Introduction
Pancreatic cancer is a leading cause of
cancer-related death, largely due to metastatic dissemination, and
it has the worst prognosis of any major tumor type, with a 5-year
survival rate of ~5% (1). Patients
with pancreatic cancer respond poorly to existing chemotherapeutic
agents and radiation due to the high degree of hypoxia in
pancreatic tumors (2,3). Hypoxia-inducible factor-1α (HIF-1α)
and vascular endothelial growth factor (VEGF) are ROS-related
proteins that are both regulated by Ape1 redox signaling (4). Furthermore, ROS have been shown to
switch on hypoxia-dependent epithelial-mesenchymal transition (EMT)
in cancer cells (5). Abnormal
activation of the WNT/β-catenin pathway is also associated with
pancreatic cancer (6). Because ROS
are important intracellular messengers that can modulate
WNT/β-catenin signaling by a redox mechanism (7), we hypothesized that there may be
crosstalk between Ape1 redox signaling and WNT/β-catenin signaling
in pancreatic cancer cells.
Ape1 is a multifunctional protein that regulates a
wide variety of important cellular functions (8). It functions as an
apurinic/apyrimidinic endonuclease in base excision repair (BER) of
DNA lesions and as a redox-modifying factor in eukaryotic
transcriptional regulation (9–13).
For example, the redox function of Ape1 can stimulate the
DNA-binding activity of HIF-1α, nuclear factor
kappa-light-chain-enhancer of activated B cells (NF-κB), activator
protein 1 (AP-1), and p53 (12–16).
A recent report also showed that Ape1 is an essential factor
stabilizing telomeric DNA. Its deficiency is associated with
telomere dysfunction and segregation defects in immortalized cells
maintaining telomeres by either the alternative lengthening of
telomere pathway or telomerase expression. The DNA repair and
N-terminal acetylation domains are required for Ape1 function at
telomeres. Ape1 associates with telomere proteins in U2OS cells,
and Ape1 depletion causes dissociation of telomeric repeat-binding
factor 2 (TRF2) from telomeres (17). Ape1 also regulates endothelial
nitric oxide (eNO) production and vascular tone (18), and interacts with nucleophosmin
(NPM1) within nucleoli, where it not only plays a role in the rRNA
quality control process, but also where its BER activity is
stimulated in cells (19–21). Furthermore, Ape1 can be
phosphorylated by cyclin-dependent kinase 5 (Cdk5) complexes, thus,
reducing its apurinic/apyrimidinic (AP) endonuclease activity,
which results in the accumulation of DNA damage and contributes to
neuronal death (22). Accumulating
evidence suggests that Ape1 is upregulated in pancreatic cancers
(23), and it can affect
WNT/β-catenin signaling target genes, such as cyclin D1 and
c-myc, in different ways (24,25).
In addition, small interfering RNA targeting Ape1 enhances the
sensitivity of human pancreatic cancer cells to gemcitabine in
vitro (26). Single nucleotide
polymorphisms (SNPs) in the Ape1 gene are also associated
with breast cancer risk in a Chinese population (27), suggesting Ape1 plays a role in
multiple types of cancer. Nevertheless, the functional role of Ape1
in tumor pathogenesis and progression remains unclear.
In the present study, we define a critical pathway
by which Ape1 regulates WNT/β-catenin signaling through its redox
function. Inhibition of the redox functional domain of Ape1 by
E3330 resulted in the upregulation of β-catenin and its target gene
c-myc, but not cyclin D1. The level of cyclin D1 was positively
correlated with Ape1, but not c-myc. Treating cells with a
combination of an Ape1 inhibitor and a WNT inhibitor was more
effective at inhibiting pancreatic cell proliferation, compared
with either inhibitor alone. These data indicate that using a
combination of these inhibitors will enhance their efficacy in
pancreatic cancer therapy.
Materials and methods
Cell lines
SW-1990 and Panc-1 were purchased from, and
authenticated by the American Type Culture Collection (ATCC,
Manassas, VA, USA). Cells were maintained at 37°C in 5%
CO2 and grown in Dulbecco’s modified Eagle’s medium
(DMEM; HyClone, Logan, UT, USA) with 10% fetal bovine serum (FBS;
Invitrogen, Carlsbad, CA, USA).
Plasmid construction
Cysteines 65 and 93 in human Ape1 were mutated using
the Quick Change Site-Directed Mutagenesis kit (Stratagene, La
Jolla, CA, USA) to generate Ape1 (C65/93A). Ape1 (ΔNLS) encodes
Ape1 with a 20-aa deletion of the putative N-terminal nuclear
localization signal. All mutations and deletions were verified by
DNA sequencing. Wild-type Ape1 (WT), Ape1 (C65/93A), and Ape1
(ΔNLS) were cloned into the pDsRed-N1 expression vector by standard
cloning methods, as previously described (18).
Cell transfections
SW-1990 cells were transfected with each Ape1
plasmid (2 μg) using Lipofectamine 2000 (Invitrogen). Forty-eight
hours after transfection with plasmids, stable transfected cell
lines were selected for by growing the cells in G418 (800 μg/ml)
for one month. The cells were imaged with a fluorescence microscope
(Carl Zeiss, Oberkochen, Germany). The stable transfected cells
were then harvested for analysis by RT-PCR, western blot analysis
and flow cytometry.
RNA interference
SW-1990 and Panc-1 cells were transfected with 40 nM
siApe1 using Lipofectamine 2000 (Invitrogen), as described by the
manufacturer. The siRNA sequence targeting human Ape1 was
previously described (17). The
transfected cells were maintained for 48 h, and then harvested for
analysis by RT-PCR and western blot analysis.
RNA isolation and RT-PCR
SW-1990 and Panc-1 cells were harvested after
incubating with E3330 and/or IWR-1 for 48 h. Total RNA was isolated
using TRIzol reagent (Invitrogen) according to the manufacturer’s
instructions. Single-stranded cDNA was synthesized in a 20 μl final
volume containing 1 μg of total RNA, 4 μl of 5× reverse
transcriptase buffer, 4 μl of 2.5 mM dNTP, 1 μl of oligo-dT (100
pmol/μl), 1 μl of RNase inhibitor (4 units/μl), and 1 μl of AMV
reverse transcriptase (RT) (5 units/μl) at 42°C for 1 h. Then, the
reaction mixture was boiled for 5 min to inactivate the RT and
quickly chilled on ice. The gene-specific primers used for PCR
amplification are listed in Table
I. PCR was performed using a thermal cycler (Applied
Biosystems, Foster City, CA, USA). PCR products were analyzed by
electrophoresis on a 1.5% agarose gel.
 | Table IPrimers for RT-PCR. |
Table I
Primers for RT-PCR.
| Gene | Primer
sequence | Size (bp) | Temperature
(°C) |
|---|
| Ape1 | Forward:
ACTTCAGGAGCTGCCTGGACT | | |
| Reverse:
AATGCAGGTAACAGAGAGTGGGA | 564 | 56 |
|
β-catenin | Forward:
TGATGGAGTTGGACATGGCCATGG | | |
| Reverse:
CAGACACCATCTGAGGAGAACGCA | 570 | 62.5 |
| c-myc | Forward:
GCGTCCTGGGAAGGGAGATCCGGAGC | | |
|
Reverse:TTGAGGGGCATCGTCGCGGGAGGCTG | 328 | 62.5 |
| Cyclin
D1 | Forward:
GAGAACAAACAGATCATCCGCA | | |
| Reverse:
GCTTCGATCTGCTCCTGG | 242 | 56 |
| GAPDH | Forward:
GAAGGTGAAGGTCGGAGTCA | | |
| Reverse:
TTCACACCCATGACGAACAT | 402 | 60 |
In vitro cell growth assay
In vitro growth assays were performed with
SW-1990 and Panc-1 cells that were exposed to varying
concentrations of E3330 and/or IWR-1. Briefly, either SW-1990 or
Panc-1 cells were cultured in 96-well plates (4,000 cells/well) for
12 h, treated with E3330 and/or IWR-1 which Diluted with DMSO at
the indicated dose for 48 h, and then the number of viable cells
was determined using the nonradioactive Cell Counting kit-8 (CCK-8;
Dojindo, Kyushu, Japan). All assays were repeated five times.
Western blot analysis
Cells were treated with E3330 and/or IWR-1 for 48 h
and then lysed in RIPA buffer (Thermo Fisher Scientific, Waltham,
MA, USA). Protein concentrations were measured using the BCA
protein assay kit (Thermo Fisher Scientific). Samples were resolved
through a 10% SDS-PAGE gel and transferred to PVDF membranes
(Bio-Rad Laboratories, Hercules, CA, USA). The membrane was blocked
in Tris-buffered saline containing 0.05% Tween-20 with 5% non-fat
skim milk for 1 h at room temperature, and then the membrane was
incubated with primary antibody overnight at 4°C. After three
washes in Tris-buffered saline containing 0.1% Tween-20 (TBST), the
membrane was incubated with horse-radish peroxidase-conjugated
secondary antibody for 1 h at room temperature. After three washes
in TBST, the membrane was visualized by enhanced chemiluminescence
using the ChemiDoc XRS+ imaging system (Bio-Rad Laboratories). The
following antibodies were used: GAPDH, Ape1, β-catenin, cyclin D1
(Cell Signaling Technology, Danvers, MA, USA), and c-myc (Abcam,
Burlingame, CA, USA).
Detection of reactive oxygen species by
flow cytometry
Cells were treated with E3330 and/or IWR-1 for 48 h,
washed with PBS, and resuspended in DMEM. Then, the cells were
incubated in 0.5 μM DCFH-DA (Beyotime Institute of Biotechnology,
Jiangsu, China) for 30 min at 37°C. ROS fluorescence intensity was
determined by flow cytometry with an excitation at 488 nm and an
emission at 525 nm using a FACSCalibur flow cytometer
(Becton-Dickinson, Franklin Lakes, NJ, USA).
Detection of cell cycle by flow
cytometry
Cell cycle analysis was performed on SW-1990 and
Panc-1 cells following a 48-h incubation with E3330 and/or IWR-1.
The cells were fixed in chilled methanol overnight before staining
with 50 μg/ml propidium iodide (Nanjing KeyGen Biotech, Co., Ltd.,
Jiangsu, China) in the presence of 20 μg/ml RNase (Beyotime
Institute of Biotechnology) and 0.1% NP-40 (Sigma). Analysis was
performed immediately after staining using a FACSCalibur flow
cytometer (Becton-Dickinson).
Statistical analysis
Statistical analysis was performed using a GraphPad
Prism v5.0 statistical software package, and a Student’s test was
used to test the probability of significant differences between
samples. The cut-off for statistical significance was set at
P<0.05.
Results
Inhibition of Ape1 redox function with
E3330 suppresses the growth of human pancreatic cancer cell lines
through cyclin D1 downregulation
To determine the function of the Ape1 redox domain
in human pancreatic cancer cells, we treated SW-1990 and Panc-1
cells with E3330, a small molecule inhibitor of the Ape-1 redox
domain. We measured the in vitro growth of SW-1990 and
Panc-1 cells that were exposed to increasing doses of E3330 using
the Cell Counting kit-8 (CCK-8) assay (Fig. 1A). There was a significant
inhibition in the growth of SW-1990 and Panc-1 cells at doses of
E3330 >10 μmol/l. Based on this result, we analyzed the level of
the cell proliferation-related gene cyclin D1, and we found that
cyclin D1 was reduced at both the mRNA and protein level with
increasing concentrations of E3330 (Fig. 1B–E). These alterations in cyclin D1
levels were accompanied by the upregulation of ROS (Fig. 1F).
E3330 treatment upregulates WNT/β-catenin
signaling through upregulation of intracellular ROS levels
E3330 treatment of pancreatic cancer cell lines
resulted in reduced levels of cyclin D1, a well-known WNT/β-catenin
target gene. Therefore, we examined the level of β-catenin, a key
member of the WNT/β-catenin signaling pathway, and another
WNT/β-catenin downstream target gene, c-myc. β-catenin and c-myc
were upregulated at both the mRNA and protein level in an E3330
dose-dependent manner (Fig. 1B–E).
ROS have been shown to function as intracellular messengers to
augment WNT/β-catenin signaling by modulating the redox-dependent
interaction between nucleoredoxin (NRX) and dishevelled (Dvl)
(28,29). Therefore, we hypothesize that E3330
augments WNT/β-catenin signaling by inhibiting the redox function
of Ape1 and upregulating intracellular ROS levels.
The WNT/β-catenin signaling inhibitor
IWR-1 suppresses growth of pancreatic cancer cell lines
IWR-1 is a WNT/β-catenin signaling inhibitor that
induces stabilization of Axin proteins via a direct interaction,
thus leading to enhanced β-catenin destruction (30). To determine the effect of IWR-1 on
the in vitro growth of SW-1990 and Panc-1 cells, we exposed
each cell line to increasing doses of IWR-1 and measured growth
effects using a CCK-8 assay (Fig.
2A). There was a significant inhibition in the growth of both
SW-1990 and Panc-1 cells at doses of IWR-1 >20 μmol/l.
WNT/β-catenin signaling-associated protein levels were examined,
and we found that β-catenin, c-myc and cyclin D1 were decreased
with increasing doses of IWR-1 (Fig.
2D and E). However, the level of β-catenin mRNA was not
altered (Fig. 2B and C).
Combined treatment with E3330 and IWR-1
downregulates cyclin D1 more effectively and blocks G1-to-S
progression
E3330 treatment suppressed the growth of pancreatic
cancer cells and reduced levels of cyclin D1. However, E3330
treatment also increased WNT/β-catenin pathway genes, such as
β-catenin and c-myc. To determine whether inhibition of the
WNT/β-catenin pathway can enhance the growth suppressive effects of
E3330, we treated pancreatic cancer cells with a combination of
E3330 and IWR-1. SW-1990 and Panc-1 cells were exposed to varying
doses of E3330 and IWR-1 in combination and growth effects were
measured using the CCK-8 assay. There was a significant inhibition
in the growth of SW-1990 and Panc-1 cells that were treated with
IWR-1 (15 μmol/l) and E3330 (10 μmol/l) (Fig. 3A). Ape1 and WNT/β-catenin signaling
genes were examined by RT-PCR and western blot analysis. The
upregulation of β-catenin and c-myc that was previously observed
with E3330 treatment was effectively inhibited by the addition of
IWR-1. Cyclin D1 downregulation was also enhanced by the combined
inhibitor treatment, compared with that observed with individual
inhibitor treatments (Fig. 3B–E).
In addition, G1-to-S cell cycle progression was blocked with
decreasing levels of cyclin D1 (Fig.
3G). These data suggest that E3330 and IWR-1 are promising
candidates for a novel combinatorial therapeutic strategy.
Overexpression of Ape1 affects
WNT/β-catenin signaling and cellular ROS levels
To validate the role of Ape1 in WNT/β-catenin
signaling activation, we cloned wild-type Ape1 (WT) and two mutant
Ape1 cDNAs into the pDsRed-N1 expression vector for
overexpression studies (Fig. 4A).
To generate mutant Ape1, we developed a construct with
mutated redox-sensitive cysteines (C65/93A), and a construct with a
20-aa deletion of the putative N-terminal nuclear localization
signal (ΔNLS) of Ape1. Each of these plasmids was
transfected into SW-1990 cells. Ape1 (WT) and Ape1 (C65/93A) were
largely localized to the nucleus, whereas Ape1 (ΔNLS) was largely
localized to the cytoplasm (Fig.
4B). The ROS levels in Ape1 transfected cells were determined
using DCFH-DA and flow cytometry. Overexpression of Ape1 (WT) and
Ape1 (ΔNLS), but not Ape1 (C65/93A), reduced the level of
intracellular ROS (Fig. 4C). We
examined the level of Ape1 and WNT/β-catenin signaling genes by
RT-PCR and western blot analysis. Overexpression of all of the Ape1
constructs in SW-1990 cells resulted in the downregulation of
c-myc, but overexpression of Ape1 (ΔNLS) resulted in the greatest
reduction of c-myc. Cyclin D1 was upregulated in SW-1990 cells
expressing Ape1 (WT) and Ape1 (ΔNLS), but not Ape1 (C65/93A). These
alterations occurred both at the mRNA and protein level (Fig. 4D and E). We believe that c-myc
down-regulation may be due to the ability of Ape1 to cleave
c-myc mRNA (31).
Ape1 is required for inhibiting
WNT/β-catenin signaling
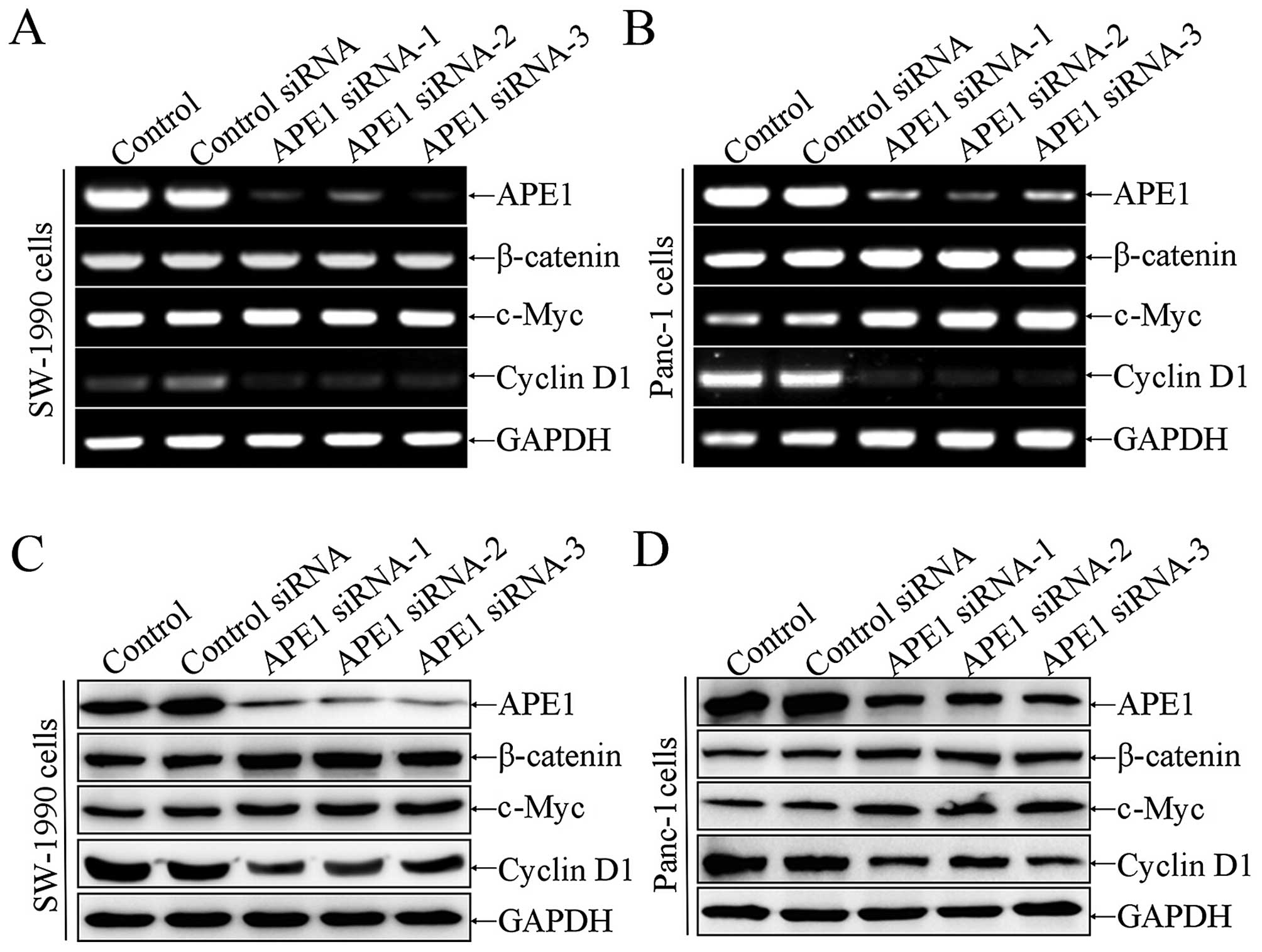
To confirm the function of Ape1 in WNT/β-catenin
signaling, Ape1 was knocked down by siRNA in SW-1990 and
Panc-1 cells. Ape1 and several genes of the WNT/β-catenin pathway
were examined by RT-PCR and western blot analysis, and we found
that β-catenin and c-myc were upregulated at protein level more
than mRNA level (Fig. 5A and B),
whereas cyclin D1 was downregulated both at the mRNA and protein
level (Fig. 5A–D). These data
further suggest that Ape1 is an inhibitor of WNT/β-catenin
signaling.
Ape1 is downregulated by high oxidative
stress
To determine the effect of oxidative stress on Ape1,
SW-1990 and Panc-1 cells were treated with increasing doses of
H2O2 for 2 h, and then cultured for 48 h to
establish a hyperoxia model. ROS levels were upregulated in SW-1990
and Panc-1 cells, but cell viability was reduced as the
concentration of H2O2 increased (Fig. 6A). We performed RT-PCR and western
blot analysis, and unexpectedly found that Ape1 levels decreased as
the concentration of H2O2 increased, at both
the mRNA and protein level. In addition, β-catenin protein, but not
mRNA, was upregulated, c-myc was upregulated at the mRNA and
protein level and cyclin D1 was downregulated (Fig. 6B–E). The cyclin D1 promoter
contains many transcription factor binding sites, including
β-catenin/TCF and NF-κB binding sequences (7,32).
Because Ape1 can activate NF-κB through its redox activity
(33), we believe that cyclin D1
expression is mainly controlled by NF-κB signaling in pancreatic
cancer cells (Fig. 7).
Discussion
Pancreatic cancer is the fourth most common cause of
cancer-related deaths in the United States (34) and the twelfth worldwide. Pancreatic
cancer has an extremely poor prognosis: for all stages combined,
the 1- and 5-year relative survival rates are 25 and 6%,
respectively. Therefore, further study of this disease in cellular
biology and pathophysiology may increase the possibility of finding
new treatments, and consequently, improving the prognosis and
survival of patients. We recently identified aberrant expression of
Ape1 in pancreatic cancer cells (23) and colon cancer stem cells (27). In the present study, we begin to
elucidate the function of Ape1 in pancreatic cancer cells and
demonstrate crosstalk between the Ape1-mediated redox signaling and
WNT signaling pathways. Furthermore, we show that treating
pancreatic cancer cells with a combination of Ape1 and WNT
inhibitors had an enhanced effect on growth inhibition.
Ape1 is a multifunctional protein involved in the
maintenance of genomic integrity and in the regulation of gene
expression. Pursuing Ape1 inhibition as a potential strategy for
cancer cell therapy is justified, based on the following
observations. Ape1 expression and activity are upregulated, or
dysregulated, in many types of cancer, including prostate, bladder,
ovarian, cervical, pancreatic, colon, and non-small cell lung
cancer, as well as germ cell tumors. In addition, our previous
studies demonstrated the role of Ape1 in regulating cancer cell
growth and tumor angiogenesis in both pancreatic cancer (4,23)
and colon cancer stem cells (27).
Because WNT signaling is also important in cancer
cells, we wanted to determine whether Ape1 regulates WNT/β-catenin
signaling in cancer cells. WNT paracrine factors are cysteine-rich
glycoproteins that bind to the Frizzled protein, a transmembrane
receptor. The binding of WNT to its receptors can stimulate at
least three distinct signaling pathways: the β-catenin pathway, the
planar cell polarity (PCP) pathway, and the Ca2+ pathway
(35). β-catenin is a central
component of the WNT pathway, and it forms a complex with members
of the TCF family of transcription factors in the nucleus to
control the transcription of target genes (36). WNT signaling controls critical
biological phenomena throughout development and in adult tissues,
and it is a highly conserved pathway across all species. In
parallel, aberrant WNT signaling underlies a wide range of
pathologies in humans (37).
Aberrant activation of WNT signaling is involved in the development
of several epithelial tumors, including thyroid cancer (38). WNT signaling has been shown to
regulate telomerase in cancer cells, and β-catenin has an important
role in the maintenance of mitochondrial homeostasis (36). Importantly, WNT/β-catenin signaling
is aberrant in pancreatic carcinoma (39). The formation of non-adherent tumor
spheres by human pancreatic cancer cells is associated with the
upregulation of multiple WNT signaling genes, and pancreatic
circulating tumor cells (CTCs) also show enriched expression of WNT
signaling genes (40).
Redox balance underlies cellular homeostasis, and
cancer initiation and progression has been linked to the disruption
of redox balance and oxidative stress (28,41).
In the present study, we demonstrate that ROS can modulate
signaling by the WNT/β-catenin pathway. The present study provides
interesting new insight into crosstalk between redox and
WNT/β-catenin signaling in normal physiology and cancer. The
WNT/β-catenin signaling pathway can be regulated by redox signaling
through the redox-sensitive association of NRX with Dvl (28). NRX is a thioredoxin-related,
redox-regulating protein that inhibits WNT/β-catenin signaling
through Dvl (7). Oxidative stress
inhibits the interaction between NRX and Dvl, which suggests that
treatment with H2O2 may activate
WNT/β-catenin signaling by releasing the NRX-mediated block on Dvl
activity. Therefore, ROS may augment WNT/β-catenin signaling by
modulating the redox-dependent interaction between NRX and Dvl
(7).
Our previous study demonstrated that Ape1 is highly
expressed in pancreatic cancer cell lines, and that inhibition of
Ape1 redox activity significantly inhibits pancreatic cell
proliferation (23). However, the
question of whether Ape1-mediated redox signaling regulated the
WNT/β-catenin pathway in cancer cells remained unclear. Further
questions regarding whether an Ape1 redox inhibitor could act
synergistically with a WNT inhibitor in the growth inhibition of
cancer cells also needed to be addressed. In the present study, we
identified a regulatory role for Ape1 in WNT/β-catenin signaling
through its redox functional domain in pancreatic cancer cells. Our
observations suggest that Ape1 acts as an inhibitor of
WNT/β-catenin signaling, because β-catenin was upregulated when
Ape1 mRNA was depleted by siRNA. Furthermore, E3330
inhibition of the redox function of Ape1 in pancreatic cancer cells
increased intracellular ROS levels and led to the upregulation of
β-catenin. Although E3330 inhibited proliferation of pancreatic
cancer cells, the increased β-catenin upon E3330 treatment could
enhance cancer cell metastasis, which would ultimately result in a
worse prognosis for the patient. Therefore, administration of E3330
alone in the treatment of pancreatic cancer is likely not ideal. We
found that the combination of E3330 and the WNT/β-catenin signaling
inhibitor IWR-1, effectively blocked the upregulation of β-catenin
upon E3330 administration, and enhanced the growth suppression of
pancreatic cancer cells.
In summary, although E3330 inhibited SW1990
pancreatic cancer cell proliferation in a single dose, it also
upregulated β-catenin expression in those cells, which may protect
the cells from further growth inhibition. Therefore, a combination
of an Ape1 and a WNT/β-catenin inhibitor had a stronger effect in
inhibiting pancreatic cell proliferation, indicating that this
inhibitor combination may offer a more promising treatment option
in pancreatic cancer therapy.
Acknowledgements
The present study was supported by the Shanghai
Science and Technology Commision (11ZR1434800).
Abbreviations:
|
AP-1
|
activator protein 1
|
|
Ape1/Ref-1
|
apurinic/apyrimidinic endonuclease
1/redox factor-1
|
|
BER
|
base excision repair
|
|
Cdk5
|
cyclin-dependent kinase 5
|
|
CTCs
|
circulating tumor cells
|
|
Dvl
|
dishevelled
|
|
E3330
|
(2E)-2-[(4,5-dimethoxy-2-methyl-3,6-dioxo-1,4-cyclohexadien-1-yl)methylene]-undecanoic
acid
|
|
EMT
|
epithelial-mesenchymal transition
|
|
eNO
|
endothelial nitric oxide
|
|
HIF-1α
|
hypoxia inducing factor
|
|
IWR-1
|
4-(1,3,3a,4,7,7a-hexahydro-1,3-dioxo-4,7-methano-2H-isoindol-2-yl)-N-8-quinolinyl-benzamide
|
|
NF-κB
|
nuclear factor
kappa-light-chain-enhancer of activated B cells
|
|
NPM1
|
nucleophosmin
|
|
NRX
|
nucleoredoxin
|
|
PCP
|
planar cell polarity
|
|
ROS
|
reactive oxygen species
|
|
TRF2
|
telomeric repeat-binding factor 2
|
|
VEGF
|
vascular endothelial growth factor
|
References
|
1
|
Haeno H, Gonen M, Davis MB, Herman JM,
Iacobuzio-Donahue CA and Michor F: Computational modeling of
pancreatic cancer reveals kinetics of metastasis suggesting optimum
treatment strategies. Cell. 148:362–375. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Fishel ML, Jiang Y, Rajeshkumar NV,
Scandura G, Sinn AL, He Y, Shen C, Jones DR, Pollok KE, Ivan M, et
al: Impact of APE1/Ref-1 redox inhibition on pancreatic tumor
growth. Mol Cancer Ther. 10:1698–1708. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Guillaumond F, Leca J, Olivares O, Lavaut
MN, Vidal N, Berthezène P, Dusetti NJ, Loncle C, Calvo E, Turrini
O, et al: Strengthened glycolysis under hypoxia supports tumor
symbiosis and hexosamine biosynthesis in pancreatic adenocarcinoma.
Proc Natl Acad Sci USA. 110:3919–3924. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zou GM, Karikari C, Kabe Y, Handa H,
Anders RA and Maitra A: The Ape-1/Ref-1 redox antagonist E3330
inhibits the growth of tumor endothelium and endothelial progenitor
cells: Therapeutic implications in tumor angiogenesis. J Cell
Physiol. 219:209–218. 2009. View Article : Google Scholar
|
|
5
|
Cannito S, Novo E, Compagnone A, Valfrè di
Bonzo L, Busletta C, Zamara E, Paternostro C, Povero D, Bandino A,
Bozzo F, et al: Redox mechanisms switch on hypoxia-dependent
epithelial-mesenchymal transition in cancer cells. Carcinogenesis.
29:2267–2278. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zhang Y, Morris JP IV, Yan W, Schofield
HK, Gurney A, Simeone DM, Millar SE, Hoey T, Hebrok M and Pasca di
Magliano M: Canonical wnt signaling is required for pancreatic
carcinogenesis. Cancer Res. 73:4909–4922. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Korswagen HC: Regulation of the
Wnt/beta-catenin pathway by redox signaling. Dev Cell. 10:687–688.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zou GM, Luo MH, Reed A, Kelley MR and
Yoder MC: Ape1 regulates hematopoietic differentiation of embryonic
stem cells through its redox functional domain. Blood.
109:1917–1922. 2007. View Article : Google Scholar
|
|
9
|
Mol CD, Izumi T, Mitra S and Tainer JA:
DNA-bound structures and mutants reveal abasic DNA binding by APE1
and DNA repair coordination (corrected). Nature. 403:451–456. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Luo M, Delaplane S, Jiang A, Reed A, He Y,
Fishel M, Nyland RL II, Borch RF, Qiao X, Georgiadis MM, et al:
Role of the multifunctional DNA repair and redox signaling protein
Ape1/Ref-1 in cancer and endothelial cells: Small-molecule
inhibition of the redox function of Ape1. Antioxid Redox Signal.
10:1853–1867. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Vasko MR, Guo C and Kelley MR: The
multifunctional DNA repair/redox enzyme Ape1/Ref-1 promotes
survival of neurons after oxidative stress. DNA Repair (Amst).
4:367–379. 2005. View Article : Google Scholar
|
|
12
|
Fishel ML, He Y, Reed AM, Chin-Sinex H,
Hutchins GD, Mendonca MS and Kelley MR: Knockdown of the DNA repair
and redox signaling protein Ape1/Ref-1 blocks ovarian cancer cell
and tumor growth. DNA Repair (Amst). 7:177–186. 2008. View Article : Google Scholar
|
|
13
|
Tell G, Quadrifoglio F, Tiribelli C and
Kelley MR: The many functions of APE1/Ref-1: Not only a DNA repair
enzyme. Antioxid Redox Signal. 11:601–620. 2009. View Article : Google Scholar
|
|
14
|
Kelley MR and Fishel ML: DNA repair
proteins as molecular targets for cancer therapeutics. Anticancer
Agents Med Chem. 8:417–425. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Georgiadis MM, Luo M, Gaur RK, Delaplane
S, Li X and Kelley MR: Evolution of the redox function in mammalian
apurinic/apyrimidinic endonuclease. Mutat Res. 643:54–63. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Fishel ML, He Y, Smith ML and Kelley MR:
Manipulation of base excision repair to sensitize ovarian cancer
cells to alkylating agent temozolomide. Clin Cancer Res.
13:260–267. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Madlener S, Ströbel T, Vose S, Saydam O,
Price BD, Demple B and Saydam N: Essential role for mammalian
apurinic/apyrimidinic (AP) endonuclease Ape1/Ref-1 in telomere
maintenance. Proc Natl Acad Sci USA. 110:17844–17849. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Jeon BH, Gupta G, Park YC, Qi B, Haile A,
Khanday FA, Liu YX, Kim JM, Ozaki M, White AR, et al:
Apurinic/apyrimidinic endonuclease 1 regulates endothelial NO
production and vascular tone. Circ Res. 95:902–910. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Vascotto C, Fantini D, Romanello M,
Cesaratto L, Deganuto M, Leonardi A, Radicella JP, Kelley MR,
D’Ambrosio C, Scaloni A, et al: APE1/Ref-1 interacts with NPM1
within nucleoli and plays a role in the rRNA quality control
process. Mol Cell Biol. 29:1834–1854. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Poletto M, Vascotto C, Scognamiglio PL,
Lirussi L, Marasco D and Tell G: Role of the unstructured
N-terminal domain of the hAPE1 (human apurinic/apyrimidinic
endonuclease 1) in the modulation of its interaction with nucleic
acids and NPM1 (nucleophosmin). Biochem J. 452:545–557. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Vascotto C, Lirussi L, Poletto M,
Tiribelli M, Damiani D, Fabbro D, Damante G, Demple B, Colombo E
and Tell G: Functional regulation of the apurinic/apyrimidinic
endonuclease 1 by nucleophosmin: Impact on tumor biology. Oncogene.
33:2876–2887. 2014. View Article : Google Scholar
|
|
22
|
Huang E, Qu D, Zhang Y, Venderova K, Haque
ME, Rousseaux MW, Slack RS, Woulfe JM and Park DS: The role of
Cdk5-mediated apurinic/apyrimidinic endonuclease 1 phosphorylation
in neuronal death. Nat Cell Biol. 12:563–571. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zou GM and Maitra A: Small-molecule
inhibitor of the AP endonuclease 1/REF-1 E3330 inhibits pancreatic
cancer cell growth and migration. Mol Cancer Ther. 7:2012–2021.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wang YT, Tzeng DW, Wang CY, Hong JY and
Yang JL: APE1/Ref-1 prevents oxidative inactivation of ERK for
G1-to-S progression following lead acetate exposure. Toxicology.
305:120–129. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lee HM, Yuk JM, Shin DM, Yang CS, Kim KK,
Choi DK, Liang ZL, Kim JM, Jeon BH, Kim CD, et al:
Apurinic/apyrimidinic endonuclease 1 is a key modulator of
keratinocyte inflammatory responses. J Immunol. 183:6839–6848.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Xiong GS, Sun HL, Wu SM and Mo JZ: Small
interfering RNA against the apurinic or apyrimidinic endonuclease
enhances the sensitivity of human pancreatic cancer cells to
gemcitabine in vitro. J Dig Dis. 11:224–230. 2010.PubMed/NCBI
|
|
27
|
Lou D, Zhu L, Ding H, Dai HY and Zou GM:
Aberrant expression of redox protein Ape1 in colon cancer stem
cells. Oncol Lett. 7:1078–1082. 2014.PubMed/NCBI
|
|
28
|
Funato Y, Michiue T, Asashima M and Miki
H: The thioredoxin-related redox-regulating protein nucleoredoxin
inhibits Wnt-beta-catenin signalling through dishevelled. Nat Cell
Biol. 8:501–508. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tetsu O and McCormick F: Beta-catenin
regulates expression of cyclin D1 in colon carcinoma cells. Nature.
398:422–426. 1999. View
Article : Google Scholar : PubMed/NCBI
|
|
30
|
Chen B, Dodge ME, Tang W, Lu J, Ma Z, Fan
CW, Wei S, Hao W, Kilgore J, Williams NS, et al: Small
molecule-mediated disruption of Wnt-dependent signaling in tissue
regeneration and cancer. Nat Chem Biol. 5:100–107. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Barnes T, Kim WC, Mantha AK, Kim SE, Izumi
T, Mitra S and Lee CH: Identification of Apurinic/apyrimidinic
endonuclease 1 (APE1) as the endoribonuclease that cleaves c-myc
mRNA. Nucleic Acids Res. 37:3946–3958. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Guttridge DC, Albanese C, Reuther JY,
Pestell RG and Baldwin AS Jr: NF-kappaB controls cell growth and
differentiation through transcriptional regulation of cyclin D1.
Mol Cell Biol. 19:5785–5799. 1999.PubMed/NCBI
|
|
33
|
Wu HH, Cheng YW, Chang JT, Wu TC, Liu WS,
Chen CY and Lee H: Subcellular localization of apurinic
endonuclease 1 promotes lung tumor aggressiveness via NF-kappaB
activation. Oncogene. 29:4330–4340. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Hariharan D, Saied A and Kocher HM:
Analysis of mortality rates for pancreatic cancer across the world.
HPB (Oxford). 10:58–62. 2008. View Article : Google Scholar
|
|
35
|
Kikuchi A, Kishida S and Yamamoto H:
Regulation of Wnt signaling by protein-protein interaction and
post-translational modifications. Exp Mol Med. 38:1–10. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Hoffmeyer K, Raggioli A, Rudloff S, Anton
R, Hierholzer A, Del Valle I, Hein K, Vogt R and Kemler R:
Wnt/β-catenin signaling regulates telomerase in stem cells and
cancer cells. Science. 336:1549–1554. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Clevers H and Nusse R: Wnt/β-catenin
signaling and disease. Cell. 149:1192–1205. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Sastre-Perona A and Santisteban P: Role of
the wnt pathway in thyroid cancer. Front Endocrinol (Lausanne).
3:312012.
|
|
39
|
Zeng G, Germinaro M, Micsenyi A, Monga NK,
Bell A, Sood A, Malhotra V, Sood N, Midda V, Monga DK, et al:
Aberrant Wnt/beta-catenin signaling in pancreatic adenocarcinoma.
Neoplasia. 8:279–289. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Yu M, Ting DT, Stott SL, Wittner BS,
Ozsolak F, Paul S, Ciciliano JC, Smas ME, Winokur D, Gilman AJ, et
al: RNA sequencing of pancreatic circulating tumour cells
implicates WNT signalling in metastasis. Nature. 487:510–513. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Grek CL and Tew KD: Redox metabolism and
malignancy. Curr Opin Pharmacol. 10:362–368. 2010. View Article : Google Scholar : PubMed/NCBI
|