Introduction
Non-small cell lung carcinoma (NSCLC) is a leading
cause of cancer death worldwide, and represents close to 90% of all
lung cancers with a poor prognosis. The progression of lung cancer
is a complex and multistep process where several mechanisms such as
transformation, hypoxia, invasion, migration and metastasis are
known to be the main hallmarks, especially metastasis, which is
responsible for 90% of cancer-related mortality (1). Clinical data have shown that most
lung cancer patients eventually suffer relapse and/or metastasis
after complete excision of the cancer, even if they were at stage
I-A (2). Research on NSCLC
metastasis has recently expanded to tumor-microenvironment, which
include the stromal cellular compartment and extra-cellular matrix
components. Metastasis (from initial angiogenesis, intravasation,
to extravasate into a distant tissue) is an inefficient process and
few released cancer cells complete the entire process,
micro-environmental interactions assist each of these steps
(3). It is necessary to better
understand the mechanisms by which tumor cells co-operate with the
micro-environment.
Together with other immune cells, tumor cells and
interstitial cytokines, the tumor associated macrophages (TAMs) can
be brought to form a tumor immune microenvironment (4). TAMs refer to a kind of macrophages
which are migrated and infiltrated in local tumor during the
occurrence and development of tumors (5). TAMs are known to be crucial cells in
lung cancer as they are in close proximity to tumor cells compared
to other stromal cells (1).
According to the activation type of macrophage, it can be divided
into two main types M1 and M2. It is generally considered that
these two types of macrophages are antagonistic (6,7). M1
phenotype is activated by interferon (IFN)-γ, LPS and tumor
necrosis factor (TNF)-α (8,9).
This phenotype is associated with the expression of interleukin
(IL)-12, TNF-α and inducible nitric oxide synthase (iNOS)(10,11)
and also with extended survival time in NSCLC patients (12). M2 phenotype possesses effects of
inflammation inhibition by producing anti-inflammatory cytokines
such as IL-4 and IL-10 (7,11,13).
This phenotype can promote tumor growth and sustain tumor survival
(14). They also play a vital role
in increasing angiogenesis via VEGF, which is a prominent mediator
of angiogenesis (15). Given the
above, the inducing of M1 and inhibition of M2 polarization may
help alleviate the metastasis of NSCLC.
Puerarin
[4H-1-benzopyran-4-one,8-β-D-glucopyranosyl-7-hydroxy-3-(4-hydroxyphenyl),
C21H20O9] is the major bioactive
ingredient isolated from the root of traditional Chinese medicine
Ge-gen (Radix Puerariae, RP) (16). Due to its wide spectrum of
pharmacological properties (e.g., cardioprotection,
neuroprotection, antioxidant, anticancer, alleviating pain and
inhibiting alcohol intake), puerarin has been widely used in the
treatment of various deseases, including cardiovascular, diabetes,
Parkinson's disease, endometriosis and cancer (17). The antitumor activity of puerarin
has been reported on human lung carcinoma A549 cell line (18). However, the effect of puerarin on
NSCLC metastasis has rarely been reported.
The present study applied puerarin in the context of
NSCLC, we found that puerarin reduced the tumor growth in NSCLC
xenograft model. Besides, puerarin acted directly on macrophages to
inhibit macrophage polarized to M2 phenotype and suppressed cell
invasion and migration. These inhibition effects may function
through inactivating MEK/ERK 1/2 pathway. Taken together, our
results suggest that puerarin may provide novel insight into the
mechanism and treatment of NSCLC.
Materials and methods
NSCLC xenografts
All the animals involved in the present study were
purchased from the the Institute of Zoology, Chinese Academy of
Medical Sciences (NOD/SCID mice, clean, 8-week-old and weighing
20–22 g). Human NSCLC cells A549, obtained from the American Type
Culture Collection (ATCC; Manassas, VA, USA), were digested by the
pancreatic enzymes and the final concentration was adjusted to
1×106/ml. On day 0, the mice (n=50) were narcotized with
200 l of 0.75% sodium pentobarbital solution per mouse and the
subcutaneous injection were conducted of 5×106 A549
cells. The mice in the treatment group (n=25) were injected with
puerarin (40 mg/kg body weight) every other day, while the mice
(n=25) in model group received the same volume injection of
phosphate-buffered saline (PBS). After the development of a
palpable tumor, the tumor volume was monitored every 6 days,
briefly, tumor was isolated from five mice in each group, the
volume was assessed by the following formula: tumor volume
(mm3) = maximal length (mm) × perpendicular width
(mm)2/2. All mice were assigned to euthanasia at the end
of the measurements. On day 30, tumor tissues in model mice and
puerarin treating mice were collected. All animal experiments were
performed according to the current guidelines and under a protocol
approved by the Institutional Animal Care and Use Committee.
Flow cytometry
Macrophages from local tumor tissue in model mice
were considered as NSCLC model group; macrophages from lung tissues
in healthy mice were considered as healthy control group;
macrophages from the tumor tissue in puerarin treating mice were
regarded as NSCLC model + puerarin group. The suspensions of
macrophages from each group were prepared by grinding the organ
through a 40-mm nylon mesh in medium. For cell surface staining,
cells were stained for 20 min at room temperature (RT) in 1%
BSA-PBS buffer with the following panel of antibodies: Alexa Fluor
488-anti-F4/80 (Serotec, Oxford, UK) and PE-CD197, PE-iNOS,
PE-CD40, PE-CD206, PE-Arg-1 and PE-CD163 (eBioscience, San Diego,
CA, USA), followed by flow cytometry (BD Biosciences, San Jose, CA,
USA). Data were analyzed using CellQuest software.
Western blotting
Macrophages were lysed in lysis buffer (Beyotime
Institute of Biotechnology, Haimen, China) supplemented with 1 mM
phenylmethanesulfonyl fluoride (PMSF). The protein concentration
was determined using the BCA protein assay (Tiangen Biotech, Co.,
Ltd., Beijing, China). Twenty micrograms of protein in each sample
was separated by 12% SDS-PAGE and electro-transferred to
polyvinylidene fluoride (PVDF) membranes (Millipore, Billerica, MA,
USA) for immunoblotting. The following primary antibodies were
used: anti-IFN-γ (1:1,000, ab175878; Abcam), anti-TNF-α (1:1,000,
ab6671; Abcam), anti-IL-12 (1:500, ab9992; Abcam), anti-IL-10
(1:500, ab34843; Abcam), anti-IL-1β (Santa Cruz Biotechnology,
Santa Cruz, CA, USA), anti-TGF-β (1:500, ab66043; Abcam), anti-VEGF
(1:1,000, ab32152; Abcam), anti-MMP-9 (1:100, ab73734; Abcam),
anti-ICAM-1 (1:20, ab20; Abcam), anti-MEK (1:20,000, ab178876;
Abcam), anti-p-MEK (1:500, ab194754; Abcam), anti-ERK1/2 (1:100,
ab54230; Abcam), anti-p-ERK1/2 (Santa Cruz Biotechnology) and
anti-GAPDH (1:500, ab8245; Abcam), which was used as the internal
reference. After incubation with the appropriate horseradish
peroxidase (HRP)-conjugated secondary antibody, proteins were
detected using a ChemiDoc XRS imaging system and Quantity One
analysis software (Bio-Rad Laboratories, San Francisco, CA,
USA).
Preparation of purified macrophages
Macrophages were prepared as previously reported
(19). Macrophages from tumor
tissue in model mice were seeded in 6-well tissue culture plates
(2×106 cells/ml; Corning, Inc., Corning, NY, USA) and
allowed to adhere for 1 h. Adherent cells were purified by positive
selection using CD68 MicroBeads (Miltenyi Biotec, Leiden, The
Netherlands). The purity of CD68+ cells was evaluated by
flow cytometry (CD68+F4/80+ cells >96%).
The purified macrophages were incubated with puerarin (40
µM) for 48 h. The number of M2 phenotype (Arg-1+)
was measured by flow cytometry. The level of M2 cytokines (IL-10,
IL-4 and TGF-β) was detected by western blot analysis.
Transwell invasion assay
Transwell membranes coated with Matrigel
(Becton-Dickinson, Franklin Lakes, NJ, USA) were used to assay
invasion of NSCLC cells in vitro. A549 cells pretreated with
or without puerarin (40 µM) for 24 h were plated at
2×104/well in the upper chamber in serum-free medium,
20% fetal bovine serum (FBS) was added to the medium in the lower
chamber. After incubating for 24 h, non-invading cells were removed
from the top well with a cotton swab, while the bottom cells were
fixed in 95% ethanol, stained with hematoxylin. The cell numbers
were determined by counting the penetrating cells under a
microscope at ×200 magnification on 10 random fields in each well.
Each experiment was performed in triplicate.
Wound healing assay
The CytoSelect 24-Well Wound Healing assay (Cell
Biolabs, Inc., San Diego, CA, USA) was used to analyze the
migration of NSCLC cells. The assay was performed according to the
manufacturer's recommendations using 2×103 A549 cells
pre-treated with or without puerarin (40 µM) for 24 h/well.
Image acquisition of wound fields was done after the removal of
inserts (0 h) and wound closure documentation was completed after
24 h with a phase-contrast microscope (Leica DM IL; Leica
Microsystems, Wetzlar, Germany) equipped with a digital camera
(Leica DFC300 FX). Image analysis was conducted with Adobe
Photoshop CS7 software.
Immunofluorescence staining
Purified macrophages (2×103 cells/well)
were cultured on 8-well chamber CultureSlides (Becton-Dickinson,
Bedford, MA, USA). After 8 h, cells were fixed in 3%
paraformaldehyde in PBS at room temperature for 8 min, then
permeabilized with 0.2% Triton X-100 for 15 min at room
temperature. After washing in PBS, the cells were incubated with
primary mouse anti-ERK monoclonal antibody (1 mg/ml; Transduction
Laboratories, Lexington, KY, USA) at 4°C overnight. After washing,
cells were incubated with biotinylated goat anti-mouse IgG (Pierce,
Rockford, IL, USA) at room temperature for 1 h. The
immunoreactivity was revealed using Alexa568-conjugated
streptavidin (Molecular Probes, Eugene, OR, USA) and cells were
counterstained with 10 mg/ml DAPI. The cells were examined under a
Nikon fluorescence microscope (Image Systems, Columbia, MD,
USA).
Statistical analysis
All results are presented as mean ± SD from a
minimum of three replicates. Differences between the groups were
evaluated by the SPSS version 15.0 statistical software with the
Student's t-test when comparing only two groups or assessed by
one-way ANOVA when more than two groups were compared. Differences
were considered statistically significant at P<0.05.
Results
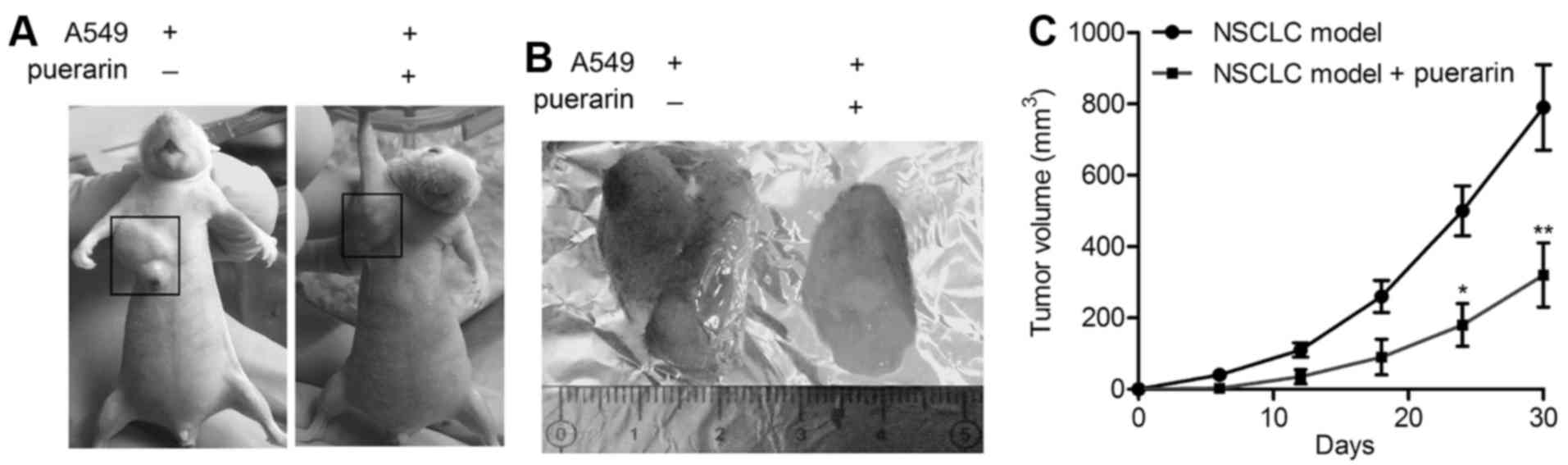
Puerarin reduces tumor growth of NSCLC
xenograft model
NSCLC xenograft model was established by
subcutaneous injection of A549 cells into NOD/SCID mice. The
representative images of NSCLC xenograft model and isolated tumors
are shown in Fig. 1A and B. The
tumor growth was slower in puerarin group compared with model
group, and the tumor volumes were reduced significantly in NSCLC
mice under treatment with puerarin (P<0.05; Fig. 1C). The results displayed the
antitumor effect of puerarin in the NSCLC xenograft model.
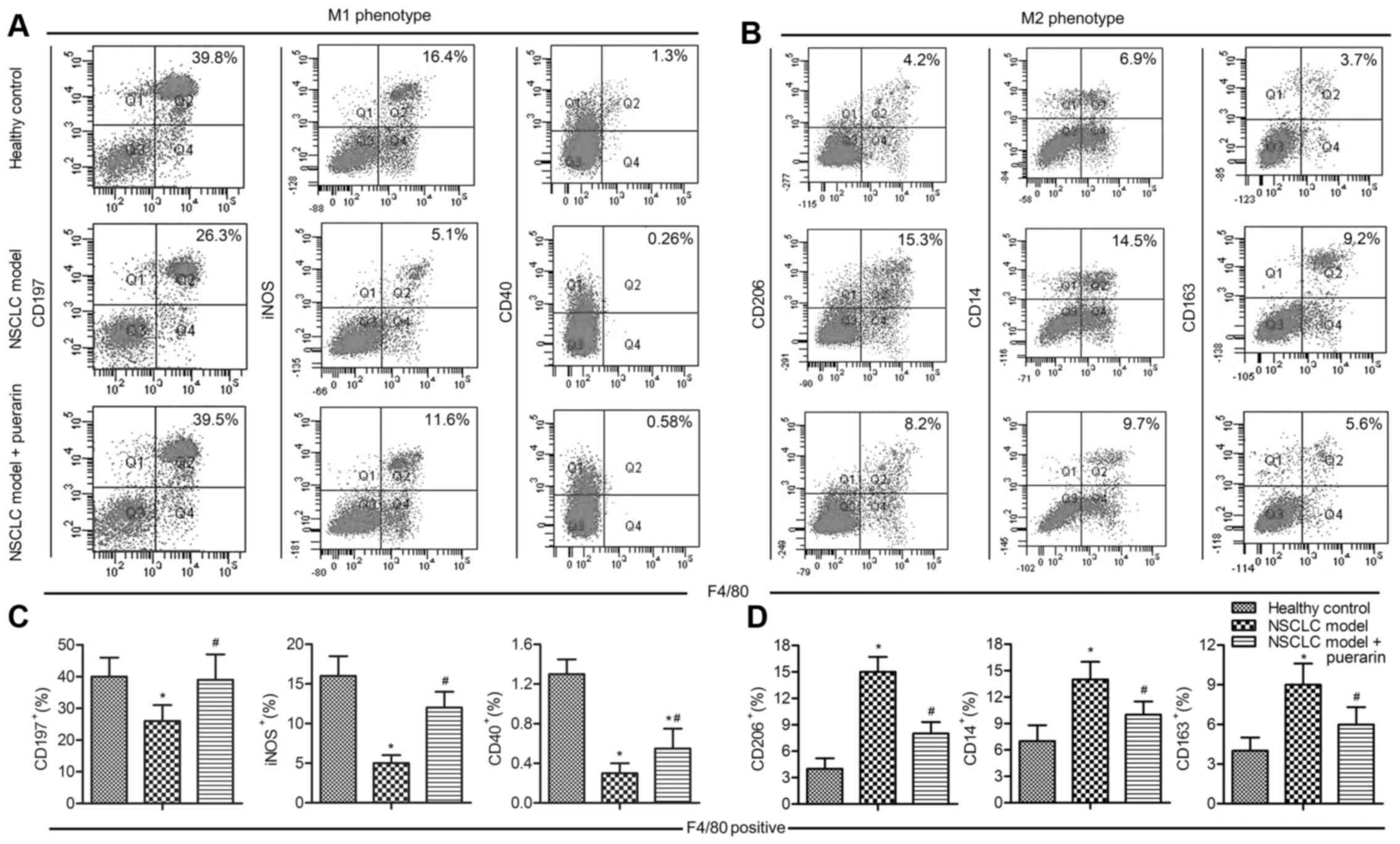
Puerarin inhibits macrophages polarized
to M2 phenotype
TAMs are abundant components of NSCLC, the
polarization status (M1/M2) has pro-inflammatory or pro-tumoral
properties (20). We next explored
whether the antitumor role of puerarin was involved in regulating
macrophage polarization. The proportions of M1 markers
(CD197+, iNOS+ and CD40+) and M2
markers (CD206+, Arg-1+ and
CD163+) were detectd by flow cytometry. The results
showed that M1 markers were decreased in model group compared with
the control group (P<0.05), and the treatment with puerarin in
NSCLC model elevated M1 markers (P<0.05; Fig. 2A and C). On the contrary, M2
markers (CD206+, Arg-1+ and
CD163+) were increased in model group compared with the
control group (P<0.05), but strongly decreased adding puerarin
in NSCLC model (P<0.05; Fig. 2B and
D). These results suggested that puerarin exert antitumor
effect via inhibiting macrophage polarized to M2 phenotype.
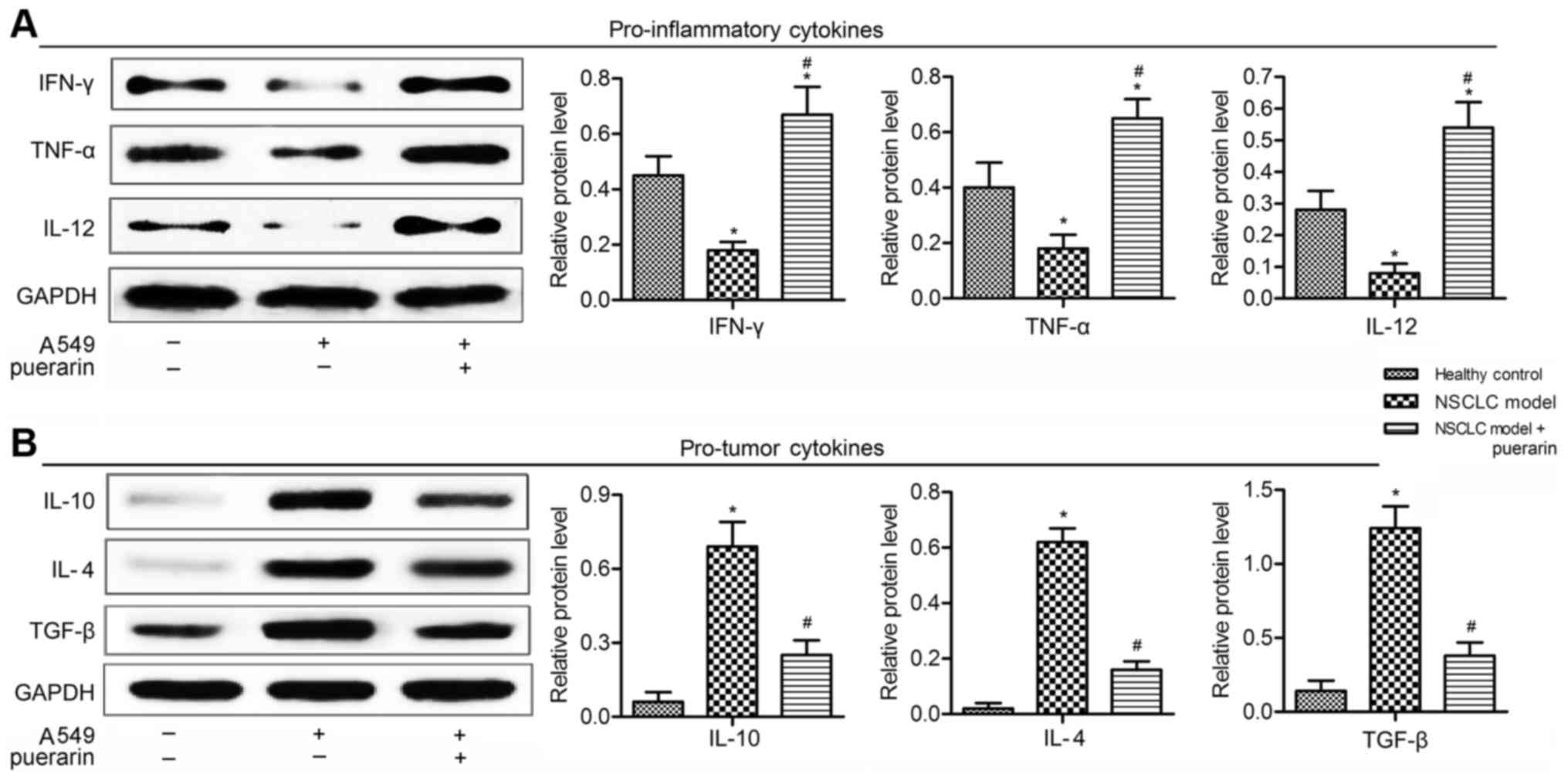
Puerarin increases pro-inflammatory
cytokines and decreases pro-tumor cytokines
Polarized macrophages differ in terms of effector
function, cytokine and chemokine production (21). Here, the expression level of
cytokines in NSCLC xenograft model was detected by western blot
analysis. Levels of pro-inflammatory cytokines (IFN-γ, TNF-α and
IL-12) were decreased in model group compared with the control
group (P<0.05), but were promoted in puerarin group compared to
the model group (P<0.05; Fig.
3A). The expression of tumorigenesis-related cytokines (IL-10,
IL-4 and TGF-β) was elevated in model group in comparison with the
control group (P<0.05), but the levels were significantly
reduced in NSCLC model treated with puerarin (P<0.05; Fig. 3B). These results indicated puerarin
exhibited antitumor effect by inflammatory cytokines and decreased
tumorigenesis-related cytokines.
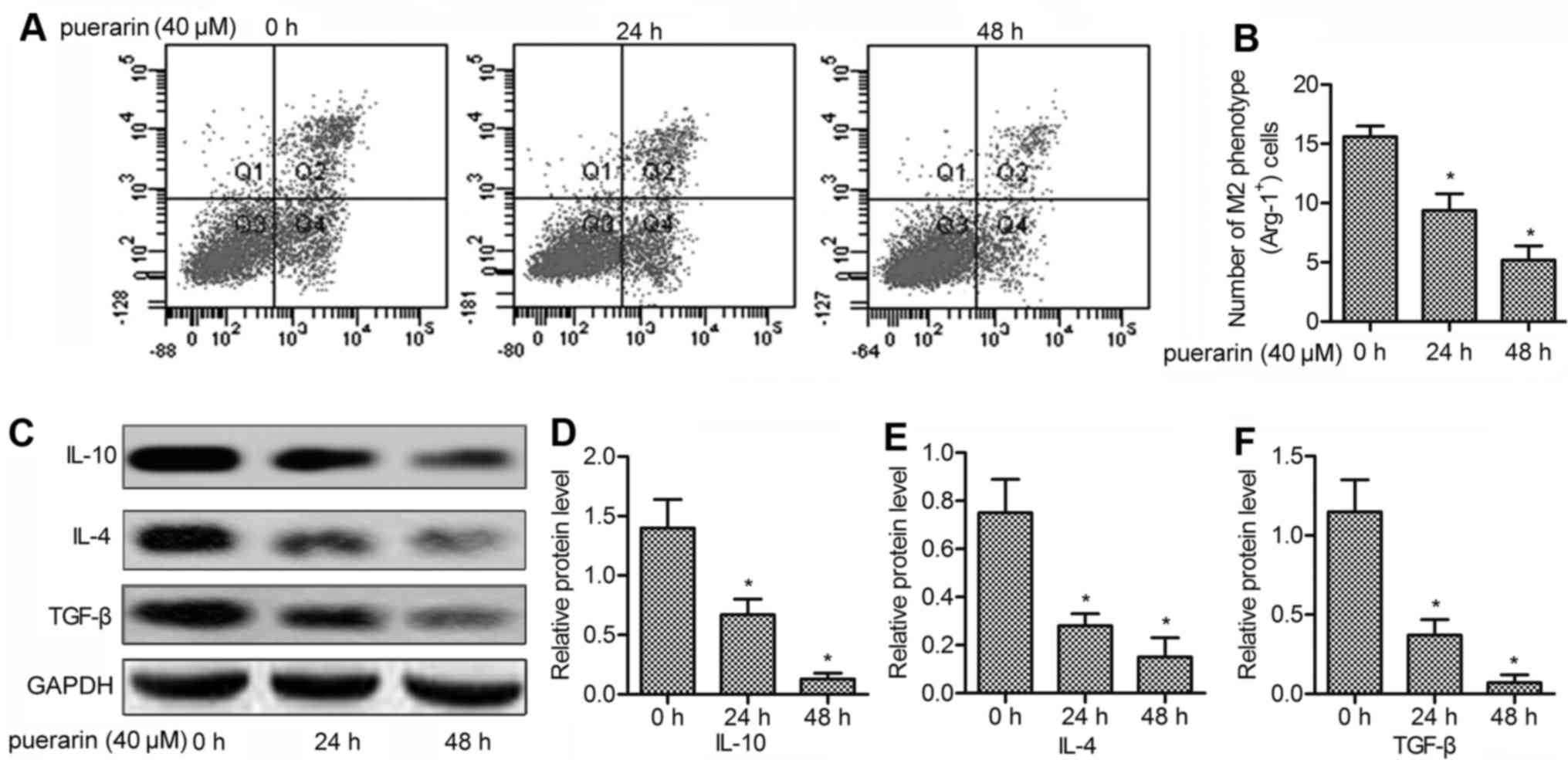
Puerarin acts directly on macrophages to
inhibit M2 polarization
To explore whether puerarin directly acts on
macrophages, we purified macrophages from model mice. An in
vitro experiment was conducted to investigate the role of
puerarin on M2 polarization. The results showed that the number of
M2 phenotype (Arg-1+) was significantly reduced under
treatment with puerarin for 24 h (P<0.05; Fig. 4A), and the number was further
decreased after 48 h (P<0.05; Fig.
4B). To confirm the inhibitory effect of puerarin on M2
polarization, the level of M2 cytokines (IL-10, IL-4 and TGF-β) was
detected by western blot analysis. The results displayed that the
expression of IL-10, IL-4 and TGF-β was strongly inhibited adding
puerarin for 24 and 48 h (P<0.05; Fig. 4C–F). Taken together, our results
clearly demonstrated that puerarin inhibited macrophages polarized
to M2 phenotype and did not require the participation or the
auxiliary of other cells.
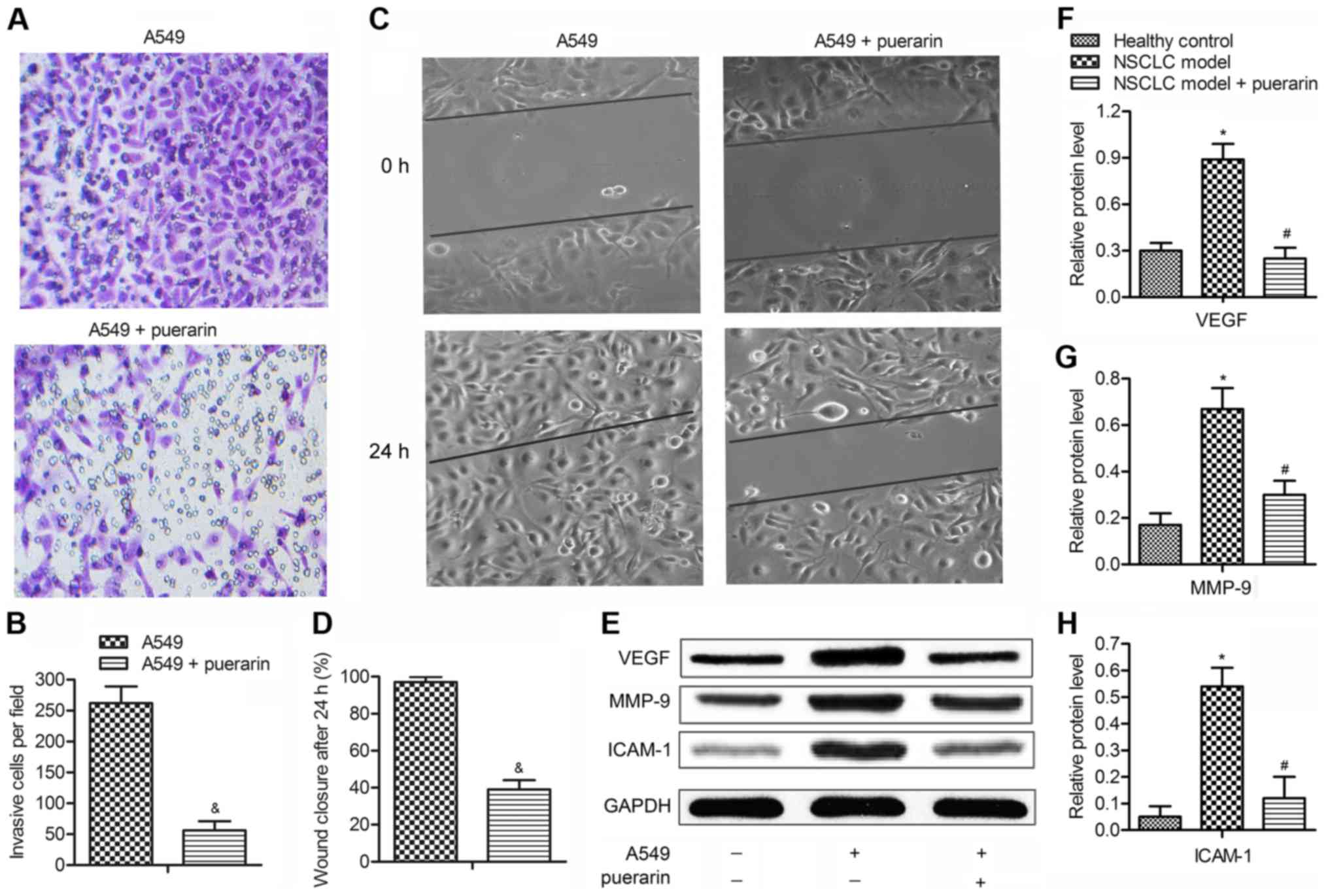
Puerarin suppresses the invasion and
migration of NSCLC cells
A report indicated that high density of M2
macrophages is associated with the metastasis in NSCLC patients
(22). The effect of puerarin on
the motility of NSCLC cells (A549) was measured by Transwell assays
and scratch assays. The number of invasive cells was decreased by
2.5 times adding puerarin (40 µM) in A549 cells (P<0.05;
Fig. 5A and B). The results of
scratch assays agreed with the Transwell assays. The A549 group
showed a complete closure of the gap, whereas puerarin increased
the gap by ~50% compared with model group (P<0.05; Fig. 5C and D). These observations
suggested that puerarin was a negative metastatic regulator of
NSCLC. The expression of tumor metastasis-related proteins (VEGF,
MMP-9 and ICAM-1) were then determined by western blot analysis
(Fig. 5E). The results indicated
that the levels of the three proteins were increased in model group
compared with the control group (P<0.05), but puerarin
significantly restrained their expression compared with the model
group (P<0.05; Fig. 5F–H). The
suppression of puerarin on migration-related protein expressions
further confirming the inhibition effect of puerarin on the
metastasis of NSCLC.
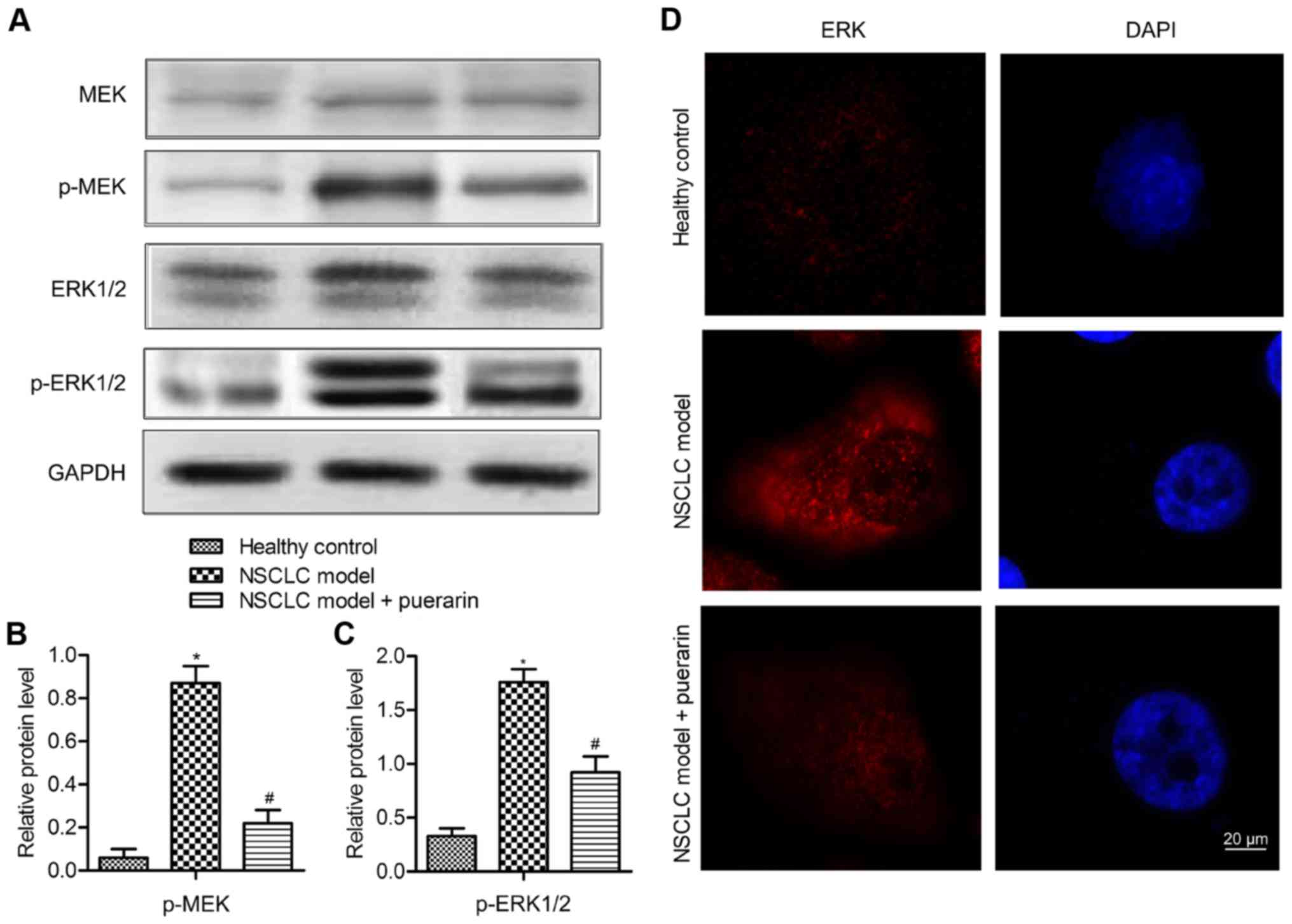
Puerarin restrains the activation of
MEK/ERK1/2 pathway
To investigate the mechanism of puerarin on the
metastasis of NSCLC, the level of MEK, ERK1/2 and their
phosphorylated forms was measured by western blot analysis
(Fig. 6A). The results displayed
that the expression of p-MEK and p-ERK 1/2 was enhanced in model
group compared with the control group (P<0.05), indicating that
the MEK/ERK 1/2 pathway was activated in the NSCLC model. But the
level of p-MEK and p-ERK 1/2 was reduced significantly in NSCLC
model treated with puerarin (P<0.05) (Fig. 6B and C). To verify the inactivating
role of puerarin, the subcellular localization of ERK was measured.
Cells in the control group showed membrane ERK staining with
minimal cytoplasmic or nuclear staining, the model group displayed
predominantly nuclear ERK staining. However, the cells in the
puerarin group displayed reduced cytoplasmic and nuclear staining
of ERK as compared with the model group (Fig. 6D). These results indicated that
puerarin restrained the activation of MEK/ERK 1/2 pathway through
inhibition of the ERK nucleus translocation.
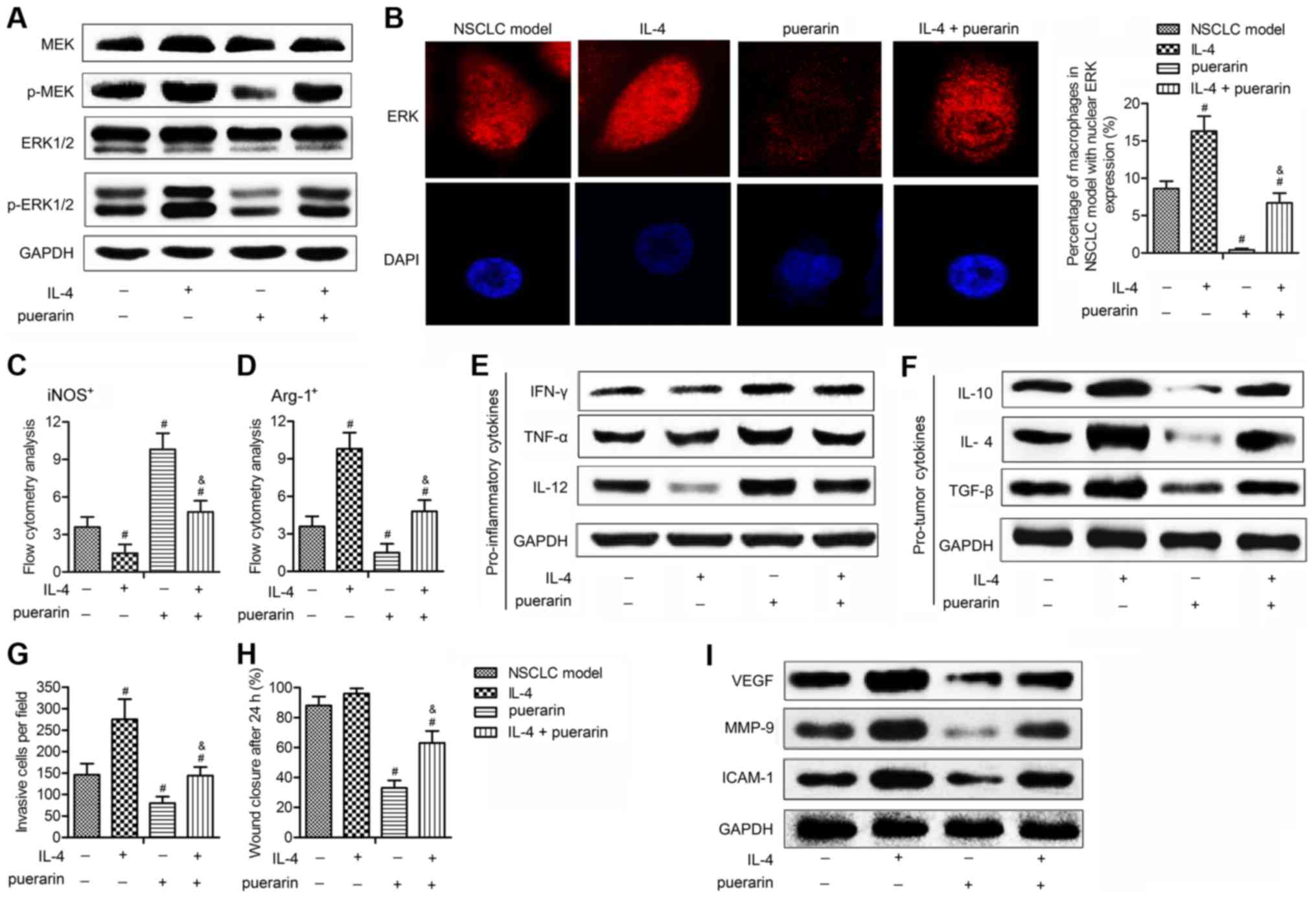
IL-4-induced M2 macrophage polarization
and metastasis are partially offset by puerarin through MEK/ERK1/2
pathway
To convince that puerarin exerts antitumor effect on
NSCLC is through inactivating MEK/ERK 1/2 pathway, we applied IL-4,
which has been reported to mediate M2 macrophage polarization and
promote tumor metastases via activating ERK pathway (23,24).
As shown in Fig. 7A and B, IL-4
upregulated the level of p-MEK and p-ERK 1/2, and a large
accumulation of ERK staining in cytoplasm and nucleus was detected
in NSCLC model induced by IL-4, indicating the strong activation of
MEK/ERK 1/2 pathway. However, the expression of p-MEK and p-ERK 1/2
and the percentage of cells with nucleus ERK expression was
significantly inhibited adding puerarin in IL-4-induced NSCLC
model. These findings convinced the inhibitory effect of puerarin
on MEK/ERK 1/2 pathway. Besides, puerarin increased
iNOS+ macrophages (Fig.
7C), which were reduced by IL-4, whereas the elevated
Arg-1+ macrophages induced by IL-4 were down-regulated
by puerarin (Fig. 7D).
Consistently, IL-4 reduced pro-inflammatory markers (IFN-γ, TNF-α
and IL-12) (Fig. 7E) and increased
pro-tumor markers (IL-10, IL-4 and TGF-β) (Fig. 7F), these effects were offset adding
puerarin in IL-4 induced NSCLC model. Besides, the promoting effect
of IL-4 on metastasis was restrained by puerarin (Fig. 7G). In addition, puerarin reduced
the invasive numbers, slowing the wound healing (Fig. 7H) and inhibited expression of tumor
metastasis-related proteins (VEGF, MMP-9 and ICAM-1) (Fig. 7I). Taken together, these results
are convincing that puerarin suppressed M2 macrophage polarization
and metastasis of NSCLC via inactivating MEK/ERK 1/2 pathway.
Discussion
Although early-stage non-small cell lung cancer
(NSCLC) can be cured by surgical resection, a substantial fraction
of patients ultimately dies due to distant metastasis (25). Therefore, identifying novel
molecules that can repress the invasiveness and metastasis of NSCLC
will facilitate the development of new anti-lung cancer strategies
(26). As a well known isoflavone
C-glycoside, puerarin is available in common foods and has been
shown to possess beneficial effects for various patients (17). The present study examined the
effect of puerarin on macrophage polarization and metastasis of
NSCLC.
Accumulated studies have demonstrated anticancer
activity of puerarin in animal models as well as in a variety of
cancer cell lines. Puerarin was suggested as a safe and effective
chemosensitive agent in the treatment of human esophageal cancer,
for puerarin inhibited proliferation of Eca-109 esophageal cancer
cells in vitro and in vivo, and did not increase the
side-effects of chemotherapy (27). The same behavior of puerarin was
observed in gastric carcinoma (28), hepatocellular carcinoma (29) and colon cancer (30). Recently, an in vitro and
in vivo animal study showed that puerarin significantly
inhibited tumor growth in A549 cells (18). Consistently, our research exhibited
that puerarin suppressed tumor growth and tumor volumes in NSCLC
xenograft model, suggesting the antitumor effect of puerarin in
NSCLC.
The TAMs can secrete a variety of cytokines, which
play a key role in the formation of tumor microenvironment and
tumor invasion and metastasis (31). The function of M1 and M2
macrophages is entirely different in the tumor microenvironment
(6). To investigate the effect of
puerarin on M1 and M2 marker expression in TAM populations, we
selected M1 markers (e.g., CD197 and iNOS) and M2 markers (e.g.,
CD206 and Arg-1), based on a large amount of literature in the
relevant field of macrophage polarization (6,7,32,33).
iNOS was suggested as an important mediator that may alter the TAM
phenotype and eventually improve tumor suppressing function
(34). TAMs that express iNOS were
associated with extended survival in patients with NSCLC (12). A report indicated that iNOS
expression was decreased in tissue from NSCLC patients with
adenocarcinoma and squamous cell carcinoma compared to non-tumor
tissues (7). A previous study
measured reduced iNOS expression in TAMs that were directly
isolated from the tumor in tumor-bearing mice (35). In accordance with these reports,
our research demonstrated that M1 macrophages (CD197+,
iNOS+ and CD40+) were decreased in NSCLC
model, while puerarin strongly increased M1 macrophages. These
results indicated that puerarin may alter the TAM phenotype through
upregulating M1 macrophages. The expression of the M2 marker in
TAMs was significantly correlated to poor prognosis and lymph node
metastasis in patients with advanced adenocarcinoma (14). A report exhibited that the
expression of M2 marker CD163 was significantly increased in all
NSCLC subtypes (7) and in patients
with progressive disease (36).
Similarly, the M2 markers (CD206+, Arg-1+ and
CD163+) were increased in NSCLC model in the present
study. However, puerarin strongly decreased the expression of M2
markers. Considering the percentage of TAMs within a tumor
microenvironment has been linked with tumor metastasis (14). Our results suggested the
anti-metastasis effect of puerarin is through elevating M1 markers
and inhibiting M2 markers.
The presence of cytokines is essential for the
initiation of immune responses (37). Th1 cells have been found to play a
major role in anti-tumor immunity, whereas Th2 cells are known to
act as the helper cells that influence B-cell development and
produce anti-inflammatory cytokines (38). Pro-inflammatory cytokines such as
TNF-α and IFN-γ are known to stimulate Th1 cells (19,39).
In contrast, Th2 cells produce anti-inflammatory cytokines such as
IL-4 and IL-10 (13,24). TGF-β has been suggested to induce
an M2-like phenotype characterized by upregulation of the
anti-inflammatory cytokine IL-10 and downregulation of the
pro-inflammatory cytokines TNF-α and IL-12 (40). IL-10 itself can also promote M1 to
M2 transition (41). In this
study, puerarin enhanced the expression of antitumor cytokines
(IFN-γ, TNF-α and IL-12), reduced the level of anti-inflammatory
cytokines (IL-10, IL-4 and TGF-β). The following in vitro
experiments showed that puerarin significantly reduced
Arg-1+ macrophages and the level of M2 markers. These
results indicated that puerarin was able to skew macrophage
populations back to M1 subsets to stimulate antitumor effects
within the tumor microenvironment, by directly acting on
macrophages.
Puerarin can inhibit the adhesion, invasion and
migration of HO-8910 cells, plays an antagonist effect against the
stimulation of estrogen on the malignant behavior of tumor cells
(42). A report also indicated
that puerarin suppressed the tissue invasion and the
vascularization of ectopic endometrial tissues stimulated by
17β-estradiol, by decreasing the angiogenesis factors MMP-9, ICAM-1
and VEGF (43). Based on the
research, puerarin suppressed the invasion and migration of A549
cells, inhibited the expressions of tumor metastasis-related
proteins (VEGF, MMP-9 and ICAM-1) in macrophages. These results
confirmed that puerarin was a negative metastatic regulator of
NSCLC.
The activation of MEK/ERK 1/2 pathway is closely
related to NSCLC metastasis. A report indicated that the activation
of ERK signaling skewing macrophage polarization away from the M1-
to a tumor-promoting M2-like phenotype, and high density of M2
macrophages was associated with metastasis in NSCLC patients
(22). Previous research also
demonstrated that suppressing ERK1/2 led to suppression of various
critical proteins for A549 invasion and migration (44). The results in this study indicated
that puerarin restrained the activation of MEK/ERK 1/2 pathway
through inhibition of ERK nucleus translocation. Besides,
IL-4-induced M2 macrophage polarization and metastasis were
partially offset by puerarin through restraining MEK/ERK 1/2
pathway. These results are convincing that puerarin suppressed M2
macrophage polarization and metastasis of NSCLC via inactivating
the MEK/ERK 1/2 pathway.
In conclusion, the present study explored puerarin
on macrophage polarization and metastasis of NSCLC. We found that
puerarin suppressed tumor growth in NSCLC xenograft model,
increased M1 macrophages, decreased M2 markers, enhanced the
expression of antitumor cytokines, reduced the level of pro-tumor
cytokines. Besides, puerarin suppressesed the invasion and
migration of NSCLC macrophages, inhibited the expressions of
angiogenesis factors. Finally, IL-4-induced M2 macrophage
polarization and metastasis were partially offset by puerarin
through restraining MEK/ERK 1/2 pathway. These results validated
that puerarin is able to skew macrophage populations back to M1
subsets to stimulate anti-tumor effects, and suggest puerarin is a
negative metastatic regulator of NSCLC.
Abbreviations:
|
NSCLC
|
non-small cell lung carcinoma
|
|
TAMs
|
tumor associated macrophages
|
|
iNOS
|
inducible nitric oxide synthase
|
|
IFN-γ
|
interferon-γ
|
|
TNF-α
|
tumor necrosis factor-α
|
|
IL
|
interleukine
|
|
TGF-β
|
transforming growth factor-β
|
|
VEGF
|
vascular endothelial growth factor
|
|
MMP-9
|
matrix metalloproteinase-9
|
|
ICAM-1
|
intercellular adhesion molecule-1
|
|
MEK
|
mitogen-activated extracellular
signal-regulated kinase
|
|
ERK
|
extracellular signal-regulated
kinase
|
Acknowledgments
All authors would like to thank the members of the
Liaocheng People's Hospital, for providing helpful discussions and
technical support concerning the present study.
References
|
1
|
Kim R, Emi M, Tanabe K and Arihiro K:
Tumor-driven evolution of immunosuppressive networks during
malignant progression. Cancer Res. 66:5527–5536. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Harpole DH Jr: Prognostic modeling in
early stage lung cancer: An evolving process from histopathology to
genomics. Thorac Surg Clin. 17:167–173. viii2007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wood SL, Pernemalm M, Crosbie PA and
Whetton AD: The role of the tumor-microenvironment in lung
cancer-metastasis and its relationship to potential therapeutic
targets. Cancer Treat Rev. 40:558–566. 2014. View Article : Google Scholar
|
|
4
|
Rhee I: Diverse macrophages polarization
in tumor microenvironment. Arch Pharm Res. 39:1588–1596. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Shigeoka M, Urakawa N, Nishio M, Takase N,
Utsunomiya S, Akiyama H, Kakeji Y, Komori T, Koma Y and Yokozaki H:
Cyr61 promotes CD204 expression and the migration of macrophages
via MEK/ERK pathway in esophageal squamous cell carcinoma. Cancer
Med. 4:437–446. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
6
|
Liu B, Wang X, Chen TZ, Li GL, Tan CC,
Chen Y and Duan SQ: Polarization of M1 tumor associated macrophage
promoted by the activation of TLR3 signal pathway. Asian Pac J Trop
Med. 9:484–488. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Almatroodi SA, McDonald CF, Darby IA and
Pouniotis DS: Characterization of M1/M2 tumour-associated
macrophages (TAMs) and Th1/Th2 cytokine profiles in patients with
NSCLC. Cancer Microenviron. 9:1–11. 2016. View Article : Google Scholar :
|
|
8
|
Ferrante CJ, Pinhal-Enfield G, Elson G,
Cronstein BN, Hasko G, Outram S and Leibovich SJ: The
adenosine-dependent angiogenic switch of macrophages to an M2-like
phenotype is independent of interleukin-4 receptor alpha (IL-4Rα)
signaling. Inflammation. 36:921–931. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Moore SM, Holt VV, Malpass LR, Hines IN
and Wheeler MD: Fatty acid-binding protein 5 limits the
anti-inflammatory response in murine macrophages. Mol Immunol.
67:265–275. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Choi JW, Kwon MJ, Kim IH, Kim YM, Lee MK
and Nam TJ: Pyropia yezoensis glycoprotein promotes the M1 to M2
macrophage phenotypic switch via the STAT3 and STAT6 transcription
factors. Int J Mol Med. 38:666–674. 2016.PubMed/NCBI
|
|
11
|
Tran TH, Rastogi R, Shelke J and Amiji MM:
Modulation of macrophage functional polarity towards
anti-inflammatory phenotype with plasmid DNA delivery in CD44
targeting hyaluronic acid nanoparticles. Sci Rep. 5:166322015.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ohri CM, Shikotra A, Green RH, Waller DA
and Bradding P: Macrophages within NSCLC tumour islets are
predominantly of a cytotoxic M1 phenotype associated with extended
survival. Eur Respir J. 33:118–126. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
da Silva MD, Bobinski F, Sato KL, Kolker
SJ, Sluka KA and Santos AR: IL-10 cytokine released from M2
macrophages is crucial for analgesic and anti-inflammatory effects
of acupuncture in a model of inflammatory muscle pain. Mol
Neurobiol. 51:19–31. 2015. View Article : Google Scholar
|
|
14
|
Zhang B, Yao G, Zhang Y and Gao J, Yang B,
Rao Z and Gao J: M2-polarized tumor-associated macrophages are
associated with poor prognoses resulting from accelerated
lymphangiogenesis in lung adenocarcinoma. Clinics (Sao Paulo).
66:1879–1886. 2011. View Article : Google Scholar
|
|
15
|
Chen PC, Cheng HC, Wang J, Wang SW, Tai
HC, Lin CW and Tang CH: Prostate cancer-derived CCN3 induces M2
macrophage infiltration and contributes to angiogenesis in prostate
cancer microenvironment. Oncotarget. 5:1595–1608. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang S, Chen S, Shen Y, Yang D, Liu X,
Sun-Chi AC and Xu H: Puerarin induces angiogenesis in myocardium of
rat with myocardial infarction. Biol Pharm Bull. 29:945–950. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhou YX, Zhang H and Peng C: Puerarin: A
review of pharmacological effects. Phytother Res. 28:961–975. 2014.
View Article : Google Scholar
|
|
18
|
Chen T, Chen H, Wang Y and Zhang J: In
vitro and in vivo antitumour activities of puerarin 6″-O-xyloside
on human lung carcinoma A549 cell line via the induction of the
mitochondria-mediated apoptosis pathway. Pharm Biol. 5:1–7.
2016.
|
|
19
|
Liu C, Li Y, Yu J, Feng L, Hou S, Liu Y,
Guo M, Xie Y, Meng J, Zhang H, et al: Targeting the shift from M1
to M2 macrophages in experimental autoimmune encephalomyelitis mice
treated with fasudil. PLoS One. 8:e548412013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Becker M, Müller CB, De Bastiani MA and
Klamt F: The prognostic impact of tumor-associated macrophages and
intra-tumoral apoptosis in non-small cell lung cancer. Histol
Histopathol. 29:21–31. 2014.
|
|
21
|
Deepak P, Kumar S and Acharya A:
Interleukin-13 neutralization modulates interleukin-13 induced
suppression of reactive oxygen species production in peritoneal
macrophages in a murine T-cell lymphoma. Cell Immunol. 251:72–77.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhang J, Cao J, Ma S, Dong R, Meng W, Ying
M, Weng Q, Chen Z, Ma J, Fang Q, et al: Tumor hypoxia enhances
non-small cell lung cancer metastasis by selectively promoting
macrophage M2 polarization through the activation of ERK signaling.
Oncotarget. 5:9664–9677. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Park SJ, Lee KP, Kang S, Lee J, Sato K,
Chung HY, Okajima F and Im DS: Sphingosine 1-phosphate induced
anti-atherogenic and atheroprotective M2 macrophage polarization
through IL-4. Cell Signal. 26:2249–2258. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ferreira R, Lively S and Schlichter LC:
IL-4 type 1 receptor signaling up-regulates KCNN4 expression, and
increases the KCa3.1 current and its contribution to migration of
alternative-activated microglia. Front Cell Neurosci. 8:1832014.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ji P, Diederichs S, Wang W, Böing S,
Metzger R, Schneider PM, Tidow N, Brandt B, Buerger H, Bulk E, et
al: MALAT-1, a novel noncoding RNA, and thymosin beta4 predict
metastasis and survival in early-stage non-small cell lung cancer.
Oncogene. 22:8031–8041. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shi L, Zhang B, Sun X, Lu S, Liu Z, Liu Y,
Li H, Wang L, Wang X and Zhao C: MiR-204 inhibits human NSCLC
metastasis through suppression of NUAK1. Br J Cancer.
111:2316–2327. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang J, Yang ZR, Guo XF, Song J, Zhang JX,
Wang J and Dong WG: Synergistic effects of puerarin combined with
5-fluorouracil on esophageal cancer. Mol Med Rep. 10:2535–2541.
2014.PubMed/NCBI
|
|
28
|
Guo XF, Yang ZR, Wang J, Lei XF, Lv XG and
Dong WG: Synergistic antitumor effect of puerarin combined with
5-fluorouracil on gastric carcinoma. Mol Med Rep. 11:2562–2568.
2015.
|
|
29
|
Zeng YP, Yang ZR, Guo XF, Jun W and Dong
WG: Synergistic effect of puerarin and 5-fluorouracil on
hepatocellular carcinoma. Oncol Lett. 8:2436–2442. 2014.PubMed/NCBI
|
|
30
|
Wang Y, Ma Y, Zheng Y, Song J, Yang X, Bi
C, Zhang D and Zhang Q: In vitro and in vivo anticancer activity of
a novel puerarin nanosuspension against colon cancer, with high
efficacy and low toxicity. Int J Pharm. 441:728–735. 2013.
View Article : Google Scholar
|
|
31
|
Ruffell B, Affara NI and Coussens LM:
Differential macrophage programming in the tumor microenvironment.
Trends Immunol. 33:119–126. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Li D, Duan M, Feng Y, Geng L, Li X and
Zhang W: MiR-146a modulates macrophage polarization in systemic
juvenile idiopathic arthritis by targeting INHBA. Mol Immunol.
77:205–212. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Yang L, Wang F, Wang L, Huang L, Wang J,
Zhang B and Zhang Y: CD163+ tumor-associated macrophage
is a prognostic biomarker and is associated with therapeutic effect
on malignant pleural effusion of lung cancer patients. Oncotarget.
6:10592–10603. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Deng X, Zhang P, Liang T, Deng S, Chen X
and Zhu L: Ovarian cancer stem cells induce the M2 polarization of
macrophages through the PPARγ and NF-κB pathways. Int J Mol Med.
36:449–454. 2015.PubMed/NCBI
|
|
35
|
Dinapoli MR, Calderon CL and Lopez DM: The
altered tumoricidal capacity of macrophages isolated from
tumor-bearing mice is related to reduce expression of the inducible
nitric oxide synthase gene. J Exp Med. 183:1323–1329. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
de Gaetano M, Crean D, Barry M and Belton
O: M1- and M2-type macrophage responses are predictive of adverse
outcomes in human atherosclerosis. Front Immunol. 7:2752016.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Belardelli F and Ferrantini M: Cytokines
as a link between innate and adaptive antitumor immunity. Trends
Immunol. 23:201–208. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Seo N and Tokura Y: Downregulation of
innate and acquired antitumor immunity by bystander gammadelta and
alphabeta T lymphocytes with Th2 or Tr1 cytokine profiles. J
Interferon Cytokine Res. 19:555–561. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Iwamoto S, Iwai S, Tsujiyama K, Kurahashi
C, Takeshita K, Naoe M, Masunaga A, Ogawa Y, Oguchi K and Miyazaki
A: TNF-alpha drives human CD14+ monocytes to
differentiate into CD70+ dendritic cells evoking Th1 and
Th17 responses. J Immunol. 179:1449–1457. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Zhang F, Wang H, Wang X, Jiang G, Liu H,
Zhang G, Wang H, Fang R, Bu X, Cai S, et al: TGF-β induces M2-like
macrophage polarization via SNAIL-mediated suppression of a
pro-inflammatory phenotype. Oncotarget. 13:105612016.
|
|
41
|
Deng B, Wehling-Henricks M, Villalta SA,
Wang Y and Tidball JG: IL-10 triggers changes in macrophage
phenotype that promote muscle growth and regeneration. J Immunol.
189:3669–3680. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Han J, Yu CQ and Shen W: Inhibitory
effects of puerarin on invasion and metastasis of oophoroma cells
HO-8910. Zhongguo Zhong Xi Yi Jie He Za Zhi. 29:632–635. 2009.In
Chinese. PubMed/NCBI
|
|
43
|
Wang D, Liu Y, Han J, Zai D, Ji M, Cheng
W, Xu L, Yang L, He M, Ni J, et al: Puerarin suppresses invasion
and vascularization of endometriosis tissue stimulated by
17β-estradiol. PLoS One. 6:e250112011. View Article : Google Scholar
|
|
44
|
Lee SH, Jaganath IB, Manikam R and Sekaran
SD: Inhibition of Raf-MEK-ERK and hypoxia pathways by Phyllanthus
prevents metastasis in human lung (A549) cancer cell line. BMC
Complement Altern Med. 13:2712013. View Article : Google Scholar : PubMed/NCBI
|