Introduction
Alpha-fetoprotein (AFP)-producing gastric cancer
(AFPGC) is a relatively rare type of malignancy, which comprises
1–8% of all gastric cancers (1–3). AFPGC is
characterized by a high incidence of metastases to the liver and
lymph nodes, and a poor prognosis (1–3).
Currently, there is no standard therapy for this challenging
subtype of gastric cancer. Histopathologically, AFPGC is known to
have high proliferative activity, reduced apoptotic rate and rich
neovascularization, which influence the clinical aggressiveness of
the disease (3,4). High expression of vascular endothelial
growth factor (VEGF) is considered one of the main reasons for the
rich neovascularization in AFPGC (3–5).
Over the past few decades, molecular-targeted
therapy has led to improved survival of patients with various types
of cancer, including gastric cancer (6). A humanized anti-human epidermal growth
factor receptor 2 (HER2) monoclonal antibody (namely trastuzumab)
in combination with chemotherapy demonstrated a survival benefit
for patients with advanced HER2-positive gastric cancer (7). A monoclonal antibody for VEGF receptor-2
(namely ramucirumab) also demonstrated survival benefit for
patients with advanced gastric cancer following first-line
chemotherapy (8,9).
Ramucirumab binds to VEGF receptor-2 and inhibits
VEGF-induced angiogenesis (8,9). Ramucirumab is the first anti-angiogenic
drug approved for the treatment of advanced gastric cancer;
however, there is no validated predictor for selecting patients who
will benefit from ramucirumab therapy to date (8,9). Although
AFPGC has rich neovascularization (3,4), the
clinical efficacy of ramucirumab in patients with AFPGC has not yet
been reported. In the current case report, the case of a
chemotherapy-resistant recurrent AFPGC patient who received
ramucirumab monotherapy is presented.
Case report
A 56-year-old man with upper abdominal pain was
referred to Jikei University Hospital (Tokyo, Japan) in January
2014. The patient had no significant family or medical history,
except well-controlled hypertension. Gastrointestinal endoscopy
revealed a 5-cm elevated tumor with a central depression (Borrmann
type 3) on the posterior wall of the gastric body, which was
histologically diagnosed as moderately differentiated tubular
adenocarcinoma. HER2 immunostaining was performed using the
HercepTest kit (Dako; Agilent Technologies, Inc., Santa Clara, CA,
USA) and scored as 1+, according to the manufacturer's instruction
(10). Computed tomography (CT)
revealed thickening of the gastric wall and enlargement of lymph
nodes on the lesser curvature. There were no signs of liver
metastases or liver cirrhosis. The results of a complete blood
count and blood chemistry were as follows: Hemoglobin 14.4 g/dl
(normal range, 13.5–16.5 g/dl), white blood cell count 6,300/µl
(normal, 3,300-8,600/µl), platelet count 227,000/µl (normal,
150,000-350,000/µl), aspartate aminotransferase 22 U/l (normal,
10–33 U/l), alanine aminotransferase 14 U/l (normal, 6–35 U/l),
total protein 7.3 g/dl (normal, 6.7–8.3 g/dl), albumin 4.2 g/dl
(normal, 3.5–5.2 g/dl), blood urea nitrogen 11 mg/dl (normal, 8–20
mg/dl), creatinine 0.55 mg/dl (normal, 0.50–1.10 mg/dl), and
C-reactive protein (CRP) 0.08 mg/dl (normal, 0–0.30 mg/ml).
Hepatitis B surface antigen and hepatitis C antibody were negative.
The patient's serum AFP and carcinoembryonic antigen (CEA) levels
were increased to 910 ng/ml (normal range, 0–10 ng/ml) and 17.0
ng/ml (normal range, 0.0–5.8 ng/ml), respectively.
The patient received two courses of neoadjuvant
chemotherapy with S-1 (tegafur/gimeracil/oteracil potassium) plus
cisplatin (11), which resulted in
stable disease, and then underwent distal gastrectomy with D2 lymph
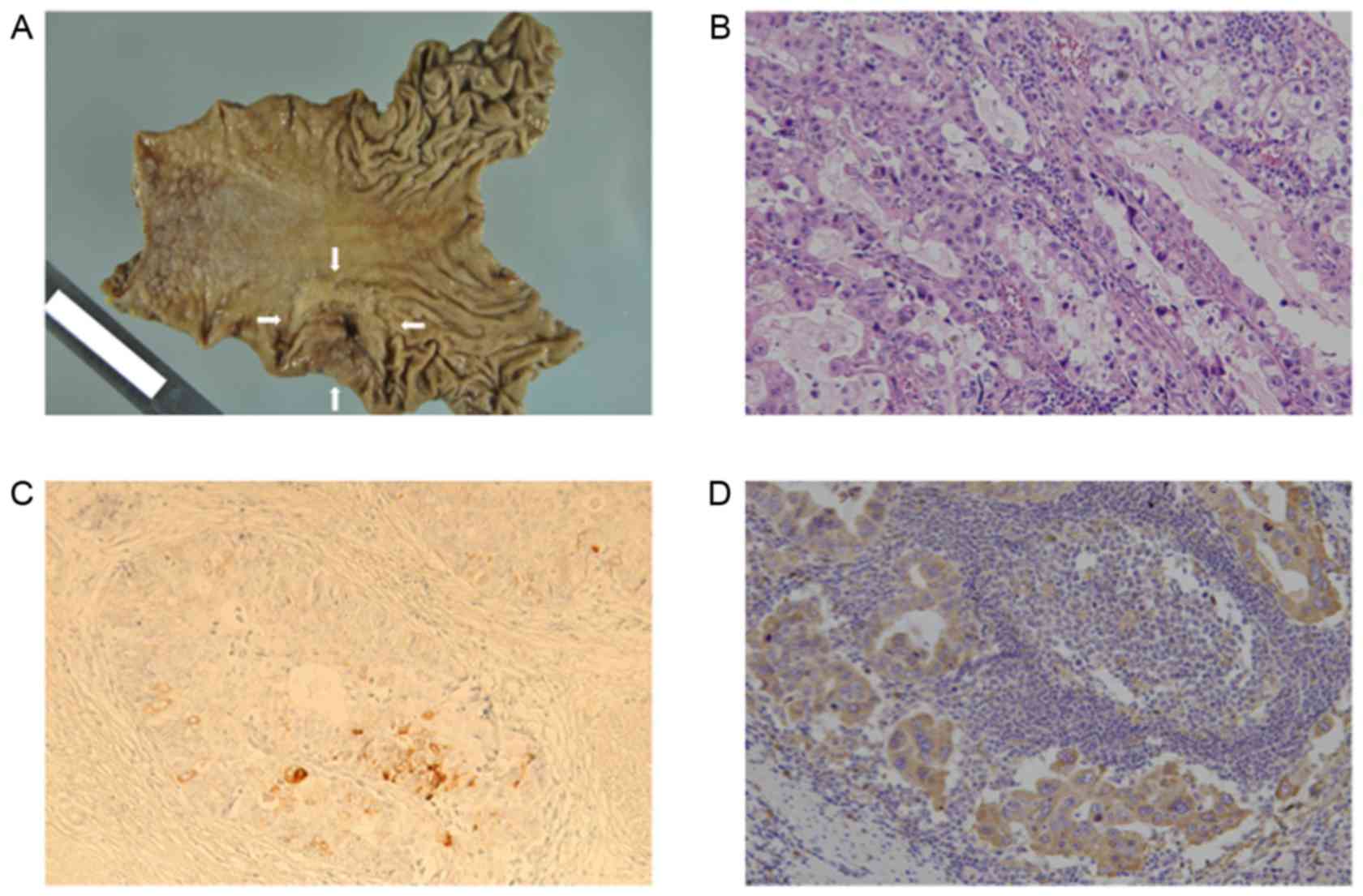
node dissection (Fig. 1A).
Histopathologically, the tumor was predominantly composed of well-
to moderately-differentiated adenocarcinoma with papillary and
tubular proliferation (Fig. 1B). The
specimen also contained minor components of poorly differentiated
adenocarcinoma with signet-ring cells. The typical histological
feature of AFPGC is a liver-like structure called a ‘hepatoid’, but
the specimen did not contain a hepatoid component (12). Immunohistological staining for AFP and
VEGF was performed by the avidin-biotin complex (ABC) method.
Following deparaffinization and rehydration, 4-µm thick sections
were treated with 0.3% H2O2 for 30 min and
blocked with 1% bovine serum albumin in phosphate-buffered saline
(PBS) for 30 min. Then, sections were rinsed in PBS and treated
with antibodies against AFP (dilution, 1:100; cat. no A008; Dako;
Agilent Technologies, Inc.) or VEGF (dilution, 1:100; cat. no R11;
Immuno-Biological Laboratories Co., Ltd., Gunma, Japan) for 1 h at
room temperature in a humidified chamber, followed by incubation
with biotinylated IgG (dilution, 1:100; Vector Laboratories,
Burlingame, CA, USA) for 30 min at room temperature. The sections
were developed using ABC-peroxidase and diaminobenzidine (both
Vector Laboratories). AFP-positive foci were observed (Fig. 1C) and VEGF expression was positive in
the cytoplasm of the tumor cells (Fig.
1D). The final stage was T3, N2, M0, stage IIIA, according to
the 7th edition of the Union for the International Cancer Control
classification (13), and a curative
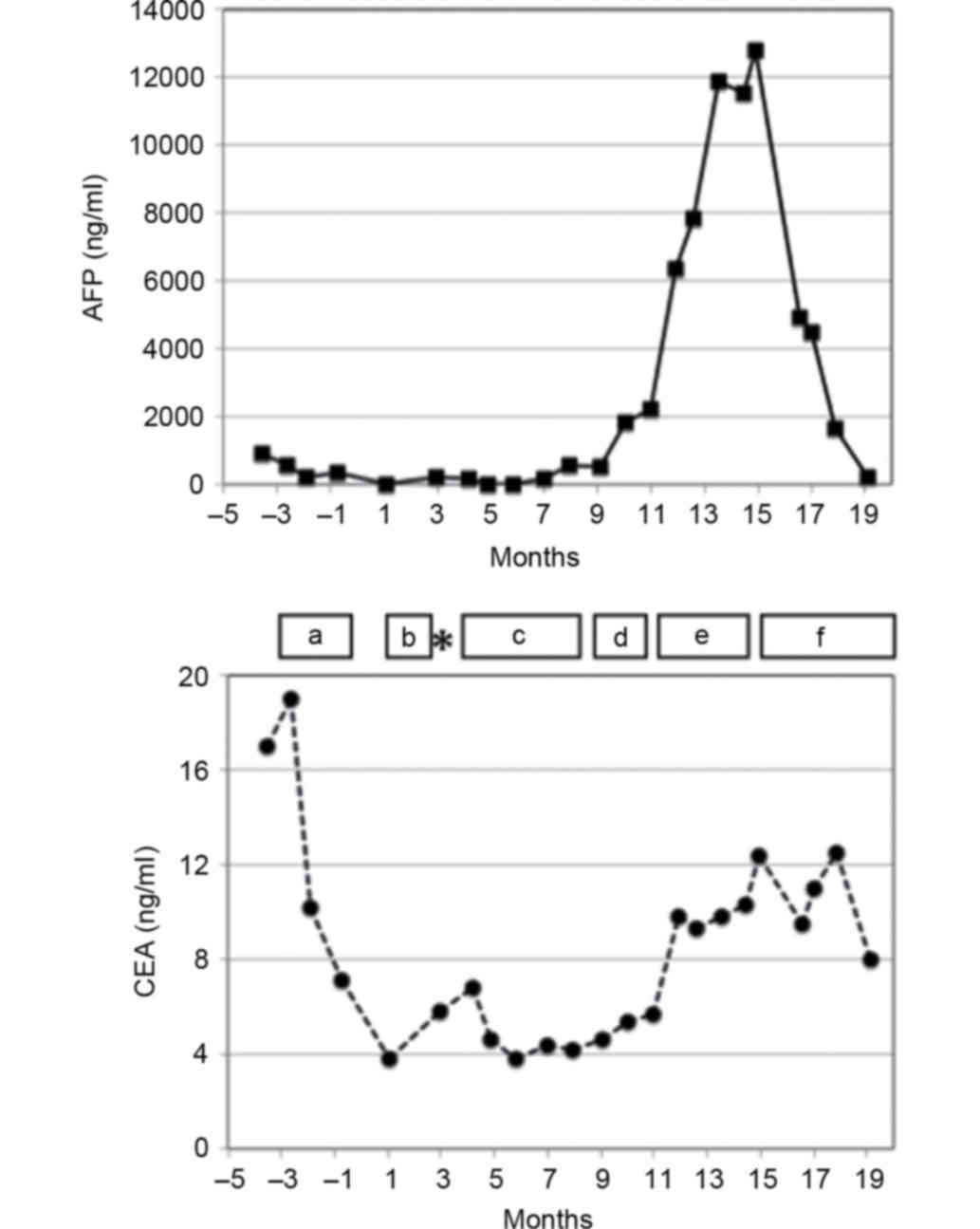
resection was confirmed. A month after the surgery, the patient's
serum AFP and CEA levels were decreased to 15 and 3.8 ng/ml,
respectively. The patient was started on adjuvant therapy with S-1
(14) (Fig.
2).
Following a course of S-1 therapy (3 months after
the surgery), CT examination revealed a 22-mm para-aortic lymph
node. The serum AFP levels were increased to 207 mg/ml, and the
patient was diagnosed as having a recurrence of AFPGC. The patient
was then treated with five courses of weekly paclitaxel (14), three courses of irinotecan (15), and two courses of sequential
methotrexate-5-fluorouracil chemotherapy (16). However, CT scan revealed that the
metastatic para-aortic lymph node was gradually increasing in size,
while the serum AFP level was increased to a maximum of 12,800
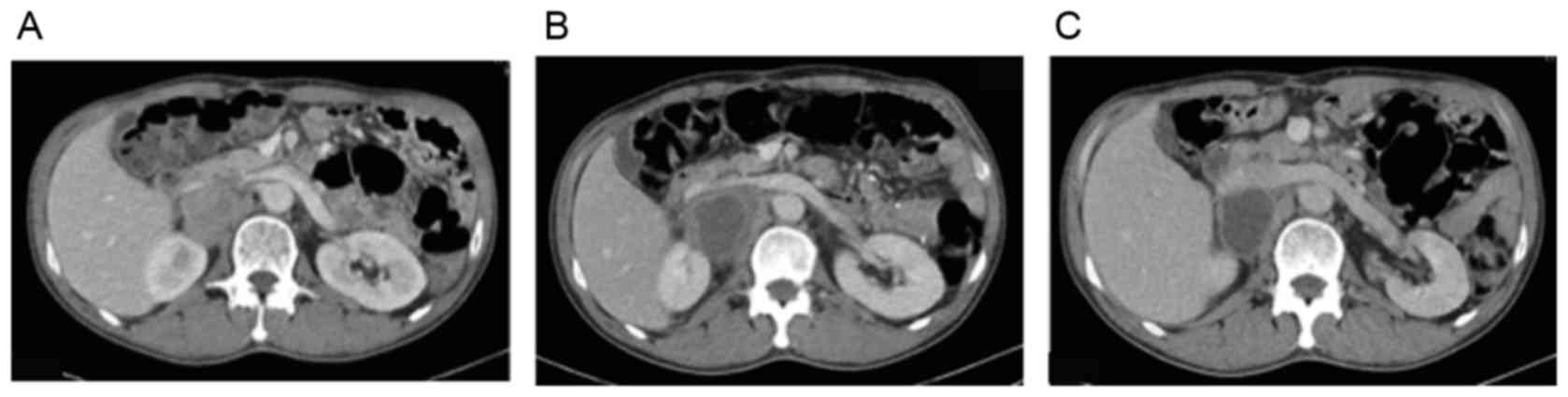
ng/ml (Figs. 2 and 3A).
The patient was then treated with ramucirumab
monotherapy (7). Following six doses
of ramucirumab, the patient presented with back pain and low-grade
fever. Laboratory tests revealed an increased CRP level, at 11.23
mg/dl (normal range, 0–0.30 mg/ml). Contrast-enhanced CT revealed a
larger para-aortic lymph node with adjacent fat stranding, in
addition to a significant decrease of enhancement in the middle of
the lymph node, suggesting attenuation of vascularity (Fig. 3B). The patient was prescribed
celecoxib for his back pain, and ramucirumab was discontinued for a
month. Follow-up CT revealed a marked reduction of enhancement in
the lymph node, suggesting an episode of central necrosis, while
the serum AFP level decreased rapidly to 225 ng/ml (Figs. 2 and 3C). The patient proceeded with ramucirumab
and has so far survived for 16 months after recurrence. Written
informed consent was obtained from the patient for publication of
this case report.
Discussion
AFPGC is considered a highly aggressive type of
gastric cancer. The 5-year survival rate and median survival time
are 42% and 29 months, respectively, even in patients who undergo
curative surgical resection (17).
Although molecular-targeted therapy has led to survival improvement
of patients with various types of cancer (7,18–20) there is no validated application of
tailored therapy for this challenging subtype of gastric cancer. In
the present case, the tumor cells were positive for VEGF. AFPGC is
known to express high levels of VEGF or its isoform VEGF-C, and is
considered to have increased neovascularization (4,5,21). Since VEGF and VEGF-C serve as
angiogenic factors in the tumor microenvironment (22), their inhibition may exhibit
anti-angiogenic and anti-tumor activities on AFPGC. However, only a
few cases of the efficacy of an anti-angiogenic multi-kinase
inhibitor (namely sorafenib) in AFPGC patients have been reported
thus far (23,24). The clinical efficacy of ramucirumab, a
monoclonal antibody for VEGF receptor-2, in AFPGC patients has not
yet been reported. Sorafenib inhibits the signaling of the VEGF
receptor and the platelet-derived growth factor receptor, but its
specific targets are not fully understood yet. Sorafenib also
inhibits the activity of the Raf/mitogen-activated protein kinase
kinase/extracellular signal-regulated kinase and Janus
kinase/Signal Transducers and Activator of Transcription signaling
pathways (25,26). Therefore, it is not clear that the
efficacy of sorafenib in the previous AFPGC cases was due to its
anti-angiogenic properties or its direct pro-apoptotic effects.
In the present case, the patient's metastatic lymph
node exhibited marked central necrosis following six doses of
ramucirumab monotherapy, which is similar to the effect of
anti-angiogenic drugs on hepatocellular carcinoma (HCC), another
AFP-producing cancer (27). Sorafenib
has been reported to have a survival benefit in patients with
advanced HCC (28). However, the
response rate to sorafenib in that study was only 2%, according to
Response Evaluation Criteria in Solid Tumors (RECIST) (28). It has been demonstrated that the
change in vascularization of the lesion is the crucial point for
evaluating the response to anti-angiogenic drugs; thus, a modified
RECIST was proposed (27). According
to RECIST evaluation, the present patient's metastatic lymph node
was defined as stable disease without any change in its greatest
dimension, while according to the modified RECIST, the lesion was
defined as partial response because the maximal dimension of
continuous enhancement in the lesion was markedly reduced. In
second-line treatment of advanced HCC, ramucirumab may have a
survival benefit only for patients with high serum AFP levels
(29).
Recent advances in the understanding of molecular
pathogenesis have led to the development of molecular-targeted
therapy in various types of cancer, including gastric cancer
(7,18–20). In
first-line treatment of HER2-positive advanced gastric cancer, the
addition of trastuzumab to chemotherapy improves overall survival
(7). Ramucirumab improves survival in
the second-line setting of advanced gastric cancer; however,
predictive biomarkers have not yet been clearly defined (30). In the present case report, the patient
with chemotherapy-resistant recurrent AFPGC exhibited a significant
response to ramucirumab monotherapy. In line with previous reports
on AFPGC, the present patient's tumor cells were positive for VEGF.
AFP production in gastric cancer may represent one of the
predictors of ramucirumab sensitivity. The present case report
demonstrates a potential new treatment option for AFPGC.
References
|
1
|
Kono K, Amemiya H, Sekikawa T, Iizuka H,
Takahashi A, Fujii H and Matsumoto Y: Clinicopathologic features of
gastric cancers producing alpha-fetoprotein. Dig Surg. 19:359–365;
discussion 365. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Inoue M, Sano T, Kuchiba A, Taniguchi H,
Fukagawa T and Katai H: Long-term results of gastrectomy for
alpha-fetoprotein-producing gastric cancer. Br J Surg.
97:1056–1061. 2010. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Li XD, Wu CP, Ji M, Wu J, Lu B, Shi HB and
Jiang JT: Characteristic analysis of α-fetoprotein-producing
gastric carcinoma in China. World J Surg Oncol. 11:2462013.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Koide N, Nishio A, Igarashi J, Kajikawa S,
Adachi W and Amano J: Alpha-fetoprotein-producing gastric cancer:
Histochemical analysis of cell proliferation, apoptosis and
angiogenesis. Am J Gastroenterol. 94:1658–1663. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Takahashi Y, Ohta T and Mai M:
Angiogenesis of AFP producing gastric carcinoma: Correlation with
frequent liver metastasis and its inhibition by anti-AFP antibody.
Oncol Rep. 11:809–813. 2004.PubMed/NCBI
|
|
6
|
Yuan DD, Zhu ZX, Zhang X and Liu J:
Targeted therapy for gastric cancer: Current status and future
directions (Review). Oncol Rep. 35:1245–1254. 2016.PubMed/NCBI
|
|
7
|
Bang YJ, Van Cutsem E, Feyereislova A,
Chung HC, Shen L, Sawaki A, Lordick F, Ohtsu A, Omuro Y, Satoh T,
et al: Trastuzumab in combination with chemotherapy versus
chemotherapy alone for treatment of HER2-positive advanced gastric
or gastro-oesophageal junction cancer (ToGA): A phase 3,
open-label, randomised controlled trial. Lancet. 376:687–697. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Fuchs CS, Tomasek J, Yong CJ, Dumitru F,
Passalacqua R, Goswami C, Safran H, dos Santos LV, Aprile G, Ferry
DR, et al: Ramucirumab monotherapy for previously treated advanced
gastric or gastro-oesophageal junction adenocarcinoma (REGARD): An
international, randomised, multicentre, placebo-controlled, phase 3
trial. Lancet. 383:31–39. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wilke H, Muro K, Van Cutsem E, Oh SC,
Bodoky G, Shimada Y, Hironaka S, Sugimoto N, Lipatov O, Kim TY, et
al: Ramucirumab plus paclitaxel versus placebo plus paclitaxel in
patients with previously treated advanced gastric or
gastro-oesophageal junction adenocarcinoma (RAINBOW): A
double-blind, randomised phase 3 trial. Lancet Oncol. 15:1224–1235.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Rüschoff J, Hanna W, Bilous M, Hofmann M,
Osamura RY, Penault-Llorca F, van de Vijver M and Viale G: HER2
testing in gastric cancer: A practical approach. Mod Pathol.
25:637–650. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Okabe H, Hata H, Ueda S, Zaima M, Tokuka
A, Yoshimura T, Ota S, Kinjo Y, Yoshimura K and Sakai Y; Kyoto
University Surgical Oncology Group (KUSOG), : A phase II study of
neoadjuvant chemotherapy with S-1 and cisplatin for stage III
gastric cancer: KUGC03. J Surg Oncol. 113:36–41. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kinjo T, Taniguchi H, Kushima R, Sekine S,
Oda I, Saka M, Gotoda T, Kinjo F, Fujita J and Shimoda T:
Histologic and immunohistochemical analyses of
α-fetoprotein-producing cancer of the stomach. Am J Surg Pathol.
36:56–65. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
http://link.springer.com/article/10.1245/s10434-010-0985-4
|
|
14
|
Sakuramoto S, Sasako M, Yamaguchi T,
Kinoshita T, Fujii M, Nashimoto A, Furukawa H, Nakajima T, Ohashi
Y, Imamura H, et al: Adjuvant chemotherapy for gastric cancer with
S-1, an oral fluoropyrimidine. N Engl J Med. 357:1810–1820. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hironaka S, Ueda S, Yasui H, Nishina T,
Tsuda M, Tsumura T, Sugimoto N, Shimodaira H, Tokunaga S, Moriwaki
T, et al: Randomized, open-label, phase III study comparing
irinotecan with paclitaxel in patients with advanced gastric cancer
without severe peritoneal metastasis after failure of prior
combination chemotherapy using fluoropyrimidine plus platinum: WJOG
4007 trial. J Clin Oncol. 31:4438–4444. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hamaguchi T, Shirao K, Yamamichi N, Hyodo
I, Koizumi W, Seki S, Imamura T, Honma H, Ohtsu A, Boku N, et al: A
phase II study of sequential methotrexate and 5-fluorouracil
chemotherapy in previously treated gastric cancer: A report from
the gastrointestinal Oncology group of the Japan clinical oncology
group, JCOG 9207 trial. Jpn J Clin Oncol. 38:432–437. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Adachi Y, Tsuchihashi J, Shiraishi N,
Yasuda K, Etoh T and Kitano S: AFP-producing gastric carcinoma:
Multivariate analysis of prognostic factors in 270 patients.
Oncology. 65:95–101. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Coiffier B, Thieblemont C, Van den Neste
E, Lepeu G, Plantier I, Castaigne S, Lefort S, Marit G, Macro M,
Sebban C, et al: Long-term outcome of patients in the LNH-98.5
trial, the first randomized study comparing rituximab-CHOP to
standard CHOP chemotherapy in DLBCL patients: A study by the Groupe
d'Etudes des Lymphomes de l'Adulte. Blood. 116:2040–2045. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Mitsudomi T, Kosaka T, Endoh H, Horio Y,
Hida T, Mori S, Hatooka S, Shinoda M, Takahashi T and Yatabe Y:
Mutations of the epidermal growth factor receptor gene predict
prolonged survival after gefitinib treatment in patients with
non-small-cell lung cancer with postoperative recurrence. J Clin
Oncol. 23:2513–2520. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Van Cutsem E, Köhne CH, Láng I, Folprecht
G, Nowacki MP, Cascinu S, Shchepotin I, Maurel J, Cunningham D,
Tejpar S, et al: Cetuximab plus irinotecan, fluorouracil and
leucovorin as first-line treatment for metastatic colorectal
cancer: Updated analysis of overall survival according to tumor
KRAS and BRAF mutation status. J Clin Oncol. 29:2011–2019. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kamei S, Kono K, Amemiya H, Takahashi A,
Sugai H, Ichihara F, Fujii H and Matsumoto Y: Evaluation of VEGF
and VEGF-C expression in gastric cancer cells producing
alpha-fetoprotein. J Gastroenterol. 38:540–547. 2003.PubMed/NCBI
|
|
22
|
Ichikura T, Tomimatsu S, Ohkura E and
Mochizuki H: Prognostic significance of the expression of vascular
endothelial growth factor (VEGF) and VEGF-C in gastric carcinoma. J
Surg Oncol. 78:132–137. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
23
|
Koneri K, Hirono Y, Fujimoto D, Sawai K,
Morikawa M, Murakami M, Goi T, Iida A, Katayama K and Yamaguchi A:
Five-year survival of alpha-fetoprotein-producing gastric cancer
with synchronous liver metastasis: A case report. J Gastric Cancer.
13:58–64. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Fang YU, Wang L, Yang N, Gong X, Zhang YU
and Qin S: Successful multimodal therapy for an
α-fetoprotein-producing gastric cancer patient with simultaneous
liver metastases. Oncol Lett. 10:3021–3025. 2015.PubMed/NCBI
|
|
25
|
Wilhelm SM, Adnane L, Newell P, Villanueva
A, Llovet JM and Lynch M: Preclinical overview of sorafenib, a
multikinase inhibitor that targets both Raf and VEGF and PDGF
receptor tyrosine kinase signaling. Mol Cancer Ther. 7:3129–3140.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Blechacz BR, Smoot RL, Bronk SF, Werneburg
NW, Sirica AE and Gores GJ: Sorafenib inhibits signal transducer
and activator of transcription-3 signaling in cholangiocarcinoma
cells by activating the phosphatase shatterproof 2. Hepatology.
50:1861–1870. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Edeline J, Boucher E, Rolland Y, Vauléon
E, Pracht M, Perrin C, Le Roux C and Raoul JL: Comparison of tumor
response by Response Evaluation Criteria in Solid Tumors (RECIST)
and modified RECIST in patients treated with sorafenib for
hepatocellular carcinoma. Cancer. 118:147–156. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Llovet JM, Ricci S, Mazzaferro V, Hilgard
P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A,
et al: Sorafenib in advanced hepatocellular carcinoma. N Engl J
Med. 359:378–390. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhu AX, Park JO, Ryoo BY, Yen CJ, Poon R,
Pastorelli D, Blanc JF, Chung HC, Baron AD, Pfiffer TE, et al:
Ramucirumab versus placebo as second-line treatment in patients
with advanced hepatocellular carcinoma following first-line therapy
with sorafenib (REACH): A randomised, double-blind, multicentre,
phase 3 trial. Lancet Oncol. 16:859–870. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kim C, Mulder K and Spratlin J: How
prognostic and predictive biomarkers are transforming our
understanding and management of advanced gastric cancer.
Oncologist. 19:1046–1055. 2014. View Article : Google Scholar : PubMed/NCBI
|