Introduction
Teratoma is a tumor of embryonic origin, composed of
various tissues derived from three germ layers (ectoderm, endoderm
and mesoderm), which differentiate to form somatic tissues
(1). The teratoma may comprise
either well-differentiated (mature) or primitive (immature) tissue.
The tumor is diagnosed as an immature teratoma if there are areas
of tissue immaturity in the stromal, epithelial or neural
component. The ectodermal components typically include squamous
epithelium and neural tissue. Although neural tissue is usually a
major component of immature and mature teratoma, a retinal
structure is rarely identified in teratoma tissues.
Sacrococcygeal teratoma appears on the caudal
portion of the newborn and accounts for 40% of all teratomas in the
first two decades of life, followed by ovarian teratoma (37%)
(2). The incidence of the disease
is between 1/35,000 and 1/40,000 live births and has a 4:1
female:male ratio (3). Almost all
cases of sacrococcygeal teratoma are diagnosed in the perinatal
period, in contrast to ovarian teratoma which are rare within the
first 24 months of life and seldom appear prior to the age of 6
years (2). Prenatal examination,
including abdominal ultrasonography, may detect the sacrococcygeal
mass, which is treated with surgical resection following birth
(4). The prognosis depends on the
extent and histological scoring of the tumor, with an excellent
prognosis if the tumor has no immature components and adequate
resection is performed (3).
In the present study, we report a case of
sacrococcygeal teratoma including the features of a retina-like
structure. The similarities between the retina-like structure
within the teratoma and the normal mammalian retina are
investigated with regard to tissue structure and protein expression
(5).
The study was conducted with the approval of the
Ethics Committee of the University of Gifu, and consent was
obtained from the patient and her family.
Patients and methods
Clinical history
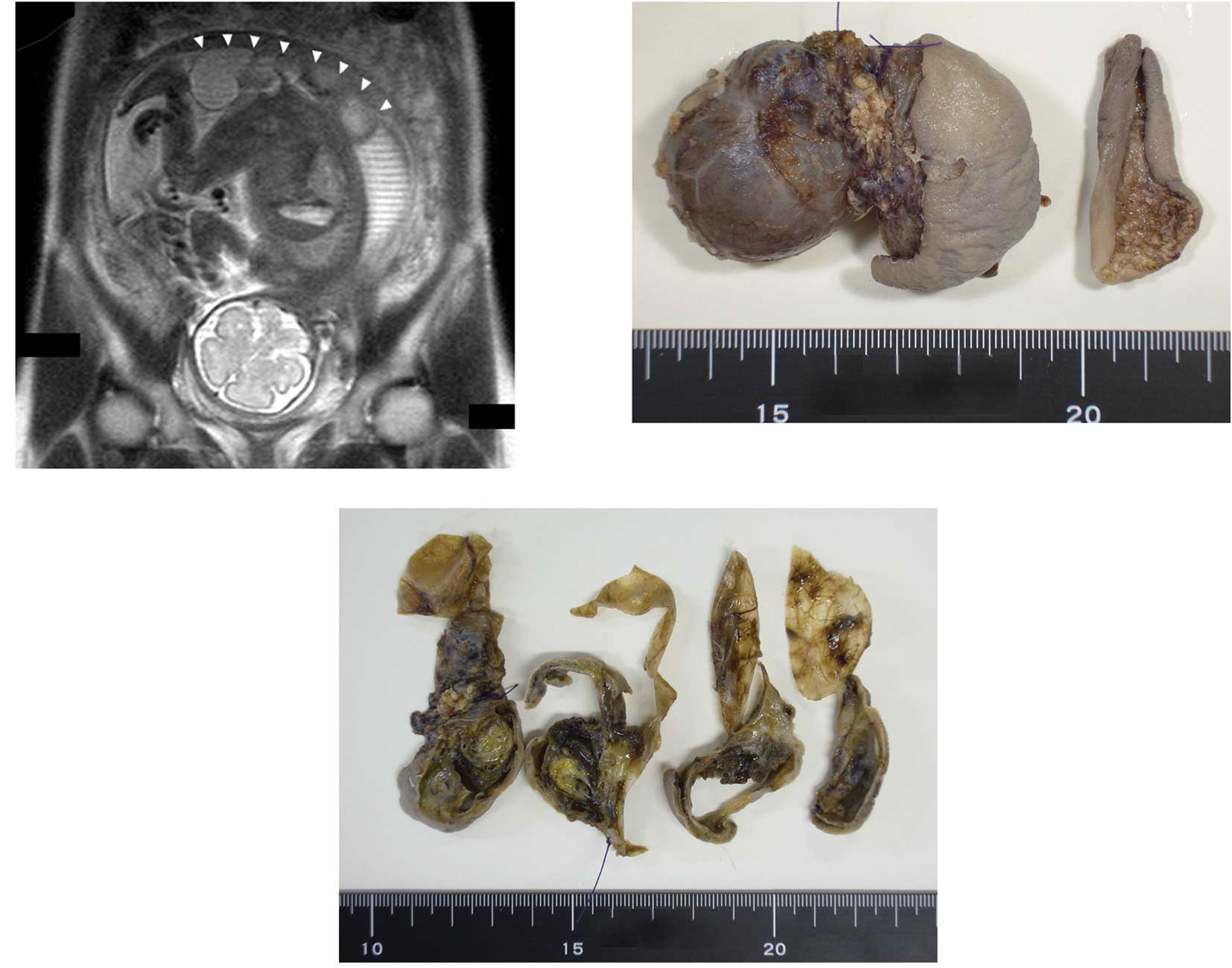
Ultrasonic examination revealed a mass on the
buttocks of a fetus within the uterus of a 29-year-old woman. At 28
weeks of pregnancy, magnetic resonance imaging (MRI) revealed that
the mass had solid, cystic components and it appeared that the
majority of the tumor was located outside of the fetal body
(Fig. 1A). One week later, a
Caesarean section was performed as the fetus was showing signs of
distress. The newborn was premature (29 w, 3 d), of low birth
weight (1,152 g) and had a mass on her back. Following several
treatments during the perinatal period, including intratracheal
intubation and surfactant therapy for respiratory distress syndrome
(RDS), surgical resection was performed 10 days after delivery. The
majority of the mass appeared to be located outside of the pelvis
and was completely resected. Analysis of the peripheral blood of
the newborn revealed an elevation in the α-fetoprotein level at
birth (680,000 ng/ml), which then decreased eight weeks after
surgery (63,000 ng/ml). The newborn experienced no major problems
following surgery and was discharged from the hospital when she had
a body weight of 2,369 g.
Tissue sections and staining
The tissue specimens were formalin-fixed and
paraffin-embedded. The tissue sections were stained with
hematoxylin and eosin (H&E), Kluver-Barrera staining and silver
staining. To determine the cell types of the retina-like structure
of the teratoma, we also used immuno- histochemical staining with
monoclonal or polyclonal antibodies against pax6 (1:100, Chemicon,
Temecula, CA, USA), synaptophysin (1:200 Dako, Carpinteria, CA,
USA), β-tubulin (1:1000 Covance, Princeton, NJ, USA), Hu C/D (1:100
Molecular Probes, Carlsbad, CA, USA), Nestin (1:400 Chemicon), GFAP
(1:300 Dako), RPE 65 (1:200 Millipore, Billerica, MA, USA), HMB45
(1:200 Dako) and Ki-67 (1:200 Dako).
Results
Macroscopic observations
Macroscopically, the mass had a grayish tense cyst 3
cm in diameter and slightly hard components covered with skin
(Fig. 1B). On the cut surface, the
cyst had a thin wall with scattered petechial hemorrhagic lesions
and the other section contained solid dark-yellow components with
thick walls that also contained a cystic lesion (Fig. 1C).
Histological observations
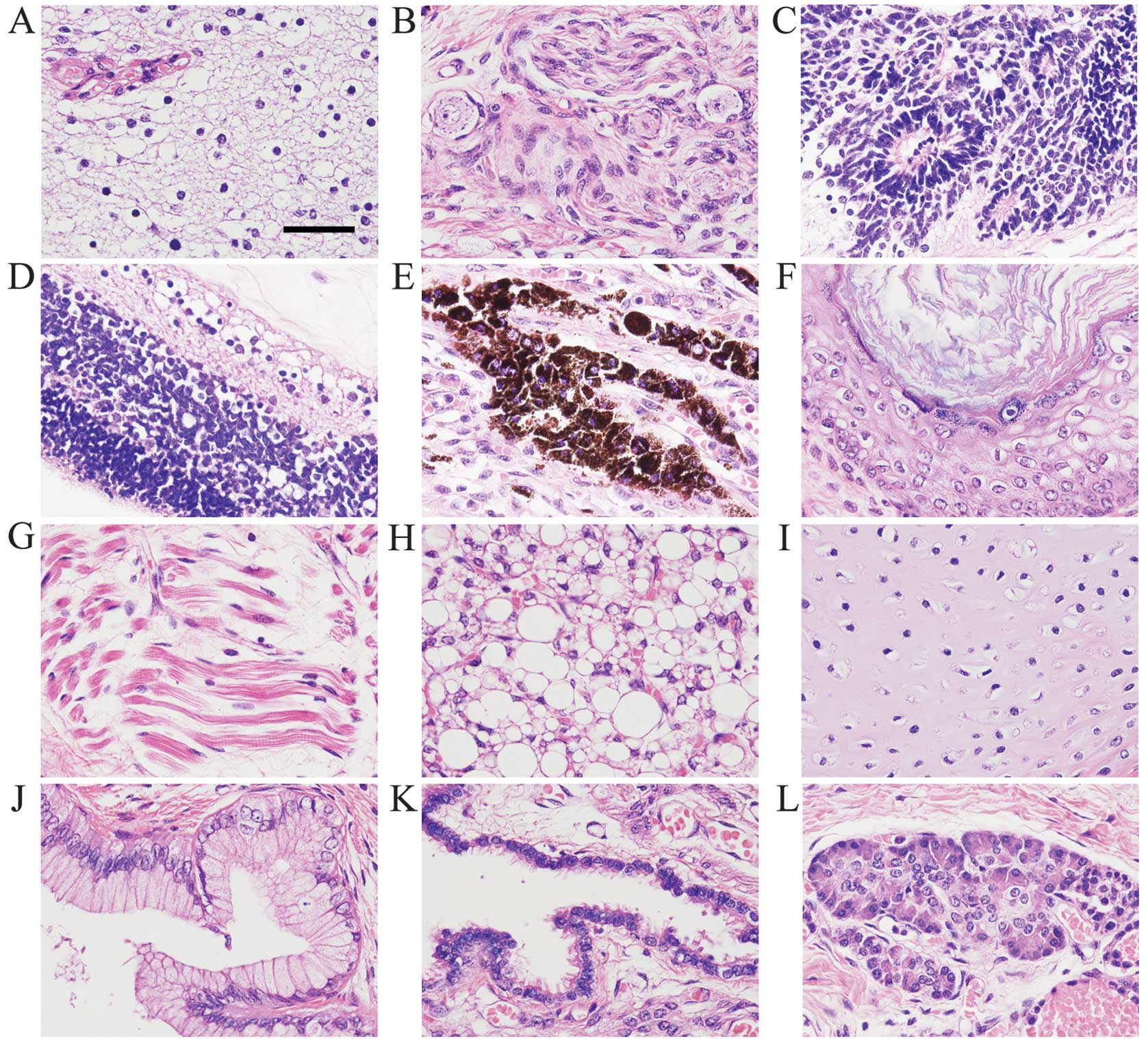
Histologically, the cysts were covered with fibrous
tissue and the solid components contained glandular, brain, neural,
epithelial, cartilaginous and adipose tissues (Fig. 2A, B and E-L). The mass also had
primitive neuroepithelium consisting of small hyperchromatic cells
arranged in rosettes (Fig. 2C).
Consequently, the infant was diagnosed as having mature cystic
teratoma containing immature components. In one section of the
capsules, we observed cells arranged linearly along a fibrous
membrane which had a multilayer structure resembling the retina
(Fig. 2D).
Comparison with normal mouse retina
There are several cell types in the normal mouse and
human retina, including ganglion, bipolar and photoreceptor cells.
The retina-like structure in the teratoma of the present case
(Fig. 3 capital letters) and a
mouse retina as a control (Fig. 3
small letters) are shown with several methods of staining. In
H&E staining, the retina-like structure of the teratoma
(Fig. 3A) shows three cell layers,
which resemble the ganglion cell and inner and outer nuclear layers
in the normal mouse retina (Fig.
3a). The fine fibrous layer, which resembles optic nerve fibers
and was positive for nestin (Fig.
3G) and GFAP (Fig. 3H), is
connected to the layer with irregularly arranged nuclei, which
resembles the ganglion cell layer and was positive for Pax6
(Fig. 3B) and HuC/D (Fig. 3F). The second layer has dense cells
with small, round nuclei and scanty cytoplasm which were focally
positive for Pax6 (Fig. 3B),
synaptophysin (Fig. 3C), β-tubulin
(Fig. 3D) and HuC/D (Fig. 3F), which is similar to the inner
nuclear layer of the mouse retina. The cells of the nuclear layer
opposite to the stromal tissue have nuclei that are similar to
those of photoreceptors and were positive for synaptophysin
(Fig. 3C). There were no clear
cones and rods connected to the photoreceptor cell bodies (Fig. 3A, E and I). Among the three cell
layers, we observed a bundle of fine eosinophilic fibers resembling
the inner and outer plexiform layers.
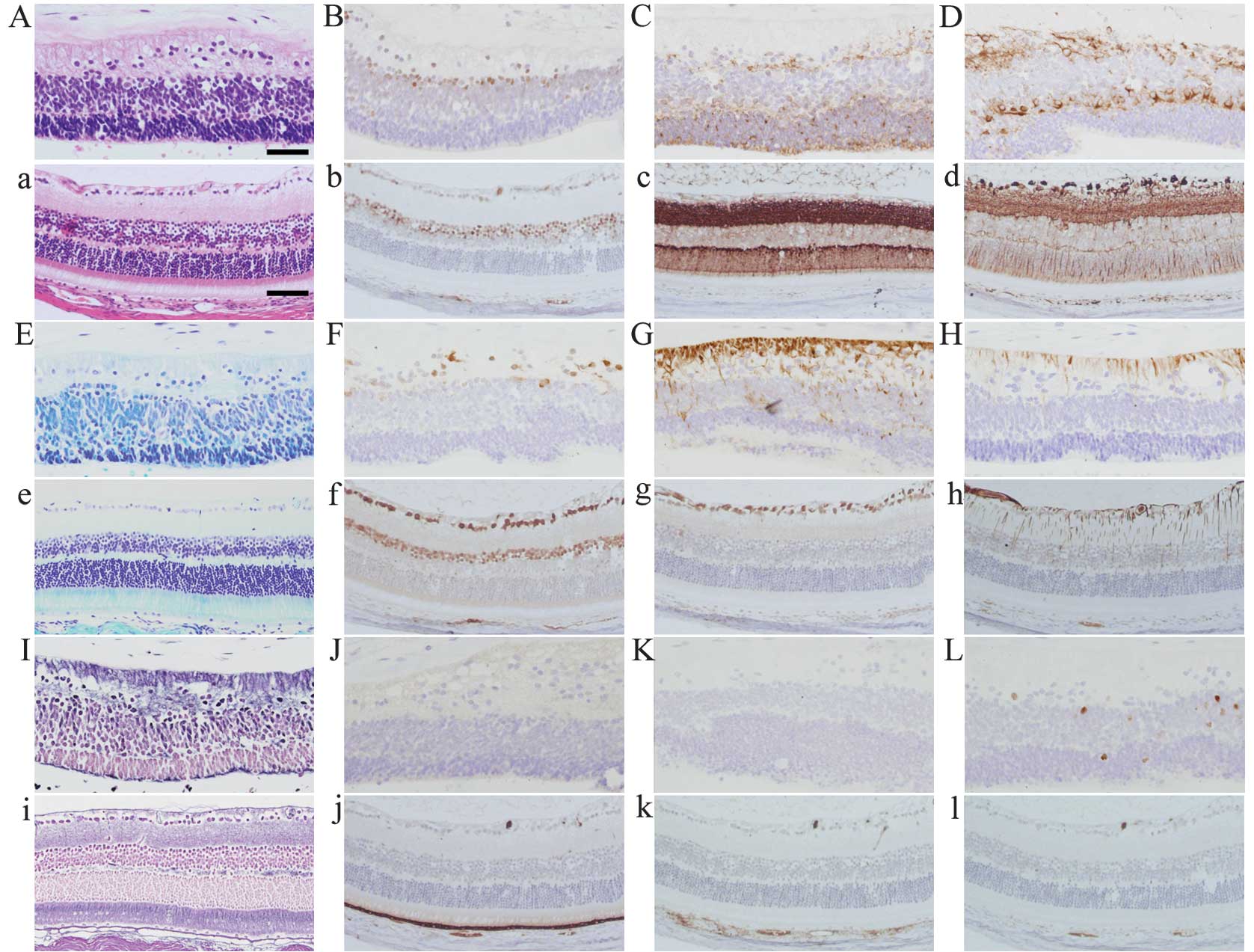 | Figure 3Histopathological images of the
retina-like structure in the human teratoma (A-L) with a normal
mouse retina as a control (a-l). (A, a) H&E staining, (E, e)
Kluver-Barrera staining, (I, i) silver staining.
Immunohistochemistry for (B, b) Pax6, (C, c) synaptophysin, (D, d)
β-tubulin, (F, f) HuC/D, (G, g) nestin, (H, h) GFAP, (J, j) RPE65,
(K, k) HMB45 and (L, l) Ki-67. Scale bars in (A) 50 μm; in (a), 75
μm. |
The retinal pigment epithelium, which is positive
for RPE65 and HMB45 in the normal mouse retina (Fig. 3j and k), was not identified in the
retina-like structure of the teratoma (Fig. 3J and K). There were few
proliferating cells positive for Ki-67 in the retina-like structure
of the teratoma and these cells were not observed in the normal
mouse retina.
Discussion
Although tumors located in the sacrococcygeal region
have been reported since the 19th century, the first review to
elaborate on sacrococcygeal teratoma was by Deveer and Browder in
1937 (6). The American Academy of
Pediatrics Surgical Section (AAPSS) classified sacrococcygeal
teratomas into four groups according to the extent of the tumor and
its histological appearance (3).
The extent of the tumor is described as follows: Type I,
predominantly external with minimal presacral component; Type II,
external but with significant intrapelvic extension; Type III,
apparent externally but predominantly a pelvic mass extending into
the abdomen; Type IV, presacral with no external components
(7). The histological findings are
graded as follows: Grade 0, tumor contains only mature tissue;
Grade 1, tumor contains rare foci of immature tissues; Grade 2,
tumor contains moderate quantities of immature tissues; Grade 3,
tumor contains large quantities of immature tissue with or without
malignant yolk sac elements (8).
The present case was diagnosed as Type I and Grade 1 according to
the AAPSS classification.
Teratomas with a retina-like structure have been
reported and some of the cases showed a complete or partial eye
structure (9). A retrospective
search in our medical center revealed that 2 out of 7 cases of
sacrococcygeal teratoma of newborns had retina-like structures; one
is the present case and the other showed an incompletely stratified
retinal structure associated with retinal markers. The two cases
also had immature neural components, including true rosettes and
primitive neural tubes. However, 4 out of 40 cases of ovarian
teratoma had immature neural components and no retinal structure
was found in the tissues. These observations indicate that the
immature components of teratomas are associated with the formation
of the retina-like structure and that the occurrence of the
teratoma at the sacrococcygeal site is significant in the induction
of a retinal structure.
Results of our histological and immunohistochemical
analysis reveal that a retina-like structure of the teratoma
resembled almost all of the characteristics of normal retina, with
the exception of rods and cones. To form such a structure, there
must be certain factors which polarize and differentiate the cells.
In the teratoma, stromal tissue, including arteries feeding the
retina-like structure, was connected to the side of the ganglion
cell layer, which is the same as the normal retina fed by the
central retinal artery. Moreover, the stromal tissue adjacent to
the retina-like structure had only a few vessels, in contrast to
that of the less differentiated or immature neural components. This
observation shows that polarized structures, including the normal
retina and retina-like structure, need a gradient of concentration
of oxygen or blood flow during formation and structural
development, especially under hypoxic conditions (10). In addition, retinopathy of
prematurity is thought to be the result of excessive oxygen
exposure (11) and may be relevant
to the formation of proliferative lesions, since varying oxygen
concentrations may influence microvascular formation. A precise and
careful search of more cases is needed to clarify the hypothesis of
retinal differentiation within the teratoma.
In conclusion, we have reported a case of
sacrococcygeal teratoma with a retina-like structure in a newborn
and suggested the importance of blood supply and oxygen
circumstance in the formation of the stratified structure. This
finding may aid the development of efficient methods to solve the
problem of condition setting in retinal transplantation (12–14)
using embryonic stem (ES) cell (15) or induced pluripotent stem (iPS) cell
(16) technology, which may cause
teratogenic transformation.
References
|
1
|
Azizkhan RG and Caty MG: Teratomas in
childhood. Curr Opin Pediatr. 8:287–292. 1996. View Article : Google Scholar
|
|
2
|
Tapper D and Lack EE: Teratomas in infancy
and childhood. A 54-year experience at the Children's Hospital
Medical Center. Ann Surg. 198:398–410. 1983.
|
|
3
|
Tuladhar R, Patole SK and Whitehall JS:
Sacrococcygeal teratoma in the perinatal period. Postgrad Med J.
76:754–759. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Winderl LM and Silverman RK: Prenatal
identification of a completely cystic internal sacrococcygeal
teratoma (type IV). Ultrasound Obstet Gynecol. 9:425–428. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Aoki H, Hara A, Niwa M, Yamada Y and
Kunisada T: In vitro and in vivo differentiation of human embryonic
stem cells into retina-like organs and comparison with that from
mouse pluripotent epiblast stem cells. Dev Dyn. 238:2266–2279.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Deveer JA and Browder J: Sacrococcygeal
teratoma. Ann Surg. 105:408–417. 1937. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Murphy JJ, Blair GK and Fraser GC:
Coagulopathy associated with large sacrococcygeal teratomas. J
Pediatr Surg. 27:1308–1310. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Graf JL, Housely HT, Albanese CT, Adzick
NS and Harrison MR: A surprising histological evolution of preterm
sacrococcygeal teratoma. J Pediatr Surg. 33:177–179. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sergi C, Ehemann V, Beedgen B, Linderkamp
O and Otto HF: Huge fetal sacrococcygeal teratoma with a completely
formed eye and intratumoral DNA ploidy heterogeneity. Pediatr Dev
Pathol. 2:50–57. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Claxton S and Fruttiger M: Oxygen modifies
artery differentiation and network morphogenesis in the retinal
vasculature. Dev Dyn. 233:822–828. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhang W, Ito Y, Berlin E, Roberts R and
Berkowitz BA: Role of hypoxia during normal retinal vessel
development and in experimental retinopathy of prematurity. Invest
Ophthalmol Vis Sci. 44:3119–3123. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hara A, Niwa M, Kunisada T, et al:
Embryonic stem cells are capable of generating a neuronal network
in the adult mouse retina. Brain Res. 999:216–221. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hara A, Niwa M, Kumada M, et al:
Intraocular injection of folate antagonist methotrexate induces
neuronal differentiation of embryonic stem cells transplanted in
the adult mouse retina. Brain Res. 1085:33–42. 2006. View Article : Google Scholar
|
|
14
|
Hara A, Taguchi A, Aoki H, et al: Folate
antagonist, methotrexate induces neuronal differentiation of human
embryonic stem cells transplanted into nude mouse retina. Neurosci
Lett. 477:138–143. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sadowski D, Kiel ME, Apicella M, Arriola
AG, Chen CP and McKinnon RD: Teratogenic potential in cultures
optimized for oligodendrocyte development from mouse embryonic stem
cells. Stem Cells Dev. 19:1343–1353. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kooreman NG and Wu JC: Tumorigenicity of
pluripotent stem cells: biological insights from molecular imaging.
J R Soc Interface. 7(Suppl 6): S753–S763. 2010. View Article : Google Scholar : PubMed/NCBI
|