Introduction
Gastric cancer is the second leading cause of death
from cancer in the world with low survival and high recurrence
especially for the advanced patients (1–3).
Signet ring cell gastric cancer (SRCGC) is a histologic diagnosis
based on microscopic characteristics as described by the World
Health Organization (4). It is
reported that the diffuse type gastric cancer, especially the
signet ring cell subtype, has become more prevalent and incidence
of SRCGC has an abrupt increase of 99.8% in recent years (5). Moreover, it is more malignant in
clinic and has worse prognosis than other forms of gastric cancer
(6). Cisplatin (DDP),
5-fluorouracil (5-FU), mitomycin (MMC), and epirubicin (EPI) are
the most effective anticancer drugs used in chemotherapy of gastric
cancer. However, >70% of patients are resistant to these drugs
(7) and the mechanism for
drug-resistance is not yet fully understood.
The cancer stem cells (CSCs) theory provides new
insight into cancer drug-resistance, initiation, development, and
metastasis (8,9). A number of studies have shown that
CD44-positive stem cells play an important role in drug resistance
in breast cancer and multiple myeloma (10,11).
For gastric cancer, CD44 has been demonstrated to be the most
widely used marker for cancer stem cells (12). CD44-positive cells have been
demonstrated to exist in many gastric cancer cell lines and show
spheroid colony formation ability in vitro and tumorigenic
ability in vivo. CD133 is another widely used stem cell
marker associated with poor survival in gastric patients (13) and it has not been fully studied in
cell lines. Function of stem cells in drug resistance for gastric
cancer is not fully understood.
Gastric cancer has been demonstrated to contain CSCs
in surface marker assays in many cell lines. Interestingly, the CSC
is heterogeneous in different gastric cell lines (12,14,15),
and its proportion is low in most of them. Among these cell lines
only one of the SRCGC has been studied. Some SRCGC cell lines have
been established only in Japan but have not been fully studied
(16–18). The study of gastric cancer stem
cells has been delayed due to a lack of proper cell line models.
However, more cell lines are urgently needed because of the
heterogeneity in gastric cancer and increasing incidence of
SRCGC.
The main purpose of our study was to establish a new
SRCGC cell line model for drug resistance and CSCs study. We
cultured cells from 6 patients with SRCGC and successfully
established one reliable cell line named as GCSR1. Its
chemotherapeutic response to most widely used drugs was evaluated
and the proportions of CD44 and CD133 were determined. The results
show that the cell line was drug-resistant and contained abundant
cancer stem cells. Thus, the GCSR1 is a useful tool for the
drug-resistance and cancer stem cell studies.
Materials and methods
Patients
The cell line was derived from the malignant primary
site from a 57-year-old Chinese male patient with gastric cancer,
of which the pathological diagnosis was SRCGC. The patient
underwent surgical resection and received FOLFOX6 treatment after
the surgery. The study was approved by the Ethics Committee of
Zhejiang University, and informed consent was signed by the patient
prior to the study.
Establishment of the cell line
Neoplastic tissue was obtained from the primary site
of the gastric cancer patient and xenografted into nude mice. When
the xenografts reached ~10 mm in diameter, the tumors were excised
to create the cell line GCSR1. Xenografts were minced by sterile
blades, and cells were placed in 1640 medium with 20% fetal bovine
serum (FBS). The cells were purified with differential adhesion
technique (19). The cells grew
well at 37°C in a humidified atmosphere containing 5%
CO2 and 95% air. Without interruption, >90 serial
passages were successively carried out during a period of >2
years. The cells continue to exhibit stable growth.
Cell culture
The human gastric cancer cell lines SGC7901 and
KATO-III were kept in our laboratory. SGC7901 was cultured under
adherent conditions in RPMI-1640 (Hyclone) with 10%
heat-inactivated fetal bovine serum (FBS, Gibco) at 37°C and 5%
CO2. KATO-III was cultured in DMEM (Hyclone) with 10%
heat-inactivated FBS (Gibco) at 37°C and 5% CO2.
Cell proliferation assay
Cells were placed on a 96-well plate (2,000
cells/well) and 42 parallel wells were subjected to the cell
proliferation assay for 7 days, 6 wells each. Cell-Counting kit
(CCK-8) reagent (BestBio) was added into the well and incubated for
3 h before reading (450 nm) using a multiwell spectrophotometer
(Bio-Rad).
Karyotyping
G-banding was conducted for karyotyping of GCSR1.
GCSR1 cells were cultured in a 37°C CO2 incubator and 24
h later exponentially growing cells were exposed to colcemid (0.8
μg/ml) for 5 h at 37°C for cell cycle arrest at metaphase.
Following hypotonic treatment and fixation, the slides were made by
dropping cells on glass slides and stained by Giemsa using a
routine laboratory procedure.
Soft agar colony formation assay
Cells (500–1,000) were suspended in 0.3% agarose
(0815, Amresco) in RPMI-1640 containing 20% FBS. This suspension
was overlaid onto a solid layer of 0.6% agarose in a 6-well plate.
The cells were treated with fresh RPMI-1640 containing 20% FBS
every other day. Three weeks later, the cells were photographed
under a microscope.
Mutation analysis of P53 and P16
Genomic DNA was extracted from GCSR1 using AxyPrep
DNA Gel Extraction kit. Polymerase chain reactions (PCR) of P53
exons 5–8 and P16 exons 1 and 3 were performed. PCR products were
sequenced (Invitrogen). The oligonucleotide primers were 5-GGA ATT
CCT CTT CCT GCA GTA CTC C-3 (forward) and 5-GGA ATT CAG TTG CAA ACC
A-3 (reverse) for exons 5 and 6 of P53 (378 bp); 5-GGA ATT CTC CTA
GGT TGG CTC TGA C-3 (forward) and 5-GGA ATT CCT GCT TGC TTA CCT CGC
T-3 (reverse) for exons 7 and 8 of P53 (590 bp); 5-TCT GCG GAG AGG
GGG AGA GCA G-3 (forward) and 5-GCG CTA CCT GAT TCC AAT TC-3
(reverse) for exon 1 of P16 (280 bp); 5-GGA TGT TCC ACA CAT CTT
TG-3 (forward) and 5-ATG AAA ACT ACG AAA GCG GG-3 (reverse) for
exon 3 of P16 (189 bp). The PCR conditions were: 30 cycles of 30
sec denaturation at 95°C, 30 sec annealing at 55°C for P53 and 58°C
for P16, 40 sec extension at 74°C, and 10 min final extension at
72°C. Subsequently, PCR products were loaded onto gels running for
20 min at 70 V.
Flow cytometry analysis
Seventy-ninety percent confluent cells in a 100-mm
cell plate were dissociated from plates using trypsin-EDTA
(Invitrogen), centrifuged, and then stained with CD44 (11-0441, BD
Biosciences), isotype control antibody CD44 (11-4031, BD
Biosciences) or CD133 (130-080-801, Miltenyi Biotec), isotype
control antibody CD133 (130-092-212, Miltenyi Biotec) at 4°C for 30
min. Analyses were performed using a BD FACSCanto II (BD
Biosciences).
Tumorigenicity in nude mice
To determine the tumorigenicity of the cell lines
in vivo, 6×106 viable cells were suspended in 200
μl PBS and subcutaneously injected into 5 nude mice. The mice were
examined every three days.
Chemosensitivity assays
The anticancer drugs, 5-FU (F6627), DDP (P4394), MMC
(Y0000378Y) and EPI (E9406) were from Sigma-Aldrich. All drugs were
used as recommended by the supplier. Sensitivity of GCSR1 cells to
each drug was examined using the CCK-8 kit (BestBio). Briefly,
cells were plated at a density of 2×104 cells per well
in 96-well plates containing 100 μl of culture medium. After 24-h
incubation at 37°C, drugs were added into wells at a total volume
of 10 μl per well and incubated for an additional 72 h. CCK-8
reagent was then added into each well and incubated for 3 h before
reading at a wavelength of 450 nm using a multiwell
spectrophotometer (Bio-Rad). IC50 values were calculated
using GraphPad Prism 5 (GraphPad software) at least three
independent experiments.
Migration and invasion assays
Briefly, 8×105 cells were plated on upper
chambers of Transwell plates (Corning) and 700 μl of RPMI-1640 with
10% FBS was added to the lower chambers. The plates were then
placed in an incubator at 37°C with 5% CO2 for 12 h.
After incubation, the cells remaining in the upper
chamber were carefully removed, and the Transwell membranes were
fixed with dehydrated ethanol and stained with 0.2% crystal violet.
To count the fixed cells, photographs were taken from 5 independent
fields at ×200 magnification. Independent experiments were
performed in triplicate.
For the cell invasionassay, Matrigel (356234, BD
Biosciences) was thawed on ice at 4°C overnight and diluted with
serum-free medium at a ratio of 1:9. The Transwell chambers were
coated with 40 μl of diluted Matrigel in a 24-well plate and
incubated at 37°C for 2 h before 1.5×105 cells in
serum-free RPMI-1640 medium were seeded into the prepared Transwell
chambers. Then, 700 μl of 1640 with 10% FBS was added to the lower
chambers. The 24-well plate was incubated at 37°C with 5%
CO2 for 12 h. Cells were stained and counted as in the
migration assay.
Western blotting
Western blotting was performed with a SDS-PAGE
electrophoresis system. Briefly, 20-μg protein samples were
resuspended in a reduced sample buffer, and then electrophoresed on
an 8–10% Tris gel with Tris running buffer, blotted to PVDF
membrane, and sequentially probed with primary antibodies against
Her2 (1:200, SC-284, Santa Cruz Biotechnology), EGFR (1:10,000,
ab32077, Abcam), E-cad (1:1,000, ab1416, Abcam), vimentin (1:200,
Santa Cruz Biotechnology). Secondary antibodies coupled to
horseradish peroxidase (HRP) were detected through autoradiography
using enhanced chemiluminescence (ECL Plus, Millipore).
Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (1:5,000) (KC-5G4,
Kangchen) was used as a loading control.
Statistical analysis
The unpaired 2-tailed t-test was used with GraphPad
Prism 5. P<0.01 was considered statistically significant.
Results
Morphological features
The culture cells from passages 38 and 70 were
analyzed by a microscope and H&E staining for the morphological
feature to confirm that the cell line was signet ring cell origin
and stable in vitro. GCSR1 presented various shapes under
the microscope at passage 38 (Fig.
1A) and the morphology was similar with passage 70 (Fig. 1B). H&E staining of cells at
passages 38 (Fig. 1C) and 70
(Fig. 1D) both showed typical
signet ring cell morphology, in which the cells were filled with
mucin vacuoles that pushed the nucleus to one side. Further, the
cell culture implantation and primary tumor from animals were
analyzed by H&E staining to confirm the cell line was
pathologically identical to primary tumor, which showed that GCSR1
cells (Fig. 1E) were similar to
the primary tumor in vivo (Fig.
1F).
Proliferation features
The GCSR1 cells remained in culture for >90
passages and became a stable cell line. It proliferated with a
doubling time of approximately 67.65 h (Fig. 2A) in normal culture and
demonstrated anchorage-independence and is able to form colonies in
soft agar (Fig. 2B and C).
Karyotype analysis
Karyotype analysis showed that GCSR1 cells were
aneuploid with a median chromosome number 65 (XXY). There were
multiple copies of chromosomes A1–A3, B4, B5, C7–C12, E17 and E18;
deletions of C6, D13 and G22; and some unknown chromosomes in GCSR1
(Fig. 3).
Sequencing analysis of P53 and P16
mutations
Mutation analysis was performed in GCSR1 cells for
the tumor suppressor genes, P53 and P16, respectively. P53 gene
exhibited CAT→CGT missense mutation (histidine→arginine) in codon
193 of exon 6 (Fig. 4A), while no
other mutations in exon 5 or exons 7 and 8 were found (data not
shown). Homozygous loss of codons 1 and 3 was observed in P16
(Fig. 4B).
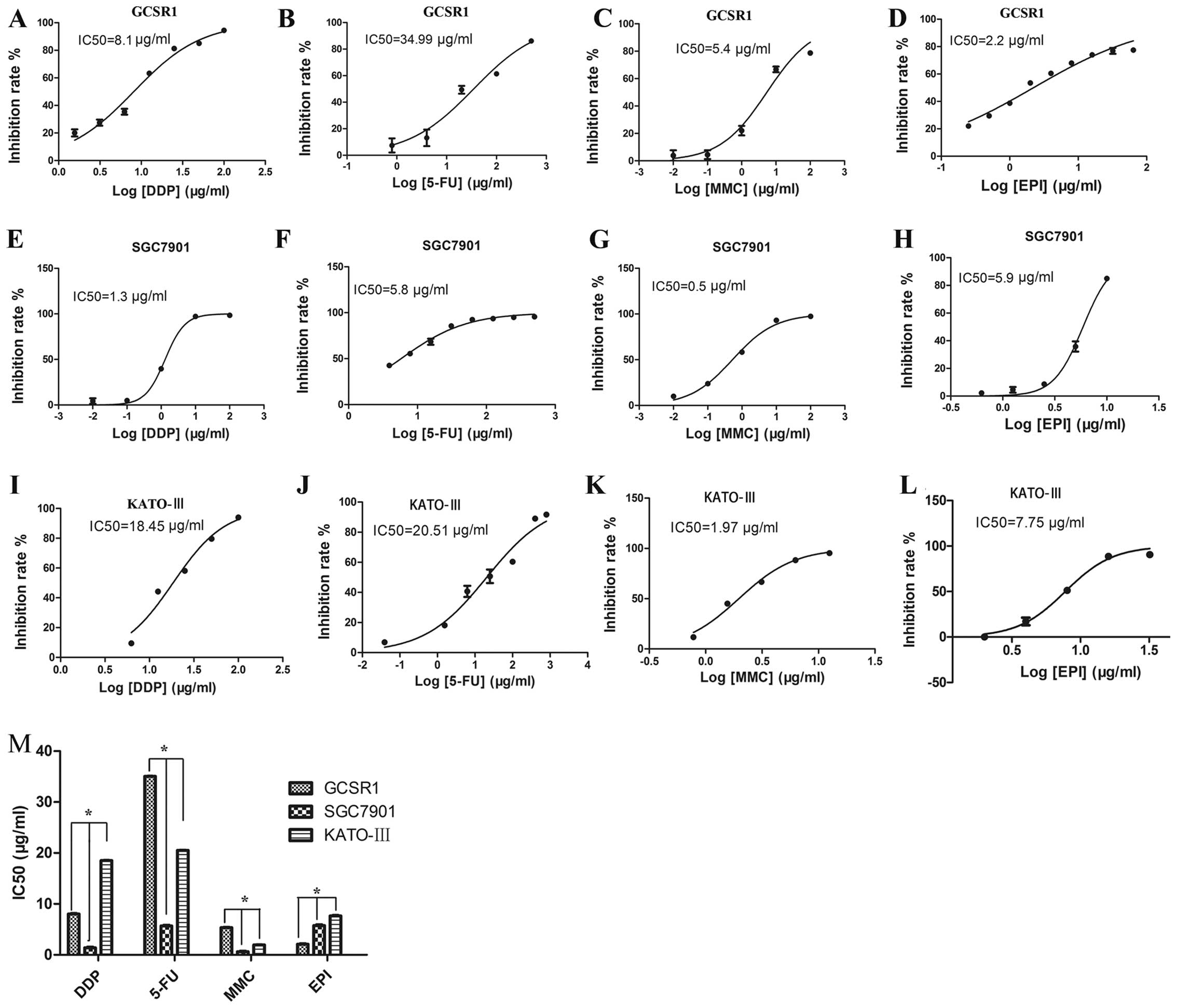
Drug sensitivity assay
To clarify the drug sensitivity of GCSR1 cells, DDP,
5-FU, MMC and EPI were examined. The SGC7901 (20) (intestinal gastric cancer) and
KATO-III (16) (signet ring cell
gastric cancer) cell lines were used as positive control. The
IC50 of GCSR1 to DDP, 5-FU, MMC, and EPI were 8.1,
34.99, 5.4 and 2.2 μg/ml, respectively, after 72-h exposure
(Fig. 5A–D). The IC50
of SGC7901 to DDP, 5-FU, MMC, and EPI were 1.3, 5.8, 0.5 and 5.9
μg/ml, respectively (Fig. 5E–H).
The IC50 of KATO-III cells to DDP, 5-FU, MMC and EPI
were 18.45, 20.51, 1.97 and 7.75 μg/ml, respectively (Fig. 5I–L). GCSR1 cells were more
resistant than the SGC7901 cells to three drugs: 6.20-fold for DDP,
6.0-fold for 5-FU and 10.8-fold for MMC (Fig. 5M); and more resistant than the
KATO-III cells to two drugs: 1.7-fold for 5-FU and 2.74-fold for
MMC (Fig. 5M). These results
indicate that GCSR1 is a multi-drug resistant cell line.
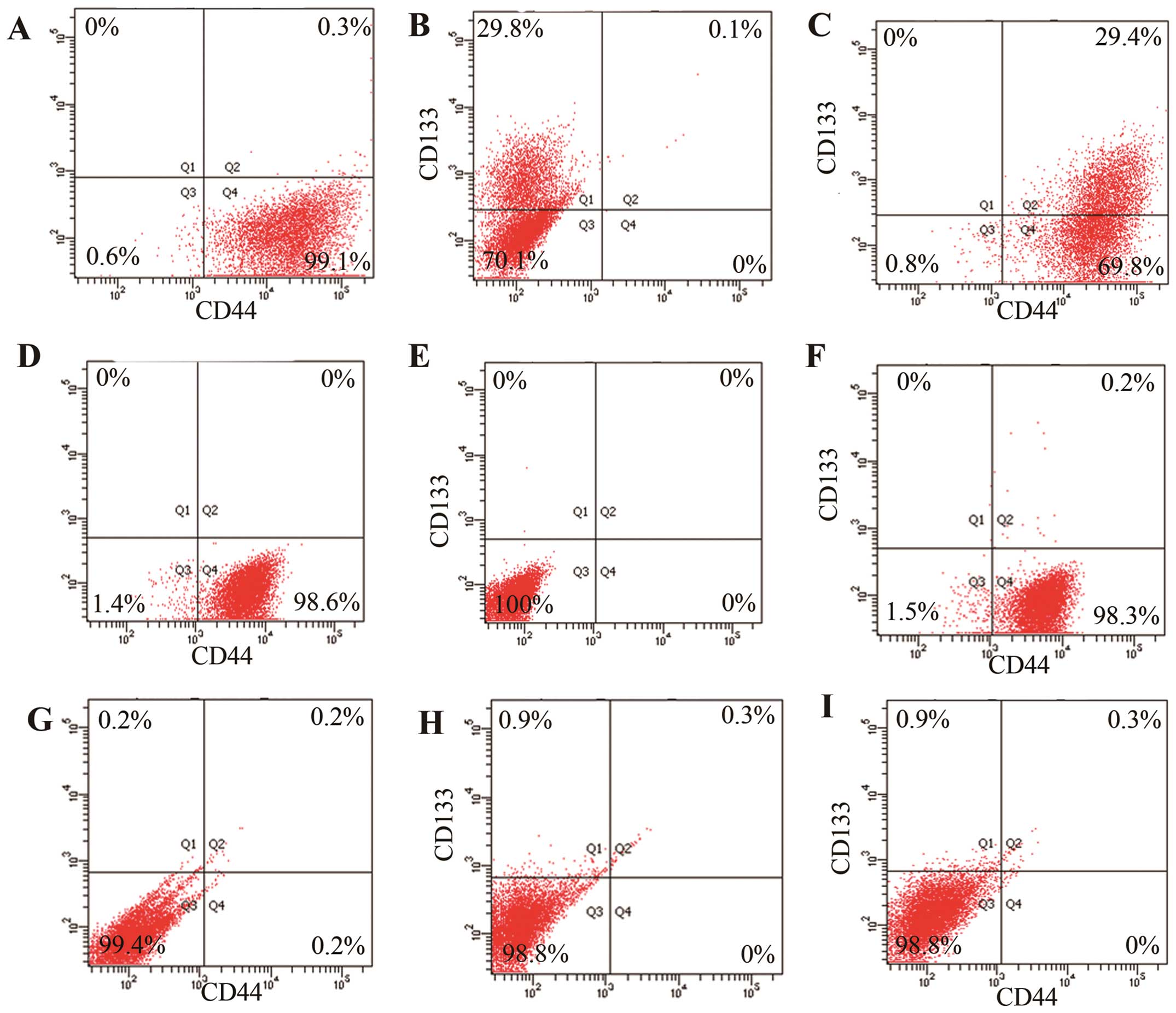
CD44 and CD133 expression assay
Flow cytrometry was performed to clarify the
proportion of CSCs that was high in GCSR1, the results showed that
99.2% of GCSR1 cells were CD44-positive (Fig. 6A) and 29.5% were CD133-positive
(Fig. 6B). Interestingly, almost
all the CD133-positive cells were also CD44-positive (Fig. 6C). By contrast, 98.6% of SGC7901
cells were CD44-positive (Fig. 6D)
and 0% were CD133-positive (Fig.
6E) and 0.2% were CD44+CD133+ (Fig. 6F). In the cell line KATO-III, only
0.2% of the cells were CD44-positive (Fig. 6G), 0.9% were CD133-positive
(Fig. 6H), and 0.3% were
CD44+CD133+ (Fig. 6I). These results show that high
proportion of stem cells partially explained the drug resistance in
GCSR1 cells.
Tumorigenicity in vivo
In order to confirm that the GCSR1 cells have higher
proportion of stem cells, we examined self-renewal ability in
vivo, which is the most important characteristic of CSCs
(21). Viable cells
(6×106) were implanted by s.c. injection into 5 nude
mice. The GCSR1 cells demonstrated a significant tumor growth, with
100% tumor formation rate (Fig.
7A), while only 60% was seen in SGC7901 (Fig. 7B) and 0% in KATO-III (data not
shown). Tumor volumes in GCSR1 (Fig.
7C) were larger than SGC7901 (Fig.
7D) and KATO-III (data not shown). These data indicate that
GCSR1 had abundant stem cells.
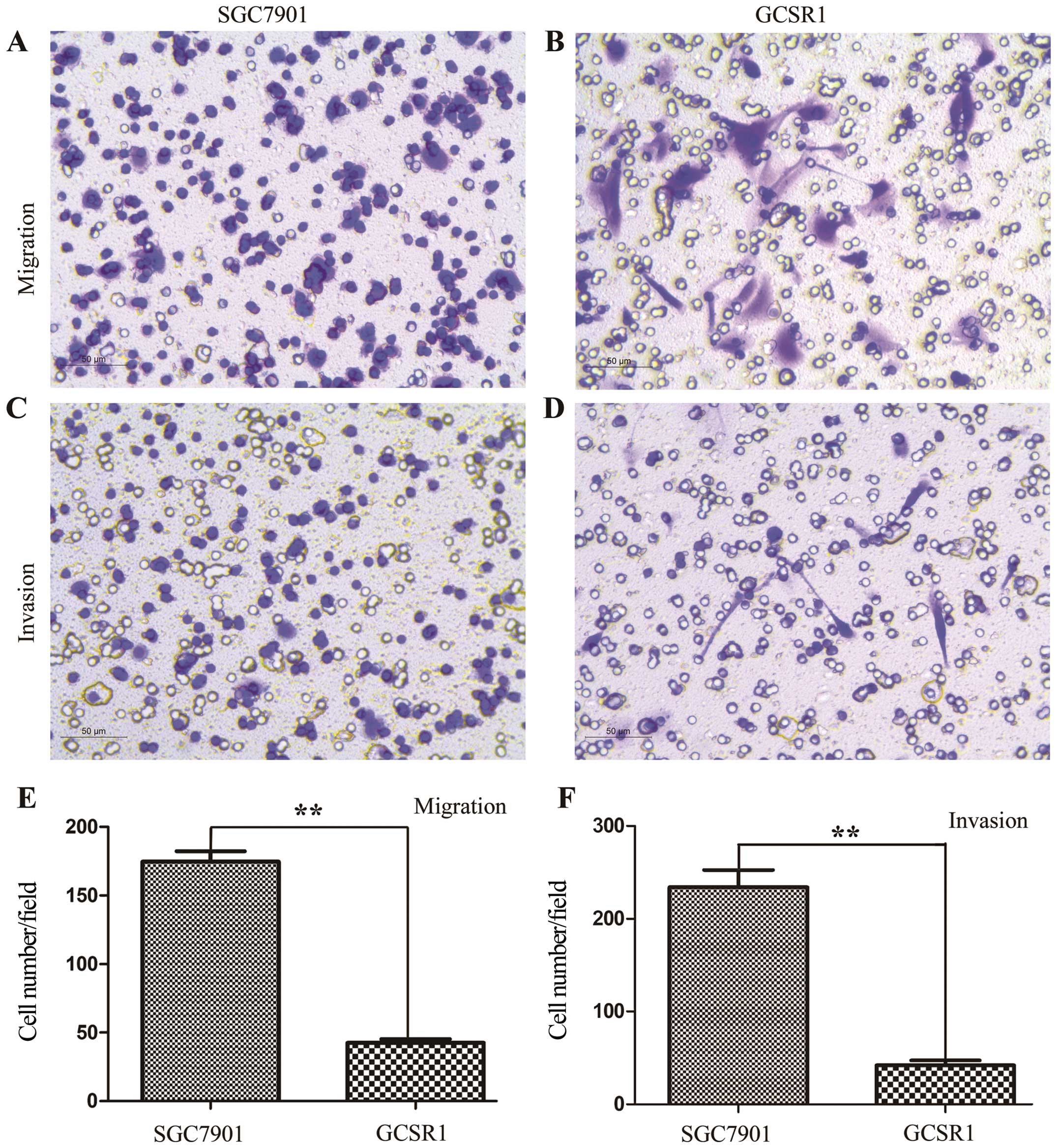
Migration and invasion assay
In vitro transwell migration and invasion
assays were used to examine the migratory and invasive potential of
the GCSR1 cells. GCSR1 showed both migration and invasion after 12
h, which were weaker than those in SGC7901 (Fig. 8), while KATO-III showed no
migration or invasion (data not shown).
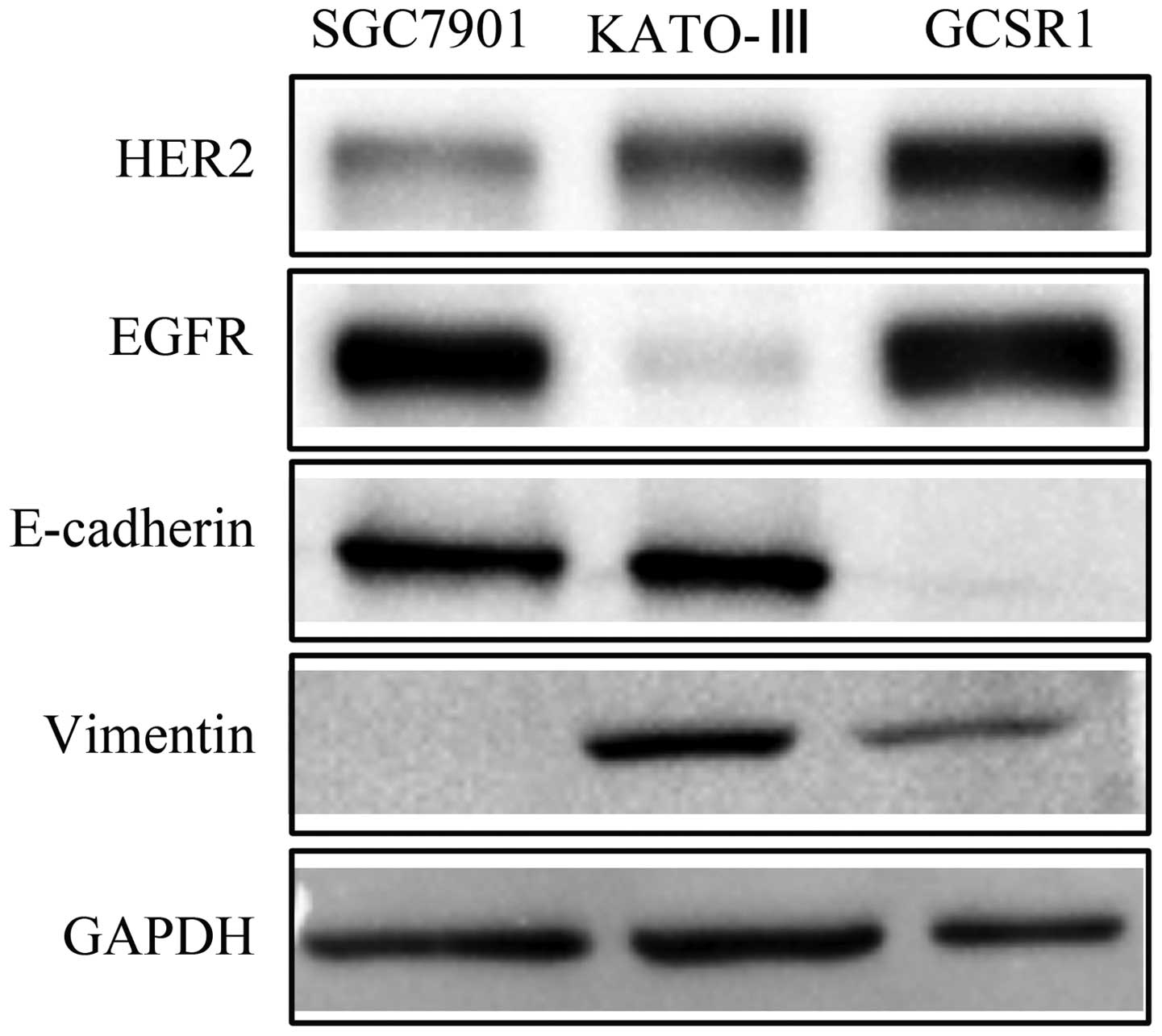
Western blots of HER2, EGFR and EMT
markers
HER pathway played a critical role in stem cell
survival (22) and trastuzumab has
been proved to target HER2 effectively in advanced gastric cancer
(23). EMT markers were reported
to be associated with drug resistance and stem cell phonotype
(24–28). The expression of these proteins is
important for understanding drug resistance and finding potential
drug target markers for drug resistant cell line GCSR1. Western
blot analyses revealed high expression of HER2 and EGFR in GCSR1,
compared with SGC7901 and KATO-III as positive control (Fig. 9). These results demonstrate that
HER2 and EGFR can be potential therapy targets in GCSR1. Western
blot analyses also revealed that GCSR1 had acquired the expression
of fibroblastic-related protein such as vimentin and lost the
expression of epithelial specific gene E-cad (Fig. 9), indicating that GCSR1 was
transformed to myofibroblast-like cells via epithelial-mesenchymal
transition.
Discussion
Chemotherapy is a major treatment strategy for
gastric cancer, but its efficacy is poor. The main reason for drug
failure is multidrug resistance (MDR), of which the mechanisms in
gastric cancer are complex and have not been clarified. A number of
studies have been conducted to reveal the cellular mechanisms of
drug resistance in gastric cancer. The most widely used
experimental models are the acquired drug-resistant cell lines,
which were selected in the presence of cytotoxic agents. By
comparing the naive parental cells to these drug-resistant cell
lines using cellular and molecular biology tools, many
drug-resistance associated molecules have been revealed (29). The classical drug-resistant
molecules are P-glycoprotein/ABCB1 and MRP1/ABCC1, which played
important roles in mediating MDR in some gastric cancers (30). However, the drug resistance is
complex in clinic and has not been fully solved. New experimental
models, especially primary drug resistance cell lines established
from clinical samples is required for drug resistance study. GCSR1,
which is a new cell line established from a SRCGC patient, showed
stronger multi-drug resistance to chemotherapies compared to other
cell lines such as SGC7901 and KATO-III. Our results show that
GCSR1 holds high homogeneity to the original tumor tissues, so it
can represent not only the primary tumor, but also the primary drug
resistant model, better than some other established cell lines. It
is a useful model for investigation of the mechanisms of gastric
cancer drug resistance.
Cancer stem cells have been studied in SGC7901 and
KATO-III (12,15), and the results show that these two
cell lines contained CD44+ cells but showed weak
tumorigenicity in vivo. By contrast, GCSR1 had higher
proportions of CD44 stem cells (99.8%) and showed self-renewal
ability both in vitro and in vivo. High proportion of
stem cells partially explains the malignancy and drug resistance in
GCSR1. Our laboratory has also established a patient-derived human
tumor tissue (PDTT) model (31),
which is derived from the same patient as the GCSR1 cells.
Therefore, further study will need to test the GCSR1 cell line
together with the stable PDTT model to confirm the stem cell
function in drug resistance.
Targeting therapies have become more attractive as
therapeutic strategies in gastric cancer and HER2 is of particular
interest as a drug target especially for advanced gastric cancer
(23). We tested HER2 expression
in GCSR1 and found it was highly expressed compared with that in
SGC7901 and KATO-III. The results indicate that trastuzumab, a
monoclonal antibody that interferes with the HER2 receptor, may be
considered for the treatment of GCSR1.
Recently, intra-tumor heterogeneity has been
clarified for cancer treatment, which was a topic in AACR Annual
Meeting 2014. Thus, an in vitro model that can mimic the
situation of intra-tumor heterogeneity is urgently needed. However,
long-term cell lines in vitro lead to the selection of
subpopulations over time. On the contrary, primary cell lines keep
the heterogeneity of the primary tumor and thus provide a useful
tool for further study. Moreover, we also established further nine
sub-clones from the early passage cells from the same tumor tissue
along with GCSR1. These sub-clones display significant
heterogeneity in many aspects. We are now conducting more
experiments to further study intra tumor heterogeneity of SRCGC. In
conclusion, GCSR1 is a well-established, well-characterized
multi-drug resistant cell line with abundant cancer stem cells.
Acknowledgements
This study was supported by the funds from National
Natural Science Foundation of China (no. 81272676); National
Science and Technology Major Project of the Ministry of Science and
Technology of China (no. 2013ZX09506015); Medical Science and
Technology Project of Zhejiang Province (no. 2011ZDA009). The
authors would like to thank Qin-Yuan Lou, Chi-Hong Zhu and Yu-Tian
Ling for their assistance during this study.
References
|
1
|
Jemal A, Center MM, DeSantis C and Ward
EM: Global patterns of cancer incidence and mortality rates and
trends. Cancer Epidemiol Biomarkers Prev. 19:1893–1907. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Cunningham D, Allum WH, Stenning SP,
Thompson JN, van de Velde CJ, Nicolson M, Scarffe JH, Lofts FJ,
Falk SJ, Iveson TJ, et al: Perioperative chemotherapy versus
surgery alone for resectable gastroesophageal cancer. N Engl J Med.
355:11–20. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Macdonald JS, Smalley SR, Benedetti J,
Hundahl SA, Estes NC, Stemmermann GN, Haller DG, Ajani JA,
Gunderson LL, Jessup JM, et al: Chemoradiotherapy after surgery
compared with surgery alone for adenocarcinoma of the stomach or
gastroesophageal junction. N Engl J Med. 345:725–730. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Jass JR, Sobin LH and Watanabe H: The
World Health Organization’s histologic classification of
gastrointestinal tumors. A commentary on the second edition.
Cancer. 66:2162–2167. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Henson DE, Dittus C, Younes M, Nguyen H
and Albores-Saavedra J: Differential trends in the intestinal and
diffuse types of gastric carcinoma in the United States, 1973–2000:
increase in the signet ring cell type. Arch Pathol Lab Med.
128:765–770. 2004.PubMed/NCBI
|
|
6
|
Taghavi S, Jayarajan SN, Davey A and
Willis AI: Prognostic significance of signet ring gastric cancer. J
Clin Oncol. 30:3493–3498. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Shah MA and Kelsen DP: Gastric cancer: a
primer on the epidemiology and biology of the disease and an
overview of the medical management of advanced disease. J Natl
Compr Cancer Netw. 8:437–447. 2010.
|
|
8
|
Gilbert CA and Ross AH: Cancer stem cells:
cell culture, markers, and targets for new therapies. J Cell
Biochem. 108:1031–1038. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Passegue E, Rafii S and Herlyn M: Cancer
stem cells are everywhere. Nat Med. 15:232009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Palyi-Krekk Z, Barok M, Isola J, Tammi M,
Szollosi J and Nagy P: Hyaluronan-induced masking of ErbB2 and
CD44-enhanced trastuzumab internalisation in trastuzumab resistant
breast cancer. Eur J Cancer. 43:2423–2433. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bjorklund CC, Baladandayuthapani V, Lin
HY, Jones RJ, Kuiatse I, Wang H, Yang J, Shah JJ, Thomas SK, Wang
M, et al: Evidence of a role for CD44 and cell adhesion in
mediating resistance to lenalidomide in multiple myeloma:
therapeutic implications. Leukemia. 28:373–383. 2014. View Article : Google Scholar
|
|
12
|
Takaishi S, Okumura T, Tu S, Wang SS,
Shibata W, Vigneshwaran R, Gordon SA, Shimada Y and Wang TC:
Identification of gastric cancer stem cells using the cell surface
marker CD44. Stem Cells. 27:1006–1020. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chen S, Hou JH, Feng XY, Zhang XS, Zhou
ZW, Yun JP, Chen YB and Cai MY: Clinicopathologic significance of
putative stem cell marker, CD44 and CD133, in human gastric
carcinoma. J Surg Oncol. 107:799–806. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
She JJ, Zhang PG, Wang X, Che XM and Wang
ZM: Side population cells isolated from KATO III human gastric
cancer cell line have cancer stem cell-like characteristics. World
J Gastroenterol. 18:4610–4617. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Xue Z, Yan H, Li J, Liang S, Cai X, Chen
X, Wu Q, Gao L, Wu K, Nie Y, et al: Identification of cancer stem
cells in vincristine preconditioned SGC7901 gastric cancer cell
line. J Cell Biochem. 113:302–312. 2012. View Article : Google Scholar
|
|
16
|
Yanagihara K, Seyama T, Tsumuraya M,
Kamada N and Yokoro K: Establishment and characterization of human
signet ring cell gastric carcinoma cell lines with amplification of
the c-myc oncogene. Cancer Res. 51:381–386. 1991.PubMed/NCBI
|
|
17
|
Murakami H, Nakanishi H, Tanaka H, Ito S,
Misawa K, Ito Y, Ikehara Y, Kondo E and Kodera Y: Establishment and
characterization of novel gastric signet-ring cell and non
signet-ring cell poorly differentiated adenocarcinoma cell lines
with low and high malignant potential. Gastric Cancer. 16:74–83.
2013. View Article : Google Scholar
|
|
18
|
Sekiguchi M, Sakakibara K and Fujii G:
Establishment of cultured cell lines derived from a human gastric
carcinoma. Jpn J Exp Med. 48:61–68. 1978.PubMed/NCBI
|
|
19
|
Kreider BQ, Messing A, Doan H, Kim SU,
Lisak RP and Pleasure DE: Enrichment of Schwann cell cultures from
neonatal rat sciatic nerve by differential adhesion. Brain Res.
207:433–444. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lin CH, Fu ZM, Liu YL, Yang JL, Xu JF,
Chen QS and Chen HM: Investigation of SGC-7901 cell line
established from human gastric carcinoma cells. Chin Med J (Engl).
97:831–834. 1984.
|
|
21
|
Kreso A and Dick JE: Evolution of the
cancer stem cell model. Cell Stem Cell. 14:275–291. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ithimakin S, Day KC, Malik F, Zen Q,
Dawsey SJ, Bersano-Begey TF, Quraishi AA, Ignatoski KW, Daignault
S, Davis A, et al: HER2 drives luminal breast cancer stem cells in
the absence of HER2 amplification: implications for efficacy of
adjuvant trastuzumab. Cancer Res. 73:1635–1646. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Bang YJ, Van Cutsem E, Feyereislova A,
Chung HC, Shen L, Sawaki A, Lordick F, Ohtsu A, Omuro Y, Satoh T,
et al: Trastuzumab in combination with chemotherapy versus
chemotherapy alone for treatment of HER2-positive advanced gastric
or gastro-oesophageal junction cancer (ToGA): a phase 3,
open-label, randomised controlled trial. Lancet. 376:687–697. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Iseri OD, Kars MD, Arpaci F, Atalay C, Pak
I and Gunduz U: Drug resistant MCF-7 cells exhibit
epithelial-mesenchymal transition gene expression pattern. Biomed
Pharmacother. 65:40–45. 2011. View Article : Google Scholar
|
|
25
|
McConkey DJ, Choi W, Marquis L, Martin F,
Williams MB, Shah J, Svatek R, Das A, Adam L, Kamat A, et al: Role
of epithelial-to-mesenchymal transition (EMT) in drug sensitivity
and metastasis in bladder cancer. Cancer Metastasis Rev.
28:335–344. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shah AN, Summy JM, Zhang J, Park SI,
Parikh NU and Gallick GE: Development and characterization of
gemcitabine-resistant pancreatic tumor cells. Ann Surg Oncol.
14:3629–3637. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang Z, Li Y, Kong D, Banerjee S, Ahmad A,
Azmi AS, Ali S, Abbruzzese JL, Gallick GE and Sarkar FH:
Acquisition of epithelial-mesenchymal transition phenotype of
gemcitabine-resistant pancreatic cancer cells is linked with
activation of the notch signaling pathway. Cancer Res.
69:2400–2407. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan
A, Zhou AY, Brooks M, Reinhard F, Zhang CC, Shipitsin M, et al: The
epithelial-mesenchymal transition generates cells with properties
of stem cells. Cell. 133:704–715. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhao Y, You H, Liu F, An H, Shi Y, Yu Q
and Fan D: Differentially expressed gene profiles between multidrug
resistant gastric adeno-carcinoma cells and their parental cells.
Cancer Lett. 185:211–218. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Szakacs G, Paterson JK, Ludwig JA,
Booth-Genthe C and Gottesman MM: Targeting multidrug resistance in
cancer. Nat Rev Drug Discov. 5:219–234. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Jin K, He K, Han N, Li G, Wang H, Xu Z,
Jiang H, Zhang J and Teng L: Establishment of a PDTT xenograft
model of gastric carcinoma and its application in personalized
therapeutic regimen selection. Hepatogastroenterology.
58:1814–1822. 2011. View Article : Google Scholar : PubMed/NCBI
|