Introduction
The immunological mechanisms controlling human
papillomavirus (HPV) infection are poorly understood. Initial
infection of basal keratinocytes does not appear to activate a
local immune response and HPV-associated skin lesions can persist
for months or even years in immunocompetent subjects (1). Successful viral evasion from immune
controlling mechanisms is likely the result of a number of factors
including the low level of viral protein expression, nuclear
localization of viral proteins in the basal layer of infected
epithelium and/or the absence of cytolysis or cytopathic death
resulting from viral replication and assembly (2). Accordingly, there is little or no
release of the proinflammatory cytokines necessary for activation
and migration of antigen-presenting cells. In addition, because HPV
does not undergo a systemic or viremic phase of infection, the
opportunities for immune cells to capture virus and present viral
antigens in the lymph nodes are limited (3).
Previous studies have described a localized immune
dysfunction accompanying cervical HPV infection. This dysfunction
is characterized by quantitative and qualitative alterations of
CD4+ T lymphocytes in the area of infection. (4,5) In
addition, it has been shown that the number and distribution of
Langerhans cells (LCs) are abnormal in HPV-infected preneoplastic
and neoplastic cervical epithelium (6–10).
LCs are included in the lineage of immature dendritic cells (DCs)
(11) and are considered to be the
most important professional antigen-presenting cells in the mucosal
immune system (12,13).
Several paradigms have been proposed to explain LC
depletion in the area of an HPV infection. For example, LC
depletion might result from the creation of a locally unfavorable
environment fostered by infected epithelial cells and fibroblasts.
HPV-infected keratinocytes are known to express low levels of major
histocompatibility complex restriction elements and do not present
the necessary levels of costimulatory molecules for naive T-cell
priming (14). Additionally,
production of granulocyte-macrophage colony stimulating factor
(GM-CSF) (15) as well as type I
interferon (16,17) is significantly decreased in
HPV-infected keratinocytes. Such an immunosuppressive environment
might lead to a decrease in intraepithelial DC maturation and
prevent migration of DCs and macrophages to infected tissue. In
addition, HPV may be capable of non-productively infecting DCs and
producing a direct cytotoxic effect (7,9).
Several lines of indirect evidence support this possibility. For
example, DCs are located in the basal and middle third of the
squamous epithelium and commonly project numerous cytoplasmic
processes into more superficial cell layers (9). Therefore, the proximity of DCs to
basal epithelial cells, which are considered to be the primary
targets for HPV infection, could make DCs accessible for infection.
Previous studies have shown that DCs express the α6 integrin
receptor, which is associated with viral entry (18,19).
Additionally, human monocytes and macrophages are known to be
susceptible to bovine papillomavirus infection in vitro
(20).
Spontaneous regression of flat warts is believed to
be associated with activation of a cellular immune response against
HPV-infected keratinocytes (21).
Initiation of this response depends upon HPV antigen presentation
by DCs. The contribution of HPV-infected basal keratinocytes as
non-professional antigen-presenting cells is most likely
insignificant in light of their low-level expression of E6 and E7
as well as their lack of the receptors necessary to mount an
effective immune response (22,23).
Therefore, it is possible that professional antigen presenting
cells such as LCs and DCs transport HPV antigens from the skin or
mucosal tissues to draining lymph nodes where the antigens are
presented to specific naive T cells (11). This view is supported by studies
in vitro, which show that DCs are capable of initiating an
immune response after priming with viral proteins E6 and E7
(24). However, if this occurs, it
most likely happens at late stages of infection in that
seroconversion associated with high-risk forms of HPV typically
takes nine months on average from the first detection of HPV DNA in
a cervical biopsy (25).
Taken together, published studies indicate that DC
activation is a crucial factor in anti-HPV protection. Lack of an
in vitro system for productive infection of DCs with HPVs,
prompted us to examined human DCs transfected with constructs
containing the HPV genome. Our data suggest that HPV transfection
of DCs leads to changes in DC migratory activity and cytokine
production, which potentially may suppress or delay an immune
response to viral antigens. Additionally, changes in cytokine
production by HPV-transformed human foreskin fibroblasts (HFFs) and
human cervical epithelial cells reveals that DC migratory and
antigen-presenting functions may be impaired by the suppressive
effects of cytokines produced by HPV-infected epithelial and
stromal cells.
Materials and methods
Cell lines and media
The DC line RAN1 was previously generated from cells
obtained from a patient with histiocytic lymphoma by using several
rounds of selection and limiting dilution in order to obtain
monoclonality (52). HFFs were
from Cellular Engineering Technologies, Coralville, IA, USA; the
HPV18-infected C4-II cell line was obtained from ATCC
(ATCC® CRL 1595TM); and normal human cervical
cells epithelial cells (NHCEC) cells were originally purchased from
Bio-Whittaker (now Lonza, Inc., Allendale, NJ, USA). All cell
lines, except NHCEC cells, were grown in Dulbecco’s modified
Eagle’s medium (DMEM) supplemented with glutamine and 20% fetal
bovine serum (FBS, Atlanta Biologicals, Norcross, GA, USA). KGM-2
medium containing epidermal growth factor, insulin, hydrocortisone
and bovine pituitary extract (Lonza Inc.) was used to grow NHCEC
cells.
Plasmid and transfection
The GFP Fusion TOPO cloning vector (Life
Technologies, Carlsbad, CA, USA) was used to create a GFP-HPV18
plasmid containing the full length HPV18 genome. The HPV18 genome
was purchased from ATCC (ATCC 45152™). Originally linearized at the
unique EcoRI site and cloned into the pBR322 vector
(53), the HPV18 genome DNA was
digested with XmnI and NheI, purified and ligated
into the NheI-EcoRV-cut GFP Fusion TOPO cloning vector.
DCs or HFFs were seeded into 24-well plates
(2.0×104 cells per well) and grown to 75–80% confluency.
Transfection of cells was performed using Lipofectamine™ reagent
(Life Technologies). GFP-HPV18 plasmid DNA (2 μg) and
Lipofectamine reagent (10 μl) were mixed in serum-reduced
medium (Opti-MEM® medium, Life Technologies) and
incubated for 45 min at 21°C. Cell monolayers, washed once with
Opti-MEM medium, were overlaid with 1 ml of preformed lipid-DNA
complexes. After 5 h of incubation at 37°C in 5% CO2, 1
ml of culture medium containing 20% FBS was added. Culture medium
was refreshed 24 h after initiation of transfection. Cells were
harvested at 72 h post-transfection and grown in selection medium
containing G418 sulfate (Geneticin®, Life Technologies).
The appropriate concentration of selection antibiotic was
determined in prior experiments by titration of LD50 and varied
depending on the cell line (2.5 mg/ml for DCs and 250 μg/ml
for HHFs). The appearance of transfected colonies was detected by
light microscopy 5–7 days after initiating selection. HHFs and DCs
were transfected with GFP Fusion TOPO cloning plasmid to generate
stably transfected cell lines.
RT-PCR detection of E6 and E7
transcripts
Total RNA from C4-II cells and HPV18-transfected DCs
and HFFs was extracted using TRIzol® reagent (Life
Technologies). DNA contamination of RNA samples was prevented by
treatment with DNase (Life Technologies) according to the
manufacturer’s recommendation. cDNA was amplified using Super
Script kit (Life Technologies) according to the manufacturer’s
instructions. Ten microliters of cDNA was mixed with 4 μl
10X PCR buffer, 2 μl 25 mM MgCl2, 0.5 μl
(100 pmol) of each primer, 32.7 μl distilled water and 0.3
μl (2.5 U) Taq DNA polymerase. The reaction mixture was then
subjected to 35 cycles, each consisting of 30 sec at 94°C, 30 sec
at 50°C and 2 min at 72°C. The amplified products were sequenced to
confirm HPV18 DNA. Primers used were:
5′-GTGAGAAACACACCACAATACTATG-3′ forward and
5′-CTCTGCGTCGTTGGAGTCGTTC-3′ reverse, which amplify the E6 product
of 328 bp and 5′-CGGACACACAAAGGACAGGG-3′ forward and
5′-GACGCAGAGAAACACAAGTATAATA-3′ reverse which amplified the E7
products of 485- or 302-bp (spliced) fragments.
Immunoprecipitation and western blot
analysis
Expression of HPV18 proteins was evaluated by
immunoprecipitation and western blotting. HPV18-transfected and
non-transfected cells were collected after trypsinization, pelleted
(1,000 rpm, 5 min) and lysed by suspension in 200 μl of PBS
with 1% Triton X-100 and protease inhibitors (1:10; Complete Mini,
Roche, Indianapolis, IN, USA). Cell debris was removed by
centrifugation at 3,000 rpm for 5 min and supernatants were stored
at −70°C prior to use. Protein G Plus-Agarose (P-A, 100 μl;
Santa Cruz Biotechnology, Santa Cruz, CA, USA) was washed twice in
PBS (pH 7.2) and incubated with monoclonal anti-L1 HPV coat protein
antibody (Novocastra, Burlingame, CA, USA) or anti-E6-protein
monoclonal antibody (Calbiochem, La Jolla, CA, USA) for 2 h at room
temperature. Protein G Plus-Agarose-antibody complexes (P-AA) were
washed twice in PBS (pH 7.2) and incubated 12 h with clarified cell
supernatant at 4°C. P-AA complexes with bound antigen were
centrifuged briefly at 1,000 rpm for 5 min, washed in PBS (pH 7.2)
twice and incubated with 1% SDS loading buffer for 5 min at room
temperature. Agarose beads were removed by centrifugation and
samples were analyzed by SDS-PAGE electrophoresis. Proteins were
standardized using a modified Lowry protein assay (Pierce,
Rockford, IL, USA) and was verified by Coomassie staining of the
gel. Additionally, quantitative loading of proteins was determined
by probing the nitrocellulose membrane with anti-actin antibodies
(Santa Cruz Biotechnology). Proteins were electroblotted (15 min, 4
mA/cm2) onto nitrocellulose membranes (Bio-Rad,
Hercules, CA, USA) and blocked for 1 h at room temperature with 5%
milk in Tris-buffered saline (TBS) and 0.5% Tween-20. After three
washes, membranes were incubated with monoclonal anti-HPV L1
antibodies (1:50; Novocastra) or anti-E6-protein monoclonal
antibodies (1:25; Calbiochem) for 12 h at room temperature.
Antigen-antibody complexes were identified with HRP-conjugated goat
anti-mouse antibodies and developed using DAB (3,
3′-diaminobenzidine) substrate (Vector Laboratories, Inc.,
Burlingame, CA, USA).
Cytokine ELISA
Tumor necrosis factor-α (TNF-α) interleukin-6
(IL-6), transforming growth factor-β (TGF-β) and monocyte
chemotactic protein-1 (MCP-1) ELISA kits were purchased from Life
Technologies. The selection of cytokines for this study was based
upon published data suggesting their involvement in mechanisms
controlling tumor growth or regression (14,30,36–38).
Transfected and non-transfected cells (106 cells/well)
were placed in 12-well plates and grown to confluency and activated
with lipopolysaccharide (1 μg/ml; LPS; Sigma Chemical Co.,
St. Louis, MO, USA). Forty-eight hours after addition of LPS,
growth medium was collected and stored at −80°C until analyzed.
Southern blot analysis
An HPV18 32P-end-labeled DNA probe was
generated using a DNA labeling kit (Ready-To-Go, GE Healthcare Life
Sciences Inc., Piscataway, NJ, USA), 50 μCi
[α32P]-dCTP and ∼50 ng of HPV18 plasmid DNA. After a
15-min incubation at 37°C, MicroSpin G-50 columns (GE Healthcare
Life Sciences Inc.) were used to purify the labeled DNA. A DNA
isolation kit (Puregene® DNA, Qiagen, Valencia, CA, USA)
was used for extraction of DNA from HPV-transfected cell lines.
Approximately 10 μg of DNA was digested with EcoR1
restriction enzyme (New England Biolab; Beverly, MA, USA) and
resolved on a 1.0% agarose gel. Denatured DNA (0.25 N HCl for 10
min) was transferred by capillary to nylon membranes (ZetaProbe
Nylon, Bio-Rad). The membranes were incubated 14 h at 55°C with
labeled HPV18 DNA (5 ng) in 10 ml of hybridization buffer [1.5X
SSPE, 7% SDS, 10% (wt/vol) polyethylene glycol 4000]. The blots
were washed twice for 15 min each at room temperature with 2X
SSC-1% SDS and then twice with 0.1X SSC-1% SDS for 25 min each at
55°C. The blots were then exposed to X-ray film for 15 h at
−80°C.
Northern blot analysis
In order to recover total intracellular RNA, cells
were washed with PBS (pH 7.2) and lysed with TRIzol reagent
according to the manufacturer’s instructions. RNA (20–50 μg)
was denatured for 15 min at 70°C in RNA buffer [50% formamide, 5%
formaldehyde, 1X morpholinepropanesulfonic acid (MOPS)], separated
on a 3% formaldehyde-agarose gel and transferred to a nylon
membrane (ZetaProbe Nylon). Membranes were hybridized with
32P-labeled plasmid for 14 h at 60°C and washed as
described above. A 32P-labeled 18S ribosomal RNA probe
(Ambion, Austin, TX, USA) was used to standardize loading.
Colony formation assay
HPV18 transfected and nontransfected cells
(106/well in 12-well plates) were placed in
methylcellulose medium (3 ml; Life Technologies) containing 20% FBS
(Atlanta Biologicals) and L-glutamine (Life Technologies). After 14
days of culture, colonies were counted under light microscopy.
Chemotaxis assay
The in vitro migration of DCs was assayed in
24-well transwell cell culture chambers (54). DCs were washed and suspended in
DMEM, supplemented with 20% FBS. Filter inserts (8 μm) were
placed into the cell culture plate wells to create upper and lower
compartments. DCs (5×104) were applied into the upper
chamber, while the lower chamber was filled with 200 μl of
medium (DMEM + 20% FBS) containing 50 μg of GM-CSF. After 4
h of incubation (37°C, 5% CO2), cells in the upper side
of the filter were removed and filters were scraped to remove
residual cells. Filters were fixed (3:1 methanol/acetone mixture, 5
min) and cellular nuclei were stained with hematoxylin. Five random
microscopic fields were counted per filter to evaluate the number
of migrated cells.
Statistical analysis
Statistical significance was assessed by the
Student’s paired t-test and considered significant at
P<0.05.
Results
Characterization of HPV18-transfected
DCs
Because of difficulties in propagating
papillomaviruses and generating infectious virus in vitro,
we used DNA transfection to deliver viral DNA into DCs. One week
after transfection, colonies of DCs were isolated and subcultured
for an additional 2–3 passages. A stably transfected DC line, which
persistently maintained the HPV18 genome, was utilized for
experiments reported herein.
It has been reported that HPV DNA regularly becomes
integrated into host cell DNA (1).
Therefore, it was of interest to determine if our HPV18 DNA
construct is maintained as a plasmid or integrated into the genome.
Total DNA from transfected DCs was isolated as described in
Materials and methods. Southern blot analysis revealed integration
of HPV18 DNA into the DC genome; no extra-chromosomal plasmid could
be detected in DNA samples isolated from transfected cells
following cleavage with EcoRI (Fig. 1, lane 1). Transfection did not
induce differentiation of DCs to a more mature or macrophage-like
phenotype or change their growth pattern (data not shown).
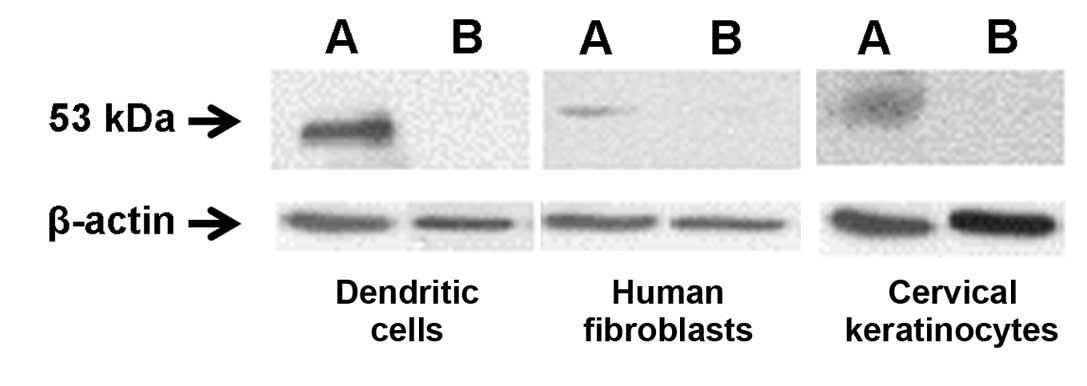
L1 is the major HPV capsid protein and is highly
conserved among all papillomaviruses. Since expression of
papilloma-virus L1 protein is important for development of an
anti-HPV immune response (26–29),
we sought to establish if HPV18-transfected cells express this
protein. Immunoprecipitation and western blot analysis established
that L1 protein was present only in extracts of transfected cells
(Fig. 2). To confirm these data,
we further analyzed extracts for the presence of L1 mRNA. Total RNA
from transfected and non-transfected cells was collected and
subjected to northern blot analysis. Consistent with our western
blot results, HPV L1 specific transcripts were detected only in
HPV18 transfected DCs, whereas no HPV RNA was found in
non-transfected DC control (Fig.
3).
It is well documented that HPV18 E6 and E7 proteins
are important in cell transformation; therefore, transcription of
E6 and E7 genes was analyzed by RT-PCR. Amplicons consistent with
the expected RT-PCR product of the E6 and E7 gene (∼302 and 485 bp
for E6 and ∼328 bp for E7) were detected in transfected DC cells
(Fig. 4A, lane 1 and B, lane 1).
Sequencing of the excised bands confirmed these results.
In view of the possibility that expression of viral
transcripts or proteins could influence immune functions, IL-6,
MCP-1, TGF-β and TNF-α expression was analyzed in transfected and
non-transfected DCs. Selection of cytokines was based upon previous
reports on their importance in HPV-associated cervical neoplasia
(30,31) or their in vitro expression
following DC exposure to HPV proteins (24,32).
To determine the effect of HPV18 transfection on cytokine
production, transfected DCs, non-transfected DCs and DCs
transfected with empty vector were cultured to confluency. As a
positive control for cytokine production, LPS (1 μg/ml) was
added to the medium of both transfected and non-transfected cell
lines. Transfection with the empty vector did not influence
cytokine production (data not shown). IL-6, TGF-β and TNF-α
production was significantly increased in supernatants of
HPV18-transfected DCs compared to non-transfected controls
(Fig. 5A–C). LPS stimulation
resulted in upregulation of IL-6 and TNF-α by both non-transfected
and transfected cells; however, the increases in IL-6 and TNF-α
production following LPS stimulation were greater in
HPV-transfected cells than in nontransfected controls. LPS
treatment of non-transfected DCs increased TGF-β production, as did
transfection with the HPV construct. However, LPS stimulation of
HPV18 transfected DCs did not cause further upregulation of TGF-β
compared to non-transfected cells. The concentration of MCP-1 was
significantly decreased in supernatants of HPV-transfected DCs
versus non-transfected cells (Fig.
5B).
Migration of DCs from the site of infection to the
regional lymph nodes is essential for induction of an effective
immune response; therefore, changes in the migratory function due
to HPV infection, may potentially impact the outcome of an anti-HPV
immune response. To determine if HPV18 transfection might influence
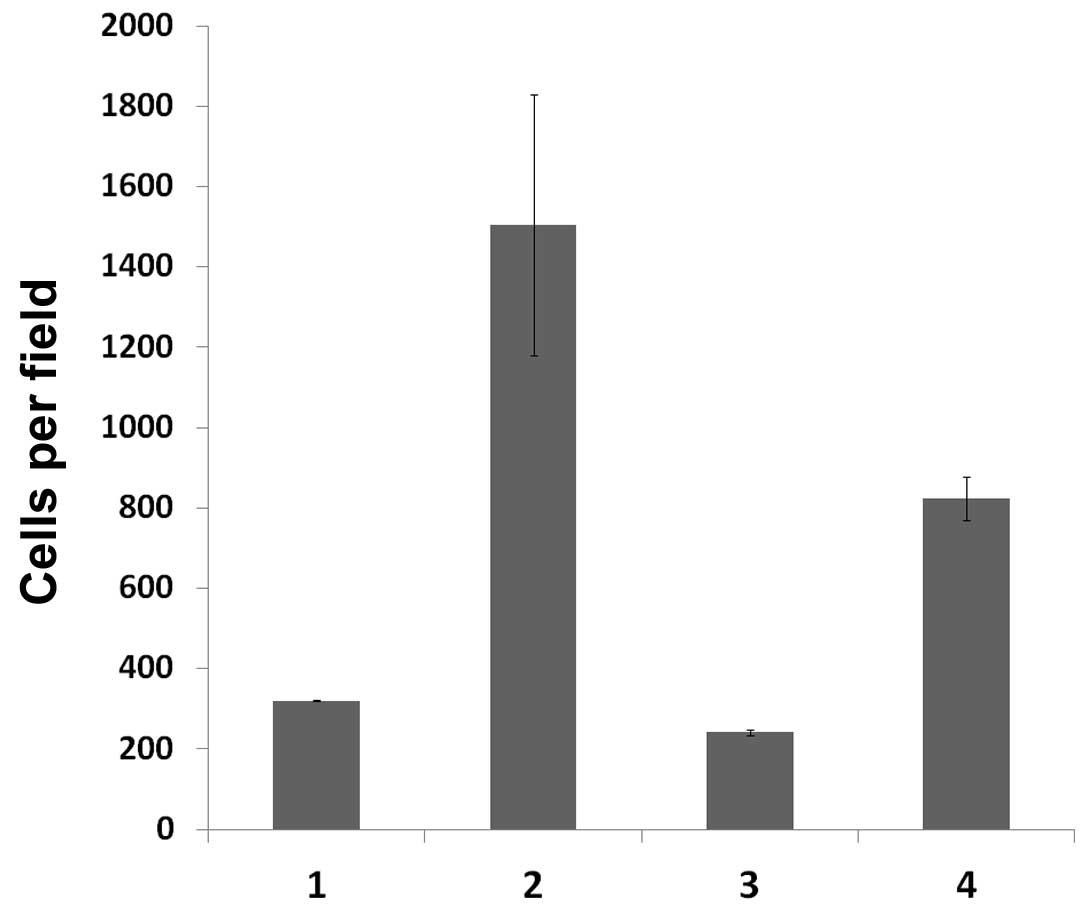
the migration of DCs, we conducted migration assays using a Boyden
microchemotaxis chamber. Chemotaxis was significantly enhanced by
addition of GM-CSF to the medium (Fig.
6). Spontaneous motility of DCs transfected with HPV18 was
significantly higher than that of non-transfected cells. However,
the chemotactic response of transfected DCs was significantly lower
than that of non-transfected cells, although it remained higher
than the response to non-conditioned medium. The presence of the
empty vector control did not affect DC migration (data not
shown).
Characterization of human foreskin
fibroblasts (HFF) transfected with HPV18 DNA and HPV18-infected
human cervical epithelial cells (C4-II)
HPV18 infects basal epithelial cells of the cervix,
therefore, cytokines produced by cervical epithelial cells and
fibroblasts may influence the function of local DCs. For this
reason, we sought to determine if HPV18 transformation promoted
cytokine production in human fibroblasts and cervical epithelial
cells. The HPV18-infected human cervical epithelial cell line
(C4-II) has been shown to maintain an expression profile that
accurately represents cervical cancer when compared to normal
cervix biopsies (33). Therefore,
this cell line was used to evaluate the effects of virus
transformation on cytokine expression and growth patterns of
cervical epithelial cells. Normal human cervical epithelial cells
(NHCEC) were used as a control.
Human fibroblasts were transfected with HPV-GFP
plasmid and cultured for selection of stably transfected cell
colonies. A transformed cell line (HPV18-HFF) that persistently
maintained the HPV18 genome was isolated and used in all further
experiments. Southern blot analysis revealed genomic integration of
viral DNA in transfected HFFs that was consistent with that
observed in HPV18-infected C4-II cells (Fig. 1, lane 2). Additionally, transfected
HFFs and HPV18-infected C4-II cells both expressed HPV18 L1 protein
(Fig. 2), as detected by
immunoprecipitation and western blotting.
In order to determine if transcriptional activation
of the E6 and E7 genes could be detected in transfected HFFs and
transformed C4-II cells, we again performed RT-PCR as described
above. Consistent with our observations for DCs, transcriptional
activation of the E6 and E7 genes was identified in HFFs and C4-II
cells (Fig. 4). Significantly,
HPV18 transfection resulted in transformation of HFFs as suggested
by the colony formation assay (Fig.
7). Transfected HFFs were able to support anchorage-independent
growth in soft agarose medium. In contrast, non-transfected HFFs
failed to produce colonies in soft agarose. It has been reported
that expression of E6 and E7 genes affect cell growth properties
such as saturation density and colony formation in soft agarose
(34,35), consistent with our
observations.
In view of our observation that HFFs and cervical
epithelial cells were able to support the expression of some HPV18
proteins, we sought to determine if transfection with the HPV18
construct would influence cytokine production. Culture media from
HPV18-transformed (HPV18-HFFs and C4-II) and non-transformed cells
(HFFs and NHCEC) was analyzed for cytokine production by ELISA.
HFFs transfected with empty vector were used as a negative control
in all experiments and LPS treatment was used as a positive
control. When compared to non-transfected cells, those transfected
with empty vector consistently showed no difference in cytokine
production (data not shown).
When compared to non-transformed cells, all
transformed cells showed statistically significant differences in
cytokine production (Fig. 5), with
the greatest difference being a 60-fold increase in IL-6 production
(Fig. 5E and I). Interestingly,
transformed keratinocytes were refractory in MCP-1 and TGF-β
production, even upon LPS stimulation, when compared to their
non-transformed counterparts (Fig. 5J
and K). Additionally, upon LPS stimulation, transformed
fibroblasts displayed decreased TNF-α production when compared to
non-transformed cells (Fig.
5H).
Discussion
In order to investigate the possible consequence of
HPV infection on DC functions we used transfection as a form of
viral genome delivery. Transfection of DCs and expression of viral
proteins did not result in cell death; conversely, we were able to
develop several lines of DCs containing the complete HPV18 genome.
Expression of L1 major capsid protein was revealed in western blot
analyses, as were L1 transcripts by Southern blotting. Although
evidence of E6 or E7 protein production was lacking in transfected
DCs, all transfected cell lines expressed HPV18 E6 and E7
transcripts, suggesting possible low-level protein expression below
the sensitivity of our detection method. Expression of the L1
protein by DCs may be important for stimulation of an antibody
response against HPV. Indeed, Steele and Gallimore (28) observed the production of specific
antibodies during the course of HPV infection, the majority of
which were antiviral capsid proteins. They further reported that as
papillomas progressed to carcinomas, there was a marked increase in
the response to L1 and L2 suggesting a change in interaction
between virus-infected host cells and the host’s immune system
(26,27). However, it is believed that
antibodies to capsid proteins are not sufficient to totally
suppress infection (30) because
of the limitation of HPV capsid protein expression to superficial
epithelial cells, which do not represent the site of virus
replication (3).
In our experiments, HPV18 transfection of DCs
resulted in increased production of cytokines involved in DC
migration. This phenomenon was accompanied by increased spontaneous
migration of DCs after HPV18 transfection compared to
nontransfected cells, suggesting that HPV18 transfection could
activate cellular mechanisms controlling DC migration. It has been
shown that TNF-α and IL-6 contribute to the migration of DCs
through the dermis in vivo and in vitro (36–38).
Constitutive and inducible expression of IL-6 by DCs could
facilitate the induction of cutaneous immune responses (36). Importantly, DCs are reported to be
a significant source of IL-6 in lymph nodes during the primary
immune responses to cutaneous antigens (39). Our data show that HPV18
transfection increases production of TNF-α and IL-6 by DCs. This
result suggests that HPV18-transfected DCs are potentially capable
of inducing an immune response to viral proteins as a result of
increased migratory activity and upregulation of cytokines involved
in antigen priming of T cells in lymph nodes. Additionally,
HPV-transfected DCs released significantly more inflammatory
cytokines (TNF-α and IL-6) after LPS stimulation than
non-transfected cells. These data suggest that papillomavirus may
act as a cofactor for induction of proinflammatory cytokines in DCs
when they are exposed to secondary inflammatory stimuli.
Keratinocytes are the primary target for
papillomavirus infection (23).
Previous studies have shown that keratinocytes secrete a variety of
lymphokines that are critical components of the host response to
infection or injury (40,41). HPV infection may result in changes
in cytokine production creating a favorable environment for viruses
to evade clearance by immune cells (3,14).
In this study, we found that cervical epithelial keratinocytes and
HFFs support expression of viral proteins. Although, HPV18
transfection altered cytokine production in keratinocytes and HFFs,
these changes were cell-type specific.
Since several reports proposed MCP-1 and TGF-β
involvement in the pathogenesis of HPV-associated cervical
neoplasms (30,31), we studied production of these
cytokines by HPV transfected cells. HPV18 infection of cervical
keratinocytes significantly decreased production of MCP-1. Previous
studies showed that MCP-1 plays an important role in recruitment of
monocytes to areas of virus entry and activates these cells to
secrete a variety of cytokines and chemokines (42,43).
It is believed that monocytes attracted into infected tissue can
differentiate into a variety of cells, including DCs (44). Thus, a local decrease in MCP-1
might alter the mechanism of DC recruitment into tumor tissue
leading to a decreased number of local DCs. Kleine and coworkers
(45) reported rapid growth of
tumors in nude mice inoculated with MCP-1-negative HeLa cells and
suggested that this effect was a result of a decrease in
macrophages migrating into the tumor tissue due to low levels of
MCP-1 produced by the tumor cells.
Decreases in TGF-β production by transformed
keratinocytes may also contribute to long-term maintenance of
papillomavirus infection. TGF-β is an important factor in
differentiating HPV-infected keratinocytes. For example, TGF-β
treatment of cultured monolayers is accompanied by downregulation
of HPV E6 and E7 transcription (46). Treatment of organotypic cultures
with TGF-β results in renewed ability of keratinocytes to express
morphological and biochemical markers, characteristic of a more
normal and differentiated phenotype (47). Previous studies established that
TGF-β stimulation is requisite for the development of DCs in
vitro and in vivo (48,49)
and is involved in the reciprocal control of DC chemotaxis
(50). In addition, TGF-β inhibits
upregulation of critical T-cell costimulatory molecules on the
surface of DCs. Furthermore, TGF-β appears to enhance antigen
processing and costimulatory functions of epithelial LCs (reviewed
by Strobl and Knapp) (51). Thus,
a decrease in local TGF-β as a result of HPV infection may abrogate
mechanisms of immune recognition and/or stimulation and suppress DC
migratory ability.
The reaction of human HFFs to HPV18 transformation
differed markedly from that of cervical keratinocytes. For example,
HPV18-HFFs produced significantly more MCP-1 and TGF-β than
non-transformed cells, whereas cervical keratinocytes responded in
the opposite manner. The increases in cytokine production by
HPV18-HFFs suggest that HPV transformation may lead to the
development of a localized tissue environment supportive for
increased DC numbers and differentiation. HFFs are a main component
of the subepithelial stroma. Previous studies have shown that the
immunological reactions of tumor tissue and peritumoral stroma may
be different. For example, Beutner and Tyring (22) found that, if immature DCs are
predominantly localized in breast carcinoma tissue, mature DCs
adhere selectively to peritumoral areas. Mota and coworkers
(14) discovered that
CD1+ DCs were occasionally detected in cervical
intraepithelial neoplastic lesions; however, leucocytes with an
activated phenotype accumulated immediately below the epithelial
lesions in subepithelial stroma (24). These observations and data
presented here, are consistent with the proposal that HFFs
expressing papillomavirus proteins develop an immunoactivating
cytokine profile; nevertheless, additional investigations are
necessary to delineate the role of stromal HFFs in pathogenesis of
papillomavirus infection.
Acknowledgements
Work conducted in the laboratory of
V.C.L. was supported, in part, by an award from the U.S. Department
of Health and Human Services, HERSA award C76HF19321; work
conducted in the laboratory of A.A.R. by FCP No. 14.A18.21.1930,
Russian Federation President Grant for young scientists (Dr.Sci.)
MД-433.2013.4, as well as the Federal Center of Collective Use and
Pharmaceutical Research and Education Center, Kazan (Volga Region)
Federal University, Kazan, Russia.
References
|
1.
|
zur Hausen H: Papillomavirus infections -
a major cause of human cancers. Biochim Biophys Acta. 1288:F55–F78.
1996.PubMed/NCBI
|
|
2.
|
Kanodia S, Fahey LM and Kast WM:
Mechanisms used by human papillomaviruses to escape the host immune
response. Curr Cancer Drug Targets. 7:79–89. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
3.
|
Frazer IH: The role of the immune system
in anogenital human papillomavirus. Australas J Dermatol. 39(Suppl
1): S5–S7. 1998.PubMed/NCBI
|
|
4.
|
al-Saleh W, Giannini SL, Jacobs N, et al:
Correlation of T-helper secretory differentiation and types of
antigen-presenting cells in squamous intraepithelial lesions of the
uterine cervix. J Pathol. 184:283–290. 1998. View Article : Google Scholar
|
|
5.
|
Coleman N, Birley HD, Renton AM, et al:
Immunological events in regressing genital warts. Am J Clin Pathol.
102:768–774. 1994.PubMed/NCBI
|
|
6.
|
al-Saleh W, Delvenne P, Arrese JE, Nikkels
AF, Pierard GE and Boniver J: Inverse modulation of intraepithelial
Langerhans’ cells and stromal macrophage/dendrocyte populations in
human papillomavirus-associated squamous intraepithelial lesions of
the cervix. Virchows Arch. 427:41–48. 1995.
|
|
7.
|
McArdle JP and Muller HK: Quantitative
assessment of Langerhans’ cells in human cervical intraepithelial
neoplasia and wart virus infection. Am J Obstet Gynecol.
154:509–515. 1986.
|
|
8.
|
Morelli AE, Sananes C, Di Paola G, Paredes
A and Fainboim L: Relationship between types of human
papillomavirus and Langerhans’ cells in cervical condyloma and
intraepithelial neoplasia. Am J Clin Pathol. 99:200–206. 1993.
|
|
9.
|
Morris HH, Gatter KC, Stein H and Mason
DY: Langerhans’ cells in human cervical epithelium: an
immunohistological study. Br J Obstet Gynaecol. 90:400–411.
1983.
|
|
10.
|
Viac J, Guerin-Reverchon I, Chardonnet Y
and Bremond A: Langerhans cells and epithelial cell modifications
in cervical intraepithelial neoplasia: correlation with human
papillomavirus infection. Immunobiology. 180:328–338. 1990.
View Article : Google Scholar
|
|
11.
|
Abbas AK, Lichtman AH and Pober JS:
Cellular and Molecular Immunology. Saunders; Philadelphia, PA:
1997
|
|
12.
|
Breathnach SM: The Langerhans cell. Br J
Dermatol. 119:463–469. 1988. View Article : Google Scholar
|
|
13.
|
Melief CJ: Dendritic cells as specialized
antigen-presenting cells. Res Immunol. 140:902–906; discussion
918–926, 1989.
|
|
14.
|
Mota F, Rayment N, Chong S, Singer A and
Chain B: The antigen-presenting environment in normal and human
papillomavirus (HPV)-related premalignant cervical epithelium. Clin
Exp Immunol. 116:33–40. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
15.
|
Hubert P, van den Brule F, Giannini SL,
Franzen-Detrooz E, Boniver J and Delvenne P: Colonization of in
vitro-formed cervical human papillomavirus-associated
(pre)neoplastic lesions with dendritic cells: role of
granulocyte/macrophage colony-stimulating factor. Am J Pathol.
154:775–784. 1999. View Article : Google Scholar
|
|
16.
|
Arany I, Goel A and Tyring SK: Interferon
response depends on viral transcription in human
papillomavirus-containing lesions. Anticancer Res. 15:2865–2869.
1995.PubMed/NCBI
|
|
17.
|
Park JS, Kim EJ, Kwon HJ, Hwang ES,
Namkoong SE and Um SJ: Inactivation of interferon regulatory
factor-1 tumor suppressor protein by HPV E7 oncoprotein.
Implication for the E7-mediated immune evasion mechanism in
cervical carcinogenesis. J Biol Chem. 275:6764–6769. 2000.
View Article : Google Scholar
|
|
18.
|
Evander M, Frazer IH, Payne E, Qi YM,
Hengst K and McMillan NA: Identification of the alpha6 integrin as
a candidate receptor for papillomaviruses. J Virol. 71:2449–2456.
1997.PubMed/NCBI
|
|
19.
|
Price AA, Cumberbatch M, Kimber I and Ager
A: Alpha 6 integrins are required for Langerhans cell migration
from the epidermis. J Exp Med. 186:1725–1735. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Bonnez W, Reichman RC, Strussenberg J and
Roberts NJ Jr: In vitro interactions between bovine papillomavirus
and human monocytes and macrophages. Intervirology. 32:246–252.
1991.PubMed/NCBI
|
|
21.
|
Tagami H, Oguchi M and Ofuji S: The
phenomenon of spontaneous regression of numerous flat warts:
immunohistological studies. Cancer. 45:2557–2563. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
22.
|
Beutner KR and Tyring S: Human
papillomavirus and human disease. Am J Med. 102:9–15. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
23.
|
Shah KV and Howley PM: Papillomaviruses.
Fields BN, Knipe DM and Howley M: Fields Virology. 3rd edition.
Lippincott-Raven Publishers; Philadelphia, PA: pp. 2077–2109.
1996
|
|
24.
|
Le Buanec H, D’Anna R, Lachgar A, et al:
HPV-16 E7 but not E6 oncogenic protein triggers both cellular
immunosuppression and angiogenic processes. Biomed Pharmacother.
53:424–431. 1999.
|
|
25.
|
Carter JJ, Koutsky LA, Hughes JP, et al:
Comparison of human papillomavirus types 16, 18, and 6 capsid
antibody responses following incident infection. J Infect Dis.
181:1911–1919. 2000. View
Article : Google Scholar : PubMed/NCBI
|
|
26.
|
Lin YL, Borenstein LA, Selvakumar R, Ahmed
R and Wettstein FO: Progression from papilloma to carcinoma is
accompanied by changes in antibody response to papillomavirus
proteins. J Virol. 67:382–389. 1993.PubMed/NCBI
|
|
27.
|
Park JS, Park DC, Kim CJ, et al:
HPV-16-related proteins as the serologic markers in cervical
neoplasia. Gynecol Oncol. 69:47–55. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
28.
|
Steele JC and Gallimore PH: Humoral assays
of human sera to disrupted and nondisrupted epitopes of human
papillomavirus type 1. Virology. 174:388–398. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
29.
|
Suzich JA, Ghim SJ, Palmer-Hill FJ, et al:
Systemic immunization with papillomavirus L1 protein completely
prevents the development of viral mucosal papillomas. Proc Natl
Acad Sci USA. 92:11553–11557. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
30.
|
Frazer IH, Thomas R, Zhou J, et al:
Potential strategies utilised by papillomavirus to evade host
immunity. Immunol Rev. 168:131–142. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
31.
|
Rosl F, Lengert M, Albrecht J, et al:
Differential regulation of the JE gene encoding the monocyte
chemoattractant protein (MCP-1) in cervical carcinoma cells and
derived hybrids. J Virol. 68:2142–2150. 1994.PubMed/NCBI
|
|
32.
|
Santin AD, Hermonat PL, Ravaggi A, et al:
Development, characterization and distribution of adoptively
transferred peripheral blood lymphocytes primed by human
papillomavirus 18 E7 - pulsed autologous dendritic cells in a
patient with metastatic adenocarcinoma of the uterine cervix. Eur J
Gynaecol Oncol. 21:17–23. 2000.
|
|
33.
|
Carlson MW, Iyer VR and Marcotte EM:
Quantitative gene expression assessment identifies appropriate cell
line models for individual cervical cancer pathways. BMC Genomics.
8:1172007. View Article : Google Scholar : PubMed/NCBI
|
|
34.
|
Miyasaka M, Takami Y, Inoue H and Hakura
A: Rat primary embryo fibroblast cells suppress transformation by
the E6 and E7 genes of human papillomavirus type 16 in somatic
hybrid cells. J Virol. 65:479–482. 1991.
|
|
35.
|
Yutsudo M, Okamoto Y and Hakura A:
Functional dissociation of transforming genes of human
papillomavirus type 16. Virology. 166:594–597. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
36.
|
Cumberbatch M, Dearman RJ and Kimber I:
Constitutive and inducible expression of interleukin-6 by
Langerhans cells and lymph node dendritic cells. Immunology.
87:513–518. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
37.
|
Saitoh A, Yasaka N, Osada A, Nakamura K,
Furue M and Tamaki K: Migration of Langerhans cells in an in vitro
organ culture system: IL-6 and TNF-alpha are partially responsible
for migration into the epidermis. J Dermatol Sci. 19:166–174. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
38.
|
Stoitzner P, Zanella M, Ortner U, et al:
Migration of langerhans cells and dermal dendritic cells in skin
organ cultures: augmentation by TNF-alpha and IL-1beta. J Leukoc
Biol. 66:462–470. 1999.PubMed/NCBI
|
|
39.
|
Hope JC, Cumberbatch M, Fielding I,
Dearman RJ, Kimber I and Hopkins SJ: Identification of dendritic
cells as a major source of interleukin-6 in draining lymph nodes
following skin sensitization of mice. Immunology. 86:441–447.
1995.PubMed/NCBI
|
|
40.
|
Gibran NS, Ferguson M, Heimbach DM and
Isik FF: Monocyte chemoattractant protein-1 mRNA expression in the
human burn wound. J Surg Res. 70:1–6. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
41.
|
Shreedhar V, Giese T, Sung VW and Ullrich
SE: A cytokine cascade including prostaglandin E2, IL-4, and IL-10
is responsible for UV-induced systemic immune suppression. J
Immunol. 160:3783–3789. 1998.PubMed/NCBI
|
|
42.
|
Mantovani A, Bottazzi B, Colotta F,
Sozzani S and Ruco L: The origin and function of tumor-associated
macrophages. Immunol Today. 13:265–270. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
43.
|
Miller MD and Krangel MS: Biology and
biochemistry of the chemokines: a family of chemotactic and
inflammatory cytokines. Crit Rev Immunol. 12:17–46. 1992.PubMed/NCBI
|
|
44.
|
Muller WA and Randolph GJ: Migration of
leukocytes across endothelium and beyond: molecules involved in the
transmigration and fate of monocytes. J Leukoc Biol. 66:698–704.
1999.PubMed/NCBI
|
|
45.
|
Kleine K, Konig G, Kreuzer J, Komitowski
D, Zur Hausen H and Rosl F: The effect of the JE (MCP-1) gene,
which encodes monocyte chemoattractant protein-1, on the growth of
HeLa cells and derived somatic-cell hybrids in nude mice. Mol
Carcinog. 14:179–189. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
46.
|
Braun L, Durst M, Mikumo R, Crowley A and
Robinson M: Regulation of growth and gene expression in human
papillomavirus-transformed keratinocytes by transforming growth
factor-beta: implications for the control of papillomavirus
infection. Mol Carcinog. 6:100–111. 1992. View Article : Google Scholar
|
|
47.
|
Ozbun MA and Meyers C: Transforming growth
factor beta1 induces differentiation in human
papillomavirus-positive keratinocytes. J Virol. 70:5437–5446.
1996.PubMed/NCBI
|
|
48.
|
Borkowski TA, Letterio JJ, Farr AG and
Udey MC: A role for endogenous transforming growth factor beta 1 in
Langerhans cell biology: the skin of transforming growth factor
beta 1 null mice is devoid of epidermal Langerhans cells. J Exp
Med. 184:2417–2422. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
49.
|
Geissmann F, Prost C, Monnet JP, Dy M,
Brousse N and Hermine O: Transforming growth factor beta1, in the
presence of granulocyte/macrophage colony-stimulating factor and
interleukin 4, induces differentiation of human peripheral blood
monocytes into dendritic Langerhans cells. J Exp Med. 187:961–966.
1998. View Article : Google Scholar
|
|
50.
|
Sato K, Kawasaki H, Nagayama H, et al:
TGF-beta 1 reciprocally controls chemotaxis of human peripheral
blood monocyte-derived dendritic cells via chemokine receptors. J
Immunol. 164:2285–2295. 2000. View Article : Google Scholar
|
|
51.
|
Strobl H and Knapp W: TGF-beta1 regulation
of dendritic cells. Microbes Infect. 1:1283–1290. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
52.
|
Nunez R, Sanchez M, Wild P, Filgueira L
and Nunez C: Characterisation of two human dendritic cell-lines
that express CD1a, take-up, process and present soluble antigens
and induce MLR. Immunol Lett. 61:33–43. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
53.
|
Cole ST and Danos O: Nucleotide sequence
and comparative analysis of the human papillomavirus type 18
genome. Phylogeny of papillomaviruses and repeated structure of the
E6 and E7 gene products. J Mol Biol. 193:599–608. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
54.
|
Dunzendorfer S, Kaser A, Meierhofer C,
Tilg H and Wiedermann CJ: Dendritic cell migration in different
micropore filter assays. Immunol Lett. 71:5–11. 2000. View Article : Google Scholar : PubMed/NCBI
|