Introduction
A prominent role of apoptosis is the suicidal
destruction of dangerous cells, such as cancer cells. Caspase-8
(casp-8) is an initiator caspase, initially identified as a
critical effector of death receptors, present in many normal and
cancerous cell types (1). However,
casp-8 is not always involved in cell death signaling and, in some
cancer cells, its activity seems to work under non-apoptotic
conditions (2). Since cancer cell
growth also depends on evasion of apoptosis, one could argue that,
in cancer cells, caspases are frequently inactivated. Conversely,
in cancer cells, the executioner and initiator caspases are only
rarely mutated in specific types of cancer. In tumors such as colon
carcinoma, which progresses through multiple stages, the loss of
casp-8 is rare. Whereas, in tumors at rapid progression such as
neuroblastoma (3,4), medulloblastoma (5) and glioblastoma (6), as well as in small cell lung
carcinoma (7), lack of casp-8
expression is very frequent. Inactivation of the casp-8 gene was
also reported for human hepatocellular carcinomas (8) and advanced gastric cancer (9).
Breast cancer (BC) is the second leading cause of
cancer death in women worldwide. It is exceeded only by lung
cancer. In year 2015, the estimated number of new cases of invasive
breast cancer expected in the United States is 231,840, and ~40,000
women are expected to die from breast cancer in the same year
(10). Triple-negative breast
cancer (TNBC), that accounts for ~10–20% of all breast cancer cases
(11), is clinically aggressive
form of BC, which do not express estrogen, progesterone and
HER-2/neu receptors (12); as a
consequence, they are unresponsive to endocrine agents or
trastuzumab (13) and are treated
with combined therapies such as surgery, radiation therapy and
chemotherapy (14). Overall, TNBC
represents the poorest prognosis form and patients with TNBC are at
higher risk of early recurrence, mainly in the lungs, brain, and
soft tissue (15). Thus, there is
an urgent need for new therapies.
Recently, we focused our studies on TNBC cells where
the effects of various anticancer drugs were evaluated, showing the
potent cytotoxic effect exerted by parthenolide and by SAHA-TRAIL
and SAHA-parthenolide combination, and also elucidating their
molecular mechanism of action (16–19).
Here, we aimed at determining the role played by casp-8 in TNBC
MDA-MB-231 cells. We evaluated mRNA and protein levels of casp-8
showing that, with respect to the normal human breast epithelial
cell line HMEC, there was an overexpression of casp-8. To further
evaluate the role of casp-8 in MDA-MB-231 cells, we silenced casp-8
using a specific small interfering-RNA (si-RNA). Under these
conditions, we demonstrated that casp-8-knockdown decreased
MDA-MB-231 cell growth rate and delayed G0/G1 to S-phase
transition; moreover, it potently increased the expression of the
cell cycle inhibitors p21 and p27 and those of the
hypophosphorylated-active form of pRb. It also augmented the
migratory and invasive capacity of the cells and the expression of
a number of related genes. Altogether, our results showed that, in
MDA-MB-231 cells, casp-8 played some non-canonical roles which
should be better explored to understand whether it might be
identified as a molecular therapeutic target.
Materials and methods
Cell cultures
The human breast cancer cell line MDA-MB-231 was
purchased from ICLC (Interlab Cell Line Collection, Genova, Italy).
Cells were maintained in DMEM/high glucose, supplemented with 10%
heat-inactivated fetal bovine serum (FBS), L-glutamine (2 mM),
non-essential amino acid solution (1%), penicillin (100 IU/ml) and
streptomycin (100 μg/ml) (Euroclone, Pero, Italy). HMEC (human
mammary epithelial cells) were purchased from Lonza (Lonza,
Euroclone) and maintained according to the manufacturer's
instructions. Cells were incubated in a humidified atmosphere of
95% air plus 5% CO2 at 37°C. Media were replaced every
three days. Cellular morphology was evaluated using a Leica DM IRB
inverted microscope (Leica Microsystems S.r.l., Milan, Italy);
photographs were captured with a computer-imaging system (Leica
DC300F camera).
Pharmacological inhibition of
caspases
MDA-MB-231 cells were seeded in 6-well plates with a
density of 1×105/well in DMEM as described above. After
24 h from seeding, cells were treated with the pan-caspases
inhibitor z-VAD-FMK (50 μM) (Promega, Milan, Italy) or the specific
caspase-8 inhibitor z-IETD-FMK (50 μM) (Santa Cruz Biotechnology,
Santa Cruz, CA, USA) and incubated for 24 h.
Growth curve and cell viability
assays
Total cell number and viability were evaluated by
trypan blue (Sigma-Aldrich) exclusion counting as previously
described (20).
EdU cell proliferation assay
EdU staining was conducted using Click-iT™ EdU Alexa
Fluor imaging kit (Invitrogen, Life Technologies Ltd., Monza,
Italy) according to the manufacturer's protocol. Briefly, cells
were seeded at density of 5×103 cells/well in 96-well
plates and, after silencing procedures, cells were incubated with
10 μM EdU solution for 2 h. Then, cells were washed in PBS, fixed
in 3.7% formaldehyde in PBS for 15 min at RT. After washing with 3%
bovine serum albumin (BSA) in PBS, cells were permeabilized with
0.5% Triton X-100 in PBS for 20 min. Permeabilization buffer was
removed washing cells with 3% BSA in PBS and then incubated with
Click-it reaction cocktail prepared as indicated in instruction's
kit, for 30 min protecting from light. The cells were washed with
3% BSA in PBS. Then, cells were counterstained with 5 μg/ml Hoechst
33342 for 30 min for DNA staining. The slides were washed three
times with PBS and examined under a fluorescent microscope Leica
DM-IRB.
Transient downregulation of casp-8 by
small interfering RNA (siRNA)
Cells were seeded into 6-well plates with a density
of 1×105/well in DMEM-high glucose as previously
described. Transfection procedure was performed 24 h after seeding;
specific siRNAs directed against casp-8 (Santa Cruz Biotechnology)
were transfected for 5 h into the cells at a final concentration of
50 nM, in the presence of 5 μl Lipofectamine 2000 suspension
(Invitrogen, Life Technologies Ltd.) in a final volume of 1 ml
antibiotic- and serum-free DMEM. At the end, the reaction was
stopped replacing the culture medium with DMEM + 10% FBS. Cells
were examined for casp-8 downregulation and other properties, at
the time indicated in the single experiments. siRNA, consisting in
a scramble sequence, was used as a negative control.
Western blot analysis
Cells were detached by trypsinization (0.025%
trypsin-EDTA in PBS) and lysed in RIPA buffer containing protease
inhibitors cocktail (Sigma-Aldrich S.r.l., Milan, Italy) for 30 min
and sonicated three times for 10 sec. Protein concentration was
examined by Bradford Protein Assay (Bio-Rad Laboratories S.r.l.,
Segrate, Milan, Italy). Then, 20 μg of total proteins were resolved
by SDS-PAGE and transferred to a nitrocellulose membrane (Bio-Rad).
Immunodetection analysis was performed using primary antibodies
against caspase-8 (#9746), p21 (#2946), p27 (#2552) by Cell
Signaling Technology, Euroclone. pRb (sc-102), VEGFA (sc-7269),
HMGA2 (sc-30223), KLF4 (sc-20691), CXCR4 (sc-53534), MMP2
(sc-10736) by Santa Cruz Biotechnology. Versican V1 (ab19345) by
Abcam Inc. Prodotti Gianni, Milan, Italy) and the appropriate
horse-radish peroxidase-conjugated secondary antibodies (Santa
Cruz). Immunoreactive signals were detected using enhanced
chemiluminescence (ECL) reagents (Bio-Rad). The correct protein
loading was confirmed by stripping the immunoblot and reprobing
with primary antibody for actin (Sigma-Aldrich). Bands were
visualized and photographed with Chemi Doc XRS (Bio-Rad).
Quantification was performed using Quantity One software.
Flow cytometry analysis
Cell cycle and proliferation analyses were performed
as previously described (21).
RNA extraction and real-time RT-PCR
RNA was extracted with RNeasy mini kit and reverse
transcription reaction was done using QuantiTect Reverse
Transcription kit (Qiagen, Milan, Italy), according to the
manufacturer's instructions. Briefly, 0.8 μg RNA was reverse
transcribed incubating a 20-μl reaction mix in a thermal cycler at
42°C for 25 min. The cDNA was used for quantitative analysis by
real-time PCR using iQ-SYBR Green Supermix (Bio-Rad) and PCR
primers (QuantiTect Primer assay; Qiagen). The primers used were:
casp-8 (QT00052416), P21 (QT00062090), P27 (QT00998445), RB1
(QT00066899), VEGFA (QT01010184), cMYC (QT00035406), CTNNB1
(QT00077882), KLF4 (QT00061033), HMGA2 (QT01157674), CXCR4
(QT00223188). Thermal cycling protocol was programmed as follows:
95°C for 10 min; 95°C for 30 sec, 60°C for 60 sec, 72°C for 30 sec
for 40 cycles and a final extension at 72°C for 5 min. To determine
primer specificity, three stages (95°C for 15 sec, 60°C for 20 sec
and 95°C for 15 sec, with a ramping time of 20 min) were added at
the end of the PCR to obtain dissociation curves for each gene.
Melting curve analyses were done for all samples. No RT control was
included to verify RNA signal specificity, for each gene tested.
All real-time PCR reactions are performed in triplicate. Real-time
PCR and data collection were performed on an IQ5 cycler instrument
(Bio-Rad) qPCR data were analyzed by IQ5 cycler software. The
relative amount of mRNAs was normalized to GAPDH (QT01192646). Data
were calculated using the comparative 2−ΔΔCt method.
Wound healing assay
For the wound healing assay, MDA-MB-231 cells were
transfected with caspase-8-siRNA or scramble siRNA. After 24 h,
transfected cells were harvested, counted and re-seeded into 6-well
plates at the density of 106 cells/well. After 24 h, the
confluent cells were scratched with a 200-μl pipette tip to
generate the wound. One hour before scratching, the medium was
replaced with medium containing 0.1% FBS to minimize the cell
proliferation. Cells were monitored for 24 h post-wounding using
Leica DM-IRB microscope; images were acquired on a Leica DC300F
digital camera using Leica IM50 software. The wound area closure
was determined by ImageJ software and reported as percentage of the
total area of wound as previously described (20).
Transwell migration assay
For the Transwell migration assay, MDA-MB-231 cells
were transfected with casp-8-siRNAorscr-siRNA. After 24 h,
transfected cells were harvested, counted and re-seeded at
1×105 cells/well in the upper chamber of a 6-well
invasion chamber system (BD Biosciences, Discovery Labware,
Becton-Dickinson, Buccinasco, Italy), in serum-free DMEM. DMEM
supplemented with 10% FBS was placed in the bottom well as
chemoattractant. Plates were incubated at 37°C with 5%
CO2, in humidified air for 48 h. Non-migratory cells in
the upper chamber were then removed with a cotton-tip applicator.
Migrated cells on the lower surface were stained with Hoechst 33342
(2.5 μg/ml; Sigma-Aldrich) for 10 min and then visualized under a
fluorescence microscope. The number of migrating cells was
determined by counting in five random fields on each membrane.
Statistical analysis
Data, presented as mean ± SD, were analyzed using
the two-tailed Student's t-test using Microsoft Excel. Differences
were considered significant at P<0.05.
Results
Expression of casp-8 in MDA-MB-231 breast
cancer cells compared with normal human breast epithelial cell line
HMEC
We initially evaluated the levels of casp-8 in
MDA-MB-231 breast cancer cells in comparison with non-transformed
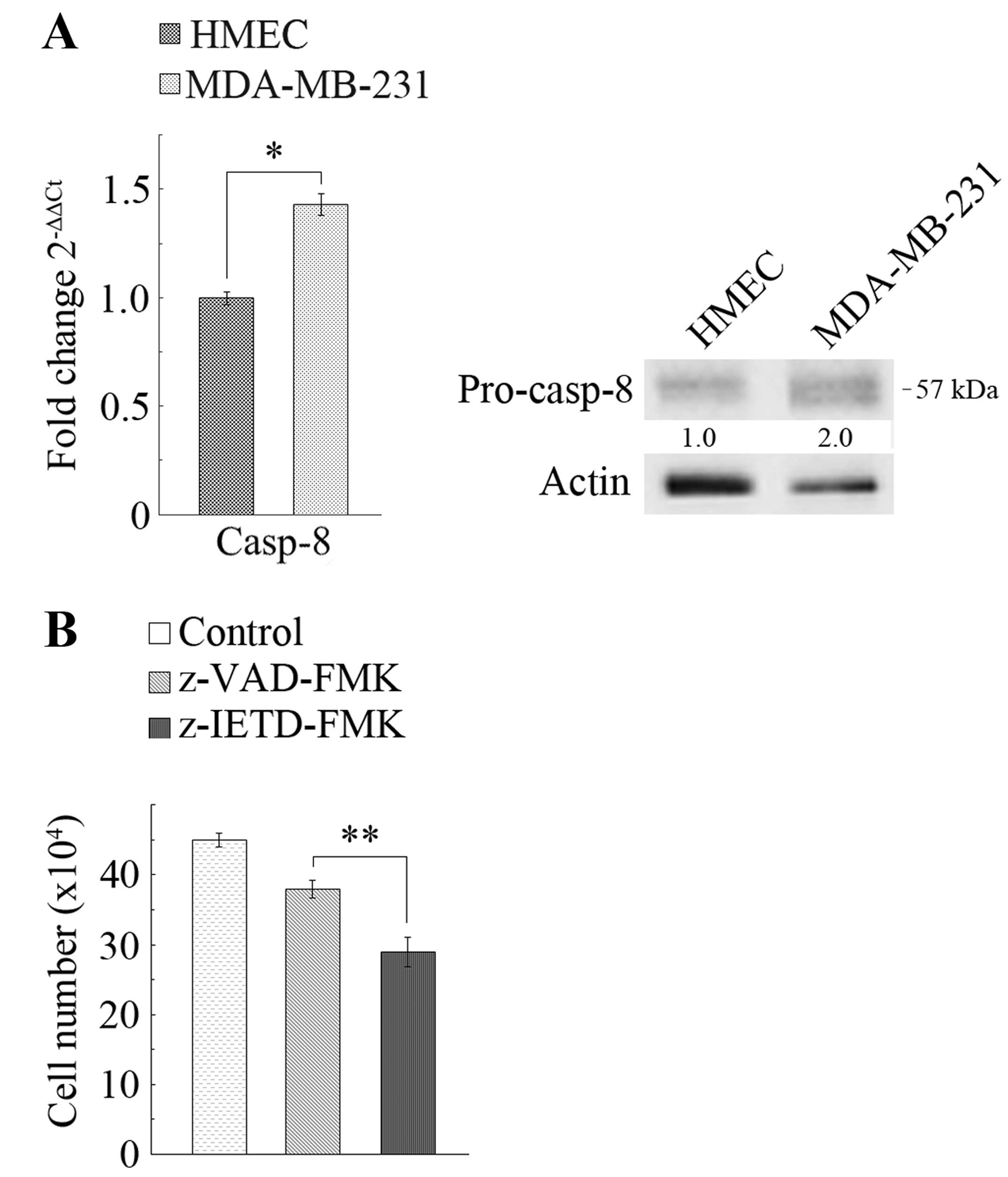
human mammary epithelial cells HMEC. As Fig. 1A shows, casp-8 mRNA was expressed
in both cell lines, however, in MDA-MB-231 cells, the levels were
much higher (1.4-fold) than in HMEC cells. The figure also shows
(western blot analysis) that the levels of casp-8 protein were
significantly higher in MDA-MB-231 than in HMEC cells. As a
preliminary molecular approach to study the involvement of casp-8
in the apoptotic cascade, we evaluated the effects of Z-VAD-FMK, an
irreversible pan-caspase inhibitor, and of Z-IETD-FMK, a specific
casp-8 inhibitor, on MDA-MB-231 cell number. Surprisingly, we
observed (Fig. 1B) that the
treatment determined a decrease in cell number, whereas it did not
induce loss of cell viability (data not shown). In particular,
after 24-h treatment, Z-VAD-Fmk determined a decrease in cell
number of ~15%, while Z-IETD-FMK determined a decrease of ~35%. As
a results of the inhibitory effects on canonical caspase, in
contrast, an increase in cell number was expected, while the
observed decreases suggested that, in TNBC MDA-MB-231 cells, casp-8
could exert unusual roles. To assess such hypothesis, we silenced
casp-8.
Casp-8-knockdown by casp-8-siRNA
As reported in Materials and methods, casp-8 was
transiently depleted by small interfering RNA (si-RNA). After
transfection of the cells with casp-8-siRNA or scrambled siRNA
(scr-siRNA), the level of endogenous casp-8 mRNA was checked.
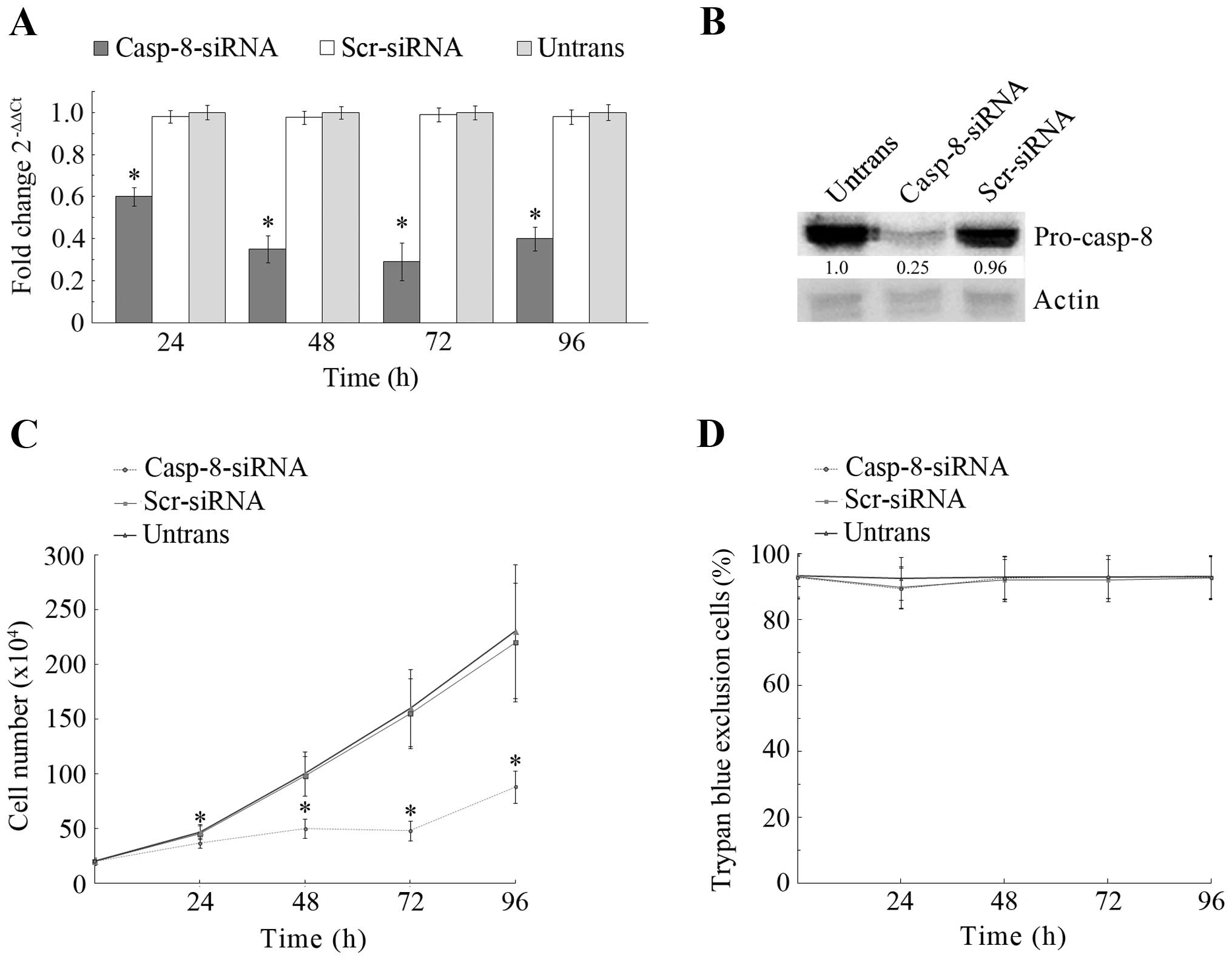
Between 24 and 96 h after casp-8 siRNA transfection, qRT-PCR
(Fig. 2A) shows that, in cells
transfected with casp-8-siRNA, the levels of casp-8 mRNA
progressively lowered with respect to both untransfected cells and
cells transfected with scr-siRNA. The effect of gene silencing was
already evident 24 h after transfection (40% decrease of casp-8
mRNA), increased after 48 h (65%) and reached its maximum after 72
h, when a 71% decrease was observed. Thereafter, casp-8 mRNA level
increased, so that at 96 h after transfection, its decreased value
was ~60%. This suggested that, at that time, the transient
silencing started to recover and that the optimal silencing
efficiency was maintained until 72 h after transfection. Western
blot analysis, which was performed at 72 h after transfection
(Fig. 2B), showed that casp-8
protein decreased by 75%, suggesting that it was reduced similarly
to its mRNA. Importantly, the knockdown of mRNA and protein was
specific as no reduction was observed in cells transfected with
scr-siRNA. Thus, 72 h of casp-8-knockdown was the silencing time
employed for our subsequent experiments.
Casp-8-knockdown strongly reduced
MDA-MB-231 growth rate without affecting cell viability
To evaluate whether casp-8-knockdown modified the
growth rate of MDA-MB-231 cells, untransfected cells and cells
transfected with scr-siRNA or with casp-8 siRNA were analysed (0–96
h) for cell proliferation. Through cell counting, we discovered
(Fig. 2C) that upon casp-8
knockdown, cell number progressively lowered with respect to
untransfected or scr-siRNA-transfected cells. In particular, at 24,
48 and 72 h after transfection, growth rate decreased by 20, 50 and
70% respectively; then, at 96 h, the decrease slightly diminished
(62%) with an overall trend, which strongly agreed with what was
observed for casp-8 mRNA after the casp-8 si-RNA transfection.
Interestingly, the strong reduction in growth rate induced by
casp-8-knockdown did not affect cell viability, as the trypan blue
exclusion assay showed (Fig.
2D).
Casp-8-knockdown delays G0/G1 to S-phase
transition
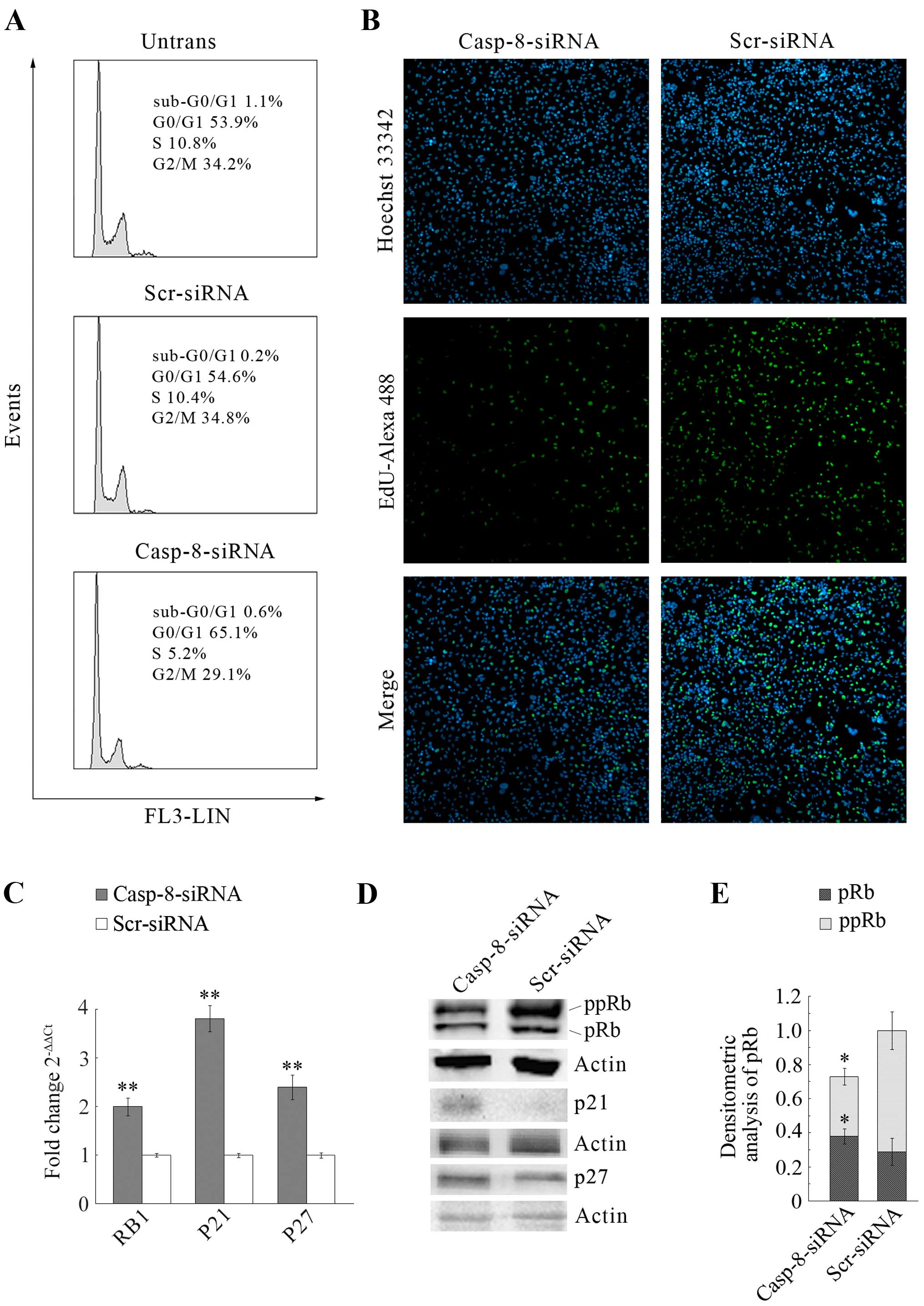
The evaluation of the effect of casp-8-knockdown on
cell cycle, in order to distinguish cells in the different phases
of the cell cycle, was performed by flow cytometric analysis of
cellular DNA content in casp-8 silenced cells. The analysis was
also performed in both untransfected and scr-siRNA-transfected
cells. In Fig. 3A, the DNA
histogram displays that casp-8-knockdown induced a significant
accumulation of cells in G0-G1 phase (11%), with corresponding
decrease in percentage of S phase and G2-M phases.
For all the results obtained so far, both
untransfected and scr-siRNA-transfected samples were used as
control. However, due to the fact that statistically significant
differences (P>0.05) have not been observed between
untransfected cells and cells transfected with the scr-siRNA,
henceforth, only scr-siRNA were used as control. Under these
conditions, cells were microscopically observed at 72 h post casp-8
transfection and analysed for cells in the S-phase of the cell
cycle (EdU positivity). This assay, which allows for a more
accurate detection of DNA synthesis in proliferating cells, showed
that, in agreement with cell cycle study, casp-8-knockdown cells
were strongly less EdU-positive than control cells (Fig. 3B). Overall, these findings indicate
that in MDA-MB-231 cells, casp-8 knockdown consistently reduced
cell proliferation by delaying G0-G1 to S phase transition.
Casp-8-knockdown increases the expression
of key cell cycle regulators
To reveal the molecular mechanism of cell cycle
arrest in the G0/G1 phase, the key molecular protagonists of cell
cycle control were evaluated, particularly, the level of RB1 gene
expression and of p21 and p27, the cyclin-CDK complex inhibitors.
As reported in Fig. 3C,
quantification by qRT-PCR analysis of RB1, p21 and p27, showed
that, in MDA-MB-231 cells, casp-8-knockdown increases their
expression by 2.1, 3.8 and 2.4-fold, respectively. Moreover, by
western blot analysis, the level of the corresponding proteins was
evaluated. As reported in Fig. 3D,
the analysis of the protein pRb allowed us to identify two distinct
molecular forms migrating at different electrophoretic speeds, a
slower form (upper band) and a faster form (lower band). These two
forms might correspond to the hyper-phosphorylated-inactive form
(ppRb) and to the hypo-phosphorylated-active form (pRb),
respectively. As the figure shows, casp-8-knockdown increased the
hypophosphorylated/ active form of the protein, while it strongly
decreased the levels of the hyperphosphorylated/inactive form. The
same figure also shows that, the levels of p21 and p27, similarly
to their mRNA levels, strongly increased after casp-8-knockdown.
Densitometric analysis of the described proteins is reported in
Fig. 3E. Overall, these results
suggest that the delay in G0–G1 to S-phase transition caused by
casp-8 knockdown might arise from the strong upregulation of p21
and p27, with consequent decrease in pRb phosphorylation.
Casp-8 knockdown affects metastatic
capacity of MDA-MB-231 cells
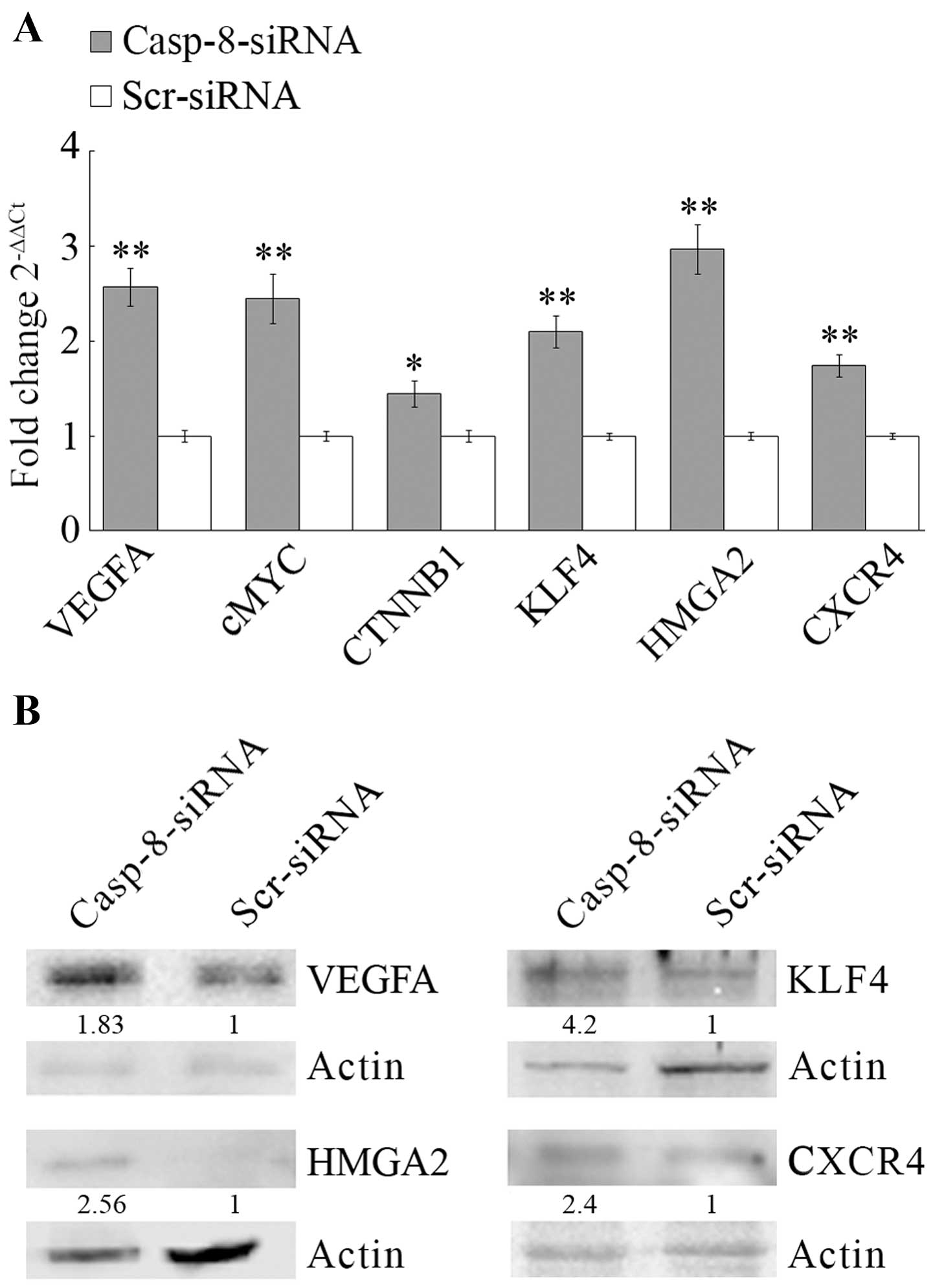
To assess the metastatic capacity of MDA-MB-231
cells, the gene expression profiles of the meta-static markers
after casp-8 knockdown were also measured. As reported in Fig. 4A, the qRT-PCR analysis of a panel
of meta-static markers showed a potent increase in the expression
of all the metastatic stimulator genes studied. In particular,
casp-8 knockdown increased the expression of VEGFA (2.6-fold),
C-MYC (2.4-fold), CTNNB1 (1.4-fold), Krüppel-like factor 4 (KLF4)
(2.1-fold), HMGA2 (2.9-fold), and CXCR4 (1.7-fold). Western blot
analysis of VEGFA, HMGA2, KLF4 and CXCR4, showed similar increases
in protein levels (Fig. 4B).
Casp-8 knockdown strongly influences
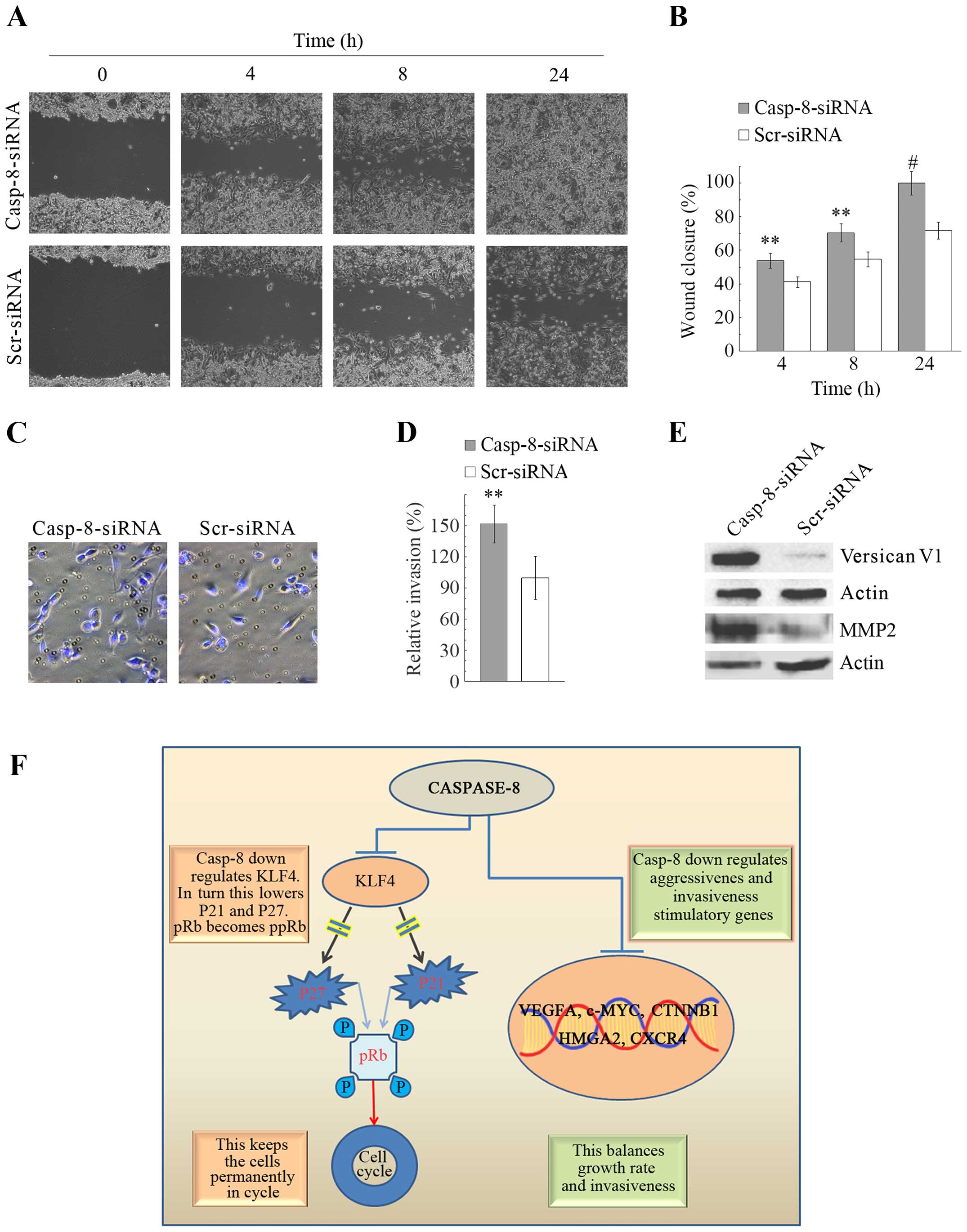
migratory and invasive capacity of MDA-MB-231 cells
To evaluate whether casp-8 knockdown influences the
motility and invasivness of MDA-MB-231 cells, scratch/wound healing
and Matrigel Transwell invasion assays were performed,
respectively. In agreement with the increased metastatic properties
induced by casp-8 knockdown, the data from the wound-healing repair
assay obtained at 4, 8 and 24 h after scratching, proved that, in
comparison with the control cells, casp-8 knockdown cells were
distinctively more migratory, with a wound closure per field of
1.23-, 1.27- and 1.42-fold (P<0.01), respectively (Fig. 5A and B). These results were also
strengthened by the experiments performed with Matrigel, which
showed a significant increase in the invasivity capacity of casp-8
knockdown cells in comparison with the control cells (Fig. 5C and D).
Recently, it has been shown that versican, a large
chondroitin sulfate proteoglycan belonging to the family of
lecticans, strongly promotes cell migration and invasion in a
number of cancer cells (22,23).
Thus, it was pertinent to verify whether such involvement was also
possible in MDA-MB-231 cells. Western blot analysis of the levels
of versican showed that these cells lightly expressed the versican
V1 isoform and that casp-8 knockdown strongly increased its levels
(Fig. 5E). The same figure also
demonstrated a strong increase in the levels of the
metalloproteinase MMP2. Overall, the data demonstrate that casp-8
knockdown strongly increases migratory and metastatic capacities of
MDA-MB-231 cells.
Discussion
Casp-8 plays an important role as an initiator
caspase in apoptosis, therefore, promoting casp-8 expression might
seem good for use in cancer treatment. However, assigning to casp-8
a general antitumor role may be misleading, as dysregulation of
programmed cell death components could result in a switch to a
non-apoptotic signaling pathway to promote tumor growth, survival
and cell migration associated with metastasis (2). Moreover, in tumors, casp-8 is not
generally lost and often, its expression is heterogeneous and
sometimes very elevated (24,25).
Overall, as a number of malignant tumors tolerate very high levels
of casp-8, it seems likely that the apparent upregulation of this
protein in these tumors may represent exploitation of one or more
non-apoptotic roles of casp-8 (25). Thus, before any therapeutic
strategies are hypothesized to increase casp-8 expression, the
reported knowledge must be taken into account.
In this report, we demonstrated that in MDA-MB-231
breast cancer cell line, casp-8 exhibits RNA and protein levels
markedly higher than in the normal human breast epithelial cell
line HMEC.
In the preliminary approach to evaluate the
canonical involvement of casp-8 in apoptosis, we were surprised to
find that treatments with casp-8 inhibitors, rather than causing an
increase in cell number, significantly reduced MDA-MB-231 cell
proliferation. This addressed our study on non-canonical roles
played by casp-8 in these cells and the results obtained have
reconciled this initially unexpected finding by showing that in
these cells apoptosis is minimal.
Here, for the first time, we demonstrated that in
MDA-MB-231 cells, silencing of casp-8 by siRNA induced a potent
decrease in cell growth rate accompanied by a delayed G0/G1 to
S-phase transition. Such a delay was accompanied by a significant
increase in the expression of the key cell cycle inhibitors p21 and
p27 and by increased levels of the hypophosphorylated-active form
of pRb.
It has been reported that during cancer progression,
dysregulation of oncogenic or tumor suppressive transcription
factors, collectively influenced invasion-metastasis cascade
(26). Despite major advances in
understanding the molecular and genetic basis of cancer, metastasis
remains the cause of 90% of cancer-related mortality (27). So, understanding the molecular
mechanisms of metastasis initiation and progression may be critical
for individualizing new therapeutic strategies. Our results showed
that the delayed G0/G1-S-phase transition observed in
casp-8-knockdown MDA-MB-231 cells, was accompanied by a powerful
increase in the expression of a number of factors (VEGFA, C-MYC,
CTNNB1, KLF4, HMGA2, CXCR4, VERSICAN V1 and MMP2) known to strongly
promote cell migration and metastasis in numerous tumors (22,23,26,28).
This could provide MDA-MB-231 cells with particularly high
migratory/metastatic capacity, as suggested by wound healing and
Transwell assays, showing that, in comparison with control cells,
casp-8 knockdown cells were distinctively more migratory, and by
the marked increase in the levels of versican V1, which is known to
strongly promote cell migration and invasion in a number of cancer
cells. In agreement with this result, we also observed a strong
increase in the levels of MMP2, a metalloproteinase, which has been
reported to have a close association with tumor size in 135
patients with infiltrative breast carcinomas (28). Among the above-mentioned
overexpressed factors, KLF4, known to explicate a dual function,
both as a transcriptional activator and repressor, seemed to play
pivotal roles in mediating cell growth inhibition. Indeed, KLF4 may
bind and activate the promoters of the p21 and p27 thus, blocking
cell cycle progression from the G1 phase to S phase (29–32).
Interestingly, in 100 patients with breast cancer, where the
expression of KLF4 was determined by tissue microarray analysis, it
has been shown that KLF4 is a favorable prognostic indicator
(33).
In conclusion, in light of the unusual roles played
by casp-8 in various cancer cell types, and of the results by us
obtained in MDA-MB-231 cells after casp-8-knockdown, we suggest
that in MDA-MB-231 cells, casp-8 could be sucked towards
non-canonical functions. In these cells, casp-8 could be aimed at
maintaining a balance between cell proliferation and invasiveness.
To this aim, downregulating KLF4, which in turn may downregulate
p21 and p27 with consequent hyperphosphorylation and inactivation
of pRb, would be an excellent machinery exploited by casp-8 to keep
MDA-MB-231 cells permanently in cycle. Simultaneously, casp-8 could
tightly control the levels of the stimulatory aggressiveness and
invasiveness as VEGFA, C-MYC, CTNNB1, HMGA2, CXCR4, VERSICAN V1 and
MMP2, so that the cancer cells could balance growth rate and
invasiveness (Fig. 5F). We believe
that the unusual roles played by casp-8 in MDA-MB-231 cells should
be further explored, in order to understand whether and how it
might be identified as a molecular therapeutic target.
Acknowledgements
This study was supported in part by the European
Regional Development Fund, European Territorial Cooperation
2007–2013, CCI 2007 CB 163 PO 037, OP Italia-Malta 2007–2013 and by
the Italian Ministry of Education, University and Research (MIUR)
ex-60%, 2012; Dr D. Carlisi was a recipient of a fellowship granted
by MIUR (contract no. 82, January 23, 2014); Dr R. Di Fiore and Dr
R. Drago-Ferrante were recipients of fellowships granted by the
European Regional Development Fund, European Territorial
Cooperation 2007–2013, CCI 2007 CB 163 PO 037, OP Italia-Malta
2007–2013; Dr M. Montalbano is a PhD student supported by ‘Italian
Ministry of Education, University and Research’ (MIUR); Dr M.
Morreale is a recipient of a contract for research support granted
by the European Regional Development Fund, European Territorial
Cooperation 2007–2013, CCI 2007 CB 163 PO 037, OP Italia-Malta
2007–2013.
References
|
1
|
Kim HS, Lee JW, Soung YH, Park WS, Kim SY,
Lee JH, Park JY, Cho YG, Kim CJ, Jeong SW, et al: Inactivating
mutations of caspase-8 gene in colorectal carcinomas.
Gastroenterology. 125:708–715. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Torres VA, Mielgo A, Barbero S, Hsiao R,
Wilkins JA and Stupack DG: Rab5 mediates caspase-8-promoted cell
motility and metastasis. Mol Biol Cell. 21:369–376. 2010.
View Article : Google Scholar :
|
|
3
|
Stupack DG, Teitz T, Potter MD, Mikolon D,
Houghton PJ, Kidd VJ, Lahti JM and Cheresh DA: Potentiation of
neuroblastoma metastasis by loss of caspase-8. Nature. 439:95–99.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Teitz T, Wei T, Valentine MB, Vanin EF,
Grenet J, Valentine VA, Behm FG, Look AT, Lahti JM and Kidd VJ:
Caspase 8 is deleted or silenced preferentially in childhood
neuroblastomas with amplification of MYCN. Nat Med. 6:529–535.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Pingoud-Meier C, Lang D, Janss AJ, Rorke
LB, Phillips PC, Shalaby T and Grotzer MA: Loss of caspase-8
protein expression correlates with unfavorable survival outcome in
childhood medulloblastoma. Clin Cancer Res. 9:6401–6409.
2003.PubMed/NCBI
|
|
6
|
Skiriute D, Vaitkiene P, Saferis V,
Asmoniene V, Skauminas K, Deltuva VP and Tamasauskas A: MGMT,
GATA6, CD81, DR4, and CASP8 gene promoter methylation in
glioblastoma. BMC Cancer. 12:2182012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hopkins-Donaldson S, Ziegler A, Kurtz S,
Bigosch C, Kandioler D, Ludwig C, Zangemeister-Wittke U and Stahel
R: Silencing of death receptor and caspase-8 expression in small
cell lung carcinoma cell lines and tumors by DNA methylation. Cell
Death Differ. 10:356–364. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Soung YH, Lee JW, Kim SY, Sung YJ, Park
WS, Nam SW, Kim SH, Lee JY, Yoo NJ and Lee SH: Caspase-8 gene is
frequently inactivated by the frameshift somatic mutation
1225_1226delTG in hepatocellular carcinomas. Oncogene. 24:141–147.
2005. View Article : Google Scholar
|
|
9
|
Soung YH, Lee JW, Kim SY, Jang J, Park YG,
Park WS, Nam SW, Lee JY, Yoo NJ and Lee SH: Caspase-8 gene is
inactivated by somatic mutations in gastric carcinomas. Cancer Res.
65:815–821. 2005.PubMed/NCBI
|
|
10
|
American Cancer Society. Surveillance and
Heath Services Research. 2015, http://www.cancer.org/research/cancerfactsstatistics/cancerfactsfigures2015/.
|
|
11
|
Zhang J, Wang Y, Yin Q, Zhang W, Zhang T
and Niu Y: An associated classification of triple negative breast
cancer: The risk of relapse and the response to chemotherapy. Int J
Clin Exp Pathol. 6:1380–1391. 2013.PubMed/NCBI
|
|
12
|
Bauer KR, Brown M, Cress RD, Parise CA and
Caggiano V: Descriptive analysis of estrogen receptor
(ER)-negative, progesterone receptor (PR)-negative, and
HER2-negative invasive breast cancer, the so-called triple-negative
phenotype: a population-based study from the California cancer
Registry. Cancer. 109:1721–1728. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bayraktar S and Glück S: Molecularly
targeted therapies for metastatic triple-negative breast cancer.
Breast Cancer Res Treat. 138:21–35. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yagata H, Kajiura Y and Yamauchi H:
Current strategy for triple-negative breast cancer: Appropriate
combination of surgery, radiation, and chemotherapy. Breast Cancer.
18:165–173. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Pogoda K, Niwińska A, Murawska M and
Pieńkowski T: Analysis of pattern, time and risk factors
influencing recurrence in triple-negative breast cancer patients.
Med Oncol. 30:3882013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
D'Anneo A, Carlisi D, Lauricella M, Puleio
R, Martinez R, Di Bella S, Di Marco P, Emanuele S, Di Fiore R,
Guercio A, et al: Parthenolide generates reactive oxygen species
and autophagy in MDA-MB231 cells. A soluble parthenolide analogue
inhibits tumour growth and metastasis in a xenograft model of
breast cancer. Cell Death Dis. 4:e8912013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Carlisi D, Lauricella M, D'Anneo A,
Emanuele S, Angileri L, Di Fazio P, Santulli A, Vento R and
Tesoriere G: The histone deacetylase inhibitor suberoylanilide
hydroxamic acid sensitises human hepatocellular carcinoma cells to
TRAIL-induced apoptosis by TRAIL-DISC activation. Eur J Cancer.
45:2425–2438. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lauricella M, Ciraolo A, Carlisi D, Vento
R and Tesoriere G: SAHA/TRAIL combination induces detachment and
anoikis of MDA-MB231 and MCF-7 breast cancer cells. Biochimie.
94:287–299. 2012. View Article : Google Scholar
|
|
19
|
Carlisi D, Lauricella M, D'Anneo A,
Buttitta G, Emanuele S, di Fiore R, Martinez R, Rolfo C, Vento R
and Tesoriere G: The synergistic effect of SAHA and parthenolide in
MDA-MB231 breast cancer cells. J Cell Physiol. 230:1276–1289. 2015.
View Article : Google Scholar
|
|
20
|
Di Fiore R, Marcatti M, Drago-Ferrante R,
D'Anneo A, Giuliano M, Carlisi D, De Blasio A, Querques F, Pastore
L, Tesoriere G, et al: Mutant p53 gain of function can be at the
root of dedifferentiation of human osteosarcoma MG63 cells into
3AB-OS cancer stem cells. Bone. 60:198–212. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Di Fiore R, Drago-Ferrante R, Pentimalli
F, Di Marzo D, Forte IM, D'Anneo A, Carlisi D, De Blasio A,
Giuliano M, Tesoriere G, et al: MicroRNA-29b-1 impairs in vitro
cell proliferation, self-renewal and chemoresistance of human
osteosarcoma 3AB-OS cancer stem cells. Int J Oncol. 45:2013–2023.
2014.PubMed/NCBI
|
|
22
|
Gorter A, Zijlmans HJ, van Gent H, Trimbos
JB, Fleuren GJ and Jordanova ES: Versican expression is associated
with tumor-infiltrating CD8-positive T cells and infiltration depth
in cervical cancer. Mod Pathol. 23:1605–1615. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ricciardelli C, Sakko AJ, Ween MP, Russell
DL and Horsfall DJ: The biological role and regulation of versican
levels in cancer. Cancer Metastasis Rev. 28:233–245. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Jäger R and Zwacka RM: The enigmatic roles
of caspases in tumor development. Cancers (Basel). 2:1952–1979.
2010. View Article : Google Scholar
|
|
25
|
Stupack DG: Caspase-8 as a therapeutic
target in cancer. Cancer Lett. 332:133–140. 2013. View Article : Google Scholar
|
|
26
|
Ell B and Kang Y: Transcriptional control
of cancer metastasis. Trends Cell Biol. 23:603–611. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Weigelt B, Peterse JL and van ‘t Veer LJ:
Breast cancer metastasis: Markers and models. Nat Rev Cancer.
5:591–602. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
28
|
Nakopoulou L, Tsirmpa I, Alexandrou P,
Louvrou A, Ampela C, Markaki S and Davaris PS: MMP-2 protein in
invasive breast cancer and the impact of MMP-2/TIMP-2 phenotype on
overall survival. Breast Cancer Res Treat. 77:145–155. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tiwari N, Meyer-Schaller N, Arnold P,
Antoniadis H, Pachkov M, van Nimwegen E and Christofori G: Klf4 is
a transcriptional regulator of genes critical for EMT, including
Jnk1 (Mapk8). PLoS One. 8:e573292013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Shi Y and Ai W: Function of KLF4 in stem
cell biology. Biochemistry, Genetics and Molecular Biology
‘Pluripotent Stem Cells’. Bhartiya D and Lenka N: InTech; 2013
|
|
31
|
Rowland BD, Bernards R and Peeper DS: The
KLF4 tumour suppressor is a transcriptional repressor of p53 that
acts as a context-dependent oncogene. Nat Cell Biol. 7:1074–1082.
2005. View
Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wei D, Kanai M, Jia Z, Le X and Xie K:
Kruppel-like factor 4 induces p27Kip1 expression in and suppresses
the growth and metastasis of human pancreatic cancer cells. Cancer
Res. 68:4631–4639. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Nagata T, Shimada Y, Sekine S, Hori R,
Matsui K, Okumura T, Sawada S, Fukuoka J and Tsukada K: Prognostic
significance of NANOG and KLF4 for breast cancer. Breast Cancer.
21:96–101. 2014. View Article : Google Scholar
|