Introduction
Cancer cells must create mechanisms with which to
evade the host immune system to manifest as neoplastic entities
fully. This process, also known as ‘immune evasion’, is a
characteristic of malignancies. Cancer immune escape, initially
described in solid tumors, is a feature of several hematological
malignancies (1). When cellular
stress or tissue injury occurs, molecules known as
damage-associated molecular patterns (DAMPs) are generated. These
molecules are known as endogenous danger signals as they trigger
potent inflammatory reactions by triggering the innate immune
system during non-infectious inflammation (2-4).
DAMPs are molecules generated upon cellular stress or tissue injury
(3,4). Previous research has demonstrated that
DAMPs, by causing inflammation, play a key role in the
pathophysiology of human disorders (5). DAMPs include fibrinogen and uric acid.
By activating the NOD-like receptor protein 3, uric acid encourages
T-cells to release interleukin (IL)-1β, while fibrinogen stimulates
macrophages to release inflammatory cytokines through Toll-like
receptor (TLR)2 or 4 (2,6).
Although DAMPs contribute to the defense system of
the host, they promote pathological inflammatory responses. The
well-known promoters of carcinogenesis include IL-1, IL-6 and
lymphotoxin (LT)-β. DAMPs produce IL-1, IL-6, LT-β, IFN-γ, TNF and
transforming growth factor (TGF)-β when they activate inflammatory
pathways (7,8). Examples of these DAMPs include
high-mobility group box 1 (HMGB1), S100 proteins and heat-shock
proteins (HSPs). Adenosine, uric acid, ATP, IL-1α and
immunosuppression all contribute to carcinogenesis through
angiogenesis, inflammation and tumor cell proliferation. In this
instance, it appears that DAMPs promote the growth of tumors during
the initial phases of carcinogenesis (9). Fibrinogen and uric acid, two DAMPs that
are assessed and suitable therapeutic targets, were selected for
analysis in the present study. The aim of the present study was to
examine the association between serum danger molecules and the
survival outcomes of patients with diffuse large B cell lymphoma
(DLBCL) at the time of diagnosis.
Patients and methods
Study design and data collection
The present study was carried out retrospectively.
An evaluation was conducted on 122 patients with DLBCL who were
diagnosed at Selcuk University Hospital (Konya, Turkey) between
2011 and 2024. The hospital database provided the demographic
information of the patients. As per the Declaration of Helsinki
1964, all ethical issues were rigorously adhered to. It was
determined from the patient records that all the study subjects had
provided informed consent at the time of hospitalization, prior to
the administration of chemotherapy, and before receiving any other
pertinent diagnostic or therapeutic standard of care, by the Selcuk
Medical School's standard of care. Ethics committee approval was
received from the Selçuk University Faculty of Medicine (dated
October 22, 2024; no. 2024/533). The laboratory results of the
patients diagnosed with DLBCL, which were examined in detail at the
time of diagnosis, were examined retrospectively. The inclusion
criteria for the study were that the patients were >18 years of
age, had a diagnosis of DLBCL, and the laboratory parameters
analyzed during diagnosis could be obtained from the hospital
database. Patients whose data could not be accessed were excluded
from the study.
Patients, disease characteristics and
definitions
Between 2011 and 2024, 122 patients with DLBCL who
received care in the Hematology Clinic of Selçuk University were
assessed. The study participants were adults who had just been
diagnosed with DLBCL and were being treated in the Department of
Hematology of Selçuk University. All patients had a pathological
diagnosis. Bone marrow aspiration and biopsy were performed for
staging purposes in each patient diagnosed with DLBCL.
Additionally, each patient underwent positron emission tomography
for staging purposes.
High and low fibrinogen and uric acid levels were
determined according to the median fibrinogen and uric acid levels
of all patients. Patients with fibrinogen levels >424 mg/dl at
the time of diagnosis were evaluated as patients with high
fibrinogen levels, while patients with fibrinogen levels ≤424 mg/dl
were evaluated as patients with low fibrinogen levels. Patients
with uric acid levels >5 mg/dl at the time of diagnosis were
evaluated as patients with high uric acid levels, while patients
with uric acid levels ≤5 mg/dl were evaluated as those with low
uric acid levels.
Overall survival (OS) and progression-free survival
(PFS) were used to evaluate the survival outcomes of the patients.
OS was evaluated as the time during which patients were still alive
after receiving the diagnosis of DLBCL. PFS was evaluated as the
period during which the disease did not worsen or progress in
patients with DLBCL during and after treatment.
Statistical analyses
The statistical analyses utilized IBM Corp.'s SPSS
25 (IBM Corp.). Both analytical and visual approaches (such as
probability maps and histograms) were used to explore the
variables. The Kolmogorov-Smirnov/Shapiro-Wilk test was utilized to
confirm the normality of the data. Chi-squared tests were used to
perform statistical comparisons of categorical data. The Student's
t-test was employed to compare continuous numerical data between
two independent samples. Kaplan-Meier analysis was employed to
conduct survival analyses. The log rank test was used to determine
significance. Cox regression analysis was used to provide a
multivariate study of survival factors. The multivariate analysis
comprised parameters that exhibited P≤0.15 in the univariate
analysis. A value of P<0.05 was considered to indicate a
statistically significant difference.
Results
Patients and disease
characteristics
A total of 122 patients were enrolled in the present
study between 2011 and 2024. The median age of the patients was 59
years (range, 22-87 years) and 56.6% (n=69) of the patients were
female. All patients (100%) were diagnosed with DLBCL. The median
fibrinogen level was 424 mg/dl (range, 205-1,165 mg/dl) and the
median uric acid level was 5.0 mg/dl (range, 1.5-12.9 mg/dl) at the
time of diagnosis. In total, 53 patients (43.4%) were evaluated as
ECOG PS 1. According to the Ann Arbor staging system, 64 patients
(52.5%) were evaluated as stage IV according to the PET CT and bone
marrow biopsy results. Extranodal involvement was present in 84
patients (68.9%) at the time of diagnosis. The laboratory and
clinical characteristics of the patients are summarized in Table I.
 | Table ILaboratory and clinical
characteristics of all the patients in the present study. |
Table I
Laboratory and clinical
characteristics of all the patients in the present study.
| Parameter | Value; n (%) or
median (range) |
|---|
| Age (median years,
range) | 58 (22-87) |
| Sex
(female/male) | 69 (56.6)/53
(43.4) |
| Hemoglobin
(mg/dl) | 12.4 (5.7-17.9) |
| Leucocyte
(x109/l) | 7.4 (1.2-77.2) |
| Neutrophil
(x109/l) | 5.1 (0.9-20.0) |
| Monocytes
(x109/l) | 1.4 (0.1-16.1) |
| Lymphocytes
(x109/l) | 0.6 (0.08-46.4) |
| Platelets
(x109/l) | 263 (42-691) |
| Sedimentation rate
(mm/h) | 23 (2-120) |
| C reactive protein
(mg/l) | 20.5 (2.1-396) |
| Ferritin
(ng/m/l) | 192.4
(5.8-15,000) |
| Fibrinogen
(mg/dl) | 424 (205-1,165) |
| Uric acid
(mg/dl) | 5.0 (1.5-12.9) |
| Albumin (g/dl) | 3.8 (2.2-4.9) |
| Lactate dehydrogenase
(U/l) | 291 (122-4,065) |
| B12 (pg/ml) | 308 (75-1,965) |
| ECOG performance
score (0/1/2/3) | 30 (24.6)/53
(43.4)/24 (19.7)/15 (12.3) |
| Stage
(I/II/III/IV) | 11 (9.0)/26 (21.3)/21
(17.2)/64 (52.5) |
| International
Prognostic Score (low/low-intermediate/high-intermediate/high) | 35 (28.7)/39
(32.0)/26 (21.3)/22 (18.0) |
| Revised IPI (very
good/good/poor) | 5 (4.1)/71 (58.2)/46
(37.7) |
| NCCN IPI
(low/low-intermediate/high-intermediate/high) | 10 (8.2)/68 (55.7)/32
(26.2)/12 (9.8) |
| Extranodal
involvement | 84 (68.9%) |
Laboratory and clinical
characteristics of the patients according to danger molecule
levels
In the patients with high fibrinogen levels at the
time of diagnosis, age, neutrophil counts, platelet counts,
sedimentation, C-reactive protein (CRP) levels, ECOG PS, disease
stage, International Prognostic Index (IPI), Revised-International
Prognostic Index (R-IPI), and National Comprehensive Cancer
Network-International Prognostic Index (NCCN-IPI) scores were
higher. By contrast, the albumin levels were significantly lower in
patients with high fibrinogen levels. The relapse and mortality
rates were also higher in patients with high fibrinogen levels
(Table II).
 | Table IILaboratory and clinical
characteristics according to the fibrinogen level at diagnosis. |
Table II
Laboratory and clinical
characteristics according to the fibrinogen level at diagnosis.
| Parameter | Patients with a
fibrinogen level ≤424 mg/dl at diagnosis; n, (%), median
(range) | Patients with a
fibrinogen level >424 mg/dl at diagnosis; n, (%), median
(range) | P-value |
|---|
| No. of patients
(%) | 61 (50%) | 61 (50%) | |
| Age in years. median
(range) | 53 (22-85) | 61 (22-87) | 0.003 |
| Sex
(female/male) | 33 (47.8)/28
(52.8) | 36 (52.2)/25
(47.2) | 0.58 |
| Hemoglobin
(mg/dl) | 12.7 (5.7-17.9) | 12.1 (7.0-15.7) | 0.06 |
| Leucocytes
(x109/l) | 6.5 (4.2-20.5) | 8.3 (1.2-77.2) | 0.08 |
| Neutrophils
(x109/l) | 4.5 (1.7-18.6) | 5.8 (0.9-20.0) | 0.03 |
| Monocytes
(x109/l) | 0.5 (0.08-13.0) | 0.7 (0.1-46.4) | 0.36 |
| Lymphocytes
(x109/l) | 1.4 (0.2-5.3) | 1.5 (0.1-16.1) | 0.47 |
| Platelets
(x109/l) | 238 (42-496) | 267 (102-691) | 0.04 |
| Sedimentation rate
(mm/h) | 18 (2-78) | 29 (4-120) | <0.001 |
| C reactive protein
(mg/l) | 10 (2.1-396) | 34.7 (2.9-245) | 0.03 |
| Ferritin
(ng/m/l) | 110 (12.6-2,000) | 214 (5.8-14,000) | 0.24 |
| Uric acid
(mg/dl) | 5 (2.3-10.0) | 5.2 (1.5-12.9) | 0.41 |
| Albumin (g/dl) | 4.1 (2.4-4.9) | 3.7 (2.2-4.7) | 0.003 |
| Lactate
dehydrogenase (U/l) | 267
(153-1,980) | 314
(122-4,065) | 0.11 |
| B12 (pg/ml) | 294 (75-727) | 312
(158-1,965) | 0.08 |
| ECOG performance
score (0/1/2/3) | 15 (24.6)/33
(54.1)/9 (14.8)/4 (6.6) | 15 (24.6)/20
(32.8)/15 (24.6)/11 (18.0) | 0.04 |
| Stage
(I/II/III/IV) | 6 (9.8)/21
(34.4)/12 (19.7)/22 (36.1) | 5 (8.2)/5 (8.2)/9
(14.8)/42 (68.9) | 0.001 |
| International
prognostic score (low/low-intermediate/high-intermediate/high) | 25 (41.0)/23
(37.7)/8 (13.1)/5 (8.2) | 10 (16.4)/16
(26.2)/18 (29.5)/17 (27.9) | <0.001 |
| Revised IPI (very
good/good/poor) | 20
(32.8)/32(52.5)/9 (14.8) | 8
(13.1)/29(47.5)/24 (39.3) | 0.002 |
| NCCN IPI
(low/low-intermediate/high-intermediate/high) | 7 (11.5)/20
(32.8)/19 (31.1)/15 (24.6) | 3 (4.9)/10
(16.4)/19 (31.1)/29 (47.5) | 0.023 |
| Extranodal
involvement, yes/no | 37 (60.7)/24
(39.3) | 47 (77.0)/14
(23%) | 0.05 |
| Relapse rate,
yes/no | 3 (4.9)/58
(95.1) | 13 (21.3)/48
(78.7) | 0.007 |
| Mortality rate,
yes/no | 5 (8.2)/56
(91.8) | 30 (49.2)/31
(50.8) | <0.001 |
In patients with high uric acid levels at the time
of diagnosis, age and lactate dehydrogenase (LDH) levels were
higher, while B12 levels were significantly lower than in patients
with low uric acid levels. The mortality rates were also higher in
patients with high uric acid levels (Table III).
 | Table IIILaboratory and clinical
characteristics according to uric acid level at diagnosis. |
Table III
Laboratory and clinical
characteristics according to uric acid level at diagnosis.
| Parameter | Patients with uric
acid ≤5 mg/dl at diagnosis; n, (%), median (range) | Patients with uric
acid >5 mg/dl at diagnosis; n, (%), median (range) | P-value |
|---|
| No. of patients
(%) | 64 (52.4%) | 58 (47.6%) | |
| Age in years,
median (range) | 54 (22-86) | 60 (32-87) | 0.03 |
| Sex
(female/male) | 33 (51.6)/31
(48.4) | 36 (62.1)/22
(37.9) | 0.24 |
| Hemoglobin
(mg/dl) | 12.3
(817-15.7) | 12.4
(5.7-15.9) | 0.65 |
| Leucocytes
(x109/l) | 7.4 (3.6-23.6) | 7.4 (1.2-77.2) | 0.40 |
| Neutrophils
(x109/l) | 5.1 (1.7-20.0) | 5.0 (0.9-15.3) | 0.62 |
| Monocytes
(x109/l) | 0.6 (0.08-1.6) | 0.67
(0.10-46.4) | 0.17 |
| Lymphocytes
(x109/l) | 1.4 (5.3-0.26) | 1.4
(0.10-16.1) | 0.26 |
| Platelets
(x109/l) | 266 (24-664) | 233 (70-691) | 0.43 |
| Sedimentation rate
(mm/h) | 21.5 (2-120) | 24 (2-97) | 0.93 |
| C reactive protein
(mg/l) | 15.9 (2.1-245) | 23 (2.5-396) | 0.77 |
| Ferritin
(ng/m/l) | 199
(5.8-2,000) | 169
(17-15,000) | 0.57 |
| Albümin (g/dl) | 3.8 (2.2-4.9) | 3.9 (2.2-4.7) | 0.83 |
| Lactate
dehydrogenase (U/l) | 254
(122-1,980) | 381
(172-4,065) | 0.007 |
| B12 (pg/ml) | 308 (75-1,965) | 305 (140-652) | 0.02 |
| ECOG performance
score (0/1/2/3) | 20 (31.3)/28
(43.8)/11 (17.2)/5 (7.8) | 10 (17.2)/25
(43.1)/13 (22.4)/10 (17.2) | 0.16 |
| Stage
(I/II/III/IV) | 7 (10.9)/15
(23.4)/12 (18.8)/30 (46.9) | 4 (6.9)/11 (19.0)/9
(15.5)/34 (58.6) | 0.61 |
| International
prognostic score (low/low-intermediate/high-intermediate/high) | 21 (32.8)/23
(35.9)/13 (20.3)/7 (10.9) | 14 (24.1)/16
(27.6)/13 (22.4)/15 (25.9) | 0.15 |
| Revised IPI (very
good/good/poor) | 16 (25.0)/35
(54.7)/13 (20.3) | 12 (20.7)/26
(44.8)/20 (34.5) | 0.21 |
| NCCN IPI
(low/low-intermediate/high-intermediate/high) | 7 (10.9)/19
(29.7)/19 (29.7)/19 (29.7) | 3 (5.2)/11
(19.0)/19 (32.8)/25 (43.1) | 0.23 |
| Extranodal
involvement, yes/no | 43 (67.2)/21
(32.8) | 41 (70.7)/17
(29.3) | 0.67 |
| Relapse rate,
yes/no | 9 (14.1)/55
(85.9) | 7 (12.1)/51
(57.9) | 0.74 |
| Mortality rate,
yes/no | 12 (18.8)/52
(71.2) | 23 (39.7)/35
(60.3) | 0.001 |
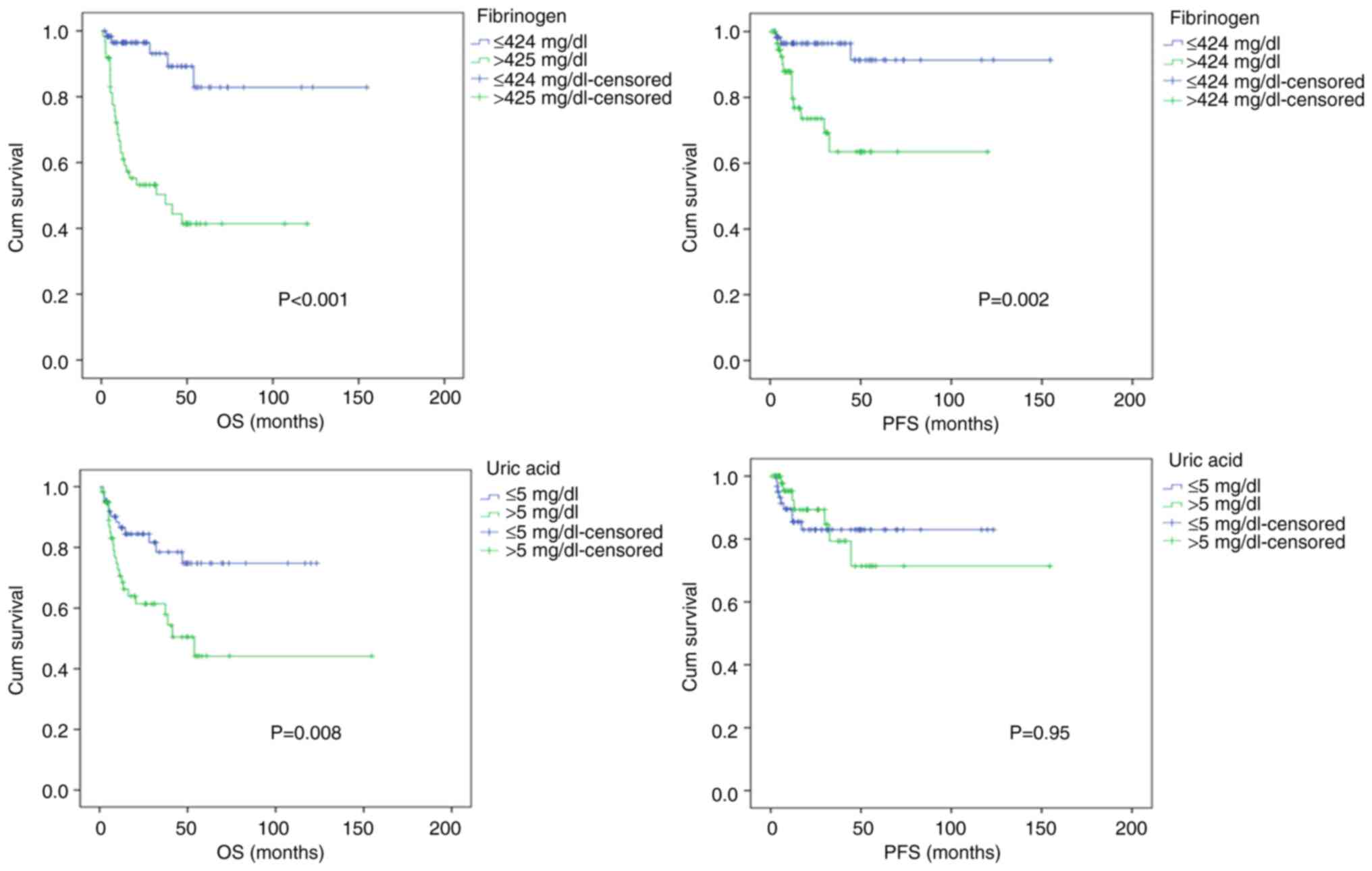
Overall outcomes
The median OS rate was 20 months (range, 0.8-154.5
months) for all patients. Of the 122 patients included in the
study, 61 patients (50%) had fibrinogen levels ≤424 at the time of
diagnosis. The other 61 patients (50%) had fibrinogen levels
>424. The 5-year OS rates of the patients who had a fibrinogen
level ≤424 mg/dl and of those who had a fibrinogen level >424
mg/dl at the time of diagnosis were 82 and 41%, respectively
(P<0.001). The 5-year PFS rates of the patients who had a
fibrinogen level ≤424 mg/dl and of those who had a fibrinogen level
>424 mg/dl at the time of diagnosis were 91 and 63%,
respectively (P=0.002) (Fig. 1).
When the patients were evaluated in terms of uric
acid, another danger molecule, 64 patients (52.5%) had uric acid
levels ≤5 mg/dl, while 58 patients (47.5%) had uric acid levels
>5 mg/dl at the time of diagnosis. The 5-year OS rates of the
patients with a uric acid level ≤5 mg/dl and those with a uric acid
level >5 mg/dl at the time of diagnosis were 74 and 44%,
respectively (P=0.008). The 5-year PFS rates of the patients who
had a uric acid level ≤5 mg/dl those who had a uric acid level
>5 mg/dl at the time of diagnosis were 83 and 71%, respectively
(P=0.95) (Fig. 1).
Cox regression analyses
In univariate analyses, the variables that affected
OS were determined to be an age ≤58 years (P=0.001), fibrinogen
levels ≤424 mg/dl (P<0.001), uric acid levels ≤5 mg/dl)
(P=0.008), an ECOG performance score of 0, 1, 2 and 3)
(P<0.001), a disease stage of I, II, III and IV) (P=0.01), an
International Prognostic Score of low, low-intermediate,
high-intermediate, high) (P<0.001), a revised IPI of very
good/good/poor (P<0.001), NCCN IPI of low, low-intermediate,
high-intermediate, high (P<0.001), extranodal involvement
(P=0.14) and treatment regimen (P=0.12). Cox regression analysis
revealed that fibrinogen levels ≤424 mg/dl) (P=0.002), a revised
IPI (very good/good/poor; P=0.03) and treatment regimen (R-CHOP;
P=0.04) were the parameters predicting OS (Table IV).
 | Table IVUnivariate and multivariate analyses
(Cox model) of overall survival and progression-free survival. |
Table IV
Univariate and multivariate analyses
(Cox model) of overall survival and progression-free survival.
| A, Parameters for
overall survival |
|---|
| | | | Multivariate
analyses |
|---|
| Parameter | Favorable
factors | Univariate analyses
P-value | Hazard ratio | 95% confidence
interval | P-value |
|---|
| Age | ≤58 years | 0.001 | 1.261 | 0.431-3.692 | 0.67 |
| Sex | | 0.81 | - | - | - |
| Fibrinogen
level | ≤424 mg/dl | <0.001 | 5.583 | 1.904-16.378 | 0.002 |
| Uric acid
level | ≤5 mg/dl | 0.008 | 1.688 | 0.771-3.696 | 0.19 |
| ECOG performance
score (0, 1, 2, 3) | | <0.001 | 1.565 | 0.931-2.631 | 0.09 |
| Stage (I, II, III,
IV) | | 0.01 | 1.018 | 0.539-1.922 | 0.95 |
| International
Prognostic Score (low, low-intermediate, high-intermediate,
high) | | <0.001 | 0.979 | 0.434-2.207 | 0.95 |
| Revised IPI (very
good/good/poor) | | <0.001 | 2.210 | 1.079-4.526 | 0.03 |
| NCCN IPI (low,
low-intermediate, high-intermediate, high) | | <0.001 | 1.107 | 0.435-1.747 | 0.69 |
| Extranodal
involvement | None | 0.14 | 1.107 | 0.452-2.711 | 0.82 |
| Treatment
regimen | R-CHOP | 0.12 | 2.367 | 1.029-5.443 | 0.04 |
| B, Parameters for
progression-free survival |
| | | | Multivariate
analyses |
| Parameter | Favorable
factors | Univariate analyses
P-value | Hazard ratio | 95% confidence
interval | P-value |
| Age | ≤58 years | 0.08 | 1.179 | 0.327-4.248 | 0.80 |
| Gender | Male | 0.03 | 3.529 | 0.945-13.176 | 0.06 |
| Fibrinogen
level | ≤424 mg/dl | 0.002 | 5.222 | 1.258-21.676 | 0.02 |
| Uric acid
level | ≤5 mg/dl | 0.95 | - | - | - |
| ECOG performance
score (0, 1, 2, 3) | | 0.01 | 1.798 | 0.778-4.155 | 0.17 |
| Stage (I, II, III,
IV) | | 0.07 | 1.394 | 0.648-3.003 | 0.39 |
| International
Prognostic Score (low, low-intermediate, high-intermediate,
high) | | <0.001 | 3.262 | 0.916-11.621 | 0.06 |
| Revised IPI (very
good/good/poor) | | 0.02 | 0.436 | 0.155-1.224 | 0.11 |
| NCCN IPI (low,
low-intermediate, high-intermediate, high) | | 0.003 | 0.800 | 0.314-2.042 | 0.64 |
| Extranodal
involvement | None | 0.35 | - | - | - |
| Treatment
regimen | R-CHOP | 0.54 | - | - | - |
In the univariate analyses, the variables that
affected PFS were determined to be an age ≤58 years (P=0.08), the
male sex (P=0.03), fibrinogen levels ≤424 mg/dl (P=0.002), an ECOG
performance score (0, 1, 2, 3; P=0.01), disease stage (I, II, III,
IV; P=0.07), an International Prognostic Score (low,
low-intermediate, high-intermediate, high; P<0.001), a revised
IPI (very good/good/poor; P=0.02) and NCCN IPI (low,
low-intermediate, high-intermediate, high; P=0.003). Cox regression
analysis revealed only the fibrinogen level (≤424 mg/dl) (P=0.02),
as a parameter predicting PFS (Table
IV).
Discussion
Numerous DAMPs have been found since Matzinger
(10) developed the hazard model,
and the number of DAMPs is continually increasing (4,5,10,11).
Following tissue damage or cell death, DAMPs are released from the
intracellular or extracellular area (11). Macrophages can identify these DAMPs,
and various pathways, such as TLRs and inflammasomes, can cause an
inflammatory response (11,12). DAMPs can come from a variety of
sources and include plasma proteins such as fibrinogen, Gc-globulin
and serum amyloid A, as well as extracellular proteins such as
biglycan and tenascin C and intracellular proteins, such as HMGB1,
histones, S100 proteins and HSPs (11,13,14).
The role and mechanisms of DAMPs in the etiology of
cancer remain a matter of debate. By causing persistent
inflammation, which is a complex risk factor for tumor growth,
DAMPs may influence tumor progression (7,9). To the
best of our understanding, well-known promoters of carcinogenesis
include IL-1, IL-6 and LT-β (7-9).
DAMPs that trigger inflammatory pathways and produce IL-1, IL-6,
LT-β, IFN-γ, TNF and TGF-β include HMGB1, S100 proteins and HSPs
(9). Through inflammation,
immunosuppression, angiogenesis and tumor cell proliferation, ATP,
IL-1α, adenosine and uric acid also contribute to carcinogenesis
(9). According to this, DAMPs appear
to promote the growth of tumors during the early phases of
carcinogenesis (9).
Plasma glycoproteins, such as fibrinogen are mostly
produced by liver cells, although they are also produced by tumor
and epithelial cells (15).
Fibrinogen is physiologically focused on wound healing, coagulation
and platelet aggregation (16).
Fibrinogen is one of the positive acute phase proteins, known for
their increased quantities during inflammation (17). Shehata et al (18) revealed an association between a
number of clinical and laboratory traits of patients with DLBCL and
hyperfibrinogenemia. Additionally, they evaluated the potential
significance of hyperfibrinogenemia in the prognoses of these
individuals (18). According to
their study, there was a strong association between the baseline
fibrinogen level and laboratory markers, such as platelet count,
serum LDH levels, B2-microglobulin and red cell distribution width.
There were no statistically significant associations observed
between baseline fibrinogen levels and OS, PFS, or responsiveness
to therapy (18). In another study,
in patients with non-Hodgkin lymphoma, Wang et al (19) found a strong association between the
number of extranodal locations implicated and hyperfibrinogenemia.
Additionally, they discovered a propensity for patients with DLBCL
with baseline hyperfibrinogenemia to have a lower 2-PFS (19). Troppan et al (20) revealed that a high fibrinogen plasma
level was associated with decreased 5-year OS and a 5-year PFS in
patients with DLBCL (P<0.001). Furthermore, in their study, in a
multivariate analysis, elevated serum fibrinogen levels were found
to be an independent marker of poor clinical outcomes: 5-year OS
[hazard ratio (HR), 1.69; 95% confidence interval (CI), 1.06-2.72,
P=0.029] and 5-year PFS (HR, 1.68; 95% CI, 1.08-2.61, P=0.021)
(20).
On the other hand, the ultimate byproduct of purine
metabolism, uric acid, demonstrates a high nucleic acid turnover
rate in cellular components. Consequently, elevated uric acid
levels in the blood are frequently observed in individuals with
solid tumors or hematological malignancies. Crucially, multiple
studies have shown that high uric acid levels are associated with a
poor prognosis of patients with several solid cancer types, such as
pancreatic, esophageal and colon cancer, as well as an increased
risk of cancer incidence and mortality (21-23).
Prochazka et al (24)
demonstrated that patients with DLBCL with elevated uric acid
levels before therapy had a statistically significant and
clinically significant low OS and PFS. To date, only a limited
number of studies have examined the association between
pre-treatment uric acid levels and disease prognosis in patients
with DLBCL. Graft vs. host disease (GvHD) and allogeneic
hematopoietic stem cell transplantation (allo-HCT) cause injured
tissue to emit fibrinogen and uric acid (25). Previously, Çelik et al
(25) assessed the effects of
fibrinogen levels and uric acid on GvHD survival. The levels of
uric acid and fibrinogen were assessed on day 0 of allo-HCT and on
days 0, 7, 14 and 28 of GvHD. In multivariable models, fibrinogen
GvHD day 0 was the independent predictor of OS, PFS, and
non-relapse mortality (NRM). Additionally, OS and NRM were
independently predicted by uric acid GvHD on day 28. According to
that study, a considerably shorter OS and greater NRM were linked
to hypouricemia and hypofibrinogenemia (25).
The present study evaluated the effects of
fibrinogen and uric acid levels, which are danger molecules at the
time of diagnosis, on survival outcomes in 122 patients with DLBCL.
Patients with higher fibrinogen levels at the time of diagnosis
were older, and these patients also had higher neutrophil counts,
platelet counts, sedimentation rates and CRP levels at the time of
diagnosis, while the albumin levels were lower. When evaluated
clinically, the ECOG performance score, disease stage,
International Prognostic Score, revised IPI, NCCN IPI, relapse rate
and mortality rate were higher in patients with high fibrinogen
levels. Patients with higher uric acid levels at the time of
diagnosis were also older, and these patients also had higher LDH
levels and lower vitamin B12 levels at diagnosis. When evaluated
clinically, only the mortality rate was higher in patients with
high uric acid levels, unlike the fibrinogen level. OS and PFS were
found to be statistically significantly higher in patients with
lower fibrinogen levels at the time of diagnosis. OS was
statistically significantly higher in patients with lower uric acid
levels at the time of diagnosis, while no statistically significant
difference was observed in PFS. When evaluated with multivariate
analysis, lower fibrinogen levels at the time of diagnosis, lower
R-IPS scores, and chemotherapy protocol (R-CHOP) were found to have
improved OS rates. In the multivariate analysis, only lower
fibrinogen levels at diagnosis were associated with better PFS
rates.
The present study had some limitations. Only
fibrinogen and uric acid levels were evaluated among the danger
molecules. Since this was a retrospective study, other danger
molecules could not be evaluated. However, since fibrinogen and
uric acid are easily accessible tests and can be studied in almost
every patient, it is deemed that this study may prove to be useful
for clinical practice.
In conclusion, the present study demonstrated that
high uric acid and fibrinogen levels were negative prognostic
factors for OS and PFS in patients with DLBCL. These data may
indicate that uric acid and fibrinogen levels at the time of
diagnosis may provide predictive information to the existing
library of clinical risk prediction tools in DLBCL.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
RC was a main contributor to the conception of the
study, as well as to the literature search for related studies. AEC
and CS were involved in the literature review, in the writing of
the manuscript, and in the analysis and interpretation of the
patients' data. RC and CS collected data. RC and CS confirm the
authenticity of all the raw data. All authors have read and
approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved by Selçuk University
Faculty of Medicine Ethic Committee (dated October 22, 2024; no
2024/533). Written consent was obtained from the participants.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Pizzi M, Boi M, Bertoni F and Inghirami G:
Emerging therapies provide new opportunities to reshape the
multifaceted interactions between the immune system and lymphoma
cells. Leukemia. 30:1805–1815. 2016.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Roh JS and Sohn DH: Damage-associated
molecular patterns in inflammatory diseases. Immune Netw.
18(e27)2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Chen GY and Nuñez G: Sterile inflammation:
Sensing and reacting to damage. Nat Rev Immunol. 10:826–837.
2010.PubMed/NCBI View
Article : Google Scholar
|
|
4
|
Vénéreau E, Ceriotti C and Bianchi ME:
DAMPs from cell death to new life. Front Immunol.
6(422)2015.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Land WG: The role of damage-associated
molecular patterns (DAMPs) in human diseases: Part II: DAMPs as
diagnostics, prognostics and therapeutics in clinical medicine.
Sultan Qaboos Univ Med J. 15:e157–e170. 2015.PubMed/NCBI
|
|
6
|
Braga TT, Forni MF, Correa-Costa M, Ramos
RN, Barbuto JA, Branco P, Castoldi A, Hiyane MI, Davanso MR, Latz
E, et al: Soluble uric acid activates the NLRP3 inflammasome. Sci
Rep. 7(39884)2017.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Grivennikov SI, Greten FR and Karin M:
Immunity, inflammation, and cancer. Cell. 140:883–899.
2010.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Haybaeck J, Zeller N, Wolf MJ, Weber A,
Wagner U, Kurrer MO, Bremer J, Iezzi G, Graf R, Clavien PA, et al:
A lymphotoxin-driven pathway to hepatocellular carcinoma. Cancer
Cell. 16:295–308. 2009.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Hernandez C, Huebener P and Schwabe RF:
Damage-associated molecular patterns in cancer: A double-edged
sword. Oncogene. 35:5931–5941. 2016.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Matzinger P: Tolerance, danger, and the
extended family. Annu Rev Immunol. 12:991–1045. 1994.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Schaefer L: Complexity of danger: The
diverse nature of damage-associated molecular patterns. J Biol
Chem. 289:35237–35245. 2014.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Zhang X and Mosser DM: Macrophage
activation by endogenous danger signals. J Pathol. 214:161–178.
2008.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Smiley ST, King JA and Hancock WW:
Fibrinogen stimulates macrophage chemokine secretion through
toll-like receptor 4. J Immunol. 167:2887–2894. 2001.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Ye RD and Sun L: Emerging functions of
serum amyloid A in inflammation. J Leukoc Biol. 98:923–929.
2015.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Lawrence SO and Simpson-Haidaris PJ:
Regulated de novo biosynthesis of fibrinogen in extrahepatic
epithelial cells in response to inflammation. Thromb Haemost.
92:234–243. 2004.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Polterauer S, Grimm C, Seebacher V, Concin
N, Marth C, Tomovski C, Husslein H, Leipold H, Hefler-Frischmuth K,
Tempfer C, et al: Plasma fibrinogen levels and prognosis in
patients with ovarian cancer: A multicenter study. Oncologist.
14:979–985. 2009.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Xu Q, Yan Y, Gu S, Mao K, Zhang J, Huang
P, Zhou Z, Chen Z, Zheng S, Liang J, et al: A novel
inflammation-based prognostic score: The fibrinogen/albumin ratio
predicts prognoses of patients after curative resection for
hepatocellular carcinoma. J Immunol Res.
2018(4925498)2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Shehata AMF, Aldesoky AI and Gohar SF:
Plasma fibrinogen level as possible prognostic biomarker in diffuse
large B-cell lymphoma. Hematology. 24:103–107. 2019.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Wang JW, Li KC, Shi Z and Tang X: Clinical
significance of plasma fibrinogen level in patients with
non-Hodgkin's lymphoma. Tumor. 34:370–373. 2014.
|
|
20
|
Troppan KT, Melchardt T, Wenzl K, Schlick
K, Deutsch A, Bullock MD, Reitz D, Beham-Schmid C, Weiss L,
Neureiter D, et al: The clinical significance of fibrinogen plasma
levels in patients with diffuse large B cell lymphoma. J Clin
Pathol. 69:326–330. 2016.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Stotz M, Szkandera J, Seidel J, Stojakovic
T, Samonigg H, Reitz D, Gary T, Kornprat P, Schaberl-Moser R,
Hoefler G, et al: Evaluation of uric acid as a prognostic
blood-based marker in a large cohort of pancreatic cancer patients.
PLoS One. 9(e104730)2014.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Yan S, Zhang P, Xu W, Liu Y, Wang B, Jiang
T, Hua C, Wang X, Xu D and Sun B: Serum uric acid increases risk of
cancer incidence and mortality: A systematic review and
meta-analysis. Mediators Inflamm. 2015(764250)2015.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Chen YF, Li Q, Chen DT, Pan JH, Chen YH,
Wen ZS and Zeng WA: Prognostic value of pre-operative serum uric
acid levels in esophageal squamous cell carcinoma patients who
undergo R0 esophagectomy. Cancer Biomark. 17:89–96. 2016.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Prochazka KT, Melchardt T, Posch F,
Schlick K, Deutsch A, Beham-Schmid C, Weiss L, Gary T, Neureiter D,
Klieser E, et al: NCCN-IPI score-independent prognostic potential
of pretreatment uric acid levels for clinical outcome of diffuse
large B-cell lymphoma patients. Br J Cancer. 115:1264–1272.
2016.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Çelik S, Kaynar L, Güven ZT, Atasever
Duran K, Kontaş O, Keklik M and Ünal A: The effect of
danger-associated molecular patterns on survival in acute graft
versus host disease. Bone Marrow Transplant. 59:189–195.
2024.PubMed/NCBI View Article : Google Scholar
|