Introduction
Human cancers constitute a notable burden on
societies. In less developed countries, gastric cancer among males
is one of the leading causes of cancer-associated mortalities
(1). Due to its genetic complexity
and heterogeneity, advances in the treatment of gastric cancer have
been limited thus far (2). Therefore,
the identification of specific biomarkers is crucial for the
development of individualized treatments, which are required for
the effective and precise management of gastric cancer in
patients.
The poly (adenosine diphosphate-ribose) polymerase
(PARP) proteins are a family of 17 enzymes involved in the
regulation of transcription, DNA damage response, genome stability,
cell cycle, energy metabolism, cell death and tumorigenesis
(3–5).
PARP-1 was the first PARP enzyme identified over 50 years ago and
has been the subject of several studies (6–8). PARP-1
binds single- or double-stranded DNA breaks; its activity increases
as required to maintain genomic integrity (9,10). It has
previously been demonstrated that PARP-1 is overexpressed in
numerous types of tumors, including malignant melanomas, colorectal
cancer, breast cancer, testicular tumors and
lymphangioleiomyomatosis, and that it is associated with
invasiveness and poor clinical prognosis (11–15).
Therefore, PARP-1 may be a potential anticancer target (16,17). PARP
inhibitors are also currently used in combination with
chemotherapeutic agents to increase tumor responses (18–20).
PARP-1 single nucleotide polymorphisms, including
PARP-1 2819G, PARP-1 762Val/Ala and PARP-1 rs1136410, were
previously demonstrated to be associated with gastric cancer
susceptibility and lymph node metastasis in gastric cancer
(21–23). Le et al (24) demonstrated that PARP-1 inhibitors
enhance the cytotoxicity of cisplatin in human gastric cancer
cells. Liu et al (25)
previously demonstrated that the cochinchina momordica seed extract
significantly inhibited the survival rate of human gastric cancer
cells by downregulating PARP expression. However, the protein
expression pattern of PARP-1 in gastric cancer patients requires
further study. It remains to be determined whether the expression
levels of PARP-1 are associated with the tumorigenesis and
progression of gastric cancer.
In the present study, tissue microarray-based
immunohistochemistry was used to determine the expression of PARP-1
in 564 gastric cancer tissue samples and 335 tumor-adjacent tissue
samples. The aim of the current study was to analyze the
association between the expression levels of PARP-1 and the
clinicopathological features and prognosis of gastric cancer
patients.
Materials and methods
Patients and tissue samples
Human gastric cancer tissue samples were obtained
from 564 patients (405 males and 159 females; age range, 29–82
years) with primary gastric tumors who underwent D1 or D2 radical
gastrectomy surgery at the First Affiliated Hospital of Dalian
Medical University (Dalian, China) between 2011 and 2013. The
gastric tissues outside the cancer loci were selected as the
tumor-adjacent tissue samples; 335 tumor-adjacent tissue samples
from these patients were collected as controls. The diagnosis of
gastric cancer was confirmed by pathological staining.
Clinicopathological data including patient age, gender, tumor
location, tumor size, histological differentiation, invasion depth,
Helicobacter pylori (H. pylori) infection, ascites,
lymphatic invasion, lymph node metastasis, distant metastasis and
tumor-node-metastasis (TNM) stage (26) were retrospectively retrieved from the
medical records. The patients did not undergo radiotherapy or
chemotherapy prior to surgery. Outcomes of interest included the
overall survival (OS) and disease-free survival (DFS) rates. OS was
defined as the duration from diagnosis to the last follow-up, or to
mortality. DFS was calculated as the time from the initial
diagnosis to local recurrence, or distant metastasis.
The patients consented to have tissue samples
collected at the time of admission for surgery, according to
protocols authorized by the Regional Human Ethics Committee of
Dalian Medical University. The Medical Ethics Committee of Dalian
Medical University approved the present study. Due to the
retrospective nature of the present study, the Ethics Committees
waived the requirement for written informed consent from the
patients.
Tissue microarray (TMA)-based
immunohistochemistry
Tissue cores were extracted from formalin-fixed and
paraffin-embedded tissue blocks containing the tumor tissue samples
and the tumor-adjacent tissue samples and arrayed into a paraffin
recipient block. Sections (4 µm thick) were obtained from the TMA
blocks using a microtome, mounted on poly-L-lysine-coated glass
slides and used for immunohistochemistry.
TMA sections were washed in xylene, rehydrated in a
graded ethanol series and washed in tap water. The tissue sections
were then heated in 100 ml 10 mM sodium citrate buffer (pH 6.0) in
a microwave oven (high power, 700 W for 3 min; medium power, 400 W
for 3 min; low power, 100 W for 3 min; a total of 9 min) to
retrieve antigen. The sections were then incubated at 37°C for 30
min in 3% H2O2/methanol to block endogenous
peroxidase activity. Nonspecific protein binding sites were blocked
by 10% normal goat serum (Boster Biological Technology, Ltd.,
Wuhan, China) at 37°C for 30 min. The sections were incubated in a
primary polyclonal rabbit anti-human antibody against PARP-1 (cat.
ab6079; dilution 1:200; Abcam, Cambridge, UK) overnight at 4°C, and
subsequently incubated with a biotinylated goat anti-rabbit
secondary antibody (cat. no. SAEP031; dilution, 1:150; Wuhan
Elabscience Biotechnology Co., Ltd., Wuhan, China) for 30 min and
streptavidin horseradish peroxidase (LSAB kit; Dako, Glostrup,
Denmark) for an additional 30 min. Sections were stained with
3,3-diaminobenzidine at room temperature for 50–60 sec,
counterstained with hematoxylin, dehydrated with graded ethanol and
mounted with neutral resin. For the negative controls, the primary
antibodies were replaced with phosphate-buffered saline.
Evaluation of
immunohistochemistry
Two pathologists blinded to the experimental
conditions examined the final effective immunostaining under a
light microscope (Eclipse 50i; Nikon Corporation, Tokyo, Japan).
The intensity of immunoreactivity was graded on a scale of 0–3, as
follows: 0) For no visible staining, i) for low staining, ii) for
moderate staining and iii) for high staining. The percentage of the
stained nuclei in gastric cancer gland cells and normal gland cells
was assigned using 5% increments. Five random, non-overlapping
fields were defined and 40 cells for each field (a total of 200
cells) were selected in order to calculate the percentage of
stained cells present in each sample. The immunoreactivity score
(IRS) was determined by multiplying the intensity score and the
percentage of stained nuclei, giving a minimum IRS score of 0 and a
maximum of 300%. Receiver operating characteristic (ROC) curve
analysis was performed to determine an optimal cutoff IRS for
PARP-1 expression. Plotting the sensitivity and specificity for
each outcome under study generated the ROC curves.
Statistical analysis
The Pearson's χ2 test and the Fisher's
exact probability test were used to determine significant
differences between the categorical data. The Mann-Whitney U test
was used to detect differences in the IRS or lymph node metastasis
number between various groups. The Wilcoxon rank-sum test was used
to compare the IRS of gastric cancer tissues with tumor-adjacent
tissues, for the paired tissue samples. Kaplan-Meier survival plots
were generated and comparisons between the survival curves were
determined with a log-rank test. Cox's proportional hazards
regression model was used to evaluate the association between the
potential confounding variables and the prognosis (OS or DFS). Only
those cases with complete data on all the variables (n=153) were
included in the multivariate analyses. The data were processed
using SPSS 16.0 software (SPSS, Inc., Chicago, IL, USA). P<0.05
was considered to indicate statistical significance.
Results
Clinicopathological characteristics of
patients with gastric cancer
The clinicopathological characteristics of 564
patients with gastric cancer are summarized in Table I. For these 564 patients, the features
of tumor location of 498 patients (88.3%), tumor size of 522
patients (92.6%), histological differentiation of 555 patients
(98.4%), depth of tumor invasion of 529 patients (93.8%), status of
H. pylori infection of 297 patients (52.7%), presence of
ascites of 479 patients (84.9%), lymphatic invasion of 540 patients
(95.7%), lymph node metastasis of 553 patients (98.0%), distant
metastasis of 434 patients (77.0%) and TNM stage of 481 patients
(85.3%) were recorded. The average age (mean ± standard deviation)
of gastric cancer patients in the present study was 60.1±10.4 years
(range, 29–82 years). The histological differentiation of the
cancers was determined in 555 patients as follows: 14.6% high
differentiation (n=81), 21.4% moderate differentiation, (n=119) and
64.0% low differentiation (n=355). The depth of tumor invasion was
evaluated in 529 patients as follows: 10.8% T1 (n=57, tumor invades
the mucosa or submucosa), 15.9% T2 (n=84, tumor invades the
muscularis propria), 64.1% T3 (n=339, tumor invasion of the
serosa), and 9.3% T4 (n=49, tumor invades the adjacent organs and
structures). The status of H. pylori infection was defined
by a 13C-urea breath test. Of the total group, 208
patients (70.0%) were diagnosed with H. pylori infection while 89
patients (30.0%) were negative for H. pylori infection. The
stage of the cancer was evaluated in 481 patients according to the
TNM staging system as follows: 13.7% Stage I (n=66), 24.9% stage II
(n=120), 35.3% stage III (n=170) and 26.0% stage IV (n=125).
Ascites, lymphatic invasion, lymph node metastasis and distant
metastasis occurred in 176 (36.7%) of 479 patients, 203 (37.6%) of
540 patients, 316 (57.1%) of 553 patients and 148 (34.1%) of 434
patients, respectively.
 | Table I.Clinicopathological characteristics
of patients with gastric cancer. |
Table I.
Clinicopathological characteristics
of patients with gastric cancer.
| Features | Total | Number | % |
|---|
| Age (years) | 564 |
|
|
|
≤60 |
| 296 | 52.5 |
|
>60 |
| 268 | 47.5 |
| Gender | 564 |
|
|
|
Male |
| 405 | 71.8 |
|
Female |
| 159 | 28.2 |
| Tumor location | 498 |
|
|
|
Lower |
| 257 | 51.6 |
|
Middle |
| 199 | 40.0 |
|
Upper |
| 42 | 8.4 |
| Tumor size
(cm) | 522 |
|
|
|
≤5.0 |
| 240 | 46.0 |
|
>5.0 |
| 282 | 54.0 |
| Histological
differentiation | 555 |
|
|
|
High |
| 81 | 14.6 |
|
Moderate |
| 119 | 21.4 |
|
Low |
| 355 | 64.0 |
| Invasion depth | 529 |
|
|
| T1 |
| 57 | 10.8 |
| T2 |
| 84 | 15.9 |
| T3 |
| 339 | 64.1 |
| T4 |
| 49 | 9.3 |
| H. pylori
infection | 297 |
|
|
|
Negative |
| 89 | 30.0 |
|
Positive |
| 208 | 70.0 |
| Ascites | 479 |
|
|
|
Negative |
| 303 | 63.3 |
|
Positive |
| 176 | 36.7 |
| Lymphatic
invasion | 540 |
|
|
|
Negative |
| 337 | 62.4 |
|
Positive |
| 203 | 37.6 |
| Lymph node
metastasis | 553 |
|
|
|
Negative |
| 237 | 42.9 |
|
Positive |
| 316 | 57.1 |
| Distant
metastasis | 434 |
|
|
|
Negative |
| 286 | 65.9 |
|
Positive |
| 148 | 34.1 |
| TNM stage | 481 |
|
|
| I |
| 66 | 13.7 |
| II |
| 120 | 24.9 |
|
III |
| 170 | 35.3 |
| IV |
| 125 | 26.0 |
Follow-up information was available for 523 patients
with gastric cancer. During the follow-up period of 0–95 months,
relapses occurred in 246 cases and mortality occurred in 208 cases.
The 5-year survival rate was determined to be 46.5%. The mean OS
time was 65.8 months (95% confidence interval (CI); 62.6–68.9
months) and the mean DFS time was 60.5 months (95% CI; 57.3–63.7
months) respectively.
PARP-1 overexpression in breast
cancer
Using immunohistochemistry, the expression levels of
PARP-1 in 564 gastric cancer tissue samples and 335 tumor-adjacent
control tissue samples were studied. Positive PARP-1 staining in
gastric tissues appeared as brown particles that were primarily
nuclear (Fig. 1). The gastric cancer
tissue samples exhibited significantly more intense staining for
PARP-1 expression, as compared with tumor-adjacent tissue samples
(P<0.001; Fig. 2A). There was also
a significant increase in the IRS of PARP-1 expression among paired
samples (P<0.001; Fig. 2B).
The ROC analysis was performed to determine an
optimal cutoff score for PARP-1 expression; the ROC curves for the
clinicopathological features with statistical significance are
indicated in Fig. 3. The area under
the curve for the lymph node metastasis status had the largest
relative area (Fig. 3A). Based on
this outcome, a cutoff score of 175% was selected for PARP-1
expression levels: The low and high expression levels of PARP-1
were defined as IRS≤175% and IRS>175%, respectively. A total of
298 (52.8%) tumors exhibited low expression levels of PARP-1 and
266 (47.2%) tumors exhibited high expression levels of PARP-1.
Association of PARP-1 expression with
clinicopathological characteristics of gastric cancer
The association between PARP-1 expression levels and
the clinicopathological characteristics of gastric cancer were
investigated (Table II). Statistical
analysis indicated that PARP-1 expression levels were increased
with the following: Decreased differentiation (P<0.001),
increased depth of invasion (P=0.037), presence of lymphatic
invasion (P<0.001), presence of lymph node metastasis
(P<0.001) and advanced TNM stage (P=0.015). In addition, high
PARP-1 expression levels were increased among patients with H.
pylori infection (P=0.032). The expression levels of PARP-1
were not identified to be significantly associated with age,
gender, tumor location, tumor size, distal metastasis or presence
of ascites in the patients (P>0.05).
 | Table II.Association of the expression of
PARP-1 with clinicopathological features of gastric cancer. |
Table II.
Association of the expression of
PARP-1 with clinicopathological features of gastric cancer.
|
| High
expression |
|
|---|
|
|
|
|
|---|
| Features | n | % |
P-valuea |
|---|
| Age at diagnosis
(years) |
|
| 0.105 |
|
≤60 | 130 | 43.9 |
|
|
>60 | 136 | 50.7 |
|
| Gender |
|
| 0.575 |
|
Male | 194 | 47.9 |
|
|
Female | 72 | 45.3 |
|
| Tumor location |
|
| 0.057 |
|
Lower | 111 | 43.2 |
|
|
Middle | 104 | 52.3 |
|
|
Upper | 15 | 35.7 |
|
| Tumor size
(cm) |
|
| 0.123 |
|
≤5.0 | 108 | 45.0 |
|
|
>5.0 | 146 | 51.8 |
|
| Histologic
differentiation |
|
|
<0.001b |
|
High | 18 | 22.2 |
|
|
Moderate | 53 | 44.5 |
|
|
Low | 190 | 53.5 |
|
| Invasion depth |
|
| 0.037b |
| T1 | 20 | 35.1 |
|
| T2 | 36 | 42.9 |
|
| T3 | 163 | 48.1 |
|
| T4 | 31 | 63.3 |
|
| H.
pylori |
|
| 0.032b |
|
Negative | 38 | 42.7 |
|
|
Positive | 117 | 56.2 |
|
| Ascites |
|
| 0.253 |
|
Negative | 142 | 46.9 |
|
|
Positive | 73 | 41.5 |
|
| Lymphatic
invasion |
|
|
<0.001b |
|
Negative | 136 | 40.4 |
|
|
Positive | 118 | 58.1 |
|
| Lymph node
metastasis |
|
|
<0.001b |
|
Negative | 84 | 35.4 |
|
|
Positive | 180 | 57.0 |
|
| Distant
metastasis |
|
| 0.263 |
|
Negative | 123 | 43.0 |
|
|
Positive | 72 | 48.6 |
|
| TNM stage |
|
| 0.015b |
|
I~II | 76 | 40.9 |
|
|
III~IV | 154 | 52.2 |
|
The association between PARP-1 expression levels and
lymph node metastasis in gastric cancer patients was also
investigated (Fig. 4). The numbers of
lymph nodes positive for metastasis were recorded from 553 gastric
cancer patients, and ranged from 0 to 33 lymph nodes among the 553
patients. Compared with PARP-1 low expression levels, PARP-1 high
expression levels were associated with a larger number of positive
lymph nodes (P<0.001; Fig. 4A).
Furthermore, lymph node metastasis-positive tumors exhibited a
higher IRS of PARP-1 expression (P<0.001; Fig. 4B).
Association of PARP-1 expression with
the survival of patients with gastric cancer
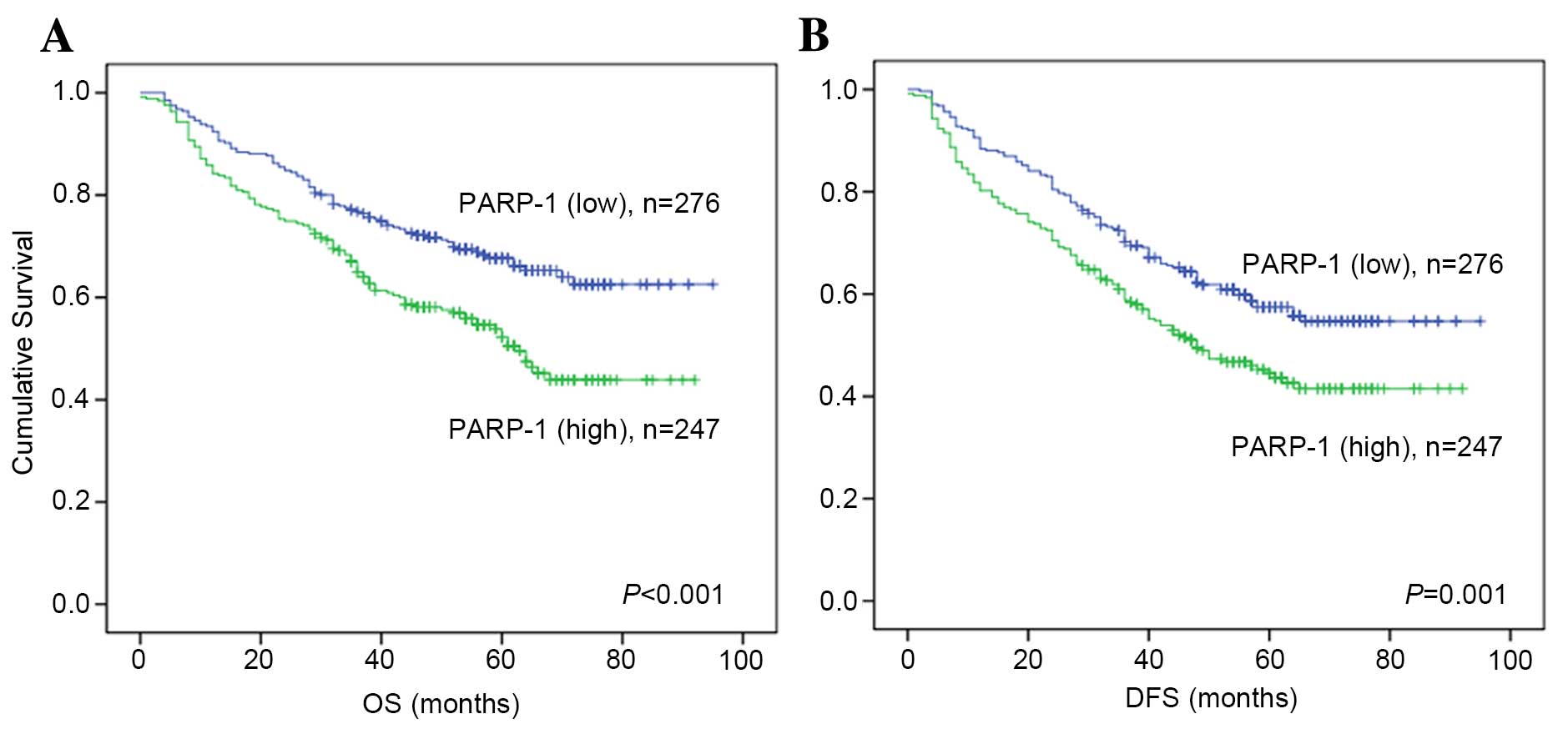
Kaplan-Meier analysis and a log-rank test were used
to evaluate the association between the expression levels of PARP-1
protein and patient mortality. High expression levels of PARP-1
were associated with significantly shorter OS time (P<0.001;
Fig. 5A) and DFS time (P=0.001;
Fig. 5B).
Univariate analysis of the potential prognostic
impact of the clinicopathological parameters identified age,
gender, tumor size, histological differentiation, invasion depth,
presence of ascites, lymph node metastasis, TNM stage and PARP-1
expression as significantly associated with OS and DFS in gastric
cancer patients (Table III).
Subsequently, multivariate Cox regression models (using the same
clinicopathological features) revealed that only histological
differentiation, presence of ascites, lymph node metastasis and TNM
stage remained as independent prognostic factors (Table IV).
 | Table III.Univariate Cox regression analysis of
clinicopathological data associated with OS and DFS in gastric
cancer. |
Table III.
Univariate Cox regression analysis of
clinicopathological data associated with OS and DFS in gastric
cancer.
|
|
| OS | DFS |
|---|
|
|
|
|
|
|---|
| Factors | n | RR (95% CI) | P-value | RR (95% CI) | P-value |
|---|
| Age, years | 523 |
| 0.013a |
| 0.005a |
|
≤60/>60 |
| 1.412
(1.075–1.856) |
| 1.428
(1.111–1.835) |
|
| Gender | 523 |
| 0.017a |
| 0.025a |
|
Female/male |
| 0.673
(0.486–0.932) |
| 0.715
(0.532–0.959) |
|
| Tumor location | 457 |
| 0.732 |
| 0.905 |
|
Upper/middle/lower |
| 1.041
(0.828–1.307) |
| 1.013
(0.822–1.247) |
|
| Tumor size, cm | 484 |
|
<0.001a |
|
<0.001a |
|
>5.0/≤5.0 |
| 1.958
(1.460–2.624) |
| 1.896
(1.453–2.475) |
|
| Histologic
differentiation | 514 |
| 0.018a |
| 0.011a |
|
High/moderate/low |
| 0.779
(0.633–0.959) |
| 0.785
(0.650–0.947) |
|
| Invasion depth | 495 |
| 0.005a |
| 0.003a |
|
T4/T3/T2/T1 |
| 1.317
(1.088–1.596) |
| 1.309
(1.097–1.561) |
|
| H.
pylori | 268 |
| 0.172 |
| 0.048a |
|
Positive/negative |
| 1.341
(0.880–2.045) |
| 1.492
(1.00–2.218) |
|
| Ascites | 439 |
| 0.001a |
|
<0.001a |
|
Yes/no |
| 1.622
(1.221–2.155) |
| 1.623
(1.249–2.107) |
|
| Lymphatic
invasion | 500 |
| 0.290 |
| 0.102 |
|
Yes/no |
| 1.164
(0.879–1.541) |
| 1.240
(0.958–1.605) |
|
| Lymph node
metastasis | 512 |
|
<0.001a |
|
<0.001a |
|
Yes/no |
| 4.991
(3.476–7.165) |
| 4.979
(3.594–6.898) |
|
| Distant
metastasis | 400 |
| 0.263 |
| 0.116 |
|
Yes/no |
| 1.192
(0.876–1.623) |
| 1.257
(0.945–1.671) |
|
| TNM stage | 442 |
|
<0.001a |
|
<0.001a |
|
IV/III/II/I |
| 1.524
(1.309–1.775) |
| 1.493
(1.299–1.715) |
|
| PARP-1
expression | 523 |
|
<0.001a |
| 0.001a |
|
Positive/negative |
| 1.685
(1.280–2.218) |
| 1.507
(1.172–1.936) |
|
 | Table IV.Multivariate Cox regression analysis
of OS and DFS in gastric cancer. |
Table IV.
Multivariate Cox regression analysis
of OS and DFS in gastric cancer.
|
| OS | DFS |
|---|
|
|
|
|
|---|
| Factors | RR (95% CI) | P | RR (95% CI) | P |
|---|
| Age, years
(≤60/>60) | 1.063
(0.676–1.669) | 0.792 | 1.144
(0.753–1.738) | 0.528 |
| Gender
(female/male) | 0.855
(0.516–1.417) | 0.543 | 0.904
(0.568–1.438) | 0.670 |
| Tumor size, cm
(>5.0/≤5.0) | 1.447
(0.838–2.499) | 0.185 | 1.346
(0.815–2.222) | 0.246 |
| Histological
differentiation | 1.435
(1.012–2.033) | 0.042a | 1.376
(1.001–1.891) | 0.049a |
| Invasion depth
(T4/T3/T2/T1) | 1.136
(0.796–1.621) | 0.483 | 1.218
(0.879–1.689) | 0.236 |
| H. pylori
(positive/negative) | 1.260
(0.740–2.144) | 0.395 | 1.366
(0.838–2.225) | 0.211 |
| Ascites
(yes/no) | 1.614
(1.011–2.576) | 0.045a | 1.686
(1.098–2.588 | 0.017a |
| Lymph node
metastasis (yes/no) | 3.432
(1.784–6.603) |
<0.001a | 4.010
(2.204–7.295) |
<0.001a |
| TNM stage
(IV/III/II/I) | 1.411
(1.057–1.883) | 0.019a | 1.402
(1.080–1.821) | 0.011a |
| PARP-1
expression | 1.642
(0.994–2.712) | 0.053 | 1.354
(0.863–2.122) | 0.187 |
Subgroup analysis of the association
between PARP-1 expression levels and the survival of gastric cancer
patients
The present study used Kaplan-Meier analysis to
investigate the association of PARP-1 expression levels with OS and
DFS in subgroups of gastric cancer patients, which were categorized
according to clinicopathological parameters. The results of the
current study indicated that high expression levels of PARP-1 were
associated with shorter OS (P<0.001; Fig. 6A) and DFS (P<0.001; Fig. 6B) in the subset of patients with H.
pylori infection. However, in those patients without H.
pylori infection, the expression levels of PARP-1 were not
significantly associated with OS or DFS (OS, P=0338; DFS, P=0.999;
Fig. 6C and D). In addition, high
expression levels of PARP-1 were associated with shorter OS
(P=0.001; Fig. 7A) and DFS (P=0.006;
Fig. 7B) in the subset of patients
with an advanced TNM stage, but not with an early TNM stage (OS,
P=0.834; DFS, P=0.955; Fig. 7C and
D). The impact of PARP-1 expression levels on the prognosis was
not significantly different between the subgroups of gastric cancer
patients categorized according to age, gender, tumor size, tumor
location, invasion depth, histological differentiation, presence of
ascites, distant metastasis, lymphatic invasion or lymph node
metastasis.
Discussion
PARP-1 is the most abundant and best characterized
nuclear enzyme of the PARP superfamily (27). PARP-1 binds single- or double-stranded
DNA breaks in response to stresses and functions to maintain
genomic integrity (10). This role
has been the focus of a number of studies in the field of oncology
(28,29). However, the role of PARP-1 in gastric
cancer tumorigenesis remains to be determined. The present study
investigated PARP-1 expression in gastric cancer for the first
time. The present study has demonstrated that gastric cancer
tissues exhibit significantly higher immunoreactivity of PARP-1,
compared with tumor-adjacent tissues, indicating that PARP-1
overexpression may contribute to gastric cancer malignancy. These
results are consistent with those of previous studies, which
demonstrated that PARP-1 was upregulated in numerous types of tumor
(11,15,30–32).
The present study analyzed the association between
PARP-1 expression levels and various clinicopathological features
in patients with gastric cancer. It was demonstrated that increased
PARP-1 expression levels are associated with increased depth of
invasion, lymphatic invasion, lymph node metastasis and advanced
TNM stage. Furthermore, high PARP-1 expression levels were
associated with a larger number of lymph node metastases,
suggesting that PARP-1 over-expression increased invasion and
metastasis in gastric cancer. Concordant with these results,
Rodríguez et al (33)
previously demonstrated that inhibition of PARP-1 expression
suppressed the invasion and colonization of distal organs in
melanoma cells. In addition, Li et al (34) demonstrated that inhibition of PARP
expression attenuated the adhesion of mouse colon carcinoma cells
to the extracellular matrix and decreased their migration and
invasion through Matrigel, suggesting that PARP-1 is important in
controlling the migration and invasion of certain cancers. PARP-1
has been previously demonstrated to regulate cell invasion and
metastasis through the modulation of epithelial-mesenchymal
transition-induced malignant transformation or the regulation of
the activity of the nuclear factor kappa-light-chain-enhancer of
activated B cells (33–36). Further studies are required to
determine whether a similar mechanism of PARP-1 occurs in gastric
cancer.
The present study demonstrated that increased PARP-1
expression levels were associated with lower histological
differentiation in gastric cancer. This result is concordant with
the previously demonstrated inverse correlation between the degree
of cell differentiation and PARP-1 activity (37). Barboro et al (38) demonstrated that higher PARP expression
levels were detected in a less-differentiated PC3 cell line, as
compared with a more-differentiated LNCaP prostate carcinoma cell
line. It is possible that PARP-1 upregulation is involved in
maintaining the stemness of cells, therefore exhibiting an
association with lower differentiation in certain cancer cells.
Concordant with this hypothesis, Chiou et al (39) previously demonstrated that the
activation of PARP-1 promoted induced pluripotent stem cell
production and helped to maintain a pluripotent state.
PARP-1 expression has been previously demonstrated
to be associated with the poor prognosis of numerous tumor types,
including early breast cancer and non-small cell lung cancer
(13,40–42).
However, Aiad et al (43)
demonstrated that high nuclear PARP-1 expression levels were
significantly associated with improved OS in locally advanced
breast cancer; Klauschen et al (44) demonstrated that low nuclear expression
levels of PARP were associated with a poor prognosis in pancreatic
cancer. These previous studies indicated that PARP-1 expression had
differing roles in between different tumor types and stages of the
tumors. The present study demonstrated that high PARP-1 expression
levels are associated with significantly reduced DFS and OS in
gastric cancer patients. Furthermore, high expression levels of
PARP-1 were demonstrated to be associated with a poor prognosis in
a subset of patients with an advanced TNM stage (III–IV), but not
early TNM stage (I–II). In addition, a univariate Cox regression
analysis identified that high PARP-1 expression levels are
associated with a poor prognosis for gastric cancer patients.
Therefore, PARP-1 expression levels may have a prognostic value in
gastric cancer, particularly for those patients with an advanced
TNM stage. However, a multivariate analysis determined that PARP-1
expression levels were not independent prognostic factors in
gastric cancer, which may be due to a significant association
between PARP-1 overexpression and tumor invasion and metastasis of
gastric cancer.
H. pylori may induce apoptosis of gastric
epithelial cells (45,46), and H. pylori infection is a
risk factor associated with gastric cancer (47,48). Chen
et al (49) previously
demonstrated that incubating BGC-823 gastric cancer cells with
H. pylori extract induced a breakdown of caspase-1 and
caspase-3, but not of PARP. Nossa et al (50) subsequently demonstrated that PARP-1
became activated in H. pylori infected gastric epithelial
cells. Notably, the present study similarly demonstrated that
PARP-1 expression levels were significantly increased in H.
pylori infected gastric cancer cells. Regarding pathogenic
infection, Hassumi-Fukasawa et al (51) demonstrated a significant positive
association between PARP-1 expression levels and human papilloma
virus positivity, in high-grade squamous intraepithelial lesions of
the uterine cervix. Therefore, PARP-1 expression levels may be
involved in host cell responses to pathogen infection. Furthermore,
the current study demonstrated that the expression of PARP-1 is
associated with significantly shorter OS and DFS in gastric cancer
patients with H. pylori infection, but not in patients
without H. pylori infection. These results suggest that the
upregulation or activation of PARP-1 in response to H.
pylori infection may be one mechanism underlying the
association of PARP-1 expression levels and poor prognosis in
gastric cancer patients. Concordant with a previous study (52), the results of the present study
indicate the potential applications of PARP-1 targeted therapy for
treating H. pylori-mediated gastric cancer.
In conclusion, the present study was a novel
investigation into PARP-1 expression patterns in gastric cancer,
and the association between PARP-1 expression levels and the
clinicopathological features and prognosis of gastric cancer
patients. The results of the current study demonstrated that PARP-1
expression levels are significantly higher in gastric cancer
tissues as compared with tumor-adjacent tissues; high PARP-1
expression levels are associated with H. pylori infection,
lower histological differentiation, tumor invasion and metastasis,
and poor prognosis in gastric cancer. Furthermore, it was
demonstrated that high PARP-1 expression levels are associated with
increased mortality in the subgroups of patients with H.
pylori infection and an advanced TNM stage. The results of the
present study suggest that the inhibition of PARP-1 may suppress
tumor invasion and metastasis and improve histological
differentiation and the survival rate in gastric cancer. The
targeting PARP-1 may be an effective therapeutic strategy for the
treatment of gastric cancer, particularly of H.
pylori-positive or advanced-stage gastric cancer.
Acknowledgements
This study was supported by the Science and
Technology Department Foundation of Liaoning Province (grant no.
2013225021).
Glossary
Abbreviations
Abbreviations:
|
AUCs
|
areas under curve
|
|
CI
|
confidence interval
|
|
DFS
|
disease-free survival
|
|
EMT
|
epithelial-mesenchymal transition
|
|
H. pylori
|
Helicobacter pylori
|
|
IRS
|
immunoreactivity score
|
|
OS
|
overall survival
|
|
PARP-1
|
poly (adenosine diphosphate-ribose)
polymerase 1
|
|
ROC
|
receiver operating characteristic
|
|
RR
|
relative risk
|
|
TNM
|
tumor-node-metastasis
|
|
TMA
|
tissue microarray
|
References
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lordick F, Allum W, Carneiro F, Mitry E,
Tabernero J, Tan P, Van Cutsem E, van de Velde C and Cervantes A:
Unmet needs and challenges in gastric cancer: The way forward.
Cancer Treat Rev. 40:692–700. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hakmé A, Wong HK, Dantzer F and Schreiber
V: The expanding field of poly (ADP-ribosyl)ation reactions.
‘Protein modifications: Beyond the usual suspects’ review series.
EMBO Rep. 9:1094–1100. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hottiger MO, Hassa PO, Luscher B, Schuler
H and Koch-Nolte F: Toward a unified nomenclature for mammalian
ADP-ribosyltransferases. Trends Biochem Sci. 35:208–219. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Michels J, Vitale I, Saparbaev M, Castedo
M and Kroemer G: Predictive biomarkers for cancer therapy with PARP
inhibitors. Oncogene. 33:3894–3907. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chambon P, Weill JD and Mandel P:
Nicotinamide mononucleotide activation of new DNA-dependent
polyadenylic acid synthesizing nuclear enzyme. Biochem Biophys Res
Commun. 11:39–43. 1963. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sugimura T and Miwa M: Poly (ADP-ribose):
Historical perspective. Mol Cell Biochem. 138:5–12. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Davies JR, Jewell R, Affleck P, Anic GM,
Randerson-Moor J, Ozola A, Egan KM, Elliott F, García-Casado Z,
Hansson J, et al: Inherited variation in the PARP1 geneand survival
from melanoma. Int J Cancer. 135:1625–1633. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bacalini MG, Di Lonardo D, Catizone A,
Ciccarone F, Bruno T, Zampieri M, Guastafierro T, Calabrese R,
Fanciulli M, Passananti C, et al: Poly (ADP-ribosyl)ation affects
stabilization of Che-1 protein in response to DNA damage. DNA
Repair (Amst). 10:380–389. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Schreiber V, Dantzer F, Ame JC and de
Murcia G: Poly (ADP-ribose): Novel functions for an old molecule.
Nat Rev Mol Cell Biol. 7:517–528. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Staibano S, Pepe S, Lo Muzio L, Somma P,
Mascolo M, Argenziano G, Scalvenzi M, Salvatore G, Fabbrocini G,
Molea G, et al: Poly(adenosine diphosphate-ribose) polymerase 1
expression in malignant melanomas from photoexposed areas of the
head and neck region. Hum Pathol. 36:724–731. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nosho K, Yamamoto H, Mikami M, Taniguchi
H, Takahashi T, Adachi Y, Imamura A, Imai K and Shinomura Y:
Overexpression of poly(ADP-ribose) polymerase-1 (PARP-1) in the
early stage of colorectal carcinogenesis. Eur J Cancer.
42:2374–2381. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Rojo F, Garcia-Parra J, Zazo S, Tusquets
I, Ferrer-Lozano J, Menendez S, Eroles P, Chamizo C, Servitja S,
Ramírez-Merino N, et al: Nuclear PARP-1 protein overexpression is
associated with poor overall survival in early breast cancer. Ann
Oncol. 23:1156–1164. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Mego M, Cierna Z, Svetlovska D, Macak D,
Machalekova K, Miskovska V, Chovanec M, Usakova V, Obertova J,
Babal P and Mardiak J: PARP expression in germ cell tumours. J Clin
Pathol. 66:607–612. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sun Y, Gallacchi D, Zhang EY, Reynolds SB,
Robinson L, Malinowska IA, Chiou TT, Pereira AM, Li C, Kwiatkowski
DJ, et al: Rapamycin-resistant poly (ADP-ribose) polymerase-1
overexpression is a potential therapeutic target in
lymphangioleiomyomatosis. Am J Respir Cell Mol Biol. 51:738–749.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Weaver AN and Yang ES: Beyond DNA Repair:
Additional functions of PARP-1 in cancer. Front Oncol. 3:2902013.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Rouleau M, Patel A, Hendzel MJ, Kaufmann
SH and Poirier GG: PARP inhibition: PARP1 and beyond. Nat Rev
Cancer. 10:293–301. 2010. View
Article : Google Scholar : PubMed/NCBI
|
|
18
|
Benafif S and Hall M: An update on PARP
inhibitors for the treatment of cancer. Onco Targets Ther.
8:519–528. 2015.PubMed/NCBI
|
|
19
|
O'Shaughnessy J, Osborne C, Pippen JE,
Yoffe M, Patt D, Rocha C, Koo IC, Sherman BM and Bradley C:
Iniparib plus chemotherapy in metastatic triple-negative breast
cancer. N Engl J Med. 364:205–214. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kummar S, Ji J, Morgan R, Lenz HJ, Puhalla
SL, Belani CP, Gandara DR, Allen D, Kiesel B, Beumer JH, et al: A
phase I study of veliparib in combination with metronomic
cyclophosphamide in adults with refractory solid tumors and
lymphomas. Clin Cancer Res. 18:1726–1734. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhang Q, Li Y, Li X, Zhou W, Shi B, Chen H
and Yuan W: PARP-1 Val762Ala polymorphism, CagA+ H.
pylori infection and risk for gastric cancer in Han Chinese
population. Mol Biol Rep. 36:1461–1467. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kim J, Pyun JA, Cho SW, Lee K and Kwack K:
Lymph node metastasis of gastric cancer is associated with the
interaction between poly (ADP-ribose) polymerase 1 and matrix
metallopeptidase 2. DNA Cell Biol. 30:1011–1017. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
He W, Liu T, Shan Y, Zhu K and Li Y: PARP1
polymorphisms increase the risk of gastric cancer in a Chinese
population. Mol Diagn Ther. 16:35–42. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Le TV, Suh JH, Kim N and Park HJ: In
silico identification of poly (ADP-ribose)polymerase-1 inhibitors
and their chemosensitizing effects against cisplatin-resistant
human gastric cancer cells. Bioorg Med Chem Lett. 23:2642–2646.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Liu HR, Meng LY, Lin ZY, Shen Y, Yu YQ and
Zhu YZ: Cochinchina momordica seed extract induces apoptosis and
cell cycle arrest in human gastric cancer cells via PARP and p53
signal pathways. Nutr Cancer. 64:1070–1077. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Fielding JW, Roginski C, Ellis DJ, Jones
BG, Powell J, Waterhouse JA and Brookes VS: Clinicopathological
staging of gastric cancer. Br J Surg. 71:677–680. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Krishnakumar R and Kraus WL: The PARP side
of the nucleus: Molecular actions, physiological outcomes, and
clinical targets. Mol Cell. 39:8–24. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Cepeda V, Fuertes MA, Castilla J, Alonso
C, Quevedo C, Soto M and Pérez JM: Poly (ADP-ribose) polymerase-1
(PARP-1) inhibitors in cancer chemotherapy. Recent Pat Anticancer
Drug Discov. 1:39–53. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Mégnin-Chanet F, Bollet MA and Hall J:
Targeting poly(ADP-ribose) polymerase activity for cancer therapy.
Cell Mol Life Sci. 67:3649–3662. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Shimizu S, Nomura F, Tomonaga T, Sunaga M,
Noda M, Ebara M and Saisho H: Expression of poly(ADP-ribose)
polymerase in human hepatocellular carcinoma and analysis of biopsy
specimens obtained under sonographic guidance. Oncol Rep.
12:821–825. 2004.PubMed/NCBI
|
|
31
|
Miwa M and Masutani M:
PolyADP-ribosylation and cancer. Cancer Sci. 98:1528–1535. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Brustmann H: Poly(adenosine
diphosphate-ribose) polymerase expression in serous ovarian
carcinoma: Correlation with p53, MIB-1, and outcome. Int J Gynecol
Pathol. 26:147–153. 2007.PubMed/NCBI
|
|
33
|
Rodriguez MI, Peralta-Leal A, O'Valle F,
Rodriguez-Vargas JM, Gonzalez-Flores A, Majuelos-Melguizo J, López
L, Serrano S, de Herreros AG, Rodríguez-Manzaneque JC, et al:
PARP-1 regulates metastatic melanoma through modulation of
vimentin-induced malignant transformation. PLoS Genet.
9:e10035312013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Li M, Threadgill MD, Wang Y, Cai L and Lin
X: Poly(ADP-ribose) polymerase inhibition down-regulates expression
of metastasis-related genes in CT26 colon carcinoma cells.
Pathobiology. 76:108–116. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Pu H, Horbinski C, Hensley PJ, Matuszak
EA, Atkinson T and Kyprianou N: PARP-1 regulates
epithelial-mesenchymal transition (EMT) in prostate tumorigenesis.
Carcinogenesis. 35:2592–2601. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Lönn P, van der Heide LP, Dahl M, Hellman
U, Heldin CH and Moustakas A: PARP-1 attenuates Smad-mediated
transcription. Mol Cell. 40:521–532. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Virág L and Szabó C: The therapeutic
potential of poly(ADP-ribose) polymerase inhibitors. Pharmacol Rev.
54:375–429. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Barboro P, Repaci E, D'Arrigo C and Balbi
C: The role of nuclear matrix proteins binding to matrix attachment
regions (Mars) in prostate cancer cell differentiation. PLoS One.
7:e406172012. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Chiou SH, Jiang BH, Yu YL, Chou SJ, Tsai
PH, Chang WC, Chen LK, Chen LH, Chien Y and Chiou GY: Poly
(ADP-ribose) polymerase 1 regulates nuclear reprogramming and
promotes iPSC generation without c-Myc. J Exp Med. 210:85–98. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Xie KJ, He HE, Sun AJ, Liu XB, Sun LP and
Dong XJ: Expression of ERCC1, MSH2 and PARP1 in non-small cell lung
cancer and prognostic value in patients treated with platinum-based
chemotherapy. Asian Pac J Cancer Prev. 15:2591–2596. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Donizy P, Pietrzyk G, Halon A, Kozyra C,
Gansukh T, Lage H, Surowiak P and Matkowski R: Nuclear-cytoplasmic
PARP-1 expression as an unfavorable prognostic marker in lymph
node-negative early breast cancer: 15-year follow-up. Oncol Rep.
31:1777–1787. 2014.PubMed/NCBI
|
|
42
|
Goncalves A, Finetti P, Sabatier R,
Gilabert M, Adelaide J, Borg JP, Chaffanet M, Viens P, Birnbaum D
and Bertucci F: Poly (ADP-ribose) polymerase-1 mRNA expression in
human breast cancer: A meta-analysis. Breast Cancer Res Treat.
127:273–281. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Aiad HA, Kandil MA, El-Tahmody MA,
Abulkheir IL, Abulkasem FM, Elmansori AA and Aleskandarany MA: The
prognostic and predictive significance of PARP-1 in locally
advanced breast cancer of Egyptian patients receiving neoadjuvant
chemotherapy. Appl Immunohistochem Mol Morphol. 23:571–579. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Klauschen F, von Winterfeld M, Stenzinger
A, Sinn BV, Budczies J, Kamphues C, Bahra M, Wittschieber D,
Weichert W, Striefler J, et al: High nuclear
poly-(ADP-ribose)-polymerase expression is prognostic of improved
survival in pancreatic cancer. Histopathology. 61:409–416. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Moss SF, Calam J, Agarwal B, Wang S and
Holt PR: Induction of gastric epithelial apoptosis by Helicobacter
pylori. Gut. 38:498–501. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Wagner S, Beil W, Westermann J, Logan RP,
Bock CT, Trautwein C, Bleck JS and Manns MP: Regulation of gastric
epithelial cell growth by Helicobacter pylori: Offdence for a major
role of apoptosis. Gastroenterology. 113:1836–1847. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Atherton JC: The pathogenesis of
Helicobacter pylori-induced gastro-duodenal diseases. Annu Rev
Pathol. 1:63–96. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Polk DB and Peek RM Jr: Helicobacter
pylori: Gastric cancer and beyond. Nat Rev Cancer. 10:403–414.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Chen Y, Wang Y, Xu W and Zhang Z: Analysis
on the mechanism of Helicobacter pylori-induced apoptosis in
gastric cancer cell line BGC-823. Int J Mol Med. 16:741–745.
2005.PubMed/NCBI
|
|
50
|
Nossa CW, Jain P, Tamilselvam B, Gupta VR,
Chen LF, Schreiber V, Desnoyers S and Blanke SR: Activation of the
abundant nuclear factor poly(ADP-ribose) polymerase-1 by
Helicobacter pylori. Proc Natl Acad Sci USA. 106:19998–20003. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Hassumi-Fukasawa MK, Miranda-Camargo FA,
Zanetti BR, Galano DF, Ribeiro-Silva A and Soares EG: Expression of
BAG-1 and PARP-1 in precursor lesions and invasive cervical cancer
associated with human papillomavirus (HPV). Pathol Oncol Res.
18:929–937. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Nossa CW and Blanke SR: Helicobacter
pylori activation of PARP-1: Usurping a versatile regulator of host
cellular health. Gut Microbes. 1:373–378. 2010. View Article : Google Scholar : PubMed/NCBI
|