Introduction
Renal cell carcinoma (RCC) is a common malignant
tumor of the urinary system. One out of nearly a million
individuals succumb to the disease each year worldwide and the
incidence is on the increase (1).
RCC patients frequently present with subclinical disease and 20–30%
of patients are admitted (2).
However, traditional radical nephrectomy in RCC in the early stage
has a positive effect, which is not the case in advanced stage and
metastatic RCC. Simultaneously, RCC is not sensitive to
radiotherapy and chemotherapy (3).
Since RCC is a highly immunogenic tumor, advances in molecular and
immune biology allow for the potential use of tumor vaccines as
immune therapy (4). G250/MN/CA
IX (MN antigen receptor/carbonic anhydrase-9) is one of the
tumor markers that possess favorable tumor specificity (5). The specific expression of
G250/MN/CA IX in RCC renders it a key target for cancer
diagnosis and treatment. In this study, a eukaryotic expression
vector, containing the G250/MN/CA IX and human
granulocyte-macrophage colony stimulating factor
(hGM-CSF) genes, was constructed and transfected into human
embryonic kidney 293 (HEK 293) cells. The fusion protein expression
and immunoreactivity were then detected to establish anti-renal
cell carcinoma vaccines for studies based on the G250
gene.
Materials and methods
Materials
E. coli Top 10, vector pVAX1,
recombinant plasmid pORF-hGM-CSF and pcDNA3.0-G250
were obtained from the Department of Microbiology and Immunology,
Medical College, Jinan University, Guangzhou, China. Ex-Taq
DNA polymerase, T4 DNA ligase, restriction enzymes
XbaI, HindIII and KpnI, 1 kb DNA marker, 100
bp DNA marker, plasmid mini and gel extraction kits were from
Takara Bio Inc., Japan. The target gene sequencing analysis was
from Shanghai Sangon Biological Engineering Technology and Services
Company, China. Lipofectamine-2000 and Opti-MEM were from
Invitrogen Corporation (Carlsbad, CA, USA), and the
immunohistochemical kit was from Zhongshan Company, China. The
ELISA kit was purchased from R&D Inc., USA, and the G250
antibody was from Abcam Inc., USA.
Construction and identification of
recombinant plasmids
Based on the CDS sequence of the G250 gene in
Gene Bank (NCBI: BC014950), primer 5.0 was used to design the
primers 5′-GCAAGCTTTTCCAATGCACGTACAG-3′ (forward) and
5′-TCGGGTACCGGCTCCAGTCTC-3′ (reverse) with the appropriate
restriction endonuclease sites and omission of the termination
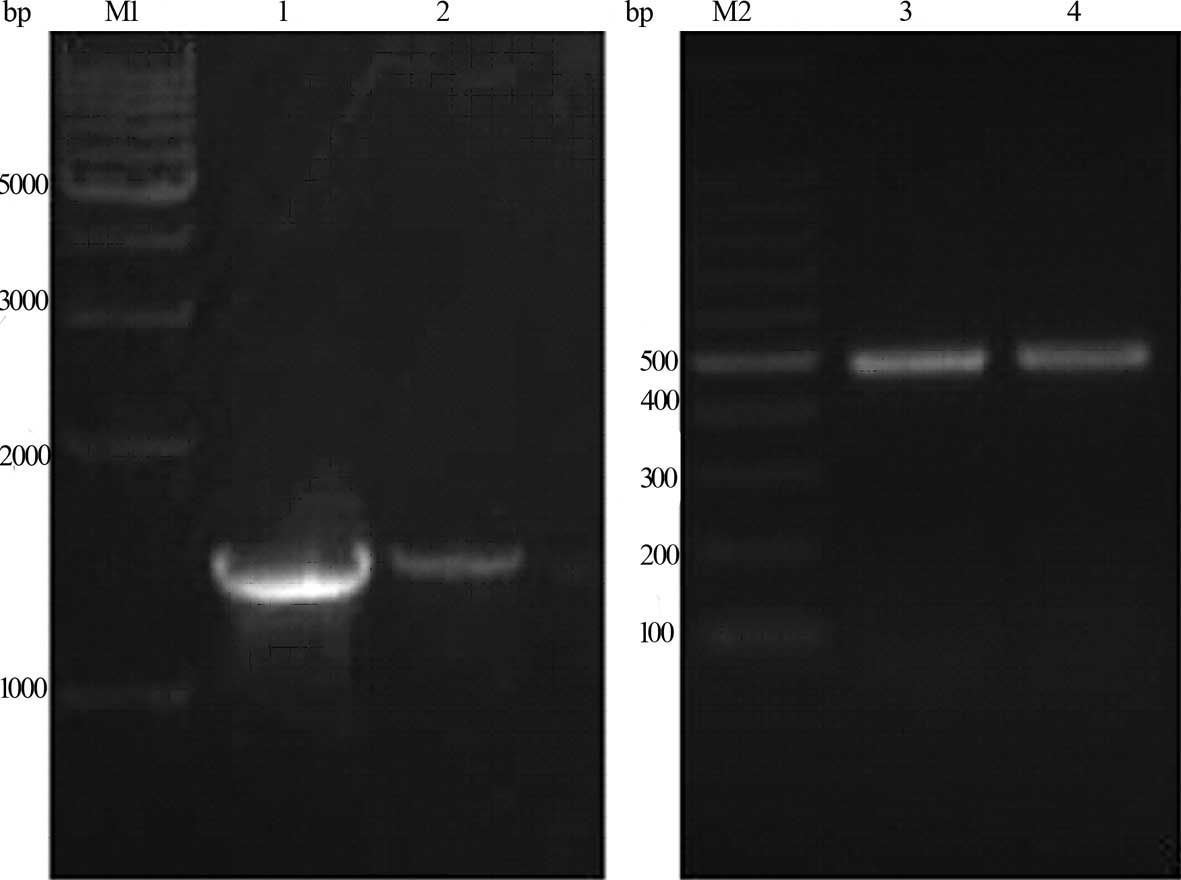
codon which was used to amplify G250 (Fig. 1). The PCR fragments and plasmid
pVAX1 were digested by KpnI and HindIII. The
cleaved products were ligated using T4 DNA ligase at 16°C
overnight. The ligated products were transformed into the competent
E. coli Top 10, and antibiotic selection and the restriction
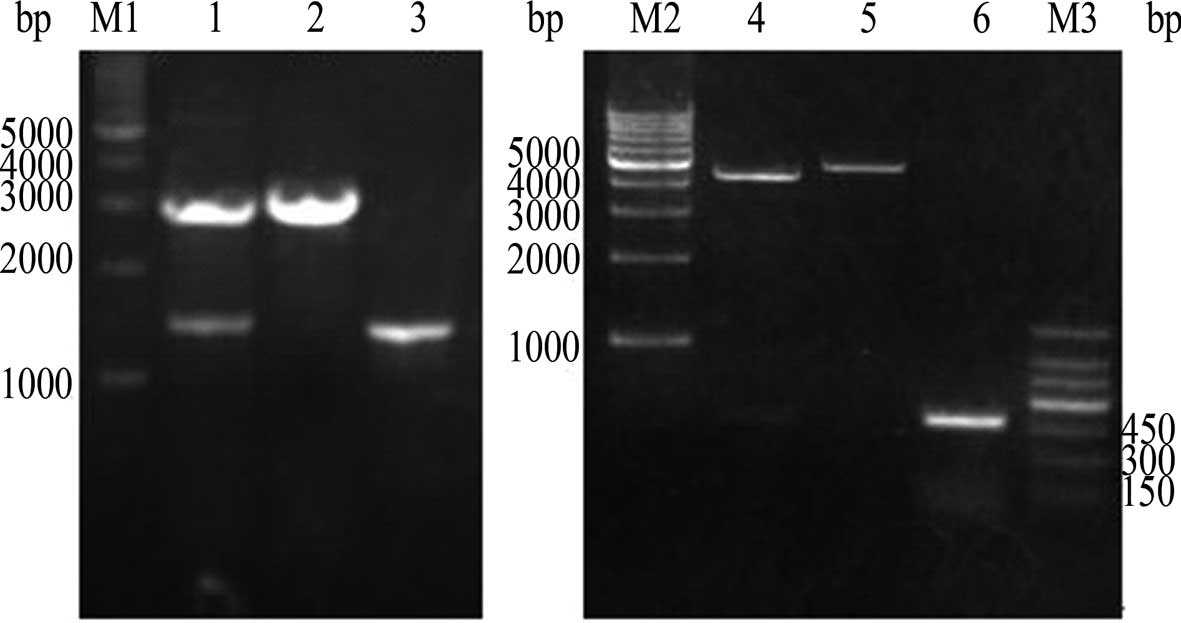
endonuclease assay (Fig. 2) were
used to screen and identify positive clones. DNA sequencing
analysis was performed using Sanger dideoxy chain termination.
The coding sequences of the hGM-CSF fragments
were synthesized by PCR from pORF-hGM-CSF using specific
primers: 5′-TATGGTACCGGATCAGGAGGTTCTATGTGG CTGCAGAGCCT-3′ (forward)
and 5′-GGGTCTAGATATCA TGTCGAGCTAGCGAATTCACT-3′ (reverse), which
were cloned into the KpnI and XbaI sites of
pVAX1-G250 using standard cloning techniques (Fig. 1). The recombinant plasmids were
purified and double digested with KpnI and XbaI
(Fig. 2). The procedure involving
the sequence analysis of the recombinant plasmids was terminated by
the Shanghai Bio-Engineering Company and recombinant plasmid
pVAX1-G250-GM was successfully constructed.
Cell transfection
HEK 293 cells were digested with 0.25% trypsin and
diluted to 1–4×105 cells/ml. The cells were plated in
6-well plates with 2 ml medium per well. When the cells achieved
60–70% confluence, 4 μg purified plasmid was transfected to the
prepared cells using 8 μl lipofectamine-2000 reagent. After 48 h,
the living cells were examined directly and photographed under an
inverted fluorescence microscope.
Immunocytochemistry staining
Non-transfected cells were regarded as the blank
comparison and pVAX1-transfected cells as the negative
comparison. Immunocytochemistry staining was performed according to
the manufacturer's instructions and mouse anti-G250 antibody
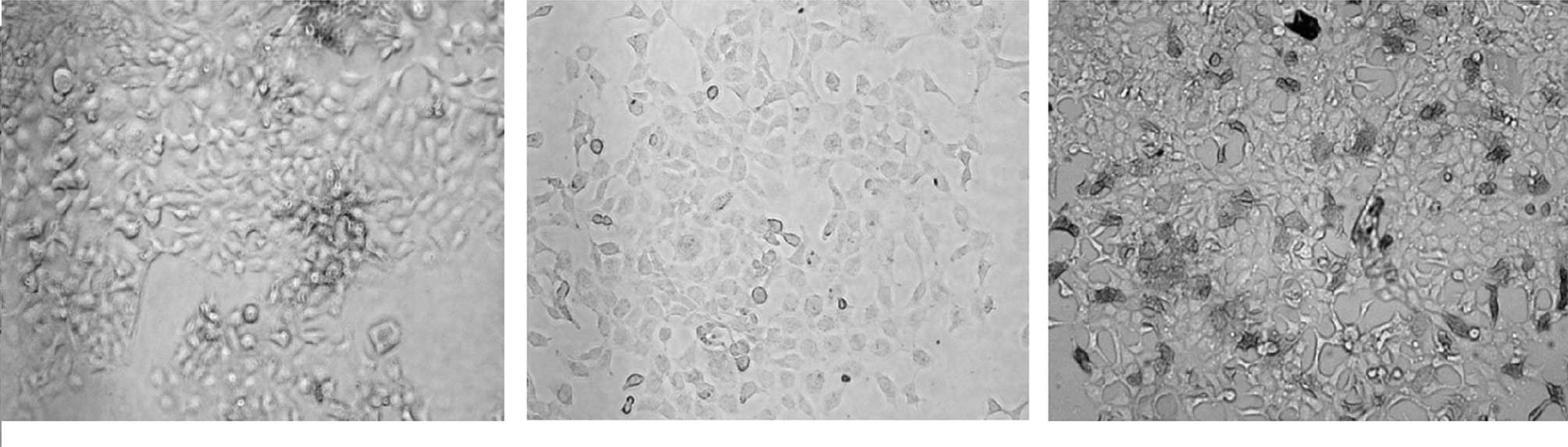
was used as the primary antibody (Fig.
3).
Atomic force microscopy
After being dried, samples were immunoblotted,
visualized using DAB chromogen and scanned by atomic force
microscopy (AFM) (AutoProbe CP Research, ThermoMicroscopes Inc.,
USA). The samples were placed in XY scanning stage, and then the
monitor was used to locate the scanning area of the sample and
scanning image. The test was conducted using 100 μm scanners, UL20B
silicon probe, and a force constant of 2.8 N/m. Equipment
configuration software (ThermoMicroscopes pro-scan image processing
software version 2.1) was used for image data acquisition and
processing. Images were smoothed to eliminate the scan direction
and ensure a low level of background noise (Fig. 4).
ELISA
Double-antibody sandwich ELISA was used to detect
the hGM-CSF protein level. The results were shown as the χ̄ ± s and
the significant level of difference between the values was analyzed
using SPSS 13.0 software. P<0.05 was considered to be
statistically significant (Table I,
Fig. 5).
 | Table IExpression value of hGM-CSF protein by
ELISA. |
Table I
Expression value of hGM-CSF protein by
ELISA.
| Group | 24-h hGM-CSF
(pg/ml) | 48-h hGM-CSF
(pg/ml) |
|---|
| pVAX1 | 53.97±0.53 | 54.10±0.79 |
|
pVAX1CAIX-hGM | 482.47±5.86a | 513.36±4.45a |
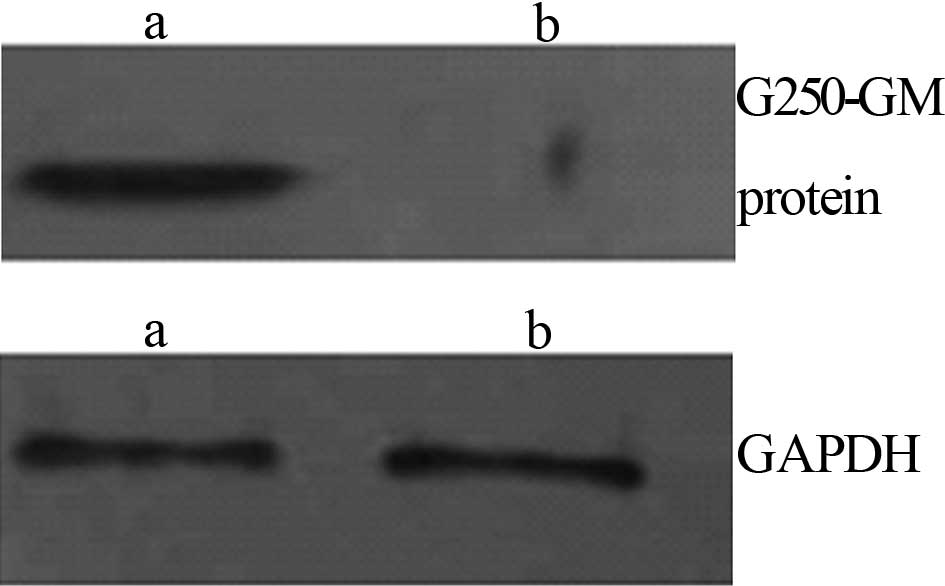
Western blotting
A Western blot analysis of the fusion proteins was
performed according to the standard method. The purified protein
was separated on 120 g/l SDS-PAGE and transferred to a
nitrocellulose membrane. Anti-G250 antibody at 1:1,000
dilution was used as the primary antibody to detect the G250
protein. The blots were developed using the ECL method with
HRP-labeled anti-goat IgG at a dilution of 1:6,000 (Fig. 6).
Results
PCR products of G250 and hGM-CSF
genes
Results of an agarose gel electrophoresis assay
showed that the size of the specific PCR amplification products
were ~1.48 and 0.48 kb for the G250 and hGM-CSF
genes, respectively. The results were in agreement with the
anticipated fragment.
Recombinant plasmid detection by
restriction enzyme digestion
pVAX1-G250 was double digested by KpnI
and HindIII. Results of an agarose gel electrophoresis assay
showed the two fragments to be ~3.0 and 1.5 kb for the G250
and hGM-CSF genes, respectively. pVAX1-G250-GM was
double digested by KpnI and XbaI, and the two
fragments were found to be ~4.5 and 0.48 kb for the G250 and
hGM-CSF genes, respectively, were noted.
Sequencing identification
The recombinant plasmid pVAX1-G250-GM was
examined by sequencing. The results showed that it was identical to
the reported G250 gene sequence (NCBI accession: BC014950)
and the hGM-CSF gene sequence (NCBI accession: M10663).
Identification of fusion protein by
immunocytochemistry staining
Immunocytochemistry staining results showed that the
expression of the G250 protein was negative in the transfected
pVAX1 group. However, in the
pVAX1-G250-GM-transfected group the expression of G250
protein was positive (Fig. 3).
Atomic force microscopy
Normal HEK 293 cells had a smooth surface, with a
rich-pseudopod extension and a cell height that reached 1,250 μm.
When transfected with pVAX1, the cell surfaces became rough
and the cell height was 1,161 μm. pVAX1-G250-GM was
transfected into cells and the cell height became significant
(~7,450 nm).
ELISA analysis
The expression values of the hGM-CSF protein of the
pVAX1 control group were 53.97±0.53 and 54.10±0.79 pg/ml (χ̄
± s, n=3), whereas the expression values of the hGM-CSF protein of
the pVAX1-G250-GM-transfected group were 482.47±5.86 and
513.36±4.45 pg/ml. The results were considered to be statistically
significant (P<0.05).
Western blotting
The proteins which bound to the G250 antibody were
detected following recombinant plasmid pVAX1-G250-hGM
transfection into the HEK 293 cells.
Discussion
RCC is a highly immunogenic tumor that induces the
host immune response. RCC primary tumors and their metastasis were
found to be relatively stable over time. The tumors occasionally
spontaneously regressed, indicating that immune mechanisms play a
key role in RCC. G250/MN/CA IX, a good tissue-specific
RCC-associated antigen, has been identified and cloned from a
variety of RCC cell lines. This antigen has 459 amino acids lying
in the plasma and nuclear membranes, with a molecular weight
between 58 and 54 ku (6). Clear
cell carcinoma of the kidneys, and the majority of other types of
RCC, express G250 antigen. Approximately 88% of tumor
metastases also express the G250 antigen (7), which regulates cell proliferation in
hypoxic conditions. G250 possesses HLA-A2.1-restricted
epitopes (8), and the G250
transduction in peripheral blood mononuclear cells produced
cytotoxic T cells, which inhibited the tumor cells expressed in the
growth of G250. For these reasons, G250 is a key target for
RCC immunotherapy (9).
In recent years, increasing importance has been
attached to gene therapy as adjuvant treatment in DNA vaccination.
As a preponderant adjuvant, hGM-CSF has the ability to
activate endotheliocytes and macrophagocytes through a variety of
mechanisms. It also regulates the amount of and function of
antigen-presenting cells and enhances the cytoactive level of
cytotoxic T lymphocyte and natural killer cells to strengthen the
immune level. hGM-CSF is considered to be an adjuvant
therapy that assists in the preparation of DNA vaccination and more
favorable experimental results (10,11).
Tani et al (12) prepared
hGM-CSF gene-modified autologous tumor cells from type IV
tumors and used gene-modified tumor cells to treat RCC patients.
The results showed that the vaccine induced a tumoral immune
response, indicating that it was able to enhance the anti-tumor
effect. Simultaneously, hGM-CSF combined with autologous
tumor cells increased the number of lymphocytes, resulting in slow
disease progression and extended patient survival (13).
Concerning safety requirements, we used vector
pVAX1, which is approved by the FDA and can be applied to
the human body. Linkers were added when the upstream primers were
designed in order to amplify hGM-CSF. To produce G250
protein, the linkers were added between the fusion proteins and the
hGM-CSF fold in natural three-dimensional structures while
maintaining intrinsic immunogenicity. To assess the character of
the fusion proteins, we transfected the recombinant plasmids into
HEK 293 cells using the liposome transfection method and detected
the proteins with immunocytochemistry staining and Western
blotting. The results showed that cells transfected with
recombinant plasmids in immunocytochemistry expressed protein,
whereas the control group had no significant change in color. This
expression shows that G250 protein is immunoreactive.
Immunocytochemistry usually uses conventional light or electron
microscopy. These classical methods lack dynamic, three-dimensional
and single-molecule measurement ability. This study utilized AFM, a
novel surface imaging technique that detected nanometer positioning
resolution, in order to scale antigen-antibody complex positioning,
qualitative, quantitative and three-dimensional display on the
membrane surface. AFM detects, not only single-molecule single
atoms, but also specific biological molecules or non-specific
binding between the three-dimensional morphology of the dynamic
process of change. Cells transfected with recombinant plasmid
ultrastructures exhibit surface roughness and the height changes
significantly, due to the recombinant plasmid which, since it
contains G250, binds with the antibody, thereby reducing the
interaction between the cells. Notably, the use of ELISA has shown
an accurate expression of hGM-CSF fusion protein.
In conclusion, we constructed a eukaryotic vector
containing G250 and hGM-CSF and detected the
expression of the two proteins. However, further investigations
regarding immunogenicity and safety of the vaccine are required, as
well as examination of the vaccination of the anti-G250 antigen and
its immune response mechanism of biological immunotherapy in
RCC.
Acknowledgements
This study was supported by the Key specialist of
Guangdong Provincial 11th Five-Year Plan.
References
|
1
|
Kirkali Z, Tuzel E and Mungan MU: Recent
advances in kidney cancer and metastatic disease. BJU Int.
88:818–824. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ljungberg B, Hanbury DC, Kuczyk MA,
Merseburger AS, Mulders PF, Patard JJ and Sinescu IC: European
Association of Urology Guideline Group for renal cell carcinoma.
Eur Urol. 51:1502–1510. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bleumer I, Oosterwijk E and De Mulder PF:
Immunotherapy for renal cell carcinoma. Eur Urol. 44:65–75. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Van Poppel H, Joniau S and van Gool SW:
Vaccine therapy in patients with renal cell carcinoma. European
Urology. 55:1333–1344. 2009.PubMed/NCBI
|
|
5
|
Potter CP and Harris AL: Diagnostic,
prognostic and therapeutic implications on carbonic anhydrases in
cancer. Br J Cancer. 89:2–7. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Opavský R, Pastoreková S, Zelník V,
Gibadulinová A, Stanbridge EJ, Závada J, Kettmann R and Pastorek J:
Human MN/CA9 gene, a novel member of the carbonic anhydrase family:
structure and exon to protein domain relationships. Genomics.
33:480–487. 1996.PubMed/NCBI
|
|
7
|
Oosterwijk E, Ruiter DJ, Hoedemaeker PJ,
Pauwels EK, Jonas U, Zwartendijk J and Warnaar SO: Monoclonal
antibody G250 recognizes a determinant present in renal cell
carcinoma and absent from normal kidney. Int J Cancer. 38:489–494.
1986. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Vissers JL, De Vries IJ, Schreurs MW,
Engelen LP, Oosterwijk E, Figdor CG and Adema GJ: The renal cell
carcinoma-associated antigen G250 encodes a human leukocyte antigen
(HLA)-A2.1-restricted epitope recognized by cytotoxic T
lymphocytes. Cancer Res. 59:5554–5559. 1999.
|
|
9
|
Dorai T, Sawczuk IS, Pastorek J, Wiernik
PH and Dutcher JP: The role of carbonic anhydrase IX overexpression
in kidney cancer. Eur J Cancer. 41:2935–2947. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cruciani M, Mengoli C, Serpelloni G, Mazzi
R, Bosco O and Malena M: Granulocyte macrophage colony-stimulating
factor as an adjuvant for hepatitis B vaccination: a meta-analysis.
Vaccine. 25:709–718. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Miyashita T, Shah FA, Marti G, Wang J,
Armstrong T, Bonde P, Gibson MK, Yoshimura K, Montgomery EA, Duncan
MD, Jaffee EM and Harmon JW: Vaccine impedes the development of
reflux-induced esophageal cancer in a surgical rat model: efficacy
of the vaccine in a pre-Barrett's esophagus setting. J Gastrointest
Surg. 12:2–9. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tani K, Azuma M, Nakazaki Y, et al: Phase
I study of autologous tumor vaccines transduced with the GM-CSF
gene in four patients with stage IV renal cell cancer in Japan:
clinical and immunological findings. Mol Ther. 10:799–816. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Schwaab T, Tretter CP, Gibson JJ, Cole BF,
Schned AR, Harris R, Wallen EM, Fisher JL, Waugh MG, Truman D,
Stempkowski LM, Crosby NA, Heaney JA and Ernstoff MS: Immunological
effects of granulocyte-macrophage colony-stimulating factor and
autologous tumor vaccine in patients with renal cell carcinoma. J
Urol. 171:1036–1042. 2004. View Article : Google Scholar : PubMed/NCBI
|