Introduction
Gastric cancer is the third leading cause of
tumor-related death, killing >730,000 people a year worldwide
(1). However, the incidence of
gastric cancer ranks only fifth globally, reaching 1,000,000 new
cases/year (2). Notably, there are
differences in the incidence of gastric cancer in different regions
and races. In the United States, the incidence of gastric cancer in
White American patients is low, with 25,000 new cases of gastric
cancer each year, but it causes 10,700 deaths/year (3). Moreover, 75% of White American
patients with gastric cancer have progressive disease, and the 5
year survival rate is only 30.4% (3). For African American patients, the
incidence rate for gastric cancer is 11.8 (95% CI, 10.3-13.2) per
100,000 person-years (3). In
addition, >70% of new cases of gastric cancer occur in
developing countries, with ~50% of cases in eastern Asia, mainly in
China, and the occurrence and mortality of gastric cancer in China
accounts for 42.6 and 45.0% of the worldwide total, respectively
(4). China ranks fifth in gastric
cancer incidence and sixth in mortality in the world among 183
countries (4). Overall, there are
notable sex differences in the occurrence of gastric cancer, with a
previous study indicating that gastric cancer occurs significantly
more frequently in male patients than in female patients, with a
ratio of 2:1 (5). However, the
impact of sex differences on gastric cancer is not clear, nor is it
clear whether sex differences occur in gastric cancer patients of
different races. Overall, research in this area is still lacking,
and further studies are needed to analyze such differences.
Clarification of the factors that lead to sex and ethnic
differences in gastric cancer may offer new insight into the key
pathways involved.
To obtain survival and tumor-related data from
gastric cancer patients of different ethnic backgrounds, data from
two large centers, the Gastric Cancer Research of Sun Yat-Sen
University and the Surveillance Epidemiology and End Results (SEER)
databases, were utilized. The SEER database is part of the National
Cancer Institute and contains records of the pathogenesis,
treatment, pathology and prognosis of millions of patients in the
United States from 1973 onwards (6). In addition to analyzing differences
in patient survival and prognosis, mRNA and DNA methylation data
from gastric cancer tissue samples were obtained from The Cancer
Genome Atlas (TCGA) database to identify relevant molecular
mechanisms affecting sex differences among different races.
Materials and methods
Study subjects
During the period 1973-2013, 150,264 patients with
gastric cancer were registered in the SEER database. In the present
study, patients with gastric cancer and complete information were
included as the study subjects. Patients with no pathological
diagnosis (n=526), patients without pathological staging
(n=21,646), patients with tumors at the esophagogastric junction
(n=849), patients with neuroendocrine tumors (n=604), patients with
a rare pathological type (n=7,513), patients with unknown surgical
status (n=4,683), patients with unknown radiotherapy (n=2,664) and
patients with unknown ethnicity or ethnic origin (n=21,626) were
excluded. Ultimately, 90,153 patients with gastric cancer were
included. Among them, there were 76,942 White patients and 13,211
Black patients, with 49,614 males and 27,328 females in the former
and 7,985 males and 5,226 females in the latter. The data obtained
from the SEER database included patient demographics, primary tumor
location, tumor morphology, diagnosis phase, first course of
treatment and tracked patient follow up (https://seer.cancer.gov/).
The Center for Gastric Cancer Research of Sun
Yat-Sen University collected follow up data for patients with
gastric cancer between 1994 and 2017, including general demographic
data, location of primary tumor, family history, Borrmann
classification, surgical status and tumor characteristics. The
present study included 3,113 patients with gastric cancer who were
enrolled in the Center for Gastric Cancer Research at Sun Yat-Sen
University between 1994 and 2017, and had complete follow-up
information. Patients with no pathological diagnosis (n=201),
patients without pathological staging (n=59), patients with rare
pathological types (n=34) and patients with unknown surgical status
(n=39) were excluded (2,780 patients; Fig. 1). In the Asian population, there
were 1,845 male patients and 935 female patients.
TCGA contained information for 396 patients with
gastric cancer. Patients with unknown ethnicities were excluded
(n=55), leading to a total of 341 patients, including 256 White
patients, 11 Black patients and 74 Asian patients.
Clinicopathological parameters and ethics
statement
Because the American Joint Committee on Cancer
(AJCC) staging system was not used before 2004, patients in the
SEER data base before 2004 were mainly classified according to SEER
staging (7). SEER staging includes
localized, regional and distant classifications. For patients after
2004, TNM staging was performed according to the 2004 AJCC standard
(7), and this applied to most of
the data from Sun Yat-Sen University Center for Gastric Cancer
Research (7). Patients from the
Sun Yat-Sen University Center for Gastric Cancer Research before
2004 were reclassified according to their data. Age was divided
into categories of ≥65 years old and <65 years old. The
diagnosis of gastric cancer was based on the standards of the
International Classification of Diseases for Oncology (ICD-O). None
of the information obtained from the SEER database contains any
identifiers, and all information is public. Due to the
retrospective nature of the study, informed consent from the
patients was not required. This analysis did not involve
interaction with human subjects or the use of personally
identifiable information. Prior to analysis, the patient
records/information was anonymized and deidentified, and these
methods were performed in accordance with the study protocol
approved by the ethics committee. The Ethics Committee of the First
Affiliated Hospital of Sun Yat-Sen University approved the use of
the aforementioned databases in the present study.
Screening of differentially expressed
genes (DEGs)
The RNA-Seq by Expectation Maximization method was
used to normalize the level 3 transcriptome data of the dataset,
and all gene expression values were logarithmically trans formed.
Approximate data were normally distributed after quantile
normalization (8). In the present
study, the R program package limma v3.28.14 (https://www.bioconductor.org/packages/devel/bioc/vignettes/limma/inst/doc/users-guide.pdf)
was used to analyze gene expression data for male and female
patient of different ethnicities, and mRNAs satisfying P<0.01,
false discovery rate (FDR) <0.01, and |log2 fold change
(FC)|>2 were further investigated, where P<0.05 indicates
that the hypothesis test was statistically significant and FDR is a
control index for the hypothesis test error rate. As an evaluation
index of the selected differential genes, the number of false
rejections was proportional to the number of rejected null
hypotheses. FC is generally used to describe the degree of change
from an initial to a final value. In the present study, the ratio
of the tumor tissue gene expression value to the normal tissue gene
expression value, also called the difference multiple, was used.
Heat maps of the differential genes were constructed in R
(https://cran.r-project.org/web/packages/pheatmap/pheatmap.pdf)
for easy visualcomparison.
Preprocessing of DNA methylation chip
data
Whole-genome DNA methylation analysis of the data
set was performed using the Illumina Infinium Human Methylation 450
Bead Chip method (Illumina, Inc.), and raw data (primary data in
TCGA) were processed using the R package minfi version 1.20.0
(9). Subsequent background
subtraction, quantile normalization and quality control were
performed. A low-quality probe was removed if it met the following
criteria: i) An undetectable rate (P>0.05) in ≥5% of the total
sample; ii) a variance coefficient <5%; or iii)
single-nucleotide polymorphisms (SNPs) located in the detected CpG
dinucleotide (10). β-mixture
quantile normalization was used for further Type I and II probe
correction (11). The DNA
methylation data of the validation set were standardized (12), and the methylation and expression
data of the core genes were correlated. VanderWeele's mediation
analysis was used to investigate whether the prognostic effects of
seven DNA methylation sites were mediated by affecting the
corresponding mRNA expression (13).
Functional enrichment and pathway
analysis of DEGs
Gene Ontology (GO) analysis was performed on the
identified DEGs using the Database for Annotation, Visualization
and Integrated Discovery (DAVID 6.8; https://david.ncifcrf.gov/), which provides a
comprehensive and systematic annotation tool for elucidating the
biological significance of genes. GO analysis includes molecular
function, biological process and cellular component aspects
(14). DEGs detected using DESeq2
software v3.6 (http://bioconductor.org/packages/stats/bioc/DESeq2/)
were used as enrichment prospects, human database reference genes
were used as the enrichment analysis background, P<0.05 was used
to screen significantly enriched GO terms, and the number of DEGs
in each significantly enriched GO term was counted (15). The results were visualized using
the R Goplot package v1.0.2 (https://www.rdocumentation.org/packages/GOplot/versions/1.0.2).
Kaplan-Meier plotter
The Kaplan-Meier plotter (http://kmplot.com/analysis/) is a platform that
contains gene expression information for 10 tumors and 1,065
clinical survival data points for patients with gastric cancer.
This website was used to obtain core gene expression and patient
survival prognosis information in an effort to identify the reasons
for differences in the survival of gastric cancer patients of
different races and sexes (16).
To assess the prognostic value of a particular gene, patient
samples were divided into two groups based on the median expression
level of the gene (high vs. low expression). Core genes were
uploaded to the database to obtain a Kaplan-Meier survival map and
this map was used to analyze the overall survival (OS) of patients
with gastric cancer. The 95% CI, P-value and hazard ratio (HR) were
calculated and are displayed on the graphs.
Statistical analyses
Differences in the clinicopathological parameters
between groups were calculated using Pearson's χ2 test.
Survival analysis was performed to calculate cancer-specific
survival (CSS) and OS. CSS was defined as the time from the initial
diagnosis of primary gastric cancer to the death of a patient due
to the tumor. OS was defined as the time from the initial diagnosis
of primary gastric cancer to death for any reason. The survival
rate was statistically examined using the Kaplan-Meier method, and
the differences in survival curves were statistically analyzed
using the log-rank test. Propensity analysis is a statistical
method used to generate matched patients when comparing long term
survival in observational nonrandomized controlled trials. A
one-to-one match of nonpermutation was performed using the nearest
neighbor matching method (17).
All factors with an impact on survival prognosis were included in
the propensity score matching (Figs.
S1-S5). The caliper width was 0.05 times the standard deviation
of the logit of the propensity score, and deviations of the
co-construction variable of >99% were eliminated (17). Spearman's rank correlation (rs) was
used to investigate the relationship between methylation and gene
expression. Univariate analysis of continuous and binary
categorical dependent variables was performed using simple linear
analysis and logistic regression analysis. All statistical analyses
were performed using SPSS Statistical Software version 20.0 (IBM
Corp.) and R version 3.3.0 (The R Foundation; https://www.rproject.org/foundation/).
Two-tailed P<0.05 was considered to indicate a statistically
significant difference.
Results
Clinicopathological characteristics of
the study subjects
The total number of subjects was 93,278, among which
2,857 were Asian, 13,223 were Black and 77,198 were White. In the
Asian population, there were 1,893 male patients and 964 female
patients, and the male to female ratio was 1.96. In the Black
population, there were 7,993 male and 5,241 female patients, and
the male to female ratio was 1.53. There were 49,766 male and
27,432 female patients with gastric cancer in the White population,
with a male to female ratio of 1.81. Compared with White male
patients, White female patients had a lower degree of
differentiation (59.43 vs. 63.70%), a higher incidence of signet
ring cell carcinoma (9.91 vs. 15.28%), a lower incidence of lymph
node metastasis (35.35 vs. 28.22%), T1 stages (23.08 vs. 24.37%)
and decreased distant metastasis (34.91 vs. 31.32%) (Table I). Moreover, the primary site of
the tumor more frequently occurred in the gastric antrum (13.59 vs.
21.62%), and more female patients underwent radical gastrectomy
(46.47 vs. 49.45%) (Tables I and
II). Among Black patients, the
degree of differentiation, the incidence of signet ring cell
carcinoma, lymph node metastasis and T stage were similar to those
of White patients. However, the primary site of gastric cancer in
Black patients was more likely to be associated with the gastric
antrum; additionally, the incidence of cancer in the gastric antrum
in male and female patients was similar (27.19 vs. 29.80%), and
fewer patients underwent radical gastrectomy (51.68 vs. 49.45%)
(Table II). A rare pathological
type occurred more frequently in female patients than in male
patients (12.40 vs. 14.77%) (Tables
I and II). Table III illustrates the
clinicopathological features of Asian patients with gastric cancer.
The incidence of the primary tumor site in the corpus was higher
among females (16.15 vs. 30.70%), as was the incidence of diffuse
infiltrative gastric cancer (9.11 vs. 14.97%). The general
characteristics of patients with gastric cancer of different ethnic
backgrounds showed that the effects of different ethnicities on
gastric cancer are varied.
 | Table IDemographics according to gender for
White and Black gastric cancer patients (1973-2003). |
Table I
Demographics according to gender for
White and Black gastric cancer patients (1973-2003).
| Characteristic
No. | White gastric
cancer patients
| Black gastric
cancer patients
|
|---|
Unadjusted
| Adjusted
| Unadjusted
| Adjusted
|
|---|
| M | SD/% | F | SD/% | P‑value | M | SD/% | F | SD/% | M | SD/% | F | SD/% | P‑value | M | SD/% | F | SD/% |
|---|
| 28,775 | 63.86 | 16,281 | 36.14 | | 9,975 | 50 | 9,975 | 50 | 4,469 | 61.40 | 2,810 | 38.60 | | 1,188 | 50 | 1,188 | 50 |
|---|
| Age, years | | | | | | | | | | | | | | | | | | |
| ≥65 | 10,184 | 35.39 | 4,290 | 26.35 | <0.01 | 3,028 | 30.36 | 3,028 | 30.36 | 1959 | 43.84 | 953 | 33.91 | <0.01 | 437 | 36.78 | 437 | 36.78 |
| <65 | 18,591 | 64.61 | 11,991 | 73.65 | | 6,947 | 69.64 | 6,947 | 69.64 | 2,510 | 56.16 | 1,857 | 66.09 | | 751 | 63.22 | 751 | 63.22 |
| Tumor grade | | | | | | | | | | | | | | | | | | |
| 1 | 1,910 | 6.64 | 1,037 | 6.37 | <0.01 | 510 | 5.11 | 510 | 5.11 | 371 | 8.30 | 217 | 7.72 | <0.01 | 49 | 4.12 | 49 | 4.12 |
| 2 | 8,151 | 28.33 | 3,901 | 23.96 | | 2461 | 24.67 | 2461 | 24.67 | 1324 | 29.63 | 807 | 28.72 | | 334 | 28.11 | 334 | 28.11 |
| 3 | 17,101 | 59.43 | 10,371 | 63.70 | | 6,597 | 66.14 | 6,597 | 66.14 | 2,575 | 57.62 | 1,664 | 59.22 | | 786 | 66.16 | 786 | 66.16 |
| 4 | 1,613 | 5.61 | 972 | 5.97 | | 407 | 4.08 | 407 | 4.08 | 199 | 4.45 | 122 | 4.34 | | 19 | 1.60 | 19 | 1.60 |
| Primary site | | | | | | | | | | | | | | | | | | |
| Cardia | 10,784 | 37.48 | 2,727 | 16.75 | <0.01 | 2,240 | 22.46 | 2,240 | 22.46 | 532 | 11.90 | 227 | 8.08 | <0.01 | 86 | 7.24 | 86 | 7.24 |
| Fundus | 1,257 | 4.37 | 799 | 4.91 | | 445 | 4.46 | 445 | 4.46 | 188 | 4.21 | 118 | 4.20 | | 31 | 2.61 | 31 | 2.61 |
| Body | 1,662 | 5.78 | 1,328 | 8.16 | | 719 | 7.21 | 719 | 7.21 | 333 | 7.45 | 238 | 8.47 | | 72 | 6.06 | 72 | 6.06 |
| Gastric
antrum | 4,028 | 14.00 | 3,523 | 21.64 | | 2,007 | 20.12 | 2,007 | 20.12 | 1,164 | 26.05 | 829 | 29.50 | | 417 | 35.10 | 417 | 35.10 |
| Pylorus | 863 | 3.00 | 649 | 3.99 | | 321 | 3.22 | 321 | 3.22 | 194 | 4.34 | 158 | 5.62 | | 53 | 4.46 | 53 | 4.46 |
| Lesser
curvature | 2,542 | 8.83 | 1,676 | 10.29 | | 971 | 9.73 | 971 | 9.73 | 598 | 13.38 | 343 | 12.21 | | 157 | 13.22 | 157 | 13.22 |
| Greater
curvature | 1,148 | 3.99 | 860 | 5.28 | | 442 | 4.43 | 442 | 4.43 | 230 | 5.15 | 125 | 4.45 | | 34 | 2.86 | 34 | 2.86 |
| Overlapping
lesion | 2,422 | 8.42 | 1,737 | 10.67 | | 987 | 9.89 | 987 | 9.89 | 494 | 11.05 | 277 | 9.86 | | 116 | 9.76 | 116 | 9.76 |
| Stomach | 4,069 | 14.14 | 2,982 | 18.32 | | 1,843 | 18.48 | 1,843 | 18.48 | 736 | 16.47 | 495 | 17.62 | | 222 | 18.69 | 222 | 18.69 |
| Histology | | | | | | | | | | | | | | | | | | |
|
Adenocarcinoma | 20,438 | 71.03 | 10,229 | 62.83 | <0.01 | 7,114 | 71.32 | 7,114 | 71.32 | 3,032 | 67.85 | 1,770 | 62.99 | <0.01 | 929 | 78.20 | 929 | 78.20 |
| Signet ring cell
carcinoma | 2,852 | 9.91 | 2,487 | 15.28 | | 1,403 | 14.07 | 1,403 | 14.07 | 465 | 10.41 | 399 | 14.20 | | 147 | 12.37 | 147 | 12.37 |
| Mucinous
carcinoma | 1,218 | 4.23 | 628 | 3.86 | | 214 | 2.15 | 214 | 2.15 | 203 | 4.54 | 109 | 3.88 | | 13 | 1.09 | 13 | 1.09 |
| Linitis
plastica | 523 | 1.82 | 536 | 3.29 | | 220 | 2.21 | 220 | 2.21 | | | | | | | | | |
| Adenocarcinoma
intestinal | 1,022 | 3.55 | 535 | 3.29 | | 217 | 2.18 | 217 | 2.18 | 215 | 4.81 | 117 | 4.16 | | 26 | 2.19 | 26 | 2.19 |
| Other | 2,722 | 9.46 | 1,866 | 11.46 | | 807 | 8.09 | 807 | 8.09 | 554 | 12.40 | 415 | 14.77 | | 73 | 6.14 | 73 | 6.14 |
| SEER historic
stage | | | | | | | | | | | | | | | | | | |
| Distant | 10,126 | 35.19 | 5,253 | 32.26 | <0.01 | 3,455 | 34.64 | 3,455 | 34.64 | 1,677 | 37.53 | 889 | 31.64 | <0.01 | 422 | 35.52 | 422 | 35.52 |
| Localized | 5,579 | 19.39 | 3,566 | 21.90 | | 2,030 | 20.35 | 2,030 | 20.35 | 897 | 20.07 | 650 | 23.13 | | 222 | 18.69 | 222 | 18.69 |
| Regional | 10,399 | 36.14 | 5,525 | 33.94 | | 3,456 | 34.65 | 3,456 | 34.65 | 1,450 | 32.45 | 911 | 32.42 | | 404 | 34.01 | 404 | 34.01 |
| Unstaged | 2,671 | 9.28 | 1,973 | 12.12 | | 1,034 | 10.37 | 1,034 | 10.37 | 445 | 9.96 | 360 | 12.81 | | 120 | 10.10 | 120 | 10.10 |
| Sequence with
surgery | | | | | | | | | | | | | | | | | | |
| No radiation | 25,482 | 88.56 | 15,077 | 92.60 | <0.01 | 9,273 | 92.96 | 9,273 | 92.96 | 4,088 | 91.47 | 2,577 | 91.71 | 0.94 | 1,121 | 94.36 | 1,121 | 94.36 |
| Radiation after
surgery | 690 | 2.40 | 149 | 0.92 | | 74 | 0.74 | 74 | 0.74 | 355 | 7.94 | 217 | 7.72 | | 66 | 5.56 | 66 | 5.56 |
| Radiation prior to
surgery | 2,603 | 9.05 | 1,055 | 6.48 | | 628 | 6.30 | 628 | 6.30 | 26 | 0.58 | 16 | 0.57 | | 1 | 0.08 | 1 | 0.08 |
| Gastrectomy | | | | | | | | | | | | | | | | | | |
| No | 12,089 | 42.01 | 6,781 | 41.65 | 0.46 | 4,192 | 42.03 | 4,192 | 42.03 | 1,915 | 42.85 | 1,179 | 41.96 | 0.45 | 508 | 42.76 | 508 | 42.76 |
| Yes | 16,686 | 57.99 | 9,500 | 58.35 | | 5,783 | 57.97 | 5,783 | 57.97 | 2,554 | 57.15 | 1,631 | 58.04 | | 680 | 57.24 | 680 | 57.24 |
| Radiation | | | | | | | | | | | | | | | | | | |
| No | 22,022 | 76.53 | 13,783 | 84.66 | <0.01 | 8,532 | 85.53 | 8,532 | 85.53 | 3,701 | 82.81 | 2,381 | 84.73 | 0.03 | 1,074 | 90.40 | 1,074 | 90.40 |
| Yes | 6,753 | 23.47 | 2,498 | 15.34 | | 1,443 | 14.47 | 1,443 | 14.47 | 766 | 17.14 | 429 | 15.27 | | 114 | 9.60 | 114 | 9.60 |
 | Table IIDemographics according to gender for
White and Black GC patients (2004-2013). |
Table II
Demographics according to gender for
White and Black GC patients (2004-2013).
| Characteristic
No. | White GC patients
| Black GC patients
|
|---|
Unadjusted
| Adjusted
| Unadjusted
| Adjusted
|
|---|
| M | SD/% | F | SD/% | P‑value | M | SD/% | F | SD/% | M | SD/% | F | SD/% | P‑value | M | SD/% | F | SD/% |
|---|
| 20,839 | 65.24 | 11,047 | 34.76 | | 2,704 | 50 | 2,704 | 50 | 3,516 | 59.24 | 2,416 | 40.76 | | 183 | 50 | 183 | 50 |
|---|
| Age, years | | | | | | | | | | | | | | | | | | |
| ≥65 | 8,022 | 38.50 | 3,567 | 32.29 | <0.01 | 812 | 30.03 | 812 | 30.03 | 2,522 | 71.73 | 1,739 | 71.98 | 0.89 | 155 | 84.70 | 155 | 84.70 |
| <65 | 12,871 | 61.76 | 7,480 | 67.71 | | 1,892 | 69.97 | 1,892 | 69.97 | 994 | 28.27 | 680 | 28.15 | | 28 | 15.30 | 28 | 15.30 |
| Tumor grade | | | | | | | | | | | | | | | | | | |
| 1 | 1,017 | 4.88 | 600 | 5.43 | <0.01 | 105 | 3.88 | 105 | 3.88 | 175 | 4.98 | 129 | 5.34 | <0.01 | 4 | 2.19 | 4 | 2.19 |
| 2 | 6,025 | 28.91 | 2,533 | 22.93 | | 732 | 27.07 | 732 | 27.07 | 1,079 | 30.69 | 653 | 27.03 | | 58 | 31.69 | 58 | 31.69 |
| 3 | 13,327 | 63.95 | 7,633 | 69.10 | | 1,859 | 68.75 | 1,859 | 68.75 | 2,189 | 62.26 | 1,576 | 65.23 | | 121 | 66.12 | 121 | 66.12 |
| 4 | 524 | 2.51 | 281 | 2.54 | | 8 | 0.30 | 8 | 0.30 | 73 | 2.08 | 61 | 2.52 | | 0 | 0.00 | 0 | 0.00 |
| Primary site | | | | | | | | | | | | | | | | | | |
| Cardia | 9,778 | 46.92 | 2,620 | 23.72 | <0.01 | 1,087 | 40.20 | 1,087 | 40.20 | 579 | 16.47 | 252 | 10.43 | <0.01 | 15 | 8.20 | 15 | 8.20 |
| Fundus | 847 | 4.06 | 507 | 4.59 | | 67 | 2.48 | 67 | 2.48 | 139 | 3.95 | 94 | 3.89 | | 2 | 1.09 | 2 | 1.09 |
| Body | 1,515 | 7.27 | 1,246 | 11.28 | | 221 | 8.17 | 221 | 8.17 | 365 | 10.38 | 289 | 11.96 | | 25 | 13.66 | 25 | 13.66 |
| Gastric
antrum | 2,833 | 13.59 | 2,388 | 21.62 | | 598 | 22.12 | 598 | 22.12 | 956 | 27.19 | 720 | 29.80 | | 71 | 38.80 | 71 | 38.80 |
| Pylorus | 448 | 2.15 | 352 | 3.19 | | 32 | 1.18 | 32 | 1.18 | 145 | 4.12 | 125 | 5.17 | | 1 | 0.55 | 1 | 0.55 |
| Lesser
curvature | 1,244 | 5.97 | 821 | 7.43 | | 131 | 4.84 | 131 | 4.84 | 376 | 10.69 | 241 | 9.98 | | 15 | 8.20 | 15 | 8.20 |
| Greater
curvature | 618 | 2.97 | 499 | 4.52 | | 53 | 1.96 | 53 | 1.96 | 141 | 4.01 | 100 | 4.14 | | 2 | 1.09 | 2 | 1.09 |
| Overlapping
lesion | 1,368 | 6.56 | 965 | 8.74 | | 134 | 4.96 | 134 | 4.96 | 309 | 8.79 | 218 | 9.02 | | 10 | 5.46 | 10 | 5.46 |
| Stomach | 2,242 | 10.76 | 1,849 | 16.74 | | 381 | 14.09 | 381 | 14.09 | 506 | 14.39 | 380 | 15.73 | | 42 | 22.95 | 42 | 22.95 |
| Histology | | | | | | | | | | | | | | | | | | |
|
Adenocarcinoma | 12,774 | 61.30 | 5,445 | 49.29 | <0.01 | 1,842 | 68.12 | 1,842 | 68.12 | 2,006 | 57.05 | 1,211 | 50.12 | <0.01 | 127 | 69.40 | 127 | 69.40 |
| Signet ring cell
carcinoma | 3,262 | 15.65 | 2,672 | 24.19 | | 534 | 19.75 | 534 | 19.75 | 502 | 14.28 | 502 | 20.78 | | 20 | 10.93 | 20 | 10.93 |
| Mucinous
carcinoma | 1,338 | 6.42 | 834 | 7.55 | | 54 | 2.00 | 54 | 2.00 | 119 | 3.38 | 74 | 3.06 | | 1 | 0.55 | 1 | 0.55 |
| Adenocarcinoma,
intestinal type | 1,782 | 8.55 | 975 | 8.83 | | 158 | 5.84 | 158 | 5.84 | 438 | 12.46 | 283 | 11.71 | | 8 | 4.37 | 8 | 4.37 |
| Other | 1,737 | 8.34 | 1,121 | 10.15 | | 116 | 4.29 | 116 | 4.29 | 451 | 12.83 | 349 | 14.45 | | 27 | 14.75 | 27 | 14.75 |
| T stage | | | | | | | | | | | | | | | | | | |
| T1 | 4,810 | 23.08 | 2692 | 24.37 | <0.01 | 787 | 29.11 | 787 | 29.11 | 857 | 24.37 | 649 | 26.86 | <0.01 | 68 | 37.16 | 68 | 37.16 |
| T2a | 1,585 | 7.61 | 868 | 7.86 | | 177 | 6.55 | 177 | 6.55 | 265 | 7.54 | 181 | 7.49 | | 9 | 4.92 | 9 | 4.92 |
| T2b | 4,669 | 22.41 | 2,174 | 19.68 | | 527 | 19.49 | 527 | 19.49 | 789 | 22.44 | 486 | 20.12 | | 28 | 15.30 | 28 | 15.30 |
| T3 | 2,803 | 13.45 | 1,566 | 14.18 | | 270 | 9.99 | 270 | 9.99 | 410 | 11.66 | 294 | 12.17 | | 10 | 5.46 | 10 | 5.46 |
| T4 | 2,400 | 11.52 | 1,342 | 12.15 | | 199 | 7.36 | 199 | 7.36 | 499 | 14.19 | 324 | 13.41 | | 10 | 5.46 | 10 | 5.46 |
| TX | 4,626 | 22.20 | 2,405 | 21.77 | | 744 | 27.51 | 744 | 27.51 | 696 | 19.80 | 485 | 20.07 | | 58 | 31.69 | 58 | 31.69 |
| N stage | | | | | | | | | | | | | | | | | | |
| N0 | 8,187 | 39.29 | 4,827 | 43.70 | <0.01 | 1,325 | 49.00 | 1,325 | 49.00 | 1,464 | 41.64 | 4,827 | 43.70 | <0.01 | 106 | 57.92 | 106 | 57.92 |
| N1 | 7,366 | 35.35 | 3,118 | 28.22 | | 754 | 27.88 | 754 | 27.88 | 1,142 | 32.48 | 3,118 | 28.22 | | 35 | 19.13 | 35 | 19.13 |
| N2 | 1,660 | 7.97 | 898 | 8.13 | | 120 | 4.44 | 120 | 4.44 | 317 | 9.02 | 898 | 8.13 | | 6 | 3.28 | 6 | 3.28 |
| N3 | 649 | 3.11 | 386 | 3.49 | | 31 | 1.15 | 31 | 1.15 | 121 | 3.44 | 386 | 3.49 | | 0 | 0.00 | 0 | 0.00 |
| NX | 3,031 | 14.54 | 1,818 | 16.46 | | 474 | 17.53 | 474 | 17.53 | 472 | 13.42 | 1,818 | 16.46 | | 36 | 19.67 | 36 | 19.67 |
| M stage | | | | | | | | | | | | | | | | | | |
| M0 | 12,327 | 59.15 | 6,727 | 60.89 | <0.01 | 1,698 | 62.80 | 1,698 | 62.80 | 2,091 | 59.47 | 6,727 | 60.89 | <0.01 | 106 | 57.92 | 106 | 57.92 |
| M1 | 7,274 | 34.91 | 3,460 | 31.32 | | 805 | 29.77 | 805 | 29.77 | 1,218 | 34.64 | 3,460 | 31.32 | | 62 | 33.88 | 62 | 33.88 |
| MX | 1,292 | 6.20 | 860 | 7.78 | | 201 | 7.43 | 201 | 7.43 | 207 | 5.89 | 860 | 7.78 | | 15 | 8.20 | 15 | 8.20 |
| Tumor size | | | | | | | | | | | | | | | | | | |
| ≤1 cm | 11,071 | 53.13 | 5,608 | 50.76 | <0.01 | 1,415 | 52.33 | 1,415 | 52.33 | 1,870 | 53.19 | 5,608 | 50.76 | <0.01 | 88 | 48.09 | 88 | 48.09 |
| >1 cm | 9,822 | 47.13 | 5,439 | 49.24 | | 1,289 | 47.67 | 1,289 | 47.67 | 1,646 | 46.81 | 5,439 | 49.24 | | 95 | 51.91 | 95 | 51.91 |
| Sequence with
surgery | | | | | | | | | | | | | | | | | | |
| No radiation | 17,489 | 83.92 | 9,622 | 87.10 | <0.01 | 2,406 | 88.98 | 2,406 | 88.98 | 2,997 | 85.24 | 9,622 | 87.10 | <0.01 | 173 | 94.54 | 173 | 94.54 |
| Radiation after
surgery | 2,117 | 10.16 | 1,203 | 10.89 | | 202 | 7.47 | 202 | 7.47 | 465 | 13.23 | 1,203 | 10.89 | | 9 | 4.92 | 9 | 4.92 |
| Radiation prior to
surgery | 1,287 | 6.18 | 222 | 2.01 | | 96 | 3.55 | 96 | 3.55 | 54 | 1.54 | 222 | 2.01 | | 1 | 0.55 | 1 | 0.55 |
| Gastrectomy | | | | | | | | | | | | | | | | | | |
| No | 11,210 | 53.79 | 5,584 | 50.55 | <0.01 | 1,414 | 52.29 | 1,414 | 52.29 | 1,699 | 48.32 | 5,584 | 50.55 | <0.01 | 83 | 45.36 | 83 | 45.36 |
| Yes | 9,683 | 46.47 | 5,463 | 49.45 | | 1,290 | 47.71 | 1,290 | 47.71 | 1,817 | 51.68 | 5,463 | 49.45 | | 100 | 54.64 | 100 | 54.64 |
| Radiation | | | | | | | | | | | | | | | | | | |
| No | 14,062 | 67.48 | 8,518 | 77.11 | <0.01 | 2,173 | 80.36 | 2,173 | 80.36 | 2,616 | 74.40 | 8,518 | 77.11 | <0.01 | 171 | 93.44 | 171 | 93.44 |
| Yes | 6,831 | 32.78 | 2,529 | 22.89 | | 531 | 19.64 | 531 | 19.64 | 900 | 25.60 | 2,529 | 22.89 | | 12 | 6.56 | 12 | 6.56 |
 | Table IIIDemographics according to gender for
Asian GC patient (1994-2017). |
Table III
Demographics according to gender for
Asian GC patient (1994-2017).
| Characteristic | Unadjusted
| Adjusted
|
|---|
| M | SD/% | F | SD/% | P-value | M | SD/% | F | SD/% | P-value |
|---|
| No. | 1,845 | 66.37 | 935 | 33.63 | | 293 | 50 | 293 | 50 | |
| Age, years | | | | | | | | | | |
| ≥65 | 1,213 | 65.75 | 711 | 76.04 | <0.001 | 233 | 79.52 | 233 | 79.52 | 1.000 |
| <65 | 632 | 34.25 | 224 | 23.96 | | 60 | 20.48 | 60 | 20.48 | |
| Family history | | | | | | | | | | |
| No | 1,670 | 90.51 | 852 | 91.12 | 0.834 | 283 | 96.59 | 283 | 96.59 | |
| Yes | 168 | 9.11 | 83 | 8.88 | | 10 | 3.41 | 10 | 3.41 | |
| Tumor grade | | | | | | | | | | |
| 1 | 93 | 5.04 | 103 | 11.02 | <0.001 | 3 | 1.02 | 3 | 1.02 | 1.000 |
| 2 | 527 | 28.56 | 137 | 14.65 | | 51 | 17.41 | 51 | 17.41 | |
| 3 | 1,149 | 62.28 | 667 | 71.34 | | 223 | 76.11 | 223 | 76.11 | |
| 4 | 76 | 4.12 | 28 | 2.99 | | 16 | 5.46 | 16 | 5.46 | |
| Ascites | | | | | | | | | | |
| No | 1,630 | 88.35 | 815 | 87.17 | 0.329 | 289 | 98.63 | 289 | 98.63 | 1.000 |
| Serous fluid | 186 | 10.08 | 109 | 11.66 | | 4 | 1.37 | 4 | 1.37 | |
| Hemorrhagic | 29 | 1.57 | 11 | 1.18 | | 0 | 0.00 | 0 | 0.00 | |
| Primary site | | | | | | | | | | |
| Cardia | 630 | 34.15 | 227 | 24.28 | <0.001 | 68 | 23.21 | 68 | 23.21 | 1.000 |
| Corpus | 298 | 16.15 | 287 | 30.70 | | 75 | 25.60 | 75 | 25.60 | |
| Pylorus | 759 | 41.14 | 402 | 42.99 | | 150 | 51.19 | 150 | 51.19 | |
| Stomach | 58 | 3.14 | 19 | 2.03 | | 0 | 0.00 | 0 | 0.00 | |
| Histology | | | | | | | | | | |
|
Adenocarcinoma | 1,413 | 76.59 | 688 | 73.58 | <0.001 | 261 | 89.08 | 261 | 89.08 | 1.000 |
| Signet ring cell
carcinoma | 179 | 9.70 | 127 | 13.58 | | 29 | 9.90 | 29 | 9.90 | |
| Mucinous
carcinoma | 72 | 3.90 | 32 | 3.42 | | 1 | 0.34 | 1 | 0.34 | |
| Other | 181 | 9.81 | 88 | 9.41 | | 2 | 0.68 | 2 | 0.68 | |
| T stage (AJCC,
2004) | | | | | | | | | | |
| T1 | 204 | 11.06 | 132 | 14.12 | 0.137 | 40 | 13.65 | 40 | 13.65 | |
| T2 | 237 | 12.85 | 119 | 12.73 | | 25 | 8.53 | 25 | 8.53 | |
| T3 | 848 | 45.96 | 414 | 44.28 | | 170 | 58.02 | 170 | 58.02 | |
| T4 | 556 | 30.14 | 270 | 28.88 | | 58 | 19.80 | 58 | 19.80 | |
| N stage (AJCC,
2004) | | | | | | | | | | |
| N0 | 414 | 22.44 | 212 | 22.67 | 0.532 | 68 | 23.21 | 68 | 23.21 | 1.000 |
| N1 | 770 | 41.73 | 403 | 43.10 | | 157 | 53.58 | 157 | 53.58 | |
| N2 | 645 | 34.96 | 316 | 33.80 | | 68 | 23.21 | 68 | 23.21 | |
| N3 | 16 | 0.87 | 4 | 0.43 | | 0 | 0.00 | 0 | 0.00 | |
| M stage (AJCC,
2004) | | | | | | | | | | |
| M0 | 1,389 | 75.28 | 713 | 76.26 | 0.492 | 271 | 92.49 | 271 | 92.49 | 1.000 |
| M1 | 419 | 22.71 | 209 | 22.35 | | 22 | 7.51 | 22 | 7.51 | |
| MX | 37 | 2.01 | 13 | 1.39 | | 0 | 0.00 | 0 | 0.00 | |
| Borrmann | | | | | | | | | | |
| Massive type | 102 | 5.53 | 32 | 3.42 | <0.001 | 5 | 1.71 | 5 | 1.71 | 1.000 |
| Localized
type | 436 | 23.63 | 211 | 22.57 | | 69 | 23.55 | 69 | 23.55 | |
| Infiltrating
ulcerative type | 1,023 | 55.45 | 484 | 51.76 | | 199 | 67.92 | 199 | 67.92 | |
| Diffuse
infiltrative type | 168 | 9.11 | 140 | 14.97 | | 14 | 4.78 | 14 | 4.78 | 1.000 |
| Mixed type | 116 | 6.29 | 68 | 7.27 | | 6 | 2.05 | 6 | 2.05 | |
| Gastrectomy | | | | | | | | | | |
| No | 207 | 11.22 | 120 | 12.83 | 0.213 | 1 | 0.34 | 1 | 0.34 | 1.000 |
| Yes | 1,638 | 88.78 | 815 | 87.17 | | 292 | 99.66 | 292 | 99.66 | |
Survival analysis of subgroups according
to race and sex
As shown in Figs. 2
and 3, the survival prognosis of
female gastric cancer patients was better than that of male
patients among Whites. Fig. 2A and
B compare the OS and CSS of patients with different gastric
cancer types between 1973 and 2003. The survival time of male
patients was significantly lower than that of female patients (OS:
HR of death=1.06, 95% CI=1.039-1.081, P<0.001; CSS: HR of
death=1.084, 95% CI=1.059-1.111, P<0.001), which remained after
matching (OS: HR of death=1.072, 95% CI=1.041-1.103, P<0.001;
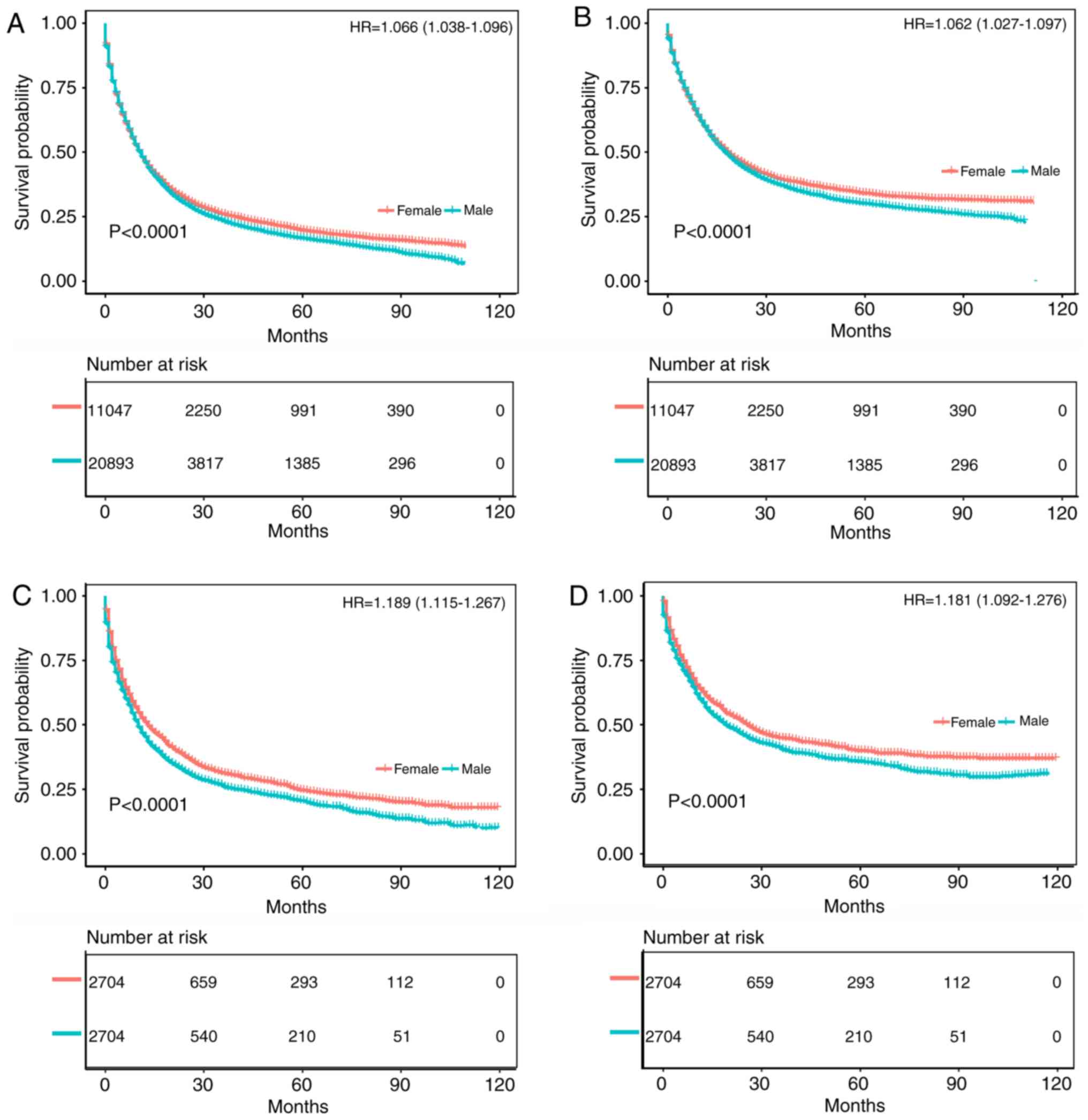
CSS: HR of death=1.084, 95% CI=1.048-1.122, P<0.001) (Fig. 2C and D). Fig. 3A and B compare the OS and CSS of
patients with different gastric cancer types between 2004 and 2013,
revealing that the survival time of male patients was significantly
lower than that of female patients (OS: HR of death=1.066, 95%
CI=1.038-1.096, P<0.001; CSS: HR of death=1.062, 95%
CI=1.027-1.097, P<0.001), which again remained after matching
(OS: HR of death=1.189, 95% CI=1.115-1.267, P<0.001; CSS: HR of
death=1.181, 95% CI=1.092-1.276, P<0.001) (Fig. 3C and D). Fig. S1A and B displays the trend in HRs
for OS and CSS among male and female patients (P<0.0001), with
significantly better results for the latter.
The survival prognosis for female gastric cancer
patients was not greatly different from that of male patients among
Black patients (Figs. 4 and
5). Fig. 4A and B compare the OS and CSS of
patients with different gastric cancer types between 1973 and 2003,
wherein the survival time of male patients was significantly lower
than that of female patients (OS: HR of death=1.097, 95%
CI=1.045-1.152, P<0.001; CSS: HR of death=1.085, 95%
CI=1.022-1.151, P=0.0058). After matching, the OS of male patients
was still significantly lower than that of female patients (HR of
death=1.182, 95% CI=1.087-1.284, P<0.001), but there was no
significant difference in CSS (HR of death=1.059, 95%
CI=0.957-1.172, P=0.260) (Fig. 4C and
D). Fig. 5A and B compare the
OS and CSS of patients with different gastric cancer types between
2004 and 2013. Although the OS of male patients was significantly
lower than that of female patients (HR of death=1.135, 95%
CI=1.068-1.205, P<0.001), there was no significant difference in
CSS (HR of death=1.066, 95% CI=0.992-1.147, P=0.077). Furthermore,
there was no significant difference in survival between the two
groups after matching (OS: HR of death=1.15, 95% CI=0.896-1.477,
P=0.25; CSS: HR of death=0.914, 95% CI=0.666-1.254, P=0.570)
(Fig. 5C and D). Fig. S1C and D illustrates the trend in
HRs for OS and CSS among male and female patients, with no
significant differences between the groups.
Among Asian patients, the survival prognosis of
female gastric cancer patients was worse than that of male patients
(Fig. 6). The comparison of the OS
and CSS of patients with different gastric cancer types between
1994 and 2017 is shown in Fig. 6A and
B. The survival time of female patients was significantly lower
than that of male patients (OS: HR of death=1.147, 95%
CI=1.032-1.274, P=0.011; CSS: HR of death=1.114, 95%
CI=1.007-1.233, P=0.036), and the survival time of female patients
was significantly lower than that of male patients after matching
(OS: HR of death=1.342, 95% CI=1.046-1.722, P=0.021; CSS: HR of
death=1.329, 95% CI=1.042-1.695, P=0.023) (Fig. 6C and D). The trend in HRs for OS
and CSS among male and female patients is illustrated in Fig. S1E and F (P<0.0001), with
significantly better results for men than women.
All 12 clinicopathological factors, including T
stage, N stage, M stage, tumor grade, marital status, primary tumor
location, pathological type, ethnicity, age group, SEER historic
stage, ascites status and Borrmann classification, were significant
in the univariate analysis (Figs. S2
S6). Figs. S2 and S3 indicate that for White patients,
female sex was an independent protective factor for survival.
However, as shown in Figs. S4 and
S5, the prognosis of women
between 1973 and 2003 was better than that of men among Black
patients, whereas sex-related survival was not significant between
2004 and 2013. In Asians, male sex was an independent protective
factor for survival in gastric cancer (Fig. S6).
Functional enrichment and pathway
analysis of DEGs
Regarding White patients, the mRNA expression data
of 161 male and 95 female gastric cancer tissue samples were
summarized after pretreatment and the R package limma was used for
differential expression analysis according to the top 50 log2 FC
values. Genes were mapped using heat maps, in which 50 mRNAs were
upregulated and 50 mRNAs downregulated. The specific genes are
shown in Fig. S7. Among them,
gray indicates male, and yellow indicates female. To further
investigate the role of differential genes in White patients with
gastric cancer, DAVID was used for GO enrichment analysis.
According to the results, in addition to enrichment of the sex
differentiation pathway, the DEGs were mainly enriched in ‘RNA
binding', ‘transcription', ‘oxidoreductase activity incorporation
of two atoms of oxygen', and ‘rRNA binding' (Fig. S8). The mRNA expression data for
Black patients were also summarized, comprising 8 male and 3 female
gastric cancer tissue samples. The specific genes are shown in
Fig. S9. As above, gray indicates
male, and yellow indicates female. Based on the GO enrichment
analysis, DEGs were mainly enriched in ‘cornified envelope',
‘endopeptidase inhibitor activity', ‘structural molecule activity',
‘extracellular region' and ‘extracellular space', in addition to
enrichment of the sex differentiation pathway (Fig. S10). Similarly, the mRNA expression
data for Asian patients involved 48 male and 26 female gastric
cancer tissue samples. The specific genes are shown in Fig. S11, where gray indicates male and
yellow female. In addition to enrichment of the sex differentiation
pathway, DEGs were mainly enriched in ‘RNA binding', ‘extracellular
region', ‘acute phase response', ‘cell surface receptor linked
signal transduction' and ‘G protein coupled receptor protein
signaling pathway', according to the GO enrichment analysis
(Fig. S12).
Relationship between CpG methylation,
gene expression and prognosis
Methylation of seven CpG loci and expression of
quantitative trait loci were assessed in the gene set. Because
promoter hypermethylation plays an important role in the
inactivation of cancer associated genes, there was particular
interest in genes with low methylation and high expression levels.
In subsequent analyses, these genes may be used to screen candidate
diagnostic biomarkers (18). Among
White patients, the methylation level of the CpG locus in the
promoter and first exon regions was inversely correlated with the
expression of corresponding genes, such as glycogenin 2 pseudogene
1 (GYG2P1; rs=0.216, P<0.05), ribosomal protein S4 Y-linked 1
(RPS4Y1; rs= 0.515, P<0.05), taxilin-γ (TXLNG; rs=0.129,
P<0.05) and eukaryotic translation initiation factor 1A X-linked
(EIF1AX; rs=0.231, P<0.05). In the KMPlot database, OS data were
available for 876 patients with gastric cancer, and GYG2P1 (HR of
death=1.600, 95% CI=1.340-1.920, P<0.001), RPS4Y1 (HR of
death=1.370, 95% CI=1.110-1.690, P=0.003), TXLNG (HR of
death=0.790, 95% CI=0.630-0.980, P=0.031) and EIF1AX (HR of
death=0.470, 95% CI=0.390-0.560, P<0.001) were differentially
expressed. GYG2P1, RPS4Y1, TXLNG and EIF1AX were indicated to be
upregulated by DNA hypomethylation. GYG2P1 and RPS4Y1 were highly
expressed in male patients and were significantly associated with a
poor prognosis. By contrast, high expression of TXLNG and EIF1AX in
female patients was associated with a favor able prognosis
(Fig. 7).
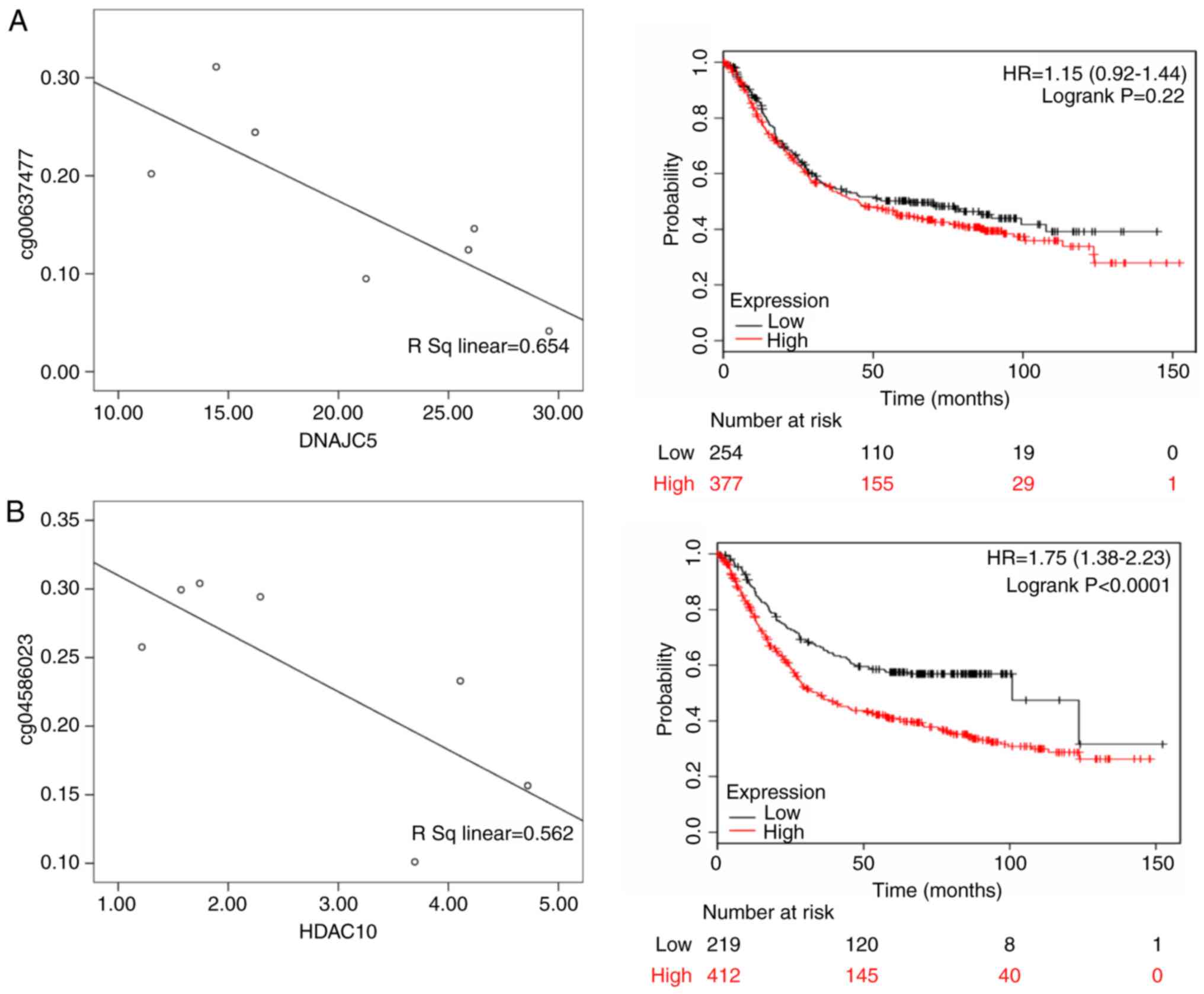
For Black patients, DnaJ heat shock protein family
(Hsp40) member C5 (DNAJC5; rs=0.654, P<0.05), histone
deacetylase 10 (HDAC10; rs=0.562, P<0.05), neogenin 1 (NEO1; rs=
0.720, P<0.05) and SMG5 nonsense mediated mRNA decay factor
(SMG5; rs=0.726; P<0.05) were differentially expressed.
According to the KMPlot database, 876 patients with gastric cancer
had OS data, including those with differential expression of DNAJC5
(HR of death=1.15, 95% CI= 0.920-1.440, P= 0.220), HDAC10 (HR of
death=1.750, 95% CI=1.380-2.230, P<0.001), NEO1 (HR of
death=1.320, 95% CI=1.120-1.570, P=0.001) and SMG5 (HR of
death=1.580, 95% CI=1.320-1.890, P<0.001). Among these genes,
DNAJC5, HDAC10, NEO1 and SMG5 were highly expressed in male
patients and were significantly associated with a poor prognosis
(Fig. 8).
Thymosin β4 Y-linked (TMSB4Y; rs=0.341, P<0.05),
ubiquitously transcribed tetratricopeptide repeat containing,
Y-linked (UTY; rs= 0.461, P<0.05), zinc finger protein Y-linked
(ZFY; rs=0.208, P<0.05) and zinc finger protein 787 (ZNF787;
rs=0.338, P<0.05) were differentially expressed in Asian
patients, and there were 876 gastric cancer patients with OS data
from KMPlot database. ZFY (HR of death=0.840, 95% CI=0.670-1.050,
P=0.12), TMSB4Y (HR of death=0.790, 95% CI=0.660-0.930, P=0.006),
UTY (HR of death=1.130, 95% CI= 0.950-1.350, P= 0.170) and ZNF787
(HR of death=1.830, 95% CI=1.540-2.170, P<0.001) were
differentially expressed. ZFY, TMSB4Y and UTY were highly expressed
in male patients, and TMSB4Y was significantly associated with a
better prognosis; ZNF787 was highly expressed in female patients
and associated with a poor prognosis (Fig. 9).
Discussion
The present study analyzed 93,278 gastric cancer
patients in the SEER and Sun Yat-Sen University Center for Gastric
Cancer Research databases, with 2,857 Asian, 13,223 Black and
77,198 White patients. In the Asian population, the male to female
ratio was 1.96; that of the Black population was 1.53, and that of
the White population was 1.81. This ratio is similar to that in a
previous study, indicating that the incidence of gastric cancer in
men is almost twice that in women (3). Based on the general tumor
characteristics of gastric cancer patients of the different ethnic
groups, it was found that for White patients, the tumor grade of
male patients was worse than that of female patients; this
difference was not significant for Black patients. Conversely, the
tumor grade of Asian female patients was worse than that of Asian
male patients. This may be an important factor leading to the
effect of sex on the prognosis of gastric cancer. Despite the few
studies on the tumor character istics of gastric cancer in
different sexes, Aron et al (19) studied patients with renal cell
carcinoma in the SEER database, and found that tumor size and tumor
stage were lower among females compared to males, which is similar
to the results for gastric cancer in the present study.
Furthermore, there are few studies on the prognosis of gastric
cancer in the different sexes. However, Kim et al (20) analyzed 4,722 patients undergoing
radical gastrectomy, including 3,136 males and 1,586 females, and
found that younger female patients had a higher incidence of signet
ring cell carcinoma than did men, and that the prognosis of gastric
cancer among women was worse than that among men. Li et al
(21) also investigated 96,501
gastric cancer patients, including 61,639 males and 34,862 females,
reporting that the former presented with larger tumors, higher
stages and higher grade gastric cancer; in addition, OS and CSS
were better in women, with a significant difference. These studies
suggested that sex has a significant effect on gastric cancer.
To date, the impact of ethnicity on gastric cancer
has been assessed in a considerable number of reports. Dong et
al (3) conducted a
retrospective cohort study of the comprehensive healthcare system
in southern California between 2008 and 2014 by assessing the
incidence of gastric cancer in different races, and found that race
is indeed a risk factor. Non-Hispanic Black patients had an odds
ratio (OR) of 1.500, White Hispanic patients had an OR of 1.400 and
Asian patients had an OR of 1.500 (3). The above data showed that the
incidence of gastric cancer in Asian populations is high. In fact,
75% of gastric cancer patients in the world are of Asian descent
(5). In addition to these racial
differences, the impact of sex on gastric cancer is also worthy of
further study. Globally, the incidence of gastric cancer in women
is almost half that in men. However, a study from South Korea found
that although the incidence in women is lower than that in men, the
prognosis of women is generally poor and the average age of onset
is low (55.0 years for females and 57.9 years for males) (20). A study in the United States between
2010 and 2014 reported that the incidence of gastric cancer in male
Asian patients was 14/100,000, and that in female patients was
8/100,000, with mortality rates of 7.1/100,000 and 4.3/100,000 for
males and females, respectively. The incidence of gastric cancer in
males of the African American population was 13.6/100,000 and that
in females was 7.8/100,000, with mortality rates of 8.6/100,000 and
4.1/100,000 for males and females, respectively. Lastly, the
incidence of gastric cancer in males of the White American
population was 8.2/100,000 and that in females was 3.7/100,000; the
mortality rate was 3.4/100,000 for males and 1.7/100,000 for
females (22). In fact, the
effects of sex on gastric cancer in patients of different ethnic
backgrounds vary considerably. The present study showed that the
prognosis of White female patients with gastric cancer in the
United States was better than that of White male patients. By
contrast, the difference between men and women may not be
significant among Black gastric cancer patients, but for Chinese
patients, the prognosis of females was worse than that of males.
These conclusions are consistent with the conclusions of the
previous studies mentioned above.
Different populations carry different gene
mutations, and gene expression also varies, which may explain why
different populations have differences in cancer risk. For example,
Loh et al (23) studied
polymorphisms in gastric cancer genes in different ethnic groups by
evidence-based medicine. A total of 203 related studies were
included, and 225 polymorphisms in 95 genes were evaluated; the
results showed that race can have a marked impact on the risk of
gastric cancer. The present study also evaluated differential gene
expression between ethnicities and found significant differences
among White, Black and Asian patients. Among them, the DEGs between
White male and female patients were mainly enriched in RNA binding
and transcription pathways. A total of four core genes were
identified, GYG2P1, RPS4Y1, TXLNG and EIF1AX, according to the
relationship between gene expression and DNA methylation (Table IV). GYG2P1 (24), RPS4Y1 (25), TXLNG (26) and EIF1AX (26) are mainly involved in tumor RNA
binding and transcriptional path ways, as well as the survival
prognosis of patients with different sexes of gastric cancer. In
general, the impact of these core genes on tumors may be an
important factor leading to poor prognosis in males. The DEGs
between Black male and female patients were mainly enriched in cell
structural changes, and DNAJC5, HDAC10, NEO1 and SMG5 were
identified by the same means described above (Table IV). However, there is little
research on the relationship between DNAJC5 and SMG5 in tumors, and
tumor-related effects are not yet clear (27). Although studies have shown that
high expression of HDAC10 can improve prognosis, high expression of
this gene was reported to be associated with a poor prognosis
(28). Increased expression of
NEO1 increases the sensitivity of gastric cancer cells to
carboplatin (29). Overall, these
genes may play a role by altering the structural function of cells,
though the influence of these genes on the survival prognosis of
gastric cancer in different sexes remains unclear. For Asian
patients, the DEGs between the sexes were mainly enriched in cell
surface receptor-associated signal transduction and the G protein
coupled receptor protein signaling, and four core genes were
screened, TMSB4Y, UTY, ZFY and ZNF787, based on the relationship
between gene expression and DNA methylation (Table IV). TMSB4Y is significantly
related to the inhibition of tumor migration, suggesting that it
may be a tumor suppressor gene (30). In this study, it was also found
that elevated expression of TMSB4Y can improve the prognosis of
patients. UTY and ZFY are mainly involved in apoptosis in stem
cells in males (31), yet their
impact on cancer is still unclear. ZNF can promote the migration
and invasion of gastric cancer (32). The present study found that
elevated expression of ZNF787 affects the prognosis of patients.
The impact of these core genes on tumors may be an important factor
leading to poor prognosis in females.
 | Table IVKnown function/phenotype of
Protein-protein interaction analysis of differential genes between
of gender in different races of patients with gastric cancer. |
Table IV
Known function/phenotype of
Protein-protein interaction analysis of differential genes between
of gender in different races of patients with gastric cancer.
| Author, year | Protein | Protein
function | Expression | (Refs.) |
|---|
| Loh et al,
2009 | GYG2P1 | Glycogenin 2
pseudogene 1. The expression of pseudogenes is significantly
related to the development and prognosis of tumors. | White male | (23) |
| Lou et al,
2019 | RPS4Y1 | RPS4Y1 is a full
name of ribosomal protein S4 Y-linked 1, which is a member. of the
ribosomal protein S4E family. The role of RPS4Y1 is similar to that
of RPS4X; its high level of expression is associated with poor
prognosis of intrahepatic cholangiocarcinoma and non mucinous
carcinoma | White male | (24) |
| Jung et al,
2011 | TXLNG | Located in Xp22.2,
the full name of taxilin-γ, which encodes a member of the
paclitaxel family. The encoded protein binds to the c terminal coil
region of the diamine family members 1a, 3a and 4a and may play a
role in intracellular vesicle transport, which is mediated by
lipopolysaccharide and may be involved in Cell cycle
regulation. | White female | (25) |
| Yu et al,
2005 | EIF1AX | EIF1AX is located
at Xp22.12 and is called the eukaryotic translation initiation.
factor 1A X-linked. This gene encodes an important eukaryotic
translation initiation factor. The expression of EIF1AX is an
important factor in translation which affect tumor progression and
prognosis | White female | (26) |
| Benitez et
al, 2017 | DNAJC5 | Located at
20q13.33, the full name of DnaJ heat shock protein family (Hsp40)
member C5, which is a member of the J protein family. The
relationship between this gene and tumors, and the tumor-related
effects are not yet clear. | Black male | (27) |
| Tao et al,
2017 | HDAC10 | Located at
22q13.33, the full name of histone deacetylase 10, which encodes a
protein belonging to the family of histone deacetylases, members of
which deacetylate lysine residues at the n terminal portion of the
core histone. | Black male | (28) |
| Qu et al,
2018 | NEO1 | Located at 15q24.1,
the full name of neogenin 1, which encodes a cell surface protein
that is a member of the immunoglobulin superfamily. Increased
expression of NEO1 increases the sensitivity of gastric cancer
cells to carboplatin. | Black male | (29) |
| Benitez et
al, 2017 | SMG5 | Located in 1q22,
the full name is nonsense mediated mRNA decay factor, and SMG5 is
involved in non-sensory-mediated mRNA decay. | Black male | (27) |
| Wong et al,
2015 | TMSB4Y | Located in
Yq11.221, the full name of thymosin beta 4 Y-linked, which is
located in a specific region of the Y chromosome and encodes an
actin-separating protein. The expression of TMSB4Y is significantly
related to the inhibition of tumor migration. | Asian male | (30) |
| Tricoli et
al, 1993 | UTY | Located in
Yq11.221, the full name of ubiquitously transcribed
tetratricopeptide repeat containing, Y-linked, which encodes a
protein containing a repeat of tetra quartin, thought to be
involved in protein protein-interactions. | Asian male | (31) |
| Tricoli et
al, 1993 | ZFY | Located in Yp11.2,
the full name zinc finger protein Y-linked, which encodes a
zinc-containing finger protein that acts as a transcription factor
and is mainly involved in apoptosis of male stem cells. | Asian male | (31) |
| Li et al,
2014 | ZNF787 | Located at
19q13.43, the full name of zinc finger protein 787. Some
abnormalities in ZNF protein can increase the expression of MMP 2,
MMP-9 and ICAM-1, reduce the expression of timp-1, and promote the
migration and invasion of gastric cancer. | Asian female | (32) |
Although the present study provided a comprehensive
analysis of the prognosis of patients with different ethnic back
grounds based on the world's largest database and data from China's
largest center for gastric cancer research, there are several
limitations. First, because the staging standards were different at
different periods, SEER data were divided into 1973-2003 and
2004-2013 periods to include more patients and bolster the results.
Due to the limitations of follow up in the databases, some data
were not available, such as the situation of patients with respect
to chemotherapy, which may have an impact on the conclusion.
Second, as the classification of patients in the SEER database does
not distinguish between Asians and Indians and South Americans,
analysis of data for the Asian population from the SEER database is
not possible. Third, this study was a retrospective study, and thus
there may be some bias in the statistical results. Fourth, a period
of 40 years was examined because of differences in treatment and
diet, and potentially variable environmental factors. These factors
could affect the accuracy of the results. Given that the present
study was retrospective, the data collection was not strictly
randomized; therefore, the presence of confounding factors is
unavoidable. Furthermore, whether sex differences are the cause of
differences in the prognosis of patients with gastric cancer cannot
be directly determined.
In the present study, it was found that the effects
of sex on the prognosis of gastric cancer patients of different
ethnicities differed. It was concluded that differences in
molecular mechanisms between the two sexes among gastric cancer
patients of different ethnic groups are the main reasons for the
observed variability.
Supplementary Data
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
CZ and HL conceived and designed the study. HL, CW,
ZW, ZG, WC and YH performed the data analysis. HL, CW and ZW wrote
the paper. All authors read and approved the manuscript.
Ethics approval and consent to
participate
The information of for White and Black patients with
gastric cancer obtained from the SEER database does not contain any
identifiers and is public. Due to the retrospective nature of the
study, informed consent was not required. All SEER database
information was deidentified by the United States National Cancer
Institute review board approval. This analysis did not involve
interaction with human subjects or the use of person ally
identifiable information. The research on the Asian cohort was
approved by the ethics committee of the First Affiliated Hospital
of Sun Yat-Sen University, and written informed consent was
obtained from all of these patients.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgments
Not applicable.
References
|
1
|
Allemani C, Matsuda T, Di Carlo V,
Harewood R, Matz M, Nikšić M, Bonaventure A, Valkov M, Johnson CJ,
Estève J, et al: Global surveillance of trends in cancer survival
2000-14 (CONCORD -3): Analysis of individual records for 37 513 025
patients diagnosed with one of 18 cancers from 322 population based
registries in 71 countries. Lancet. 391:1023–1075. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Dong E, Duan L and Wu BU: Racial and
Ethnic minorities at increased risk for gastric cancer in a
regional US population study. Clin Gastroenterol Hepatol.
15:511–517. 2017. View Article : Google Scholar
|
|
4
|
Ferlay J, Soerjomataram I, Dikshit R, Eser
S, Mathers C, Rebelo M, Parkin DM, Forman D and Bray F: Cancer
incidence and mortality worldwide: Sources methods and major
patterns in GLOBOCAN 2012. Int J Cancer. 136:E359–E386. 2015.
View Article : Google Scholar
|
|
5
|
Karimi P, Islami F, Anandasabapathy S,
Freedman ND and Kamangar F: Gastric cancer: Descriptive
epidemiology, risk factors, screening, and prevention. Cancer
Epidemiol Biomarkers Prev. 23:700–713. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hwang KT, Kim J, Jung J, Chang JH, Chai
YJ, Oh SW, Oh S, Kim YA, Park SB and Hwang KR: Impact of breast
cancer subtypes on prognosis of women with operable invasive breast
cancer: A population-based study using SEER database. Clin Cancer
Res. 25:1970–1979. 2019.
|
|
7
|
Greene FL, Compton CC, Fritz AG, Shah JP
and Winchester DP: AJCC Cancer Staging Atlas. Springer; New York,
NY: 2006, View Article : Google Scholar
|
|
8
|
Li B and Dewey CN: RSEM: Accurate
transcript quantification from RNA-Seq data with or without a
reference genome. BMC Bioinformatics. 12:3232011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Aryee MJ, Jaffe AE, Corrada-Bravo H,
Ladd-Acosta C, Feinberg AP, Hansen KD and Irizarry RA: Minfi: A
flexible and comprehensive bioconductor package for the analysis of
infinium DNA methylation microarrays. Bioinformatics. 30:1363–1369.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sandoval J, Mendez-Gonzalez J, Nadal E,
Chen G, Carmona FJ, Sayols S, Moran S, Heyn H, Vizoso M, Gomez A,
et al: A prog nostic DNA methylation signature for stage I
non-small-cell lung cancer. J Clin Oncol. 31:4140–4147. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Teschendorff AE, Marabita F, Lechner M,
Bartlett T, Tegner J, Gomez-Cabrero D and Beck S: A beta-mixture
quantile normalization method for correcting probe design bias in
Illumina Infinium 450 k DNA methylation data. Bioinformatics.
29:189–196. 2013. View Article : Google Scholar :
|
|
12
|
Langevin SM, Butler RA, Eliot M, Pawlita
M, Maccani JZ, McClean MD and Kelsey KT: Novel DNA methylation
targets in oral rinse samples predict survival of patients with
oral squamous cell carcinoma. Oral Oncol. 50:1072–1080. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Böhnke JR: Explanation in causal
inference: Methods for mediation and interaction. Q J Exp Psychol
(Hove). 69:1243–1244. 2016. View Article : Google Scholar
|
|
14
|
Gene Ontology Consortium: The gene
ontology (GO) project in 2006. Nucleic Acids Res. 34:D322–D326.
2006. View Article : Google Scholar :
|
|
15
|
Ashburner M, Ball CA, Blake JA, Botstein
D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT,
et al: Gene ontology: Tool for the unification of biology. The gene
ontology consortium. Nat Genet. 25:25–29. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Györffy B, Lanczky A, Eklund AC, Denkert
C, Budczies J, Li Q and Szallasi Z: An online survival analysis
tool to rapidly assess the effect of 22,277 genes on breast cancer
prognosis using microarray data of 1,809 patients. Breast Cancer
Res Treat. 123:725–731. 2010. View Article : Google Scholar
|
|
17
|
Austin PC: Optimal caliper widths for
propensity-score matching when estimating differences in means and
differences in proportions in observational studies. Pharm Stat.
10:150–161. 2011. View
Article : Google Scholar :
|
|
18
|
Herman JG and Baylin SB: Gene silencing in
cancer in association with promoter hypermethylation. N Engl J Med.
349:2042–2054. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Aron M, Nguyen MM, Stein RJ and Gill IS:
Impact of gender in renal cell carcinoma: An analysis of the SEER
database. Eur Urol. 54:133–140. 2008. View Article : Google Scholar
|
|
20
|
Kim HW, Kim JH, Lim BJ, Kim H, Kim H, Park
JJ, Youn YH, Park H, Noh SH, Kim JW and Choi SH: Sex disparity in
gastric cancer: Female sex is a poor prognostic factor for advanced
gastric cancer. Ann Surg Oncol. 23:4344–4351. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li H, Wei Z, Wang C, Chen W, He Y and
Zhang C: Gender differences in gastric cancer survival: 99,922
cases based on the SEER database. J Gastrointest Surg. Jul
25–2019.Epub ahead of print. View Article : Google Scholar
|
|
22
|
Ashktorab H, Kupfer SS, Brim H and
Carethers JM: Racial disparity in gastrointestinal cancer risk.
Gastroenterology. 153:910–923. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Loh M, Koh KX, Yeo BH, Song CM, Chia KS,
Zhu F, Yeoh KG, Hill J, Iacopetta B and Soong R: Meta-analysis of
genetic polymorphisms and gastric cancer risk: Variability in
associations according to race. Eur J Cancer. 45:2562–2568. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lou W, Ding B and Fan W: High expression
of pseudogene PTTG3P indicates a poor prognosis in human breast
cancer. Mol Ther Oncolytics. 14:15–26. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Jung Y, Lee S, Choi HS, Kim SN, Lee E,
Shin Y, Seo J, Kim B, Jung Y, Kim WK, et al: Clinical validation of
colorectal cancer biomarkers identified from bioinformatics
analysis of public expression data. Clin Cancer Res. 17:700–709.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yu VW, Ambartsoumian G, Verlinden L, Moir
JM, Prud'homme J, Gauthier C, Roughley PJ and St-Arnaud R: FIAT
represses ATF4-mediated transcription to regulate bone mass in
transgenic mice. J Cell Biol. 169:591–601. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Benitez BA and Sands MS: Primary
fibroblasts from CSPα mutation carriers recapitulate hallmarks of
the adult onset neuronal ceroid lipofuscinosis. Sci Rep.
7:63322017. View Article : Google Scholar
|
|
28
|
Tao X, Yan Y, Lu L and Chen B: HDAC10
expression is associated with DNA mismatch repair gene and is a
predictor of good prognosis in colon carcinoma. Oncol Lett.
14:4923–4929. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Qu H, Sun H and Wang X: Neogenin-1
promotes cell proliferation, motility, and adhesion by
up-regulation of zinc finger E-box binding homeobox 1 via
activating the Rac1/PI3K/AKT pathway in gastric cancer cells. Cell
Physiol Biochem. 48:1457–1467. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wong HY, Wang GM, Croessmann S, Zabransky
DJ, Chu D, Garay JP, Cidado J, Cochran RL, Beaver JA, Aggarwal A,
et al: TMSB4Y is a candidate tumor suppressor on the Y chromosome
and is deleted in male breast cancer. Oncotarget. 6:44927–44940.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Tricoli JV and Bracken RB: ZFY gene
expression and retention in human prostate adenocarcinoma. Genes
Chromosomes Cancer. 6:65–72. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Li Y, Tan BB, Zhao Q, Fan LQ, Wang D and
Liu Y: ZNF139 promotes tumor metastasis by increasing migration and
invasion in human gastric cancer cells. Neoplasma. 61:291–298.
2014. View Article : Google Scholar : PubMed/NCBI
|