by the National Institutes of Health in 1996 and
approved by the Institutional Animal Ethics Committee for the Guide
for the Care and Use of Laboratory Animals of Huai'an First
People's Hospital, Nanjing Medical University (Huai'an, China).
SKH-1 hairless mice were then randomly divided into 4 groups with
15 mice per group. Mice were exposed to UVB lamps (GL20SE; Sankyo
Denki Co., Ltd., Kanagawa, Japan) equipped with a controller to
modulate UV dosage, with a distance of 20 cm between the target
skin and the light source. The treatment groups (n=15 per group)
were as follows: Group 1, untreated animals (Con); group 2, animals
irradiated with UVB only (UVB); group 3, UVB irradiation with
application of 15 mg/kg juglanin (cat. no. 5041-67-8; purity ≥98%;
Shanghai YuanMu Biological Technology Co., Ltd., Shanghai, China)
by gavage (UVB+15); group 4, UVB irradiation with application of
juglanin (30 mg/kg) by gavage (UVB+30), which was subsequent to 30
min UVB (50 mJ/cm2). Murine skin exposure consisted of
50 mJ/cm2 UVB following juglanin treatment for 1 h, 3
times per week for 10 consecutive weeks. During the period of
exposure, mice were individually housed in stainless steel
chambers. A non-irradiated group of animals was included as a UVB
negative control. After 10 weeks of exposure with or without
juglanin treatment, the animals were euthanized 24 h after the
final UVB irradiation, and the dorsal skin tissues from the mice
were excised, maintained in liquid nitrogen and stored at -80°C for
subsequent research. A total of 6 skin tissue samples (1×1
cm2) were selected randomly and fixed in 4% formalin for
48 h at room temperature and subsequently embedded in paraffin for
immunohistochemical analysis.
Cell culture and treatment
Human epidermal cells, HaCaT, were purchased from
the American Type Culture Collection (Manassas, VA, USA). Cells
were cultured in Dulbecco's modified Eagle's medium supplemented
with 10% fetal bovine serum (Invitrogen; Thermo Fisher Scientific,
Inc., Waltham, MA, USA), penicillin (100 U/ml) and streptomycin
(100 µg/ml) at 37°C in an atmosphere of 5% CO2.
The HaCaT cells were cultured until 80% confluence and then
pretreated with various concentrations of juglanin (80 and 160
µM). Following a 12-h incubation at 37°C, the culture medium
was replaced with 1.5 ml phosphate-buffered saline (PBS). HaCaT
cells were then exposed to 15 mJ/cm2 UVB (light source,
312 nm). After UVB exposure, the cells were treated with various
concentrations of juglanin (80 and 160 µM) in serum-free
DMEM medium (Gibco; Thermo Fisher Scientific, Inc.) for another 12
h (21,23). Mitogen activated protein kinases
(MAPK) inhibitors of SB203580 [high-performance liquid
chromatography (HPLC) ≥98%], PD98059, SP600125 (HPLC, ≥98%), and
NF-κB (p65) inhibitor, JSH-23 (HPLC, ≥98%), were purchased from
Sigma-Aldrich (Merck KGaA, Darmstad, Germany). The inhibitors were
dissolved in dimethyl sulfoxide (DMSO; Beyotime Institute of
Biotechnology, Shanghai, China) and added to the culture media. The
cells were pre-treated with each inhibitor at the indicated
concentrations [SB203580 (50 nM), PD98059 (10 nM) and SP600125 (50
µM)] for 1 h and/or with juglanin for 12 h, and exposed to
15 mJ/cm2 UVB irradiation for 24 h, followed by
incubation for another 12 h in the absence or presence of juglanin
(160 µM) at 37°C. After 48 h treatment, the cells were
harvested for further study.
Intracellular ROS analysis
Following incubation with various concentrations of
juglanin (0, 80 and 160 µM), 5×105 cells/200
µl HaCaT cells were re-incubated with dihydroethidium (DHE)
for 10 min at room temperature using a Reactive Oxygen Species
Assay kit (cat. no. C1300; BestBio, Shanghai, China) according to
the manufacturer's protocol in Hanks' balanced salt solution for 30
min at 37°C. Following incubation, the fluorescence of DHE was
measured spectro-fluorimeterically at 535 nm excitation and 610 nm
emission wavelengths.
Cell viability assays
A
3-(4,5-dimethylthiazol-2-yl)-2,5-di-phenyltetrazolium bromide (MTT;
cat. no. C0009; Beyotime Institute of Biotechnology) colorimetric
assay kit was used to measure cell viability, according to the
manufacturer's protocol. A total of 103 HaCaT cells were
maintained until 80% confluence and then pretreated with different
concentrations (0, 5, 10, 20, 40, 80 and 160 µM) of juglanin
for various durations (0, 6, 12, 24, 36 and 48 h). Cell medium was
replaced with 150 µl MTT and incubated at 37°C for an
additional 4 h. Once the HaCaT cells were washed using PBS twice,
the insoluble formazan crystals were dissolved in 200 µl
DMSO. The absorbance was read by spectrophotometry at a wavelength
of 540 nm using a Multiskan EX microplate reader (Thermo Fisher
Scientific, Inc.). The data were represented as percentage cell
viability compared with the untreated control group.
Biochemical indicator analysis
The activities of enzymatic antioxidants in skin
tissue samples, including superoxide dismutase (SOD), catalase
(CAT) and glutathione peroxides (GPx) were analyzed using SOD (cat.
no. A001-1-1), CAT (cat. no. A007-1-1) and GPx (cat. no. A005)
assay kits (all from Nanjing Jiancheng Bioengineering Institute,
Nanjing, China), respectively. The levels of reduced glutathione
(GSH) and malondialdehyde (MDA) in skin tissue samples were
measured using GSH (cat. no. A006-2) and MDA (cat. no. A003-1)
assay kits (Nanjing Jiancheng Bioengineering Institute) according
to the manufacturer's protocol. The thiobarbituric acid reactive
substance (TBARS) levels in the skin tissues were calculated using
a mouse TBARS ELISA kit (cat. no. xy-E10068; Runyubio-Technology
Co., Ltd., Shanghai, China) according to the manufacturer's
protocol.
ELISA for transforming growth factor-β1
(TGF-β1) assessment
The skin samples were frozen in liquid nitrogen and
crushed into a powder using a multibead shocker. The powder was
dissolved in cell lysis buffer for Western and IP (Beyotime
Institute of Biotechnology) with protease inhibitor cocktail (Roche
Diagnostics, Indianapolis, IN, USA). The skin extract was prepared
by centrifugation at 12,000 × g for 10 min at 4°C, and the
supernatant was stored for further experiments. The sample protein
concentrations were calculated using a BCA protein assay kit (cat.
no. 23250; Thermo Fisher Scientific Inc.) according to the
manufacturer's protocol. TGF-β1 protein expression levels were
assessed using a Mouse ELISA kit (R&D Systems, Inc.,
Minneapolis, MN, USA) according to the manufacturer's protocol.
Transepidermal water loss (TEWL)
analysis
TEWL (g/m2/h) is a marker of epidermal
skin barrier function, and was measured using a Tewameter™ 300
(Courage + Khazaka Electronic GmbH, Köln, Germany). Briefly, under
isoflurane anesthesia, a medical adhesive tape (Honsmed, Shanghai,
China) was used to apply a gentle pressure on the mouse skin; then
it was removed. Mice were tape-stripped 5 times. TEWL was then
recorded using a Tewameter™ 300.
Calculation of wrinkle formation in the
dorsal skin
The dorsal skin samples of mice were collected
following the final UVB irradiation and replicated using a silicone
product under isoflurane anesthesia. The representative images of
the mouse skin replica were analyzed using a 3D Magic Mirror Skin
Analyzer Machine SW-26A equipped with Skin-Visiometer VL 650
software (Sunwein-tech, Guangdong, China). The parameters to
evaluate skin wrinkles were wrinkle volume ratio, wrinkle area
ratio, total groove volume ratio and the number of wrinkles.
Assessment of ROS generation
ROS generation in cells was measured with DHE using
a Dihydroethidium kit (cat. no. S0063; Beyotime Institute of
Biotechnology), purchased from Beyotime Institute of Biotechnology
according to the manufacturer's protocol. Following the various
treatments, cells were incubated with DHE (10 µM) for 30 min
under 37°C, and the cells were washed with serum-free DMEM medium
three times. The fluorescence intensity was observed under a
confocal microscope (Olympus Corporation, Tokyo, Japan) at a
magnification of ×200.
Immunohistochemical (IHC) analysis
The fixed skin tissue samples obtained from mice
were embedded in paraffin blocks following euthanasia. Skin samples
were dehydrated in ascending concentrations of ethanol (80, 95 and
100%), cleared in xylene, embedded in Paraplast (Sigma Aldrich;
Merck KGaA) and 3-µm thick sections were cut. Skin sections
were deparaffinized and stained for 15 min with hematoxylin and
eosin (H&E) at room temperature. The thickness of the skin
epidermis was assessed using Magnuspro software (version 3.0; Plan
Achromate, Olympus, Germany). Epidermal thickness of the
H&E-stained sections was further assessed with ImageJ Software
(version 1.47; National Institutes of Health, Bethesda, MD, USA).
For immunohistochemical images, the skin tissue sections were
exposed to HCl (3.5 M) for 20 min at room temperature and washed
using PBS three times. Subsequently, the skin tissue sections were
treated with peroxidase (0.3%) to diminish endogenous peroxidase
activity. Tissue sections were incubated with normal goat serum
(5%) for 30 min followed by incubation with primary antibodies
[inducible nitric oxide synthase (iNOS; cat. no. 13120) and TGF-β1
(cat. no. 3711); Cell Signaling Technology, Inc., Danvers, MA,
USA)] at 1:100 dilution for 2 h at room temperature. The section
was then incubated with goat anti-rabbit immunoglobulin G (IgG)
horseradish peroxidase (HRP)-conjugated secondary antibody (1:250;
cat. no. 150077; Abcam, Cambridge, MA, USA) at room temperature for
30 min. Diaminobenzidine (ChemService, Inc., West Chester, PA, USA)
served as the chromogen according to the manufacturer's
protocol.
Immunofluorescent analysis of skin
tissue
The skin tissue sections were dried for 10 min at
room temperature, fixed with chilled acetone for 10 min at -20°C,
and washed with PBS three times (5 min per wash). The
pre-incubation was conducted with 5% normal rabbit serum
(SouthernBiotech, Birmingham, AL, USA) at room temperature for 1 h,
and sections were incubated with the respective specific
antibodies: Polyclonal rabbit anti-phosphorylated (p)-NF-κB (cat.
no. 86299; dilution, 1:50; Abcam), polyclonal rabbit anti-iNOS
(cat. no. 13120; dilution, 1:100; CST Biological Reagents Co.,
Ltd., Shanghai, China) at 4°C overnight. Subsequently, the slides
were washed with PBS three times and treated with HRP-conjugated
anti-rabbit monoclonal IgG secondary antibody (cat. no. A0208;
dilution: 1:200; Beyotime Institute of Biotechnology) for 15 min at
room temperature. Sections were counterstained for cell nucleus
detection using 4',6-diamidino-2-phenylindole (Thermo Fisher
Scientific, Inc.) solution for 5 min at room temperature, washed
with PBS three times (5 min per wash), and mounted in
17984-25-Fluoromount-G™ Slide Mounting Medium (Southern Biotech,
Birmingham, AL, USA). All fluorescent images were obtained using a
fluorescence microscope (BX53, Olympus, Japan) at a magnification
of ×200.
Determination of myeloperoxidase (MPO)
activity
Tissue MPO was evaluated according to the protocol
provided by the MPO kit's manufacturer (USCNK, Wuhan, China). This
assay provides a fluorescence-based method for detecting the MPO
activity in tissue lysates.
Western blot analysis
Following various cell treatments as described
previously in the cell culture and treatment sections, all HaCaT
cells were collected for western blot analysis. Briefly, HaCaT
cells were pelleted and lysed with cell lysis reagent (0.216% Beta
glycerophosphate, 0.19% Sodium orthovanadate, 0.001% Leupeptin,
0.38% EGTA, 10% Triton-X-100, 3.15% Tris HCl, 8.8% Sodium chloride,
0.29% Sodium EDTA, 1.12% Sodium pyrophosphate decahydrate; Beyotime
Institute of Biotechnology) in the presence of protease inhibitor
cocktail (Beyotime Institute of Biotechnology). Mice were
euthanized and dorsal-skin tissue samples were isolated. The skin
tissue samples were homogenized into 10% (w/v) hypotonic buffer [25
mM Tris-HCl (pH 8.0), 1 mM EDTA, 5 µg/ml leupeptin, 1 mM
Pefabloc SC, 50 µg/ml aprotinin, 5 µg/ml soybean
trypsin inhibitor and 4 mM benzamidine] to yield a homogenate. The
final supernatants from the cells and skin tissue samples were
obtained by centrifugation at 12,000 × g for 20 min at 4°C. Protein
content was calculated using a BCA Protein Assay kit (cat. no.
23250; Thermo Fisher Scientific, Inc.). The extracted proteins were
denatured at 100°C for 5 min. Proteins (40 µg/well) were
loaded in 10% Tris-SDS gel and transferred to polyvinylidene
difluoride membranes, (EMD Millipore, Billerica, MA, USA) using the
wet transfer system. After blocking for 2 h at 37°C with 5% skimmed
milk in Tris-buffered saline with 0.1% Tween-20, the membranes were
incubated overnight at 4°C with anti-protein primary antibodies
against p-NF-κB (cat. no. ab86299; dilution, 1:1,000), NF-κB (cat.
no. ab16502; dilution, 1:1,000) (both from Abcam), p-ERK1/2 (cat.
no. 8544; dilution, 1:1,000), ERK1/2 (cat. no. 4695; dilution,
1:1,000) (both from CST Biological Reagents Co., Ltd.), p-JNK (cat.
no. ab47337; dilution, 1:1,000), Nrf2 (cat. no. ab62352; dilution,
1:1,000), JNK (cat. no. 112501; dilution 1:1,000) (all from Abcam),
p-p38 (cat. no. 4511; dilution, 1:1,000), p38 (cat. no. 8690;
dilution, 1:1,000), COX2 (cat. no. 12282; dilution, 1:1,000), IL-1β
(cat. no. 12703; dilution, 1:1,000), TNF-α (cat. no. 11948;
dilution, 1:1,000), iNOS (cat. no. 13120; dilution, 1:1,000),
TGF-β1 (cat. no. 3711; dilution, 1:1,000) (all from CST Biological
Reagents Co., Ltd.), and GAPDH (cat. no. sc293335; dilution, 1:500;
Santa Cruz Biotechnology, Inc., Dallas, TX, USA) in blocking
buffer. The membrane was then incubated for 3 h at room temperature
with secondary anti-primary immunoglobulin G-conjugated with HRP
(dilution, 1:5,000; cat. no. GTX213110-01; GeneTex, Inc., Irvine,
CA, USA), followed by application of immobilon Western
Chemiluminescent HRP substrate (EMD Millipore). Western blot bands
were observed using an ECL Western Blotting Analysis System (GE
Healthcare, Chicago, IL, USA) and exposed to X-ray films (Kodak,
Rochester, NY, USA) for protein expression analysis (using ImageJ
software version 1.47d; National Institutes of Health, Bethesda,
MD, USA).
Small interfering RNA (siRNA)
treatment
The HaCaT cells (4.0×105) were cultured
at 37°C in 6-well plates for 24 h. Then, Nrf2 siRNA (Nrf2 siRNA,
5′-AUG GGC UAC UCG GCU AGC AAU-3; negative control, 5′-UGU AAU GGU
GCC CAG ACC G-3′) were added to the cells using the transfection
reagent Lipofectamine® 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.) according to the manufacturer's protocol. A final
concentration of 50 nM was prepared in the 6-well plates.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR) analysis
Total RNA was isolated from the skin tissue samples
and HaCaT cells using the TRIzol reagent (Nanjing KeyGen Biotech
Co., Ltd., Nanjing, China). RT-PCR was conducted using the
PrimeScript RT Reagent kit (Takara Biotechnology Co., Ltd., Dalian,
China) according to the manufacturer's protocol and cDNA then
served as the template for subsequent reactions. RT-qPCR was
conducted with SYBR Premix Ex Taq II obtained from Takara
Biotechnology Co., Ltd. The ABI-Prism 7500 Sequence Detection
System (Applied Biosystems; Thermo Fisher Scientific, Inc.)
according to the manufacturer's protocol. The primer sequences used
in the current study were commercially synthesized and presented in
Table I. The mRNA level of GAPDH
served as the loading control. The 2−ΔΔCq method was
applied to evaluate the fold changes of mRNA levels in each group
(24).
 | Table IPrimer sequences for the reverse
transcription-polymerase chain reaction. |
Table I
Primer sequences for the reverse
transcription-polymerase chain reaction.
| Items | Primer (5'→3')
|
|---|
| Forward | Reverse |
|---|
| Cyclic oxidase
2 |
TTATAGATTGATACTCACCA |
AGTGACAGTAGATCTGAGG |
| Interleukin-1β |
TGGCCTGACCACAGGAGTTA |
CTTCCGTGACTTAGTATACTAT |
| Tumor necrosis
factor-α |
GCTCTTCTCTGTCGTGTCTGCC |
AATGTATTGTTGGAATTATAC |
| Superoxide
dismutase 1 |
AGCATCCAAGTGGCAGTAA |
AGACCAACGACCAGCTCAG |
| Superoxide
dismutase 2 |
GTCTGTAGACACCGCACA |
CGAGGCCATAACTGTGACT |
| Catalase |
AGAGAGCTCGATGTTACG |
CCTTCTCGTCTGACACTGAG |
| Nuclear
factor-E2-related factor 2 |
CCAGCCGTTCTACTTCCAC |
GGAAGCACTCCAGTGGACTA |
| GAPDH |
AGAATGGCAGCTTGTGCGTG |
CTTGGAGGTGCCAACACCTC |
Statistical analysis
Data were expressed as the mean ± standard error of
the mean. Statistical analyses were performed using GraphPad Prism
(version 6.0; GraphPad Software, Inc., La Jolla, CA, USA) by
one-way analysis of variance with Dunnet's least significant
difference post-hoc test. P<0.05 was considered to indicate a
statistically significant difference.
Results
Juglanin suppresses UVB-induced skin
injury in a mouse model
Irradiation of UVB has been reported to cause
hyperplasia, cutaneous edema, leukocyte infiltration, vascular
hyperpermeability and erythema (3,5).
Thus, it is necessary to establish an effective treatment. In the
present study, the role of juglanin (Fig. 1A) in UVB-induced skin damage was
investigated in hairless mice. As demonstrated in Fig. 1B, UVB exposure led to skin damage
with higher skin thickness compared with the control group, which
was significantly reduced by juglanin administration (P<0.05 and
P<0.01 compared with the UVB group), as demonstrated by H&E
staining, indicating that juglanin ameliorated UVB-induced
hyperplasia and cell infiltration in the dermis of skin. MPO is
considered to be a marker of cutaneous infiltration induced by UVB
exposure (25). The current study
identified that MPO was upregulated by ~2.0-fold in UVB-induced
skin when compared with the control group, indicating MPO activity
resulting from UVB exposure. Notably, juglanin treatment reduced
MPO in hairless mice with UVB induction (Fig. 1C). Finally, in order to
investigate the epidermal permeability barrier function, the TEWL
of the stratum corneum was measured following UVB induction with or
without juglanin administration. As presented in Fig. 1D, TEWL was highly increased for
UVB irradiation, which was suppressed by exposure to juglanin.
Thus, juglanin may contribute to improvement of skin injury induced
by UVB irradiation.
Effects of juglanin treatment on
UVB-induced wrinkle progression in hairless mice
The process of wrinkle formation may indicate the
extent of skin damage (26). The
levels of wrinkle formation were evaluated in the present study. In
Fig. 2A, deep coarse wrinkles
were observed to be formed in mice following UVB exposure, which
was attenuated following juglanin administration. The extent of
wrinkle formation was analyzed using a 3D image analysis system.
The ratios of total groove volume (Fig. 2B), wrinkle area (Fig. 2C), wrinkle volume (Fig. 2D) and the number of wrinkles
(Fig. 2E) were significantly
increased in the UVB-treated group compared to the Con group
(P<0.01 and P<0.001 compared with the Con group). Notably,
juglanin treatment markedly reduced these indicators, indicating
its role in improving UVB-induced skin damage.
Role of juglanin in UVB-induced MDA,
anti-oxidants and TBARS levels in hairless mice
According to previous studies, UVB irradiation
induces oxidative stress, which is associated with membrane lipids
(27). In the current study, MDA
was evaluated to assess the extent of oxidative stress. In Fig. 3A, MDA levels were identified to be
increased in the UVB group, which was an increase of >2.0-fold
when compared with the control group. In addition, juglanin
apparently reduced MDA production. Furthermore, anti-oxidant
levels, including SOD, CAT, GPx and GSH, were analyzed to evaluate
the role of juglanin in regulating UVB-induced oxidative stress in
hairless mice. From Fig. 3B-E,
the SOD, CAT and GPx activities, as well as the GSH levels were
markedly reduced following UVB exposure. Treatment with juglanin
markedly restored SOD, CAT and GPx activity. The GSH levels were
also reversed, which was comparable with the UVB-alone group.
TBARS, a lipid peroxidation product, was observed at high levels
following UVB irradiation, and juglanin decreased the TBARS levels
markedly when compared with the UVB-exposed skin (Fig. 3F). These findings elucidated that
juglanin may increase anti-oxidant levels and decrease lipid
peroxidation induced by UVB, thus improving oxidative stress.
 | Figure 3Role of juglanin in UVB-induced MDA,
anti-oxidants and TBARS levels in hairless mice. (A) MDA levels,
and (B) SOD, (C) CAT, and (D) GPx activity, and (E) GSH and (F)
TBARS levels in the skin tissue of hairless mice treated under
different conditions. Data are presented as the mean ± standard
error of the mean (n=8). **P<0.01 and
***P<0.001 vs. the Con group. +P<0.05,
++P<0.01 and +++P<0.001 vs. the UVB
group. UVB, ultraviolet B; MDA, malondialdehyde; TBARS,
thiobarbituric acid reactive substance; SOD, superoxide dismutase;
CAT, catalase; GPx, glutathione peroxides; GSH, glutathione; Con,
control. |
Juglanin inhibits UVB-induced oxidative
stress in hairless mice
According to the results, oxidative stress may be
involved in juglanin-ameliorated skin damage triggered by UVB.
Therefore, the present study attempted to investigate the potential
molecular mechanism by which juglanin attenuated skin injury. In
Fig. 4A and B, IHC analysis
indicated that iNOS and TGF-β1 expression levels were markedly
induced in the UVB-alone treatment group. Notably, juglanin exerted
a suppressive role in regulating iNOS and TGF-β1 expression levels.
By contrast, Nrf2 (an essential anti-oxidative factor) was revealed
to be reduced in the skin tissue sections from mice with UVB
irradiation alone. Additionally, co-treatment with juglanin
elevated Nrf2 expression levels, indicating a potential mechanism
to inhibit oxidative stress (Fig.
4C). Furthermore, in line with the IHC findings, western
blotting confirmed the results that iNOS and TGF-β1 expression
levels were highly induced in the UVB-treated group, and were
reversed by juglanin treatment (Fig.
4D-F). In addition, the UVB-induced reduction in Nrf2 protein
expression levels was augmented by juglanin exposure (Fig. 4D and G). Thus, the findings
illustrated that juglanin-reduced oxidative stress was associated
with iNOS and TGF-β1 downregulation, as well as Nrf2
upregulation.
The MAPK signaling pathway is closely associated
with oxidative stress development (28). p38, ERK1/2, and JNK are three
significant members of the MAPK family (29). The present study identified that
p38, ERK1/2 and JNK phosphorylation levels were markedly induced in
the UVB group, according to western blotting (Fig. 5A). Juglanin administration
significantly (P<0.05, P<0.01 and P<0.001 compared with
the UVB group) reduced phosphorylation of p38, ERK1/2 and JNK
compared to UVB group (Fig.
5B-D). Thus, it is proposed that juglanin-attenuated oxidative
stress is associated with MAPK signaling pathway suppression.
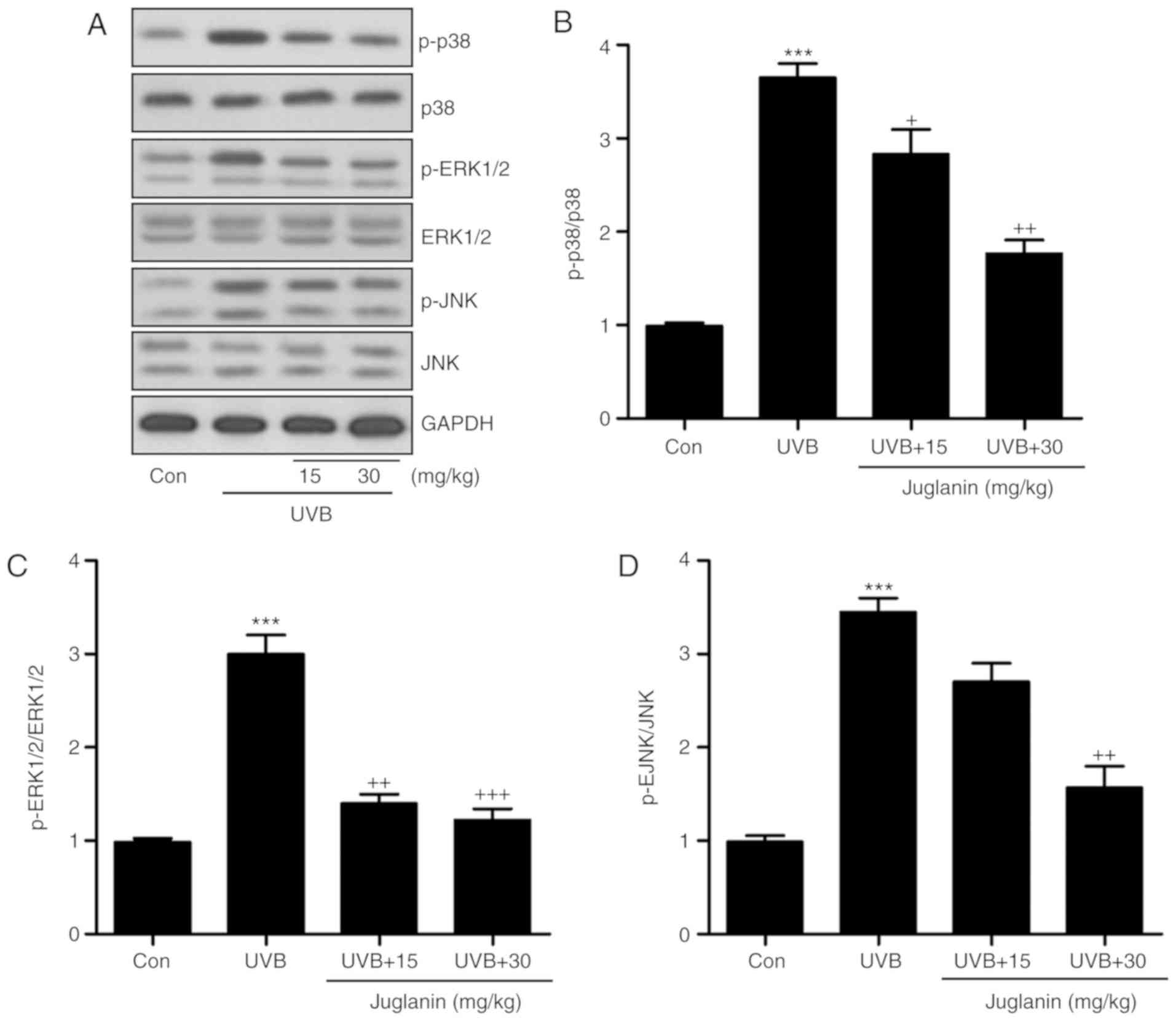 | Figure 5Juglanin impedes ROS generation via
MAPK signaling pathway suppression. (A) Western blot analysis was
applied to determine p-p38, p-ERK1/2, and p-JNK protein expression
levels. (B) The p-p38, (C) p-ERK1/2, and (D) p-JNK expression
levels were quantified according to the results of immunoblotting
analysis. Data are presented as the mean ± standard error of the
mean (n=8). ***P<0.001 vs. the Con group.
+P<0.05, ++P<0.01 and
+++P<0.001 vs. the UVB group. ROS, reactive oxygen
species; MAPK, mitogen activated protein kinases; p,
phosphorylated; ERK, extracellular signal-regulated kinase; JNK,
c-Jun N-terminal kinases; Con, control; UVB, ultraviolet B. |
Jugalanin inhibits the inflammatory
response in UVB-induced hairless mice
Pro-inflammatory cytokine release is important in
modulating tissue damage (30).
Previous reports indicated that UVB enhanced the inflammatory
response in the skin of mice, contributing to skin injury (31). Hence, the current study
investigated whether juglanin could perform its role in
ameliorating skin damage by altering the inflammatory response. As
demonstrated in Fig. 6A, western
blotting indicated that pro-inflammatory cytokines, COX2, IL-1β,
and TNF-α were highly induced in the UVB group, which was
consistent with previous studies (32). Notably, juglanin was demonstrated
to reduce the expression levels of these cytokines, indicating its
role in ameliorating the inflammatory response. NF-κB is important
for pro-inflammatory cytokine secretion via transcription
modulation (17). In Fig. 6B, NF-κB was phosphorylated in the
UVB group, which was inactivated by juglanin administration in a
dose-dependent manner. Furthermore, immunofluorescent analysis
confirmed that juglanin reduced UVB-induced NF-κB phosphorylation
(Fig. 6C). Thus, the data
indicated that juglanin reduces pro-inflammatory cytokine release
by dephosphorylating NF-κB to attenuate UVB-induced skin damage in
mice.
 | Figure 6Jugalanin inhibits the inflammatory
response in UVB-exposed hairless mice. (A) Western blot analysis
was performed to examine COX2, IL-1β and TNF-α expression levels in
the skin tissue samples isolated from mice, and the quantified
levels are presented. (B) p-NF-κB levels were measured using
western blot analysis and quantified. (C) Immunofluorescent assays
were performed to determine the NF-κB phosphorylation levels in the
skin sections isolated from the hairless mice. The positive cells
expressing p-NF-κB levels were quantified. Scale bar, 50 µm.
Data are presented as the mean ± standard error of the mean (n=8).
**P<0.01 and ***P<0.001 vs. the Con
group. +P<0.05, and ++P<0.01 vs. the
UVB group. UVB, ultraviolet B; COX2, cyclic oxidase 2; IL-1β,
interleukin-1β; TNF-α, tumor necrosis factor-α; p, phosphorylated;
NF-κB, nuclear factor-κB; Con, control. |
Juglanin ameliorates UVB-induced ROS
generation in human skin cells in vitro
In order to further confirm the effects of juglanin
on skin damage attenuation, the human epidemical cell line, HaCaT,
was included and cells were treated under various conditions. The
cytotoxicity of juglanin in HaCaT was evaluated using MTT analysis.
As shown in Fig. 7A, HaCaT were
exposed to various concentrations of juglanin (0-160 µM),
for 24 h, which was followed by cell viability analysis. No
significant difference was observed between the different groups.
Additionally, 160 µM juglanin was administered to cells for
different times as indicated. Consistently, the cell viability was
not markedly changed at the highest concentration treatment, even
after 72 h. Subsequently, HaCaT cells were exposed to 80 and 160
µM juglanin following UVB exposure for 24 h. DHE analysis
was performed to analyze ROS generation. Similar to the results
in vivo, ROS production was markedly induced in the HaCaT
cells subsequent to UVB irradiation. Notably, juglanin
administration reduced ROS generation in a dose-dependent manner
(Fig. 7B). RT-qPCR assays were
performed to investigate the anti-oxidants, SOD1, SOD2 and CAT and
Nrf2 gene expression levels. The data demonstrated that juglanin
restored SOD1, SOD2, CAT and Nrf2 mRNA expression levels that were
reduced by UVB (Fig. 7C).
Furthermore, iNOS fluorescent intensity was downregulated in
UVB-treated cells following juglanin exposure (Fig. 7D). Finally, the UVB-induced
elevated expression levels of TGF-β1 in the supernatant of cells
were reduced by co-culture with juglanin (Fig. 7E). These data illustrated that
juglanin reduced UVB-induced ROS production in vitro.
 | Figure 7Juglanin ameliorates UVB-induced ROS
generation in human skin cells in vitro. (A) Human epidermal
cells, HaCaT, were treated with various concentrations of juglanin,
ranging from 0 to 160 µm, for 24 h, followed by cell
viability analysis by MTT assay. HaCaT cells were treated with 160
µm juglanin for various durations as indicated. MTT analysis
was used to evaluate the cell viability for assessing juglanin
cytotoxicity in vitro. HaCaT cells were pretreated with
juglanin for 12 h, and exposed to 15 mJ/cm2 UVB
irradiation for 24 h, followed by incubation for another 12 h in
the absence or presence of juglanin at the indicated
concentrations. Cells were then harvested for subsequent
investigation. (B) ROS production was evaluated using DHE analysis
and the quantification of ROS levels is presented. Scale bar, 100
µm. (C) Reverse transcription-quantitative polymerase chain
reaction analysis was performed to evaluate SOD1, SOD2, CAT and
Nrf2 mRNA abundance in cells treated under different conditions.
(D) The immunofluorescent analysis was used to evaluated
iNOS-positive cells following various treatments. Scale bar, 25
µm. (E) The supernatant of cells was collected after
different treatments for TGF-β1 assessment using ELISA method and
the quantified results are presented. Data are presented as the
mean ± standard error of the mean (n=8). **P<0.01 and
***P<0.001 vs. the Con group. +P<0.05,
++P<0.01 and +++P<0.001 vs. the UVB
group. UVB, ultraviolet B; ROS, reactive oxygen species; MTT,
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; SOD,
superoxide dismutase; CAT, catalase; Nrf2, nuclear
factor-E2-related factor 2; iNOS, inducible nitric oxide synthase;
TGF-β1, transforming growth factor-β1; Con, control. |
Juglanin-induced suppression of ROS
generation is dependent on Nrf2 activity
The MAPK signaling pathway was identified to be
involved in juglanin-attenuated skin damage in vivo. Thus,
the present study attempted to further confirm its role in
vitro. Western blotting indicated that p38, ERK1/2 and JNK
phosphorylation was induced by UVB irradiation, which was
consistent with the findings in vivo. Similarly, juglanin
displayed inhibitory effects (Fig.
8A-D). Nrf2 is a key gene in the regulation of ROS generation
(33). The MAPK signaling pathway
is regulated by Nrf2 activity, as previously described (34). Thus, to further investigate the
molecular mechanism, Nrf2 was silenced using a specific siRNA
sequence. Western blotting indicated that Nrf2 knockdown was
successfully induced (Fig. 8E).
HaCaT cells were then exposed to UVB with or without juglanin and
Nrf2 silencing. The findings indicated that subsequent to Nrf2
knockdown, juglanin exhibited no significant effects on p38, ERK1/2
and JNK phosphorylation in UVB-induced HaCaT cells, indicating that
juglanin may depend on Nrf2 activity to modulate MAPK expression,
which is involved in ROS production (Fig. 8F).
 | Figure 8Juglanin-induced suppression of ROS
generation is dependent on Nrf2 activity via the inactivation of
the MAPK signaling pathway. (A) Western blot analysis was used to
calculate p38, ERK1/2 and JNK phosphorylation. (B) p-p38, (C)
p-ERK1/2, and (D) p-JNK expression levels were calculated following
immunoblotting analysis. (E) Nrf2 was silenced using a specific
Nrf2 siRNA sequence. Western blotting was performed to calculate
Nrf2 expression levels following knockdown. The representative
images are presented. (F) The HaCaT cells were pretreated with
juglanin administration in the presence or absence of Nrf2 siRNA
for 12 h, and exposed to UVB for 24 h, followed by juglanin
administration for another 12 h. Immunoblotting analysis was
performed to evaluate p-p38, p-ERK1/2 and p-JNK levels for
investigating the role of Nrf2 in juglanin-treated cells following
UVB exposure. Data are presented as the mean ± standard error of
the mean (n=8). **P<0.01 and ***P<0.001
vs. the Con group. +P<0.05, ++P<0.01
and +++P<0.001 vs. the UVB group. ROS, reactive
oxygen species; MAPK, mitogen activated protein kinase; Nrf2,
nuclear factor-E2-related factor 2; UVB, ultraviolet B; ERK,
extracellular signal-regulated kinase; JNK, c-Jun N-terminal
kinases; Con, control. |
As juglanin was revealed to suppress the
inflammatory response induced by UVB exposure in vivo,
further studies were also conducted in vitro to confirm the
results of the present study. Consistently, COX2, IL-1β and TNF-α
gene expression levels were higher in the UVB-treated group
compared with those of the control group, which were reversed
following juglanin administration (Fig. 9A). In addition, western blot
analysis confirmed the effects of juglanin on reducing
pro-inflammatory cytokine secretion in cells subsequent to UVB
irradiation (Fig. 9B and C). In
addition, NF-κB de-phosphorylation by juglanin in UVB-induced HaCaT
cells was observed (Fig. 9D). The
results indicated that juglanin, indeed, reduced the inflammatory
response by regulating NF-κB activity. Furthermore, there is a
close association between Nrf2 and NF-κB phosphorylation,
influencing pro-inflammatory cytokine secretion. Thus, Nrf2 was
silenced to further investigate the inflammatory response. As
demonstrated in Fig. 9E,
following Nrf2 knockdown and similar to MAPK expression, NF-κB
phosphorylation was not reduced following juglanin administration
in UVB-induced cells, and the pro-inflammatory cytokines
demonstrated a similar trend, which indicated that
juglanin-reversed inflammatory response may be dependent on Nrf2
activity.
 | Figure 9Juglanin reduces the inflammatory
response by suppressing NF-κB associated with Nrf2 activation. (A)
Inflammatory cytokines, including COX2, IL-1β and TNF-α, were
calculated to investigate their gene expression levels using
reverse transcription-quantitative polymerase chain reaction
analysis. (B) COX2, IL-1β and TNF-α protein expression levels in
cells treated under various conditions were assessed via western
blotting. (C) The quantification of COX2, IL-1β and TNF-α is
presented. (D) Western blotting was performed to evaluate NF-κB
phosphorylation in HaCaT cells. (E) The HaCaT cells were pretreated
with juglanin in the presence or absence of Nrf2 siRNA for 12 h,
and then exposed to UVB for 24 h, followed by juglanin
administration for another 12 h. Then, the NF-κB phosphorylation,
COX2, IL-1β and TNF-α protein expression levels were calculated by
western blot analysis. Data are presented as the mean ± standard
error of the mean (n=8). ***P<0.001 vs. the Con
group. +P<0.05, ++P<0.01 and
+++P<0.001 vs. the UVB group. NF-κB, nuclear
factor-κB; Nrf2, nuclear factor-E2-related factor 2; COX2, cyclic
oxidase 2; IL-1β, interleukin-1β; TNF-α, tumor necrosis factor-α;
p, phosphorylated; NF-κB, nuclear factor-κB; Con, control; UVB,
ultraviolet B. |
The results indicated that similar to the treatment
with each inhibitor, juglanin significantly (P<0.05, P<0.01
and P<0.001 compared with the UVB group) reduced UVB-induced
p38, ERK1/2 and JNK phosphorylation in comparison to the UVB group.
Notably, the combination of juglanin with each inhibitor further
suppressed the activation of p38, ERK1/2 and JNK (Fig. 10A-C). In addition, administration
of NF-κB inhibitor, JSH-23, and juglanin reduced the levels of
phosphorylated NF-κB. Though no significant difference was observed
between the JSH-23- and juglanin-alone treatment and the two
combinations, the downregulated expression levels were observed
(Fig. 10D). Subsequently,
pro-inflammatory cytokines were observed to be reduced by JSH-23-
and juglanin-alone treatment, and the two as a combined treatment
further impeded COX2, IL-1β and TNF-α expression levels, as
demonstrated by RT-qPCR analysis (Fig. 10E). These data indicated that
juglanin functioned as the inhibitors of MAPKs and NF-κB, thus
attenuating UVB-induced skin injury.
 | Figure 10Juglanin functions as the inhibitors
of MAPKs and NF-κB (p65) to attenuate UVB-induced skin injury in
vitro. HaCaT cells were pre-treated with (A) SB203580 (50 nM),
(B) PD98059 (10 nM) and (C) SP600125 (50 µM) for 1 h or/and
with juglanin exposure for 12 h, and then exposed to 15
mJ/cm2 of UVB irradiation for 24 h, followed by
incubation for another 12 h in the absence or presence of juglanin.
Phosphorylated p38, ERK1/2 and JNK were calculated using western
blot analysis. (D) The cells were pre-treated with NF-κB (p65)
inhibitor, JSH-23 (8 µM), for 1 h or/and with juglanin
exposure for 12 h, and then exposed to 15 mJ/cm2 of UVB
irradiation for 24 h, followed by incubation for another 12 h in
the absence or presence of juglanin (160 µM). Phosphorylated
p38 was calculated using western blot analysis. ERK1/2 activation
was measured using western blot analysis. (E) The cells after
treatment as indicated were harvested for RT-qPCR analysis of COX2,
IL-1β and TNF-α. Data are represented as the mean ± SEM, n=8.
*P<0.05, and ***P<0.001. p,
phosphorylated; ERK, extracellular signal-regulated kinase; JNK,
c-Jun N-terminal kinases; NF-κB, nuclear factor-κB; UVB,
ultraviolet B. |
Discussion
Acute or chronic exposure to UV light, particularly
UVB, results in skin damage, which may progress to skin cancer
(1-3,35).
Annually, there are increasing numbers of newly diagnosed cases of
skin cancer reported, and UV exposure is considered to be a major
factor leading to skin cancer (36). Given the increased incidence,
morbidity, and the cost of the disease, identifying safe,
chemo-preventive compounds to protect people from UV light damage
has attracted attention (37).
Natural products are widely found to inhibit various types of
cancer or disease progression to reduce the morbidity or incidence
(38,39). Recently, studies have been
conducted to investigate the role of juglanin in different types of
disease by regulating inflammatory response and ROS generation
(21,22,40). Inhibiting inflammation and
scavenging ROS generation have been applied in various types of
human chronic disease (20). In
the present study, H&E staining demonstrated that juglanin
ameliorated UVB-induced skin damage, as reduced hyperplasia and
cell infiltration were observed in the dermis of the mice, directly
indicating the potential value of juglanin in preventing skin
injury. Tissue MPO activity is an indicator of the neutrophil
infiltration at sites where inflammation is present (41). The current data indicated that
juglanin administration to mice with UVB-irradiation caused a
decrease in MPO activities, further confirming the effects of
juglanin on inflammation inhibition.
The skin exposure to UVB has a close association
with oxidative stress, which is associated with anti-oxidants and
oxidants occurring in tissues (42). Certain major anti-oxidant enzymes,
including CAT, GPx and SOD, as well as the limiting enzyme, GSH,
have been reported to be depleted in UVB-induced skin injury, which
contribute to the abnormal homeostasis of oxygen radicals in skin
tissues (43). Free radicals or
other harmful species formed upon UV irradiation exert important
effects on diminishing anti-oxidants as previously described
(44). Additionally,
anti-oxidative enzymes are crucial in DNA damage in UVB-irradiated
blood lymphocytes (45). Thus,
restoring anti-oxidants is a potential target for drug
investigation to suppress UVB-induced tissue injury (46). In the present study, UVB-exposed
mice exhibited decreased levels of SOD, CAT, GSH and GPx, while MDA
levels increased in the skin of mice, which was consistent with
previous studies. Notably, the reduced intracellular ROS generation
was observed following juglanin treatment in in vitro
studies, demonstrated by DHE analysis. Studies have reported that
the MAPK family is an essential signaling cascade involved in
signal transduction from the membrane to nucleus, influencing ROS
generation (47,48). p38, ERK1/2 and JNK, members of the
MAPK family, have been reported as key in regulating cell
proliferation (49,50). ROS generation in the present study
was further supported by p38, ERK1/2, and JNK phosphorylation in
the skin tissue of mice following UVB irradiation, which was
ameliorated by juglanin administration, which likely reduced
oxidative stress.
The induction of pro-inflammatory cytokines,
including TNF-α, IL-1β, COX-2 and iNOS has been illustrated in
pre-cancerous and malignant lesions (51). UV induction of COX2 may result in
even the earliest stages of skin injury and inflammation (52). Thus, the suppression of COX2
expression may prevent the skin damage, even cancer development.
Furthermore, UVB exposure induces the expression levels of
pro-inflammatory cytokines, IL-1β and TNF-α in the skin of mice
(53). Overexpression of TGF-β1
and iNOS in the mice skin following UVB irradiation is also
reported, which enhances skin cancer progression, metastasis as
well as epithelial-to-mesenchymal transition at later stages
(54). NF-κB has been reported in
skin damage, whose sustained activation has been elucidated in
numerous types of tumor and was involved in various stages of
carcinogenesis (55). NF-κB
phosphorylation is crucial for pro-inflammatory cytokine release
(16,56). In the present study, UVB
irradiation induced Overexpression of COX2, TNF-α, IL-1β, TGF-β1
and iNOS, in line with NF-κB activity. Notably, juglanin reduced
the levels of pro-inflammatory cytokine expression, which is
associated with NF-κB de-phosphorylation.
Recently, the photo-chemopreventive effects of the
constitutive genetic activation of Nrf2 have been reported in
photo-carcinogenesis animal models, and activation of Nrf2 has been
considered as a novel and effective molecular strategy for
cutaneous photo-protection (57,58). Furthermore, Nrf2 activation may
protect fibroblasts against the cytotoxic effects of UVB (59). Nrf2, as the major effector of ROS
in the cell, regulates numerous cellular processes (32,33). Nrf2 knockout enhances oxidative
stress, and extensive ROS generation suppresses Nrf2 activity,
which influences the downstream signaling pathways, including MAPKs
and NF-κB (34,60). To further investigate the
association between Nrf2 activity, and MAPKs and NF-κB pathways
regulated by juglanin, Nrf2 was silenced using specific siRNA
sequence in cells in vitro. The data illustrated that Nrf2
knockdown diminished the role of juglanin in reducing UVB-induced
MAPKs and NF-κB phosphorylation, which indicated that
juglanin-ameliorated skin damage induced by UVB was likely to be
dependent on Nrf2 activity, contributing to MAPKs and NF-κB
inactivation (Fig. 11).
 | Figure 11Schematic image of the effects of
juglanin on UVB-induced skin damage. Following UVB irradiation, ROS
was produced in the skin of hairless mice, contributing to MAPK
activation and inflammatory response by activating NF-κB, leading
to eventual skin damage. The process may be ameliorated by
treatment with juglanin, which is dependent on Nrf2 activity. UVB,
ultraviolet B; ROS, reactive oxygen species; MAPK, mitogen
activated protein kinase; NF-κB, nuclear factor-κB; Nrf2, nuclear
factor-E2-related factor 2; COX2, cyclic oxidase 2; IL-1β,
interleukin-1β; TNF-α, tumor necrosis factor-α; ERK, extracellular
signal-regulated kinase; JNK, c-Jun N-terminal kinases. |
In conclusion, UVB resulted in elevated ROS
generation, leading to activation of MAPKs and NF-κB and promoting
the pro-inflammatory cytokine expression, which may contribute to
inflammation in mouse skin. In addition, juglanin may suppress ROS
production via improving Nrf2 activity, thus ameliorating
UVB-induced skin damage. However, further study is still necessary
in order to comprehensively reveal the underlying molecular
mechanism by which juglanin performs its role against skin injury,
in addition to test its safety for further potential uses.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
YHK and SPX performed the experiments and wrote the
manuscript. SPX planned the study, wrote the manuscript and
organized the experiments.
Ethics approval and consent to
participate
All animal experiments were conducted according to
the Guide for the Care and Use of Laboratory Animals, issued by the
National Institutes of Health in 1996 and approved by the
Institutional Animal Ethics Committee for the Guide for the Care
and Use of Laboratory Animals of the Affiliated Huaian No. 1
People's Hospital of Nanjing Medical University (Huai'an,
China).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgments
The authors would like to thank the members of the
Affiliated Huaian No. 1 People's Hospital of Nanjing Medical
University for their guidance.
References
|
1
|
Avila Acevedo JG, Espinosa González AM, De
Maria y Campos DM, Benitez Flores Jdel C, Hernández Delgado T,
Flores Maya S, Campos Contreras J, Muñoz López JL and García Bores
AM: Photoprotection of Buddleja cordata extract against UVB-induced
skin damage in SKH-1 hairless mice. BMC Complement Altern Med.
14:2812014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Meeran SM, Akhtar S and Katiyar SK:
Inhibition of UVB-induced skin tumor development by drinking green
tea polyphenols is mediated through DNA repair and subsequent
inhibition of inflammation. J Invest Dermatol. 129:1258–1270. 2009.
View Article : Google Scholar :
|
|
3
|
Katiyar SK, Matsui MS and Mukhtar H:
Kinetics of UV light-induced cyclobutane pyrimidine dimers in human
skin in vivo: An immunohistochemical analysis of both epidermis and
dermis. Photochem Photobiol. 72:788–793. 2000. View Article : Google Scholar
|
|
4
|
Kim JE, Song D, Kim J, Choi J, Kim JR,
Yoon HS, Bae JS, Han M, Lee S, Hong JS, et al: Oral supplementation
with cocoa extract reduces UVB-induced wrinkles in hairless mouse
skin. J Invest Dermatol. 136:1012–1021. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Hwang E, Park SY, Lee HJ, Lee TY, Sun ZW
and Yi TH: Gallic acid regulates skin photoaging in UVB-exposed
fibroblast and hairless mice. Phytother Res. 28:1778–1788. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Vayalil PK, Mittal A, Hara Y, Elmets CA
and Katiyar SK: Green tea polyphenols prevent ultraviolet
light-induced oxidative damage and matrix metalloproteinases
expression in mouse skin. J Invest Dermatol. 122:1480–1487. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hong YF, Lee HY, Jung BJ, Jang S, Chung DK
and Kim H: Lipoteichoic acid isolated from Lactobacillus plantarum
down-regulates UV-induced MMP-1 expression and up-regulates type I
procollagen through the inhibition of reactive oxygen species
generation. Mol Immunol. 67:248–255. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Thomas-Ahner JM, Wulff BC, Tober KL,
Kusewitt DF, Riggenbach JA and Oberyszyn TM: Gender differences in
UVB-induced skin carcinogenesis, inflammation, and DNA damage.
Cancer Res. 67:3468–3474. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sullivan NJ, Tober KL, Burns EM, Schick
JS, Riggenbach JA, Mace TA, Bill MA, Young GS, Oberyszyn TM and
Lesinski GB: UV light B-mediated inhibition of skin catalase
activity promotes Gr-1+ CD11b+ myeloid cell
expansion. J Invest Dermatol. 132:695–702. 2012. View Article : Google Scholar
|
|
10
|
Zeng SS, Yamashita T, Kondo M, Nio K,
Hayashi T, Hara Y, Nomura Y, Yoshida M, Hayashi T, Oishi N, et al:
The transcription factor SALL4 regulates stemness of EpCAM-positive
hepatocellular carcinoma. J Hepatol. 60:127–134. 2014. View Article : Google Scholar
|
|
11
|
Cho JL, Allanson M, Domanski D, Arun SJ
and Reeve VE: Estrogen receptor-beta signaling protects epidermal
cytokine expression and immune function from UVB-induced impairment
in mice. Photoch Photobiolo Sci. 7:120–125. 2008. View Article : Google Scholar
|
|
12
|
Yasui H, Hakozaki T, Date A, Yoshii T and
Sakurai H: Real-time chemiluminescent imaging and detection of
reactive oxygen species generated in the UVB-exposed human skin
equivalent model. Biochem Biophys Res Commun. 347:83–88. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yoon WJ, Ham YM, Lee WJ, Lee NH and Hyun
CG: Brown alga Sargassum muticum inhibits proinflammatory
cytokines, iNOS and COX-2 expression in macrophage RAW 264.7 cells.
Turk J Biol. 34:25–34. 2010.
|
|
14
|
Singh T, Prasad R and Katiyar SK: Regular
intake of high-fat diet exacerbates oxidative stress-mediated DNA
hypermethylation and activation of Nrf2-Keap1 signaling in
UVB-induced skin tumors in SKH-1 hairless mice. Cancer Res.
76:43102016.
|
|
15
|
Saw CLL, Yang AY, Huang MT, Liu Y, Lee JH,
Khor TO, Su ZY, Shu L, Lu Y, Conney AH and Kong AN: Nrf2 null
enhances UVB-induced skin inflammation and extracellular matrix
damages. Cell Biosci. 4:392014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Buckman SY, Gresham A, Hale P, Hruza G,
Anast J, Masferrer J and Pentland AP: COX-2 expression is induced
by UVB exposure in human skin: Implications for the development of
skin cancer. Carcinogenesis. 19:723–729. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Muthusamy V and Piva TJ: The UV response
of the skin: A review of the MAPK, NFkappaB and TNFalpha signal
transduction pathways. Arch Dermatol Res. 302:5–17. 2010.
View Article : Google Scholar
|
|
18
|
Asgari MM, Chren MM, Warton EM, Friedman
GD and White E: Association between nonsteroidal anti-inflammatory
drug use and cutaneous squamous cell carcinoma. Arch Dermatol.
146:388–395. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Harris RE, Beebe-Donk J, Doss H and Burr
Doss D: Aspirin, ibuprofen, and other non-steroidal
anti-inflammatory drugs in cancer prevention: A critical review of
non-selective COX-2 blockade (review). Oncol Rep. 13:559–583.
2005.PubMed/NCBI
|
|
20
|
Asgari M, White E and Chren MM:
Nonsteroidal anti-inflammatory drug use in the prevention and
treatment of squamous cell carcinoma. Dermatol Surg. 30:1335–1342.
2004.PubMed/NCBI
|
|
21
|
Sun ZL, Dong JL and Wu J: Juglanin induces
apoptosis and autophagy in human breast cancer progression via
ROS/JNK promotion. Biomed Pharmacother. 85:303–312. 2017.
View Article : Google Scholar
|
|
22
|
Yang HH, Hwangbo K, Zheng MS, Son JK, Kim
HY, Baek SH, Choi HC, Park SY and Kim JR: Inhibitory effects of
juglanin on cellular senescence in human dermal fibroblasts. J Nat
Med. 68:473–480. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhou GY, Yi YX, Jin LX, Lin W, Fang PP,
Lin XZ, Zheng Y and Pan CW: The protective effect of juglanin on
fructose-induced hepatitis by inhibiting inflammation and apoptosis
through TLR4 and JAK2/STAT3 signaling pathways in fructose-fed
rats. Biomed Pharmacother. 81:318–328. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
25
|
Nicholls SJ and Hazen SL: The role of
myeloperoxidase in the pathogenesis of coronary atery disease. Jpn
J Infect Dis. 57:S21–S22. 2004.PubMed/NCBI
|
|
26
|
Hara Y, Hirao T and Iwai I: Facial
expression under stiff stratum corneum leads to strain
concentrations, followed by residual wrinkle formation. Int J
Cosmetic Sci. 39:66–71. 2017. View Article : Google Scholar
|
|
27
|
Calò R and Marabini L: Protective effect
of Vaccinium myrtillus extract against UVA- and UVB-induced damage
in a human keratinocyte cell line (HaCaT cells). J Photoch Photobio
B. 132:27–35. 2014. View Article : Google Scholar
|
|
28
|
Huang D, Ding Y, Luo WM, Bender S, Qian
CN, Kort E, Zhang ZF, VandenBeldt K, Duesbery NS, Resau JH and Teh
BT: Inhibition of MAPK kinase signaling pathways suppressed renal
cell carcinoma growth and angiogenesis in vivo. Cancer Res.
68:81–88. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Liu J, Chang F, Li F, Fu H, Wang J, Zhang
S, Zhao J and Yin D: Palmitate promotes autophagy and apoptosis
through ROS-dependent JNK and p38 MAPK. Biochem Biophys Res Commun.
463:262–267. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Fan SH, Zhang ZF, Zheng YL, Lu J, Wu DM,
Shan Q, Hu B and Wang YY: Troxerutin protects the mouse kidney from
D-galactose-caused injury through anti-inflammation and
anti-oxidation. Int Immunopharmacol. 9:91–96. 2009. View Article : Google Scholar
|
|
31
|
Ananthi S, Raghavendran HR, Sunil AG,
Gayathri V, Ramakrishnan G and Vasanthi HR: In vitro antioxidant
and in vivo anti-inflammatory potential of crude polysaccharide
from Turbinaria ornata (Marine Brown Alga). Food Chem Toxicol.
48:187–192. 2010. View Article : Google Scholar
|
|
32
|
Seo SH and Jeong GS: Fisetin inhibits
TNF-α-induced inflammatory action and hydrogen peroxide-induced
oxidative damage in human keratinocyte HaCaT cells through
PI3K/AKT/Nrf-2-mediated heme oxygenase-1 expression. Int
Immunopharmacol. 29:246–253. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kovac S, Angelova PR, Holmström KM, Zhang
Y, Dinkova-Kostova AT and Abramov AY: Nrf2 regulates ROS production
by mitochondria and NADPH oxidase. Biochim Biophys Acta.
1850:794–801. 2015. View Article : Google Scholar :
|
|
34
|
Wang P, Peng X, Wei ZF, Wei FY, Wang W, Ma
WD, Yao LP, Fu YJ and Zu YG: Geraniin exerts cytoprotective effect
against cellular oxidative stress by upregulation of Nrf2-mediated
anti-oxidant enzyme expression via PI3K/AKT and ERK1/2 pathway.
Biochim Biophys Acta. 1850:1751–1761. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Muller HK and Woods GM: Ultraviolet
radiation effects on the proteome of skin cells. Adv Exp Med Biol.
990:111–119. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Schneider MR: MicroRNAs as novel players
in skin development, homeostasis and disease. Br J Dermatol.
166:22–28. 2012. View Article : Google Scholar
|
|
37
|
Vaid M, Sharma SD and Katiyar SK:
Honokiol, a phytochemical from the Magnolia plant, inhibits
photocarcinogenesis by targeting UVB-induced inflammatory mediators
and cell cycle regulators: Development of topical formulation.
Carcinogenesis. 31:2004–2011. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Shehzad A, Lee J, Huh TL and Lee YS:
Curcumin induces apoptosis in human colorectal carcinoma (HCT-15)
cells by regulating expression of Prp4 and p53. Mol Cells.
35:526–532. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Ying TH, Yang SF, Tsai SJ, Hsieh SC, Huang
YC, Bau DT and Hsieh YH: Fisetin induces apoptosis in human
cervical cancer HeLa cells through ERK1/2-mediated activation of
caspase-8-/caspase-3-dependent pathway. Arch Toxicol. 86:263–273.
2012. View Article : Google Scholar
|
|
40
|
Paul MK, Bisht B, Darmawan DO, Chiou R, Ha
VL, Wallace WD, Chon AT, Hegab AE, Grogan T, Elashoff DA, et al:
Dynamic changes in intracellular ROS levels regulate airway basal
stem cell homeostasis through Nrf2-dependent Notch signaling. Cell
Stem Cell. 15:199–214. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Mäkelä R, Loimaala A, Nenonen A, Mercuri
M, Vuori I, Huhtala H, Oja P, Bond G, Koivula T and Lehtimäki T:
The association of myeloperoxidase promoter polymorphism with
carotid atherosclerosis is abolished in patients with type 2
diabetes. Clin Biochem. 41:532–537. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Bickers DR and Athar M: Oxidative stress
in the pathogenesis of skin disease. J Invest Dermatol.
126:2565–2575. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Braga PC, Marabini L, Wang YY, Lattuada N,
Calò R, Bertelli A, Falchi M, Dal Sasso M and Bianchi T:
Characterisation of the antioxidant effects of Aesculus
hippocastanum L. bark extract on the basis of radical scavenging
activity, the chemiluminescence of human neutrophil bursts and
lipoperoxidation assay. Eur Rev Med Pharmacol Sci. 16(Suppl 3):
S1–S9. 2012.
|
|
44
|
Acker T, Fandrey J and Acker H: The good,
the bad and the ugly in oxygen-sensing: ROS, cytochromes and
prolyl-hydroxylases. Cardiovasc Res. 71:195–207. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Ristow M: Unraveling the truth about
antioxidants: Mitohormesis explains ROS-induced health benefits.
Nat Med. 20:709–711. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Ki YW, Park JH, Lee JE, Shin IC and Koh
HC: JNK and p38 MAPK regulate oxidative stress and the inflammatory
response in chlorpyrifos-induced apoptosis. Toxicol Lett.
218:235–245. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Park EJ, Park SW, Kim HJ, Kwak JH, Lee DU
and Chang KC: Dehydrocostuslactone inhibits LPS-induced
inflammation by p38MAPK-dependent induction of hemeoxygenase-1 in
vitro and improves survival of mice in CLP-induced sepsis in vivo.
Int Immunopharmacol. 22:332–340. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Brzezianska E and Pastuszak-Lewandoska D:
A minireview: The role of MAPK/ERK and PI3K/Akt pathways in thyroid
follicular cell-derived neoplasm. Fronti Biosci (Landmark Ed).
16:422–439. 2011. View
Article : Google Scholar
|
|
49
|
Hao W, Yuan X, Yu L, Gao C, Sun X, Wang D
and Zheng Q: Licochalcone A-induced human gastric cancer BGC-823
cells apoptosis by regulating ROS-mediated MAPKs and PI3K/AKT
signaling pathways. Sci Rep. 5:103362015. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Mi Y, Xiao C, Du Q, Wu W, Qi G and Liu X:
Momordin Ic couples apoptosis with autophagy in human
hepatoblastoma cancer cells by reactive oxygen species
(ROS)-mediated PI3K/Akt and MAPK signaling pathways. Free Radic
Biol Med. 90:230–242. 2016. View Article : Google Scholar
|
|
51
|
Damiani E and Ullrich SE: Understanding
the connection between platelet-activating factor, a UV-induced
lipid mediator of inflammation, immune suppression and skin cancer.
Prog Lipid Res. 63:14–27. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Gupta SC, Patchva S and Aggarwal BB:
Therapeutic roles of curcumin: Lessons learned from clinical
trials. AAPS J. 15:195–218. 2013. View Article : Google Scholar :
|
|
53
|
Aggarwal BB, Gupta SC and Sung B:
Curcumin: An orally bioavailable blocker of TNF and other
pro-inflammatory biomarkers. Brit J Pharmacol. 16:1672–1692. 2013.
View Article : Google Scholar
|
|
54
|
Abbas S, Alam S, Pal A, Kumar M, Singh D
and Ansari KM: UVB exposure enhanced benzanthrone-induced
inflammatory responses in SKH-1 mouse skin by activating the
expression of COX-2 and iNOS through MAP kinases/NF-κB/AP-1
signalling pathways. Food Chem Toxicol. 96:183–190. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Hoesel B and Schmid JA: The complexity of
NF-κB signaling in inflammation and cancer. Mol Cancer. 12:862013.
View Article : Google Scholar
|
|
56
|
Burris RL, Ng HP and Nagarajan S: Soy
protein inhibits inflammation-induced VCAM-1 and inflammatory
cytokine induction by inhibiting the NF-κB and AKT signaling
pathway in apolipoprotein E-deficient mice. Eur J Nutr. 53:135–148.
2014. View Article : Google Scholar
|
|
57
|
Ramyaa P, Krishnaswamy R and Padma VV:
Quercetin modulates OTA-induced oxidative stress and redox
signalling in HepG2 cells-upregulation of Nrf2 expression and
downregulation of NF-κB and COX-2. Biochim Biophys Acta.
1840:681–692. 2014. View Article : Google Scholar
|
|
58
|
Agca CA, Tuzcu M, Hayirli A and Sahin K:
Taurine ameliorates neuropathy via regulating NF-κB and Nrf2/HO-1
signaling cascades in diabetic rats. Food Chem Toxicol. 71:116–121.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Poulose SM, Bielinski DF, Carey A, Schauss
AG and Shukitt-Hale B: Modulation of oxidative stress,
inflammation, autophagy and expression of Nrf2 in hippocampus and
frontal cortex of rats fed with açaí-enriched diets. Nutr Neurosci.
20:305–315. 2017. View Article : Google Scholar
|
|
60
|
Paredes-Gonzalez X, Fuentes F, Jeffery S,
Saw CL, Shu L, Su ZY and Kong AN: Induction of NRF2-mediated gene
expression by dietary phytochemical flavones apigenin and luteolin.
Biopharm Drug Dispos. 36:440–451. 2015. View Article : Google Scholar : PubMed/NCBI
|

























