Introduction
Colon cancer is one of the most common malignant
gastrointestinal tumors. The morbidity and mortality of colon
cancer in the world as well as in China are gradually increasing
(1). The majority of patients with
colon cancer are in advanced stage by the time of diagnosis
(2); thus, surgery is rarely a
sufficient treatment. Consequently, chemotherapy is markedly
important in the treatment of colon cancer. However, chemotherapy's
effectiveness is limited because colon cancer cells acquire
multiple drug resistance (MDR) (3).
To improve the effect of chemotherapy in the
treatment of colon cancer, one of the major challenges is reversing
MDR in colon cancer cells. In previous studies, calcium antagonists
such as verapamil (4) and small
interfering RNA (siRNA) targeting multidrug resistance protein 1
(MDR1) messenger RNA (mRNA) were observed to modulate
MDR1/P-glycoprotein (P-gp)-dependent MDR by downregulating the
expression of MDR1 mRNA and P-gp (5).
However, calcium antagonists may cause heart failure and
hypotension (6), and the clinical
benefit of siRNA targeting MDR1 mRNA has not been reported thus
far. Therefore, successful reversal of drug resistance still
requires new therapeutic strategies or pharmaceuticals.
Wild-type p53-induced phosphatase (Wip1) is a member
of the 2C type serine/threonine protein phosphatase family
(7). Wip1 is closely associated with
the p53 gene and it can dephosphorylate downstream proteins of the
p53 gene, thus causing the degradation of p53 protein and the
inhibition of DNA repair and cell apoptosis mediated by p53
(8–10). Thereby, Wip1 promotes tumorigenesis
(11). The Wip1 gene has been
reported to be overexpressed in a variety of tumors (12,13).
Previous studies reported an association between Wip1 and
sensitivity towards chemotherapy. For example, self-proliferation
of cancer stem cells was inhibited by downregulation of Wip1, which
improved the chemosensitivity of breast cancer cells (14). However, the correlation between the
expression of the Wip1 gene and the chemosensitivity of colon
cancer cells has not been reported yet.
In the present study, RNA interference (RNAi) was
used to observe the inhibition of the Wip1 gene induced by a
specific siRNA sequence and to investigate the influence of Wip1
gene silencing on the chemosensitivity of colon cancer cells.
Therefore, a new potential method may have been identified to
reverse the drug resistance of colon cancer cells.
Materials and methods
Cell culture
RKO and COLO 320DM human colon adenocarcinoma cell
lines were purchased from Shanghai Cell Bank of Chinese Academy of
Sciences (Shanghai, China) and were cultured in RPMI-1640 medium
(Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA)
supplemented with 10% fetal bovine serum (FBS) (Biological
Industries Israel Beit-Haemek, Kibbutz Beit-Haemek, Israel), 100
U/ml penicillin and 100 µg/ml streptomycin at 37°C in a humidified
incubator containing 5% CO2.
Western blotting
Cells were washed twice with PBS and then lysed with
100 µl lysis buffer (91 µl radioimmunoprecipitation assay buffer +
9 µl 10X protease cocktail; Beijing Kangwei Century Biotechnology
Co., Ltd., Beijing, China) on ice for 30 min. Cell lysates were
centrifuged at 14,000 × g for 20 min at 4°C. The total cellular
protein content was determined with BCA™ Protein Assay kit (Beijing
Kangwei Century Biotechnology Co., Ltd, Beijing, China).
Subsequently, cellular proteins (20 µg) were dissolved in sample
loading buffer (Beijing Kangwei Century Biotechnology Co., Ltd.)
and subjected to 10% SDS-PAGE. Proteins were electrotransferred to
polyvinylidene fluoride membranes (200 mA, 90 min). The membranes
were rinsed with PBS and blocked with 5% nonfat milk in PBS for 90
min at room temperature. The membranes were then incubated with
primary polyclonal anti-Wip1 (1:1,000; GeneTex, Inc., Irvine, CA,
USA) or monoclonal anti-GAPDH antibodies (cat. no., MB001H;
1:1,000; Bioworld Technology, Inc., St. Louis Park, MN, USA) in 5%
nonfat milk overnight at 4°C. Following primary antibody
incubation, the membranes were rinsed in TBS containing Tween-20
(TBS-T) wash buffer four times (10 min each). The membranes were
then incubated with a secondary antibody (cat. no., E030120-02;
horseradish peroxidase-conjugated goat anti-rabbit immunoglobulin
G; 1:5,000; EarthOx Life Sciences, Millbrae, CA, USA) for 1 h at
room temperature and rinsed in TBS-T wash buffer four times (5 min
each). The protein-antibody complexes were visualized by
chemiluminescence (ECL Plus Western Blotting Substrate; Thermo
Fisher Scientific, Inc.).
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNA was isolated from cells using RNAiso Plus
(Takara Biotechnology Co., Ltd., Dalian, China) according to the
manufacturer's protocol. Single stranded complementary DNA (cDNA)
was synthesized from 1 µg RNA using cDNA Synthesis kit (Takara
Biotechnology Co., Ltd.). The newly synthesized cDNA was amplified
by PCR using an iCycler iQ™ Real-Time PCR Detection System (Bio-Rad
Laboratories, Inc., Hercules, CA, USA). The reaction mixture
contained 2 µl of cDNA template, 0.4 µl of 10 µM Wip1 sense primer
(5′-CAGGGAAACTTTACCAATGAA-3′), 0.4 µl of 10 µM Wip1 antisense
primer (5′-ACGAACCAGGGCAGGTATAT-3′), 10 µl of SYBR Premix Ex Taq™
(Takara Bio Inc., Otsu, Japan) and 7.2 µl of distilled
H2O. The final reaction volume was 20 µl. Both sense
(5′-CCTGGATACCGCAGCTAGGA-3′) and antisense
(5′-GCGGCGCAATACGAATGCCCC-3′) 18S ribosomal RNA (rRNA) primers were
used as internal controls. The cycling conditions were as follows:
95°C for 30 sec to denature the cDNA and primers, followed by 40
cycles at 95°C for 5 sec and 60°C for 20 sec. Dissociation curve
analysis (15) was performed
according to the following conditions: 95°C for 0 sec, 60°C for 15
sec and 95°C for 0 sec. The amplification products of Wip1 and 18S
rRNA were 180 and 112 bp in size, respectively. The relative
expression of Wip1 mRNA was calculated by comparing it with the
mRNA expression of the negative control, which was set as 1.
siRNA synthesis and transfection
The siRNA sequences targeting Wip1 (GenBank
accession no. NM008493) were designed by Suzhou Jima Gene Co. Ltd.
(Suzhou, China). Wip1 siRNA duplexes (Wip1-811 siRNA), positive
control (GAPDH) siRNA and negative control siRNA were synthesized
by Suzhou Jima Gene Co. Ltd. For Wip1-811 siRNA, the sense strand
was 5′-GGGUGGUUCUUGGAAUUCATT-3′ and the antisense strand was
5′-UGAAUUCCAAGAACCACCCTT-3′. Cells treated with Lipofectamine™ 2000
(Thermo Fisher Scientific, Inc.) and Opti-MEM® I Reduced Serum
Medium (Thermo Fisher Scientific, Inc.) were set as the liposome
control. The siRNA duplexes were transfected using Lipofectamine™
2000 according to the protocol recommended by the manufacturer.
Serial concentrations of Wip1 siRNA, including 5, 10, 25, 50 and
100 nmol/l, were used to screen the optimal concentration of Wip1
siRNA. A series of transfection times, including 24, 48, 72 and 96
h, were used to screen the optimal transfection time.
MTS assay
Cell viability assays were performed by MTS assay.
Cells were seeded at 1×104 cells/well in 96-well
microtiter plates. After 24 h of incubation, Wip1-811 siRNA
transfection was performed. A series of diluted concentrations of
anti-tumor drugs, including 5-fluorouracil (5-FU; Sigma-Aldrich;
Merck KGaA, Darmstadt, Germany), oxaliplatin (Sigma-Aldrich; Merck
KGaA) and adriamycin (Sigma-Aldrich; Merck KGaA), were added to the
cells 24 h after transfection. Then, the cells were incubated for 3
days at 37°C in 5% CO2. Subsequently, 20 µl of MTS
solution (Promega Corporation, Madison, WI, USA) was added to the
cells and incubated for 3 h at 37°C, followed by agitation for 5
min. Optical density (OD) values were read on a Synergy HT
Multi-Detection Microplate Reader (Bio-Tek Instruments, Inc.,
Winooski, VT, USA) at a wavelength of 570 nm. Cell viability was
calculated by comparison with the OD value of the control, and the
half-maximal inhibitory concentration (IC50) of the
anti-tumor drug in each group was further calculated.
Analysis of cell apoptosis and cell
cycle
RKO colon cancer cells were plated at
5×105 cells/well in 12-well plates in RPMI-1640 medium
with 10% FBS. After 24 h of incubation, Wip1-811 siRNA transfection
was performed. 5-FU or oxalipatin were added 24 h after
transfection to a final concentration of 5 µmol/l. Then, cells were
incubated at 37°C and 5% CO2 for 48 h. Subsequently, the
cells were harvested and washed twice with cold PBS, and then
divided into two tubes. One tube of cells was used for analysis of
cell apoptosis, in which 1×106 cells/ml of cell
suspension was obtained by adding 5 µl of Annexin V-FITC (BD
Biosciences, San Jose, CA, USA) and 200 µl of 1X binding buffer (BD
Biosciences). Propidium iodide (10 µl) (Thermo Fisher Scientific,
Inc.) was next added and allowed to react in the dark for 30 min.
Next, the cell suspension was subjected to analysis of cell
apoptosis. The other tube of cells was prepared for analysis of the
cell cycle. Briefly, the cells were fixed with 2 ml of 70% (v/v)
ethanol at 4°C, and then resuspended by adding 200 µl of PBS. The
next day, the fixed cells were ready for analysis of the cell
cycle. Flow cytometry analysis was performed with a FACSVerse™ flow
cytometer (BD Biosciences). For each sample, 10,000 cells were
analyzed.
Intracellular adriamycin accumulation
assay
The fluorescence intensity of intracellular
adriamycin was determined using flow cytometry according to a
standard method (16). Cells were
plated at 2×105 cells/well in 6-well plates 24 h prior
to siRNA transient transfection, and then incubated for 48 h. Next,
adriamycin (final concentration, 10 µmol/l) was added to the cells
and incubated for 2 h. The cells were then harvested, washed twice
with cold PBS and placed in ice-water to block the reaction until
analysis. After 30 min, the fluorescence intensity of the cells was
determined by FACSVerse™ flow cytometry at an excitation wavelength
of 488 nm and a detection wavelength of 575 nm. COLO 320DM colon
cancer cells transfected with MDR1 siRNA (#4123; Thermo Fisher
Scientific, Inc.) were used as the positive control and compared
with COLO 320DM colon cancer cells (5).
Statistical analysis
All measurement data were present as mean ± standard
deviation (SD). The differences among multiple mean values were
evaluated using one-way analysis of variance (ANOVA) and post-hoc
Tukey's test. The differences between two mean values were
estimated using independent-samples t-test. All of the statistical
analyses were processed with the statistical analysis software
SPSS, version 19.0 (IBM Corp, Armonk, NY, USA).
Results
Optimal inhibitory concentration of
Wip1 siRNA on the mRNA and protein expression of Wip1 in RKO colon
cancer cells
In order to investigate the inhibitory effect of
siRNA on the mRNA and protein expression of Wip1, RKO colon cancer
cells were seeded in a 24-well plate. Serial concentrations of
Wip1-811 siRNA (range, 5–100 nmol/l) were transfected into RKO
colon cancer cells. Cells cultured in medium without liposomes or
siRNA treatment were used as controls. The liposome control was
treated only with Lipofectamine® 2000. Negative controls were
treated with a control siRNA containing no homology to any human
gene. Cells of each group were harvested 24 or 48 h after
transfection. RT-qPCR detected the expression of Wip1 mRNA. Western
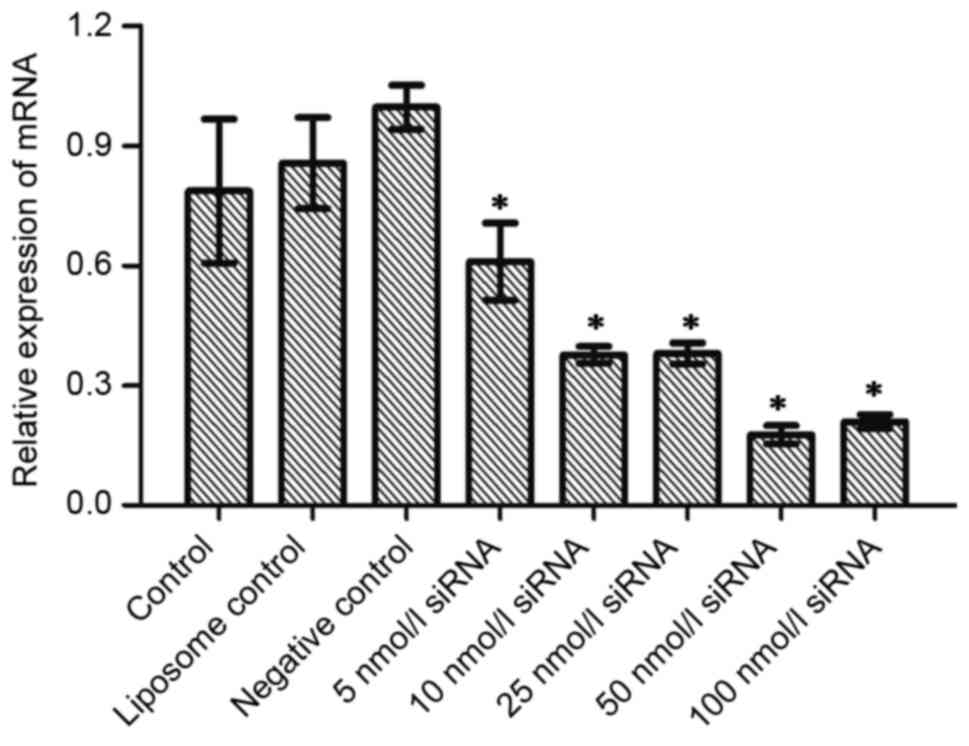
blotting examined the expression of Wip1 protein. Fig. 1A shows the influence of different
concentrations of Wip1-811 siRNA on the relative mRNA expression of
Wip1 in RKO colon cancer cells 24 h after siRNA transfection. No
significant differences were observed in Wip1 mRNA levels between
the control, liposome control and negative control groups 24 h
after transfection. However, there was a significant difference
between the Wip1 siRNA groups and the negative control group
(P<0.05; Fig. 1A). This indicated
that different concentrations of Wip1 siRNA decreased Wip1 mRNA
expression 24 h after transfection. The expression of Wip1 mRNA was
the lowest when the concentration of Wip1-811 siRNA was 50 nmol/l.
The results from RT-qPCR at 48 h post-transfection were similar to
those at 24 h post-transfection (Fig.
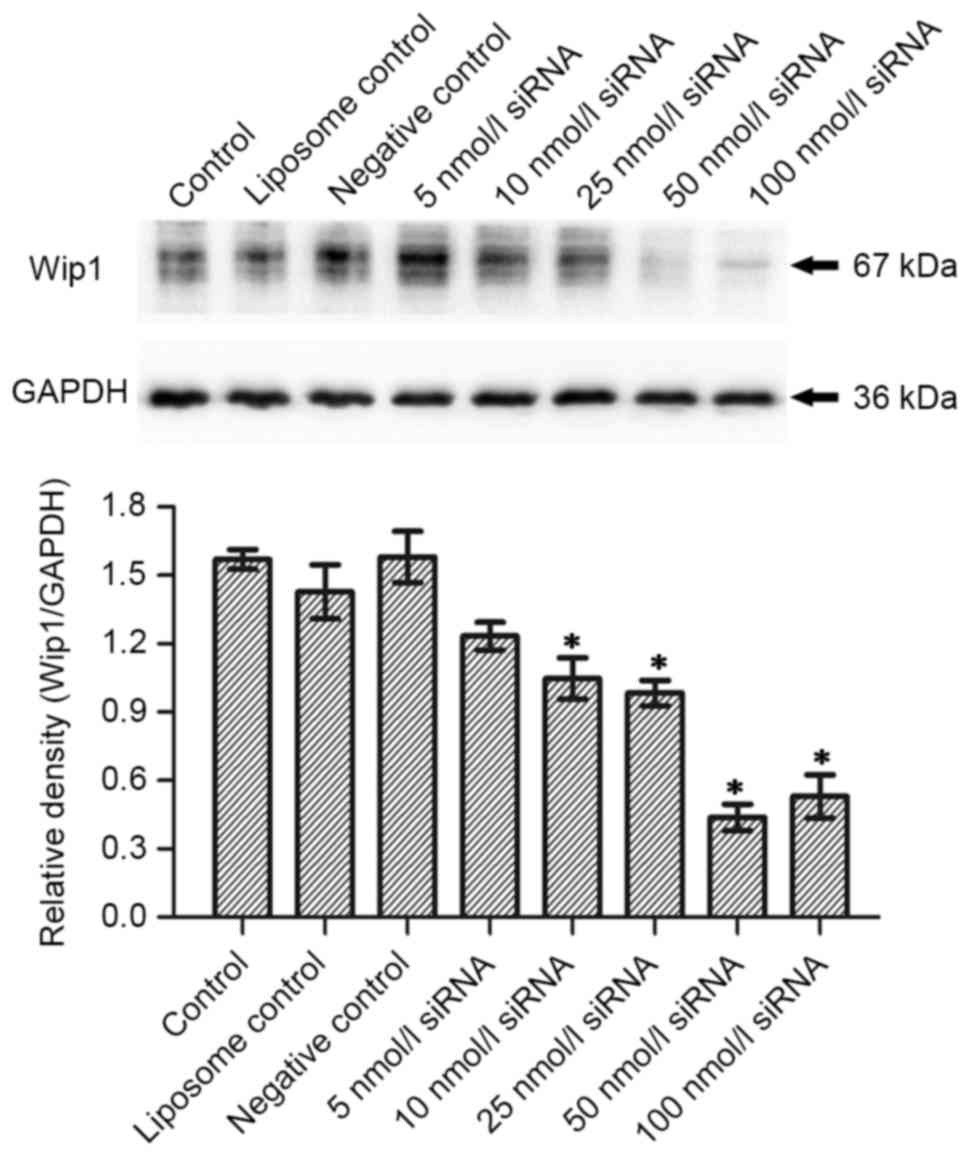
1B). No significant differences were observed in Wip1 protein
expression between the control, liposome control and negative
control groups 24 h after transfection (Fig. 2A). However, there was a significant
difference between the Wip1 siRNA groups, except for 5 nmol/l
siRNA, and the negative control group (P<0.05). This indicated
that a Wip1 siRNA range of 10–100 nmol/l inhibits Wip1 protein
expression 24 h after transfection. Wip1 protein expression was the
lowest when the concentration of Wip1-811 siRNA was 50 nmol/l
(Fig. 2A). The results from western
blot at 48 h post-transfection were similar to those at 24 h
post-transfection, except for 5 nmol/l siRNA (Fig. 2B). Therefore, the optimal
concentration of Wip1-811 siRNA appeared to be 50 nmol/l based on
the results from 24 and 48 h post-transfection.
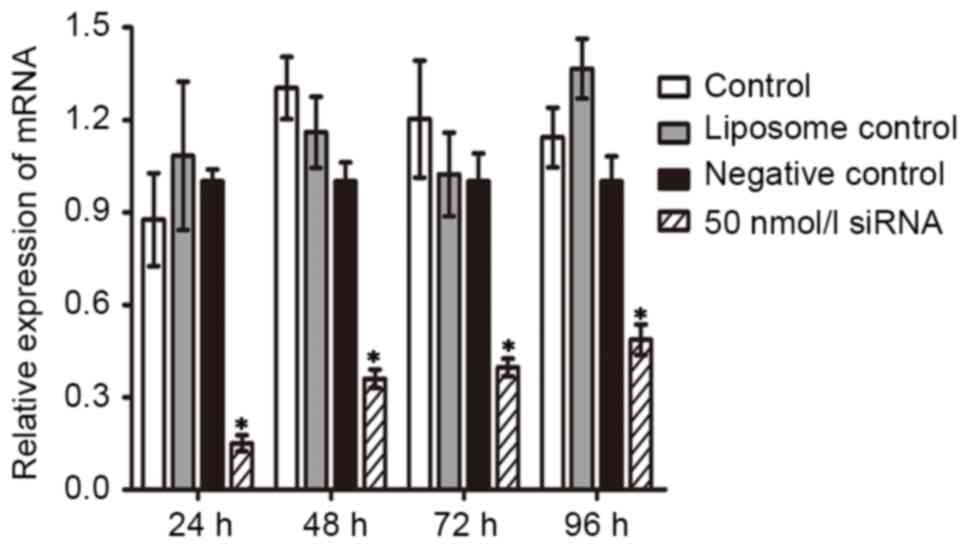
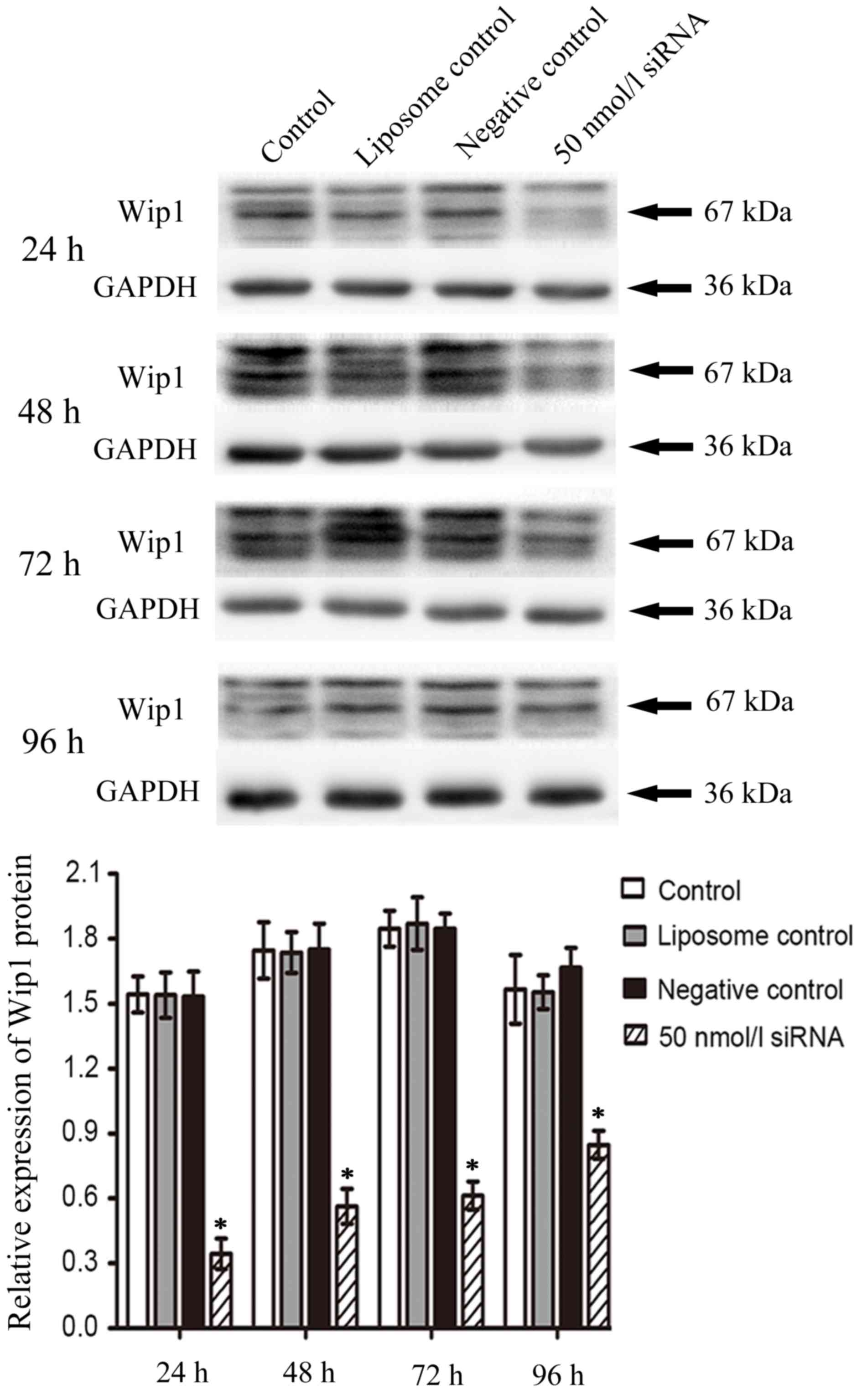
Duration of the inhibitory effect of
Wip1 siRNA on the mRNA and protein expression of Wip1 in RKO colon
cancer cells
In order to investigate the duration of the
inhibition of Wip1 siRNA on the mRNA and protein expression of Wip1
following transient transfection, 50 nmol/l of Wip1-811 siRNA was
transfected into RKO colon cancer cells. Cells were harvested at
24–96 h post-transfection. As shown by RT-qPCR in Fig. 3, the relative expression of Wip1 mRNA
in RKO colon cancer cells in the Wip1 siRNA groups was
significantly lower than that in negative control groups at any
time point. In addition, the longer the time post-transfection, the
lower the inhibitory effect of Wip1-811 siRNA. The western blot
analysis was consistent with the results from RT-qPCR (Fig. 4). According to the above results, the
optimal time of transfection of Wip1-811 siRNA was determined to be
between 24 and 48 h.
Wip1 gene silencing enhances the
chemosensitivity of RKO colon cancer cells
Wip1-811 siRNA (50 nmol/l) was transfected into RKO
colon cancer cells, and 24 h after transfection, serial
concentrations of 5-FU, oxaliplatin and adriamycin were added to
treat RKO colon cancer cells for 48 h. According to the results of
MTS assay, the cell viability of RKO colon cancer cells without
chemotherapy treatment in the Wip1-811 siRNA group exhibited no
significant difference compared with that of the control, liposome
control and negative control groups. The cell viability of RKO
colon cancer cells in these control groups and in the Wip1-811
siRNA group decreased along with increasing concentrations of the
antitumor drugs (data not shown). The IC50 of the
Wip1-811 siRNA group following treatment with 5-FU (25.32±2.59
µmol/l) was significantly decreased when compared with that of the
negative control group (56.88±6.08 µmol/l). Similarly, the
IC50 of the Wip1-811 siRNA group following treatment
with oxaliplatin (18.74±2.21 µmol/l) was significantly lower than
that of the negative control group (43.60±3.72 µmol/l). In
addition, the IC50 of the Wip1-811 siRNA group following
treatment with adriamycin (0.88±0.08 µmol/l) was significantly
lower than that of the negative control group (2.13±0.20 µmol/l)
(Table I).
 | Table I.IC50 of 5-FU, oxaliplatin and
adriamycin in different groups. |
Table I.
IC50 of 5-FU, oxaliplatin and
adriamycin in different groups.
|
| IC50
(µmol/l, mean ± standard deviation) |
|---|
|
|
|
|---|
| Group | 5-FU | Oxaliplatin | Adriamycin |
|---|
| Control | 66.38±9.26 | 45.08±4.74 | 2.22±0.21 |
| Liposome control | 59.95±6.58 | 41.80±3.74 | 2.01±0.20 |
| Negative control | 56.88±6.08 | 43.60±3.72 | 2.13±0.20 |
| Wip1-811siRNA (50
nmol/l) |
25.32±2.59a |
18.74±2.21b |
0.88±0.08c |
The results presented in Table I demonstrate that Wip1-811 siRNA alone
was not able to kill RKO colon cancer cells. However, a larger
number of RKO colon cancer cells were killed when treated with
Wip1-811 siRNA combined with 5-FU, oxaliplatin and adriamycin
compared with those in the negative control group.
Downregulation of Wip1 gene expression
increases cell apoptosis induced by chemotherapeutic drugs
In order to investigate the mechanism of Wip1 gene
silencing on the enhanced chemosensitivity of RKO colon cancer
cells, based on the results of MTS assay, 5 µmol/l of 5-FU and
oxaliplatin were applied to treat RKO colon cancer cells for 48 h
after transfection, and cell apoptosis was then detected by flow
cytometry. Figs. 5 and 6 indicate that both 5-FU and oxaliplatin
could induce cell apoptosis in RKO colon cancer cells. Wip1-811
siRNA alone could not induce cell apoptosis in RKO colon cancer
cells without treatment of chemotherapeutic drug. However, Wip1-811
siRNA could increase the cell apoptosis of RKO colon cancer cells
when in combination with antitumor drugs, compared with that of the
negative control. The results indicate that the downregulation of
Wip1 gene expression could increase the cell apoptosis of RKO colon
cancer cells induced by antitumor drugs.
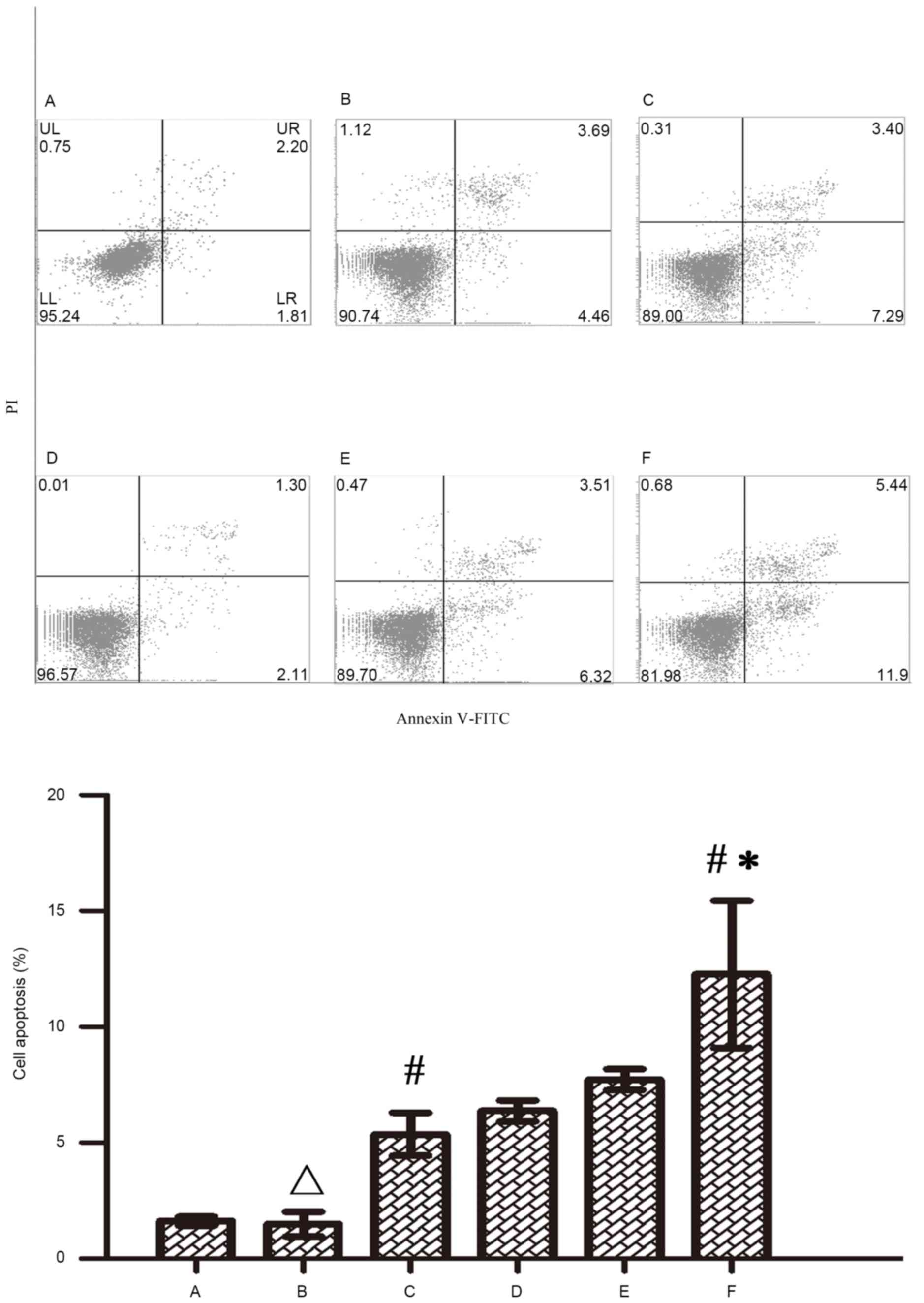 | Figure 5.Influence of Wip1-811 siRNA on cell
apoptosis in RKO colon cancer cells treated with 5-FU. A, Control;
B, Control+5-FU; C, Negative control+5-FU; D, Wip1-811 siRNA; E,
Liposome+5-FU; F, Wip1-811 siRNA+5-FU. ∆P>0.05 vs.
control; #P<0.05 vs. Wip1-811 siRNA group; *P<0.05
vs. negative control. Wip1, wild-type p53-induced phosphatase;
siRNA, small interfering RNA; 5-FU, 5-fluorouracil; Annexin V-FITC,
Annexin V-fluorescein isothiocyanate; PI, Propidium iodide; UL,
upper left; UR, upper right; LL, lower left; LR, lower right. |
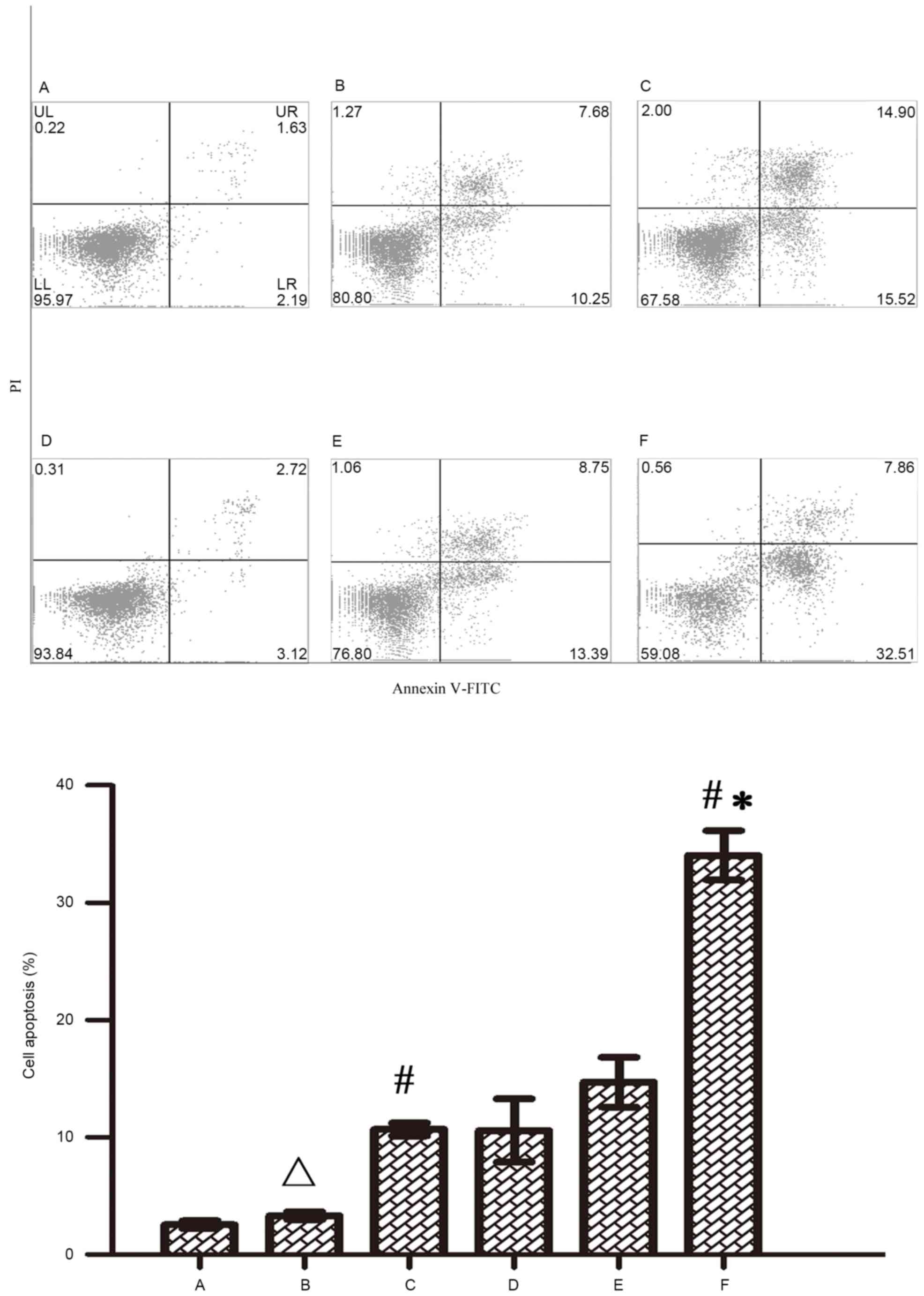 | Figure 6.Influence of Wip1-811 siRNA on cell
apoptosis in RKO colon cancer cells treated with oxaliplatin. A,
Control; B, Control+oxaliplatin; C, Negative control+oxaliplatin;
D, Wip1-811 siRNA; E, Liposome+5oxaliplatin; F, Wip1-811
siRNA+oxaliplatin. ∆P>0.05 vs. control;
#P<0.05 vs. Wip1-811 siRNA group; *P<0.05 vs.
negative control. Wip1, wild-type p53-induced phosphatase; siRNA,
small interfering RNA; Annexin V-FITC, Annexin V-fluorescein
isothiocyanate; PI, Propidium iodide; UL, upper left; UR, upper
right; LL, lower left; LR, lower right. |
Influence of Wip1 gene silencing on
the cell cycle of RKO colon cancer cells
As shown in Table II,
there was no significant difference in the cell cycle between the
siRNA group and the control group (P>0.05). This result
indicates that Wip1-811 siRNA by itself had no influence on the
cell cycle of RKO colon cancer cells in the absence of antitumor
drugs. 5-FU could induce RKO colon cancer cell arrest in G1 phase.
Therefore, 5-FU could increase the percentage of cells in G1 phase
and decrease the percentage of cells in S phase (P<0.05).
Oxaliplatin could induce RKO colon cancer cell arrest in G2/M phase
(P<0.05). However, the percentage of cells in each cell cycle
phase was not changed following Wip1 siRNA transfection in
combination with 5-FU or oxaliplatin treatment compared with that
following 5-FU or oxaliplatin treatment alone. This result
indicates that Wip1 gene silencing has no significant influence on
the cell cycle of RKO colon cancer cells induced by 5-FU or
oxaliplatin.
 | Table II.Influence of Wip1-811 siRNA on the
cell cycle of RKO colon cancer cells treated with antitumor
drugs. |
Table II.
Influence of Wip1-811 siRNA on the
cell cycle of RKO colon cancer cells treated with antitumor
drugs.
| Treatment | siRNA | G0/G1 phase (%, mean
± standard deviation) | S phase (%, mean ±
standard deviation) | G2/M phase (%, mean ±
standard deviation) |
|---|
| 5-Fluorouracil
(µmol/l) |
| 0 | Control | 50.5±7.3 | 35.0±2.5 | 14.6±7.0 |
|
| Wip-811 siRNA | 49.9±2.4a | 33.5±4.1 | 16.6±6.2 |
| 5 | Control |
60.9±3.8b | 28.9±1.5 | 10.3±5.2 |
|
| Wip-811 siRNA |
64.8±4.3c | 20.7±4.4 | 14.4±5.5 |
| Oxaliplatin
(µmol/l) |
| 0 | Control | 59.0±6.4 | 28.8±5.8 | 12.2±0.6 |
|
| Wip-811 siRNA | 54.5±4.6 | 33.8±5.3 |
11.7±1.7a |
| 5 | Control | 60.4±3.4 | 11.9±1.5 |
27.7±1.9b |
|
| Wip-811 siRNA | 58.5±3.1 | 12.3±2.7 |
28.2±6.7c |
Influence of Wip1 gene silencing on
intracellular adriamycin accumulation in RKO colon cancer
cells
To investigate if Wip1-811 siRNA could increase
intracellular antitumor drug accumulation, an intracellular
adriamycin accumulation assay was performed using COLO 320DM and
RKO colon cancer cells. Table III
shows that, when treated with adriamycin, the fluorescence
intensity of COLO 320DM colon cancer cells was 196.45 AU. However,
the fluorescence intensity increased to 410.63 AU when cells were
treated with adriamycin combined with the positive control #4123
MDR1 siRNA (P<0.05). As shown in Table III, when RKO colon cancer cells were
treated with adriamycin, adriamycin fluorescence could be detected
in all groups, including the control, liposome control and negative
control groups, as well as in the Wip1-811 siRNA group. However,
there was no significant difference in the fluorescence intensity
of adriamycin among these groups (P>0.05). This indicates that
Wip1-811 siRNA had no effect on intracellular adriamycin
accumulation in RKO colon cancer cells.
 | Table III.Fluorescence intensity of adriamycin
in RKO colon cancer cells following transfection with Wip1-811
siRNA. |
Table III.
Fluorescence intensity of adriamycin
in RKO colon cancer cells following transfection with Wip1-811
siRNA.
| Group | Fluorescence
intensity of adriamycin (AU, mean ± standard deviation) |
|---|
| Control | 395.23±29.85 |
| Liposome
control | 425.21±18.91 |
| Negative
control |
431.19±6.28a |
| Wip1-811 siRNA |
441.61±23.49b |
| COLO 320DM colon
cancer cells | 196.45±20.08 |
| Positive
control |
410.63±30.57c |
Discussion
The p53 gene is one of the most important
tumor-suppressor genes, and its mutation and functional
inactivation exist in more than half of human cancers (17). The intact function of p53 is crucial
to prevent cancer development. Wip1, which is encoded by protein
phosphatase magnesium-dependent 1 delta, is a serine/threonine
protein phosphatase belonging to the type 2C protein phosphatase
family (7). Wip1 is activated by
various stresses, and is involved in several cellular processes
such as tumorigenesis and aging (18). Wip1 is closely associated with p53
(8). Wip1, which is overexpressed in
numerous cancers, including breast cancer (19), medulloblastoma (20), pancreatic neuroendocrine tumor
(21), liver cancer (22) and stomach cancer (23), is recognized as a novel oncogene
inhibiting several p53-dependent tumor-suppressor signaling
pathways, including the Ataxia telangiectasia mutated-checkpoint
kinase 2-p53, p38 mitogen-activated protein kinase-p53 and nuclear
factor-κB signaling pathways (19).
Discher et al (14) reported
that Wip1 was associated with the chemosensitivity of cancer, since
downregulation of Wip1 gene expression could inhibit the
self-proliferation of breast cancer stem cells and enhance the
chemosensitivity of MCF-7 breast cancer cells to doxorubicin.
However, the influence of Wip1 gene on the chemosensitivity of
colon cancer cells is still unclear.
In the present study, Wip1-811 siRNA targeting Wip1
mRNA was transfected into RKO colon cancer cells to investigate the
influence of Wip1 gene silencing on the chemosensitivity of colon
cancer cells. It was observed that Wip1-811 siRNA could effectively
suppress Wip1 gene expression in RKO colon cancer cells. MTS assay
revealed that Wip1-811 siRNA alone had no influence on the cell
proliferation of RKO colon cancer cells. However, Wip1-811 siRNA
combined with 5-FU, oxaliplatin or adriamycin could kill more RKO
colon cancer cells than treatment with 5-FU, oxaliplatin or
adriamycin alone. By comparing the IC50 of 5-FU,
oxaliplatin or adriamycin in each group, it was observed that the
IC50 values of these drugs in the Wip1 siRNA group were
significantly lower than those in the three control groups. This
indicates that Wip1 gene silencing could enhance the
chemosensitivity of RKO colon cancer cells.
Cell apoptosis and cell cycle analyses by flow
cytometry revealed that Wip1-811 siRNA alone had no influence on
cell proliferation, cell apoptosis or cell cycle. However, when
combined with antitumor drugs, the cell viability of RKO colon
cancer cells in the Wip1 siRNA group was significantly lower than
that in the control groups. In addition, the cell apoptosis of RKO
colon cancer cells in the Wip1 siRNA group was significantly
increased compared with that in the control groups. There was no
significant difference on the cell cycle of RKO colon cancer cells
in the Wip1 siRNA group and the other groups. This indicated that
Wip1 siRNA enhanced the chemosensitivity of RKO colon cancer cells
via augmentation of cell apoptosis but not alteration of the cell
cycle. Nopel-Dünnebacke et al (24) reported that the downregulation of Wip1
gene expression could increase the p53 expression level and
directly induce cell apoptosis. High expression levels of p53 could
improve the outcome of chemotherapy for patients with colon cancer
who accepted regimens based on 5-FU and oxaliplatin. Bisteau et
al (25) reported that the
inhibition of Wip1 gene expression could increase the accumulation
of wild-type p53 protein and induce the cyclin D1 expression, which
was accompanied by an increased in the percentage of cells in S
phase. Therefore, the inhibition of Wip1 gene expression enhanced
the chemosensitivity of liver cancer cells. However, the present
study demonstrated that the downregulation of Wip1 gene expression
alone could not increase cell apoptosis or change the cell cycle of
RKO colon cancer cells. Thus, the alternative mechanism of enhanced
chemosensitivity in RKO colon cancer cells by Wip1 gene silencing
still requires to be further investigated. Brazina et al
(26) proposed that Wip1 was one of
the negative feedback key factors of death-associated protein 6
(Daxx) phosphorylation. High expression of Wip1 could inhibit Daxx
phosphorylation induced by DNA damage and prevent cell apoptosis
from occurring, thus increasing the resistance of cells to
stimulation (26). Wip1 gene
silencing may inhibit the self-proliferation of cancer stem cells
and therefore participate in the regulation of the chemosensitivity
of cancer cells (27).
The classical reversal of multidrug resistance is
targeting drug efflux pumps to increase intracellular antitumor
drug accumulation in cancer cells. However, in the present study,
intracellular adriamycin accumulation assay by flow cytometry
revealed that there was no significant difference between the
Wip1-811 siRNA group and the three control groups. Therefore, it
can be inferred that Wip1 gene silencing enhanced the
chemosensitivity of RKO colon cancer cells not via drug efflux
pumps.
In the present study, Wip1 siRNA was transiently
transfected into RKO colon cancer cells to successfully inhibit the
gene expression of Wip1. The downregulation of Wip1 gene expression
could enhance the chemosensitivity of RKO colon cancer cells, which
provided a new potential approach for the reversal of multidrug
resistance in colon cancer. However, siRNA transient transfection
has certain disadvantages such unsatisfactory inhibition of the
target gene and short time effects (5). Furthermore, the results of the present
study were obtained in vitro. In the future, a stable
expression vector for Wip1-811 siRNA must be constructed.
Furthermore, long-term, stable RNAi by Wip1-811 siRNA in
vivo must also be investigated.
In conclusion, Wip1-811 siRNA could induce Wip1 gene
silencing. Wip1-811 siRNA could enhance the chemosensitivity of RKO
colon cancer cells via augmentation of cell apoptosis but not
alteration of the cell cycle or intracellular antitumor drug
accumulation in RKO colon cancer cells.
Acknowledgements
The present study was supported by grant no.
[2013]163 from the Key Laboratory of Malignant Tumor Molecular
Mechanism and Translational Medicine of Guangzhou Bureau of Science
and Information Technology, and grant no. KLB09001 from the Key
Laboratory of Malignant Tumor Gene Regulation and Target Therapy of
Guangdong Higher Education Institutes.
References
|
1
|
Wiseman M: The second world cancer
research Fund/American institute for cancer research expert report:
Food, nutrition, physical activity, and the prevention of cancer: A
global perspective. Proc Nutr Soc. 67:pp. 253–256. 2008; View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Douillard JY, Cunningham D, Roth AD,
Navarro M, James RD, Karasek P, Jandik P, Iveson T, Carmichael J,
Alakl M, et al: Irinotecan combined with fluorouracil compared with
fluorouracil alone as first-line treatment for metastatic
colorectal cancer: A multiple multicentre randomised trial. Lancet.
355:1041–1047. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Redmond SM, Joncourt F, Buser K, Ziemiecki
A, Altermatt HJ, Fey M, Margison G and Cerny T: Assessment of
P-glycoprotein, glutathione-based detoxifying enzymes and
O6-alkylguanine-DNA alkyltransferase as potential indicators of
constitutive drug resistance in human colorectal tumors. Cancer
Res. 51:2092–2097. 1991.PubMed/NCBI
|
|
4
|
Van de Vrie W, Gheuens EE, Durante NM, De
Bruijn EA, Marquet RL, Van Oosterom AT and Eggermont AM: In vitro
and in vivo chemosensitizing effect of cyclosporine A on an
intrinsic multidrug-resistant rat colon tumour. J Cancer Res Clin
Oncol. 119:609–614. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Xia Z, Zhu Z, Zhang L, Royal C, Liu Z,
Chen Q and Adam BL: Specific reversal of MDR1/P-gp-dependent
multidrug resistance by RNA interference in colon cancer cells.
Oncol Rep. 20:1433–1439. 2008.PubMed/NCBI
|
|
6
|
Damiani D, Michieli M, Michelutti A, Melli
C, Cerno M and Baccarani M: D-verapamil downmodulates
P170-associated resistance to doxorubicin, daunorubicin and
idarubicin. Anticancer Drugs. 4:173–180. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Fiscella M, Zhang H, Fan S, Sakaguchi K,
Shen S, Mercer WE, Woude GF Vande, O'Connor PM and Appella E: Wip1,
a novel human protein phosphatase that is induced in response to
ionizing radiation in a p53-dependent manner. Proc Natl Acad Sci
USA. 94:pp. 6048–6053. 1997; View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Shi Y: Serine/threonine phosphatases:
Mechanism through structure. Cell. 139:468–484. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Donehower LA: Phosphatases reverse
p53-mediated cell cycle checkpoints. Proc Natl Acad Sci USA.
111:pp. 7172–7173. 2014; View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lu X, Nannenga B and Donehower LA: PPM1D
dephosphorylates Chk1 and p53 and abrogates cell cycle checkpoints.
Genes Dev. 19:1162–1174. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Demidov ON, Timofeev O, Lwin HN, Kek C,
Appella E and Bulavin DV: Wip1 phosphatase regulates p53-dependent
apoptosis of stem cell and tumorigenesis in the mouse intestine.
Cell Stem Cell. 1:180–190. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Li ZT, Zhang L, Gao XZ, Jiang XH and Sun
LQ: Expression and significance of the Wip1 proto-oncogene in
colorectal cancer. Asian Pac J Cancer Prev. 14:1975–1979. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhao M, Zhang H, Zhu G, Liang J, Chen N,
Yang Y, Liang X, Cai H and Liu W: Association between
overexpression of Wip1 and prognosis of patients with non-small
cell lung cancer. Oncol Lett. 11:2365–2370. 2016.PubMed/NCBI
|
|
14
|
Discher DE, Janmey P and Wang YL: Tissue
cells feel and repond to the stiffness of their substrate. Science.
310:1139–1143. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li J, Xu LZ, He KL, Guo WJ, Zheng YH, Xia
P and Chen Y: Reversal effects of nomegestrol acetate on multidrug
resistance in adriamycin-resistant MCF7 breast cancer cell line.
Breast Cancer Res. 3:253–263. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chang F, Syrjänen S, Kurvinen K and
Syrjänen K: The p53 tumor suppressor gene as a common cellular
target in human carcinogenesis. Am J Gastroenterol. 88:174–186.
1993.PubMed/NCBI
|
|
18
|
Lu G and Wang Y: Functional diversity of
mammalian type 2C protein phosphatase isoforms: New tales from an
old family. Clin Exp Pharmacol Physiol. 35:107–112. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yu E, Ahn YS, Jang SJ, Kim MJ, Yoon HS,
Gong G and Choi J: Overexpression of the Wip1 gene abrogates the
p38 MAPK/p53/Wip1 pathway and silences p16 expression in human
breast cancers. Breast Cancer Res Treat. 101:269–278. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Baxter EW and Milner J: p53 regulates LIF
expression in human medulloblastoma cells. J Neurooncol.
97:373–382. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hu W, Feng Z, Modica I, Klimstra DS, Song
L, Allen PJ, Brennan MF, Levine AJ and Tang LH: Gene Amplifications
in well-differentiated pancreatic neuroendocrine tumors inactivate
the P53 pathway. Genes Cancer. 1:360–368. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhu YH and Bulavin DV: Wip1-dependent
signaling pathways in health and diseases. Prog Mol Biol Transl
Sci. 106:307–325. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Fuku T, Semba S, Yutori H and Yokozaki H:
Increased wild-type p53-induced phosphatase 1 (Wip1 or PPM1D)
expression correlated with downregulation of checkpoint kinase 2 in
human gastric carcinoma. Pathol Int. 57:566–571. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Nöpel-Dünnebacke S, Schulmann K,
Reinacher-Schick A, Porschen R, Schmiegel W, Tannapfel A and
Graeven U: Prognostic value of microsatellite instability and p53
expression in metastatic colorectal cancer treated with oxaliplatin
and fluoropyrimidine-based chemotherapy. Z Gastroenterol.
52:1394–1401. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Bisteau X, Caldez MJ and Kaldis P: The
complex relationship between liver cancer and the cell cycle: A
story of multiple regulations. Cancers (Basel). 6:79–111. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Brazina J, Svadlenka J, Macurek L, Andera
L, Hodny Z, Bartek J and Hanzlikova H: DNA damage-induced
regulatoryinterplay between DAXX, p53, ATM kinase and Wip1
phosphatase. Cell Cycle. 14:375–387. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhang X, Wan G, Mlotshwa S, Vance V,
Berger FG, Chen H and Lu X: Oncogenic Wip1 phosphatase is inhibited
by miR-16 in the DNA damage signaling pathway. Cancer Res.
70:7176–7186. 2010. View Article : Google Scholar : PubMed/NCBI
|