Introduction
Programmed cell death 4 (Pdcd4) is a ubiquitously
expressed, novel tumor suppressor gene localized in chromosome
10q24, whose protein product plays a role in the suppression of
tumorigenesis and tumor progression and invasion (1–4).
Although Pdcd4 was first identified as being differentially
upregulated during apoptosis, experimental evidence established it
as a novel tumor suppressor (5–8).
Previous studies indicated that the overexpression of Pdcd4
inhibited tumor promoter TPA-induced neoplastic transformation in
JB6 and P-positive cells (4,9,10).
Pdcd4 expression is often decreased in progressed carcinomas of the
lung, ovary, breast, colon and prostate (7,11–13).
Pdcd4 protein acts as an inhibitor that suppresses protein
translation through its binding to and inhibition of the helicase
activity of eukaryotic translation initiation factor 4A (eIF4A),
the latter being a component of the protein translation complex
(4,9,14).
Certain studies investigating cellular functions of Pdcd4
demonstrated that it was capable of regulating molecules such as
p21 (7), Cdk3, ornithine
decarboxylase (1), carbonic
anhydrase II (15) and
JNK/c-Jun/AP-1 (6). It has also
been shown to suppress the expression of the invasion-related
urokinase receptor (u-PAR) gene, and to suppress
invasion/intravasation via the Sp1/Sp3 promoter motifs in cancer
(8). However, the physiological
role of Pdcd4 is not clearly understood. The purpose of the present
study was to examine the role of Pdcd4 in colorectal adenocarcinoma
(CRA). The prognosis of colorectal cancer patients varies depending
on the depth of the invasion of tumor cells and the lymph node
status, and these factors are assessed only by a histopathological
examination of surgically resected tissues (16). Pdcd4 expression and its subcellular
localization were investigated using immunohistochemistry, and
Pdcd4 expression was correlated with clinical and pathological
parameters including histology, grade, stage and overall
survival.
Materials and methods
Patients
Among patients who underwent curative surgery for
CRA at Chosun University Hospital between January 1992 and December
2001, the present study was conducted on a non-consecutive series
of 108 patients from whom paraffin-embedded tissues were obtained
and relatively well preserved, medical records were complete and
the patient status had been followed up. Patient survival was
confirmed through telephone interviews and by mail. Patients who
underwent preoperative chemoradiotherapy and emergency surgery, and
patients who had evidence of hereditary non-polyposis colorectal
cancer or familial adenomatous polyposis were excluded from the
study.
The various clinicopathological parameters of the
patients were confirmed by reviewing the patient medical records
and pathology files. The correlation of clinicopathological
parameters and immunohistochemical findings with survival was
investigated for all 108 patients. Informed consent was obtained
from each patient according to the institutional guidelines, and
the research protocols were approved by the Ethics Committee of the
University Hospital.
Histopathological analysis
Microscopic examination
Each tumor was re-evaluated by retrospective
analysis of the medical records and the tissue slide files of the
Department of Pathology. Age, gender, tumor size, histological
subtype, degree of differentiation, depth of tumor invasion, status
of lymph node metastasis and presence of distant metastasis were
assessed. Stage was defined according to the TNM staging system of
the American Joint Committee on Cancer (17). The examined tissues were fixed in
10% neutral formalin, and the prepared paraffin-embedded tissues
were sectioned at a thickness of 4–5 μm. Hematoxylin and eosin
staining was performed, and the sections were examined under a
light microscope. A representative area suitable for the study
purpose was selected, and slides were prepared for
immunohistochemical analysis.
Immunohistochemical staining
Specimens in this study were tested using a rabbit
polyclonal antibody against Pdcd4 (Abcam, Cambridge, MA, USA)
according to the manufacturer’s instructions. Immunolocalization
was performed using the mouse ImmunoCruz™ Staining System sc-2050
(Santa Cruz Biotechnology, Santa Cruz, CA, USA), according to the
manufacturer’s instructions. The staining process was performed
according to a standard protocol. Briefly, the 4-μm sections that
were obtained following formalin fixation and paraffin embedding
were deparaffinized in xylene and rehydrated with distilled water
through a graded series of ethanol solutions. The sections were
then placed in a glass jar with 10 mM citrate buffer (pH 6.0) and
were irradiated in a microwave oven for 15 min. The sections were
allowed to cool in the jar at room temperature for 20 min. The
slides were rinsed with Tris-buffered saline (TBS). A blocking
reagent was added for 10 min after quenching the endogenous
peroxidase activity in 0.3% hydrogen peroxide for 10 min. The
slides were then washed as previously described, and the slides
were subsequently subjected to the primary antibody reaction.
Immunohistochemistry was performed using the Nexes ES (Ventana,
Tucson, AZ, USA). Slides were incubated with the antibodies for 32
min. The Ventana basic DAB detection kit (catalog no. 760-001) was
the secondary detection method. This kit includes biotinylated
immunoglobulin secondary antibody, containing affinity-purified
goat anti-mouse IgG and IgM (b200 lg/ml) and goat anti-rabbit IgG
(b200 lg/ml) in phosphate buffer with preservative. Incubation was
performed for 8 min, and was followed by conjugated streptavidin
horseradish peroxidase. Slides were counterstained with hematoxylin
(Ventana catalog no. 760-2021).
Analysis and interpretation of
staining
Two pathologists, who were unaware of the clinical
course of the subjects in order to exclude subjectivity, evaluated
the results of the staining. Since Pdcd4 expression was identified
in both the nucleus and the cytoplasm, nuclear and cytoplasmic
staining was evaluated separately. Nuclear and cytoplasmic scoring
was determined according to the percentage of positive nuclear
staining and the intensity of cytoplasmic staining in cells,
respectively (18). Nuclear scoring
was as follows (18): score 1,
negative; score 2, <30%; score 3, 30 to 70%; and score 4,
>70%. Cytoplasmic scores were determined according to staining
intensity: score 1, negative; score 2, weak staining intensity;
score 3, intermediate staining intensity and score 4, strong
staining intensity, in reference to a strongly stained tissue as a
control. The overall Pdcd4 score was calculated by adding the
nuclear and cytoplasmic scores, and 4 groups were defined: negative
(total score, 1 and 2), weak staining (total score, 3 and 4),
intermediate staining (total score, 5 and 6) and strong staining
(total score, 7 and 8).
Statistical analysis
For the statistical analysis of Pdcd4 in CRA, its
association with various clinicopathological factors, and survival
rate or survival time, we used Pearson’s Chi-square test and
linear-by-linear association to compare the categorical data. The
life table and statistical significance were computed using the
Kaplan-Meier method and the log-rank test. P<0.05 was considered
to be statistically significant. The Stat View software package was
used for all statistical analyses (Abacus Concepts, Berkeley, CA,
USA).
Results
The clinical characteristics of the patients are
shown in Table I. The average age
at the time of surgery was 62.1 years and the ratio of male to
female participants was 55:53 (50.9:49.1%). Tumors were mostly
well- to moderately differentiated (88 cases, 81.5%), although 7
(6.5%) cases exhibited poor differentiation, and 13 (12.0%) cases
were mucinous adenocarcinomas. Pdcd4 was ususally detected in the
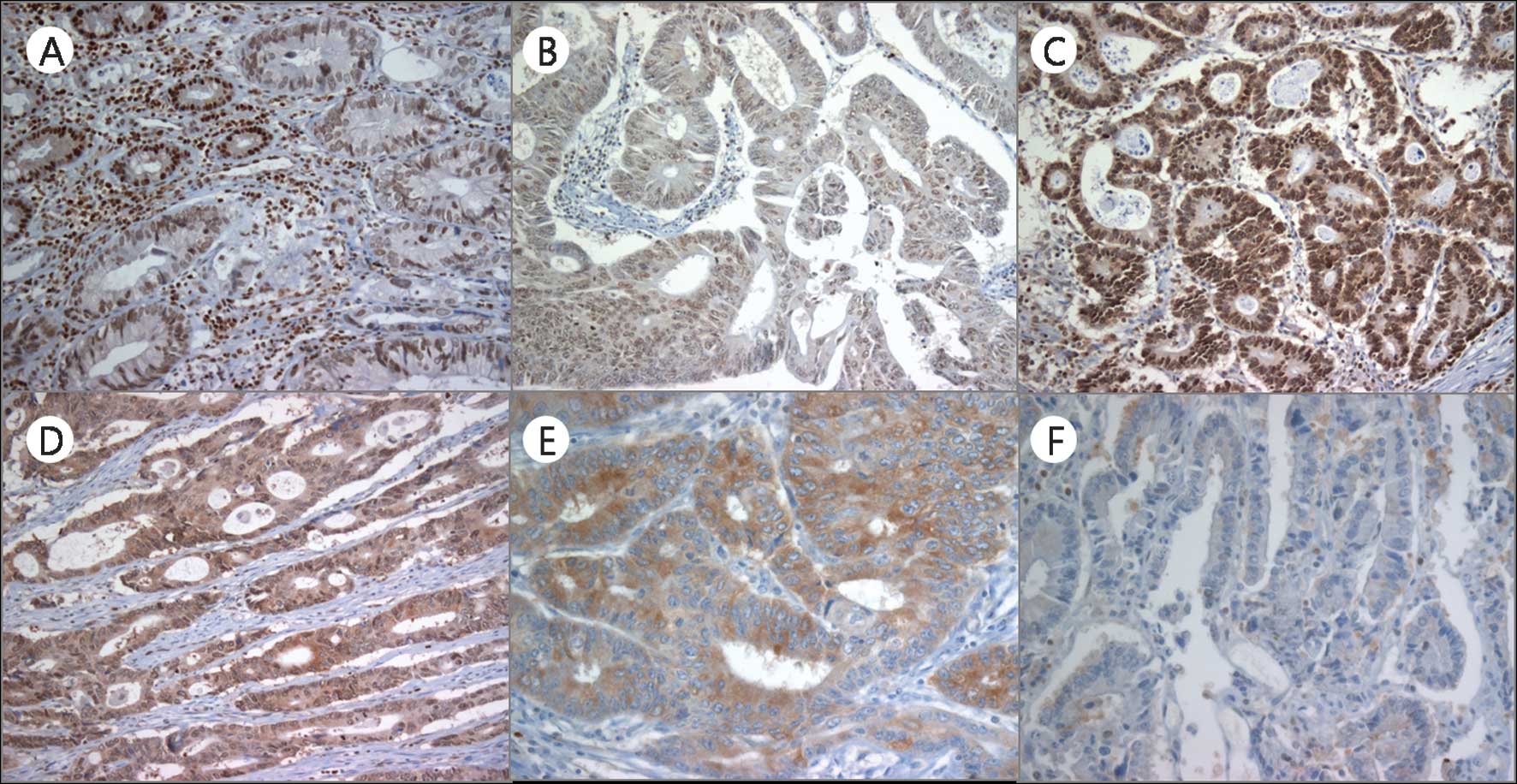
nuclei of non-neoplastic epithelial cells (Fig. 1A). However, in the cancer cells,
Pdcd4 was observed in both the nuclei and the cytoplasms, and
nuclear expression was downregulated. The nuclear, cytoplasmic and
overall Pdcd4 scores were evaluated, respectively. An overall Pdcd4
score of 0, 1, 2 and 3 was identified in 11 (10.2%), 33 (30.5%), 57
(52.8%) and 7 (6.5%) cases, respectively. In contrast to normal
epithelial cells, the majority of tumor cells had a weak to
moderate cytoplasmic staining pattern for Pdcd4. A cytoplasmic
expression score of 1, 2, 3 and 4 was observed in 63 (58.3%), 25
(23.1%), 17 (15.8%) and 3 (2.8%) cases, respectively (Table II). The cytoplasmic and overall
score revealed a tendency to increase gradually according to the
nodal status and AJCC clinical stage (linear-by-linear association,
p<0.05). The cytoplasmic score also revealed significant
difference among nodal status and AJCC clinical stages (Pearson’s
Chi-square test, p<0.05). Moreover, nuclear expression scores of
1, 2, 3 and 4 were identified in 24 (22.2%), 18 (16.7%), 20 (18.5%)
and 46 (42.6%) cases, respectively, and showed no tendency or
significant difference with any of the clinicopathological
parameters (Table II, Fig. 1B-F).
 | Table ISummary of clinicopathological
factors. |
Table I
Summary of clinicopathological
factors.
| Characteristics | n (%) |
|---|
| Age |
| ≤40 | 14 (13.0) |
| 51–59 | 28 (25.9) |
| 60–69 | 38 (35.2) |
| ≥70 | 28 (25.9) |
| Gender |
| Male | 55 (50.9) |
| Female | 53 (49.1) |
| Pathological tumor
classification (pT) |
| pT1 | 2 (1.9) |
| pT2 | 21 (19.4) |
| pT3 | 80 (74.1) |
| pT4 | 5 (4.6) |
| Pathological lymph
node classification |
| pN0 | 73 (67.6) |
| pN1 | 26 (24.1) |
| pN2 | 9 (8.3) |
| Metastasis
classification (M) |
| M0 | 104 (96.3) |
| M1 | 4 (3.7) |
| AJCC
classification |
| I | 21 (19.4) |
| IIA | 49 (45.3) |
| IIIB | 2 (1.9) |
| IIIA | 2 (1.9) |
| IIIB | 22 (20.4) |
| IIIC | 8 (7.4) |
| IV | 4 (3.7) |
| Differentiation |
| Well/Moderate | 88 (81.5) |
| Poor | 7 (6.5) |
| Mucinous | 13 (12.0) |
 | Table IISummary of Pdcd4 expression and
correlation with clinicopathologic parameters. |
Table II
Summary of Pdcd4 expression and
correlation with clinicopathologic parameters.
| n | Pdcd4 expression | Nuclear
expression | Cytoplasmic
expression |
|---|
|
|
|
|---|
| 0A | 1A | 2A | 3A | P-value | 1 | 2 | 3 | 4 | P-value | 1 | 2 | 3 | 4 | P-value |
|---|
| T stage | | | | | | | | | | | | | | | | |
| 1 | 2 | 0 | 1 | 1 | 0 | 0.443 | 1 | 0 | 0 | 1 | 0.018d | 1 | 0 | 1 | 0 | 0.903 |
| 2 | 21 | 3 | 8 | 9 | 1 | | 5 | 2 | 10 | 4 | | 13 | 4 | 3 | 1 | |
| 3 | 80 | 8 | 20 | 46 | 6 | | 17 | 14 | 9 | 40 | | 45 | 20 | 13 | 2 | |
| 4 | 8 | 0 | 4 | 1 | 0 | | 1 | 2 | 1 | 1 | | 4 | 1 | 0 | 0 | |
| N stage | | | | | | | | | | | | | | | | |
| N0 | 73 | 10 | 24 | 36 | 3 | 0.133 | 17 | 12 | 14 | 30 | 0.961 | 49 | 16 | 6 | 2 | 0.011d |
| N1 | 35 | 1 | 9 | 21 | 4 | | 7 | 6 | 16 | 16 | | 14 | 9 | 11 | 1 | |
| M stage | | | | | | | | | | | | | | | | |
| M0 | 104 | 11 | 33 | 53 | 7 | 0.294 | 24 | 18 | 20 | 42 | 0.133 | 60 | 24 | 17 | 3 | 0.808 |
| M1 | 4 | 0 | 0 | 4 | 0 | | 0 | 0 | 0 | 4 | | 3 | 1 | 0 | 0 | |
| Clinical stage | | | | | | | | | | | | | | | | |
| Highd | 72 | 10 | 24 | 35 | 3 | 0.114 | 17 | 12 | 14 | 29 | 0.906 | 49 | 15 | 6 | 2 | 0.009d |
| Lowd | 36 | 1 | 9 | 22 | 4 | | 7 | 6 | 6 | 17 | | 14 | 10 | 11 | 1 | |
|
Diff.1 | | | | | | | | | | | | | | | | |
|
W/Ma | 88 | 11 | 26 | 46 | 5 | 0.359 | 22 | 14 | 15 | 37 | 0.724 | 54 | 17 | 15 | 2 | 0.440 |
| Pb | 7 | 0 | 4 | 2 | 1 | | 1 | 2 | 1 | 3 | | 3 | 3 | 1 | 0 | |
| Mc | 13 | 0 | 3 | 9 | 1 | | 1 | 2 | 4 | 6 | | 6 | 5 | 1 | 1 | |
|
Diff.2 | | | | | | | | | | | | | | | | |
|
Non-M1 | 93 | 10 | 29 | 48 | 6 | 0.925 | 22 | 16 | 15 | 40 | 0.419 | 55 | 20 | 16 | 2 | 0.438 |
| M2 | 15 | 1 | 4 | 9 | 1 | | 2 | 2 | 5 | 6 | | 9 | 5 | 1 | 1 | |
Discussion
Pdcd4 was first identified as being differentially
upregulated during apoptosis; however, experimental evidence has
established it as a novel tumor suppressor (5–8). Pdcd4
has been identified as a suppressor of transformation (5,9,19),
tumorigenesis, progression (1),
invasion, matrix-metalloproteinase activation (3,8) and
tumor growth (20). In the present
study, the Pdcd4 expression pattern was investigated in CRA
(Fig. 1). Nuclear expression
exhibited a tendency to be lost or weak in tumor cells, whereas
cytoplasmic expression was increased. The transfer of Pdcd4 between
the nucleus and cytoplasm is understood to have a significant
effect on the regulation of its function. Pdcd4 is localized
predominantly in the nucleus under normal growth conditions but is
exported to the cytoplasm upon serum withdrawal (14). A number of authors have studied the
localization of Pdcd4 in various cell types. Yang et al
(19) reported that in the mouse
JB6 preneoplastic cell line, both nuclear and cytoplasmic
localization of Pdcd4 were identified. In their study, Yoshinaga
et al (21) found that Pdcd4
accumulated in the nucleus at the G0 phase of asynchronous cultures
of human normal fibroblasts but was localized in the cytoplasm
during the cell cycle in tumor cell lines. Goke et al
(7) reported that 6 of 7 colon
carcinomas examined revealed a complete absence of nuclear Pdcd4
staining and additional cytoplasmic staining in 4 tumors. According
to this study, epithelial cells of the prostate, breast and lung
exhibited intense nuclear but no cytoplasmic staining; a clear
shift from nuclear localization to cytoplasmic staining was
observed in all colonic adenomas investigated. In the case of
invasive breast cancer tissues, both nuclear and cytoplasmic
localization were observed. In an investigation conducted into
colorectal cancer, in addition to a decreased expression level of
Pdcd4, a significant loss of nuclear Pdcd4 from normal tissues to
colonic adenomas and carcinomas was also observed (18), supporting the hypothesis that the
intracellular localization of Pdcd4 plays a significant role in the
regulation of tumor cell progression. In the present study, similar
results to other authors were also demonstrated, in that a
differential expression pattern of Pdcd4 was found between normal
and carcinoma cells by IHC analysis. Additionally, the cytoplasmic
expression level was shown to increase according to the AJCC
clinical stage. We suggested that Pdcd4 translocates from the
nucleus to the cytoplasm during colonic cancer development. As
proposed by Zhang (23), the
accumulation of Pdcd4 in the nuclei is crucial for apoptosis, and
the regulatory mechanisms of the localization of Pdcd4 protein may
play a significant role in the induction of apoptosis in
hepatocellular carcinoma cells. Wei et al (13) speculated that the accumulation of
Pdcd4 in the nucleus negatively regulates cell proliferation while
the cytoplasmic sequestration of Pdcd4 may abolish its involvement
in ovarian cancer cells. Palamarchuk et al (22) transfected 293 cells that stably
expressed wild-type Pdcd4 and observed a primarily nuclear Pdcd4
staining pattern. However, an S457A mutant of Pdcd4 that was unable
to be phosphorylated by Akt, in contrast to the wild-type, was not
localized in the nucleus but in the cytoplasm, indicating the
significance of this amino acid and Akt phosphorylation site in the
localization of Pdcd4. However, this phenomenon requires further
confirmation using a larger patient cohort, and in vitro
studies are necessary in order to understand its regulatory
mechanism. In conclusion, both the localization and expression
level of Pdcd4 may be potential indicators of colonic cancer
progression.
The present study investigated the Pdcd4 expression
pattern in CRA using IHC, and analyzed the correlation between
Pdcd4 expression and clinical and pathological parameters,
including histology, grade, stage and overall survival. In
conclusion, in CRA, nuclear Pdcd4 expression was decreased and
inversely aberrant cytoplasmic staining was identified, which was
significantly increased according to the nodal status and AJCC
clinical stage. We suggest that Pdcd4 translocates from the nucleus
to cytoplasm in CRA progression, and that increased cytoplasmic
expression of Pdcd4 is a potential prognostic marker in CRA.
However, the regulatory mechanism of alteration of subcellular
localization remains to be further investigated.
Acknowledgements
This study was supported by the National Research
Foundation of Korea (NRF) Grant funded by the Ministry of
Education, Science and Technology (MEST) through the Research
Center for Resistant Cells (R-13-1003-09).
References
|
1
|
Jansen AP, Camalier CE and Colburn NH:
Epidermal expression of the translation inhibitor programmed cell
death 4 suppresses tumorigenesis. Cancer Res. 65:6034–6041. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hilliard A, Hilliard B, Zheng SJ, et al:
Translational regulation of autoimmune inflammation and lymphoma
genesis by programmed cell death 4. J Immunol. 177:8095–8102. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Yang HS, Matthews CP, Clair T, et al:
Tumorigenesis suppressor Pdcd4 down-regulates mitogen-activated
protein kinase kinase kinase kinase 1 expression to suppress colon
carcinoma cell invasion. Mol Cell Biol. 26:1297–1306. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Soejima H, Miyoshi O, Yoshinaga H, et al:
Assignment of the programmed cell death 4 gene (Pdcd4) to human
chromosome band 10q24 by in situ hybridization. Cytogenet Cell
Genet. 87:113–114. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Cmarik JL, Min H, Hegamyer G, et al:
Differentially expressed protein Pdcd4 inhibits tumor
promoter-induced neoplastic transformation. Proc Natl Acad Sci USA.
96:14037–14042. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bitomsky N, Bohm M and Klempnauer KH:
Transformation suppressor protein Pdcd4 interferes with
JNK-mediated phosphorylation of c-Jun and recruitment of the
coactivator p300 by c-Jun. Oncogene. 23:7484–7493. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Goke R, Barth P, Schmidt A, Samans B and
Lankat-Buttgereit B: Programmed cell death protein 4 suppresses
CDK1/cdc2 via induction of p21(Waf1/Cip1). Am J Physiol Cell
Physiol. 287:1541–1546. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Leupold JH, Yang HS, Colburn NH, Asangani
I, Post S and Allgayer H: Tumor suppressor Pdcd4 inhibits
invasion/intravasation and regulates urokinase receptor (u-PAR)
gene expression via Sp-transcription factors. Oncogene.
26:4550–4562. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yang HS, Jansen AP, Nair R, et al: A novel
transformation suppressor, Pdcd4, inhibits AP-1 transactivation but
not NF-κB or ODC transactivation. Oncogene. 20:669–676.
2001.PubMed/NCBI
|
|
10
|
Yang HS, Knies JL, Stark C and Colburn NH:
Pdcd4 suppresses tumor phenotype in JB6 cells by inhibiting AP-1
transactivation. Oncogene. 22:3712–3720. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li T, Li D, Sha J, Sun P and Huang Y:
MicroRNA-21 directly targets MARCKS and promotes apoptosis
resistance and invasion in prostate cancer cells. Biochem Biophys
Res Commun. 383:280–285. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wen YH, Shi X, Chiriboga L, Matsahashi S,
Yee H and Afonja O: Alterations in the expression of Pdcd4 in
ductal carcinoma of the breast. Oncol Rep. 18:1387–1393.
2007.PubMed/NCBI
|
|
13
|
Wei NA, Liu SS, Leung TH, et al: Loss of
Programmed cell death 4 (Pdcd4) associates with the progression of
ovarian cancer. Mol Cancer. 8:702009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bohm M, Sawicka K, Siebrasse JP,
Brehmer-Fastnacht A, Peters R and Klempnauer KH: The transformation
suppressor protein Pdcd4 shuttles between nucleus and cytoplasm and
binds RNA. Oncogene. 22:1905–1910. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lankat-Buttgereit B, Gregel C, Knolle A,
Hasilik A, Arnold R and Goke R: Pdcd4 inhibits growth of tumor
cells by suppression of carbonic anhydrase type II. Mol Cell
Endocrinol. 214:149–153. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Park JK, Hong R, Kim KJ, Lee TB and Lim
SC: Significance of p-STAT3 expression in human colorectal
adenocarcinoma. Oncol Rep. 20:597–604. 2008.PubMed/NCBI
|
|
17
|
Greene FL, Page DL, Fleming ID, et al:
AJCC Cancer Staging Manual. 6th edition. Springer-Verlag; New York:
pp. 157–164. 2002
|
|
18
|
Mudduluru G, Medved F, Grobholz R, et al:
Loss of programmed cell death 4 expression marks adenoma-carcinoma
transition, correlates inversely with phosphorylated protein kinase
B, and is an independent prognostic factor in resected colorectal
cancer. Cancer. 110:1697–1707. 2007. View Article : Google Scholar
|
|
19
|
Yang HS, Jansen AP, Komar AA, et al: The
transformation suppressor Pdcd4 is a novel eukaryotic translation
initiation factor 4A binding protein that inhibits translation. Mol
Cell Biol. 23:26–37. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chen Y, Knosel T, Kristiansen G, et al:
Loss of Pdcd4 expression in human lung cancer correlates with
tumour progression and prognosis. J Pathol. 200:640–646. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yoshinaga H, Matsuhashi S, Fujiyama C and
Masaki Z: Novel human Pdcd4 (H731) gene expressed in proliferative
cells is expressed in the small duct epithelial cells of the breast
as revealed by an anti-H731 antibody. Pathol Int. 49:1067–1077.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Palamarchuk A, Efanov A, Maximov V,
Aqeilan RI, Croce CM and Pekarsky Y: Akt phosphorylates and
regulates Pdcd4 tumor suppressor protein. Cancer Res.
65:11282–11286. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhang H, Ozaki I, Mizuta T, et al:
Involvement of programmed cell death 4 in transforming growth
factor-beta1-induced apoptosis in human hepatocellular carcinoma.
Oncogene. 25:6101–6112. 2005. View Article : Google Scholar : PubMed/NCBI
|