Gastric cancer (GC) ranks fifth in malignancy
incidence worldwide and is the fourth leading cause of
cancer-related death globally (1). GC is a heterogeneous disease with
multiple histological and molecular subtypes (2), necessitating, for optimal
investigation of GC initiation and progression, the establishment
of reliable preclinical model systems that reflect the
heterogeneity of primary tumors. Individual treatment of GC, due to
disease heterogeneity, varies greatly in clinical practice
(3). In addition, the mechanisms
of GC development remain to be fully elucidated. Although multiple
anti-cancer drugs have been evaluated in Phase I clinical trial
safety testing, only a small number have been successful in Phase
II and III clinical trial efficacy testing (4). The high failure rates observed in
clinical trials highlight the importance of good preclinical models
to better predict clinical outcomes. Patient-derived tumor
xenografts (PDXs), in which tumor fragments from cancer patients
are transplanted directly into immunodeficient mice, are one such
model used in precision medicine. Patient-derived organoids (PDOs),
established by three-dimensional (3D) culture in a matrix, also
function well as an in vitro model for cancer treatment.
Intra- and inter-tumor heterogeneity of the primary tumor is
largely conserved in PDX and PDO model systems (5-10),
which can retain the morphologic and genetic features of the
original tumors. Therefore, PDX and PDO models have great potential
as preclinical research tools for studying individualized tumor
progression and therapy resistance. Given recent advances in both
scientific understanding and technology, PDO and PDX models for GC
have facilitated more in-depth research and individualized
precision treatment (Fig. 1).
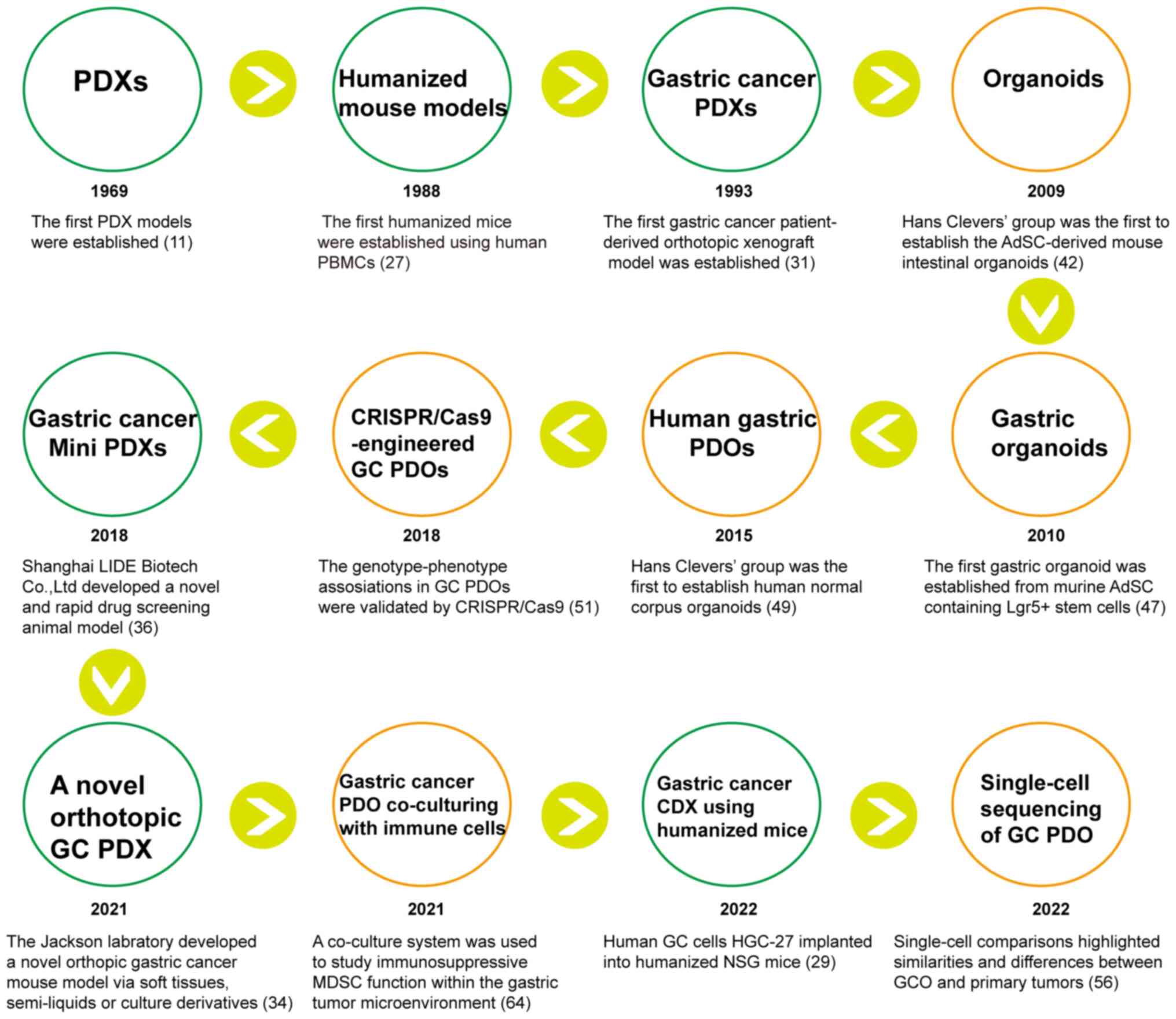
Important discoveries made in GC research using these preclinical
models are summarized in Table I.
These models have enhanced our comprehension of GC progression and
metastasis mechanisms and have been used to forecast patient
therapeutic response to anti-cancer compounds, including
immunotherapy drugs (Table
II).
Animal models have a crucial role in studying the
biological behavior and molecular mechanisms of carcinogenesis and
evaluating drug effectiveness. Cancer cell lines are often
transplanted into immunodeficient mice to generate a model with
easy manipulation and accessibility. However, this model loses the
phenotypic and genetic heterogeneity, as well as the tumor
microenvironment (TME) of the original tumors. A main advantage of
PDXs in cancer research is that the tumor's histopathological
architecture, cancer cells and surrounding stromal cells are
largely preserved. Evidence suggests that the characteristics of
PDX models are highly similar to those of parental tumors and their
response to anti-cancer drugs is also similar to that of patients.
PDXs were first reported in 1969 when the Danish scholar Rygaard
transplanted human colon adenocarcinoma masses into nude mice
(11). As with other tumor PDX
models, immunodeficient mice have been used to establish PDX models
of GC. The immunocompromised mouse strains widely used for PDX
models are as follows: i) Nude mice, which lack a thymus and are
unable to produce T cells, resulting in defective adaptive immune
responses (12); ii) severe
combined immune deficiency (SCID) mice, which lack both functional
T and B lymphocytes (13). Human
tumor engraftment efficiency is higher in SCID mice than in nude
mice (14). Furthermore,
SCID/beige mice, in addition to lacking T and B cells, have a
severe deficiency of natural killer (NK) cell function, so the
engraftment rate of human cancer cells is enhanced in SCID/beige
mice compared to SCID mice (15,16); iii) nonobese diabetic (NOD)/SCID,
interleukin 2 receptor (IL2R)-γnull (NSG or NOG) mice
and NOD/SCID Jak3null (NOJ), in which T-, B- and NK-cell
activity are completely absent, may markedly improve the efficiency
of xenotransplantation (17-19); iv) BALB/c
Rag-2null/IL2R-γnull and
Rag-2null/Jak3null, in which
macrophage-mediated phagocytosis of human cells may be reduced
(20-22); and v) nude
Rag-2null/Jak3null mice, established by
crossing BALB/c Rag-2null/Jak3null mice and
BALB/c nude mice, all serve as powerful tools for evaluating human
tumor-host interactions (23).
Choi et al (24)
successfully established 15 GC PDX models with passaging to
maintain tumors in nude or NOG mice (24.2%, 15/62); the genetic and
histological characteristics of the primary tumors and PDX models
were highly consistent. Karalis et al (25) established 23 PDX models from
Western patients with GC with various ethnic backgrounds. In
theory, highly immunosuppressed mouse strains may allow for higher
tumor engraftment rates, but tumors implanted into NSG (16%) and
nude (21%) mice had a similar engraftment rate, possibly because,
as the immunodeficiency level increases in the recipient mice, the
likelihood of developing B-cell lymphoma also increases, and the
presence of B-cell lymphoma hinders the generation of solid tumor
PDXs. Corso et al (10)
generated a wide, multilevel platform of GC models, including 100
PDXs, organoids and primary cell lines. This PDX platform was the
widest in an academic institution, and included all GC histologic
and molecular types identified by The Cancer Genome Atlas. They
also conducted a transcriptomic analysis of PDXs to identify a
microsatellite instable (MSI) signature with the potential to
assist in the development of precision medicine for GC (10).
The TME includes the extracellular matrix and
stromal cells, which include cancer-associated fibroblasts (CAFs),
immune cells, pro-inflammatory cells and other components. The
interaction between the TME and tumor cells has a prominent role in
tumor progression, metastasis and therapeutic response. However,
during xenograft growth, human stromal cells originally present in
patient-derived tumors are gradually replaced by murine
counterparts, which may hinder the analysis of tumor-stroma
interaction in humans, as certain cytokines from mouse stroma may
not have an impact on human carcinoma cells in PDX models (26). To overcome this limitation in PDX
models, humanized mouse models have been generated. Researchers
engrafted the human immune system and human tumor tissues in animal
models, allowing the human immune system to reconstitute in the
immunodeficient mice with patient tumor engraftment. Improved
humanized mouse models have also been developed, such as i) the
human peripheral blood lymphocytes (Hu-PBL) model, ii) the
Hu-CD34+ model and iii) the bone marrow-liver-thymus
(BLT) mice model. In 1988, Mosier et al (27) established the first of these, the
Hu-PBL model, by injecting peripheral blood mononuclear cells
(PBMCs) intraperitoneally (i.p.) or intravenously (i.v.) into SCID
mice. After the transplantation of PBMC, human CD3+ T
cells could be detected within one week, and ~50% of human
CD45+ cells could be detected in the peripheral blood of
mice after approximately four weeks. The advantage of this approach
was that PBMCs were readily available and easy to manipulate, but
the transplanted mice developed lethal graft vs. host disease
(GVHD) within 2-3 weeks caused by the human T cells attacking mouse
tissue, limiting the model's utility. More importantly, these mice
are incapable of mounting adaptive immune responses with their
engrafted immune systems. With the Hu-CD34+ model,
immunodeficient mice were first given sublethal irradiation to
deplete mouse hematopoietic stem cells (HSCs). Then, human
CD34+ HSCs from human umbilical cord blood, adult bone
morrow, granulocyte colony-stimulating factor-mobilized PBMCs or
fetal liver was injected i.v. or i.p. into newborn or adult
immunodeficient mice. In this model, the CD34+ HSCs can
differentiate into various mature blood cells, such as T cells, B
cells, NK cells or myeloid cells. However, human-derived T-cell
development was low due to the lack of a human thymus. To address
this problem, Lan et al (28) established the Hu-BLT model in
2006, in which immunodeficient mice (NOD/SCID) were also treated
with sublethal (2-3 Gy) whole-body irradiation, after which human
fetal liver and thymus tissue were transplanted into the subrenal
capsule of adult immunodeficient mice, and autologous
CD34+ human HSCs from the same fetal liver or bone
marrow were injected i.v. into the mice, resulting in a stable
model 3-4 months after transplantation. This method achieved a
significant reduction in GVHD symptoms, but the limited donor
source and the complexity of establishment have restricted the use
of this model to study the human immune microenvironment and
infectious diseases. To our knowledge, GC PDX models using
humanized mice have not been reported thus far. However, a
cell-derived xenograft (CDX) model of GC using humanized mice has
been used to evaluate the biological roles of Zinc Finger Protein
64 (ZFP64) in GC for nab-paclitaxel resistance (29). In this study, 3-week-old NSG mice
were injected with cord blood-derived CD34+HSCs;
subsequently, human GC HGC-27 cells were subcutaneously implanted
in the humanized mice. The integration of tumor progression
analysis and humanized mouse models offered a novel approach for
evaluating tumor cell drug resistance, as well as the role of the
immune system in response to chemotherapy.
PDX models can be established by orthotopic or
heterotopic (e.g. subcutaneous, intravenous or intraperitoneal
injection) implantation. Heterotopic engraftments with subcutaneous
injection of patient-derived cancer tissues have been widely used,
as it is easier to manipulate and to monitor tumor growth. However,
compared with heterotopic engraftments, orthotopic models are more
clinically relevant and more suitable for the interpretation of the
mechanisms of cancer metastasis, development and progression
(30). In 1993, Furukawa et
al (31) established the
first patient-derived orthotopic nude mouse models of GC. In total,
tissues from 36 patients with advanced GC were transplanted
orthotopically into nude mice, yielding 20 tumors (56%, 20/36). GC
commonly metastasizes to the liver, lymph nodes, peritoneum, lung
and bone, either through direct invasion or via distant metastases
by lymphatic, hematogenous or intraperitoneal spread (32). Hepatic metastases, observed in
~50% of patients with GC, are the most common distant metastases,
with a survival rate of 4% at five years (33). In one study, the tumor tissues of
five patients with clinical liver metastases also developed liver
metastases in nude mice (31).
Existing orthotopic implantation methods of GC are used to
establish orthotopic stomach tumor models for studying cancer
biology or organ metastasis. However, only certain types of
malignant material have been successfully transplanted, such as
single-cell suspensions or a firm fragment of tumor. In 2021, the
Jackson Laboratory developed a novel, completely closed, orthotopic
GC animal model in NSG mice using diverse tumor materials, such as
soft tissues, semi-liquids or culture derivatives (34). This novel method overcame the
weaknesses of the existing methodologies that supported using only
single-cell suspensions or a firm tumor fragment. Although their
approach required advanced surgical techniques, this procedure can
generate an appropriate animal model for numerous research
purposes, including exploration of biomarker functions, testing the
efficacy of anti-tumor drugs and utilizing GC organoids.
PDXs have emerged as valuable models for predicting
drug responses in GC treatment. However, their limitations,
including being time-consuming and having a lower engraftment rate,
hinder their clinical application in patients with advanced GC due
to rapid disease progression. Thus, there is an urgent need for a
rapid and dependable alternative approach to evaluating drug
sensitivity. The hollow fiber assay has been proposed as a
preliminary screening tool for anticancer agents to identify
sensitive tumor cell lines (35),
but this approach did not have high similarity with clinical
results. In 2018, Shanghai LIDE Biotech Co., Ltd. developed a rapid
drug screening model named OncoVee® MiniPDX (36). In this model, hollow fiber
capsules were filled with patient-derived GC tumors and then
implanted subcutaneously into mice, and they are permitted to grow
for 7 days. The system has shown high similarity between compound
responses of miniPDX and corresponding PDX. Several study groups
have also reported the use of miniPDX models for the treatment of
GC and found that drug screening through this system can provide
significant benefits for patients with GC (37-41). MiniPDX in combination with
next-generation sequencing (NGS) can be used to rapidly evaluate
drug sensitivity in patients with GC and identify key genetic
mutations (39). A single-arm,
open-label phase I clinical study utilizing miniPDX models to
evaluate HER2-negative medium-advanced GC/gastroesophageal junction
cancer chemotherapy regimens and targeted agents resulted in
favorable antitumor activity and safety outcomes (41). In the future, it will presumably
be possible to co-transfer fresh cancer tissues with autoimmune
cells (PBMCs or tumor-infiltrating lymphocytes) from the same
patient with GC into minicapsules and engraft them into
immunodeficient mice, which can capture the human TME to a maximum
extent, allowing for the evaluation of the efficacy of
immunotherapy drugs.
In the last decade, organoids have been established
successfully, serving as a 3D cell cultivation system derived from
adult stem cells (AdSCs) or pluripotent stem cells. In 2009, Hans
Clevers' group was the first to establish the AdSC-derived organoid
system, in which mouse intestinal organoids were cultured in medium
containing the specific growth factors required for growth of
intestinal stem cells (42).
Since then, organoid research has expanded to various organs or
corresponding tumors, including liver (43), kidney (44), lung cancer (45), breast cancer (8) and pancreatic cancer (46). The first gastric organoid culture
derived from murine adult stem cells was established using antrum
glands containing leucine rich repeat containing G protein-coupled
receptor 5-positive stem cells. In these cultures, markers of chief
cells (pepsinogen C) and mucus neck cells (mucin 6) were observed
(47). The same conditions were
used for murine corpus organoids derived from Troy+ stem
cells, also resulting in expression of chief cell and mucus neck
cell markers (48). Subsequently,
human gastric corpus organoid culture protocols were established
based on the murine protocol (49). These normal gastric organoids,
with characteristics similar to those of parental tissues, are a
useful tool to study Helicobacter pylori infection (49). Vlachogiannis et al
(50) reported the first human GC
PDO biobank from patients with metastatic, heavily pretreated
colorectal and gastroesophageal cancer recruited from Phase I/II
clinical trials. In their study, patient drug responses in the
clinic were also observed in the PDOs, indicating their potential
use in personalized medicine. As organoid technologies have
matured, several independent study groups have successfully
generated patient-derived GC organoids (9,51,52). Yan et al (9) established the largest GC biobank
consisting of 46 molecularly characterized GC PDOs, including most
known molecular subtypes of GC, such as Epstein-Barr
virus-positive, MSI, intestinal/chromosomal unstable and
diffuse/genomically stable. The cluster regularly interspaced short
palindromic repeats (CRISPR)-CRISPR-associated protein 9 (Cas 9)
system, originally identified in bacteria as a defense mechanism
against phage infection and plasmid transfer, has been repurposed
as a potent RNA-guided DNA genome editing technology for various
applications, such as gene editing, epigenome editing and
transcriptional perturbation (53). Nanki et al (51) established a living biobank of 37
patient-derived GC organoid lines that included diverse
histological and genetic subtypes. They demonstrated
genotype-phenotype associations in GC organoids and validated their
findings through the CRISPR-Cas9-engineered gastric organoids with
different GC mutations (51).
AT-rich interaction domain 1A (ARID1A) helps regulate gene
expression that drives oncogenesis or tumor suppression (54). However, the oncogenic consequences
of ARID1A mutation in human cells remain poorly defined due
to a lack of accurate genetic models. Lo et al (55) used CRISPR/Cas9 to knock out
ARID1A in primary TP53−/− GC, causing morphologic
dysplasia, tumorigenicity and mucinous differentiation. When
Wnt/β-catenin was activated genetically, mucinous differentiation
was rescued, but not hyperproliferation. This phenotype-genotype
association suggests alternative pathways of ARID1A-mediated
transformation. An independent research group confirmed the
association of ARID1A loss with the induction of a mucinous
phenotype (56).
The presence of stromal cells in the TME, such as
endothelial cells, immune cells and CAFs, contributes significantly
to tumorigenesis, metastasis and treatment resistance (57). Tumors expressing programmed cell
death-ligand 1 (PD-L1) interact with CD8+ cytotoxic T
lymphocytes (CTLs) expressing programmed cell death protein 1
(PD-1) to inhibit CTL proliferation and survival, leading to tumor
evasion of immune surveillance, which in turn leads to increased
proliferation of cancer cells (58,59). More than 40% of patients with GC
have tumors that express PD-L1 (60). However, only 22% (8/36) of
patients with GC have had an overall response to anti-PD-1 antibody
pembrolizumab (61). Therefore,
improved preclinical models are needed that can predict the
efficacy of immune therapies to enhance the survival of patients
with GC. In most organoid models, these crucial components of the
TME are absent. Given the lack of immune cells, a co-culture model
system was established to overcome this drawback. Co-culturing
cancer organoids with immune cells or fibroblasts provided a
valuable tool for investigating the TME and molecular interactions
in cancer treatment. Current checkpoint blockade immunotherapy has
shown remarkable efficacy in unblocking T cells that are negatively
controlled, leading to T cell-mediated anticancer responses.
Several studies of cancer precision medicine have utilized
co-culturing of GC PDOs with immune cells in combination with
checkpoint blockade inhibitors. Chakrabarti et al (62) established a GC patient-derived
organoids/immune cell co-culturing system. Before co-culturing
organoids with CTLs, researchers pulsed antigen-presenting
dendritic cells (DCs) with tumor antigens and then cultured
autologous CTLs with the DCs to increase cytolytic activity and
proliferation of tumor-specific T lymphocytes. Using this
autologous organoid/immune cell co-culture system, they found that
HER2 expression may promote immune evasion in GC that was mediated
by PD-L1. This co-culturing strategy provided a suitable
preclinical model for studying the effect of anti-HER2-targeted
therapy in combination with anti-PD-L1 immunotherapy for patients
with GC (63). In addition, this
system was used to investigate the differentiation and
immunosuppressive function of myeloid-derived suppressor cells
(MDSCs) (64). In another study,
PDO/immune cell co-cultures demonstrated that gastric organoids
expressing PD-L1 were not responsive to nivolumab in vitro
when PMN-MDSCs were present. However, when PMN-MDSCs were depleted
in these co-cultures, the organoids became sensitive to
anti-PD-1/PD-L1-induced cancer cell death (64), suggesting that MDSCs with
immunosuppressive function had an important role in the TME of GC.
These studies have provided valuable insight into predicting
alternative drug regimens and studying the GC microenvironment
using GC PDOs. Thus, advances in co-culture organoid techniques may
yield additional clinical treatment strategies using targeted
therapy and immunotherapy. This platform can also benefit patients
with GC by generating individualized therapy data more rapidly than
animal models.
Single-cell RNA sequencing (scRNA-seq) is a valuable
approach that enables analysis of cancer expression profiles at the
single-cell level, allowing identification and characterization of
unique subpopulations with specific biological behaviors (65). Numerous studies examining the
heterogeneity of tumor cells and comprehensive dynamics in the TME
have been performed using scRNA-seq in GC (66-68). Jiang et al (66) were the first to evaluate the
heterogeneity of GC primary tumors and metastases in different
organs at the single-cell level, demonstrating the characteristics
of different organ-tropism metastases of GC and identifying
effective therapeutic targets. Li et al (67) utilized scRNA-seq to study the role
of CAFs in the GC TME, including their classification, function,
origin, interaction with other cell subsets and spatial
distribution in different pathological types. They found distinct
roles of CAFs in regulating various aspects of TME biology,
including immune modulation, invasion, migration and angiogenesis.
Of note, their study demonstrated that a specific type of CAFs,
known as extracellular matrix CAFs, exhibited an enhanced
chemotaxis ability for attracting M2 macrophages and their presence
was associated with poor prognosis of patients with GC (67). GC commonly metastasizes to lymph
nodes. Qian et al (68)
conducted a comprehensive analysis of the transcriptome profiles of
GC tissues of primary tumors and metastatic lymph nodes (MLNs) at
the single-cell level. They discovered that dysfunctional
neutrophil polarization and maturation had a vital role in lymph
node metastasis of GC. In addition, secreted phosphoprotein 1
(SPP1) signaling, an immune checkpoint, can be activated in MLNs.
Hence, targeting the disordered neutrophils and SPP1 signaling may
be novel strategies to treat and prevent lymph node metastasis of
GC.
Among the platforms to study GC, patient-derived
organoids have emerged as a promising system for investigating
tumor behavior and the influence of TME components. To elucidate
the stepwise progression of the disease from dysplasia to different
stages of adenocarcinoma, including well-differentiated,
poorly-differentiated and metastatic, Lu et al (69) generated a series of
genetically-edited gastric organoids in mice. Through scRNA-seq
analyses and functional studies, they identified an interaction
between tumor cells and macrophages, facilitated by integrin α6/β4
and fibronectin 1, which had an important role in promoting GC
progression and metastasis (69).
To study the extent to which GC organoid in vitro culture
affects transcriptional lineage states or cellular proportions
compared with primary cancer cells in vivo, Kumar et
al (56) performed an overall
analysis of cell states between primary GC cells and organoids by
scRNA-seq and found similarities and differences between primary GC
tissues and organoids. Similar to primary tumors, tumor PDO
epithelial cells showed upregulation of cancer-associated modules
and GC-related genes compared with normal PDO epithelial cells
(56). In addition, they found
differences between primary tumors and PDOs; for instance, stromal
and epithelial cell clusters were significantly enriched in PDOs,
while lymphoid and plasma cell clusters were depleted (56). A gene-expression comparison
between PDO and primary samples found that plasma cells exhibited
the most significant differences in gene expression profile in PDO
models, whereas epithelial signatures were relatively more
conserved. Altogether, gastric organoids are soon expected to use
combinatorial single-cell methods, including epigenetic, genetic
and transcriptional analyses, and spatial context, to further
enhance our understanding of the mechanisms underlying GC
development.
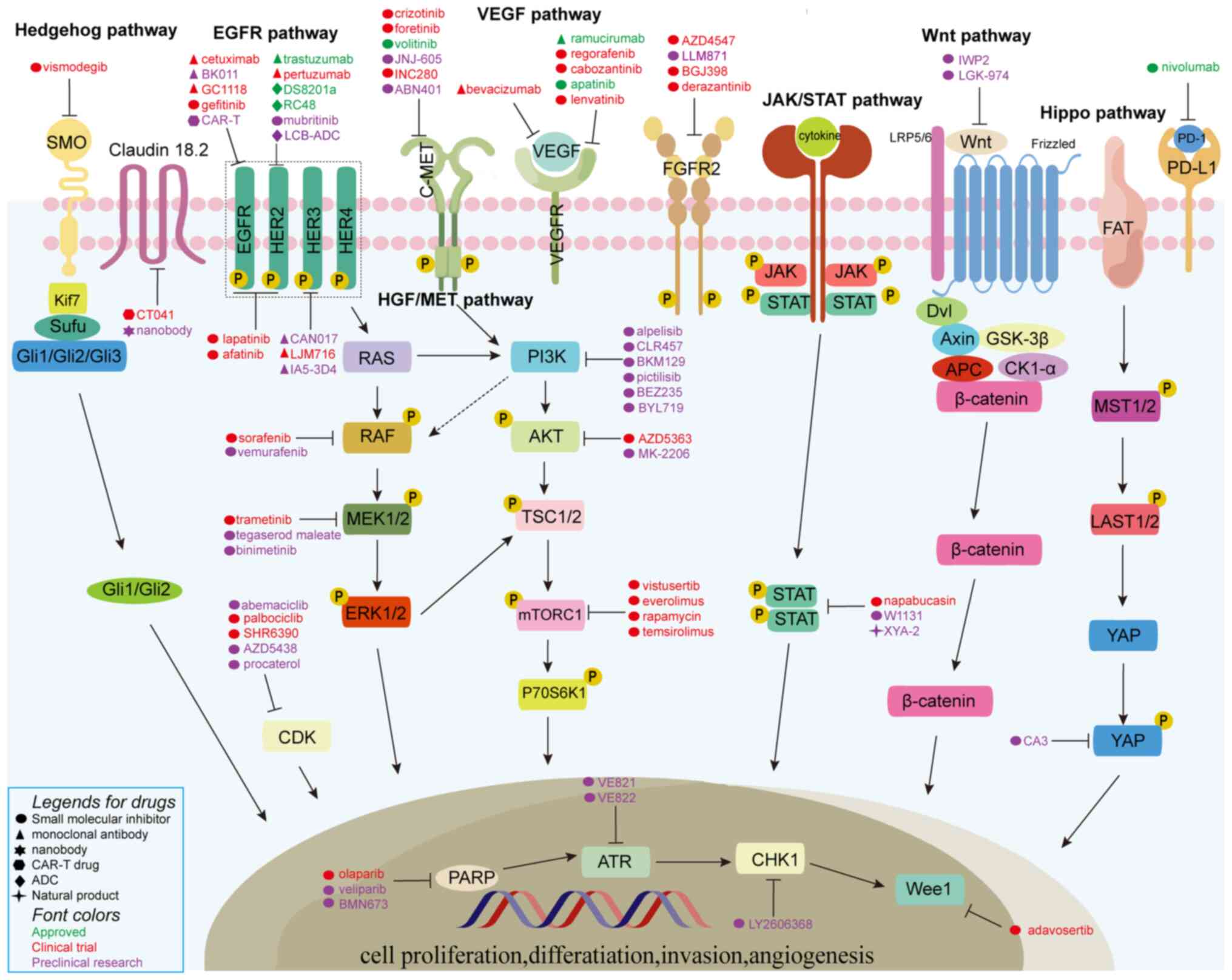
GC PDX models have the potential to emerge as
effective screening platforms for predicting clinical drug response
and determining biomarkers for drug sensitivity and resistance.
Venkatasamy et al (70)
implanted intestinal-type GC tissue samples into nude mice and
generated five PDX models with different degrees of
differentiation, including three well-differentiated, one
moderately and one poorly differentiated adenocarcinoma, which
maintained the heterogeneity and complexity of their primary
tumors. Their data highlighted the complex response of patients to
platinum-based anticancer drugs, which not only affected tumor cell
proliferation but also the TME and remote tissues. Therefore, it is
crucial to consider these factors when developing combination
treatments or new therapeutic protocols. Wang et al
(71) successfully established 13
PDX models, which included four with HER2 (12.5%, 4/32),
eight with cMet (25.0%, 8/32) and one with fibroblast growth
factor receptor 2 (FGFR2) alterations (3.1%, 1/32). These
PDX models offered an ideal platform for drug screening and
efficacy evaluation for particular patients with cMet or
FGFR2 gene amplification who may benefit from the
corresponding targeted therapies. In another study, Chen et
al (72) generated 50 GC PDX
models from patients with advanced GC and the genomic variation and
molecular profile were analyzed by NGS, in situ
hybridization and immunohistochemistry (IHC). Several drug targets,
such as MET and cyclin E1 (CCNE1), were selected and
validated in this study. Volitinib, a MET inhibitor, exhibited
potent antitumor activity in PDX models characterized by MET
overexpression or with phosphorylated MET (72), and the cyclin-dependent kinase
1/2/9 (CDK1/2/9) inhibitor AZD5438 displayed superior
antitumor activity in two PDX models with a higher copy number of
CCNE1 (72). Liu et
al (73) conducted IHC
analysis of human GC tissues to identify the expression level of
CDK12 and then used CDX and PDX models to study the gene
function and molecular interaction between CDK12 and p21-activated
kinase 2 (PAK2). They identified that the food and drug
administration-approved clinical drug procaterol may serve as a
potent CDK12 inhibitor capable of inhibiting GC-cell proliferation
and tumor growth in both models. Thus, CDK12/PAK2 can
serve as a novel therapeutic target for patients with GC.
Although PDX models have proven to be useful in drug
screening and for predicting clinical outcomes, they are not
appropriate for high-throughput drug screening. Compared to PDX
models, PDOs have the advantage of being established and expanded
more efficiently, making them suitable for conducting
high-throughput drug screening. PDOs as preclinical models for
identifying biomarkers and performing genotype-drug associations
are a relatively new area of investigation. The limited studies
conducted thus far have been promising. Chemotherapy is a primary
therapeutic strategy used to treat patients with GC, but
conventional chemotherapeutic agents often cause undesirable
adverse effects. Nanoparticles have recently emerged as potential
treatment options for GC. Compared with conventional
chemotherapeutic drugs, nanoparticles can have improved therapeutic
and pharmacologic features, while simultaneously reducing systemic
toxicity (74). Zou et al
(75) established, from
surgically resected tumor tissues and endoscopic biopsies, nine GC
PDO lines using a multiple-batch dissociation method. Two
representative paclitaxel (PTX) nanoparticles were chosen for a
comparative study and liposomal PTX was more effective than
albumin-bound PTX in killing GC PDOs in both transcriptome and
cellular levels (75). PDX models
have also been used to validate the therapeutic outcomes obtained
through intratumoral drug administration, which provided enhanced
drug concentrations at the local site with reduced systemic
toxicity (75). The evaluation of
nanoparticles using GC PDOs has been crucial to both experimental
and clinical design. Signet-ring cell carcinoma (SRCC) in advanced
GC is defined as being present in exceeding 50% of GC tumors and
was often associated with greater invasiveness and a worse
prognosis compared to other cell types (76). Recently, Li et al (77) generated four SRCC and eight
non-SRCC PDOs, performed a thorough phenotypic and genotypic
analysis, and used5-fluorouracil (5-FU), oxaliplatin, docetaxel and
irinotecan to treat SRCC and non-SRCC organoids. In addition, they
implanted GC PDOs into immunodeficient mice and successfully formed
tumors, which retained the characteristics of the primary
tumors.
Besides classical chemotherapeutics, GC PDOs can
also be treated with targeted drugs against molecular alterations.
GC organoids with AKT serine/threonine kinase 1
(AKT1) mutations were sensitive to the AKT inhibitor MK-2206
(50). Similar to clinical
outcomes, GC PDOs carrying HER2 amplification were sensitive
to trastuzumab (52). Palbociclib
or abemaciclib, which are both inhibitors of CDK4/6, can
effectively suppress the proliferation of GC organoids (9,50,52). The signal transducer and activator
of transcription 3 (STAT3) is a key oncogene, which
functions both in signal transduction and transcriptional
activation (78). In a recent
study, Ouyang et al (79)
found that STAT3 negatively regulated ferroptosis in GC.
They then developed a potent and selec- tive STAT3
inhibitor, W1131, which had powerful antitumor activity in CDX, PDX
and PDO models, suggesting that W1131 may be a novel candidate drug
or therapeutic strategy for advanced GC. Cancer stem cells (CSCs)
have a key role in the acquisition of drug resistance. However,
there is currently no biomarker capable of accurately predicting
5-FU and oxaliplatin resistance in relation to CSCs in clinical
practice. Ukai et al (80)
successfully established four 5-FU-resistant GC PDOs and performed
a microarray analysis using normal gastric organoids with matched
5-FU-resistant and parental PDOs. They determined that KH domain
containing, RNA binding, signal transduction associated 3
(KHDRBS3) may function in the acquisition of CSC-like
features, including multi-drug resistance and organoid formation by
regulating CD44 variant expression (80). Hence, KHDRBS3 may be a
potential marker for predicting treatment response and prognosis in
patients with GC. In another study, Harada et al (81) established oxaliplatin-resistant GC
organoids and evaluated their gene profiles using microarray
analysis. They found that expression of myoferlin in GC was highly
related to oxaliplatin resistance, tumor progression and
unfavorable prognosis.
In the current review, the advantages and
limitations of CDX, PDO and PDX preclinical models of GC in cancer
research and therapy development were discussed (Table III). As organoid technologies
have developed, PDO models have become robust tools for
pathogenesis research. Organoids reflect the genetic and phenotypic
heterogeneity of cancer patients and can be expanded rapidly and
modified genetically using CRISPR-Cas9 technologies. Initially, CDX
models are used for drug screening due to their uncomplicated
technology and ready cell availability. However, CDX models have a
low predictive value for clinical outcomes, rendering them
unsuitable for personalized medicine approaches. In contrast with
CDX models, PDOs are cultured in medium with various niche factors,
thereby increasing cost of maintenance. Organoids have a higher
success rate and operational convenience and are useful for
high-throughput drug screening, compared with PDX models. However,
PDX models preserve tumor heterogeneity and tumor-stromal
interactions observed in patients' tumor tissues, making them more
relevant for studying in vivo cancer biology and for
predicting clinical outcomes. Patient-derived xenograft models may
serve as an 'avatar model', meaning that PDX models derived from
cancer patients participating in a clinical trial can be subjected
to the same treatment given to the patient. This approach
facilitates the identification of new biomarkers for sensitivity or
resistance to specific anti-cancer treatments. While tumor
xenograft and organoid models lack a competent immune environment,
this limitation can be addressed by transplanting HSCs and
co-culturing with immune cells for PDX and PDO models,
respectively.
In brief, preclinical cancer research faces the
challenge of generating reliable models that closely reflect the
patient's condition, including intra-tumor heterogeneity and the
TME. Each model has its strengths and weaknesses, so combining
different preclinical models may enable better precision cancer
research. For instance, PDO models may be used for high-throughput
drug screening, followed by validation of lead candidates or
combinations using patient-derived tumor xenograft models.
Furthermore, combining drug responsiveness data from different
models can lead to more accurate predictions of drug efficacy in
clinical trials. In future investigations, scientists can, on the
one hand, improve organoid culture methods and techniques, and on
the other hand, optimize animal models for more accurate
implantation, dynamic monitoring of tumor cells and evaluating the
immune system, thereby overcoming the limitations of existing
models and developing better preclinical GC models for drug
discovery and personalized medicine.
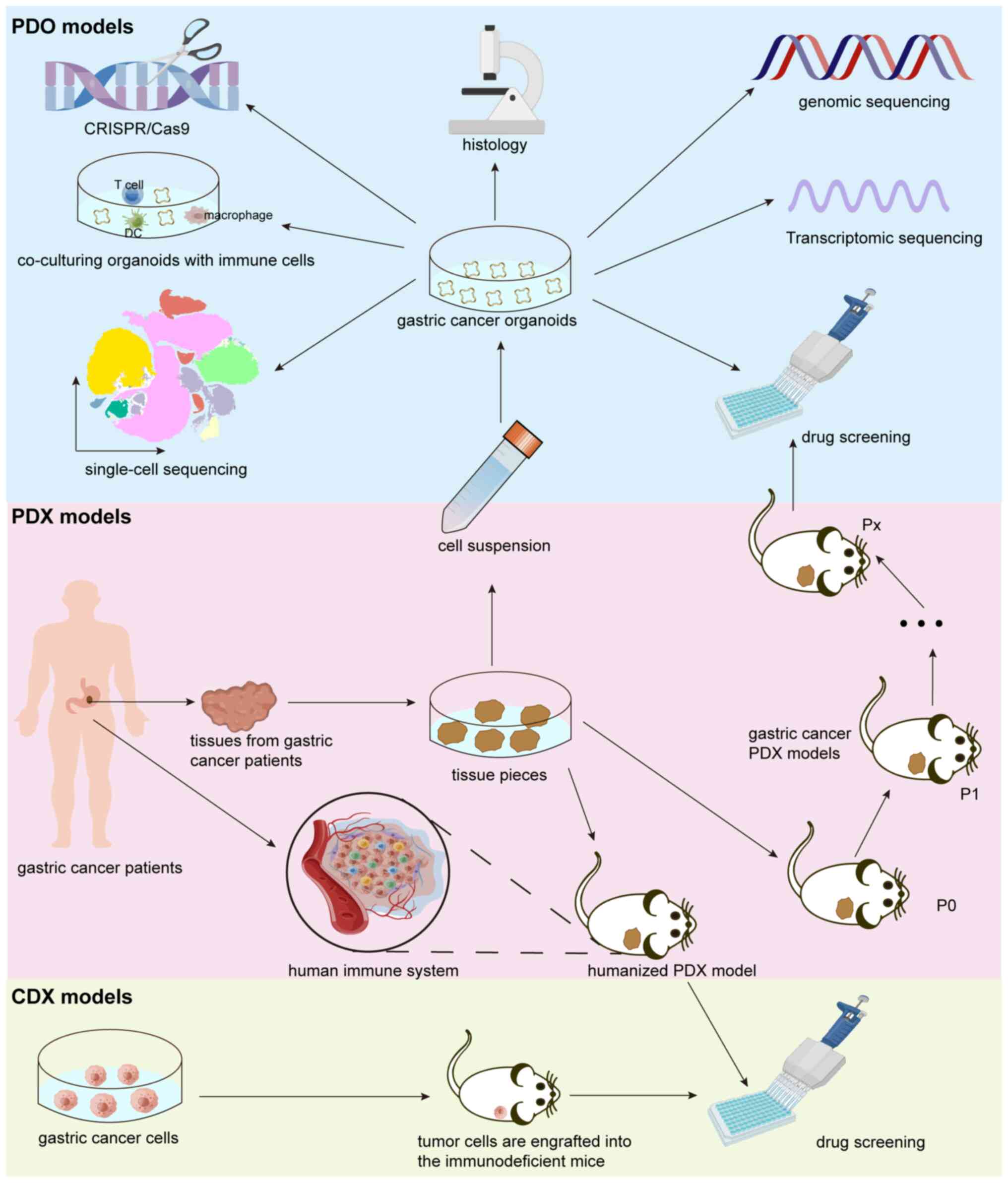
In the present review, the features of the three
mainstream preclinical GC models were highlighted and the
establishment and application of CDX, PDX and PDO model systems in
GC research were discussed (Fig.
2). GC PDX and PDO models not only reflect the morphological
and genetic characteristics of primary tumor tissues, but also
mimic therapeutic responses to anti-cancer treatments. Therefore,
both of these preclinical models may serve to predict individual
responses to diverse treatments (Fig.
3), improving personalized precision medicine. Tumor stromal
cells in the PDX models are gradually replaced during xenograft
passages. Researchers increasingly favor PDO models due to the
rapid time to be established and utility for efficient drug
screening compared to PDX models. Organoids also lack a TME.
Scientists have overcome this common problem by using humanized
mice and co-culturing immune cells to resemble the TME.
Furthermore, with the rapid development of various sequencing and
genetic editing technologies, it is possible to combine whole-exome
sequencing, single-cell sequencing and CRISPR-Cas9 with PDX as well
as PDO models to study the mechanisms of GC development more deeply
and develop individualized treatments for patients with GC.
Not applicable.
JX conceptualized the study, performed the
literature search and drafted the manuscript. JX and BY generated
the figures. FW and JY revised the manuscript. FW and JY were
responsible for project administration and funding acquisition. All
authors have read and agreed to the published version of the
manuscript. Data authentication is not applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
Not applicable.
This study was supported by the gastric cancer of National Major
Disease Multidisciplinary Collaborative Diagnosis and Treatment
Capacity Building Program (grant no. QT252), the Cancer Precision
Medical Science System and Service Platform Building-National Major
Disease Multidisciplinary Collaborative Diagnosis and Treatment
Capacity Building Program (grant no. QT264), the National Natural
Science Foundation of China (grant no. 81902680) and Xi'an Science
and Technology Association Young Talent Support Program (grant no.
095920221304).
|
1
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Smyth EC, Nilsson M, Grabsch HI, van
Grieken NC and Lordick F: Gastric cancer. Lancet. 396:635–648.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Joshi SS and Badgwell BD: Current
treatment and recent progress in gastric cancer. CA Cancer J Clin.
71:264–279. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
DiMasi JA, Feldman L, Seckler A and Wilson
A: Trends in risks associated with new drug development: Success
rates for investigational drugs. Clin Pharmacol Ther. 87:272–277.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Abdolahi S, Ghazvinian Z, Muhammadnejad S,
Saleh M, Asadzadeh Aghdaei H and Baghaei K: Patient-derived
xenograft (PDX) models, applications and challenges in cancer
research. J Transl Med. 20:2062022. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Liu Y, Wu W, Cai C, Zhang H, Shen H and
Han Y: Patient-derived xenograft models in cancer therapy:
Technologies and applications. Signal Transduct Target Ther.
8:1602023. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Mo S, Tang P, Luo W, Zhang L, Li Y, Hu X,
Ma X, Chen Y, Bao Y, He X, et al: Patient-derived organoids from
colorectal cancer with paired liver metastasis reveal tumor
heterogeneity and predict response to chemotherapy. Adv Sci
(Weinh). 9:e22040972022. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sachs N, de Ligt J, Kopper O, Gogola E,
Bounova G, Weeber F, Balgobind AV, Wind K, Gracanin A, Begthel H,
et al: A living biobank of breast cancer organoids captures disease
heterogeneity. Cell. 172:373–386.e10. 2018. View Article : Google Scholar
|
|
9
|
Yan HHN, Siu HC, Law S, Ho SL, Yue SSK,
Tsui WY, Chan D, Chan AS, Ma S, Lam KO, et al: A comprehensive
human gastric cancer organoid biobank captures tumor subtype
heterogeneity and enables therapeutic screening. Cell Stem Cell.
23:882–897. e11. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Corso S, Isella C, Bellomo SE, Apicella M,
Durando S, Migliore C, Ughetto S, D'Errico L, Menegon S, Moya-Rull
D, et al: A comprehensive PDX gastric cancer collection captures
cancer cell-intrinsic transcriptional MSI traits. Cancer Res.
79:5884–5896. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Rygaard J and Povlsen CO:
Heterotransplantation of a human malignant tumour to 'nude' mice.
Acta Pathol Microbiol Scand. 77:758–760. 1969. View Article : Google Scholar
|
|
12
|
Flanagan SP: 'Nude', a new hairless gene
with pleiotropic effects in the mouse. Genet Res. 8:295–309. 1966.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bosma GC, Custer RP and Bosma MJ: A severe
combined immunodeficiency mutation in the mouse. Nature.
301:527–530. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Taghian A, Budach W, Zietman A, Freeman J,
Gioioso D, Ruka W and Suit HD: Quantitative comparison between the
transplantability of human and murine tumors into the subcutaneous
tissue of NCr/Sed-nu/nu nude and severe combined immunodeficient
mice. Cancer Res. 53:5012–5017. 1993.PubMed/NCBI
|
|
15
|
Roder J and Duwe A: The beige mutation in
the mouse selectively impairs natural killer cell function. Nature.
278:451–453. 1979. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Mosier DE, Stell KL, Gulizia RJ, Torbett
BE and Gilmore GL: Homozygous scid/scid;beige/beige mice have low
levels of spontaneous or neonatal T cell-induced B cell generation.
J Exp Med. 177:191–194. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ito M, Hiramatsu H, Kobayashi K, Suzue K,
Kawahata M, Hioki K, Ueyama Y, Koyanagi Y, Sugamura K, Tsuji K, et
al: NOD/SCID/gamma(c)(null) mouse: An excellent recipient mouse
model for engraftment of human cells. Blood. 100:3175–3182. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Okada S, Harada H, Ito T, Saito T and Suzu
S: Early development of human hematopoietic and acquired immune
systems in new born NOD/Scid/Jak3null mice intrahepatic engrafted
with cord blood-derived CD34 + cells. Int J Hematol. 88:476–482.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Shultz LD, Lyons BL, Burzenski LM, Gott B,
Chen X, Chaleff S, Kotb M, Gillies SD, King M, Mangada J, et al:
Human lymphoid and myeloid cell development in NOD/LtSz-scid IL2R
gamma null mice engrafted with mobilized human hemopoietic stem
cells. J Immunol. 174:6477–6489. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ono A, Hattori S, Kariya R, Iwanaga S,
Taura M, Harada H, Suzu S and Okada S: Comparative study of human
hematopoietic cell engraftment into BALB/c and C57BL/6 strain of
rag-2/jak3 double-deficient mice. J Biomed Biotechnol.
2011:5397482011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Traggiai E, Chicha L, Mazzucchelli L,
Bronz L, Piffaretti JC, Lanzavecchia A and Manz MG: Development of
a human adaptive immune system in cord blood cell-transplanted
mice. Science. 304:104–107. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Iwamoto C, Takenaka K, Urata S, Yamauchi
T, Shima T, Kuriyama T, Daitoku S, Saito Y, Miyamoto T, Iwasaki H,
et al: The BALB/c-specific polymorphic SIRPA enhances its affinity
for human CD47, inhibiting phagocytosis against human cells to
promote xenogeneic engraftment. Exp Hematol. 42:163–171.e1. 2014.
View Article : Google Scholar
|
|
23
|
Gotoh K, Kariya R, Matsuda K, Hattori S,
Vaeteewoottacharn K and Okada S: A novel EGFP-expressing nude mice
with complete loss of lymphocytes and NK cells to study tumor-host
interactions. Biosci Trends. 8:202–205. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Choi YY, Lee JE, Kim H, Sim MH, Kim KK,
Lee G, Kim HI, An JY, Hyung WJ, Kim CB, et al: Establishment and
characterisation of patient-derived xenografts as paraclinical
models for gastric cancer. Sci Rep. 6:221722016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Karalis JD, Yoon LY, Hammer STG, Hong C,
Zhu M, Nassour I, Ju MR, Xiao S, Castro-Dubon EC, Agrawal D, et al:
Lenvatinib inhibits the growth of gastric cancer patient-derived
xenografts generated from a heterogeneous population. J Transl Med.
20:1162022. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yoshida GJ: Applications of
patient-derived tumor xenograft models and tumor organoids. J
Hematol Oncol. 13:42020. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Mosier DE, Gulizia RJ, Baird SM and Wilson
DB: Transfer of a functional human immune system to mice with
severe combined immunodeficiency. Nature. 335:256–259. 1988.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lan P, Tonomura N, Shimizu A, Wang S and
Yang YG: Reconstitution of a functional human immune system in
immunodeficient mice through combined human fetal thymus/liver and
CD34+ cell transplantation. Blood. 108:487–492. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhu M, Zhang P, Yu S, Tang C, Wang Y, Shen
Z, Chen W, Liu T and Cui Y: Targeting ZFP64/GAL-1 axis promotes
therapeutic effect of nab-paclitaxel and reverses immunosuppressive
microenvironment in gastric cancer. J Exp Clin Cancer Res.
41:142022. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hoffman RM: Patient-derived orthotopic
xenografts: Better mimic of metastasis than subcutaneous
xenografts. Nat Rev Cancer. 15:451–452. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Furukawa T, Kubota T, Watanabe M, Kitajima
M and Hoffman RM: Orthotopic transplantation of histologically
intact clinical specimens of stomach cancer to nude mice:
Correlation of metastatic sites in mouse and individual patient
donors. Int J Cancer. 53:608–612. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Li W, Ng JM, Wong CC, Ng EKW and Yu J:
Molecular alterations of cancer cell and tumour microenvironment in
metastatic gastric cancer. Oncogene. 37:4903–4920. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Tiberio GAM, Coniglio A, Marchet A,
Marrelli D, Giacopuzzi S, Baiocchi L, Roviello F, de Manzoni G,
Nitti D and Giulini SM: Metachronous hepatic metastases from
gastric carcinoma: A multicentric survey. Eur J Surg Oncol.
35:486–491. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kang W, Maher L, Michaud M, Bae SW, Kim S,
Lee HS, Im SA, Yang HK and Lee C: Development of a novel orthotopic
gastric cancer mouse model. Biol Proced Online. 23:12021.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Hollingshead MG, Alley MC, Camalier RF,
Abbott BJ, Mayo JG, Malspeis L and Grever MR: In vivo cultivation
of tumor cells in hollow fibers. Life Sci. 57:131–141. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhang F, Wang W, Long Y, Liu H, Cheng J,
Guo L, Li R, Meng C, Yu S, Zhao Q, et al: Characterization of drug
responses of mini patient-derived xenografts in mice for predicting
cancer patient clinical therapeutic response. Cancer Commun (Lond).
38:602018.PubMed/NCBI
|
|
37
|
Ge Y, Zhang X, Liang W, Tang C, Gu D, Shi
J and Wei X: OncoVee™-MiniPDX-guided anticancer treatment for
gastric cancer patients with synchronous liver metastases: A
retrospective cohort analysis. Front Oncol. 11:7573832022.
View Article : Google Scholar
|
|
38
|
Wang J, Huang J, Wang H, Yang W, Bai Q,
Yao Z, Li Q, Lv H, Chen B, Nie C, et al: Personalized treatment of
advanced gastric cancer guided by the MiniPDX model. J Oncol.
2022:19877052022.PubMed/NCBI
|
|
39
|
Zhu X, Xu X, Zhang B, Dong Y, Gong S, Gong
T, Zhang F and Jin C: Individualized therapy based on the
combination of mini-PDX and NGS for a patient with metastatic
AFP-producing and HER-2 amplified gastric cancer. Oncol Lett.
24:4112022. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Zhu X, Zhu Y, Chen N, Tang C and Shi J:
The drugs screened by OncoVeeTM-Mini-PDX have
significantly benefited the patient with HER2-positive advanced
gastric cancer. J Oncol Pharm Pract. 28:1435–1440. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zhang B, Li Y, Zhu X, Chen Z, Huang X,
Gong T, Zheng W, Bi Z, Zhu C, Qian J, et al:
OncoVee™-MiniPDX-guided anticancer treatment for HER2-negative
intermediate-advanced gastric cancer patients: A single-arm,
open-label phase I clinical study. Discov Oncol. 14:462023.
View Article : Google Scholar
|
|
42
|
Sato T, Vries RG, Snippert HJ, van de
Wetering M, Barker N, Stange DE, van Es JH, Abo A, Kujala P, Peters
PJ and Clevers H: Single Lgr5 stem cells build crypt-villus
structures in vitro without a mesenchymal niche. Nature.
459:262–265. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Mun SJ, Ryu JS, Lee MO, Son YS, Oh SJ, Cho
HS, Son MY, Kim DS, Kim SJ, Yoo HJ, et al: Generation of expandable
human pluripotent stem cell-derived hepatocyte-like liver
organoids. J Hepatol. 71:970–985. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Homan KA, Gupta N, Kroll KT, Kolesky DB,
Skylar-Scott M, Miyoshi T, Mau D, Valerius MT, Ferrante T,
Bonventre JV, et al: Flow-enhanced vascularization and maturation
of kidney organoids in vitro. Nat Methods. 16:255–262. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Kim M, Mun H, Sung CO, Cho EJ, Jeon HJ,
Chun SM, Jung DJ, Shin TH, Jeong GS, Kim DK, et al: Patient-derived
lung cancer organoids as in vitro cancer models for therapeutic
screening. Nat Commun. 10:39912019. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Driehuis E, van Hoeck A, Moore K, Kolders
S, Francies HE, Gulersonmez MC, Stigter ECA, Burgering B, Geurts V,
Gracanin A, et al: Pancreatic cancer organoids recapitulate disease
and allow personalized drug screening. Proc Natl Acad Sci USA.
116:26580–26590. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Barker N, Huch M, Kujala P, van de
Wetering M, Snippert HJ, van Es JH, Sato T, Stange DE, Begthel H,
van den Born M, et al: Lgr5(+ve) stem cells drive self-renewal in
the stomach and build long-lived gastric units in vitro. Cell Stem
Cell. 6:25–36. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Stange DE, Koo BK, Huch M, Sibbel G, Basak
O, Lyubimova A, Kujala P, Bartfeld S, Koster J, Geahlen JH, et al:
Differentiated Troy+ chief cells act as reserve stem cells to
generate all lineages of the stomach epithelium. Cell. 155:357–368.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Bartfeld S, Bayram T, van de Wetering M,
Huch M, Begthel H, Kujala P, Vries R, Peters PJ and Clevers H: In
vitro expansion of human gastric epithelial stem cells and their
responses to bacterial infection. Gastroenterology. 148:126–136.e6.
2015. View Article : Google Scholar
|
|
50
|
Vlachogiannis G, Hedayat S, Vatsiou A,
Jamin Y, Fernández-Mateos J, Khan K, Lampis A, Eason K, Huntingford
I, Burke R, et al: Patient-derived organoids model treatment
response of metastatic gastrointestinal cancers. Science.
359:920–926. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Nanki K, Toshimitsu K, Takano A, Fujii M,
Shimokawa M, Ohta Y, Matano M, Seino T, Nishikori S, Ishikawa K, et
al: Divergent routes toward Wnt and R-spondin niche independency
during human gastric carcinogenesis. Cell. 174:856–869.e17. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Seidlitz T, Merker SR, Rothe A, Zakrzewski
F, von Neubeck C, Grützmann K, Sommer U, Schweitzer C, Schölch S,
Uhlemann H, et al: Human gastric cancer modelling using organoids.
Gut. 68:207–217. 2019. View Article : Google Scholar
|
|
53
|
Jiang F and Doudna JA: CRISPR-Cas9
structures and mechanisms. Annu Rev Biophys. 46:505–529. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Mullen J, Kato S, Sicklick JK and Kurzrock
R: Targeting ARID1A mutations in cancer. Cancer Treat Rev.
100:1022872021. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Lo YH, Kolahi KS, Du Y, Chang CY,
Krokhotin A, Nair A, Sobba WD, Karlsson K, Jones SJ, Longacre TA,
et al: A CRISPR/Cas9-engineered ARID1A-deficient human gastric
cancer organoid model reveals essential and nonessential modes of
oncogenic transformation. Cancer Discov. 11:1562–1581. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Kumar V, Ramnarayanan K, Sundar R,
Padmanabhan N, Srivastava S, Koiwa M, Yasuda T, Koh V, Huang KK,
Tay ST, et al: Single-cell atlas of lineage states, tumor
microenvironment, and subtype-specific expression programs in
gastric cancer. Cancer Discov. 12:670–691. 2022. View Article : Google Scholar
|
|
57
|
Yuan Y, Jiang YC, Sun CK and Chen QM: Role
of the tumor microenvironment in tumor progression and the clinical
applications (review). Oncol Rep. 35:2499–3515. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Chen DS and Mellman I: Elements of cancer
immunity and the cancer-immune set point. Nature. 541:321–330.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Pardoll DM: The blockade of immune
checkpoints in cancer immunotherapy. Nat Rev Cancer. 12:252–264.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Kim JW, Nam KH, Ahn SH, Park DJ, Kim HH,
Kim SH, Chang H, Lee JO, Kim YJ, Lee HS, et al: Prognostic
implications of immunosuppressive protein expression in tumors as
well as immune cell infiltration within the tumor microenvironment
in gastric cancer. Gastric Cancer. 19:42–52. 2016. View Article : Google Scholar
|
|
61
|
Muro K, Chung HC, Shankaran V, Geva R,
Catenacci D, Gupta S, Eder JP, Golan T, Le DT, Burtness B, et al:
Pembrolizumab for patients with PD-L1-positive advanced gastric
cancer (KEYNOTE-012): A multicentre, open-label, phase 1b trial.
Lancet Oncol. 17:717–726. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Chakrabarti J, Koh V, So JBY, Yong WP and
Zavros Y: A preclinical human-derived autologous gastric cancer
organoid/immune cell co-culture model to predict the efficacy of
targeted therapies. J Vis Exp. 2021:e614432021.
|
|
63
|
Chakrabarti J, Koh V, Steele N, Hawkins J,
Ito Y, Merchant JL, Wang J, Helmrath MA, Ahmad SA, So JBY, et al:
Disruption of Her2-induced PD-L1 inhibits tumor cell immune evasion
in patient-derived gastric cancer organoids. Cancers (Basel).
13:61582021. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Koh V, Chakrabarti J, Torvund M, Steele N,
Hawkins JA, Ito Y, Wang J, Helmrath MA, Merchant JL, Ahmed SA, et
al: Hedgehog transcriptional effector GLI mediates mTOR-Induced
PD-L1 expression in gastric cancer organoids. Cancer Lett.
518:59–71. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Lawson DA, Kessenbrock K, Davis RT,
Pervolarakis N and Werb Z: Tumour heterogeneity and metastasis at
single-cell resolution. Nat Cell Biol. 20:1349–1360. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Jiang H, Yu D, Yang P, Guo R, Kong M, Gao
Y, Yu X, Lu X and Fan X: Revealing the transcriptional
heterogeneity of organ-specific metastasis in human gastric cancer
using single-cell RNA sequencing. Clin Transl Med. 12:e7302022.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Li X, Sun Z, Peng G, Xiao Y, Guo J, Wu B,
Li X, Zhou W, Li J, Li Z, et al: Single-cell RNA sequencing reveals
a pro-invasive cancer-associated fibroblast subgroup associated
with poor clinical outcomes in patients with gastric cancer.
Theranostics. 12:620–638. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Qian Y, Zhai E, Chen S, Liu Y, Ma Y, Chen
J, Liu J, Qin C, Cao Q, Chen J and Cai S: Single-cell RNA-seq
dissecting heterogeneity of tumor cells and comprehensive dynamics
in tumor microenvironment during lymph nodes metastasis in gastric
cancer. Int J Cancer. 151:1367–1381. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Lu Z, Zhong A, Liu H, Zhang M, Chen X, Pan
X, Wang M, Deng X, Gao L, Zhao L, et al: Dissecting the genetic and
microenvironmental factors of gastric tumorigenesis in mice. Cell
Rep. 41:1114822022. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Venkatasamy A, Guerin E, Reichardt W,
Devignot V, Chenard MP, Miguet L, Romain B, Jung AC, Gross I,
Gaiddon C and Mellitzer G: Morpho-functional analysis of
patient-derived xenografts reveals differential impact of gastric
cancer and chemotherapy on the tumor ecosystem, affecting immune
check point, metabolism, and sarcopenia. Gastric Cancer.
26:220–233. 2023. View Article : Google Scholar :
|
|
71
|
Wang H, Lu J, Tang J, Chen S, He K, Jiang
X, Jiang W and Teng L: Establishment of patient-derived gastric
cancer xenografts: a useful tool for preclinical evaluation of
targeted therapies involving alterations in HER-2, MET and FGFR2
signaling pathways. BMC Cancer. 17:1912017. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Chen Z, Huang W, Tian T, Zang W, Wang J,
Liu Z, Li Z, Lai Y, Jiang Z, Gao J and Shen L: Characterization and
validation of potential therapeutic targets based on the molecular
signature of patient-derived xenografts in gastric cancer. J
Hematol Oncol. 11:202018. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Liu H, Shin SH, Chen H, Liu T, Li Z, Hu Y,
Liu F, Zhang C, Kim DJ, Liu K and Dong Z: CDK12 and PAK2 as novel
therapeutic targets for human gastric cancer. Theranostics.
10:6201–6215. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Nagaraju GP, Srivani G, Dariya B,
Chalikonda G, Farran B, Behera SK, Alam A and Kamal MA:
Nanoparticles guided drug delivery and imaging in gastric cancer.
Semin Cancer Biol. 69:69–76. 2021. View Article : Google Scholar
|
|
75
|
Zou J, Wang S, Chai N, Yue H, Ye P, Guo P,
Li F, Wei B, Ma G, Wei W and Linghu E: Construction of gastric
cancer patient-derived organoids and their utilization in a
comparative study of clinically used paclitaxel nanoformulations. J
Nanobiotechnology. 20:2332022. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Huang KH, Chen MH, Fang WL, Lin CH, Chao
Y, Lo SS, Li AF, Wu CW and Shyr YM: The clinicopathological
characteristics and genetic alterations of signet-ring cell
carcinoma in gastric cancer. Cancers (Basel). 12:23182020.
View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Li G, Ma S, Wu Q, Kong D, Yang Z, Gu Z,
Feng L, Zhang K, Cheng S, Tian Y and Zhang W: Establishment of
gastric signet ring cell carcinoma organoid for the therapeutic
drug testing. Cell Death Discov. 8:62022. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Bromberg JF, Wrzeszczynska MH, Devgan G,
Zhao Y, Pestell RG, Albanese C and Darnell JE Jr: Stat3 as an
oncogene. Cell. 98:295–303. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Ouyang S, Li H, Lou L, Huang Q, Zhang Z,
Mo J, Li M, Lu J, Zhu K, Chu Y, et al: Inhibition of
STAT3-ferroptosis negative regulatory axis suppresses tumor growth
and alleviates chemoresistance in gastric cancer. Redox Biol.
52:1023172022. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Ukai S, Honma R, Sakamoto N, Yamamoto Y,
Pham QT, Harada K, Takashima T, Taniyama D, Asai R, Fukada K, et
al: Molecular biological analysis of 5-FU-resistant gastric cancer
organoids; KHDRBS3 contributes to the attainment of features of
cancer stem cell. Oncogene. 39:7265–7278. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Harada K, Sakamoto N, Ukai S, Yamamoto Y,
Pham QT, Taniyama D, Honma R, Maruyama R, Takashima T, Ota H, et
al: Establishment of oxaliplatin-resistant gastric cancer
organoids: Importance of myoferlin in the acquisition of
oxaliplatin resistance. Gastric Cancer. 24:1264–1277. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Li J, Davies BR, Han S, Zhou M, Bai Y,
Zhang J, Xu Y, Tang L, Wang H, Liu YJ, et al: The AKT inhibitor
AZD5363 is selectively active in PI3KCA mutant gastric cancer, and
sensitizes a patient-derived gastric cancer xenograft model with
PTEN loss to Taxotere. J Transl Med. 11:2412013. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Zhang M, Li B, Liao H, Chen Z, Huang W,
Yang J, Ge S, Li Z, Shen L, Zhang C and Gao J: Targeting HER3 or
MEK overcomes acquired trastuzumab resistance in HER2-positive
gastric cancer-derived xenograft. Cell Death Discov. 8:4782022.
View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Yin Y, Shen Q, Zhang P, Tao R, Chang W, Li
R, Xie G, Liu W, Zhang L, Kapoor P, et al: Chk1 inhibition
potentiates the therapeutic efficacy of PARP inhibitor BMN673 in
gastric cancer. Am J Cancer Res. 7:473–483. 2017.PubMed/NCBI
|
|
85
|
Jiang H, Shi Z, Wang P, Wang C, Yang L, Du
G, Zhang H, Shi B, Jia J, Li Q, et al: Claudin18.2-specific
chimeric antigen receptor engineered T cells for the treatment of
gastric cancer. J Natl Cancer Inst. 111:409–418. 2019. View Article : Google Scholar
|
|
86
|
Wei W, Zhang D, Zhang Y, Li L, Jin Y, An
S, Lv C, Zhao H, Wang C, Huang Y, et al: Development and comparison
of 68Ga/18F/64Cu-labeled nanobody
tracers probing Claudin18.2. Mol Ther Oncolytics. 27:305–314. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Gavine PR, Ren Y, Han L, Lv J, Fan S,
Zhang W, Xu W, Liu YJ, Zhang T, Fu H, et al: Volitinib, a potent
and highly selective c-Met inhibitor, effectively blocks c-Met
signaling and growth in c-MET amplified gastric cancer
patient-derived tumor xenograft models. Mol Oncol. 9:323–333. 2015.
View Article : Google Scholar
|
|
88
|
Gao H, Korn JM, Ferretti S, Monahan JE,
Wang Y, Singh M, Zhang C, Schnell C, Yang G, Zhang Y, et al:
High-throughput screening using patient-derived tumor xenografts to
predict clinical trial drug response. Nat Med. 21:1318–1325. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Kim J, Park KE, Jeong YS, Kim Y, Park H,
Nam JH, Jung K, Son WS, Jung HS, Lee JH, et al: Therapeutic
efficacy of ABN401, a highly potent and selective MET inhibitor,
based on diagnostic biomarker test in MET-addicted cancer. Cancers
(Basel). 12:15752020. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Lu J, Li G, He K, Jiang W, Xu C, Li Z,
Wang H, Wang W, Wang H, Teng X and Teng L: Luteolin exerts a marked
antitumor effect in cMet-overexpressing patient-derived tumor
xenograft models of gastric cancer. J Transl Med. 13:422015.
View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Wu QN, Liao YF, Lu YX, Wang Y, Lu JH, Zeng
ZL, Huang QT, Sheng H, Yun JP, Xie D, et al: Pharmacological
inhibition of DUSP6 suppresses gastric cancer growth and metastasis
and overcomes cisplatin resistance. Cancer Lett. 412:243–255. 2018.
View Article : Google Scholar
|
|
92
|
Wang CJ, Tong PJ and Zhu MY: The
combinational therapy of trastuzumab and cetuximab inhibits tumor
growth in a patient-derived tumor xenograft model of gastric
cancer. Clin Transl Oncol. 18:507–514. 2016. View Article : Google Scholar
|
|
93
|
Yu X, Chen Y, Lu J, He K, Chen Y, Ding Y,
Jin K, Wang H, Zhang H, Wang H and Teng L: Patient-derived
xenograft models for gastrointestinal tumors: A single-center
retrospective study. Front Oncol. 12:9851542022. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Park JE, Jin MH, Hur M, Nam AR, Bang JH,
Won J, Oh DY and Bang YJ: GC1118, a novel anti-EGFR antibody, has
potent KRAS mutation-independent antitumor activity compared with
cetuximab in gastric cancer. Gastric Cancer. 22:932–940. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Zhou JT, Liu JH, Song TT, Ma B, Amidula N
and Bai C: EGLIF-CAR-T cells secreting PD-1 blocking antibodies
significantly mediate the elimination of gastric cancer. Cancer
Manag Res. 12:8893–8902. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Ughetto S, Migliore C, Pietrantonio F,
Apicella M, Petrelli A, D'Errico L, Durando S, Moya-Rull D, Bellomo
SE, Rizzolio S, et al: Personalized therapeutic strategies in
HER2-driven gastric cancer. Gastric Cancer. 24:897–912. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Chen Z, Liu Z, Zhang M, Huang W, Li Z,
Wang S, Zhang C, Dong B, Gao J and Shen L: EPHA2 blockade reverses
acquired resistance to afatinib induced by EPHA2-mediated MAPK
pathway activation in gastric cancer cells and avatar mice. Int J
Cancer. 145:2440–2449. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Lau WM, Teng E, Huang KK, Tan JW, Das K,
Zang Z, Chia T, The M, Kono K, Yong WP, et al: Acquired resistance
to FGFR inhibitor in diffuse-type gastric cancer through an
AKT-independent PKC-mediated phosphorylation of GSK3β. Mol Cancer
Ther. 17:232–242. 2018. View Article : Google Scholar
|
|
99
|
McSheehy PMJ, Forster-Gross N, El Shemerly
M, Bachmann F, Roceri M, Hermann N, Spickermann J, Kellenberger L
and Lane HA: The fibroblast growth factor receptor inhibitor,
derazantinib, has strong efficacy in human gastric tumor models and
synergizes with paclitaxel in vivo. Anticancer Drugs. 34:532–543.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Li Y, Fan Y, Xu J, Huo L, Scott AW, Jin J,
Yang B, Shao S, Ma L, Wang Y, et al: GRK3 is a poor prognosticator
and serves as a therapeutic target in advanced gastric
adenocarcinoma. J Exp Clin Cancer Res. 41:2572022. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Shi J, Li F, Yao X, Mou T, Xu Z, Han Z,
Chen S, Li W, Yu J, Qi X, et al: The HER4-YAP1 axis promotes
trastuzumab resistance in HER2-positive gastric cancer by inducing
epithelial and mesenchymal transition. Oncogene. 37:3022–3038.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Ogitani Y, Aida T, Hagihara K, Yamaguchi
J, Ishii C, Harada N, Soma M, Okamoto H, Oitate M, Arakawa S, et
al: DS-8201a, a novel HER2-targeting ADC with a novel DNA
topoisomerase i inhibitor, demonstrates a promising antitumor
efficacy with differentiation from T-DM1. Clin Cancer Res.
22:5097–5108. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Chen Z, Yuan J, Xu Y, Zhang C, Li Z, Gong
J, Li Y, Shen L and Gao J: From AVATAR mice to patients: RC48-ADC
exerted promising efficacy in advanced gastric cancer with HER2
expression. Front Pharmacol. 12:7579942022. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Shin SH, Park YH, Park SS, Ju EJ, Park J,
Ko EJ, Bae DJ, Kim SY, Chung CW, Song HY, et al: An elaborate new
linker system significantly enhances the efficacy of an
HER2-antibody-drug conjugate against refractory HER2-positive
cancers. Adv Sci (Weinh). 8:e21024142021. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Wang Q, Zhang X, Shen E, Gao J, Cao F,
Wang X, Li Y, Tian T, Wang J, Chen Z, et al: The anti-HER3 antibody
in combination with trastuzumab exerts synergistic antitumor
activity in HER2-positive gastric cancer. Cancer Lett. 380:20–30.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Li M, Duan L, Wu W, Li W, Zhao L, Li A, Lu
X, He X, Dong Z, Liu K and Jiang Y: Vortioxetine hydrobromide
inhibits the growth of gastric cancer cells in vivo and in vitro by
targeting JAK2 and SRC. Oncogenesis. 12:242023. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Wang Z, Chen Y, Li X, Zhang Y, Zhao X,
Zhou H, Lu X, Zhao L, Yuan Q, Shi Y, et al: Tegaserod maleate
suppresses the growth of gastric cancer in vivo and in vitro by
targeting MEK1/2. Cancers (Basel). 14:35922022. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Zhang Q, Liu G, Liu J, Yang M, Fu J, Liu
G, Li D, Gu Z, Zhang L, Pan Y, et al: The antitumor capacity of
mesothelin-CAR-T cells in targeting solid tumors in mice. Mol Ther
Oncolytics. 20:556–568. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Cao B, Liu M, Huang J, Zhou J, Li J, Lian
H, Huang W, Guo Y, Yang S, Lin L, et al: Development of
mesothelin-specific CAR NK-92 cells for the treatment of gastric
cancer. Int J Biol Sci. 17:3850–3861. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Fukamachi H, Kim SK, Koh J, Lee HS, Sasaki
Y, Yamashita K, Nishikawaji T, Shimada S, Akiyama Y, Byeon SJ, et
al: A subset of diffuse-type gastric cancer is susceptible to mTOR
inhibitors and checkpoint inhibitors. J Exp Clin Cancer Res.
38:1272019. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Petrelli A, Rizzolio S, Pietrantonio F,
Bellomo SE, Benelli M, De Cecco L, Romagnoli D, Berrino E, Orrù C,
Ribisi S, et al: BRCA2 germline mutations identify gastric cancers
responsive to PARP inhibitors. Cancer Res. 83:1699–1710. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Khalafi S, Zhu S, Khurana R, Lohse I,
Giordano S, Corso S, Al-Ali H, Brothers SP, Wahlestedt C, Schürer S
and El-Rifai W: A novel strategy for combination of clofarabine and
pictilisib is synergistic in gastric cancer. Transl Oncol.
15:1012602022. View Article : Google Scholar
|
|
113
|
Guan X, Yang J, Wang W, Zhao B, Hu S, Yu
D, Yuan L, Shi Y, Xu J, Dong J, et al: Dual inhibition of MYC and
SLC39A10 by a novel natural product STAT3 inhibitor derived from
Chaetomium globosum suppresses tumor growth and metastasis in
gastric cancer. Pharmacol Res. 189:1067032023. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
DiPeri TP, Evans KW, Raso MG, Zhao M,
Rizvi YQ, Zheng X, Wang B, Kirby BP, Kong K, Kahle M, et al:
Adavosertib enhances antitumor activity of trastuzumab deruxtecan
in HER2-expressing Cancers. Clin Cancer Res. 29:4385–4398. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Ajani JA, Xu Y, Huo L, Wang R, Li Y, Wang
Y, Pizzi MP, Scott A, Harada K, Ma L, et al: YAP1 mediates gastric
adenocarcinoma peritoneal metastases that are attenuated by YAP1
inhibition. Gut. 70:55–66. 2021. View Article : Google Scholar
|