Introduction
Lung cancer is one of the major causes of
cancer-related mortality worldwide. Despite extensive studies
concerned with diagnostic and treatment strategies, the 5-year
survival rate for lung cancer patients is only 10% (1,2).
Na+/K+-ATPase, a sodium pump, has been
hypothesized to represent a new target for the development of
anticancer drugs. Na+/K+-ATPase is
responsible for the regulation of ion homeostasis in mammalian
cells by exporting three Na+ in exchange for two
K+. Na+/K+-ATPase is involved in
cancer progression and is significant in cell adhesion and
signaling (3,4). Cardiac glycosides, which inhibit
Na+/K+-ATPase, have been used for treating
arrhythmia and congestive heart failure for >200 years (5). Previous epidemiological studies have
implied that cardiac glycosides are capable of inhibiting the
growth of cancer cells and decreasing the incidence of certain
types of tumors, including leukemia, lymphoma, kidney tumors and
urinary tumors. Tumor metastasis is one of the main features of
malignant cancer and is the main cause of mortality in cancer
patients (6,7).
Tumors derived from the epithelium comprise 90% of
all human malignant tumors. A developmental regulatory process,
referred to as epithelial-mesenchymal transition (EMT), is
prominently implicated in the ability of transformed epithelial
cells to disseminate and become resistant to apoptosis (8–10).
During EMT, epithelial cells often lose their primary
characteristics and adopt the characteristics of mesenchymal cells
by losing their polarity and increasing invasion capabilities. EMT
may occur in a number of types of epithelial tumors, including
lung, breast, pancreatic and gastric cancer (11–13).
Non-small cell lung cancer (NSCLC) is the predominant type of lung
cancer and accounts for ∼80% of all lung cancer cases (14). The A549 cancer cell line, which has
features of type II alveolus epithelial cells, is a human NSCLC
cell line (15). Anoxia, epidermal
growth factor (EGF) and transforming growth factor (TGF)-β all
induce EMT (16). During EMT, the
expression of N-cadherin and vimentin increases, and the expression
of E-cadherin decreases (17).
Matrix metalloprotease (MMP)-2 and MMP-9 are involved in cancer
cell adhesion, invasion and migration, and have been hypothesized
to represent prognostic biomarkers for lung cancer progression. In
the present study, the effect of the
Na+/K+-ATPase inhibitor, ouabain, on the
migration of A549 cells was detected.
Materials and methods
Drugs and reagents
The following drugs and reagents were used in this
study: Ouabain (Sigma-Aldrich, St. Louis, MO, USA); RPMI-1640
medium (Gibco-BRL, Carlsbad, CA, USA); transwell chambers (Corning
Inc., Corning, NY, USA); mitomycin C (Sigma-Aldrich); trypase
(Gibco-BRL); and rabbit antibodies against MMP-2, MMP-9,
N-cadherin, E-cadherin and vimentin (Proteintech, Chicago, IL,
USA).
Cell lines
The A549 human NSCLC cell line (Institute of Cell
Biology, Chinese Academy of Science, Shanghai, China) was grown in
RPMI-1640 supplemented with 10% fetal bovine serum, 100
μg/ml penicillin and 100 μg/ml streptomycin at 37°C
in a 5% CO2 humidified atmosphere.
Wound healing assay
A549 cells that had been treated with the control
medium or medium containing EGF for 48 h were seeded in 24-well
cell culture plates and cultured to a confluent monolayer. A
pipette tip (10 μl) was used to scratch a wound on the
midline of the culture well, and the cells were pretreated with 10
μg/ml mitomycin C for 1 h at 37°C; the cells were then
washed twice with PBS. Following 15 h of culture in RPMI-1640
supplemented with 2% serum (control) or other stimulants, the
migration of the cells was evaluated by measuring the difference in
the area of the wounds with a Leica DM2500 image analysis system
(Leica, Mannheim, Germany) at 0 and 15 h.
Transwell chamber migration assay
A549 cells (2×104) that had been treated
with the control medium or with medium containing EGF for 48 h were
added to transwell chambers. Serum was added to the bottom wells of
the chambers to induce cell migration. After 15 h, the cells that
had migrated through the membrane were stained with 0.5%
methylrosaniline chloride solution and counted. Cells were counted
in five random fields and expressed as 1% of the average number of
cells/field under a light microscope.
Western blot analysis
Whole cell extracts of the cancer cells were
prepared using a whole cell extraction kit (Beyotime, Nantong,
China). In brief, cells were washed with cold PBS and scraped free
in the presence of lysis buffer (20 mmol/l MOPS, 2 mmol/l EGTA, 5
mmol/l EDTA, 30 mmol/l NaF, 40 mmol/l β-glycerophosphate, 20 mmol/l
sodium pyruvate, 0.5% Triton X-100 and 1 mmol/l sodium
orthovanadate with protease inhibitor cocktail). The lysates were
then put on ice for 30 min prior to centrifugation at 12,000 × g
for 30 min at 4°C. Protein samples were resolved by 10% SDS-PAGE
and electrotransferred to polyvinylidene fluoride (PVDF) membranes
in transfer buffer (20% methanol, 15.6 mM Tris base and 120 mM
glycine), according to standard methods. Following a 2-h incubation
in 5% non-fat dry milk blocking buffer prepared in TBS with 0.1%
Tween-20, the PVDF membranes were incubated for 15 h at 4°C with a
primary antibody diluted to 1:200–1:500 in blocking buffer.
Following three TBST buffer washes for 10 min/time, the membranes
were then incubated with anti-rabbit or anti-mouse secondary
antibodies conjugated to IRDye 700DX or 800CW (1:5,000) for 2 h at
37°C. Protein bands were detected and quantified on a two-color
Odyssey Infrared Imaging System (LI-COR Biosciences, Lincoln, NE,
USA).
Data analysis and statistics
Data are presented as the mean ± SEM. All
experiments were repeated at least three times. Differences were
analyzed with the one-way ANOVA with SPSS 13.0 software (SPSS,
Inc., Chicago, IL, USA). P<0.05 was considered to indicate a
statistically significant difference.
Results
EGF induces morphological changes in A549
cells
Morphological changes were observed in A549 cells
treated with 50 ng/ml EGF; these cells became isolated and certain
pseudopods were stretched out. In addition, the intercellular tight
junctions became loose and the shape of the cells changed from
being cobblestone-like to fibroblast-like in appearance. These
characteristic changes indicated that EGF had induced EMT in the
A549 cells (Fig. 1).
Effect of ouabain on the migration of the
A549 cell line
A wound healing assay was performed to observe the
effect of ouabain on the migration of A549 cells. To exclude the
growth inhibitory effect of ouabain on migration, the concentration
of ouabain used was 25 nM, which did not affect the growth of
cancer cells for 15 h. The effect of 25 nM ouabain on the
expression of PCNA in the A549 cells for 15 h was detected, and it
was identified that ouabain did not affect the expression of PCNA.
The A549 cells incubated in RPMI-1640 medium with or without EGF
(50 ng/ml) for 48 h were used in the experiment. The results of the
wound healing assays are presented in Fig. 2. The area that the cells had
migrated within 15 h (toward the initially scratched midline, from
the border line) was measured. The cells incubated with 50 ng/ml
EGF migrated across an area that was larger than that of the cells
incubated with the control medium, indicating that EGF stimulated
the migration of the A549 cells. The migration ratio was 83% in the
control group and almost 100% in the EGF group. Following 15 h of
treatment, ouabain not only inhibited EGF-enhanced migration, but
also inhibited basal migration of the A549 cells in the absence of
EGF. Ouabain decreased the migration ratio from 83 to 59% in the
absence of EGF and from ∼100 to 82% in the presence of EGF.
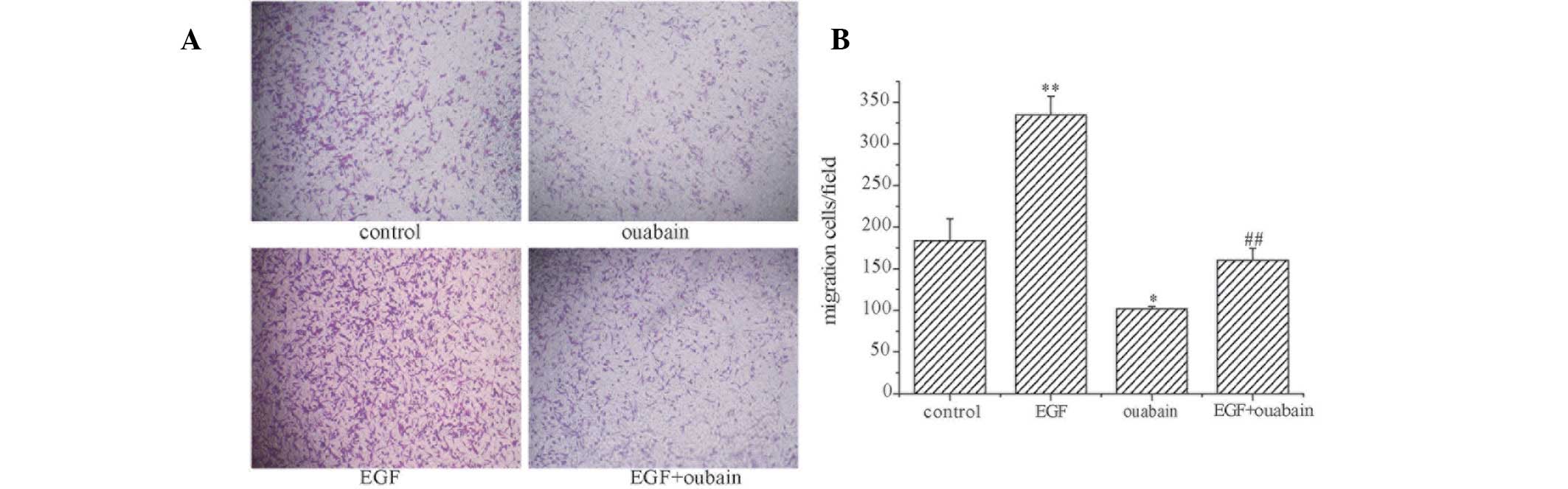
To further investigate the role of ouabain in the
migration of the A549 cells, a transwell chamber migration assay
was performed to detect the effect of ouabain on the number of
migrating cells. The results of the transwell assay were consistent
with those of the wound healing assay (Fig. 3).
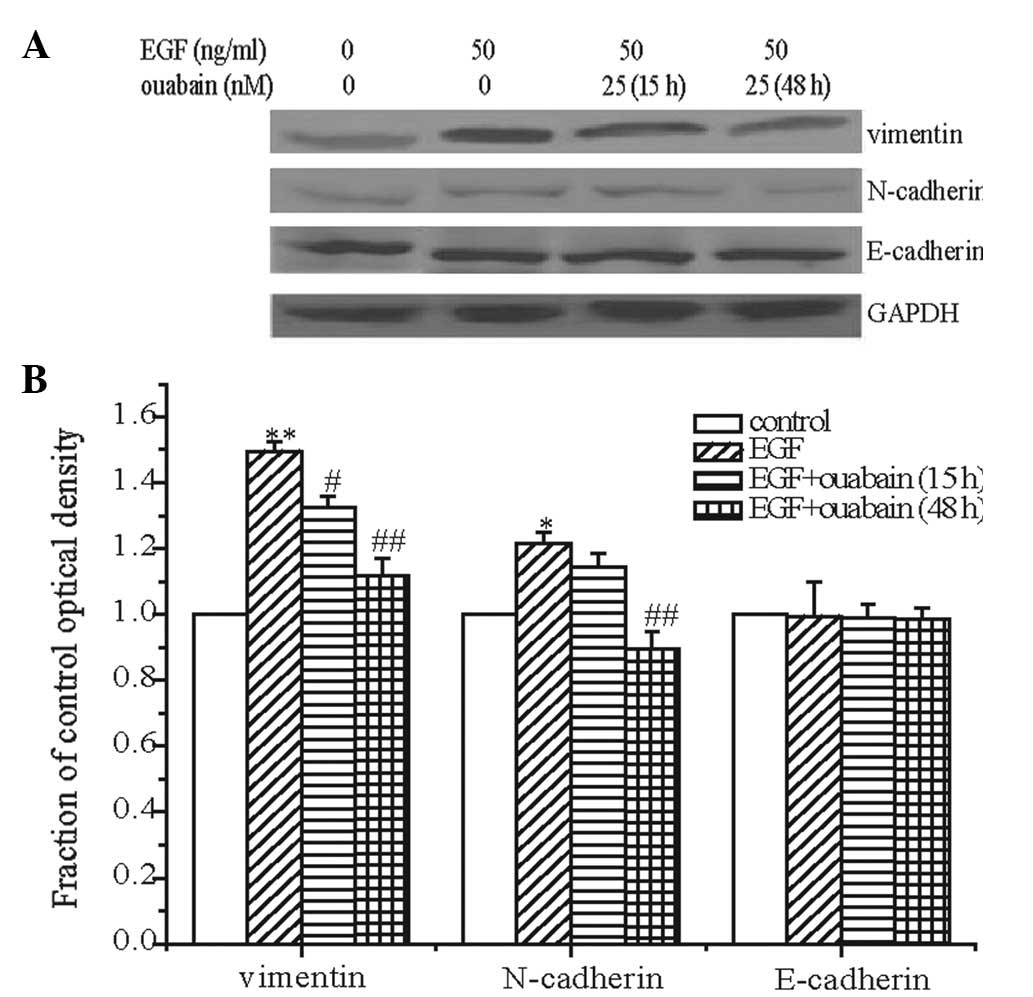
Effect of ouabain on the expression of
E-cadherin, N-cadherin and vimentin
E-cadherin, N-cadherin and vimentin are the main
biomarkers of EMT. In this study, the expression of E-cadherin,
N-cadherin and vimentin was detected following EGF treatment. EGF
significantly increased the expression of vimentin in A549 cells,
marginally (but significantly) increased the expression of
N-cadherin and exerted no effect on the expression of E-cadherin.
The enhancement of vimentin and N-cadherin indicated that EMT
occurred in the A549 cells following EGF treatment. These results
revealed that EGF induced EMT in the A549 cells, and that EMT may
be significant in EGF-induced migration of A549 cells.
Treatment with ouabain for 15 h decreased the
expression of vimentin and had no clear effect on the expression of
N-cadherin. The expression of vimentin and N-cadherin was further
detected following ouabain treatment for 48 h. The results revealed
that ouabain decreased the expression of vimentin and N-cadherin.
Ouabain had no effect on the expression of E-cadherin (Fig. 4).
Effect of ouabain on the expression of
MMP-2 and -9
MMP-2 and -9 degrade the intercellular mesenchyme
and promote tumor migration. Expression of MMP-2 and -9 was
detected following treatment with ouabain for 15 h. The results
demonstrated that ouabain downregulated the expression of MMP-2 and
-9 in the presence or absence of EGF. In addition, the expression
of MMP-2 and -9 was further detected following treatment with
ouabain for 48 h. Similarly, the results revealed that ouabain
decreased the expression of MMP-2 and -9 in the presence or absence
of EGF (Fig. 5).
Discussion
EMT describes a series of rapid changes in the
cellular phenotype. During EMT, epithelial cells downmodulate their
cell-cell adhesion structures, alter their polarity, reorganize
their cytoskeleton, become resistant to apoptosis and acquire the
capacity for differentiation. EMT is tightly regulated under normal
physiological circumstances; however, in tumor tissues, normal
regulation is lost, which results in EMT and provides the cancer
cells with the capacity for migration (18).
Numerous factors, including anoxia, TGF-β, VEGF and
EGF, induce EMT. In the present study, 50 ng/ml EGF was used to
induce EMT in the A549 cells. Following EGF treatment, the
morphology of the A549 cells became similar to that of mesenchyme
cells, the intercellular joints became loose and the cells became
isolated with stretched out pseudopods. Furthermore, the results of
the wound healing and transwell chamber migration assays revealed
that the migration velocity was increased following EGF treatment.
All results demonstrated that EMT occurred following EGF treatment,
and the expression of N-cadherin, E-cadherin and vimentin was
detected. One of the indications of EMT is cadherin transformation,
which is defined by a decrease in E-cadherin and an increase in
N-cadherin expression. Occasionally, E-cadherin expression does not
change and only that of N-cadherin increases (19,20).
E-cadherin is a calcium-dependent transmembrane glycoprotein that
is important for intercellular tight junctions in epithelial cells.
Vimentin is a skelemin protein that is highly expressed in
mesenchymal cells and rarely expressed in epithelial cells.
Vimentin is important for maintaining the structure and function of
interstitial cells (21). In the
present study, following EGF treatment, the expression of
E-cadherin did not change, the expression of N-cadherin marginally
(but significantly) increased and the expression of vimentin
significantly increased. These results indicated that EGF induced
EMT in A549 cells. The effect of ouabain on the expression of
E-cadherin, N-cadherin and vimentin was also detected. The results
revealed that ouabain lowered the expression of N-cadherin, and
vimentin and had no effect on the expression of E-cadherin.
The wound healing and transwell chamber migration
assay results demonstrated that ouabain decreased the migration
velocity of the A549 cells. Uddin et al identified that the
Na+/K+-ATPase inhibitor, marinobufagenin,
inhibited the proliferation, invasion and migration of
cytotrophoblasts via the ERK1/2 signaling pathway (22). Huang et al reported that the
Na+/K+-ATPase inhibitor, ursolic acid,
inhibited the invasion and migration of lung cancer cells by
decreasing the activity of VEGF, MMP and ICAM-1 (23). Therefore, the present study
concluded that the effect of ouabain on A549 cells may have been
due to the inhibition of Na+/K+-ATPase
activity. MMPs, which are secreted from tumor and/or stroma cells,
are involved in tumor invasion and metastasis by degrading the
extracellular matrix surrounding the tumor, particularly the
basement membrane (24). The
majority of previous studies have demonstrated that high protein
expression levels of MMP-2 and -9 correlate with a poorer prognosis
(25). In the present study, it was
indicated that ouabain downregulated the expression of MMP-2 and
-9. Therefore, the effect of ouabain on A549 migration may, in
part, be due to the effect of ouabain on MMP-2 and -9. In
conclusion, this study demonstrates that the
Na+/K+-ATPase inhibitor ouabain retards the
migration of A549 cells. The results suggest that
Na+/K+-ATPase may be a potential therapeutic
target in lung cancer.
References
|
1.
|
Parkin DM, Bray F, Ferlay J and Pisani P:
Global cancer statistics, 2002. CA Cancer J Clin. 55:74–108. 2005.
View Article : Google Scholar
|
|
2.
|
Greenlee RT, Murray T, Bolden S and Wingo
PA: Cancer statistics, 2000. CA Cancer J Clin. 50:7–33. 2000.
View Article : Google Scholar
|
|
3.
|
Yin W, Cheng W, Shen W, et al: Impairment
of Na(+),K(+)-ATPase in CD95(APO-1)-induced human T-cell leukemia
cell apoptosis mediated by glutathione depletion and generation of
hydrogen peroxide. Leukemia. 21:1669–1678. 2007.
|
|
4.
|
Einbond LS, Shimizu M, Ma H, et al: Actein
inhibits the Na+-K+-ATPase and enhances the
growth inhibitory effect of digitoxin on human breast cancer cells.
Biochem Biophys Res Commun. 375:608–613. 2008.PubMed/NCBI
|
|
5.
|
Prassas I and Diamandis EP: Novel
therapeutic applications of cardiac glycosides. Nat Rev Drug
Discov. 7:926–935. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
6.
|
Haux J: Digitoxin is a potential
anticancer agent for several types of cancer. Med Hypotheses.
53:543–548. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Xu ZW, Wang FM, Gao MJ, et al: Cardiotonic
steroids attenuate ERK phosphorylation and generate cell cycle
arrest to block human hepatoma cell growth. J Steroid Biochem Mol
Biol. 125:181–191. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Klymkowsky MW and Savagner P:
Epithelial-mesenchymal transition: a cancer researcher’s conceptual
friend and foe. Am J Pathol. 174:1588–1593. 2009.PubMed/NCBI
|
|
9.
|
Peng CW, Liu XL, Liu X and Li Y:
Co-evolution of cancer microenvironment reveals distinctive
patterns of gastric cancer invasion: laboratory evidence and
clinical significance. J Transl Med. 8:1012010. View Article : Google Scholar
|
|
10.
|
Thiery JP, Acloque H, Huang RY and Nieto
MA: Epithelial-mesenchymal transitions in development and disease.
Cell. 139:871–890. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
Espinosa-Neira R, Mejia-Rangel J,
Cortes-Reynosa P and Salazar EP: Linoleic acid induces an EMT-like
process in mammary epithelial cells MCF10A. Int J Biochem Cell
Biol. 43:1782–1791. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Kim MA, Lee HS, Lee HE, Kim JH, Yang HK
and Kim WH: Prognostic importance of epithelial-mesenchymal
transition-related protein expression in gastric carcinoma.
Histopathology. 54:442–451. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Joost S, Almada LL, Rohnalter V, et al:
GLI1 inhibition promotes epithelial-to-mesenchymal transition in
pancreatic cancer cells. Cancer Res. 72:88–99. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Walker S: Updates in non-small cell lung
cancer. Clin J Oncol Nurs. 12:587–596. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15.
|
Shintani Y, Okimura A, Sato K, et al:
Epithelial to mesenchymal transition is a determinant of
sensitivity to chemoradiotherapy in non-small cell lung cancer. Ann
Thorac Surg. 92:1794–1804. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16.
|
Hardy KM, Booth BW, Hendrix MJC, Salomon
DS and Strizzi L: ErbB/EGF signaling and EMT in mammary development
and breast cancer. J Mammary Gland Biol Neoplasia. 15:191–199.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
17.
|
Wu K and Bonavida B: The activated
NF-kappaB-Snail-RKIP circuitry in cancer regulates both the
metastatic cascade and resistance to apoptosis by cytotoxic drugs.
Crit Rev Immunol. 29:241–254. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18.
|
Kalluri R and Weinberg RA: The basics of
epithelial-mesenchymal transition. J Clin Invest. 119:1420–1428.
2009. View
Article : Google Scholar : PubMed/NCBI
|
|
19.
|
Mariotti A, Perotti A, Sessa C and Rüegg
C: N-cadherin as a therapeutic target in cancer. Expert Opin
Investig Drugs. 16:451–465. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Wheelock MJ, Shintani Y, Maeda M, Fukumoto
Y and Johnson KR: Cadherin switching. J Cell Sci. 121:727–735.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
21.
|
Kokkinos MI, Wafai R, Wong MK, Newgreen
DF, Thompson EW and Waltham M: Vimentin and epithelial-mesenchymal
transition in human breast cancer - observations in vitro and in
vivo. Cells Tissues Organs. 185:191–203. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22.
|
Uddin MN, Horvat D, Glaser SS, et al:
Marinobufagenin inhibits proliferation and migration of
cytotrophoblast and CHO cells. Placenta. 29:266–273. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
23.
|
Huang CY, Lin CY, Tsai CW and Yin MC:
Inhibition of cell proliferation, invasion and migration by ursolic
acid in human lung cancer cell lines. Toxicol In Vitro.
25:1274–1280. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24.
|
Hua H, Li M, Luo T, Yin Y and Jiang Y:
Matrix metalloproteinases in tumorigenesis: an evolving paradigm.
Cell Mol Life Sci. 68:3853–3868. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25.
|
Lin CY, Tsai PH, Kandaswami CC, et al:
Matrix metalloproteinase-9 cooperates with transcription factor
Snail to induce epithelial-mesenchymal transition. Cancer Sci.
102:815–827. 2011. View Article : Google Scholar : PubMed/NCBI
|