Introduction
On a global scale, head and neck cancer is the sixth
most common type of malignant cancer type in humans, and the major
histological type is squamous cell carcinoma (1). Over 600,000 new cases of head and
neck squamous cell carcinoma (HNSCC) occur each year, and most of
the individuals with HNSCC are already in the advanced stage by the
time of diagnosis (2). Currently,
comprehensive treatment consisting of surgery plus postoperative
chemoradiotherapy is still the most commonly used method to treat
advanced-stage HNSCC. However, the prognosis of patients in
advanced stages is not optimistic, and over 50% of patients
experience relapse and distant metastasis after treatment (3). Furthermore, tumor relapse and
metastasis caused by radiotherapy resistance are still the major
causes of failure of HNSCC treatment (4). Therefore, investigating molecular
targets that can improve the radiosensitivity of HNSCC will have
clinical significance for the effective improvement of treatment
efficacy and the prognosis of patients with HNSCC.
Fanconi anemia (FA) is an autosomal or X-linked
recessive genetic disease and is characterized by congenital
malformations, progressive bone marrow hematopoietic failure,
increased chromosomal fragility and high susceptibility to tumors
(5). In patients with FA, the
incidence of HNSCC is significantly higher than in the average
population and most patients tend to be younger (6). Patients with FA show high sensitivity
to ionizing radiation and to mitomycin C, cisplatin and other DNA
cross-linking agents. To date, 18 FA-associated genes have been
found (7,8). Proteins encoded by these genes
compose an FA signaling pathway that is involved in the repair of
DNA crosslink damage. Upstream of the FA pathway is the FA core
complex, which has E3 ubiquitin ligase activity and is composed of
at least eight FA proteins and FA-related proteins. The FA core
complex monoubiquitinates FANCD2 and FANCI (with the ID complex
formed by FANCD2 and FANCI), thus, further mobilizing downstream FA
proteins and DNA repair-related proteins; eventually, the USP1/UAF1
complex deubiquitinates the ID complex to complete the FA pathway
(9–11). The function of the FA pathway
involves cell cycle regulation, DNA damage and repair, apoptosis,
gene transcription and the maintenance of genome stability.
Inactivation of the FA pathway, which causes gene instability and
impedes the repair of DNA crosslink damage, is the molecular
mechanism of FA pathogenesis.
As a central link, FANCD2 connects the upstream and
downstream portions of the FA pathway, and its suppression will
inevitably block the transduction of FA pathway signals (12). Some scholars have found that the
downregulation of FANCD2 nuclear foci formation by phenylbutyrate
can enhance the sensitivity of HNSCC to cisplatin (13). Other studies have suggested that
FANCD2 plays an important role in the maintenance of gene stability
and DNA damage repair during radiotherapy (14). Moreover, the downregulation of
FANCD2 can enhance the 'bystander effect', which occurs during
radiotherapy (15). Therefore,
suppressing the role of FANCD2 and then downregulating the activity
of the FA pathway may enhance the sensitivity of HNSCC to
radiotherapy. However, the relationship between FANCD2 and the
sensitivity of HNSCC to radiotherapy is currently unclear.
In the present study, we used shRNA to knock down
the expression of FANCD2 in the metastatic cervical lymph
node-derived HNSCC cell line HSC-4. Through in vitro and
in vivo experiments, we investigated whether the silencing
of FANCD2 could enhance the sensitivity of HSC-4 cells to
radiotherapy; we also determined the associated mechanism, which
has provided a new molecular target for the clinical treatment of
head and neck tumors.
Materials and methods
Cell lines and cell culture
The HNSCC cell line HSC-4 was used. An experimental
group FANCD2-shRNA (HSC-4 cells transfected with an effective
FANCD2 interference sequence where the silencing effect is stable),
negative control group FANCD2-shRNA-C (HSC-4 cells transfected with
an ineffective FANCD2 interference sequence) and blank control
group HSC-4 (wild-type HSC-4 cells without any treatment) were
used. These cells were generously provided by Dr Stephan M. Feller
of the Weatherall Institute of Molecular Medicine at the University
of Oxford, UK. The cell culture conditions were as follows: cells
were cultured in high-glucose Dulbecco's modified Eagle's medium
(DMEM; Thermo Fisher Scientific, Beijing, China) supplemented with
10% fetal bovine serum (FBS; Bio-Cell, Beijing, China), 100
µg/ml streptomycin and 100 U/ml penicillin. The cells were
maintained in a thermostat incubator at 37°C in an atmosphere of 5%
CO2 and saturated humidity. Puromycin (InvivoGen, San
Diego, CA, USA), at a final concentration of 2 µg/ml, was
added to the cells of the experimental group and the negative
control group as a selection reagent.
Radiotherapy
The radiotherapy for both cells and animals was
conducted at room temperature using the medical linear electron
accelerator Elekta Precise NET (Elekta, Stockholm, Sweden) at a
dose rate of 200 cGy/min.
Cell proliferation analysis
Cell Counting kit-8 (CCK-8; Sigma-Aldrich, St.
Louis, MO, USA) was used to evaluate the impact of FANCD2 shRNA
interference on the in vitro proliferation of HSC-4 cells
after radiotherapy. Cells in the logarithmic growth phase from the
three groups were seeded in 96-well plates at a density of 3,000
cells/well. Radiotherapy of 0, 2, 5 or 8 Gy was given to the cells,
and 48 h after radiotherapy, the absorbance at 450 nm was measured.
In addition, the three groups of cells received 5 Gy of
radiotherapy, and the absorbance at 450 nm was measured at 24, 48
and 72 h after radiotherapy, which enabled the calculation of the
inhibition of the cell proliferation rate. The inhibition of the
cell proliferation rate = (1− average optical density (OD) value of
the treated group/average OD value of the blank control group) ×
100%. Finally, the concentration-inhibition rate curve was
plotted.
Colony formation assay
A colony formation assay was used to examine the
impact of FANCD2 shRNA interference on the survival rate of HSC-4
cells after radiotherapy. Cells from the three groups were seeded
into 6-cm culture plates at a predetermined number; and 24 h after
incubation, the cells received 0, 2, 5 or 8 Gy of radiotherapy,
after which the cells were routinely cultured for 13 days. Then,
the cells were fixed in methanol and stained with 1% crystal
violet-ethanol solution. The number of clones that contained >50
cells was counted under a microscope, and the colony formation rate
(colony formation rate = number of clones/number of cells seeded x
100%) and survival rate (survival rate = the number of clones in
the experimental group under a certain dose/the number of cells
seeded in this group x colony formation rate) were calculated. The
analysis was performed using the average value of the survival rate
found in three different radiation treatments.
Cell apoptosis analysis
Flow cytometry was used to examine the impact of
FANCD2 shRNA interference on the apoptosis rate of HSC-4 cells
after radiotherapy. Cells in the logarithmic growth phase from the
three groups were seeded in 6-well plates at a final concentration
of 1×105 cells/ml; when the cells became adherent and
reached a confluence of ~80%, they received an 8-Gy dose of
radiotherapy. After radiotherapy, the cells were further cultured
for 48 h before the experimental cells were harvested. The cells
were washed twice with buffer, and cells were assayed on a machine
according to the manufacturer's instructions for the Annexin V-FITC
apoptosis detection kit (Nanjing KeyGen Biotech, Co., Ltd.,
Nanjing, China). The rate of apoptosis of the cells in each group
was calculated based on the data obtained.
Cell cycle analysis
Flow cytometry was used to examine the impact of
FANCD2 shRNA interference on the cell cycle distribution of HSC-4
cells after the radiotherapy. Cells in the logarithmic growth phase
from the three groups received either 0 or 8 Gy of radiotherapy and
then, the cells were harvested 48 h after the radiotherapy. The
cells were washed twice with phosphate-buffered saline (PBS) and
were assayed on the machine according to the manufacturer's
instructions for the cell cycle assay kit (Nanjing KeyGen Biotech).
The percentage of cells in each phase of the cell cycle was then
calculated.
Animal experiments
Female BALB/c-nu nude mice that were 3 to 4 weeks
old and that weighed 18–22 g were purchased from Beijing HFK
Bioscience, Co., Ltd., Beijing, China. The mice were housed in the
specific pathogen-free (SPF) animal room of the Department of
Oncology of the Affiliated Hospital of Southwest Medical
University. All animal experimental procedures were performed in
accordance with the appropriate institution or the National
Research Council Guide for the care and use of laboratory animals
and approved by the Affiliated Hospital of Southwest Medical
University. The nude mice were randomly divided into three groups,
with five mice in each group. Cells in the logarithmic growth phase
from the three groups were used to prepare cell suspensions in
high-glucose DMEM at a concentration of 1.0×107
cells/ml. Then, 0.1 ml of the cell suspension was subcutaneously
injected into the outer side of the left thigh of the corresponding
mice, and the tumor formation rate and the tumor formation time of
each group were monitored. After visible tumors formed, the tumors
were measured every three days using a Vernier caliper, and the
tumor volume was calculated according to the formula V = π/6 × a ×
b2, where a is the longest diameter of the tumor, and b
is the short diameter perpendicular to a. When the longest diameter
of the tumor was ~4–6 mm, the radiotherapy regimen commenced, which
involved the local administration of radiotherapy to the tumor.
According to the conventional fractionated radiotherapy method, 2
Gy was given once per day for five days for a total dose of 10 Gy.
On the 10th day after the radiotherapy was terminated, the mice
were sacrificed by cervical dislocation, the tumors were removed
and weighed, and the tumor inhibition rate was calculated. Each
tumor was equally divided into two halves: one half was stored in
liquid nitrogen for western blot analysis, while the other half was
fixed in neutral-buffered formalin and paraffin-embedded for
hematoxylin and eosin (H&E) staining, terminal deoxynucleotidyl
transferase-mediated dUTP nick end labeling (TUNEL) staining and
immunohistochemistry.
TUNEL staining assay
TUNEL staining was used to examine tumor cell
apoptosis in the three groups of cells after the radiotherapy. The
protocol was performed according to the instructions of the TUNEL
apoptosis assay kit (Roche Diagnostics, Shanghai, China). When the
nucleus was stained brown, the cell was defined as an apoptotic
cell. Under a microscope at a magnification of ×400, 10 fields were
randomly selected from each slide to calculate the total number of
apoptotic cells. The apoptotic index (AI) was calculated, where the
AI = the total number of apoptotic cells/the total number of
nucleated cells x 100%.
Western blot analysis
Western blotting was used to determine the
expression level of FANCD2 protein in HSC-4 cells and the
expression of apoptosis pathway-related proteins in the tumors and
in HSC-4 cells after radiotherapy. Tumor tissue was thoroughly
ground in liquid nitrogen and lysis buffer was added. The tumor
tissue and buffer were mixed well, the sample was centrifuged and
the proteins were extracted. Cells in the logarithmic growth phase
from the three groups were seeded in 10-cm culture plates, and a
radiotherapy dose of either 0 or 8 Gy was given to each cell group.
After radiotherapy, the cells were cultured for 24 h before they
were harvested. The cells were rinsed two to three times with
pre-chilled PBS and the proteins were extracted. The protein
samples were adjusted to the same concentration, loaded onto a 10%
sodium dodecyl sulfate polyacrylamide gel electrophoresis
(SDS-PAGE) gel to separate the proteins and then transferred to a
nitrocellulose membrane, which was blocked with Tris-buffered
saline with Tween-20 (TBST) containing 5% fat-free milk. The
following primary antibodies were used: mouse anti-human FANCD2
polyclonal antibody diluted 1:300, rabbit anti-human Bax polyclonal
antibody diluted 1:200, mouse anti-human Bcl12 monoclonal antibody
diluted 1:200, mouse anti-human p38 monoclonal antibody diluted
1:200, rabbit anti-human p-p38 polyclonal antibody diluted 1:200
and mouse anti-human β-actin monoclonal antibody diluted 1:1000
(all purchased from Santa Cruz Biotechnology, Santa Cruz, CA, USA).
The membrane was incubated with the primary antibody overnight at
4°C. TBST was used to wash the membrane, and secondary antibody was
prepared as follows: goat anti-rabbit IgG diluted 1:2,000 and goat
anti-mouse IgG diluted 1:2,000 (both purchased from Santa Cruz
Biotechnology). The membrane was incubated with the secondary
antibody at room temperature for 1 h. TBST was used to wash the
membrane, and enhanced chemiluminescence (ECL) solution was used to
develop the protein bands.
Immunohistochemistry
The EnVision™ method was used for
immunohistochemistry to determine the expression levels of
apoptosis pathway-related proteins in the tumors after
radiotherapy. Paraffin-embedded tumors were continuously sectioned
at a thickness of 4 µm. Sections were routinely
deparaffinized in xylene and rehydrated in graded ethanol
solutions. Sections were then immersed into an appropriate amount
of ethylenediaminetetraacetic acid (EDTA) solution (pH 9.0) in a
slide box, which was placed inside a pressure cooker for antigen
retrieval under high temperature and pressure. The sections were
incubated with 3% H2O2 in methanol for 10 min
at room temperature to quench the endogenous peroxidase activity.
The primary antibodies were prepared as follows: rabbit anti-human
Bax polyclonal antibody was diluted 1:100, mouse anti-human Bcl2
monoclonal antibody was diluted 1:100, mouse anti-human p38
monoclonal antibody was diluted 1:100, and rabbit anti-human p-p38
polyclonal antibody was diluted 1:100 (all purchased from Santa
Cruz Biotechnology). Finally, the sections were incubated with the
appropriate secondary antibody, developed with diaminobenzidine
(DAB), counterstained in hematoxylin, dehydrated, cleared in xylene
and mounted.
Statistical analysis
SPSS 19.0 statistical analysis software and GraphPad
Prism 5 software were used to analyze the experimental results.
Experimental data are represented as mean ± SD and a one-way ANOVA
was used for data comparison. P<0.05 were considered
statistically significant.
Results
shRNA interference caused a reduction in
FANCD2 expression in HSC-4 cells
shRNA was used to silence FANCD2 expression in HSC-4
cells, while western blot analysis was used to examine FANCD2
expression in the three groups of cells. The results showed that
the level of FANCD2 expression in the experimental group was
significantly lower than the levels in the negative and blank
control groups (P<0.05; Fig.
1), which suggests that shRNA interference can effectively and
stably knock down FANCD2 gene expression in HSC-4 cells.
Silencing of FANCD2 enhanced the in vitro
proliferation inhibition effect in HSC-4 cells after
radiotherapy
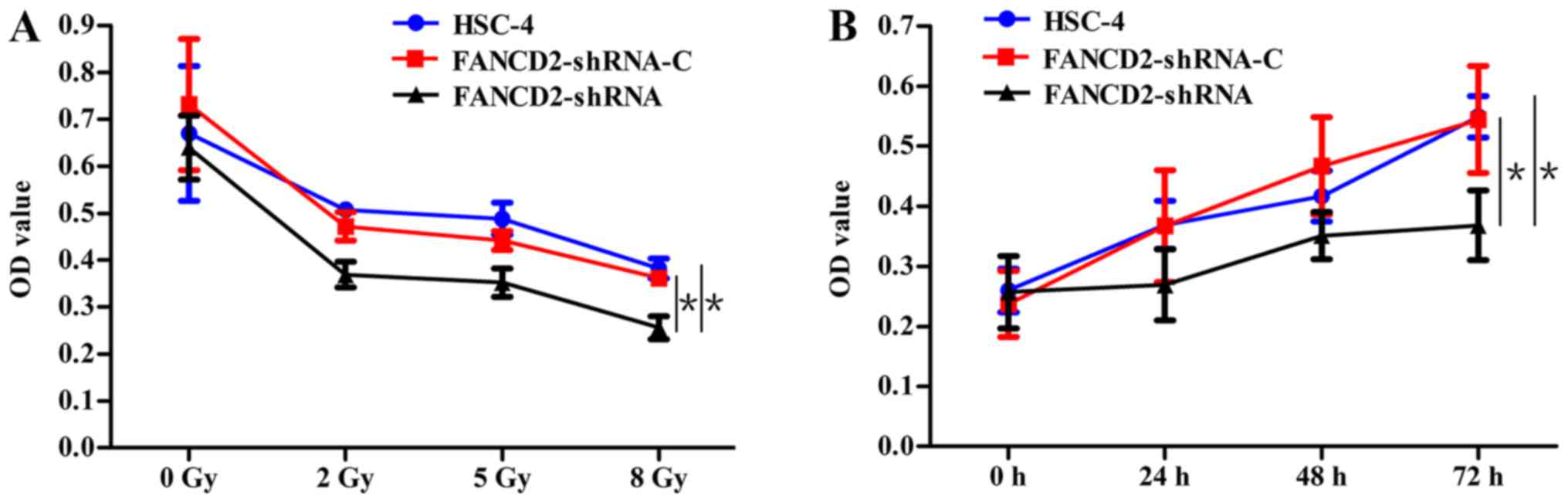
Cell proliferation experiments showed that the
proliferation levels of cells in all three groups that received
radiotherapy were lower than those in the cells that did not
receive radiotherapy (P<0.05). Moreover, the proliferation
inhibition effect was enhanced as the radiation dose increased. The
proliferation rate of the HSC-4 cells in the experimental group was
lower than that of the control groups after radiotherapy with
various dose (P<0.05), and the proliferation inhibition effect
was the most significant when the radiation dose was 8 Gy (Fig. 2A), which suggests that the
silencing of FANCD2 gene expression can enhance the effect of
proliferation inhibition in HSC-4 cells after radiotherapy and that
this effect is dose-dependent. When the radiation dose was 5 Gy for
the three groups of cells, the proliferation inhibition effect of
the experimental group was significantly enhanced compared with
that of the control groups at the three time-points of 24, 48 and
72 h (P<0.05; Fig. 2B). This
finding suggests that the silencing of FANCD2 gene expression can
enhance the effects of proliferation inhibition of HSC-4 cells
after radiotherapy and that this effect is time-dependent.
Silencing of FANCD2 reduced the survival
rate of HSC-4 cells after radiotherapy
The colony formation assay showed that the survival
rates of the three groups of cells were not significantly different
before radiotherapy was administered (P>0.05), but as the
radiotherapy dose increased, the survival rate of cells in each
group decreased. When the dose of radiotherapy was 8 Gy, the
survival rate of cells in the experimental group was significantly
lower than that of cells in the negative control and blank control
groups (P<0.05; Fig. 3), which
suggests that the silencing of FANCD2 reduced the survival rate of
HSC-4 cells after radiotherapy.
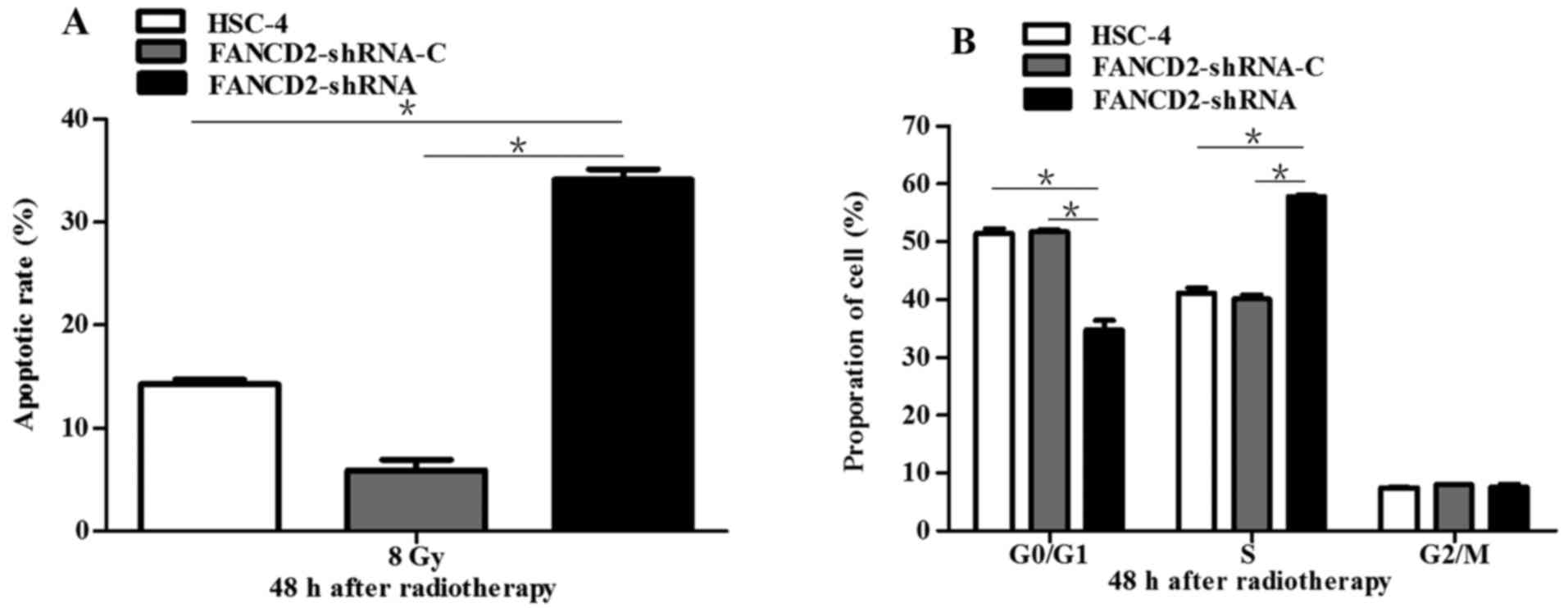
Silencing of FANCD2 enhances the
radiotherapy-induced apoptosis of HSC-4 cells and induced S phase
arrest
A dose of 8 Gy of radiation was administered to the
three groups of cells, and 48 h later, apoptosis was evaluated
using flow cytometry. The result showed that the apoptotic rate of
cells in the experimental group was significantly higher than that
of cells in the negative and blank control groups (P<0.05;
Fig. 4A), which suggests that the
silencing of FANCD2 can increase the radiotherapy-induced apoptotic
rate of HSC-4 cells and enhance their sensitivity to radiotherapy.
The three groups of cells, which had received a dose of 8 Gy, were
cultured under standard conditions for 48 h after which flow
cytometry was used to analyze the cell cycle distribution. The
result showed that after radiotherapy, the ratio of cells in G2/M
was not significantly different between the experimental group and
the control groups (P>0.05) but that the proportion of cells in
S phase was increased (P<0.05). In addition, the number of cells
entering the G0/G1 phase was decreased (P<0.05; Fig. 4B), which indicates that the
silencing of FANCD2 can induce an S phase arrest of HSC-4 cells
after radiotherapy.
Silencing of FANCD2 inhibits the HSC-4
cell growth in vivo, and increases the sensitivity of tumors to
radiotherapy
A tumor mass was visible in all 15 nude mice in the
three groups, and the tumor formation rate was 100% (Fig. 5A). Compared with the control
groups, the experimental group, in which tumors were transplanted
subcutaneously, demonstrated a prolonged tumor formation time and
slowed growth as well as significantly decreased tumor volume and
weight. The tumor formation time of HSC-4 cells in the experimental
group was 9 to 10 days with a median time of 10 days. The tumor
formation time of the negative control group and the blank group
was 5 to 7 days with a median time of 6 days. Before radiotherapy,
the tumor volume of the experimental group was 57.88±5.12
mm3, which was significantly lower than that of the
negative control group (89.05±7.44 mm3) and the blank
control group (98.83±6.31 mm3) (P<0.05; Fig. 5B). After radiotherapy, the tumor
volume of the experimental group was 11.11±5.25 mm3,
which was significantly lower than that of the negative control
group (57.96±11.36 mm3) and the blank control group
(67.42±4.40 mm3) (P<0.05; Fig. 5B). Moreover, after radiotherapy,
the tumor volume of the experimental group decreased (46.77±2.76
mm3), which was significantly more than that of the
negative control group (31.41±3.52 mm3) and the blank
control group (31.09±7.69 mm3) (P<0.05; Fig. 5B). After radiotherapy, the tumor
weight of the experimental group was 0.034±0.015 g, which was
significantly lower than that of the negative control group
(0.092±0.023 g) and the blank control group (0.100±0.045 g)
(P<0.05; Fig. 5C and D).
Lastly, the tumor weight inhibition rate of the experimental group
was 66.00±15.17%, and the volume inhibition rate was 83.52±7.78%
(P<0.05; Fig. 5C and D). These
results suggest that the silencing of FANCD2 inhibits the HSC-4
cell growth in vivo and enhances their sensitivity to
radiotherapy.
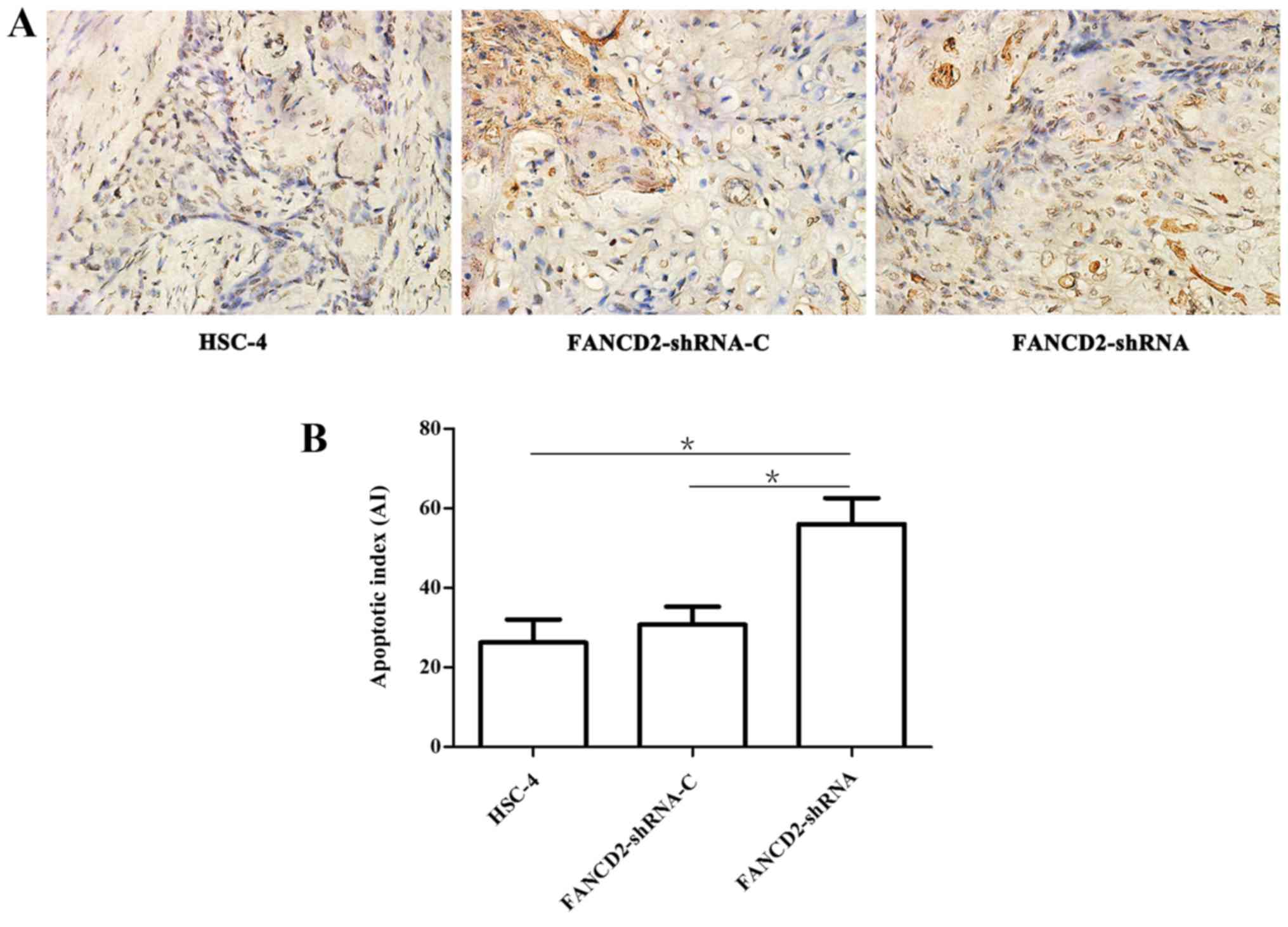
Silencing of FANCD2 increases apoptosis
in HSC-4 cell tumors after radiotherapy
A TUNEL assay showed that the three groups of
transplanted cells demonstrated different degrees of apoptosis
after radiotherapy, but the number of apoptotic cells in the
experimental group was significantly higher than that in the
negative and the blank control groups (Fig. 6A). The apoptotic index (AI) of the
experimental group was 56.02±6.51%, which was significantly higher
than that of the negative control group (30.80±4.47%) and the blank
control group (26.22±5.82%) (P<0.05; Fig. 6B).
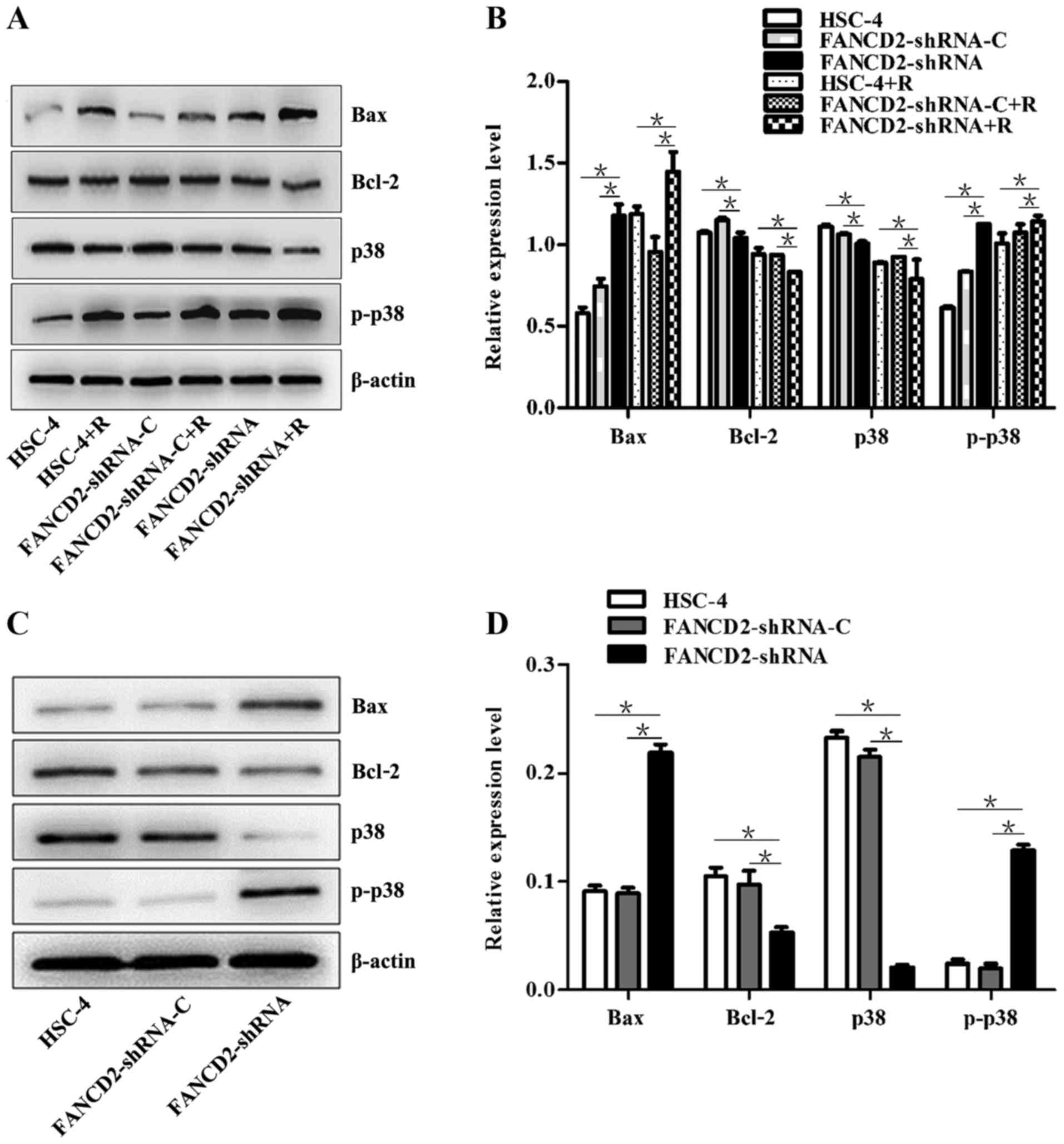
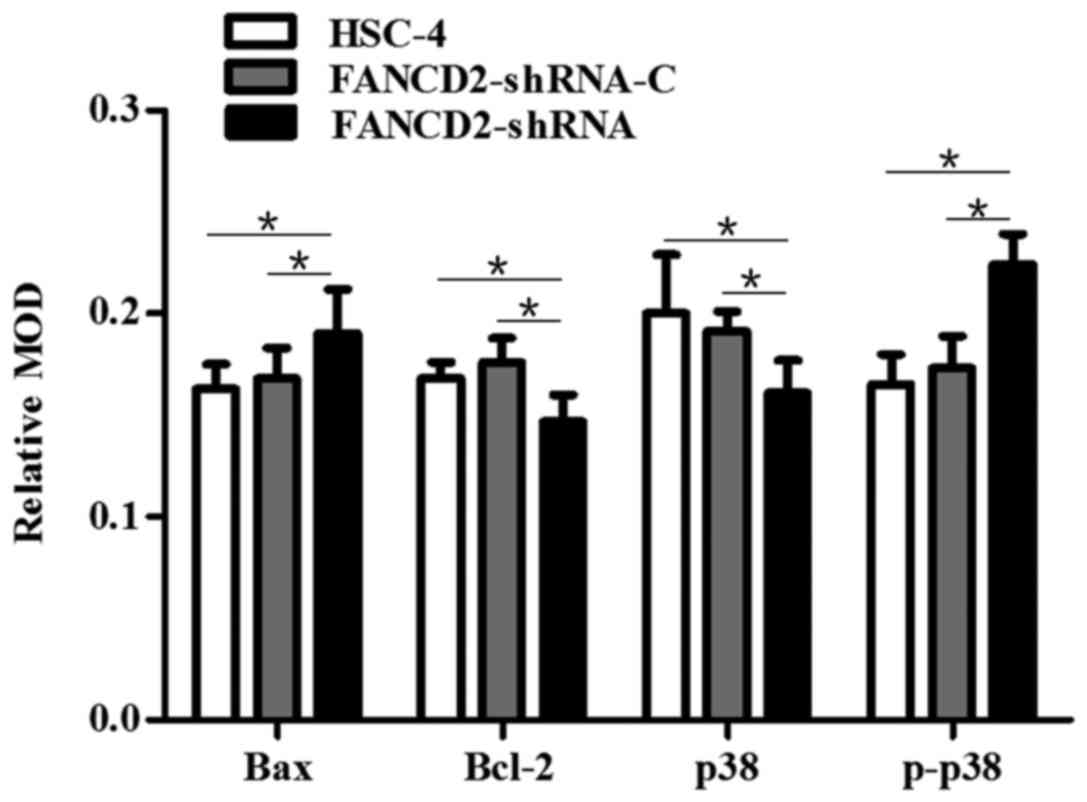
Silencing of FANCD2 increased the
Bax/Bcl2 ratio and activated the p38 MAPK signaling pathway in
HSC-4 cells after radiotherapy
Western blot analysis revealed that after the FANCD2
gene expression in HSC-4 cells was silenced by shRNA interference,
the expression of the Bax and p-p38 proteins was significantly
higher in the cells of the experimental group than in the control
groups (P<0.05), whereas the expression of the p38 and Bcl2
proteins was significantly decreased (P<0.05; Fig. 7A and B). After radiotherapy, the
same results were obtained; additionally, a significant difference
was observed in the expression of these proteins before and after
radiotherapy (P<0.05; Fig. 7A and
B). After the nude mice with transplanted HSC-4 cells received
radiotherapy, western blotting and immunohistochemistry were
performed, which showed an increase in the expression of Bax and
p-p38 proteins in the tumor tissues of mice in the experimental
group compared with the control group (P<0.05); in contrast, the
expression of p38 and Bcl2 proteins was decreased (P<0.05;
Figs. 7C and D, 8 and 9).
This finding is consistent with the results of the in vitro
cell culture experiments. These results suggest that the silencing
of FANCD2 increases the Bax/Bcl2 ratio and activates the p38 MAPK
signaling pathway in HSC-4 cells after radiotherapy.
Discussion
A study by Lyakhovich et al (16) found that the transient silencing of
FANCD2 expression using siRNA interference could increase the
sensitivity of breast cancer cells, liver cancer cells and bladder
cancer cells to γ-rays and DNA damaging agents, and such an
enhanced sensitivity effect is closely associated with the decrease
in the proliferation ability of cancer cells. The present study
found that the three groups of cells that received radiotherapy had
a lower proliferation rate compared with the group that did not
receive radiotherapy. The proliferation inhibition effect was
enhanced as the radiation dose increased, and the cell
proliferation rate was decreased as the radiotherapy duration
increased; these effects were most significant in the FANCD2
knockdown group. A colony formation assay also confirmed that the
cell survival rate of the experimental group after radiotherapy was
significantly lower than that of the negative and the blank control
groups. This finding suggests that the enhancement in the
sensitivity of HSC-4 cells to radiotherapy by FANCD2 silencing is
associated with a decrease in cell proliferation ability.
A large number of studies have found that cell cycle
regulation has a significant impact on the survival of tumor cells
and their sensitivity to radiotherapy, where G2/M phase arrest can
enhance the sensitivity of tumor cells to radiotherapy (17–19).
The present study found that after radiotherapy, the G2/M phase
ratio of HSC-4 cells in the experimental group was not
significantly different compared with that in the control groups.
In addition, the proportion of cells in S phase increased
significantly, and the number of cells entering the G0/G1 phase
decreased, which does not seem to correspond to an increased
proportion of cells in the G2/M phase. However, studies by other
scholars found that the inhibition of Bcl2 function could promote
the apoptosis of HNSCC cells, which results in S phase arrest
(20). Therefore, the increase in
the proportion of cells in S phase after radiotherapy in the
experimental group, which was found in the present study, may be
related to the downregulation of Bcl2 expression. Furthermore,
ubiquitination, which is important for the repair of DNA damage
caused by radiotherapy, occurs in the S phase of the cell cycle.
The downregulation of FANCD2 reduced the ubiquitination level of
FANCD2, which affects the DNA damage repair process and causes
cells to arrest in S phase (21).
Apoptosis is one of the important factors in the
radiosensitivity of tumor cells; and it is regulated by a
multiple-gene network, of which the Bax and Bcl2 gene families have
great importance in the molecular regulation of apoptosis. Farnebo
et al (22) found that Bax,
Bcl2, Bcl-X (L), COX-2 and p53 were closely related to the
predicted radiosensitivity of HNSCC cell lines. Some scholars
reported that the radiation-induced reduction in Bcl2 and elevation
in Bax were associated with an increase in the radiosensitivity of
tumor cells (23). Studies by
Oltvai et al (24)
demonstrated that Bax and Bcl2 existed in the form of a dimer in
cells. When the Bcl2 protein was expressed in excess, Bcl2/Bcl2
homodimers were formed, inhibiting apoptosis; however, when the
expression of the Bax protein was excessive, Bax/Bax homodimers
were formed, which also promoted apoptosis. Studies indicated that
an increase in Bax protein expression could induce an increase in
apoptosis via the activation of the caspase signal transduction
pathway (25). In the present
study, both in vivo and in vitro experiments showed
that apoptosis induced by radiotherapy in the experimental group
was significantly increased compared with that of the negative and
the blank control groups. Moreover, a western blot analysis found
that after FANCD2 expression was silenced by shRNA interference,
Bax expression was increased in the experimental group compared
with the control groups. In contrast, Bcl2 expression was decreased
in the experimental group compared with the control groups. After
the three groups of cells received radiotherapy, Bax expression was
increased in the shRNA interference group, while Bcl2 expression
was reduced; this difference in protein expression was significant
before and after treatment with radiotherapy. Thus, we speculate
that the silencing of FANCD2 can enhance radiosensitivity through
the promotion of radiotherapy-induced apoptosis in HSC-4 cells,
which is consistent with the experimental results of Van Der
Heijden et al (26). We
also speculate that this mechanism may be related to the expression
of Bax and Bcl2, which regulate the apoptosis pathway in
mitochondria.
p38 mitogen-activated protein kinase (MAPK) is one
of the members of the highly conserved threonine/serine
mitogen-activated protein kinase family, and studies have confirmed
that p38 MAPK is involved in apoptosis, cell differentiation, the
cell cycle and cell senescence (27). The inhibition of the activation of
the p38 signal transduction pathway can induce apoptosis (28). p38 MAPK could promote apoptosis of
lung adenocarcinoma cells via the regulation of the expression of
the apoptosis-related proteins Bax and Bcl2 (29). The downregulation of p38 MAPK could
reverse the radiotherapy resistance caused by the overexpression of
miRNA (30). The present study
found that after FANCD2 was silenced by shRNA interference, the
expression of the p38 protein in the cells of the experimental
group was decreased, while the expression of the p-p38 protein was
increased compared with the expression in the control groups. After
radiotherapy, the expression of the p38 and p-p38 proteins in the
HSC-4 cells in the experimental group showed a consistent trend
similar to that observed before radiotherapy. This difference in
protein expression before and after radiotherapy was significant,
which was consistent with the results of animal experiments.
Therefore, the silencing of FANCD2 can enhance the
radiotherapy-induced apoptosis of HSC-4 cells via the regulation of
the expression of p38 MAPK pathway-related proteins, leading to
increased radiosensitivity.
In summary, the present study confirms that shRNA
can stably knock down FANCD2 expression in HSC-4 cells. In both
in vivo and in vitro experiments, we found that the
silencing of FANCD2 by shRNA interference enhances the sensitivity
of metastatic lymph node-derived HSC-4 HNSCC cells to radiotherapy.
We demonstrate that the mechanism of increased radiosensitivity may
be related to the activation of the p38 MAPK signaling pathway and
the regulation of the expression of the apoptosis-related proteins
Bax and Bcl2, which has provided potential new molecular targets
for the treatment of HNSCC. Finally, this study provided a
theoretical basis for further clinical studies.
Acknowledgments
The present study was supported by grants from the
Major Project of Education Department of Sichuan Province
(12ZA076), the Health and Family Planning Commission of Sichuan
Province (120352), the Scientific Research Project of the
Affiliated Hospital of Luzhou Medical College for Returned Chinese
Scholars (2013-60-2), the Scientific Research Project of the
Southwest Medical University, and the Joint Research Project of
Science Technology Department of Sichuan Province - Office of
Science Technology of Luzhou - Luzhou Medical College (14JC0182),
China.
References
|
1
|
Wise-Draper TM, Draper DJ, Gutkind JS,
Molinolo AA, Wikenheiser-Brokamp KA and Wells SI: Future directions
and treatment strategies for head and neck squamous cell
carcinomas. Transl Res. 160:167–177. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lee JH, Song JH, Lee SN, Kang JH, Kim MS,
Sun DI and Kim YS: Adjuvant postoperative radiotherapy with or
without chemotherapy for locally advanced squamous cell carcinoma
of the head and neck: The importance of patient selection for the
postoperative chemoradiotherapy. Cancer Res Treat. 45:31–39. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Argiris A, Karamouzis MV, Raben D and
Ferris RL: Head and neck cancer. Lancet. 371:1695–1709. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Thariat J, Milas L and Ang KK: Integrating
radiotherapy with epidermal growth factor receptor antagonists and
other molecular therapeutics for the treatment of head and neck
cancer. Int J Radiat Oncol Biol Phys. 69:974–984. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kupfer GM: Fanconi anemia: A signal
transduction and DNA repair pathway. Yale J Biol Med. 86:491–497.
2013.PubMed/NCBI
|
|
6
|
Kutler DI, Auerbach AD, Satagopan J,
Giampietro PF, Batish SD, Huvos AG, Goberdhan A, Shah JP and Singh
B: High incidence of head and neck squamous cell carcinoma in
patients with Fanconi anemia. Arch Otolaryngol Head Neck Surg.
129:106–112. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Virts EL, Jankowska A, Mackay C, Glaas MF,
Wiek C, Kelich SL, Lottmann N, Kennedy FM, Marchal C, Lehnert E, et
al: AluY-mediated germline deletion, duplication and somatic stem
cell reversion in UBE2T defines a new subtype of Fanconi anemia.
Hum Mol Genet. 24:5093–5108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Rickman KA, Lach FP, Abhyankar A, Donovan
FX, Sanborn EM, Kennedy JA, Sougnez C, Gabriel SB, Elemento O,
Chandrasekharappa SC, et al: Deficiency of UBE2T, the E2 ubiquitin
ligase necessary for FANCD2 and FANCI ubiquitination, causes FA-T
subtype of Fanconi anemia. Cell Rep. 12:35–41. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kee Y and D'Andrea AD: Molecular
pathogenesis and clinical management of Fanconi anemia. J Clin
Invest. 122:3799–3806. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kim H and D'Andrea AD: Regulation of DNA
cross-link repair by the Fanconi anemia/BRCA pathway. Genes Dev.
26:1393–1408. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Jo U and Kim H: Exploiting the Fanconi
anemia pathway for targeted anti-cancer therapy. Mol Cells.
38:669–676. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wang LC and Gautier J: The Fanconi anemia
pathway and ICL repair: Implications for cancer therapy. Crit Rev
Biochem Mol Biol. 45:424–439. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Burkitt K and Ljungman M: Phenylbutyrate
interferes with the Fanconi anemia and BRCA pathway and sensitizes
head and neck cancer cells to cisplatin. Mol Cancer. 7:242008.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhu J, Su F, Mukherjee S, Mori E, Hu B and
Asaithamby A: FANCD2 influences replication fork processes and
genome stability in response to clustered DSBs. Cell Cycle.
14:1809–1822. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Burdak-Rothkamm S, Rothkamm K, McClelland
K, Al Rashid ST and Prise KM: BRCA1, FANCD2 and Chk1 are potential
molecular targets for the modulation of a radiation-induced DNA
damage response in bystander cells. Cancer Lett. 356(2 Pt B):
454–461. 2015. View Article : Google Scholar
|
|
16
|
Lyakhovich A and Surralles J: FANCD2
depletion sensitizes cancer cells repopulation ability in vitro.
Cancer Lett. 256:186–195. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Jeong YK, Kim MS, Lee JY, Kim EH and Ha H:
Metformin radiosensitizes p53-deficient colorectal cancer cells
through induction of G2/M arrest and inhibition of DNA repair
proteins. PLoS One. 10:e01435962015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Liu J, Ge YY, Zhu HC, Yang X, Cai J, Zhang
C, Lu J, Zhan LL, Qin Q, Yang Y, et al: Fenofibrate increases
radiosensitivity in head and neck squamous cell carcinoma via
inducing G2/M arrest and apoptosis. Asian Pac J Cancer Prev.
15:6649–6655. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Duangmano S, Sae-Lim P, Suksamrarn A,
Patmasiriwat P and Domann FE: Cucurbitacin B causes increased
radiation sensitivity of human breast cancer cells via G2/M cell
cycle arrest. J Oncol. 2012:6016822012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ashimori N, Zeitlin BD, Zhang Z, Warner K,
Turkienicz IM, Spalding AC, Teknos TN, Wang S and Nör JE: TW-37, a
small-molecule inhibitor of Bcl-2, mediates S-phase cell cycle
arrest and suppresses head and neck tumor angiogenesis. Mol Cancer
Ther. 8:893–903. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Taniguchi T, Garcia-Higuera I, Andreassen
PR, Gregory RC, Grompe M and D'Andrea AD: S-phase-specific
interaction of the Fanconi anemia protein, FANCD2, with BRCA1 and
RAD51. Blood. 100:2414–2420. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Farnebo L, Jerhammar F, Ceder R, Grafström
RC, Vainikka L, Thunell L, Grénman R, Johansson AC and Roberg K:
Combining factors on protein and gene level to predict
radioresponse in head and neck cancer cell lines. J Oral Pathol
Med. 40:739–746. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Findley HW, Gu L, Yeager AM and Zhou M:
Expression and regulation of Bcl-2, Bcl-xl, and Bax correlate with
p53 status and sensitivity to apoptosis in childhood acute
lymphoblastic leukemia. Blood. 89:2986–2993. 1997.PubMed/NCBI
|
|
24
|
Oltvai ZN, Milliman CL and Korsmeyer SJ:
Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that
accelerates programmed cell death. Cell. 74:609–619. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Schon EA and Manfredi G: Neuronal
degeneration and mitochondrial dysfunction. J Clin Invest.
111:303–312. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Van Der Heijden MS, Brody JR and Kern SE:
Functional screen of the fanconi anemia pathway in cancer cells by
Fancd2 immunoblot. Cancer Biol Ther. 3:534–537. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Raman M, Chen W and Cobb MH: Differential
regulation and properties of MAPKs. Oncogene. 26:3100–3112. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Chun J, Joo EJ, Kang M and Kim YS:
Platycodin D induces anoikis and caspase-mediated apoptosis via p38
MAPK in AGS human gastric cancer cells. J Cell Biochem.
114:456–470. 2013. View Article : Google Scholar
|
|
29
|
Su JC, Lin KL, Chien CM, Lu CM, Chen YL,
Chang LS and Lin SR: Novel indoloquinoline derivative, IQDMA,
induces G(2)/M phase arrest and apoptosis in A549 cells through
JNK/p38 MAPK signaling activation. Life Sci. 85:505–516. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Salim H, Akbar NS, Zong D, Vaculova AH,
Lewensohn R, Moshfegh A, Viktorsson K and Zhivotovsky B: miRNA-214
modulates radiotherapy response of non-small cell lung cancer cells
through regulation of p38MAPK, apoptosis and senescence. Br J
Cancer. 107:1361–1373. 2012. View Article : Google Scholar : PubMed/NCBI
|