Introduction
Defects of hemostasis in patients with cancer have
long been recognized; Trousseau, in 1865, was the first to note
this association, and Morrison examined altered coagulation in
patients with malignancy as early as the year 1932 (1). Since then, numerous studies have
confirmed the association between malignancy and phenomena
associated with blood coagulation. In total, ~50% of all patients
with malignant diseases and up to 95% of those with metastatic
lesions demonstrate certain abnormalities in hemostatic parameters
(2).
Activation of the coagulation cascade frequently
occurs in cancer by a number of mechanisms, including the
generation of tumor necrosis factor, tumor pro-coagulant and tissue
factor (3). The pro-coagulant state
in cancer arises from the direct capacity of tumor cells to express
and release pro-coagulant factors, including cancer pro-coagulant
and tissue factor, and to activate the hosts hemostatic system
(4).
Numerous studies have documented the association
between hemostatic abnormalities and cancer (5). In addition, various hemostasis markers
associated with tumor staging and prognosis have been previously
studied (6–10). Preoperative plasma fibrinogen levels
in gastric cancer are associated with the extent of the tumor
(10) and may be a predictor for
lymphatic metastasis (7,8). Important studies such as one by Khorana
et al (11) demonstrated that
the expression of tissue factor was associated with histological
grade in pancreatic cancer, and that tissue factor and factor VIIa
were elevated in patients with cancer (12). Elevated plasma D-dimer levels in
patients with colorectal cancer were associated with advanced tumor
stage and short postoperative survival (13). Much remains unknown regarding the
association between colorectal cancer and hemostasis markers. The
primary objective of the present study was to evaluate the
association between preoperative hemostasis markers and
clinicopathological parameters and to identify the hemostasis
marker affecting survival in patients following curative resection
for colorectal cancer.
Materials and methods
Ethical statement
The present study was conducted and coordinated in
the Department of Internal Medicine, Dong-A University Medical
Center (Busan, South Korea). The present study design was centrally
approved by the ethics committee for human research at the Medical
Faculty, Dong-A University Medical center and conformed to the
principles of the Declaration of Helsinki and its subsequent
amendments. All patients provided written informed consent prior to
registration. Study treatment was given in curative intent.
Patients
Between January 2008 and May 2012, 170 patients were
diagnosed with adenocarcinoma of the colon and rectum and underwent
curative resection at the Department of Internal Medicine, Dong-A
University Medical Center. These patients were candidates for
curative operation. All patients assessed in the present study met
the following criteria: histologically confirmed adenocarcinoma of
the colon and rectum; candidates for curative operation; age >18
years; no active infection; no serious or uncontrolled concurrent
medical illness; no history of other malignancies; no
thromboembolic events; and an Eastern Cooperative Oncology Group
performance status of 0–2 (14). The
present study was performed by means of retrospective analysis of
chart review data.
Methods
All patients underwent blood test including several
hemostasis markers was done within 3 days prior to operation for
colorectal cancer. For coagulation and fibrinolysis tests, venous
blood samples were collected in Vacuette tubes (Greiner Holding AG,
Kremsmunster, Austria). Platelet-poor plasma was obtained via 15–20
min centrifugation at room temperature (speed, 3,500 × g).
Prothrombin time (PT) and activated partial thromboplastin time
(aPTT) tests were immediately conducted. Aliquots of plasma were
transferred to plastic tubes without delay, and then stored frozen
at −80°C until analysis for fibrinogen/fibrinogen degradation
products (FDP) and D-dimer. Complete blood cell counts were
determined with an automatic cell counter with EDTA blood. PT and
aPTT were determined with a CA-1500 coagulometer (Sysmex America,
Inc. Lincolnshire, IL, USA) using Thromborel S (Dade Behring GmbH,
Marburg, Germany) and Dade Actin FS (Dade Behring GmbH) commercial
kits, respectively. Fibrinogen, FDP and D-dimer assays were
conducted using Iatron commercial kits (Mitsubishi Kagaku Iatron,
Inc., Tokyo, Japan) by CA-1500 (Sysmex America, Inc.).
Each patient underwent curative colon or rectum
resection. Tissue specimens were examined by a pathologist for the
following characteristics: depth of tumor invasion; presence of
lymph node involvement; histological type; tumor size; margin
involvement; and lymphovascular invasion. The staging of colorectal
cancer and the clinicopathological factors utilized in the present
study was based on the seventh edition of the American Joint
Committee on Cancer Staging manual (15).
Statistical analysis
All statistical calculations were carried out using
the SPSS Windows program (version 18.0; IBM SPSS, Inc., Chicago,
IL, USA). The continuous hemostasis markers are expressed as the
mean ± standard deviation and the clinicopathological parameters
are expressed as percentages. The association between the
hemostasis markers and clinicopathological parameters was assessed
with the Mann-Whitney U test. The Cox-regression analysis method
was used to estimate the association between the hemostasis markers
and overall survival. The log-rank test was used to compare the
overall survival curves between the divided groups of hemostasis
markers. P<0.05 was considered to indicate a statistically
significant difference.
Results
Patient characteristics
The baseline characteristics of the study population
are provided in Table I. Among the
170 patients analyzed, 107 patients (62.6%) were males and 63
patients (36.8%) were females. The median age of the study patients
was 63 years (range, 28–83 years). All patients underwent curative
colon or rectum resection. A total of 84 patients (49.6%) had a
tumor <5 cm in size. A total of 106 patients (62.4%) were in T3
stage, and 107 patients (62.9%) evidenced no lymph node
metastasis.
 | Table I.Characteristics of patients. |
Table I.
Characteristics of patients.
| Variables | Patients, n | % |
|---|
| Gender |
|
Male | 107 | 62.9 |
|
Female | 63 | 37.1 |
| Age, years |
| Median
(range) | 63
(28–84) |
|
|
<60 | 69 | 40.6 |
|
≥60 | 101 | 59.4 |
| Location |
|
Colon | 110 | 64.7 |
|
Rectum | 60 | 35.3 |
| Tumor size |
| <5
cm | 84 | 49.4 |
| ≥5
cm | 86 | 50.6 |
| Lymphovascular
invasion |
|
Negative | 127 | 74.7 |
|
Positive | 43 | 25.3 |
| Marginal
involvement |
|
Positive | 25 | 14.7 |
|
Negative | 137 | 80.6 |
|
Unknown | 8 | 4.7 |
|
Differentiation |
|
Well | 81 | 47.6 |
|
Moderate | 76 | 44.7 |
|
Poor | 5 | 2.9 |
|
Mucinous | 8 | 4.7 |
| T stage |
| T1 | 19 | 11.2 |
| T2 | 28 | 16.5 |
| T3 | 106 | 62.3 |
| T4 | 17 | 10 |
| N stage |
| N0 | 107 | 62.9 |
| N1 | 45 | 26.5 |
| N2 | 18 | 10.6 |
| TNM stage |
| I | 36 | 21.2 |
| II | 69 | 40.6 |
|
III | 51 | 30.8 |
| IV | 14 | 8.2 |
Hemostasis markers and
histopathological variables
In the present study, the association of hemostasis
markers with the histopathological findings was assessed. Of all
the examined hemostasis markers, fibrinogen level was associated
with tumor size (P<0.001), depth of invasion (T stage; P=0.014)
and TNM stage (P=0.048), as shown in Table II. Platelet count was associated with
tumor size (P=0.002) and depth of invasion (P=0.014). PT level was
associated with tumor size (P=0.015). D-dimer level was associated
with depth of invasion (P=0.025) and TNM stage (P=0.031). FDP level
was associated with TNM stage (P=0.002). However, aPTT level was
not associated with any histological parameters.
 | Table II.Association between hemostasis
markers and pathological stage. |
Table II.
Association between hemostasis
markers and pathological stage.
|
| P
valuea |
|---|
|
|
|
|---|
| Variables | Size | T stage | N stage | TNM stage |
|---|
| Platelet | 0.002 | 0.014 | 0.431 | 0.478 |
| PT | 0.015 | 0.076 | 0.63 | 0.168 |
| aPTT | 0.293 | 0.353 | 0.816 | 0.717 |
| Fibrinogen | <0.001 | 0.014 | 0.214 | 0.048 |
| D-dimer | 0.298 | 0.025 | 0.986 | 0.031 |
| FDP | 0.862 | 0.41 | 0.306 | 0.002 |
Hemostasis markers and survival
To clarify the association with overall survival,
the patients were divided into two groups by the value calculated
by the receiver operating characteristic curve that had highest
sensitivity and specificity (Table
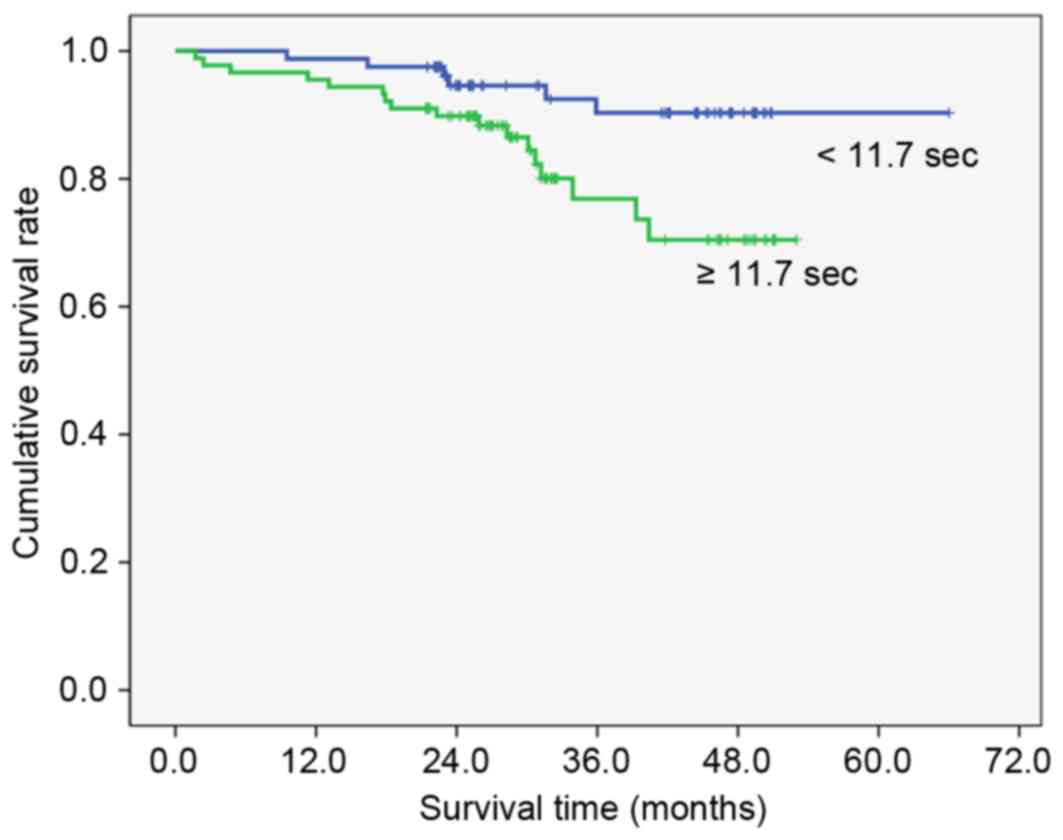
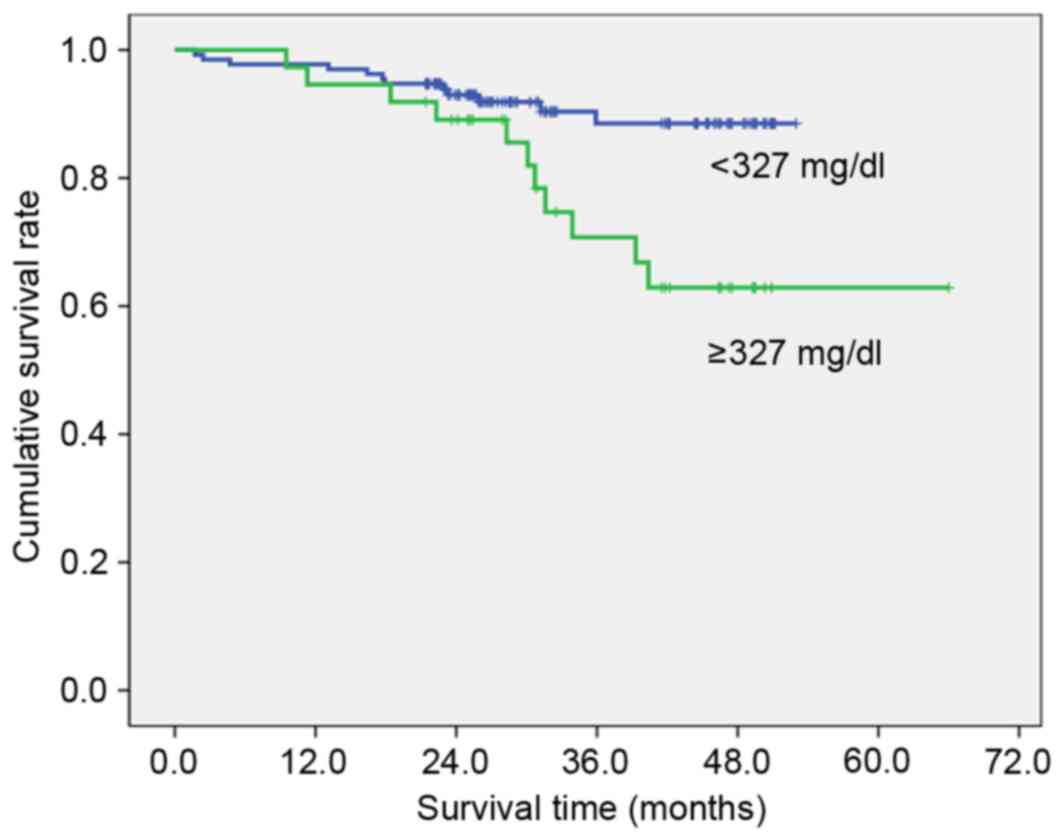
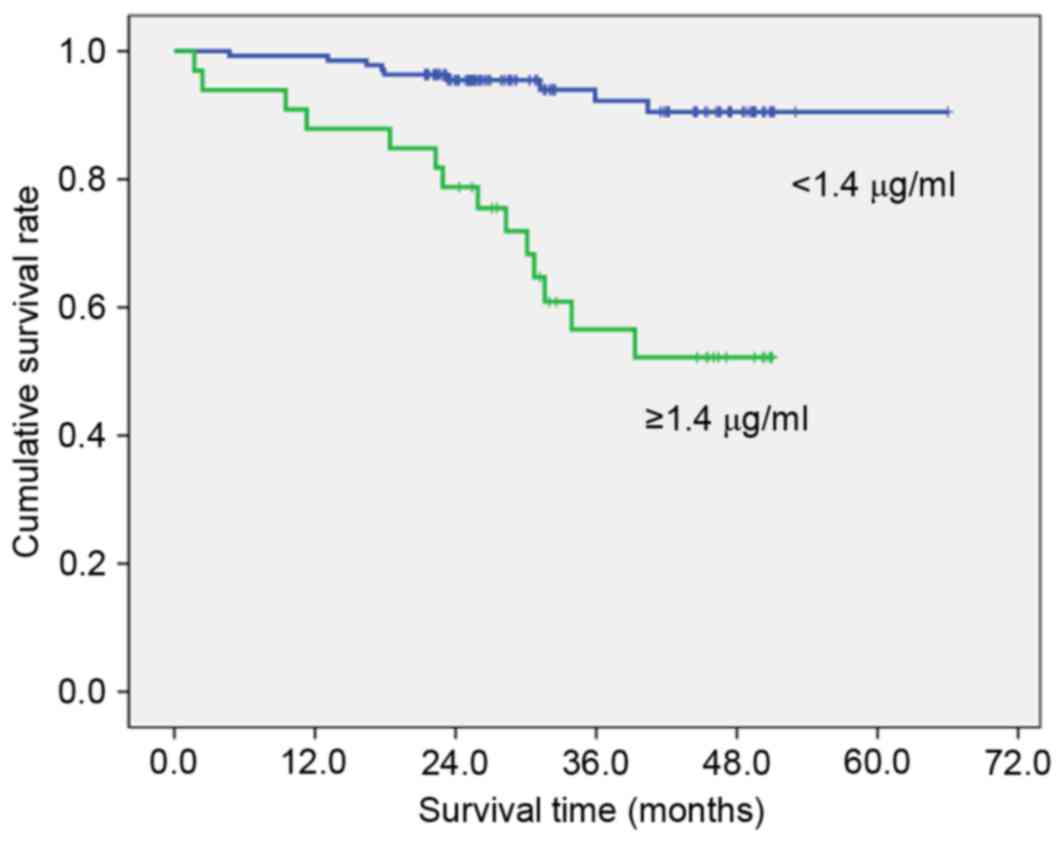
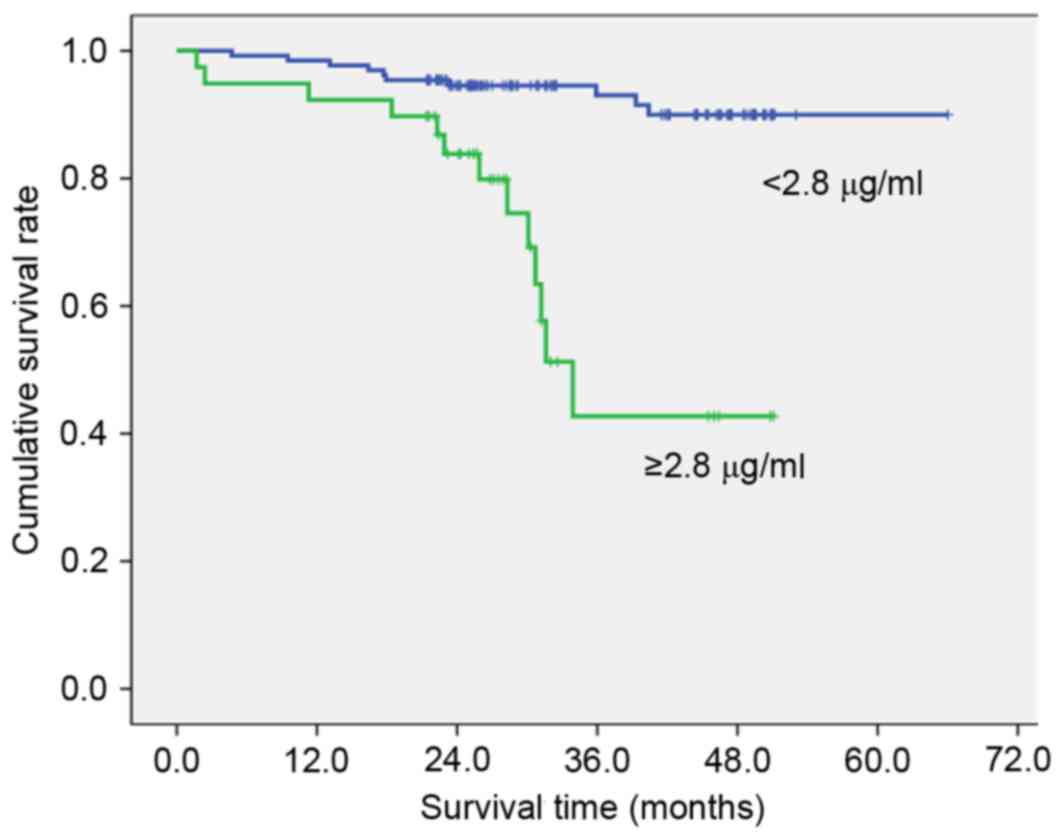
III). High preoperative PT (≥11.7 sec; Fig. 1), fibrinogen (≥327 mg/dl; Fig. 2), D-dimer (≥1.4 µg/ml; Fig. 3) and FDP (≥2.8 µg/ml; Fig. 4) showed significantly lower survival
compared with low PT, fibrinogen, D-dimer and FDP level (P=0.012,
P=007, P<0.001 and P<0.001, respectively). Postoperative
survival curves did not differ between patients with high
preoperative platelet count (PLT) (≥242×103/µl) and aPTT
(≥26.0 sec) levels and those with low PLT and PTT levels.
 | Table III.Association between hemostasis
markers and OS. |
Table III.
Association between hemostasis
markers and OS.
| Variables | Criteria by ROC
curve | No. | 3-year OS, % | Multivariate
P-valuea |
|---|
| Platelet |
≥242×103/µl | 73 | 78.7 | 0.126 |
|
|
<242×103/µl | 97 | 88.6 |
|
| PT | ≥11.7 sec | 89 | 76.9 | 0.012 |
|
| <11.7 sec | 81 | 90.3 |
|
| aPTT | ≥26.0 sec | 73 | 78.3 | 0.063 |
|
| <26.0 sec | 97 | 88.5 |
|
| Fibrinogen | ≥327 mg/dl | 37 | 70.7 | 0.007 |
|
| <327 mg/dl | 133 | 88.5 |
|
| D-dimer | ≥1.4 µg/ml | 33 | 56.6 | <0.001 |
|
| <1.4 µg/ml | 137 | 94.0 |
|
| FDP | ≥2.8 µg/ml | 39 | 42.7 | <0.001 |
|
| <2.8 µg/ml | 131 | 93.0 |
|
Discussion
Several laboratory abnormalities have been
documented in patients with cancer, including prolonged and
shortened PT, PTT, increased and decreased levels of Factor-II, V,
VIII, IX, XI, XII, fibrinogen, FDP, thrombin-anti-thrombin III
complex and thrombocytosis (16).
Activation of the clotting cascade may be due to tumor cells
themselves, or stimulation of tumor-associated inflammatory cells
(17). Accumulating evidence
indicates that hemostatic factors may interact with cancer cells
and affect their development, growth, and metastasis (18,19).
Tang et al (20) demonstrated that a high preoperative
plasma fibrinogen level is associated with distant metastasis and
impaired prognosis in colorectal cancer. Fibrinogen is a
glycoprotein synthesized by hepatocytes and is converted to fibrin
by activated thrombin generated from prothrombin by the action of
coagulation factor Xa (21). Recent
studies have shown that fibrinogen is also secreted by cancer cells
(22) and deposited independently of
thrombin cleavage (23).
Fibrin/fibrinogen deposition induces fibrinolytic activity,
resulting in the degradation of extracellular matrix. Numerous
studies have shown that elevated plasma fibrinogen levels are
useful prognostic parameters for several malignancies, including
esophageal (24), gastric (8), cervical (25) and ovarian cancer (26). Elevated fibrinogen plasma levels have
been shown to be associated with the extent of the disease and
reduced survival in patients with stomach cancer (10) or non-small cell lung cancer (27).
The results of the present study indicated that
preoperative plasma fibrinogen levels were higher in colorectal
cancer patients with large tumors, advanced depth of invasion and
TNM stage compared with patients with small tumors, limited depth
of invasion and TNM stage. This was in accordance with previously
published data on patients with other malignancies (8–10,24,28).
Of all hemostasis markers examined, D-dimer is
determined to be the smallest unique degradation product of
cross-linked fibrin resulting from the proteolytic actions of
plasmin. Plasma D-dimer levels have been shown to be increased in
patients with gastric (29),
colorectal (30), lung (31,32),
ovarian (33) and breast cancer
(34). Oya et al (13) demonstrated that D-dimer level is
associated with the depth of tumor invasion at the time of surgical
excision in patients with colorectal cancer. In the present study,
preoperative plasma D-dimer levels were increased in patients with
advanced T and TNM stages of colorectal cancer, not with lymph node
involvement. In addition, FDP level is associated with TNM stage,
but not tumor size, depth of invasion or lymph node
involvement.
Although thrombocytosis has been reported in
patients with gastric (35), lung
(36) and endometrial cancer
(37), the association between
thrombocytosis and the clinicopathological features of patients
with colorectal cancer has not been fully investigated and the
molecular mechanism of platelets affecting cancer metastasis remain
unclear. Vascularization within a tumor is a portal through which
tumor cells enter the bloodstream to disseminate. Platelets may
stabilize vessel growth during tumor development and thus
contribute to this process (36).
Platelets produce thymidine phosphorylase, which is a
platelet-derived endothelial cell growth factor with angiogenic
activity. Increased platelet count may be translated into enhanced
tumor growth. Sasaki et al (38) demonstrated that preoperative
thrombocytosis was associated with tumor size, depth of invasion,
lymph node metastasis and distant metastasis. Li et al
(36) reported that elevated
platelets enhance cancer cell migration and promote hematogenous
metastasis in advanced non-small cell lung cancer. In the present
study, the preoperative platelet counts are associated with depth
of invasion, but not tumor size, lymph node involvement or TNM
stage.
The levels of PT did not differ significantly in
cases of gastric cancer compared with patients without gastric
cancer (39). However, Ferrigno et
al (40) reported that abnormally
prolonged PT was predictive of a grave prognosis in lung cancer. In
the present study, PT levels were shown to be associated with tumor
size in colorectal cancer.
The current study demonstrated an association
between hemostasis markers and disease progression in malignancy.
Kilic et al (41) demonstrated
that postoperative survival was significantly shorter in patients
with colorectal cancer with increased preoperative D-dimer levels
compared with patients with normal preoperative D-dimer level.
Yamashita et al (8) reported
that hyperfibrinogenemia was associated with poor clinical outcome
in T2 gastric cancer. In the present study, PT, fibrinogen, FDP and
D-dimer affected the overall survival of patients. Fibrin
remodeling is involved in numerous steps of metastasis and has been
shown to perform a crucial role in the formation of new vessels
(42) Cross-linked fibrin in the ECM
serves as a stable framework for endothelial cell migration during
angiogenesis and for tumor cell migration during invasion (43). Knockout mouse models have also
revealed the importance of fibrin remodeling in tumor growth and
metastasis (44). In addition,
multivariate modeling showed an association between the presence of
elevated D-dimer levels and the presence of visceral metastases.
This association may explain why D-dimer and FDP levels were
revealed to be associated with overall survival.
Lykke et al (43) reported hemostatic alterations in
colorectal cancer. They discussed hemostatic and angiogenic system
activation using surgical trauma and postoperative infection, which
may be associated with subsequent dissemination and recurrent tumor
growth (45).
All patients who indicated adjuvant treatment
received chemotherapy and part of the patients with rectal cancer
received additional radiotherapy in the present study. However, the
possibility that adjuvant therapies may affect the disease outcomes
cannot be ignored. However, these effects were not considered on
the analysis between coagulation factor and disease outcomes.
Although various studies revealed association between coagulation
factor and clinicopathological parameters and disease outcomes,
additional studies are required to apply in routine practice. If
the sensitivity and specificity of coagulation factors for
prediction of disease outcomes are researched by meta-analysis of
various studied, these factors may be measured in routine clinical
practice.
In conclusion, preoperative plasma fibrinogen level
is significantly associated with tumor size and depth of tumor
invasion. Thus, plasma fibrinogen level may indicate increased
early tumor burden. Preoperative plasma prolonged PT level,
hyperfibrinogenemia, high D-dimer level and increased FDP level may
function as hemostasis markers that predict overall survival in
patients subsequent to undergoing curative resection for colorectal
cancer.
Acknowledgements
The present study was supported by the Dong-A
University Research Fund (Busan, Republic of Korea).
References
|
1
|
Donati MB and Lorenzet R: Thrombosis and
cancer: 40 years of research. Thromb Res. 129:348–352. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wojtukiewicz MZ, Sierko E, Klementt P and
Rak J: The hemostatic system and angiogenesis in malignancy.
Neoplasia. 3:371–384. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Falanga A, Marchetti M and Vignoli A:
Coagulation and cancer: Biological and clinical aspects. J Thromb
Haemost. 11:223–233. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Coussens LM and Werb Z: Inflammation and
cancer. Nature. 420:860–867. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Falanga A, Russo L and Milesi V: The
coagulopathy of cancer. Curr Opin Hematol. 21:423–429. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ikeda M, Furukawa H, Imamura H, Shimizu J,
Ishida H, Masutani S, Tatsuta M and Satomi T: Poor prognosis
associated with thrombocytosis in patients with gastric cancer. Ann
Surg Oncol. 9:287–291. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yamashita H, Kitayama J and Nagawa H:
Hyperfibrinogenemia is a useful predictor for lymphatic metastasis
in human gastric cancer. Jpn J Clin Oncol. 35:595–600. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yamashita H, Kitayama J, Kanno N, Yatomi Y
and Nagawa H: Hyperfibrinogenemia is associated with lymphatic as
well as hematogenous metastasis and worse clinical outcome in T2
gastric cancer. BMC Cancer. 6:1472006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yamashita H, Kitayama J, Ishikawa M and
Nagawa H: Tissue factor expression is a clinical indicator of
lymphatic metastasis and poor prognosis in gastric cancer with
intestinal phenotype. J Surg Oncol. 95:324–331. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lee SE, Lee JH, Ryu KW, Nam BH, Cho SJ,
Lee JY, Kim CG, Choi IJ, Kook MC, Park SR and Kim YW: Preoperative
plasma fibrinogen level is a useful predictor of adjacent organ
involvement in patients with advanced gastric cancer. J Gastric
Cancer. 12:81–87. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Khorana AA, Ahrendt SA, Ryan CK, Francis
CW, Hruban RH, Hu YC, Hostetter G, Harvey J and Taubman MB: Tissue
factor expression, angiogenesis, and thrombosis in pancreatic
cancer. Clin Cancer Res. 13:2870–2875. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Joanne LY, May L, Lhotak V, Shahrzad S,
Shirasawa S, Weitz JI, Coomber BL, Mackman N and Rak JW: Oncogenic
events regulate tissue factor expression in colorectal cancer
cells: Implications for tumor progression and angiogenesis. Blood.
105:1734–1741. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Oya M, Akiyama Y, Okuyama T and Ishikawa
H: High preoperative plasma D-dimer level is associated with
advanced tumor stage and short survival after curative resection in
patients with colorectal cancer. Jpn J Clin Oncol. 31:388–394.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Dewys WD, Begg C, Lavin PT, Band PR,
Bennett JM, Bertino JR, Cohen MH, Douglass HO Jr, Engstrom PF,
Ezdinli EZ, et al: Prognostic effect of weight loss prior to
chemotherapy in cancer patients. Eastern Cooperative Oncology
Group. Am J Med. 69:491–497. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
UICC, . TNM Classification of Malignant
Tumours. 7th. Wiley & Liss; New York: 2009
|
|
16
|
Gouin-Thibault I and Samama MM: Laboratory
diagnosis of the thrombophilic state in cancer patients. Semin
Thromb Hemost. 25:167–172. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lima LG and Monteiro RQ: Activation of
blood coagulation in cancer: Implications for tumour progression.
Biosci Rep. 33:e000642013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Balkwill F and Mantovani A: Inflammation
and cancer: Back to Virchow? Lancet. 357:539–545. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wang X, Wang E, Kavanagh JJ and Freedman
RS: Ovarian cancer, the coagulation pathway, and inflammation. J
Transl Med. 3:252005. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tang L, Liu K, Wang J, Wang C, Zhao P and
Liu J: High preoperative plasma fibrinogen levels are associated
with distant metastases and impaired prognosis after curative
resection in patients with colorectal cancer. J Surg Oncol.
102:428–432. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Tennent GA, Brennan SO, Stangou AJ,
O'Grady J, Hawkins PN and Pepys MB: Human plasma fibrinogen is
synthesized in the liver. Blood. 109:1971–1974. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sahni A, SimpsonHaidaris P, Sahni S, Vaday
G and Francis C: Fibrinogen synthesized by cancer cells augments
the proliferative effect of fibroblast growth factor-2 (FGF-2). J
Thromb Haemost. 6:176–183. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Guadiz G, Sporn LA and Simpson-Haidaris
PJ: Thrombin cleavage-independent deposition of fibrinogen in
extracellular matrices. Blood. 90:2644–2653. 1997.PubMed/NCBI
|
|
24
|
Takeuchi H, Ikeuchi S, Kitagawa Y, Shimada
A, Oishi T, Isobe Y, Kubochi K, Kitajima M and Matsumoto S:
Pretreatment plasma fibrinogen level correlates with tumor
progression and metastasis in patients with squamous cell carcinoma
of the esophagus. J Gastroenterol Hepatol. 22:2222–2227. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Polterauer S, Seebacher V,
Hefler-Frischmuth K, Grimm C, Heinze G, Tempfer C, Reinthaller A
and Hefler L: Fibrinogen plasma levels are an independent
prognostic parameter in patients with cervical cancer. Am J Obstet
Gynecol. 200:647.e1–e7. 2009. View Article : Google Scholar
|
|
26
|
Polterauer S, Grimm C, Seebacher V, Concin
N, Marth C, Tomovski C, Husslein H, Leipold H, Hefler-Frischmuth K,
Tempfer C, et al: Plasma fibrinogen levels and prognosis in
patients with ovarian cancer: A multicenter study. Oncologist.
14:979–985. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Li Y, Wei S, Wang J, Hong L, Cui L and
Wang C: Analysis of the factors associated with abnormal
coagulation and prognosis in patients with non-small cell lung
cancer. Zhongguo Fei Ai Za Zhi. 17:789–796. 2014.(In Chinese).
PubMed/NCBI
|
|
28
|
Ma Y, Qian Y and Lv W: The correlation
between plasma fibrinogen levels and the clinical features of
patients with ovarian carcinoma. J Int Med Res. 35:678–684. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Liu L, Zhang X, Yan B, Gu Q, Zhang X, Jiao
J, Sun D, Wang N and Yue X: Elevated plasma D-dimer levels
correlate with long term survival of gastric cancer patients. PloS
One. 9:e905472014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yamamoto M, Yoshinaga K, Matsuyama A,
Iwasa T, Osoegawa A, Tsujita E, Yamashita Y, Tsutsui S and Ishida
T: Plasma D-dimer level as a mortality predictor in patients with
advanced or recurrent colorectal cancer. Oncology. 83:10–15. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Fukumoto K, Taniguchi T, Usami N,
Kawaguchi K, Fukui T, Ishiguro F, Nakamura S and Yokoi K: The
preoperative plasma D-dimer level is an independent prognostic
factor in patients with completely resected non-small cell lung
cancer. Surg Today. 45:63–67. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
İnal T, Anar C, Polat G, Ünsal İ and
Halilçolar H: The prognostic value of D-dimer in lung cancer. Clin
Respir J. 9:305–313. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Man YN, Wang YN, Hao J, Liu X, Liu C, Zhu
C and Wu XZ: Pretreatment plasma D-dimer, fibrinogen and platelet
levels significantly impact prognosis in patients with epithelial
ovarian cancer independently of venous thromboembolism. Int J
Gynecol Cancer. 25:24–32. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Blackwell K, Haroon Z, Broadwater G, Berry
D, Harris L, Iglehart JD, Dewhirst M and Greenberg C: Plasma
D-dimer levels in operable breast cancer patients correlate with
clinical stage and axillary lymph node status. J Clin Oncol.
18:600–608. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Li FX, Wei LJ, Zhang H, Li SX and Liu JT:
Significance of thrombocytosis in clinicopathologic characteristics
and prognosis of gastric cancer. Asian Pac J Cancer Prev.
15:6511–6517. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Li Y, Miao LY, Xiao YL, Cai HR and Zhang
DP: Elevated platelets enhance cancer cell migration, promote
hematogenous metastasis and associate with a poor prognosis in
advanced non-small cell lung cancer cases. Asian Pac J Cancer Prev.
15:139–143. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Heng S and Benjapibal M: Preoperative
thrombocytosis and poor prognostic factors in endometrial cancer.
Asian Pac J Cancer Prev. 15:10231–10236. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Sasaki K, Kawai K, Tsuno NH, Sunami E and
Kitayama J: Impact of preoperative thrombocytosis on the survival
of patients with primary colorectal cancer. World J Surg.
36:192–200. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Di Micco P, Romano M, Niglio A, Nozzolillo
P, Federico A, Petronella P, Nunziata L, Di Micco B and Torella R:
Alteration of haemostasis in non-metastatic gastric cancer. Dig
Liver Dis. 33:546–550. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Ferrigno D, Buccheri G and Ricca I:
Prognostic significance of blood coagulation tests in lung cancer.
Eur Respir J. 17:667–673. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kılıc M, Yoldas O, Keskek M, Ertan T, Tez
M, Gocmen E and Koc M: Prognostic value of plasma D-dimer levels in
patients with colorectal cancer. Colorectal Dis. 10:238–241. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Südhoff T and Schneider W: Fibrinolytic
mechanisms in tumor growth and spreading. Clin Investig.
70:631–636. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Lykke J and Nielsen HJ: Haemostatic
alterations in colorectal cancer: Perspectives for future
treatment. J Surg Oncol. 88:269–275. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Bugge TH, Kombrinck KW, Xiao Q, Holmbäck
K, Daugherty CC, Witte DP and Degen JL: Growth and dissemination of
Lewis lung carcinoma in plasminogen-deficient mice. Blood.
90:4522–4531. 1997.PubMed/NCBI
|
|
45
|
Svendsen MN, Werther K, Nielsen HJ and
Kristjansen PE: VEGF and tumour angiogenesis. Impact of surgery,
wound healing, inflammation and blood transfusion. Scand J
Gastroenterol. 37:373–379. 2002. View Article : Google Scholar : PubMed/NCBI
|