Introduction
The prognosis of patients with ovarian cancer is
generally poor owing to an advanced FIGO stage at diagnosis and
often inefficient primary debulking surgery, the most important
prognostic factors in ovarian cancer (1). The presence of a residual tumor is
essentially the only factor that can be affected by the
gynecological oncologist (2).
In ovarian cancer, cutting-edge therapy consists of
radical debulking surgery that aims for complete tumor resection,
followed by chemotherapy. Despite advancements in surgical
techniques, complete debulking (no macroscopically evident residual
tumor mass) is only achieved in ~30% of ovarian carcinoma patients,
and radical tumor resection may cause considerable morbidity
(3). High morbidity is certainly
acceptable if complete debulking with its attendant survival
advantage can be achieved, but it must be viewed critically for
patients left with a residual intra-abdominal tumor and a resulting
shorter life span (4).
Such patients would be ideal candidates for
alternative therapeutic approaches, such as neoadjuvant
chemotherapy with or without subsequent surgery. However,
identification of these patients is difficult, as no standardized
selection criteria have been put forward so far. Neither
radiological imaging nor a variety of analyzed protein and gene
expression parameters have been shown to predict surgical success
accurately (5–7).
As reported previously (8), in order to predict surgical success,
we have developed the OVSCORE algorithm on the basis of a logistic
regression model involving two clinical factors and two tumor
biological factors, namely ascitic fluid volume and nuclear
grading, and kallikrein-related peptidases (KLKs), KLK6 and KLK13,
which are members of a serine protease family and are known to be
associated with ovarian cancer progression and metastasis (9,10). In
our previous trial, employing the OVSCORE, the presence of a
residual tumor mass following radical surgery could be estimated,
with good in-sample predictive performance [area under the receiver
operating characteristic curve (ROC AUC), 0.833] (8).
Other than being markers in the OVSCORE algorithm to
predict surgical success, the majority of members of the KLK-family
have been reported to be significant ovarian cancer biomarkers
(9,10). For example, high KLK5 and KLK6 tumor
tissue levels are associated with an advanced disease stage and
significantly shorter progression-free survival (PFS) and overall
survival (OS) times (11–14). Recently, we found that ovarian
cancer patients with high KLK7 tumor tissue ELISA-levels had a
two-fold lower risk of mortality or relapse compared with patients
who displayed low levels (15).
Finally, KLK13 expression in ovarian cancer tumor tissue has been
shown to correlate with early-stage disease and favorable OS
(16). These properties motivated
the evaluation of the clinical impact of the biomarkers KLK5–7 and
KLK13 in the present study.
The present study reports a retrospective validation
of OVSCORE in an independent data set comprised of 87 patients with
ovarian cancer, focusing in particular on its accuracy in
predicting the presence of a residual tumor following primary
surgery. Furthermore, the prognostic impact of established clinical
factors and the analyzed KLK5–7 and KLK13 concentrations in tumor
tissue was also analyzed in this independent collective.
Patients and methods
Patients
A total of 87 patients with ovarian cancer of
International Federation of Gynecology and Obstetrics (FIGO) stage
I–IV who were treated between 1995 and 2008 at the Department of
Obstetrics and Gynecology, Klinikum rechts der Isar, Technical
University of Munich (Munich, Germany), were enrolled in the
present retrospective biomarker study. Standard surgical procedures
were performed including pelvic and para-aortic lymphadenectomy and
if indicated, partial resection of the small and large intestine,
peritonectomy and upper abdominal surgery. All patients provided
written informed consent and the study was approved by the ethics
committee of Klinikum rechts der Isar, Technical University of
Munich. Following surgery, all patients received adjuvant
treatment, including platinum-based chemotherapy (carboplatin AUC5
and Paclitaxel, 175 mg/sqm, body surface area, every three weeks),
according to consensus recommendations at that time. The median age
of the patients at the time of surgery was 58 years (range, 25–83
years). The median time of follow-up was 50 months (range, 1–166)
for OS and 28 months (range, 1–159) for PFS. The clinical and
histomorphological factors are presented in Table I. In total, 69 tumors (79.3%) were
of the serous histotype, 9 (10.3%) were endometrioid, 4 (4.6%) were
mucinous and 5 (5.7%) belonged to other histotypes.
 | Table IPatient characteristics. |
Table I
Patient characteristics.
| Clinicopathological
parameters | n (%) |
|---|
| FIGO stage |
| I | 11 (12.6) |
| II | 4 (4.6) |
| III | 50 (57.5) |
| IV | 22 (25.3) |
| Lymph node
status |
| N0 | 27 (31.1) |
| N+ | 41 (47.1) |
| Not known | 19 (21.8) |
| Residual tumor,
mm |
| 0 | 38 (43.7) |
| >0 | 49 (56.3) |
| Ascitic fluid volume,
ml |
| None | 20 (23.0) |
| ≤500 | 22 (25.3) |
| >500 | 45 (51.7) |
| Nuclear grading |
| G1 | 3 (3.4) |
| G2 | 17 (19.5) |
| G3 | 67 (77.0) |
| Response to
chemotherapy |
| Progress | 12 (13.8) |
| No change | 3 (3.4) |
| Complete
remission | 25 (28.7) |
| Partial
remission | 5 (5.7) |
| Not known | 42 (48.3) |
| Deceased |
| No | 25 (28.7) |
| Yes | 59 (67.8) |
| Not known | 3 (3.4) |
| Relapsed |
| No | 17 (19.5) |
| Yes | 62 (71.3) |
| Not known | 8 (9.2) |
Preparation of ovarian cancer tumor
tissue extracts
Extracts from ovarian cancer primary tumor tissues
were prepared as described previously (17). Tumor tissue samples were collected
during surgery, inspected and classified by a pathologist, and then
stored in liquid nitrogen. For extraction, deep-frozen specimens of
200–500 mg weight were pulverized and then resuspended in
Tris-buffered saline [TBS; 0.02 M Tris-HCl and 0.125 M NaCl (pH
8.5)] containing 1% (w/v) of the non-ionic detergent Triton X-100
(Sigma Aldrich, Munich, Germany). Subsequent to extraction and
ultracentrifugation at 100,000 × g for 45 min, the supernatant was
collected, aliquoted and stored in liquid nitrogen until further
use.
ELISA tests for the quantitation of
KLK5–7 and KLK13 in ovarian cancer tumor tissue extracts
KLK5–7 and KLK13 antigen concentrations were
determined in ovarian cancer tumor tissue extracts by
non-commercial in-house ELISA test formats (18). For this purpose, monoclonal capture
antibodies and detection antibodies to KLK5–7 and KLK13 proteins
were generated by immunizing mice with recombinant human KLK5–7 and
KLK13. Lower detection limits of the various KLK ELISAs were 0.05
ng/ml for KLK5, 6 and 13, and 0.2 ng/ml for KLK7. In these ELISA
formats, no cross-reactivities with any other member of the human
KLK family were detected. KLK antigen values were expressed as ng
analyte/mg protein, which was determined in the tissue extracts by
the Pierce BCA method (8).
Statistical analyses
All outcome and explanatory variables were coded in
a manner consistent with our previous study (8). Outcome variables were PFS, OS and
residual tumor presence (RT); RT was defined as 1 if a macroscopic
residual tumor mass was visible and zero if completely absent.
Ascites volume, age, nuclear grade and nodal status
were coded as binary variables: Ascites, >500 vs. ≤500 ml; age,
>60 vs. ≤60 years; nuclear grade, G3 vs. G1/G2; and nodal
status, 0 for N0, otherwise 1. FIGO status was coded by three
binary indicators: Stage II/III/IV vs. I; stage III/IV vs. I/II;
and stage IV vs. I/II/III.
For validation of the OVSCORE, ‘virtual’ fractional
ranks of KLK6 and KLK13 were coded with respect to the
distributions of antigens in the original data set (8), using interpolation as required.
The OVSCORE was then computed for each patient by
multiplying the coded values of the factors by the logarithm of the
corresponding odds coefficient (8).
The procedure can be summarized by the following formula: OVSCORE =
2.57 × (A) + 1.07 × (G) + 2.14 × (rKLK6) − 2.00 × (rKLK13) − 5.41,
where ‘A’ is defined as one if ascites >500 ml and zero
otherwise, ‘G’ is one if grade is G3 and zero otherwise, ‘rKLK6’ is
the virtual fractional rank of KLK6 and ‘rKLK13’ is the virtual
fractional rank of KLK6. The fractional rank coding scheme for
validation is intended to ensure that equal values of OVSCORE in
the two independent cohorts correspond as closely as possible to
the same tumor biological interpretation, even if the disease stage
distributions in the two cohorts differ substantially.
The OVSCORE was used to construct a ROC for
prediction of surgical success (RT) in the current validation
sample. The ROC AUC was calculated as an out-of-sample performance
indicator for validation. The out-of-sample positive predictive
value (PPV) and negative predictive value (NPV) are also reported
for the median and scaled cutoffs; the scaled cutoff is defined for
this statistic as the fractional rank of OVSCORE equal to the
percentage of patients with an RT of zero.
Spearman correlations between continuous variables
and Mann-Whitney or Kruskal-Wallis tests for associations between
continuous and categorical variables were computed. Impacts on PFS
and OS were estimated by Cox’s proportional hazards regression
model using forward selection, and then expressed as hazard ratios
(HR) with respect to the defined coding.
Analyses were performed using SPSS software (SPSS,
Inc., Chicago, IL, USA). P<0.05 was used to indicate a
statistically significant difference.
Results
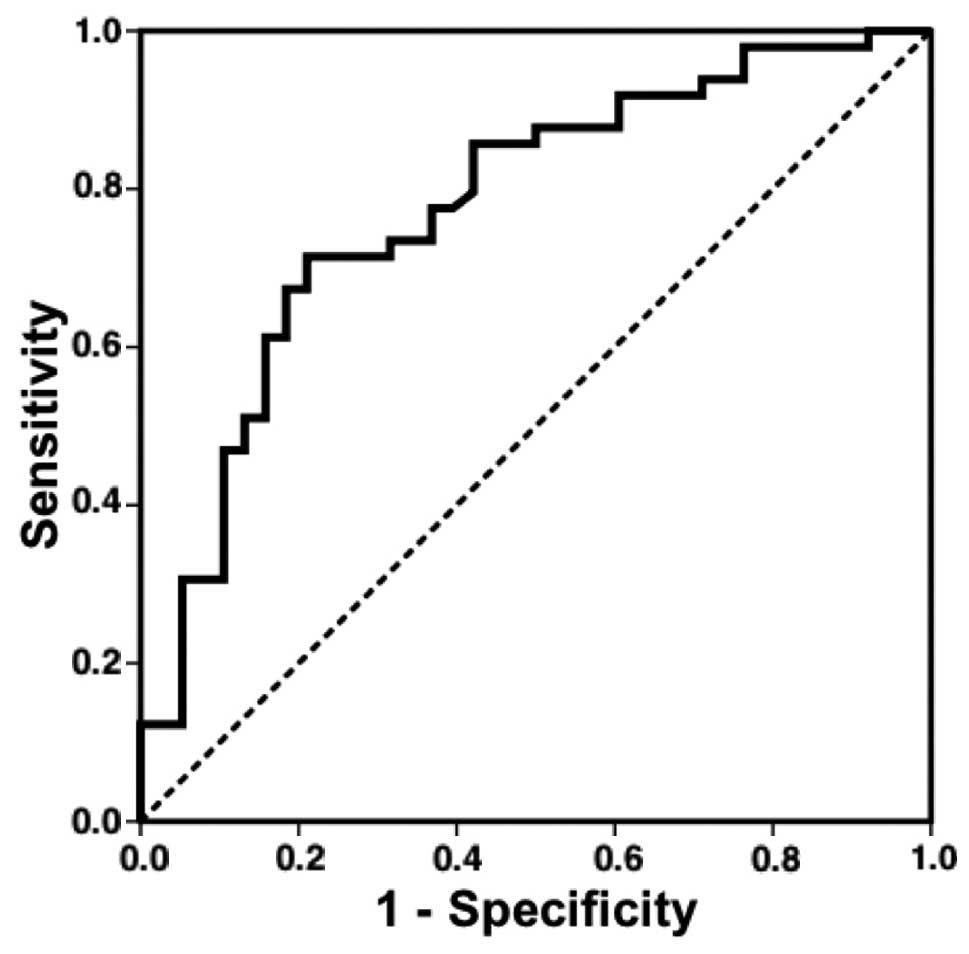
Validation of the OVSCORE algorithm to
predict surgical success in ovarian cancer patients
Based on our previous study (8), the OVSCORE algorithm was developed to
predict surgical success in ovarian cancer patients undergoing
intra-abdominal debulking surgery in order to reduce the tumor
size. The score, encompassing the clinical factors of ascitic fluid
volume and nuclear grading, plus the cancer biomarkers KLK6 and
KLK13, is designed to predict the efficiency of the intra-abdominal
debulking procedure. In the present study, the predictive
performance of the OVSCORE algorithm was tested in an independent
set of 87 primary ovarian cancer patients. The predictive
performance was comparable to that obtained in the original cohort
of 142 ovarian cancer patients: AUC ROC for pilot study, 0.833; AUC
ROC this study, 0.777 (Fig. 1). The
corresponding out-of-sample quality measures for prediction of RT
in terms of sensitivity, specificity, PPV and NPV for median and
scaled cutoffs are summarized in Table
II. Using the median cutoff, for example, one can expect that
an unfavorable (positive) OVSCORE test would be correct in
predicting a residual tumor in ~81% of cases; a favorable
(negative) OVSCORE test would be correct in predicting surgical
success in ~68% of cases.
 | Table IIOut-of-sample quality measures for
prediction of residual tumor presence. |
Table II
Out-of-sample quality measures for
prediction of residual tumor presence.
| OVSCORE | Sensitivity | Specificity | PPV | NPV |
|---|
| OVSCORE with median
cutoff, % | 71.4 | 78.9 | 81.4 | 68.20 |
| OVSCORE with scaled
cutoff, % | 73.5 | 65.8 | 73.5 | 65.80 |
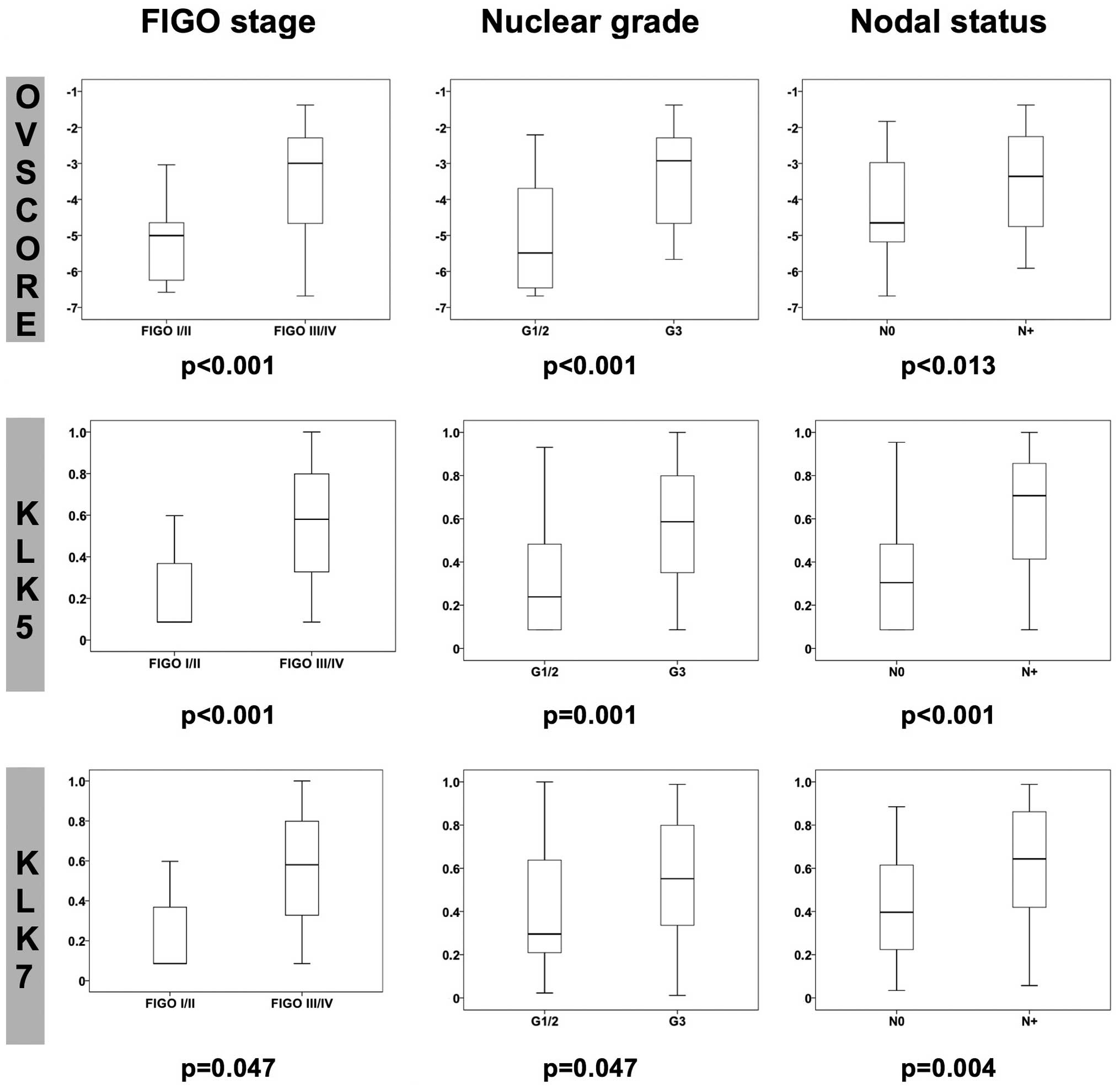
Correlations
Significant Spearman’s rank correlations
(Rs) were found between KLK5 and KLK7 (Rs,
~0.6) and between KLK7 and KLK13 (Rs, ~0.3).
Associations of the clinically established variables of FIGO stage,
nuclear grade, nodal status and ascitic fluid volume, with cancer
biomarkers KLK5–7 KLK13, serum biomarker cancer antigen (CA)-125
and the OVSCORE were quantified employing the Mann-Whitney U test
(Fig. 2): KLK5, KLK7 and the
OVSCORE (as well as CA-125 in serum, data not shown) were
associated with advanced FIGO stage (FIGO III/IV), higher nuclear
grade (G3) and positive nodal status (N+); KLK13 was
associated with nodal status (data not shown), while KLK6 was not
significantly associated with these clinical factors.
Assessment of prognostic impact of
clinical factors, CA-125, KLK5–7 and KLK13
The impact of FIGO stage, nuclear grading, nodal
status, ascitic fluid volume, residual tumor mass, CA-125, KLK5–7
and KLK13 on OS and PFS, as assessed by uni- and multivariable Cox
analysis, are summarized in Table
III. The clinical factors were significant univariate
predictors of OS and PFS. The OVSCORE itself was significantly
associated with OS and PFS in univariate analysis; KLK5–7 and KLK13
were not individually significant for either OS or PFS, whereas
CA-125 was not significant for OS or PFS, the P value for OS was
0.06.
 | Table IIIUnivariate Cox regression analysis to
determine the association of clinical factors and cancer biomarkers
with ovarian cancer patient survival (n=84). |
Table III
Univariate Cox regression analysis to
determine the association of clinical factors and cancer biomarkers
with ovarian cancer patient survival (n=84).
| Overall
survival | Progression-free
survival |
|---|
|
|
|
|---|
| Variable | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| FIGO stage | 3.5 | 1.4–8.9 | 0.007 | 2.8 | 1.2–6.6 | 0.019 |
| Nuclear grade | 3.2 | 1.5–6.7 | 0.003 | 2.6 | 1.3–5.0 | 0.007 |
| Residual tumor
mass, mm | 34.4 | 11.2–105.9 | <0.001 | 1.1 | 1.0–1.1 | <0.001 |
| Residual tumor
mass, mm (0 vs. >0) | 4.5 | 2.5–8.1 | <0.001 | 3.3 | 1.9–5.8 | <0.001 |
| Ascitic fluid
volume, ml | 2.3 | 1.3–3.9 | 0.002 | 2.2 | 1.3–3.7 | 0.003 |
| OVSCOREa | 1.4 | 1.2–1.7 | <0.001 | 1.3 | 1.1–1.6 | 0.001 |
| CA-125 | 1.4 | 1.0–1.9 | 0.060 | | | 0.100 |
The multivariate Cox model for OS contained residual
tumor mass, nuclear grade and KLK7 as independent factors. Adjusted
for residual tumor mass and nuclear grade, KLK7 entered the model
as a protective marker (HR, 0.41; Table IV). Residual tumor mass was the
only independent marker in the model for PFS.
 | Table IVMultivariable Cox regression analysis
to determine the association of clinical factors and cancer
biomarkers with ovarian cancer patient survival (n=84). |
Table IV
Multivariable Cox regression analysis
to determine the association of clinical factors and cancer
biomarkers with ovarian cancer patient survival (n=84).
| Overall
survival | Progression-free
survival |
|---|
|
|
|
|---|
| Variable | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| Residual tumor
mass, mm (0 vs >0) | 4.6 | 2.5–8.6 | <0.001 | 3.31 | 1.90–5.77 | <0.001 |
| Nuclear grade | 2.6 | 1.2–5.8 | 0.016 | - | - | NS |
| KLK7 | 0.4 | 0.2–1.0 | 0.041 | - | - | NS |
Discussion
For ovarian cancer, the major traditional prognostic
factors are FIGO stage at the time of diagnosis and size of
residual tumor mass following cytoreductive surgery (1). Other established clinical prognostic
factors are age, performance status, type of histology, nuclear
grade and presence/amount of peritoneal ascitic fluid. However,
owing to the lack of suitable biomarkers, at present, ovarian
cancer management does not use any prognostic or therapy response
predicting factors, with regard to the course of the disease or a
patient’s risk to develop disease recurrence.
In our previous study, the OVSCORE algorithm was
developed to predict the surgical outcome in the primary debulking
surgery of ovarian cancer patients (8). This score encompasses the clinically
relevant factors of ascitic fluid volume and nuclear grading, plus
two novel cancer biomarkers, the serine proteases KLK6 and
KLK13.
Broad scientific interest has been focused on the
KLKs, since the majority of the fifteen KLK family members are
believed to contribute to ovarian cancer progression and metastasis
(9,10). Four KLKs (KLK4–6 and KLK15) are
linked to the poor prognosis of ovarian cancer patients, while
higher KLK9 and KLK14 levels are associated with a favorable course
of the disease. For certain KLKs (KLK7, 8, 10, 11 and KLK13), the
clinical relevance is not yet clear, since, depending on the method
of detection (mRNA or protein, ELISA or IHC) and/or FIGO stage
(early or advanced), these KLKs can be associated with either a
poorer or more favorable disease course.
The present study validated the clinical relevance
of the multiparametric OVSCORE to predict surgical success in an
independent set of ovarian cancer patients. The performance of the
score in this set was found to be comparable to that in the
original collective of 142 ovarian cancer patients of the pilot
study (ROC AUC pilot study, 0.833; AUC ROC this study, 0.777), even
though complete debulking rates as a whole were higher in the
previous study.
The OVSCORE could easily be calculated prior to
definitive surgery by estimating the pre-operative ascitic fluid
volume by ultrasound and analyzing tumor biopsies obtained by
laparoscopy or CT-guided for nuclear grading and KLK tissue
analysis. Applying the presurgical OVSCORE, ovarian cancer patients
for whom complete tumor resection would be difficult to achieve
could be identified. Such patients could be candidates for
alternative clinical approaches, such as neoadjuvant or exclusive
chemotherapy.
Furthermore, the OVSCORE could also support
pre-operative risk stratification: e.g., in ovarian cancer patients
with substantial co-morbidity, a favorable OVSCORE would reinforce
the decision for radical surgery, whereas an unfavorable OVSCORE
would suggest considering alternative therapeutic approaches, for
instance pre-operative chemotherapy.
Apart from validating the clinical utility of the
OVSCORE, the present study evaluated the clinical impact of the
various single parameters accounting for the OVSCORE algorithm.
Large ascitic fluid volumes and higher nuclear grading are
established clinical prognostic factors known to be associated with
higher stage and inferior survival rates (1,8). As
aforementioned, KLK6 is known to be an unfavorable prognostic
ovarian cancer biomarker (13,14),
while KLK13 expression is correlated with early-stage disease and
favorable OS (16). In view of
recent studies showing that elevated KLK5 and KLK7 values are
associated with advanced stage, higher nuclear grade and a poor
prognosis in ovarian cancer patients (11,12,19–21),
the clinical impact of these two additional KLKs was tested.
In Cox’s univariate analysis, the prognostic impact
on OS and PFS by the established clinical factors of FIGO stage,
residual tumor volume subsequent to surgery, nuclear grade and
ascitic fluid volume, as well as the OVSCORE, could be confirmed,
but none of the KLKs alone or CA-125 in serum showed
significance.
In multivariate Cox analysis, only residual tumor
mass and, for OS, nuclear grade and KLK7 were of clinical
significance. Notably, in this multivariate context, KLK7 was found
to be a protective marker (HR, 0.41). This result is consistent
with our previous findings in an independent ovarian cancer patient
cohort demonstrating that higher KLK7 protein levels, as assessed
by ELISA, are associated with an improved patient outcome (15). However, there are conflicting
studies with regard to KLK7, which state that it may be associated
with a worse prognosis and a poor response to chemotherapy
(8,19,20,22–25).
However, contradictory findings have already been reported for KLK7
in breast cancer. Holzscheiter et al (26) found high full-length KLK7 mRNA
levels in breast cancer tissue to be a favorable prognostic marker,
while Talieri et al (27)
reported KLK7 expression to be associated with shorter survival. A
possible explanation for this discrepancy may be the fact that in
contrast to the cohort of Holzscheiter et al (26), most of the patients analyzed by
Talieri et al (27) were
subjected to adjuvant therapy.
The present ovarian cancer cohort was uniformly
treated by adjuvant platinum-containing polychemotherapy (except if
not indicated or not suitable). By contrast, only 30% of the
patients included in the study by Kyriakopoulou et al
(20) were treated with carboplatin
and only 16% with paclitaxel. Hence, a therapeutic effect analogous
to that hypothesized by Holzscheiter et al (26) is possible. Furthermore, in the
patient cohort analyzed by Shan et al (19), more patients were of earlier stage,
with 30% FIGO stage I/II compared with 17% FIGO stage I/II in the
present cohort, and fewer succumbed; 50% compared with 68% in the
present patient group.
Taken these findings as a whole, KLKs have recently
emerged as novel, promising predictive factors in ovarian cancer.
In particular, the present study has confirmed earlier findings
showing that KLK5 is associated with advanced and more aggressive
disease, and that KLK7 may be a favorable prognostic marker in
ovarian cancer. The key clinical finding of this study is the
validation of the previously developed OVSCORE in an independent
patient cohort. The OVSCORE could aid in the identification of
patients who do not benefit from currently recommended therapeutic
regimens.
Acknowledgements
The OVSCORE trial was designed within the
ECCO-AACR-ASCO workshop, Methods in Clinical Cancer Research in
Flims, Switzerland and was supported by a grant (no. C14–12) from
the Kommission für Klinische Forschung of the Medical Faculty of
the Technical University of Munich, Germany.
References
|
1
|
du Bois A, Reuss A, Pujade-Lauraine E, et
al: Role of surgical outcome as prognostic factor in advanced
epithelial ovarian cancer: a combined exploratory analysis of 3
prospectively randomized phase 3 multicenter trials: by the
Arbeitsgemeinschaft Gynaekologische Onkologie Studiengruppe
Ovarialkarzinom (AGO-OVAR) and the Groupe d’Investigateurs
Nationaux Pour les Etudes des Cancers de l’Ovaire (GINECO). Cancer.
115:1234–1244. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Buys SS, Partridge E, Black A, et al; PLCO
Project Team. Effect of screening on ovarian cancer mortality: the
Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening
Randomized Controlled Trial. JAMA. 305:2295–2303. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Panici BP, Maggioni A, Hacker N, et al:
Systematic aortic and pelvic lymphadenectomy versus resection of
bulky nodes only in optimally debulked advanced ovarian cancer: a
randomized clinical trial. J Natl Cancer Inst. 97:560–566. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Crawford SC, Vasey PA, Paul J, et al: Does
aggressive surgery only benefit patients with less advanced ovarian
cancer? Results from an international comparison within the
SCOTROC-1 Trial. J Clin Oncol. 23:8802–8811. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Borley J, Wilhelm-Benartzi C, Brown R and
Ghaem-Maghami S: Does tumour biology determine surgical success in
the treatment of epithelial ovarian cancer? A systematic literature
review. Br J Cancer. 107:1069–1074. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Dowdy SC, Mullany SA, Brandt KR, et al:
The utility of computed tomography scans in predicting suboptimal
cytoreductive surgery in women with advanced ovarian carcinoma.
Cancer. 101:346–352. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Axtell AE, Lee MH, Bristow RE, et al:
Multi-institutional reciprocal validation study of computed
tomography predictors of suboptimal primary cytoreduction in
patients with advanced ovarian cancer. J Clin Oncol. 25:384–389.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Dorn J, Schmitt M, Kates R, et al: Primary
tumor levels of human tissue kallikreins affect surgical success
and survival in ovarian cancer patients. Clin Cancer Res.
13:1742–1748. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Dorn J, Bayani J, Yousef GM, et al:
Clinical utility of kallikrein-related peptidases (KLK) in
urogenital malignancies. Thromb Haemost. 110:408–422. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Dorn J, Milou V, Kulasingam V, et al:
Clinical relevance of kallikrein-related peptidases in ovarian
cancer. Kallikrein-Related Peptidases. Novel Cancer-Related
Biomarkers. Magdolen V, Sommerhoff CP, Fritz H and Schmitt M: 2.
deGruyter; Berlin: pp. 145–166. 2012
|
|
11
|
Diamandis EP, Borgono CA, Scorilas A, et
al: Immunofluorometric quantification of human kallikrein 5
expression in ovarian cancer cytosols and its association with
unfavorable patient prognosis. Tumour Biol. 24:299–309. 2003.
View Article : Google Scholar
|
|
12
|
Kim H, Scorilas A, Katsaros D, et al:
Human kallikrein gene 5 (KLK5) expression is an indicator of poor
prognosis in ovarian cancer. Br J Cancer. 84:643–650. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hoffman BR, Katsaros D, Scorilas A, et al:
Immunofluorometric quantitation and histochemical localisation of
kallikrein 6 protein in ovarian cancer tissue: a new independent
unfavourable prognostic biomarker. Br J Cancer. 87:763–771. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Seiz L, Dorn J, Kotzsch M, et al: Stromal
cell-associated expression of kallikrein-related peptidase 6 (KLK6)
indicates poor prognosis of ovarian cancer patients. Biol Chem.
393:391–401. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Dorn J, Gkazepis A, Kotzsch M, et al:
Clinical value of protein expression of kallikrein-related
peptidase 7 (KLK7) in ovarian cancer. Biol Chem. 395:95–107. 2014.
View Article : Google Scholar
|
|
16
|
Scorilas A, Borgoño CA, Harbeck N, et al:
Human kallikrein 13 protein in ovarian cancer cytosols: a new
favorable prognostic marker. J Clin Oncol. 22:678–685. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Dorn J, Harbeck N, Kates R, et al: Disease
processes may be reflected by correlations among tissue kallikrein
proteases but not with proteolytic factors uPA and PAI-1 in primary
ovarian carcinoma. Biol Chem. 387:1121–1128. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Christopoulos TK and Diamandis EP:
Enzymatically amplified time-resolved fluorescence immunoassay with
terbium chelates. Anal Chem. 64:342–346. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Shan SJ, Scorilas A, Katsaros D, et al:
Unfavorable prognostic value of human kallikrein 7 quantified by
ELISA in ovarian cancer cytosols. Clin Chem. 52:1879–1886. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kyriakopoulou LG, Yousef GM, Scorilas A,
et al: Prognostic value of quantitatively assessed KLK7 expression
in ovarian cancer. Clin Biochem. 36:135–143. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Dorn J, Magdolen V, Gkazepis A, et al:
Circulating biomarker tissue kallikrein-related peptidase KLK5
impacts ovarian cancer patients’ survival. Ann Oncol. 22:1783–1790.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Psyrri A, Kountourakis P, Scorilas A, et
al: Human tissue kallikrein 7, a novel biomarker for advanced
ovarian carcinoma using a novel in situ quantitative method of
protein expression. Ann Oncol. 19:1271–1277. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Dong Y, Tan OL, Loessner D, et al:
Kallikrein-related peptidase 7 promotes multicellular aggregation
via the alpha(5)beta(1) integrin pathway and paclitaxel
chemoresistance in serous epithelial ovarian carcinoma. Cancer Res.
70:2624–2633. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Oikonomopoulou K, Li L, Zheng Y, et al:
Prediction of ovarian cancer prognosis and response to chemotherapy
by a serum-based multiparametric biomarker panel. Br J Cancer.
99:1103–1113. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Borgono CA and Diamandis EP: The emerging
roles of human tissue kallikreins in cancer. Nat Rev Cancer.
4:876–890. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
26
|
Holzscheiter L, Biermann JC, Kotzsch M, et
al: Quantitative reverse transcription-PCR assay for detection of
mRNA encoding full-length human tissue kallikrein 7: prognostic
relevance of KLK7 mRNA expression in breast cancer. Clin Chem.
52:1070–1079. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Talieri M, Diamandis EP, Gourgiotis D, et
al: Expression analysis of the human kallikrein 7 (KLK7) in breast
tumours: a new potential biomarker for prognosis of breast
carcinoma. Thromb Haemost. 91:180–186. 2004.
|