Introduction
As a malignancy of the digestive system, gastric
cancer (GC) originates in the gastric mucosal epithelium (1). Based on global cancer statistics in
2020, more than one million patients were newly diagnosed with GC
and ~769,000 patients died from GC; GC ranks fifth in incidence and
fourth in cancer-associated mortality of all types of cancer
(2). The occurrence and
development of GC are influenced by multiple factors, such as diet,
family history, consumption of alcohol and tobacco and infection of
Helicobacter pylori and Epstein-Barr virus (3). Endoscopic ultrasound is the most
efficient diagnostic methods for early-stage GC, however, a
majority of patients are diagnosed at advanced stages (4). Surgical resection is mainly applied
to treat early-stage patients, while chemotherapy proves most
efficient in advanced-staged patients (5). In spite of improvement in GC
diagnosis and treatment, the prognosis of GC still remains poor and
the 5-year survival rate is <30% (6). Hence, it is of great importance to
identify novel molecular mechanisms in GC progression.
Circular RNA (circRNA) is a type of RNA with
covalently closed structure, generated by back-splicing of
precursor mRNA (7,8). Former studies have revealed the
effect of circRNAs in GC progression: For examples, circRNA_100290
modulated by eukaryotic translation initiation factor 4A3 (EIF4A3)
can interact with microRNA (miRNA or miR)-29b-3p to target integrin
subunit alpha 11 (ITGA11), thus facilitating proliferation and
invasion of GC cells (9); circRNA
nuclear receptor interacting protein 1 targets the Akt1/mTOR
pathway by sequestering miR-149-5p to promote GC progression
(10); circRNA ELAV like RNA
binding protein 1 (circ-HuR) downregulates HuR expression by
binding to CCHC-type zinc finger nucleic acid binding protein
(CNBP), thereby suppressing metastasis, proliferation and invasion
of GC cells (11); the
circ_0004872/miR-224/Smad4/ADAR1 axis forms a negative feedback
loop to hinder GC progression (12). A recent study showed that
Helicobacter pylori infection-induced circRNA mannosidase
alpha class 1A member 2 (circMAN1A2) promotes GC progression via
sponging miR-1236-3p (13). The
aforementioned findings indicate that circRNAs are crucial
regulators in GC development, highlighting the research value of
circRNAs in GC. The present study investigated the role of a
new-found GC-associated circRPS19 selected from Gene Expression
Omnibus (GEO) database in GC cell proliferation.
Aerobic glycolysis, or the Warburg effect, was
discovered by Otto Warburg in the 1920s. Tumor cells, despite
sufficient oxygen, prefer the conversion of glucose into lactate to
generate energy (14). As reported
by previous studies, aerobic glycolysis serves a key role in a
variety of carcinomas, including GC: For example, long non-coding
RNA H19 (imprinted maternally expressed transcript interacts with
miR-519d-3p to regulate lactate dehydrogenase A (LDHA), thus
facilitating aerobic glycolysis and proliferative ability of GC
cells (15); Opa interacting
protein 5 antisense RNA 1 (OIP5-AS1) sequesters miR-186 to enhance
proliferation and aerobic glycolysis in GC cells (16); pyruvate kinase M2 (PKM2) serves an
oncogenic role in GC cells by facilitating aerobic glycolysis
(17); Hexokinase 2 (HK2), as a
rate-limiting enzyme, catalyzes glucose metabolism and cancer cells
commonly exhibit increased glucose metabolism (18). Previous research has demonstrated
that circRNA functions as a regulator of HK2 in GC cells to
facilitate aerobic glycolysis (19). Nevertheless, the association
between HK2 and circRPS19 in GC cells needs to be further
investigated.
The present current study aimed to investigate the
functions of circRPS19 in GC cell proliferation and aerobic
glycolysis and its downstream molecular mechanism.
Materials and methods
Cell culture and treatment
Human GC cell lines (AGS, HGC-27, NCI-N87 and SNU-1)
were obtained from China Center for Type Culture Collection and
normal human gastric epithelial cell line (GES-1) was obtained from
American Type Culture Collection (ATCC). All cell lines were
cultured in RPMI-1640 medium containing 10% fetal bovine serum
(FBS; Gibco; Thermo Fisher Scientific, Inc.). Cell lines were
incubated in a humid atmosphere with 5% CO2 at 37°C.
For RNA stability analysis, GC cells were treated
with 3 U/µg RNase R (9001-99-4, Sigma-Aldrich; Merck KGaA)
for 24 h at room temperature or treated with 2 µg/ml ActD
(50-76-0, Merck) for different time intervals (0, 6, 12, 18, 24 h)
at room temperature. RNA stability was determined by using RT-qPCR
as following description.
To analyze protein stability, GC cells were treated
with 5 µM cycloheximide (CHX, HY-12320, MedChemExpress) for
0, 3, 6, 9 h or treated with 20 µM MG132 (HY-13259,
MedChemExpress) for 4 h. Cells were treated with 10 µM
HBX19818 (HY-17540, MedChemExpress) to inhibit USP7 expression.
Protein levels were detected using western blot as following
description.
Data collection and bioinformatics
analysis
circRNAs significantly overexpressed in GC tissues
(under the condition of P≤0.05) were screened out from GSE83521
dataset in Gene Expression Omnibus (GEO; ncbi.nlm.nih.gov/gds). starBase database (rnasysu.com/encori/) was used to screen miRNAs that
potentially interact with both circRPS19 and USP7.
Cell transfection
Small interfering RNAs (siRNAs) targeting circRPS19
or ubiquitin-specific processing protease 7 (USP7) sequence were
synthesized for knockdown with non-targeted siRNA as the negative
control (NC). For overexpression of USP7, the full-length sequence
of USP7 was inserted into pcDNA3.1 vector to establish USP7
overexpression vector, with empty vector as NC. miR-125a-5p mimics
was synthesized and used to upregulate miR-125a-5p expression, with
mimics-NC as NC. All plasmids and vectors were synthesized and
constructed by Guangzhou RiboBio Co., Ltd. Cell transfection was
performed at 37°C using Lipofectamine 2000 (Invitrogen; Thermo
Fisher Scientific, Inc.). After being transfected with siRNAs and
pcDNA3.1 vectors for 48 h or transfected with for miRNA mimics or
inhibitor for 24 h, cells were harvested for subsequent
experiments. The duration between end of transfection of siRNAs and
pcDNA3.1 vectors and subsequent experiments was 48 h. The duration
between end of transfection of miRNA mimics or inhibitor and
subsequent experiments is 24 h. siRNA and pcDNA3.1 expression
vector (0.8/50 µl) were treated with Lipofectamine 2000 for
transfection at 24 well plates. miRNA mimics were transfected at a
final concentration of 100 nM. The sequences were as follows:
si-NC, 5′-CCG GCG GCC GCA AAT TAC GAA TGA CTC GAG TCA TTC GTA ATT
TGC GGC CGT TTT T G-3′; si-circRPS19-1: 5′-CCG GCT CAT TCG TAA CGG
CCG CAA ACT CGA GGA GTA AGC ATT GCC GGC GTT TTT TTT G-3′;
si-circRPS19-2: 5′-CCG GAT TCG TAA CGG CCG CAA ACT GCT CGA GTA AGC
ATT GCC GGC GTT TGA CTT TTT G-3′; si-circRPS19-3: 5′-CCG GGC CTC
ATT CGT AAC GGC CGC ACT CGA GCG GAG TAA GCA TTG CCG GCG TTT TTT
G-3′; mimics-NC: 5′-UCU CCG AGA UCU UAC CAC CUG GUA-3′; miR-125a-5p
mimics: 5′-UCC CUG AGA CCC UUU AAC CUG UGA-3′; inhibitor NC: 5′-UCA
UCU CAG GAA GGG UAU CGA GGA-3′; miR-125a-5p inhibitor: 5′-UCA CAG
GUU AAA GGG UCU CAG GGA-3′; si-NC: 5′-CCG GGT TAA TTT ATT ATC CAG
CAT TCT CGA GAA TGC TGG ATA ATA A AT TAA CTT TTT G-3′; si-USP7-1,
5′-CAC CGT GTA ATT TAT TAT CTT CAG CAC TCG AGT GCT GAA GAT AAT AAA
T TA CA-3′; si-USP7-2, 5′-CAC CAT TCA TAG AAC ACT CTT TGT ACT CGA
GTA CAA AGA GTG TTC TAT GAA-3′; si-USP7-3, 5′-CAC CAG TTG TAG ATG
TAA CAC TGG TCT CGA GAC CAG TGT TAC ATC TAC AAC-3′.
Reverse transcription-quantitative (RT-q)
PCR
Total RNA was extracted from AGS, HGC-27, NCI-N87,
SNU-1 or GES-1 cells using TRIzol (Invitrogen; Thermo Fisher
Scientific, Inc.). RT was performed using the PrimeScript 1st
Strand cDNA Synthesis kit (Takara Bio, Inc.) according to the
manufacturer's protocol. RT-qPCR was performed on ABI 7500 fast PCR
System with SYBR green PCR Master Mix (both Applied Biosystems;
Thermo Fisher Scientific, Inc.). The qRT-PCR reaction was performed
95°C for 5 min, followed by 40 cycles of 95°C for 10 sec and a
primer-specific annealing temperature of 60°C for 30 sec. GAPDH and
U6 were used as internal controls. Relative RNA expression was
calculated using 2−ΔΔCq method, as previously described
(20). Additionally, gel
electrophoresis was performed to identify the circular structure of
circRPS19. Briefly, circular structure of circRPS19 was validated
by PCR reactions using the HotStar HiFidelity Polymerase Kit
(#202602; Qiagen) by using pairs of both convergent and divergent
primers to amplify circRPS19. Convergent primers were all designed
within the same exon. Both cDNA and genomic DNA (gDNA) were applied
as the template for PCR analysis. The sequences for primers were as
follows (5′~3′): divergent primer for circRPS19 (forward): CAG CTG
CCA ACA AGA AGC AT; divergent primer for circRPS19 (reverse): CTG
TCC GGC GAT TCT GTC C; convergent primer for circRPS19 (forward):
ACT GAC ACC TCA GGG ACA AAG; convergent primer for circRPS19
(reverse): ATG AGG CAA TTT ATT AAC CCA GCA; RPS19 (forward): GAA
CCA GCA GGA GTT CGT CA and reverse): AGA ACC AGT TCT CAT CGT AGG G;
miR-125a-5p forward): GCCGAG TCC CTG AGA CCC TTT A; miR-125a-5p
(reverse): CTC AAC TGG TGT CGT GGA; USP7 (forward): CCG AGG ACA TGG
AGA TGG AA and reverse, TTT CGC ACA AAA CAC GGA GG; GAPDH
(forward): GAC AGT CAG CCG CAT CTT CT; GAPDH (reverse): GCG CCC AAT
ACG ACC AAA TC; U6 (forward): TCC CTT CGG GGA CAT CCG; U6
(reverse): AAT TTT GGA CCA TTT CTC GAT TTG T.
Western blotting
Total protein was isolated from AGS cells with RIPA
lysis buffer (Sigma-Aldrich; Merck KGaA), Protein concentration was
measured by using a bicinchoninic acid (BCA) Kit (Pierce
Biotechnology, Rockford, IL, USA). Protein samples (30 ng/lane)
were then separated by 10% SDS-PAGE (Bio-Rad Laboratories, Inc.).
Afterwards, the samples were transferred onto PVDF membranes, which
were blocked with 5% defatted milk in 5% TBST at room temperature
for 2 h. Subsequently, membranes were incubated with primary
antibodies, including anti-HK2 (1/1,000, ab209847, Abcam, MA, USA),
anti-6-phosphofructokinase subunit alpha (anti-PFK1, 1/1,000,
ab154804, Abcam), anti-PKM2 (1/1,000, ab85555, Abcam), anti-USP7
(1/1,000, ab264422, Abcam) and the internal reference anti-β-actin
(1:1,000, ab8227, Abcam) at 4°C overnight. After being washed in
TBST buffer, the membranes were incubated with horseradish
peroxidase (HRP)-conjugated secondary antibodies (1/2,000,
ab154804, Abcam) at room temperature for 1 h. Pierce ECL Plus
Substrate (Pierce; Thermo Fisher Scientific, Inc.) was then used to
visualize blots. Image J software v1.8.0 (National Institutes of
Health, USA) was used to quantify protein blots.
Cell Counting Kit-8 (CCK-8) assay
Transfected AGS or HGC-27 cells were inoculated into
96-well culture plates at a density of 3×103 cells/well.
GC cells were incubated with 10 µl CCK-8 solution (Dojindo
Laboratories, Inc.) for 2 h. After incubation, the absorbance at
450 nm was detected at different time points (0, 24, 48 and 72 h)
by a spectrophotometer (Thermo Fisher Scientific, Inc.).
EdU assay
Transfected AGS or HGC-27 cells were plated in
12-well plates at a density of 1×105 cells/well. GC
cells were treated with EdU reagent (Guangzhou RiboBio Co., Ltd.).
Following incubation at 37°C for 2 h, cells were washed in PBS and
fixed in 4% paraformaldehyde (Sangon Biotech Co., Ltd.) at 4°C for
30 min. The nuclei of GC cells were stained by 400 µl DAPI
(Sigma-Aldrich; Merck KGaA) at room temperature for 30 min. GC cell
proliferation was observed under a Leica GmbH fluorescence
microscope (×200) and quantified by calculating the percentage of
EdU-positive cells in five randomly selected view fields using
Image J software v1.8.0 (National Institutes of Health, Bethesda,
MD, USA).
TUNEL assay
The apoptosis of transfected AGS or HGC-27 cells was
measured by TUNEL assay. GC cells were fixed in 4% paraformaldehyde
(Sangon Biotech Co., Ltd.) at 4°C for 30 min. The fixed cells were
then washed with 5 ml cold 70% ethanol, and the dehydrated cells
were incubated at -20°C for 4 h. Cells were permeabilized with 20
µg/ml Proteinase K in PBS for 5 min. Cells were then treated
with the ProLong Antifade Kit (P-7481, Molecular Probes) and
incubated with TUNEL reaction mixture (Roche Diagnostics)
containing 200 U/ml TdT and 4 µM dUTP at room temperature
for 1 h. After removing the reaction buffer, cells were washed with
PBS for 3 times. GC cell nuclei were stained by 400 µl DAPI
(Sigma-Aldrich; Merck KGaA) that was added to the media (1:10) at
room temperature for 30 min. The percentage of TUNEL-positive cells
in five randomly selected view fields was observed under a Leica
GmbH fluorescence microscope (×200) and calculated using Image J
software v1.8.0 (National Institutes of Health, Bethesda, MD,
USA).
Flow cytometry
Flow cytometry analysis was used to detect cell
apoptosis. Briefly, cells were collected and washed twice in cold
PBS. Cells were then fixed with 4% paraformaldehyde (Sangon Biotech
Co., Ltd.) at -20°C overnight. GC cells were suspended in 600
µl eBioscience™ flow cytometry binding buffer (Invitrogen;
Thermo Fisher Scientific, Inc.) at a concentration of
106 cells/ml and then stained with 5 µl Annexin
V/FITC and 5 µl propidium iodide (both BD Biosciences) in
dark for 15 min. Samples were analyzed using a FACSAria flow
cytometry (BD Biosciences, San Jose, CA, USA). Results were
analyzed using FlowJo 7.6 software (Tree Star, Inc.).
Measurement of glucose consumption and
lactate production
The glucose consumption and lactate production of
transfected AGS or HGC-27 cells were detected by using a Glucose
Assay kit (CBA086) and a Lactate Assay Kit (MAK064; both
Sigma-Aldrich) as previously described (22).
Examination of extracellular
acidification rate (ECAR) and oxygen consumption rate (OCR)
ECAR and OCR were examined as previously reported
(22). Seahorse XF96 (cat. no.
102601-100, Seahorse Bioscience) was applied for the analysis of
ECAR and OCR in AGS or HGC-27 cells. For ECAR analysis, GC cells
were exposed to 1 µM oligomycin for elimination of
mitochondria ATP production and 150 mM 2-Deoxy-D-glucose (2-DG) at
37°C for 1 h for inhibition of aerobic glycolysis (both
Sigma-Aldrich; Merck KGaA) successively, followed by assessment
using Glycolytic Stress Test kit (Seahorse Bioscience). For OCR
analysis, GC cells were exposed to 1.0 µM oligomycin, 0.5
µM carbonyl cyanide 4-(trifluoromethoxy) phenylhydrazone
(FCCP; a protonophore uncoupler) and 0.5 µM antimycin A at
37°C for 1 h for inhibition of electron transfer chain (all
Sigma-Aldrich; Merck KGaA) successively, then measured by XF Cell
Mito Stress kit (Seahorse Bioscience). ECAR and OCR were measured
by normalizing to total protein content.
Fluorescence in situ hybridization
(FISH)
In brief, AGS cells were washed in PBS, followed by
fixation in 4% paraformaldehyde (Sangon Biotech Co., Ltd.) at 4°C
for 30 min and permeabilization in 0.5% Triton X-100 (Beijing
Leigen Biotechnology Co., Ltd.). Cells were blocked with 50
µl goat serum (Sigma-Aldrich; Merck KGaA) for 1 h at room
temperature. Primer sets were designed based on the sequence of
circRPS19 and USP7 in the pGEM-T Easy vector (Promega).
Digoxigenin-labeled DNA probes targeting circRPS19 (genomic
position: chr19:42373768-42375484; accession no. NM_001022) and
USP7 mRNA (genomic position: chr16:8985951-9057341 accession no.
NM_003470.3) or control probes synthesized by Guangzhou RiboBio
Co., Ltd. DNA probes were denatured at 75°C for 5 min. Afterwards,
the probes were incubated with GC cells in 10 µl
hybridization buffer (30% formamide, 0.9 M NaCl, 20 mM Tris-HCl at
pH 8.0, 0.01% SDS, a 1-µM probe, and MQ water) at 55°C for 4
h. To remove excess probes, samples was washed in 50 ml tubes of
washing buffer without salt [20 mM Tris-HCl, 5 mM EDTA and 0.01%
(w/v) SDS] or with 1.8 M NaCl at 47°C for 3 min.
DAPI (Sigma-Aldrich; Merck KGaA) was applied to
counterstain nuclei of GC cells at room temperature for 30 min. A
confocal microscope (×200, Zeiss AG) was employed to obtain images.
The probe sequence for circRPS19: TTT GCG GCC GTT ACG AAT GA.
To identify the co-location of USP7 and HK2
proteins, we used the anti-HK2 (1/1,000, ab209847, Abcam, MA, USA),
and anti-USP7 (1/1,000, ab264422, Abcam) to incubated cell lysates
at 4°C overnight, followed by incubation with horseradish
peroxidase (HRP)-conjugated secondary antibodies (1/2,000,
ab154804, Abcam) at room temperature for 1 h. Results were analyzed
using Image J software v1.8.0 (National Institutes of Health).
Co-immunoprecipitation (Co-IP) assay and
mass spectrometry
Co-IP assay followed by mass spectrometry was used
to identify the interacting protein of HK2. Cells were washed twice
with pre-cold PBS. Cell lysates were obtained with RIPA lysis
buffer (Sigma-Aldrich; Merck KGaA). For each IP reaction, 50
µl lysates were used. After being centrifuged at 14,000 g
for 15 min at 4°C, supernatants were collected. The supernatants
were incubated with 50 µg of the negative control anti-Flag
(1/200, 66008-3-Ig; Proteintech) or 50 µg Flag-labeled
anti-HK2 (1/200, ab209847, Abcam) at 4°C overnight, then with
protein A/G magnetic beads (Thermo Fisher Scientific) in accordance
with manufacturer's protocol. After being centrifuged at 3,000 × g
for 3 min at 4°C, the magnetic beads were deposited at the bottom
of the tube. Removing the supernatant, the magnetic beads were
washed three times in 1 ml lysis buffer. After being added 15
µl 2xSDS loading buffer, the magnetic beads were boiled for
5 min. The gel bands were excised for in-gel trypsin digestion.
Subsequently, extracted peptides were analyzed using nano-LC-MS/MS
(AB SCIEX TripleT OF 5600) for mass spectrometry. Mass spectrometry
weas conducted on an Orbitrap Fusion Tribrid mass spectrometer
(Thermo Fisher Scientific, Inc.) equipped with an Easynano UHPLC,
using a 75 µm x 250 mm Pepmap RSLC reverse phase column with
a PepMap 1000 trapping column (Thermo Fisher Scientific). The mass
spectrometer was equipped with an electrospray ionization (ESI) and
operated in the positive ion mode to monitor the m/z transitions
for all peptides and IS. Two multiple reaction monitorings were
simultaneously monitored for each peptide using manually optimized
declustering potentials (DP), collision energies (CE), collision
cell entrance (CEP) and exit potentials (CXP). Nitrogen gas was
desorbed from the SPME fibre in the injector port at 200°C, under
splitless injection mode. The spray voltage was set as 5,000 V, the
atomization pressure was set as 45 psi, the auxiliary gas pressure
was set as 10 psi. Nitrogen gas with high purity (≥99%) was used as
the carrier gas (constant flow of 1.1 ml/min; average velocity 37
cm/s).
IP
To detect ubiquitination of HK2, transfected AGS
cells were cultured for 24 h at room temperature in PBS with 0.1%
formaldehyde. Cells were treated with the proteasome inhibitor
MG132 (10 µM) at room temperature for 8 h. GC cells were
lysed in IP buffer (Beyotime Institute of Biotechnology, Shanghai,
China) with protease and protein phosphatase inhibitors (Roche
Applied Science). Cell lysates (100 ml) were then
immunoprecipitated with anti-β-actin (1/1,000, ab8227, Abcam) and
anti-HK2 (1/1,000, ab209847, Abcam) at 4°C overnight. To measure
HK2 ubiquitination, the samples were incubated with anti-ubiquitin
(1/1,000, 10201-2-AP, Proteintech Group, Inc.) 4°C overnight and
then subjected to western blot analysis as described above.
Luciferase reporter assay
The wild-type or mutant (MUT) sequences of
circRPS19/USP7 3′ untranslated region (UTR) were inserted into
pmirGLO vectors (Promega Corporation) to construct
pmirGLO-circRPS19/USP7-3′ UTR or pmirGLO-circRPS19/USP7-3′ UTR
(MUT). pmirGLO-circRPS19 and pmirGLO-circRPS19 (MUT) were
co-transfected into AGS or HGC-27 cells with miR-125a-5p mimics or
mimics-NC using Lipofectamine 2000 Transcription Reagent
(Invitrogen; Thermo Fisher Scientific, Inc.) at 37°C for 36 h.
pmirGLO-USP7-3′ UTR or pmirGLO-USP7-3′ UTR (MUT) were
co-transfected into AGS or HGC-27 cells with miR-125a-5p mimics or
mimics-NC using Lipofectamine 2000 Transcription Reagent
(Invitrogen; Thermo Fisher Scientific, Inc.) at 37°C for 48 h. The
duration between end of transfectioin and activity measurement is
48 h. After that, relative luciferase activity was measured using a
Dual-Lucy Assay kit (Progema) with Renilla luciferase
activity as the internal reference. The sequence of miR-125a-5p
mimics: 5′-UCC CUG AGA CCC UUU AAC CUG UGA-3′; the sequence of
NC-mimics: 5′-UCU CCG AGA UCU UAC CAC CUG GUA-3′.
RNA pulldown assay
RNA pulldown assay was performed in AGS or HGC-27
cells to demonstrate RNA interaction. Briefly, 1 µg
biotin-labeled circRPS19/USP7-3′ UTR synthesized by Guangzhou
RiboBio Co., Ltd. was added to 1% Triton X-100, followed by heating
at 95°C for 2 min and cooling in an ice bath for 3 min. Next, the
denatured biotinylated RNA was mixed with 15 µl streptavidin
beads (Thermo Fisher Scientific, Inc.). RNA-bead complexes were
incubated with 100 ml cell lysate obtained using RIPA lysis buffer.
After purification, RNA in pulldown products was analyzed by
RT-qPCR as described above.
RNA-binding protein IP (RIP) assay
AGS cells were lysed in RIPA buffer for RIP assay
using Magna RIP kit (MilliporeSigma). Briefly, cell lysate was
treated with RIP buffer at room temperature for 3 h and added to 10
mg/ml magnetic beads (Thermo Fisher Scientific, Inc.) conjugated
with anti-argonaute 2 (Ago2, 1/200, 04-642, MilliporeSigma) or NC
anti-IgG (1/200, AP113, MilliporeSigma). After washing the beads
with 500 µl RIP Wash Buffer, the complexes were incubated
with 150 µl proteinase K at 55°C for 30 min to remove
protein. The enrichment of circRPS19 or USP7 was analyzed by
RT-qPCR as described above.
Animal experiments
A total of 20 male BALB/C nude mice aged 4-6 weeks
and weighed 20±2 g were obtained from Shanghai Lab Animal Research
Center. All mice were raised in specific pathogen free (SPF)
condition at 22-24°C with a stable humidity (55±15%) with free
access to food/water in a 12/12 h light/dark cycle, according to
the guidelines of the local Animal Care and Use Committee at
Affiliated Hospital of Nanjing University of Chinese Medicine.
Animal experiments were approved by Ethics Committee of Affiliated
Hospital of Nanjing University of Chinese Medicine (approval NO.
2021DW-35-01, Nanjing, China). All nude mice were randomly
classified into two groups (n=10). Each mouse was subcutaneously
injected with 2×106 AGS cells transfected with si-NC or
si-circRPS19-1 (5 nmol/injection). Tumors were monitored on day 7
after injection and measured every three days. On day 28, all nude
mice were subjected to euthanasia via cervical dislocation
following intravenous injection with 30 mg/kg pentobarbital sodium
(23) and tumors were resected and
weighed. Death was confirmed by lack of spontaneous breathing for
2-3 min and loss of eye blink response. Body weight decrease of
15-20% was taken as the humane endpoint. No animals were
prematurely sacrificed according to this endpoint. Tumors were cut
into 4-µm thick small pieces. The tissues were fixed with
10% formalin for 24 h at room temperature, and then embedded in
paraffin at 58-62°C for 30 min. The sections were deparaffinized in
xylene and rehydrated in a graded series of alcohol. A steamer was
used for 20 min in an EDTA buffer for antigen retrieval at 92°C.
Next, samples were treated with 3% hydrogen peroxide in methanol at
room temparature for 20 min to block endogenous peroxidase activity
and the reaction was blocked by incubating in a 5% solution of
non-fat milk for 1 h. Slides were then incubated with polyclonal
rabbit antibodies against Ki67 (1:200; cat. no. ab16667, Abcam),
HK2 (1/500; cat. no. ab209847, Abcam), or USP7 (1/200, ab239936,
Abcam) at room temperature for 1 h, followed by incubation with the
secondary antibody HRP-polymer anti-rabbit (1/1,000, ab6721, Abcam)
at 37°C for 15 min. The samples were counterstained with DAB) at
room temperature for 5 min. Images of five typical visual fields of
each section were obtained under a light microscope (×200, DMI1,
Leica, Wetzlar, Germany) and analyzed using software (Image-Pro
Plus 6.0; Media Cybernetics, Inc.).
Statistical analysis
Unpaired Student's t test was used to analyze
differences between two groups. Data from three independent assays
are presented as the mean ± SD. SPSS v22.0 software (IBM Corp.) was
used for data analysis. One-way ANOVA followed by LSD (one control
group) or Tukey (more than one control group) post hoc test was
used for comparing >2 groups. P<0.05 was considered to
indicate a statistically significant difference.
Results
CircRPS19 has circular characteristics
and is upregulated in GC cells
circRNAs significantly overexpressed in GC tissue
were screened from GEO database. According to the GSE83521 dataset,
under the condition of P≤0.05, the top eight circRNAs with the
highest logFC values were screened, including circRNA_102614,
circRNA_103122, circRNA_102191, circRNA_100984, circRNA_102615,
circRNA_102777, circRNA_102561 and circRNA_103559 (Fig. 1A). Of these, hsa_circRNA_103122
(24), hsa_circRNA_102191
(25) and hsa_circRNA_102615
(26) have been reported in GC and
were excluded from subsequent study. RT-qPCR was used to probe the
expression levels of the other five candidates in human GC (AGS,
HGC-27, NCI-N87 and SNU-1) and normal gastric epithelial cell lines
(GES-1). circRNA_102561 was most significantly upregulated in all
four GC cell lines compared with GES-1 (Fig. 1B) and was therefore selected for
subsequent experiments. circRNA_102561 (circRPS19) originates from
exons 5 to 6 of its host gene RPS19 (Fig. 1C). PCR-agarose gel electrophoresis
and RT-qPCR were performed in GES-1 cells to validate the circular
structure of circRPS19. As shown by PCR-agarose gel, circRPS19 was
amplified by convergent and divergent primer in cDNA but only
amplified by convergent primer in gDNA (Fig. 1D). Moreover, after the treatment of
RNase R, circRPS19 expression was almost unchanged, while linear
RPS19 mRNA was nearly completely digested. Expression levels of
circRPS19 and RPS19 mRNA at 0, 6, 12, 18, 24 h in GES-1 cells
treated with Actinomycin D (ActD) were assessed. circRPS19
expression remained steady, while RPS19 mRNA level decreased with
time (Fig. 1F). Collectively, the
aforementioned results showed that circRPS19 had a circular
structure and was associated with GC.
circRPS19 knockdown inhibits GC cell
proliferation and aerobic glycolysis
As aforementioned, circRPS19 was upregulated in GC
cell lines but its specific effects on the malignant phenotype of
GC cells is unknown. It was investigated whether knockdown of
circRPS19 alters biological processes of GC cells. RT-qPCR was used
to determine the efficiency of circRPS19-1-specific siRNAs on
silencing circRPS19 in AGS and HGC-27 cells (Fig. S1A). si-circRPS19-1 and
si-circRPS19-2 were selected for subsequent assays owing to their
higher efficiency. CCK-8 and EdU assay showed that GC cell
proliferation decreased following circRPS19 knockdown (Fig. 2A and B). TUNEL assay and flow
cytometry analysis of GC cells showed that GC cell apoptosis was
enhanced following circRPS19 depletion (Fig. 2C and D). circRPS19 facilitated GC
cell proliferation but prevented GC cell apoptosis (Fig. 2A-D). Furthermore, the effect of
circRPS19 knockdown on glucose consumption and lactate production
of GC cells was assessed. Glucose consumption and lactate
production were decreased following circRPS19 knockdown (Fig. 2E and F). circRPS19 ablation led to
a decrease in ECAR but an increase in OCR (Fig. 2G and H). ECAR/OCR ratio was
decreased following circRPS19 interference (Fig. 2I). Taken together, these data
indicated that circRPS19 facilitated GC cell proliferation and
aerobic glycolysis.
 | Figure 2circRPS19 knockdown inhibits GC cell
proliferation and aerobic glycolysis. (A) Proliferation of AGS and
HGC-27 cells following circRPS19 ablation was detected by Cell
Counting Kit-8. (B) Proliferation of AGS and HGC-27 cells following
circRPS19 ablation was detected by EdU assay. (C) TUNEL assay
showed GC cell apoptosis following circRPS19 ablation (scale bar,
200 µm). (D) Flow cytometry assay showed GC cell apoptosis
following circRPS19 ablation. (E) Glucose consumption, (F) lactate
production, (G) ECAR and (H) OCR were determined in GC cells
following circRPS19 silencing. (I) ECAR/OCR ratio was assessed in
GC cells following circRPS19 silencing. *P<0.05,
**P<0.01, ***P<0.001 vs. control group.
circRPS19, circRNA ribosomal protein S19; GC, gastric cancer; ECAR,
extracellular acidification rate; OCR, oxygen consumption rate; OD,
optical density; si, small interfering; NC, negative control. |
circRPS19 upregulates HK2 protein
expression via USP7
A previous study identified three rate-limiting
enzymes in the regulation of aerobic glycolysis, including pyruvate
kinases type M2 (PKM2), HK2 and phosphofructokinase 1 (PFK1)
(14). Thus, the present study
investigated which rate-limiting enzyme was regulated by circRPS19.
RT-qPCR and western blotting showed that knockdown of circRPS19 had
no effect on mRNA levels of these three enzymes (Fig. 3A) but effectively decreased protein
levels of HK2 (Fig. 3B).
Therefore, it was investigated whether circRPS19 influenced
ubiquitination of HK2 protein. IP showed ubiquitination of HK2 was
notably increased by circRPS19 silencing (Fig. 3C). It was hypothesized circRPS19
may stabilize HK2 by directly interacting with it. However, RIP
showed that circRPS19 did not directly bind to HK2 protein
(Fig. 3D). The protein interacting
with HK2 and modulating ubiquitination of HK2 protein was assessed
by Co-IP followed by mass spectrometry. Deubiquitinating enzyme
USP7 interacted with HK2 (Fig.
3E). RT-qPCR and western blotting were performed in AGS cells
to investigate if circRPS19 affected USP7 mRNA and protein
expression. mRNA and protein levels of USP7 were significantly
decreased by circRPS19 depletion (Fig.
3F and G). FISH assay was used to investigate co-localization
of circRPS19 and USP7 mRNA in AGS cells. circRPS19 and USP7 mRNA
were co-localized in the cytoplasm (Fig. 3H). RIP showed that circRPS19 and
USP7 mRNA were more abundant in AGO2 immunoprecipitate compared
with that in IgG immunoprecipitate (Fig. 3I). In summary, circRPS19
upregulated USP7 expression to enhance HK2 protein levels and
promote aerobic glycolysis of GC cells.
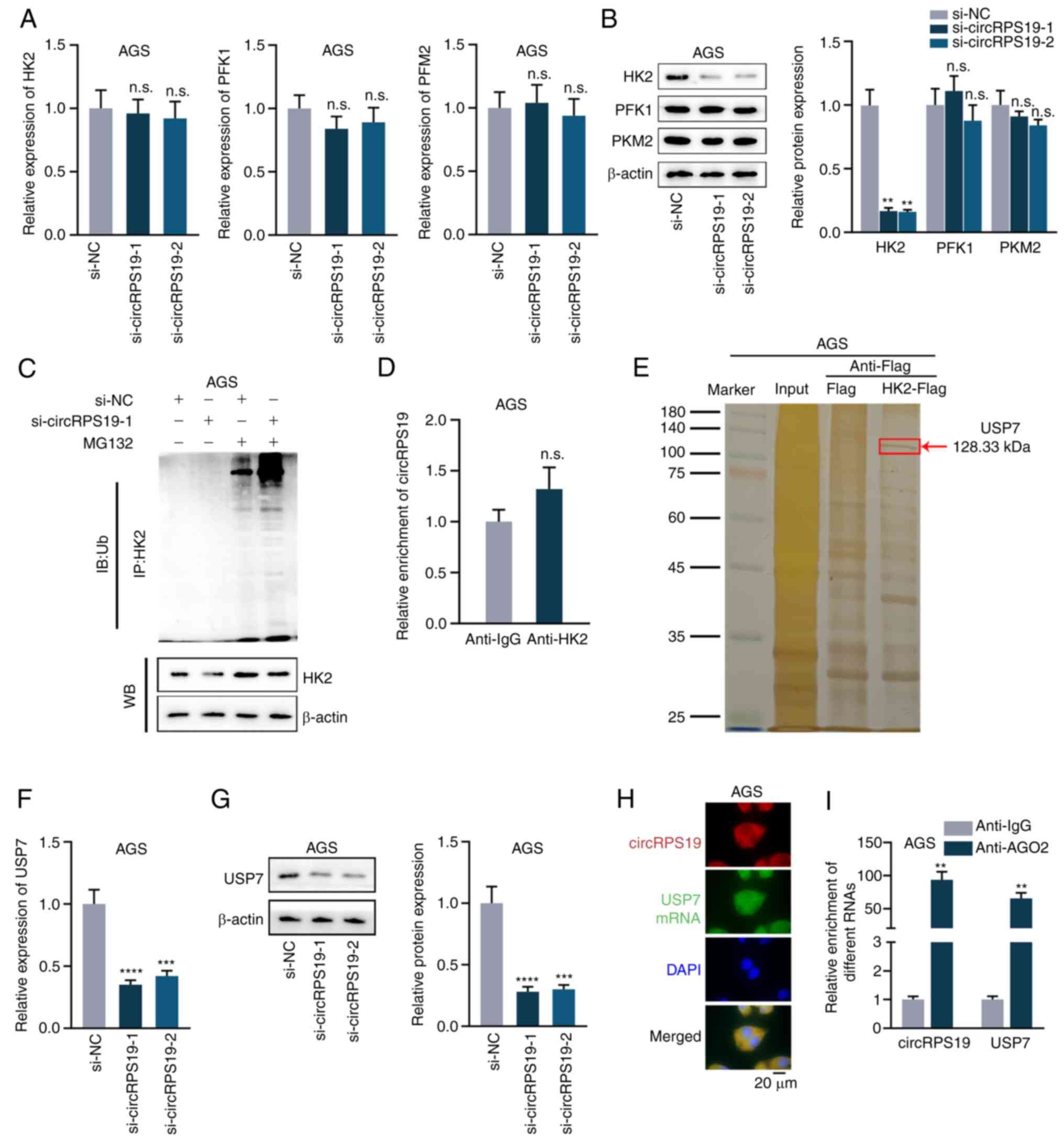 | Figure 3circRPS19 upregulates HK2 protein
expression via USP7. (A) mRNA and (B) protein levels of PKM2, HK2
and PFK1 were assessed by RT-qPCR and WB in AGS cells following
circRPS19 ablation. (C) IP showed HK2 ubiquitination in AGS cells
following circRPS19 depletion. (D) RIP assay was used to examine
the interaction between circRPS19 and HK2 protein in AGS cells. (E)
Co-IP followed by mass spectrometry analysis was used in AGS cells
to determine enzymes affecting ubiquitination of HK2 protein. (F)
mRNA and (G) protein levels of USP7 were assessed by RT-qPCR and WB
in AGS cells following circRPS19 ablation. (H) FISH assay was used
to determine co-localization of circRPS19 and USP7 mRNA in AGS
cells. (I) RIP assay was used to detect abundance of circRPS19 and
USP7 mRNA in RISC. **P<0.01,
***P<0.001, ****P<0.0001 compared with
control group. circRPS19, circRNA ribosomal protein S19; HK2,
hexokinase 2; USP7, ubiquitin-specific processing protease 7; PKM2,
pyruvate kinase M2; PFK1, 6-phosphofructokinase subunit alpha;
RT-q, reverse transcription-quantitative; IP, immunoprecipitation;
FISH, Fluorescence in situ hybridization; RISC, RNA-induced
silencing complex; n.s., not significant; si, small interfering;
NC, negative control; WB, western blot; IB, immunoblot; Ub,
ubiquitin. |
CircRPS19 upregulates USP7 expression via
competitively binding to miR-125a-5p
It was hypothesized that circRPS19 might modulate
USP7 via competitive endogenous RNA (ceRNA) network. starBase
database was used to screen miRNAs that potentially interact with
both circRPS19 and USP7. As a result, three common miRNAs
(hsa-miR-125a-5p, hsa-miR-125b-5p and hsa-miR-338-3p) were obtained
(Figs. S2A and B and 4A). RNA pulldown assay was used for
screening in AGS cells and binding affinity of miR-125a-5p with
circRPS19 was the highest compared with other miRNAs (Fig. 4B). RNA pulldown assay verified the
interaction of miR-125a-5p with circRPS19 (Fig. 4C). RNA pulldown assay in AGS and
HGC-27 cells showed that miR-125a-5p could combine with USP7 3′ UTR
(Fig. 4D). Next, miR-125a-5p was
overexpressed by transfection with miR-125a-5p mimics into AGS and
HGC-27 cells and the overexpression efficiency were detected by
RT-qPCR (Fig. S1B). As indicated
by luciferase reporter assay, upregulation of miR-125a-5p decreased
luciferase activity of pmirGLO-circRPS19/USP7-3′ UTR, while no
obvious changes were observed in MUT groups. miR-125a-5p could bind
with both circRPS19 and USP7 3′ UTR (Fig. 4E and F). RT-qPCR results indicated
that circRPS19 silencing had no effects on miR-125a-5p expression
(Fig. 4G), indicating that
circRPS19 exerted its function by competitively binding to
miR-125a-5p. mRNA and protein levels of USP7 suppressed by
circRPS19 knockdown were completely reversed by miR-125a-5p
inhibition (Fig. 4H and I).
circRPS19 upregulated USP7 expression via competitively binding to
miR-125a-5p.
 | Figure 4circRPS19 upregulates USP7 expression
via competitively binding to miR-125a-5p. (A) Common miRs
interacting with USP7 and circRPS19 predicted by starBase database
(starbase.sysu.edu.cn/). (B) RNA
pulldown assay showed the combination of circRPS19 with
miR-125a-5p, miR-125b-5p and miR-338-3p in AGS cells. (C) RNA
pulldown assay revealed the interaction of miR-125a-5p with
circRPS19 in AGS and HGC-27 cells. (D) RNA pulldown assay revealed
the interaction of miR-125a-5p with USP7 3′-UTR in AGS and HGC-27
cells. (E) Luciferase reporter assay revealed the interaction of
miR-125a-5p with circRPS19 in AGS and HGC-27 cells. (F) Luciferase
reporter assay revealed the interaction of miR-125a-5p with USP7
3′UTR in AGS and HGC-27 cells. (G) miR-125a-5p expression was
probed by RT-qPCR in AGS and HGC-27 cells subsequent to circRPS19
knockdown. (H) mRNA and (I) protein levels of USP7 were assessed by
RT-qPCR and western blot in AGS cells transfected with si-NC,
si-circRPS19-1, si-circRPS19-1 + inhibitor-NC or si-circRPS19-1 +
miR-125a-5p inhibitor. **P<0.01,
***P<0.001, ****P<0.0001 compared with
control group. circRPS19, circRNA ribosomal protein S19; USP7,
ubiquitin-specific processing protease 7; miR, microRNA; UTR,
untranslated region; RT-q, reverse transcription-quantitative; NC,
negative control; bio, biotinylated; MUT, mutant; si, small
interfering; n.s., not significant. |
circRPS19 upregulates H2K protein through
USP7-mediated deubiquitination
Based on the aforementioned interaction between USP7
and HK2, USP7-mediated HK2 deubiquitination was investigated. Co-IP
in AGS cells demonstrated an interaction between USP7 and HK2
(Fig. 5A). Immunofluorescence
assay revealed that USP7 and HK2 were co-localized in the cytoplasm
of AGS cells (Fig. 5B). To
validate the regulatory effect of USP7 on HK2, USP7 expression was
knocked down and efficiency was assessed by RT-qPCR (Fig. S1C). HK2 protein was downregulated
after USP7 interference (Fig. 5C).
Degradation rate of HK2 protein was accelerated after interference
with USP7 in CHX-treated cells (Fig.
5D). The overexpression efficiency of pcDNA3.1-USP7 was
detected by RT-qPCR in AGS and HGC-27 cells (Fig. S1D). The western blot results
showed that increased HK2 protein expression induced by USP7
overexpression was reversed by USP7 inhibitor HBX19818, indicating
that USP7 mediated deubiquitination of HK2 (Fig. 5E). IP showed that HK2
ubiquitination was notably decreased by USP7 knockdown (Fig. 5F). circRPS19 promoted USP7-mediated
deubiquitination of HK2.
 | Figure 5circRNA ribosomal protein S19
upregulates HK2 protein via USP7-mediated deubiquitination. (A)
Co-IP analysis of the interaction between USP7 and HK2 in AGS
cells. (B) Immunofluorescence assay detected the co-localization of
USP7 and HK2 in AGS cells. WB of (C) HK2 protein expression in AGS
cells and (D) degradation rate of HK2 protein in CHX-treated AGS
cells following USP7 ablation and (E) HK2 protein expression
following USP7 overexpression and HBX19818 treatment in AGS cells.
(F) IP showed HK2 ubiquitination in MG132-treated AGS cells
following USP7 depletion. *P<0.05,
**P<0.01, ***P<0.001 vs. control group.
HK2, hexokinase 2; USP7, ubiquitin-specific processing protease 7;
IP, immunoprecipitation; CHX, cycloheximide; HBX19818, a specific
inhibitor of USP7; si, small interfering; NC, negative control; WB,
western blotting; Ub, ubiquitin. |
circRPS19 facilitates GC cell
proliferation and aerobic glycolysis by modulating USP7
In vitro rescue assays were performed to
investigate whether circRPS19 promotes aerobic glycolysis of GC
cells by regulating USP7, thus facilitating GC cell progression. As
indicated by CCK-8 and EdU assay, decreased GC cell proliferation
induced by circRPS19 depletion was counteracted by USP7
overexpression (Fig. 6A and B).
TUNEL and flow cytometry showed that the effect of circRPS19
knockdown on GC cell apoptosis was counteracted by USP7
overexpression (Fig. 6C and D).
The present study investigated the roles of circRPS19 and USP7 in
aerobic glycolysis of GC cells. Glucose consumption and lactate
production suppressed by circRPS19 silencing was restored by USP7
overexpression (Fig. 6E and F). To
summarize, circRPS19 facilitated proliferation and aerobic
glycolysis but inhibited apoptosis of GC cells by modulating
USP7.
circRPS19 promotes GC cell proliferation
and aerobic glycolysis in vivo
The present study performed animal experiments to
detect the effect of circRPS19 on GC in vivo. Tumor volume
in nude mice was measured every 3 days beginning on day 7. The
growth curve showed that circRPS19 knockdown decreased tumor growth
(Fig. 7A). On day 28, tumors were
resected. Size and volume of excised tumors were reduced in
si-circRPS19-1 compared with si-NC group (Fig. 7B and C). Immunohistochemistry
showed that the expression of Ki67, HK2, and USP7 was decreased in
si-circRPS19-1 group compared with si-NC group (Fig. 7D). The effects of circRPS19 on
aerobic glycolysis of tumor cells were analyzed. Glucose
consumption and lactate production of tumor cells were decreased in
si-circRPS19-1 group in comparison with si-NC group (Fig. 7E and F). Therefore, circRPS19
promoted GC cell proliferation and aerobic glycolysis in
vivo.
Discussion
Previous studies have demonstrated that circRNAs
play either a tumor-suppressing or -promoting role in GC: For
example, circRNA circ_001988 can sequester miR-197-3p to hamper
progression of GC both in vitro and in vivo (27); circRNA circ_0004104 enhances GC
progression by sequestering miR-539-3p and regulating ring finger
protein 2 (RNF2) (28). In
addition, circRNA Ran GTPase activating protein 1 (circ-RanGAP1)
targets miR-877-3p to modulate VEGFA expression, thereby promoting
GC cell migration and invasion (29). The present study identified a novel
circRNA, circRPS19, that was notably upregulated in GC cell lines.
Furthermore, circRPS19 knockdown attenuated GC cell proliferation
both in vitro and in vivo. FISH results demonstrated
that circRPS19 and USP7 mRNA localized in the cytoplasm of GC
cells. The cytoplasmic localization is indicative of the regulation
of circRNA on mRNA at the post-transcriptional level (30). As indicated by previous studies,
circRNAs serve as ceRNAs by competitively interacting with miRNAs
to affect levels of target mRNAs: For example, circRNA DNA
topoisomerase IIA as a ceRNA increases sushi domain containing 2
(SUSD2) expression via sequestering miR-346 in glioma cells
(31); circ_0058357 competitively
combines with miR-24-3p to increase AVL9 cell migration associated
(AVL9) expression in non-small cell lung cancer cells (32); circRNA itchy E3 ubiquitin protein
ligase (circ-ITCH) acts as a sponge for miR-199a-5p to enhance
Klotho expression in GC cells (33). It was hypothesized that circRPS19
as a ceRNA binds to a certain miRNA to regulate USP7 mRNA. Through
bioinformatics analysis, miR-125a-5p was screened. The regulatory
influence of miR-125a-5p on GC progression has been studied
previously (34). Additionally,
tyrosine kinase non receptor 2 antisense RNA 1 (TNK2-AS1) targets
miR-125a-5p to modulate the PI3K/Akt pathway, thereby facilitating
epithelial-mesenchymal transition, invasion and migration of GC
cells (35). The present study
showed that miR-125a-5p bound to both circRPS19 and USP7 mRNA.
Rescue assays proved that circRPS19 upregulated USP7 mRNA
expression by competitively binding to miR-125a-5p.
Rate-limiting enzyme HK2 is a key factor in
glycolysis of GC cells (36).
According to previous studies, circRNA cullin 3 (circCUL3)
upregulates transcription factor STAT3, which facilitates HK2
transcription to increase aerobic glycolysis of GC cells (19); circRNF20 increases aerobic
glycolysis of breast cancer cells by upregulating hypoxia inducible
factor-1α, a transcription factor for HK2 (37). The present study showed that
circRPS19 promoted aerobic glycolysis of GC cells by modulating
HK2. Nevertheless, gene expression analysis showed that circRPS19
could only affect HK2 protein, excluding the possibility of ceRNA
network and transcription activation. It was hypothesized that
circRPS19 influences ubiquitination of HK2 protein via
deubiquitinating enzyme USP7. USP7 mediates ubiquitination of
target protein in cancer cells (38). The present study showed that
circRPS19 can upregulate USP7 to mediate deubiquitination of
HK2.
The present study showed that circRPS19 promotes GC
cell proliferation via aerobic glycolysis. Furthermore, circRPS19
mediated deubiquitination of HK2 by USP7, which was also regulated
by circRPS19 via ceRNA network. The present findings provide
understanding of the molecular mechanisms underlying GC, which may
facilitate development of treatment for GC.
Supplementary Data
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
SL and JQ conceived and designed the study. XZ and
JS performed experiments. XZ analyzed and interpreted data. XZ and
JQ wrote the manuscript. XZ and JS confirm the authenticity of all
the raw data. All authors have read and approved the final
manuscript.
Ethics approval and consent to
participate
The present study was reviewed and approved by the
Ethics Committee of Affiliated Hospital of Nanjing University of
Chinese Medicine (approval no. 2021DW-35-01; Nanjing, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgments
Not applicable.
Funding
The present study was supported by National Traditional Chinese
Medicine Inheritance and Innovation Platform Construction Project
by National Administration of Traditional Chinese Medicine (grant
no. Y2020CX570) and College Project of Jiangsu Province Hospital of
Chinese Medicine (No. Y2021ZR12).
References
|
1
|
Liu AG, Zhong JC, Chen G, He RQ, He YQ, Ma
J, Yang LH, Wu XJ, Huang JT, Li JJ, et al: Upregulated expression
of SAC3D1 is associated with progression in gastric cancer. Int J
Oncol. 57:122–138. 2020.PubMed/NCBI
|
|
2
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Machlowska J, Baj J, Sitarz M, Maciejewski
R and Sitarz R: Gastric cancer: Epidemiology, risk factors,
classification, genomic characteristics and treatment strategies.
Int J Mol Sci. 21:40122020. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Joshi SS and Badgwell BD: Current
treatment and recent progress in gastric cancer. CA Cancer J Clin.
71:264–279. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chen W, Zhang K, Yang Y, Guo Z, Wang X,
Teng B, Zhao Q, Huang C and Qiu Z: MEF2A-mediated lncRNA HCP5
inhibits gastric cancer progression via MiR-106b-5p/p21 axis. Int J
Biol Sci. 17:623–634. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang B, Zhang Y, Qing T, Xing K, Li J,
Zhen T, Zhu S and Zhan X: Comprehensive analysis of metastatic
gastric cancer tumour cells using single-cell RNA-seq. Sci Rep.
11:11412021. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tang X, Ren H, Guo M, Qian J, Yang Y and
Gu C: Review on circular RNAs and new insights into their roles in
cancer. Comput Struct Biotechnol J. 19:910–928. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wu T, Sun Y, Sun Z, Li S, Wang W, Yu B and
Wang G: Hsa_circ_0042823 accelerates cancer progression via
miR-877-5p/FOXM1 axis in laryngeal squamous cell carcinoma. Ann
Med. 53:960–970. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang G, Sun D, Li W and Xin Y:
CircRNA_100290 promotes GC cell proliferation and invasion via the
miR-29b-3p/ITGA11 axis and is regulated by EIF4A3. Cancer Cell Int.
21:3242021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhang X, Wang S, Wang H, Cao J, Huang X,
Chen Z, Xu P, Sun G, Xu J, Lv J and Xu Z: Circular RNA circNRIP1
acts as a microRNA-149-5p sponge to promote gastric cancer
progression via the AKT1/mTOR pathway. Mol Cancer. 18:202019.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yang F, Hu A, Li D, Wang J, Guo Y, Liu Y,
Li H, Chen Y, Wang X, Huang K, et al: Circ-HuR suppresses HuR
expression and gastric cancer progression by inhibiting CNBP
transactivation. Mol Cancer. 18:1582019. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ma C, Wang X, Yang F, Zang Y, Liu J, Wang
X, Xu X, Li W, Jia J and Liu Z: Circular RNA hsa_circ_0004872
inhibits gastric cancer progression via the miR-224/Smad4/ADAR1
successive regulatory circuit. Mol Cancer. 19:1572020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Guo R, Cui X, Li X, Zang W, Chang M, Sun
Z, Liu Z, Sun Y, Jia J and Li W: CircMAN1A2 is upregulated by
Helicobacter pylori and promotes development of gastric cancer.
Cell Death Dis. 13:4092022. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Feng J, Li J, Wu L, Yu Q, Ji J, Wu J, Dai
W and Guo C: Emerging roles and the regulation of aerobic
glycolysis in hepatocellular carcinoma. J Exp Clin Cancer Res.
39:1262020. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sun L, Li J, Yan W, Yao Z, Wang R, Zhou X,
Wu H, Zhang G, Shi T and Chen W: H19 promotes aerobic glycolysis,
proliferation, and immune escape of gastric cancer cells through
the microRNA-519d-3p/lactate dehydrogenase A axis. Cancer Sci.
112:2245–2259. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Huang J, Hou S, Xu J, Wu J and Yin J: Long
non-coding RNA OIP5-AS1 promotes cell proliferation and aerobic
glycolysis in gastric cancer through sponging miR-186. Arch Med
Sci. 17:1742–1751. 2021.
|
|
17
|
Li H, Xu H, Xing R, Pan Y, Li W, Cui J and
Lu Y: Pyruvate kinase M2 contributes to cell growth in gastric
cancer via aerobic glycolysis. Pathol Res Pract. 215:1524092019.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zheng M, Wu C, Yang K, Yang Y, Liu Y, Gao
S, Wang Q, Li C, Chen L and Li H: Novel selective hexokinase 2
inhibitor Benitrobenrazide blocks cancer cells growth by targeting
glycolysis. Pharmacol Res. 164:1053672021. View Article : Google Scholar
|
|
19
|
Pu Z, Xu M, Yuan X, Xie H and Zhao J:
Circular RNA circCUL3 accelerates the warburg effect progression of
gastric cancer through regulating the STAT3/HK2 axis. Mol Ther
Nucleic Acids. 22:310–318. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
21
|
Li J, Cheng D, Zhu M, Yu H, Pan Z, Liu L,
Geng Q, Pan H, Yan M and Yao M: OTUB2 stabilizes U2AF2 to promote
the Warburg effect and tumorigenesis via the AKT/mTOR signaling
pathway in non-small cell lung cancer. Theranostics. 9:179–195.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Li Z, Peng Y, Li J, Chen Z, Chen F, Tu J,
Lin S and Wang H: N(6)-methyladenosine regulates glycolysis of
cancer cells through PDK4. Nat Commun. 11:25782020. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Joo DT, Xiong Z, MacDonald JF, Jia Z,
Roder J, Sonner J and Orser BA: Blockade of glutamate receptors and
barbiturate anesthesia: Increased sensitivity to
pentobarbital-induced anesthesia despite reduced inhibition of AMPA
receptors in GluR2 null mutant mice. Anesthesiology. 91:1329–1341.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ding L, Zhao Y, Dang S, Wang Y, Li X, Yu
X, Li Z, Wei J, Liu M and Li G: Circular RNA circ-DONSON
facilitates gastric cancer growth and invasion via NURF complex
dependent activation of transcription factor SOX4. Mol Cancer.
18:452019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang D, Jiang X, Liu Y, Cao G, Zhang X and
Kuang Y: Circular RNA circ_HN1 facilitates gastric cancer
progression through modulation of the miR-302b-3p/ROCK2 axis. Mol
Cell Biochem. 476:199–212. 2021. View Article : Google Scholar
|
|
26
|
Chen B, Ji F, Wen X and Jin Z: Circular
RNA circ_ASAP2 promotes cell viability, migration, and invasion of
gastric cancer cells by regulating the miR-770-5p/CDK6 axis. Int J
Clin Exp Pathol. 13:2806–2819. 2020.PubMed/NCBI
|
|
27
|
Sun D, Wang G, Xiao C and Xin Y:
Hsa_circ_001988 attenuates GC progression in vitro and in vivo via
sponging miR-197-3p. J Cell Physiol. 236:612–624. 2021. View Article : Google Scholar
|
|
28
|
Yue F, Peng K, Zhang L and Zhang J:
Circ_0004104 accelerates the progression of gastric cancer by
regulating the miR-539-3p/RNF2 axis. Dig Dis Sci. 66:4290–4301.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lu J, Wang YH, Yoon C, Huang XY, Xu Y, Xie
JW, Wang JB, Lin JX, Chen QY, Cao LL, et al: Circular RNA
circ-RanGAP1 regulates VEGFA expression by targeting miR-877-3p to
facilitate gastric cancer invasion and metastasis. Cancer Lett.
471:38–48. 2020. View Article : Google Scholar
|
|
30
|
Zhou J, Zhang S, Chen Z, He Z, Xu Y and Li
Z: CircRNA-ENO1 promoted glycolysis and tumor progression in lung
adenocarcinoma through upregulating its host gene ENO1. Cell Death
Dis. 10:8852019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Sang J, Li X, Lv L, Zhang C, Zhang X and
Li G: Circ-TOP2A acts as a ceRNA for miR-346 and contributes to
glioma progression via the modulation of sushi domain-containing 2.
Mol Med Rep. 23:2552021. View Article : Google Scholar :
|
|
32
|
Wei D, Sun L and Feng W: hsa_circ_0058357
acts as a ceRNA to promote non-small cell lung cancer progression
via the hsa-miR-24-3p/AVL9 axis. Mol Med Rep. 23:4702021.
View Article : Google Scholar :
|
|
33
|
Wang Y, Wang H, Zheng R, Wu P, Sun Z, Chen
J, Zhang L, Zhang C, Qian H, Jiang J and Xu W: Circular RNA ITCH
suppresses metastasis of gastric cancer via regulating
miR-199a-5p/Klotho axis. Cell Cycle. 20:522–536. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Li R, Hu Z, Wang Z, Zhu T, Wang G, Gao B,
Wang J and Deng X: miR-125a-5p promotes gastric cancer growth and
invasion by regulating the Hippo pathway. J Clin Lab Anal.
35:e240782021. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Guo L, Ma H, Kong Y, Leng G, Liu G and
Zhang Y: Long non-coding RNA TNK2 AS1/microRNA-125a-5p axis
promotes tumor growth and modulated phosphatidylinositol 3
kinase/AKT pathway. J Gastroenterol Hepatol. 37:124–133. 2022.
View Article : Google Scholar
|
|
36
|
Wang Y, Cao B, Zhao R, Li H, Wei B and Dai
G: Knockdown of circBFAR inhibits proliferation and glycolysis in
gastric cancer by sponging miR-513a-3p/hexokinase 2 axis. Biochem
Biophys Res Commun. 560:80–86. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Cao L, Wang M, Dong Y, Xu B, Chen J, Ding
Y, Qiu S, Li L, Zaharieva EK, Zhou X and Xu Y: Circular RNA
circRNF20 promotes breast cancer tumorigenesis and Warburg effect
through miR-487a/HIF-1α/HK2. Cell Death Dis. 11:1452020. View Article : Google Scholar
|
|
38
|
Lee JE, Park CM and Kim JH: USP7
deubiquitinates and stabilizes EZH2 in prostate cancer cells. Gene
Mol Biol. 43:e201903382020. View Article : Google Scholar
|





















