Introduction
IgG4-related disease (IgG4-RD) is a rare, immune
mediated-fibroinflammatory condition that primarily affects
patients of middle to advanced age and is more common among males
than females (1). IgG4-RD was
initially described in 1951 as chronic autoimmune pancreatitis
(2,3). In 2003, upon the discovery of
extra-pancreatic lesions featuring IgG4-positive plasmacytic
infiltration in patients with autoimmune pancreatitis, IgG4-RD was
proposed as a novel clinicopathological entity (4). In 2019, IgG4-RD was formally defined by
a classification criteria (5).
IgG4-RD has been described in virtually every organ and is
characterized histologically by the infiltration of immunoglobulin
G4 (IgG4)-bearing lymphoplasmacytic cells (6). Respiratory manifestations, or
IgG4-related lung disease (IgG4-RLD), encompass various features,
such as the presence of solid nodules in the lung parenchyma, the
thickening of bronchovascular bundles and interlobular septa, and
rounded ground glass opacities (7).
The present study describes an uncommon case of IgG4-RLD with
endobronchial nodules, as well as systemic involvement.
Case report
An 84-year-old retired electrician with a previous
medical history of ischemic heart disease, hypertension and gout
was referred to the general medical outpatient clinic of Princess
Margaret Hospital, Hong Kong, SAR, China due to a 6-month history
of unquantified weight loss. He had no fever, night sweats or any
respiratory symptoms. A physical examination of his abdomen,
cardiovascular and respiratory systems did not reveal any notable
findings, apart from multiple enlarged submandibular lymph nodes.
The patient recalled a diagnosis of IgG4-RD more than a decade ago
with no treatment offered and claimed there was no marked
progression of submandibular swelling over the years.
The levels of tumor markers, including alpha fetal
protein (3 µg/l), prostate-specific antigen (<0.03 µg/l) and
carcinoembryonic antigen (1.9 µg/l) were withiun normal range. He
had an increased eosinophil count (1.9x109/l; reference
range, 0.0-0.5x109/l) and an erythrocyte sedimentation
rate of 119 mm/h (reference range, <32 mm/h). Autoimmune markers
including antinuclear, antineutrophil cytoplasmic, Sjögren
syndrome-related antigen A/Ro, Sjögren syndrome-related antigen
B/La, double-stranded DNA, ribonucleoprotein and Smith antibodies
were all negative. His serum IgG4 count was increased (2,269 mg/dl;
reference range, 9-146 mg/dl). A positron emission
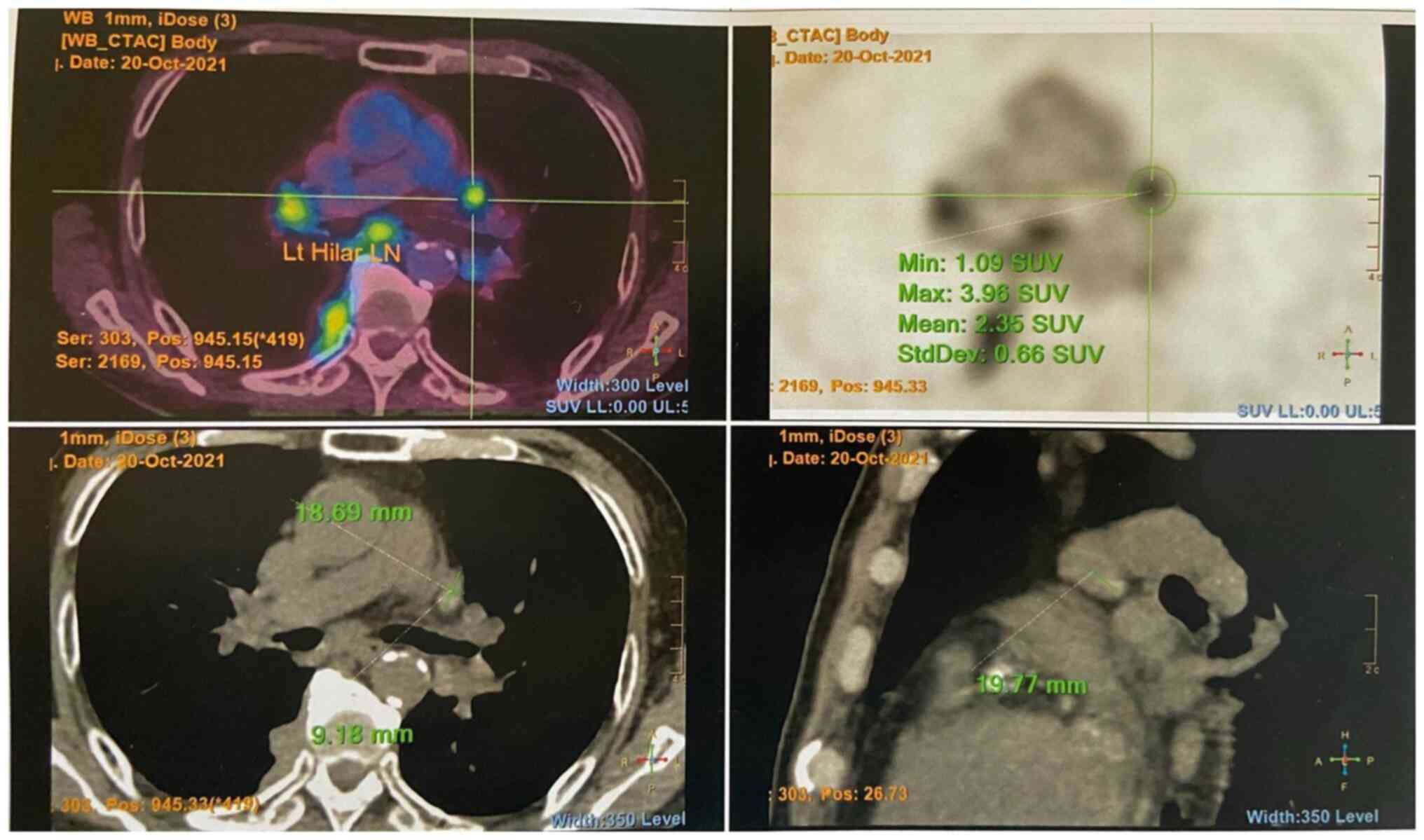
tomography-computed tomography scan revealed multiple
fludeoxyglucose F18 (FDG)-avid enlarged mediastinal, hilar,
(Fig. 1) and submandibular lymph
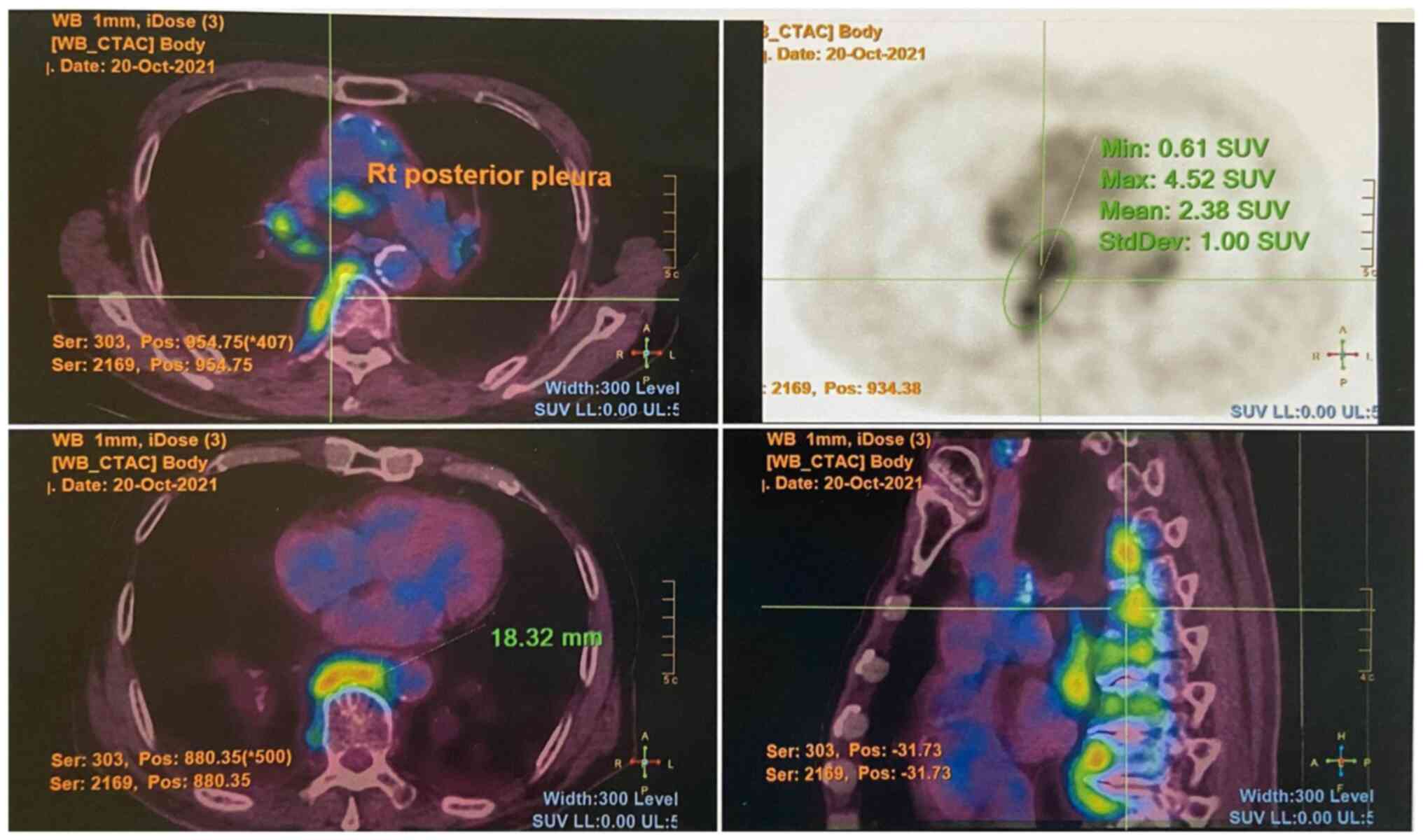
nodes, as well as an FDG-avid right parotid gland nodule. An
FDG-avid nodular thickening of the right posterior pleura was also
noted at the T9/10 level (Fig.
2).
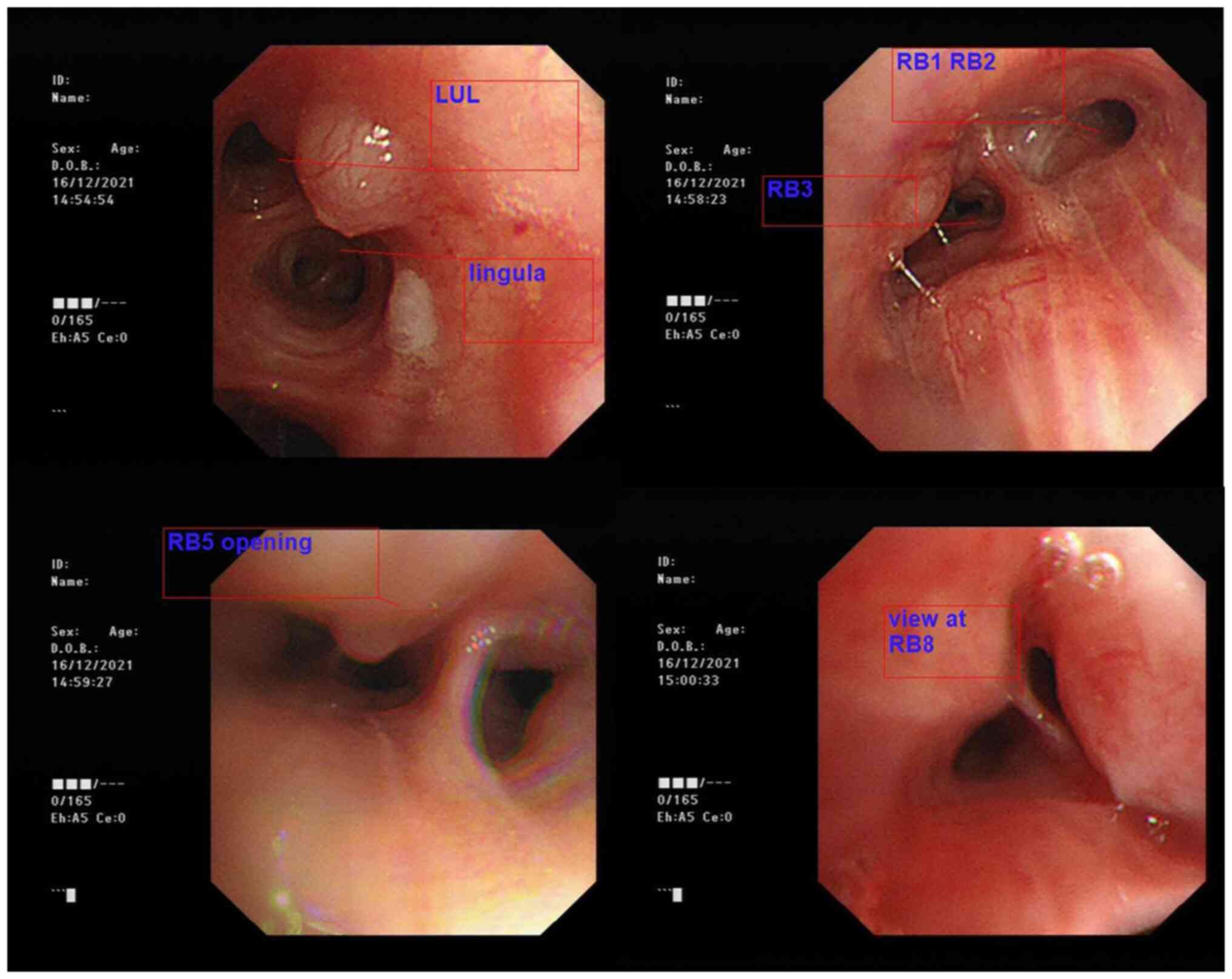
Flexible bronchoscopy incidentally identified
diffuse submucosal nodular swelling in both bronchi, featuring a
smooth surface continuous with the respiratory tract mucosa
(Fig. 3). Multiple endobronchial
biopsies of nodules were taken at the right upper lobe anterior
bronchus (RB3) and right lower lobe anterior basal bronchus (RB8).
An endobronchial ultrasound (EBUS) identified enlarged station 4R
and station 10 lymph nodes, with transbronchial needle aspiration
(TBNA) performed over the station 4R lymph node. The cytology of
the EBUS-TBNA sample only revealed tiny fragments of crushed
lymphoid tissue with no granuloma or malignant cells. The
histological analysis of multiple endobronchial biopsies revealed
benign bronchial mucosa with stromal fibrosis and containing patchy
inflammatory infiltration with marked crushing artefact (data not
shown).
In view of the diagnostic uncertainty, a left-sided
submandibular sialoadenectomy with lymph node excision was
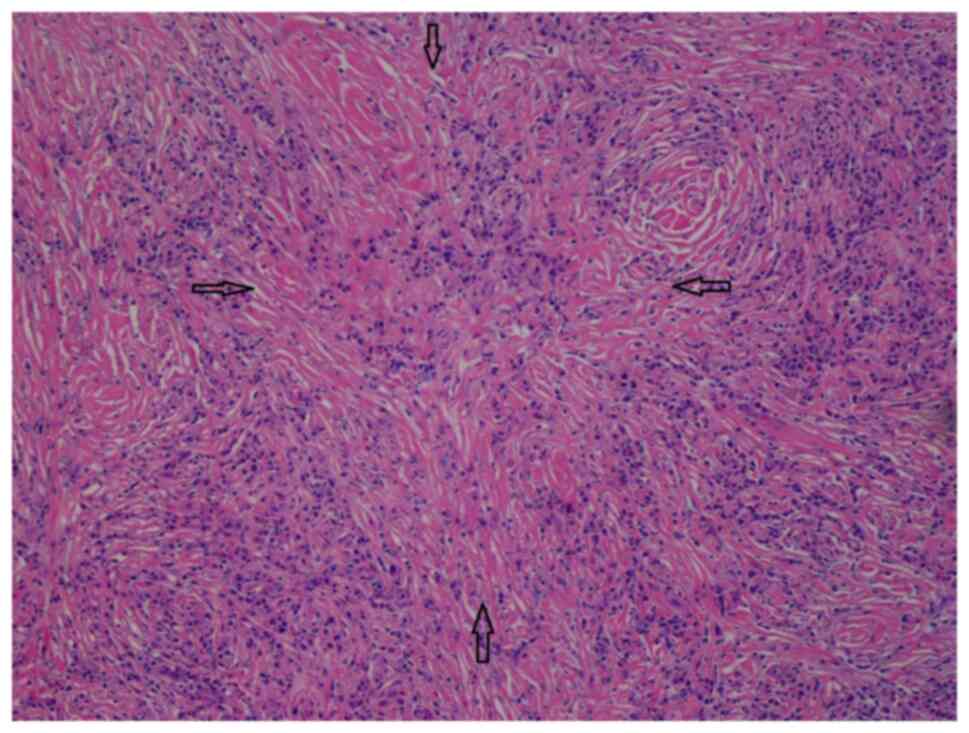
performed by the surgeons. A histological examination of the left
salivary glandular tissue (hematoxylin and eosin staining;
performed by the Histopathology Laboratory of Yan Chai Hospital,
Hong Kong, SAR, China; x100 magnification) revealed evidence of
chronic sclerosing sialadenitis (Fig.
4). The salivary gland lobules were mostly replaced by
storiform fibrosis, reactive lymphoid hyperplasia and
lymphoplasmacytic infiltration. Plasma cells were abundant.
Obliterative phlebitis was not observed. There was no evidence of
malignancy. Immunohistochemistry performed by the Histopathology
Laboratory of Yan Chai Hospital revealed a high number of IgG4
plasma cells >100/HPF in the most affected area and the IgG4:IgG
plasma cell ratio was >40% (the images for this were not
available). All these features were in-keeping with IgG4-RD.
Similar features were also observed in the sampled submandibular
lymph node.
After the diagnosis of IgG4-RD was made, the patient
was treated with prednisolone 10 mg daily (body weight, 63.5 kg)
with a gradual tapering regimen. There was a marked reduction in
submandibular swelling and his weight remained static. At the
latest follow-up, the serum IgG4 level decreased from 2,269 to 314
mg/dl after the 10 weeks of treatment.
Discussion
Various international diagnostic criteria for
IgG4-RD are available, including the 2019 American College of
Rheumatology/European League Against Rheumatism (ACR/EULAR)
Classification Criteria for IgG4-Related Disease, and the 2020
Japan College of Rheumatology's revised comprehensive diagnostic
(RCD) criteria for IgG4-RD (5,8). The RCD
criteria is the most updated criteria, requiring fulfilment of
clinical and radiological, serological and pathological features
(Table I). The case described herein
fulfils the RCD criteria of definite IgG4-RD.
 | Table IThe 2020 Revised Comprehensive
Diagnostic (RCD) criteria (8) for
IgG4-RD. |
Table I
The 2020 Revised Comprehensive
Diagnostic (RCD) criteria (8) for
IgG4-RD.
| 1. Clinical and
radiological features |
|
- One or
more organs show diffuse or localized swelling or a mass or nodule
characteristic of IgG4-RD. In single-organ involvement, lymph node
swelling is omitted. |
| 2. Serological
diagnosis |
|
- Serum IgG4
levels >135 mg/dl. |
| 3. Pathological
diagnosis |
|
- Positivity
for two of the following three criteria: |
|
-
Dense lymphocyte and plasma cell infiltration with fibrosis. |
|
-
Ratio of IgG4-positive plasma cells /IgG-positive cells greater
than 40% and the number of IgG4-positive plasma cells greater than
10 per high powered field |
|
-
Typical tissue fibrosis, particularly storiform fibrosis, or
obliterative phlebitis |
| Diagnosis: |
|
Definite: 1
+ 2 + 3 |
|
Probable: 1
+ 3 |
|
Possible: 1
+ 2 |
The diagnostic criteria used in IgG4-RLD is the same
as IgG4-RD. Due to its rarity, the exact prevalence of IgG4-RLD is
not known; however, studies have reported lung involvement in 2.4
to 27.1% of IgG4-RD cases (9).
Patients with IgG4-RLD typically exhibit minimal respiratory
symptoms. Its diagnosis is often incidental, discovered during
investigations for extrathoracic lesions or as an unexpected
finding of abnormal lung shadows (10). Characteristic features of IgG4-RLD on
computed tomography scans include hilar lymphadenopathies and the
presence of soft tissue masses in the paravertebral region
(11). Histopathological findings of
IgG4-RD are dense lymphoplasmacytic infiltrate, storiform fibrosis,
obliterative phlebitis, and increased numbers of IgG4-positive
plasma cells or an increased IgG4:IgG ratio in tissue (6).
Numerous cases of systemic IgG4-RD associated with
tracheobronchial edema, or capillary dilatation have been reported,
in addition to IgG4-RLD cases featuring isolated tracheobronchial
involvement, manifesting as mass-like lesions (7,12,13).
IgG4-RLD presents as multiple endobronchial nodules; however, it is
extremely rare. To the best of our knowledge, only two previous
articles to date have reported IgG4-RLD with multiple endobronchial
nodules (7,14). The first case reported by Wang et
al (7) involved a 52-year-old
male patient who presented with wheezing accompanied by jelly-like
sputum. His spirometry results were normal, and a computed
tomography scan of the thorax revealed left lower lung infiltrate
without obvious bronchial nodules. A bronchoscopy revealed multiple
white nodular protuberances in the trachea and bronchus, with
mucosal edema and hyperemia. He was diagnosed with IgG4-RLD
histologically through standard forceps biopsy and was treated with
prednisolone and azathioprine. There was no evidence of involvement
of other organs due to IgG4-RD (7).
The second case reported by Torii et al (14) involved a 74-year-old female patient
with known IgG4-RD affecting the parotid gland and eyelids. She had
previously been treated conservatively, with spontaneous resolution
of her symptoms. After 3 years, she presented with a recurrence of
swelling in the parotid gland and eyelids, which this time, was
accompanied by a cough, an obstructive pattern on spirometry, and a
computed tomography scan of the thorax revealed extensive multiple
nodules in the trachea and bronchi, without any masses or
ground-glass nodules in the lung parenchyma. A bronchoscopy
revealed multiple nodules with a smooth surface, continuous with
the respiratory tract mucosa. A diagnosis was made by an
endobronchial cryobiopsy and she was treated with prednisolone
(14). Both these aforementioned
cases had a resolution of endobronchial nodules demonstrated by
repeat bronchoscopies. In contrast to the two cases mentioned
above, the patient in the present study had no respiratory
symptoms. Spirometry was not performed as a result, and his
computed tomography scan of the thorax did not reveal obvious
bronchial nodules. Similar to the case reported by Torii et
al (14), the patient in the
present study exhibited systemic involvement of IgG4-RD with
endobronchial nodules that had a smooth surface continuous with the
respiratory tract mucosa.
The limitations of the present study include the
lack of a follow-up bronchoscopy to prove the resolution of
endobronchial nodules following the use of steroids, and no
pathological confirmation of IgG4 plasma cells in the endobronchial
nodules. However, it was deemed that there were no alternative
explanations of the endobronchial nodule.
A histological analysis is vital in diagnosing
IgG4-RLD, as the serum IgG4 level may be raised in several other
conditions, including multicentric Castleman's disease, repeated
infections, autoimmune diseases, cancer, primary immunodeficiencies
and systemic vasculitis (15,16). In
the case described herein, biopsies of endobronchial nodules had
crushing artefacts and were not diagnostic of IgG4-RLD. EBUS-TBNA
also failed to reveal specific features. The diagnosis was only
made following a histological examination of the left salivary
glandular tissue which revealed characteristic IgG4-RD changes. A
previous study demonstrated that histological findings from
transbronchial lung biopsies supported the diagnosis of IgG4-RLD in
47% of the cases only (17). Apart
from the obvious selection of surgical lung biopsy when diagnostic
uncertainty is met, transbronchial cryobiopsies have been reported
to successfully diagnose IgG4-RLD (12,14).
The treatment for IgG4-RLD is the same as that for
IgG4-RD. Glucocorticoids prove to be a potent remedy for IgG4-RD.
However, instances of disease resurgence often occur either during
or after the dose reduction of glucocorticoids, and these drugs
rarely result in sustained periods of remission without further
treatment (18). Rituximab, a
monoclonal anti-CD20 antibody, was shown to be effective as both
induction therapy and treatment of relapses in IgG4-RD in a
retrospective nationwide study in France (19). Inebilizumab, a monoclonal anti-CD19
antibody, demonstrated reduction in the risk of IgG4-RD flare up
and increased the likelihood of complete remission without flares
at 1 year (20). For the patient in
the present study, long-term follow-up is necessary to monitor for
disease recurrence following the discontinuation of prednisolone.
Biological agents should be considered if remission is not
achieved.
In conclusion, the present study reported a rare
presentation of IgG4-RLD with multiple endobronchial nodules and
systemic involvements, found incidentally during flexible
bronchoscopy. A histopathological examination of the endobronchial
biopsy was inconclusive and diagnosis was made after surgically
sampling the left salivary gland and submandibular lymph nodes.
This case serves the purpose of reminding physicians to consider
IgG4-RLD as a differential diagnosis when endobronchial nodules are
observed with or without systemic disease, and appropriate sampling
methods should be selected to increase its diagnostic yield.
Acknowledgements
The authors would like to express their appreciation
to Dr Ka Choi Yuen, of the Pathology Department of Yan Chai
Hospital, Hong Kong for providing and interpreting the
histopathology slides.
Funding
Funding: No funding was received
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
CCDL substantially contributed to the acquisition,
analysis and interpretation of the data and contributed to the
drafting of the manuscript. MYH, YHC and YCY analyzed the patient
data and advised on the patient's treatment. CCDL, MYH, YHC and YCY
confirm the authenticity of all the raw data, and agree to be
accountable for all aspects of the work, so that any questions
relating to research integrity or scientific accuracy in any part
of the study are appropriately investigated and resolved. All
authors have read and approved the final version of the
manuscript.
Ethics approval and consent to
participate
The patient provided written informed consent to
participate in the present study.
Patient consent for publication
The patient provided written informed consent for
the publication of any data and/or accompanying images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Uchida K, Masamune A, Shimosegawa T and
Okazaki K: Prevalence of IgG4-related disease in Japan based on
nationwide survey in 2009. Int J Rheumatol.
2012(358371)2012.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Sato T, Minegishi K, Okabe N, Osuga F,
Sudo K, Sogabe M, Endo S and Tsubochi H: Bronchial obstruction in
immunoglobulin G4-related disease. Ann Thorac Surg Short Rep,
2024.
|
|
3
|
Sarles H, Sarles JC, Muratore R and Guien
C: Chronic inflammatory sclerosis of the pancreas-an autonomous
pancreatic disease? Am J Dig Dis. 6:688–698. 1961.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Kamisawa T, Funata N, Hayashi Y, Eishi Y,
Koike M, Tsuruta K, Okamoto A, Egawa N and Nakajima H: A new
clinicopathological entity of IgG4-related autoimmune disease. J
Gastroenterol. 38:982–984. 2003.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Wallace ZS, Naden RP, Chari S, Choi H,
Della-Torre E, Dicaire JF, Hart PA, Inoue D, Kawano M, Khosroshahi
A, et al: The 2019 American college of rheumatology/European league
against rheumatism classification criteria for IgG4-related
disease. Arthritis Rheumatol. 72:7–19. 2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Stone JH, Khosroshahi A, Deshpande V, Chan
JKC, Heathcote JG, Aalberse R, Azumi A, Bloch DB, Brugge WR,
Carruthers MN, et al: Recommendations for the nomenclature of
IgG4-related disease and its individual organ system
manifestations. Arthritis Rheum. 64:3061–3067. 2012.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Wang X, Wan J, Zhao L, Da J, Cao B and
Zhai Z: IgG4-related disease with tracheobronchial miliary nodules
and asthma: A case report and review of the literature. BMC Pulm
Med. 19(191)2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Umehara H, Okazaki K, Kawa S, Takahashi H,
Goto H, Matsui S, Ishizaka N, Akamizu T, Sato Y, Kawano M, et al:
The 2020 revised comprehensive diagnostic (RCD) criteria for
IgG4-RD. Mod Rheumatol. 31:529–533. 2021.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Li PH, Ko K, Ho CT, Lau LL, Tsang RK,
Cheung T, Leung WK and Lau CS: Immunoglobulin G4-related disease in
Hong Kong: Clinical features, treatment practices, and its
association with multisystem disease. Hong Kong Med J. 23:446–453.
2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Matsui S: IgG4-related respiratory
disease. Mod Rheumatol. 29:251–256. 2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Matsui S, Hebisawa A, Sakai F, Yamamoto H,
Terasaki Y, Kurihara Y, Waseda Y, Kawamura T, Miyashita T, Inoue H,
et al: Immunoglobulin G4-related lung disease: Clinicoradiological
and pathological features. Respirology. 18:480–487. 2013.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Yamakawa H, Takemura T, Tsumiyama E, Sato
S, Akasaka K, Amano M and Matsushima H: IgG4-related bronchial
gland inflammation proved by transbronchial cryobiopsy. Am J Respir
Crit Care Med. 201:1554–1556. 2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Shrestha B, Sekiguchi H, Colby TV,
Graziano P, Aubry MC, Smyrk TC, Feldman AL, Cornell LD, Ryu JH,
Chari ST, et al: Distinctive pulmonary histopathology with
increased IgG4-positive plasma cells in patients with autoimmune
pancreatitis: report of 6 and 12 cases with similar histopathology.
Am J Surg Pathol. 33:1450–1462. 2009.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Torii A, Oshima K, Iwakoshi A and Oki M: A
case with multiple nodules and mucosal oedema of the trachea and
both bronchi induced by IgG4-related disease. BMC Pulm Med.
24(115)2024.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Otani K, Inoue D, Fujikura K, Komori T,
Abe-Suzuki S, Tajiri T, Itoh T and Zen Y: Idiopathic multicentric
Castleman's disease: A clinicopathologic study in comparison with
IgG4-related disease. Oncotarget. 9:6691–6706. 2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Ebbo M, Grados A, Bernit E, Vély F,
Boucraut J, Harlé JR, Daniel L and Schleinitz N: Pathologies
associated with serum IgG4 elevation. Int J Rheumatol. 2012:1–6.
2012.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Otani K, Inoue D, Itoh T and Zen Y:
Transbronchial lung biopsy for the diagnosis of IgG4-related lung
disease. Histopathology. 73:49–58. 2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Tanaka Y and Stone JH: Perspectives on
current and emerging therapies for immunoglobulin G4-related
disease. Mod Rheumatol. 33:229–236. 2023.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Ebbo M, Grados A, Samson M, Groh M,
Loundou A, Rigolet A, Terrier B, Guillaud C, Carra-Dallière C,
Renou F, et al: Long-term efficacy and safety of rituximab in
IgG4-related disease: Data from a French nationwide study of
thirty-three patients. PLoS One. 12(e0183844)2017.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Stone JH, Khosroshahi A, Zhang W, Della
Torre E, Okazaki K, Tanaka Y, Löhr JM, Schleinitz N, Dong L,
Umehara H, et al: Inebilizumab for treatment of IgG4-related
disease. N Engl J Med: Nov 14, 2024 (Epub ahead of print).
|