Introduction
Collision tumors refer to the occurrence of two
distinct tumors within the same organ or mass, each with separate
cellular lineages and genetic origins, but lacking a visible
transitional zone between them. This phenomenon is most commonly
observed in organs such as the liver, stomach, adrenal glands,
ovaries, lungs, kidneys and colon (1).
A collision tumor of the urinary bladder refers to a
very rare condition in which two distinct types of tumors occur
simultaneously within the same bladder. These tumors may have
different origins and growth patterns, and they may be benign or
malignant. The coexistence of two different tumor types in the same
location is what distinguishes a collision tumor from a composite
tumor, in which different cell types from the same tissues are
involved (2,3). The most common combination observed in
bladder cancer collisions is the concurrent presence of urothelial
carcinoma, which is the most common subtype of malignant bladder
tumors, and another type of tumor, such as adenocarcinoma, squamous
cell carcinoma (SCC) and other subtypes (4,5).
Urothelial carcinoma is the most common primary histological type
involving the urinary bladder, which comprises 90 to 95% of the
primary malignancies, while the other non-urothelial carcinomas,
e.g., adenocarcinoma, SCC, neuroendocrine carcinoma and lymphoma,
are uncommon and often diagnostically challenging (6,7). Both
adenocarcinoma and SCC account for 0.5-2% and 1-3% of the primary
bladder malignancies, respectively (8,9).
Oncogenic factors, such as p53, VEGF and EGFR, are known to play
critical roles in the signal transduction pathways of collision
tumors. Based on whether cancer cells can infiltrate the muscular
layer of the bladder, bladder cancer is divided into two subtypes,
namely non-muscle invasive bladder cancer (NMIBC) and muscular
invasive bladder cancer (MIBC). The transurethral resection of
bladder tumor (TURBT) is the primary treatment modality for NMIBC
(10).
The present study reports and discusses a very rare
case of a bladder collision tumor. The references have been
inspected for reliability, and the report has been written
according to the CaReL guidelines (11,12).
Case report
Patient information
A 43-year-old female patient with a history of
vesical stones was referred to the Rzgary Oncology Clinic at Rzgary
Teaching Hospital (Erbil, Iraq) after being diagnosed with bladder
carcinoma.
Clinical findings
The medical history of the patient included
controlled diabetes mellitus, hypertension and multiple sessions of
cystoscopic laser lithotripsy, as well as open vesical stone
extractions. She had previously undergone a cystoscopy-directed
TURBT to remove a mass from the urinary bladder, and a
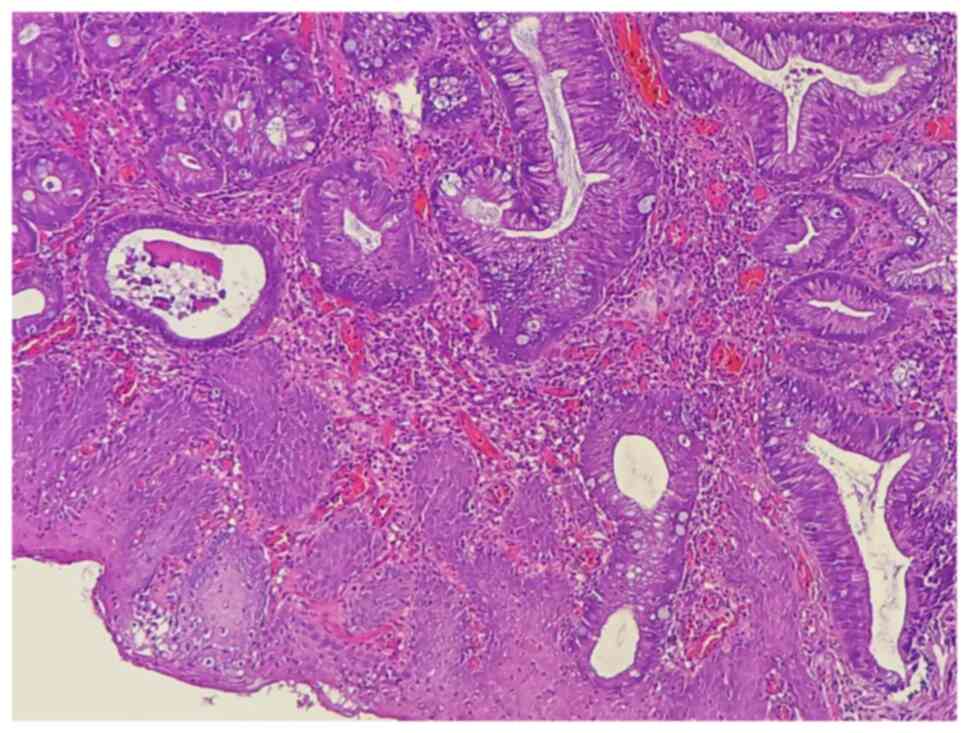
histopathological examination of the biopsy (performed at Rzgary
Oncology Laboratory), identified the tumor as a moderately
differentiated T2 adenocarcinoma (Fig.
1). Following this, the patient was referred to the Smart
Health Tower (Sulaymaniyah, Iraq) for further management.
Diagnostic assessment
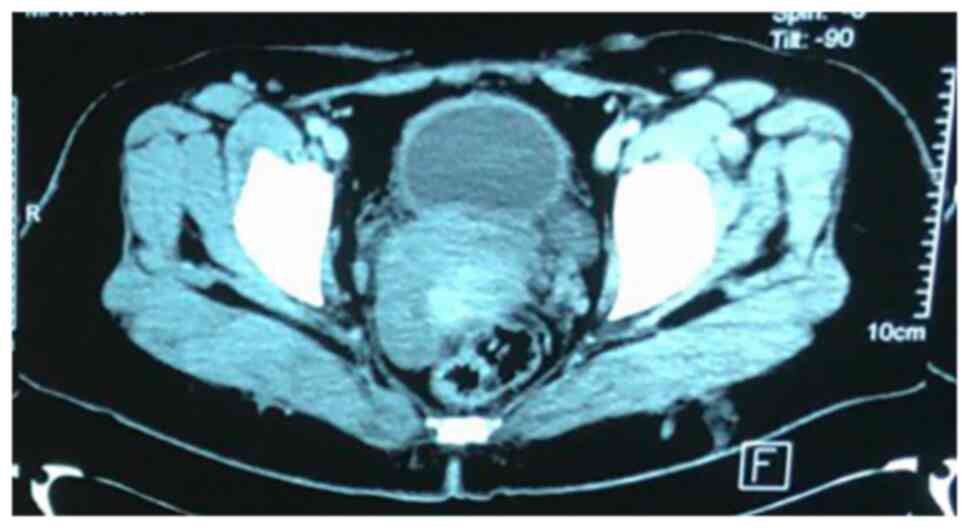
Initial diagnostic workup included an abdominal
ultrasound (U/S), which was followed by a contrast-enhanced
computed tomography (CT) scan as the previous surgery had been
based on the U/S alone. The CT scan was performed to provide a more
detailed assessment of the bladder wall and surrounding structures,
which revealed irregular, thickened bladder walls (8 mm), but no
gross mass lesion due to the previous TURBT procedure (Fig. 2). Given the history of adenocarcinoma
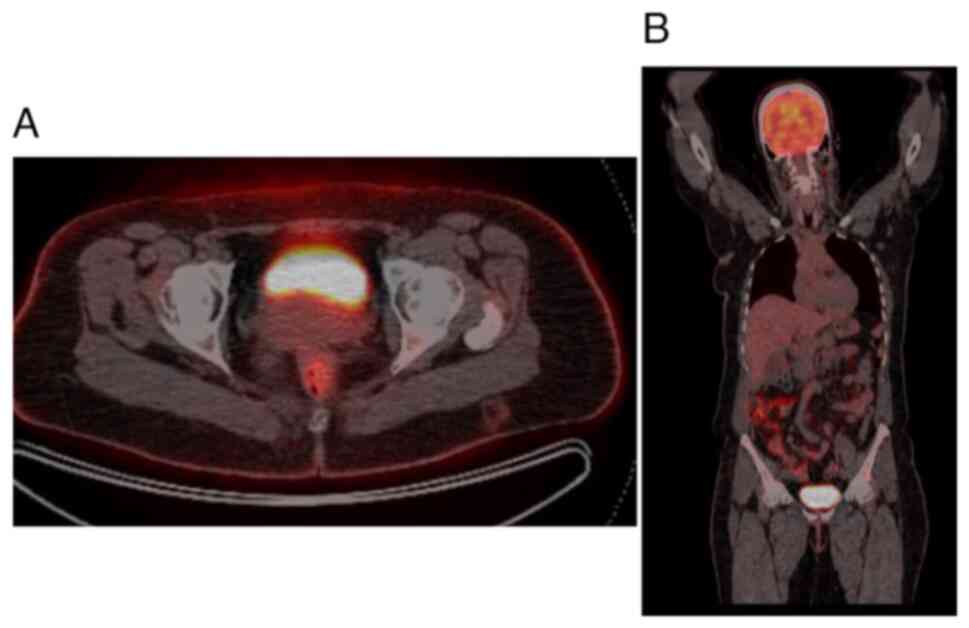
and to assess potential metastasis, a fluorodeoxyglucose positron
emission tomography (FDG-PET) was recommended by the
multidisciplinary team. The FDG-PET was crucial for detecting any
hypermetabolic nodular lesions suggestive of metastatic spread;
however, the results revealed no abnormal focal hypermetabolic
nodular lesions in the urinary bladder wall or elsewhere in the
body (Fig. 3). Based on the PET scan
findings and the initial TURBT biopsy, which revealed a T2-stage
adenocarcinoma, a radical cystectomy was advised for definitive
management.
Therapeutic intervention
The patient underwent radical cystectomy, during
which gross examination revealed a shrunken bladder (7x10 cm) with
a pale lesion at the trigone measuring approximately 25 mm. The
excised bladder specimen was fixed in 10% neutral-buffered formalin
at room temperature for 24 h before being processed using the
DiaPath Donatello automated tissue processor. The tissue underwent
dehydration in a graded alcohol series, clearing in xylene and
paraffin infiltration, followed by embedding in paraffin wax using
the Sakura Tissue-Tek embedding center. Paraffin-embedded sections,
5-µm thick, were obtained using the Sakura Accu-Cut SRM microtome,
floated in a 40-50˚C water bath, and mounted on glass slides. The
slides were then baked at 60-70˚C overnight before staining with
hematoxylin and eosin (H&E) (Bio Optica Co.) for 1-2 min at
room temperature using the DiaPath Giotto autostainer. The stained
slides were examined under a light microscope (Leica Microsystems
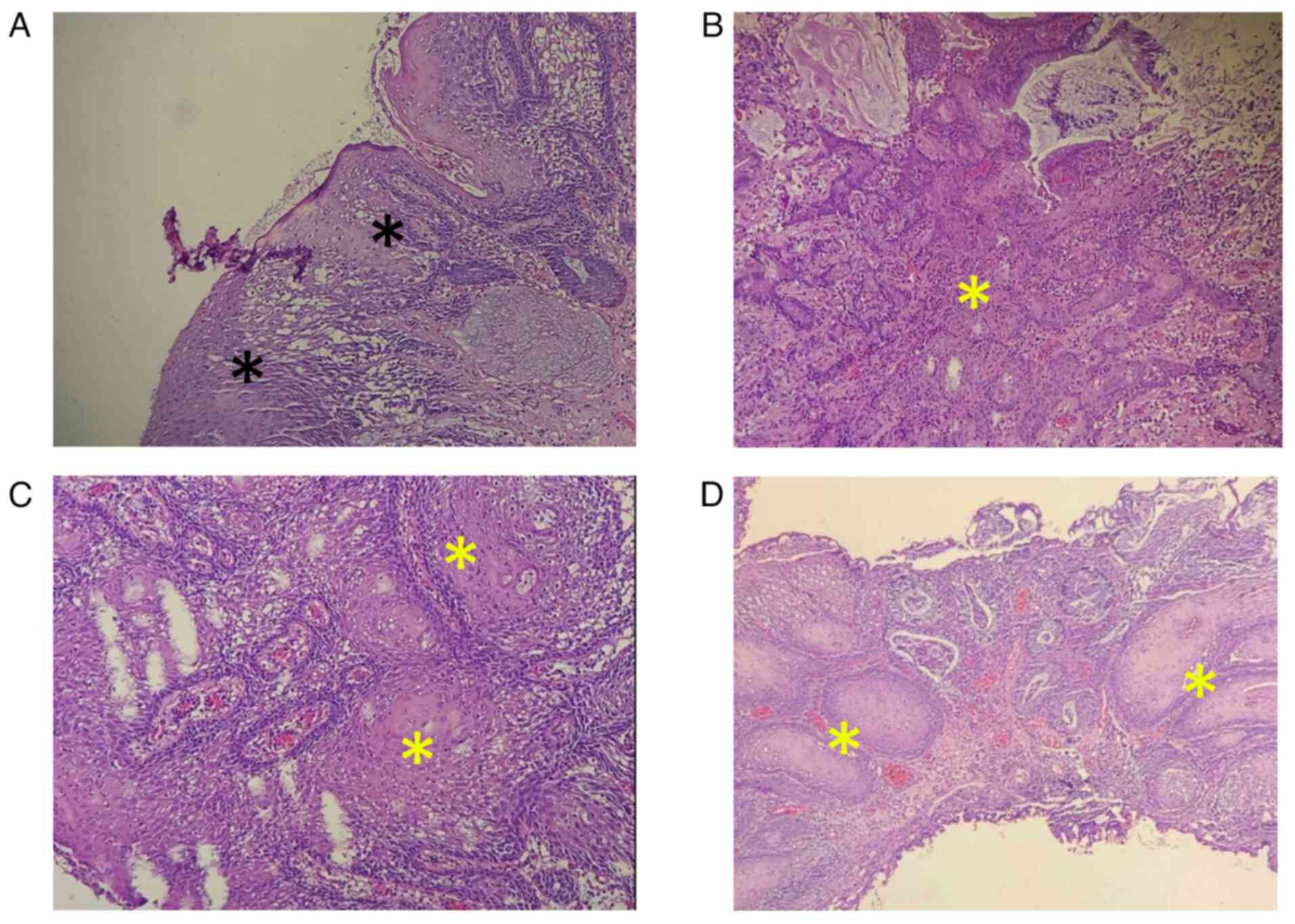
GmbH). A histopathological examination (HPE) revealed a marked
ulceration at the trigone with necrosis extending into the muscle
layer. Extensive squamous metaplasia throughout the bladder, SCC
in situ, and foci of superficially invasive SCC were
observed (Fig. 4). Glandular
metaplasia with dysplasia was also noted, extending into the distal
bladder, which exhibited significant inflammation and degenerative
changes. Additionally, two perivesical nodes were found to be free
of malignancy, and 18 lymph nodes were found after sectioning the
right and left pelvic nodes, and all were clear of malignancy. On
the TURBT, there was no residual adenocarcinoma. The final TNM
stage was poorly differentiated adenocarcinoma pT2 N0 M0 and
well-differentiated SCC T1 N0 M0 as a collision tumor. This
combination of adenocarcinoma and SCC within the same mass is a
hallmark of collision tumors, underscoring the multifocal and
independent origins of each tumor type.
Follow-up
The patient is under an extensive follow-up regimen,
which includes regular urine cytology, liver function tests, and
monitoring of creatinine and electrolytes. Imaging studies of the
chest, urinary tract, abdomen and pelvis are also performed
periodically. Additionally, annual vitamin B12 monitoring is
conducted. No recurrence was observed after 1 year of
follow-up.
Discussion
While the case presented herein is unique in its
specifics, the literature review highlighted several cases of
urinary bladder collision tumors in patients aged 50 to 74 years,
with most cases involving male patients. Common symptoms included
hematuria, sometimes accompanied by urinary urgency, dysuria, or
the presence of blood clots. Diagnostic imaging techniques, such as
CT scans, ultrasounds and MRIs, have been frequently employed to
identify the tumors and assess their extent. Surgical interventions
were the primary treatment methods, including TURBT and radical
cystectomy, often combined with pelvic lymph node dissection. In
some instances, additional treatments such as chemotherapy or
immunotherapy were utilized to manage the disease. Patient outcomes
varied; while some patients experienced recurrence or metastasis,
leading to further complications or mortality, others had
successful surgeries and remained free from recurrence for several
months to years following treatment. The majority of cases (60%)
were found to result in either recurrence or mortality, while 2
cases exhibited no recurrence following an average follow-up period
of 6.5 months (2,10,13-15)
(Table I). By contrast, the patient
in the present study remained free from recurrence during
follow-up. It is noteworthy that, although the cases reviewed
predominantly involve male patients, the present case pertains to a
female patient, thereby contributing to the diversity in the
clinical presentation of bladder collision tumors.
 | Table ISummary of several reported cases of
urinary bladder collision tumors in the literature. |
Table I
Summary of several reported cases of
urinary bladder collision tumors in the literature.
| | Imaging used for
diagnosis | | Tumor
characteristics | |
|---|
| Author(s) | Country, year of
publication | Age, years | Sex | Presentation | U/S | CT/MRI | Management | Location | Gross-section
size | HPE | Adjuvant
chemotherapy | Recurrence | (Refs.) |
|---|
| Gandhi et
al | India, 2017 | 73 | Male | Hematuria for15
days | Revealed a bladder
mass | Showed a bladder mass
measuring 5.6x2.4 cm with significant bilateral pelvic
lymphadenopathy | Radical
cystoprostatectomy with bilateral pelvic lymph node dissection and
resection of a sigmoid colon serosal deposit | Posterior wall and
dome of the bladder | 6x4.5x3 cm | Large cell
neuroendocrine carcinoma with adenocarcinoma component | Recommended but
refused by the patient | Patient died 4 months
post-surgery | (2) |
| Jiang et
al | China, 2023 | 64 | Male | Urinary urgency,
frequent urination, dysuria, and recurrent hematuria. | Not applicable | Irregular and
abnormal signal shadow in the bladder, measuring 8.4x6 cm, and
abnormal signal in the central gland of the prostate, Chemo-therapy
(gemcitabine + cisplatin) and immunotherapy (tislelizumab) | Multiple TURBT
procedure+ Radical cystectomy with pelvic lymph node resection and
cutaneous ureterostomy in May 2021 | Bladder and prostate
involvement | Up to 8.4x6 cm | Collision tumor with
small cell small cell carcinoma (SCC) and high-grade urothelial
carcinoma | Recommended but the
patient had difficulty adhering to the treatment schedule due to
economic reasons | Neck and mediastinum
lymph nodes metastases; patient died of COVID-19 | (10) |
| Brinton et
al | United States,
1970 | 50 | Male | Gross, total,
painless hematuria for 4 days | Not applicable | Not applicable | TURBT followed by
total cystectomy and ileal loop urinary diversion. | Superior to the right
ureteral orifice | 2 cm (polypoid
tumor) | Carcinosarcoma of the
bladder with muscle invasion | Not applicable | No recurrence;
patient alive and well 5 months postsurgery. | (13) |
| Chen et
al | China, 2022 | 74 | Male | Gross hematuria for 3
months worsening in the past week, occasionally accompanied by
blood clots. | Revealed a substan
tial mass in the bladder measuring 3.1x 2.4x2.1 cm with a rich
blood flow signal. Additionally, the prostate was enlarged with
intense light spots | CT scan: Indicated
focal thickening of the left posterior wall of the bladder with a
cauliflower-shaped soft-tissue density shadow protruding into the
bladder. On contrast-enhanced CT, the lesion was mildly to
moderately enhanced, suggestive of a space-occupying lesion
consistent with bladder cancer | TURBT followed by
total cystectomy | Left posterior wall
of the bladder, 1 cm lateral to the left ureteral opening | A volume of mass was
3.0x2.0x 2.0 cm | Collision tumor
between primary malignant melanoma and high-grade non-invasive
urothelial papillary carcinoma. | No chemotherapy,
immunotherapy, or gene therapy was administered. | Patient died of
systemic metastasis 31 months after surgery | (14) |
| Kitazawa et
al | Japan, 1985 | 67 | Male | Occasional small
amounts of gross hematuria and dysuria, with discharge of a
fingertip-sized mass. | Not applicable | Not applicable | TURBT, section by
section. | Right posterior wall
of the bladder | 2.1x1.8 cm; the
specimen weighed 4.0 grams. | Giant cell tumor
associated with transitional cell carcinoma. | Not applicable | No recurrence or
metastasis 8 months after TURBT. | (15) |
Collision tumors may exhibit diverse origins and
growth patterns, often involving two distinct tumor types that
originate independently within the same mass. This combination of
adenocarcinoma and SCC in the present study within the same mass is
a hallmark of collision tumors, underscoring the multifocal and
independent origins of each tumor type (2). In contrast to collision tumors,
composite tumors involve different cell types from the same
tissues. The most frequently observed combination in bladder
collision tumors, as in the present case, is the simultaneous
presence of urothelial carcinoma and another type of tumor, such as
adenocarcinoma, or SCC (3).
Adenocarcinoma of the bladder emerges from the glandular cells of
the bladder. Unlike the more prevalent transitional cell carcinoma
type, it is less common. It can be in different areas of the
bladder, such as the mucosal layer and deeper muscle layers
(16). Cigarette smoking increases
the risk of bladder cancer, as harmful chemicals in tobacco smoke
are absorbed into the bloodstream and excreted through the kidneys,
exposing the bladder lining to carcinogens. Bladder cancer is more
common in older adults, especially men. Occupational exposures to
chemicals such as aromatic amines and polycyclic aromatic
hydrocarbons, found in industries such as rubber, dye, and chemical
production also increase the risk. Prolonged exposure to chemicals
in textiles, paints, and plastics, along with chronic urinary
infections, can further increase this risk. Additionally, radiation
therapy, certain diabetes medications, and chronic Schistosoma
haematobium infection are linked to higher bladder cancer risk
(17,18). The starting point of the disease is a
genetic mutation, which can lead to uncontrolled cell growth and
division. These mutations can be inherited or occur spontaneously
during a person's lifetime; chronic inflammation and long-term
inflammation in an organ can create an environment that promotes
the growth of cancer cells (19).
The signs and symptoms of bladder collision tumors can vary. The
urine may appear pink, red, or dark brown, and the presence of
blood can be intermittent or consistent; also, there may be painful
urination, urinary urgency, and pelvic pain (4). In the present study, the patient was
diagnosed with bladder carcinoma and was referred to the Rzgary
Oncology Clinic.
The diagnosis of bladder carcinoma often involves
various tests, such as urine analysis, imaging studies, and
cystoscopy. A biopsy is the gold standard for the diagnosis
(12). Since this type of cancer is
mostly diagnosed in the advanced stages and is associated with high
recurrence rates, the understanding of gene expression regulation
is significant for early diagnosis and treatment. Epigenetic
modifications, which alter gene expression without changing the DNA
sequence, have been extensively studied as flexible and important
therapeutic targets for cancer treatments. Mutations in genes such
as FGFR3, PIK3CA, KDM6A and TP53 are common in bladder cancers and
disrupt normal gene regulation and cell growth, leading to
uncontrolled cell growth and tumor formation (20). The case described herein underwent a
CT scan with IV contrast followed by a PET scan. The HPE of the
specimen confirmed the diagnosis.
The management of bladder collision tumors includes
cancer staging and grading, the assessment of the overall health of
the patient and the consideration of the preferences of the
patient. A general outline of the management options for collision
bladder is TURBT, which is the standard initial treatment for
non-invasive or early-stage bladder cancer, radical cystectomy,
intravesical therapy and radiation therapy. Chemotherapy and
immunotherapies are used in the advanced stages. Some bladder
cancers with specific molecular features may be treated with
targeted therapies that aim to inhibit specific abnormal proteins
or pathways involved in cancer growth (21,22).
This case undergone radical cystectomy as far as it was a localized
early-stage disease.
Early detection and appropriate treatment are
crucial for better outcomes in bladder carcinoma. Regular check-ups
and prompt medical attention for concerning symptoms can improve
prognosis and increase the likelihood of successful treatment. As
with any type of cancer, a multidisciplinary approach involving
urologists, oncologists and other healthcare specialists is often
necessary to provide comprehensive care to individuals affected by
bladder carcinoma (5). Although it
is a rare disease, regular follow-up is necessary in cases of
bladder cancer (13-16,23).
The case in the present study was kept on an extensive and regular
follow-up schedule by conducting urine cytology, liver function
tests, serum creatinine and electrolytes.
In conclusion, collision tumor of the bladder is a
rare disease; extensive radiological study and careful examination
of the biopsy are necessary to diagnose the condition.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
SSO and SSF were the major contributors to the
conception of the study, as well as in the literature search for
related studies. HOA, FHK and BAA were involved in the literature
review, in the writing of the manuscript, and in the analysis and
interpretation of the patient's data. AMA, BOM, AAQ, SHK and JIH
were involved in the literature review, in the design of the study,
in revision of the manuscript and in the processing of the figures.
RMA was the pathologist who performed the histopathology diagnosis.
SHT was the radiologist who performed the assessed the case. SSO
and FHK confirm the authenticity of all the raw data. All authors
have read and approved the final manuscript.
Ethics approval and consent to
participate
The patient provided written informed consent to
participate and publish any related data in the present study.
Patient consent for publication
The patient provided written informed consent to
participate and publish any related data in the present study.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Abdullah AM, Qaradakhy AJ, Ali RM, Ali RM,
Mahmood YM, Omar SS, Nasralla HA, Muhialdeen AS, Saeed YA, Dhair HM
and Mohammed RO: Thyroid collision tumors: A systematic review.
Barw Medical J. 2:44–56. 2024.
|
|
2
|
Gandhi JS, Pasricha S and Gupta G:
Collision tumor of the urinary bladder comprising large cell
neuroendocrine carcinoma and adenocarcinoma. Asian J Oncol.
3:144–146. 2008.
|
|
3
|
Miyazaki J and Nishiyama H: Epidemiology
of urothelial carcinoma. Int J Urol. 24:730–734. 2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Radović N, Turner R and Bacalja J: Primary
‘pure’ large cell neuroendocrine carcinoma of the urinary bladder:
A case report and review of the literature. Clin Genitourin Cancer.
13:375–e377. 2015.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Rouanne M, Loriot Y, Lebret T and Soria
JC: Novel therapeutic targets in advanced urothelial carcinoma.
Crit Rev Oncol Hematol. 98:106–115. 2016.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Ploeg M, Aben KK, Hulsbergen-van de Kaa
CA, Schoenberg MP, Witjes JA and Kiemeney LA: Clinical epidemiology
of nonurothelial bladder cancer: Analysis of the Netherlands Cancer
Registry. J Urol. 183:915–920. 2010.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Tavora F and Epstein JI: Bladder cancer,
pathological classification and staging. BJU Int. 102:1216–1220.
2008.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Loriot Y, Necchi A, Park SH, Garcia-Donas
J, Huddart R, Burgess E, Fleming M, Rezazadeh A, Mellado B,
Varlamov S, et al: Erdafitinib in locally advanced or metastatic
urothelial carcinoma. N Engl J Med. 381:338–348. 2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Catto JW, Yates DR, Rehman I, Azzouzi AR,
Patterson J, Sibony M, Cussenot O and Hamdy FC: Behavior of
urothelial carcinoma with respect to anatomical location. J Urol.
177(1715)2007.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Jiang W, Pan C, Guo W, Xu Z, Ni Q and Ruan
Y: Pathologic collision of urinary bladder urothelial carcinoma
with small cell carcinoma: A case report. Diagnostic Pathology.
18(80)2023.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Abdullah HO, Abdalla BA, Kakamad FH, Ahmed
JO, Baba HO, Hassan MN, Bapir R, Rahim HM, Omar DA, Kakamad SH, et
al: Predatory Publishing Lists: A Review on the Ongoing Battle
Against Fraudulent Actions. Barw Med J. 2:26–30. 2024.
|
|
12
|
Prasad S, Nassar M, Azzam AY,
García-Muro-San José F, Jamee M, Sliman RKA, Evola G, Mustafa AM,
Abdullah HO, Abdalla BA, et al: CaReL guidelines: A consensus-based
guideline on case reports and literature review (CaReL). Barw Med
J. 2:13–19. 2024.
|
|
13
|
Brinton JA, Ito Y and Olsen BS:
Carcinosarcoma of the urinary bladder. A case report and review of
the literature. Cancer. 25:1183–1186. 1970.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Chen X, Ge H, Yang J, Huang B and Liu H:
Primary malignant melanoma of the bladder collides with high grade
non invasive urothelial papillary carcinoma: A case report. Oncol
Lett. 24:1–5. 2022.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Kitazawa M, Kobayashi H, Ohnishi Y, Kimura
K, Sakurai S and Sekine S: Giant cell tumor of the bladder
associated with transitional cell carcinoma. J Urol. 133:472–475.
1985.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Dadhania V, Czerniak B and Guo CC:
Adenocarcinoma of the urinary bladder. Am J Clin Exp Urol.
3(51)2015.PubMed/NCBI
|
|
17
|
Taguchi S, Akamatsu N, Nakagawa T, Gonoi
W, Kanatani A, Miyazaki H, Fujimura T, Fukuhara H, Kume H and Homma
Y: Sarcopenia evaluated using the skeletal muscle index is a
significant prognostic factor for metastatic urothelial carcinoma.
Clin Genitourin Cancer. 14:237–243. 2016.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Ku JH, Byun SS, Jeong H, Kwak C, Kim HH
and Lee SE: Lymphovascular invasion as a prognostic factor in the
upper urinary tract urothelial carcinoma: A systematic review and
meta-analysis. Eur J Cancer. 49:2665–2680. 2013.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Roy S and Parwani :
AVA8/arpa.2009-0713-RS adenocarcinoma of the urinary bladder. Arch
Pathol Lab Med. 135:1601–1605. 2011.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Lee KH and Song CG: Epigenetic regulation
in bladder cancer: Development of new prognostic targets and
therapeutic implications. Translational Cancer Res. 30
(Suppl):677–688. 2017.
|
|
21
|
Crabb SJ and Douglas J: The latest
treatment options for bladder cancer. Br Med Bull. 128:85–95.
2018.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Witjes JA: Bladder carcinoma in situ in
2003: State of the Art. Eur Urol. 45:142–146. 2004.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Mingomataj E, Krasniqi M, Dedushi K,
Sergeevich KA, Kust D and Qadir AA: Cancer Publications in one year
(2023): A cross-sectional study. Barw Med J. 2(3)2024.
|