SPARC-like protein 1 (SPARCL1) is a
potential tumor suppressor gene
SPARCL1, a member of the family of secreted proteins
that are acidic and rich in cysteine in the cellular matrix.
Originally termed SC1, SPARCL1 was first cloned from the rat
central nervous system and encodes an extracellular matrix
glycoprotein, similar to osteonectin/basement membrane protein
40/secreted protein acidic and rich in cysteine (SPARC) (1). Subsequently, Schraml et al
(2) and Girard et al (3) cloned the aforementioned gene from
endothelial cells in non-small cell lung cancer and high
endothelial venules in human tonsil lymphatic tissues, and termed
it MAST9 and hevin, respectively. The mRNA of the gene is 3 kb in
length and the theoretical molecular mass of the encoded protein,
SPARCL1, is ~75 kDa. However, the protein expressed in vitro
reveals molecular masses of ~75 and 150 kDa, suggesting that
SPARCL1 protein may form a homodimer in vitro (4).
SPARC is a tumor suppressor gene in cancer, and it
has been demonstrated to be involved in the regulation of tumor
progression and drug resistance (5,6). SPARCL1
exhibits 62% identity with SPARC and the two proteins share three
conservative structural domains (3),
indicating functional similarity. SPARCL1 is localized on human
chromosome 4, which contains a number of additional known tumor
suppressor genes. Thus, SPARCL1 is considered to be a potential
tumor suppressor gene and participates in tumor occurrence and
development, by regulating tumor cell viability and differentiation
(4). SPARCL1 may, additionally, be a
potential oncogene and participates in tumor occurrence and
development, by regulating tumor cell viability and affecting the
production of tumor blood vessels (7).
In the present review, the mRNA expression of
SPARCL1 in tumors was analyzed using Oncomine (www.oncomine.org/resource/login.html)
(8). As presented in Fig. 1, of the ~20 different types of solid
tumors included in the Oncomine database, SPARCL1 was downregulated
>2-fold in the majority of tumors analyzed, with the exception
of liver cancer, lymphoma and sarcoma, where SPARCL1 was
upregulated. The downregulation of SPARCL1 was marked in bladder,
breast, cervical, rectal, lung and ovarian cancer. Thus, it may be
inferred that SPARCL1 is a tumor suppressor gene in cancer.
DNA methylation may be an important
mechanism that contributes to the downregulation of SPARCL1
As presented in Table
I, SPARCL1 is upregulated in liver cancer (7); however, SPARCL1 is markedly
downregulated in prostate (9), lung
(10), ovarian (11) and a number of other types of cancer.
These results are consistent with Fig.
1. Downregulation of SPARCL1 in tumors may result from the
epigenetic mechanisms, including DNA methylation, because SPARCL1
is not a classical tumor suppressor gene exhibiting a deletion or
mutation. Isler et al (10)
used microsatellite analysis, quantitative polymerase chain
reaction and sequence analysis of all exons, including the
intron-exon junctions and a portion of the putative promoter
region, but did not identify a mutation or deletion that may be
responsible for the downregulation of SPARCL1. This was indicative
of other regulatory mechanisms resulting in the differential
expression of SPARCL1 in tumors, including epigenetic modification.
A previous study revealed that DNA methylation is the reason for
the downregulation of SPARCL1 in pancreatic, ovarian and lung
cancers (Table I), and demethylation
of the gene partially reversed the abnormal expression in
pancreatic cancer (12).
 | Table I.Summary on the SPARCL1 correlated with
tumor progression and development. |
Table I.
Summary on the SPARCL1 correlated with
tumor progression and development.
| Tumor type | SPARCL1
expression | Biological
function | Molecular
mechanism | Mechanism of gene
expression |
|---|
| Prostate cancer | Downregulation
(9) | Inhibition of cell
migration and invasion (14) | Affects migration by
regulating RhoC (19); inhibition of
the assembly of focal adhesions (20) | – |
| Colorectal
cancer | Downregulation
(13,21) | Inhibition of cell
proliferation, growth and invasion (13) | Affects tumor cell
differentiation through EMT (13) | – |
| Hilar
cholangiocarcinoma | Downregulation
(15) | Inhibition of cell
migration (15) | Inhibition of the
expression of MMP-9, MMP-2, vimentin and fibronectin (15) | – |
| Gastric cancer | Downregulation
(22,23) | – | Inactivation of its
tumor suppressor functions (22) | Loss of
heterozygosity (23) |
| Pancreatic
cancer | Downregulation
(12) | Inhibition of tumor
cell proliferation and invasion (12) | Anti-invasive
effects (12) | DNA methylation
(12) |
| Ovarian cancer | Downregulation
(11) | Associated with
multiple drug resistance (18) | Interaction with
drug resistance-related proteins including PTEN (18) | DNA methylation
(24) |
| Lung cancer | Downregulation
(10) | Decrease in
luciferase activity (10) | – | DNA methylation
(25) |
| Glioma | Downregulation
(16) | Associated with
cell cycle (16) | Anti-adhesion
(16) | – |
| Liver cancer | Upregulation
(7) | – | – | – |
| Uterine
leiomyoma | Upregulation
(26) | – | – | – |
SPARCL1 contributes to tumor development and
progression
There have been a limited number of studies on
SPARCL1, but the gene has been identified to be markedly associated
with tumor development and progression. SPARCL1 contributions to
tumor cell viability (13), migration
and invasion (12–16) and exhibits an anti-adhesive effect
(12,16). In addition, SPARCL1 may be involved in
the regulation of drug resistance in cancer. It has been identified
that SPARCL1 is a recombinant gene in the extracellular matrix of
osteosarcoma in children and is involved in the mechanism of
multiple drug resistance (17). A
previous study used comprehensive bioinformatics analysis to
identify that the SPARCL1 gene was involved in the regulation of
drug resistance in ovarian cancer (18).
The association between SPARCL1 and tumor
progression was investigated using Coremine Medical (http://www.coremine.com/medical). As presented in
Fig. 2, using SPARCL1 and cancer as
key words, SPARCL1 was identified to be associated with diagnosis,
prognosis, recurrence, invasiveness, metastasis and drug resistance
of cancer (Fig. 2A). In addition, the
associations identified in the present review, between SPARCL1 and
invasiveness, metastasis and drug resistance of cancer, were
consistent with previous studies (Table
I)(9–16,18–26).
Furthermore, analysis indicated that SPARCL1 may participate in
cancer development and progression, in 9 biological processes
(P<0.001) including cell viability, cell cycle, migration and
adhesion (Fig. 2B), which is
consistent with previous studies (Table
I). In addition, SPARCL1 and cancer were annotated with DNA
methylation, supporting the hypothesis that DNA methylation may be
an important mechanism which contributes to the downregulation of
SPARCL1.
Downregulation of SPARCL1 is associated with
poor prognosis in cancer
Previous studies indicate that downregulation of
SPARCL1 is markedly associated with poor prognosis and therefore
the gene may be a prognostic marker in cancers. In prostate cancer,
the downregulation of SPARCL1 has been markedly associated with
biochemical recurrence, metastatic disease and poor overall
survival (OS) time (19). Patients
with stage II/III colorectal cancer who possessed increased p53 and
decreased SPARCL1 expression levels exhibited ~50% decreased 3-year
survival compared with controls (27). Furthermore, in gastric cancer,
silenced expression of SPARCL1 predicted a poorer prognosis
(23).
On the basis of The Cancer Genome Atlas (TCGA)
(24) cohort data, the associations
between SPARCL1 and cancer prognosis were analyzed. The expression
value of SPARCL1 and the corresponding clinical data of each type
of cancer in the TCGA cohort was retrieved from the cBioPortal
database (cbioportal.org) (28). Expression values of SPARCL1 were
divided into high and low expression using the median as the
threshold value in a Kaplan-Meier estimator analysis, in accordance
with a previous study (29). As
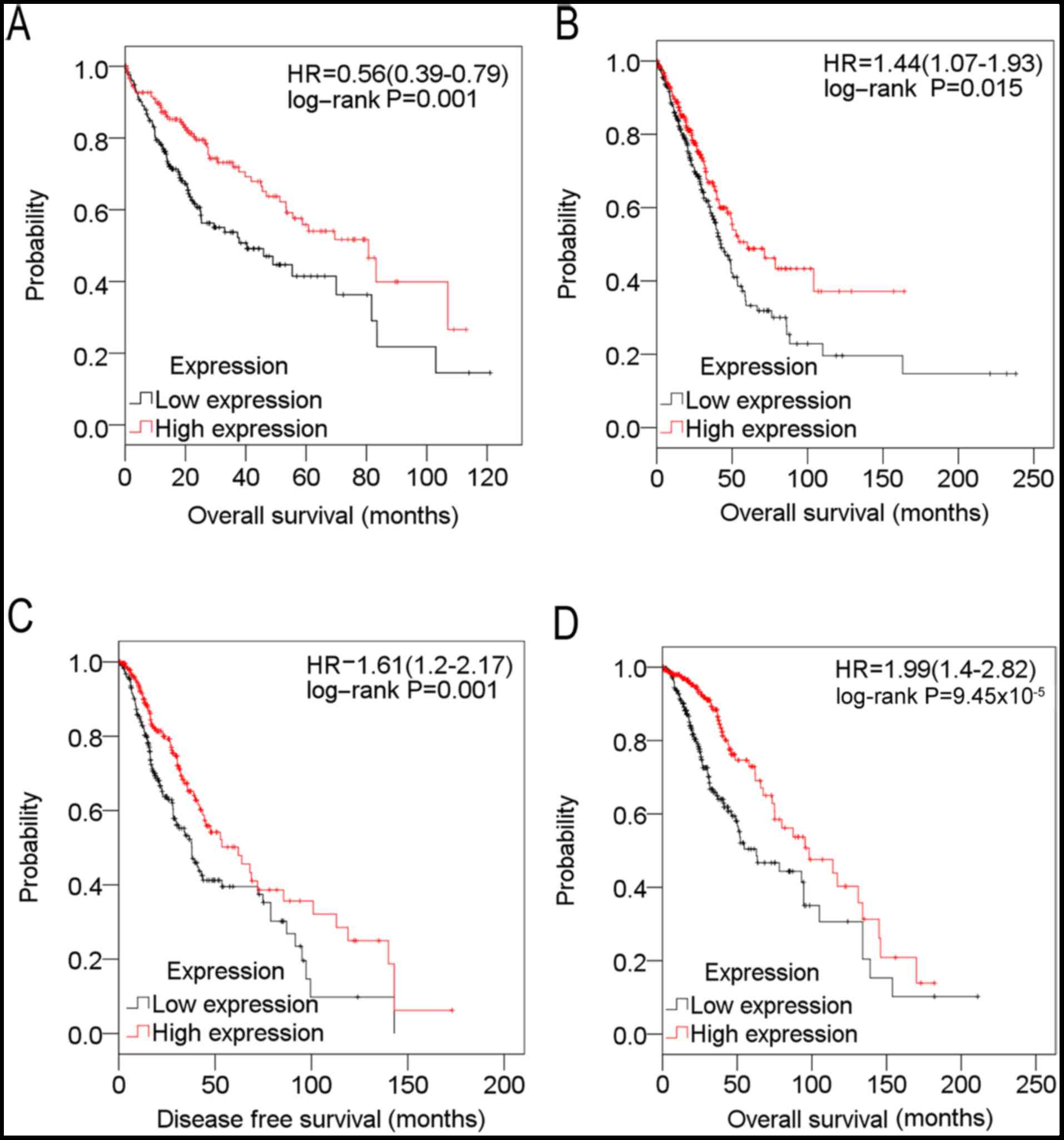
presented in Table II and Fig. 3, downregulation of SPARCL1 was
markedly associated with poor OS time in liver cancer (242 samples)
and lung cancer (324 samples), and markedly associated with poorer
disease-free survival and OS time in glioma (311 samples).
 | Table II.Association between SPARCL1
expression and prognosis in glioma, lung cancer and liver
cancer. |
Table II.
Association between SPARCL1
expression and prognosis in glioma, lung cancer and liver
cancer.
|
|
| DFS (median) | OS (median) |
|---|
|
|
|
|
|
|---|
|
| 95% confidence
interval |
|
| 95% confidence
interval |
|---|
|
|
|
|
|
|
|---|
| Disease type | Group | Estimate | Standard error | Lower boundary | Upper boundary | Estimate | Standard error | Lower boundary | Upper boundary |
|---|
| Glioma | L | 37.900 |
3.983 | 30.093 | 45.707 | 62.900 | 12.971 | 37.476 |
88.324 |
|
| H | 62.000 | 10.173 | 42.061 | 81.939 | 98.200 | 19.627 | 59.731 | 136.669 |
|
| Total | 43.500 |
4.524 | 34.633 | 52.367 | 87.400 | 10.232 | 67.346 | 107.454 |
| Lung cancer | L |
|
|
|
| 42.500 |
3.920 | 34.818 |
50.182 |
|
| H |
|
|
|
| 60.100 | 12.425 | 35.747 |
84.453 |
|
| Total |
|
|
|
| 49.200 |
3.705 | 41.938 |
56.462 |
| Liver cancer | L |
|
|
|
| 40.400 |
9.142 | 22.483 |
58.317 |
|
| H |
|
|
|
| 80.700 | 10.853 | 59.428 | 101.972 |
|
| Total |
|
|
|
| 55.600 |
8.087 | 39.749 |
71.451 |
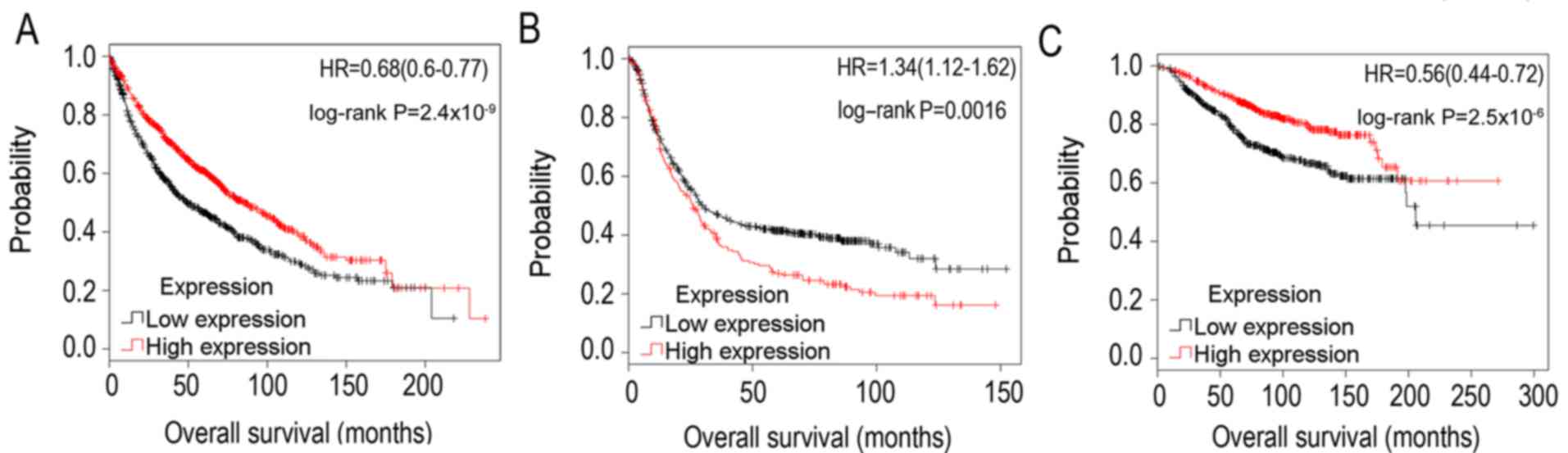
The association between SPARCL1 and OS time in lung
cancer was additionally validated using Kaplan-Meier estimator
analysis, which selected thousands of samples of ovarian, lung,
breast and gastric cancer from microarrays deposited in the TCGA
cohort and Gene Expression Omnibus profiles (30). Using the median expression as the
threshold value, it was identified that the downregulation of
SPARCL1 in lung cancer (3,021 samples) predicted decreased OS time
(Fig. 4A), which was consistent with
the results based on the TCGA cohort data (Table II and Fig.
3). Furthermore, downregulation of SPARCL1 predicted improved
OS time in gastric cancer (1,223 samples) (Fig. 4B), although this result was in
contrast with a previous study (23).
In addition, in breast cancer (2,627 samples), downregulation of
SPARCL1 predicted poorer OS time (Fig.
4C).
SPARCL1 was additionally associated with clinical
features of a number of types of tumor. Downregulation of SPARCL1
was associated with increased mortality of patients with glioma,
liver and lung cancer (P<0.05), and patients with cervical
cancer (P=0.076; Table III).
Furthermore, the downregulation rate of SPARCL1 increased
considerably for surviving patients with cervical cancer and
downregulation of the gene in ovarian cancer was markedly
associated with a lower histological grade (P<0.05; Table IV).
 | Table III.Association between SPARCL1
expression and vital status of patients with different tumors, in
accordance with The Cancer Genome Atlas cohort. |
Table III.
Association between SPARCL1
expression and vital status of patients with different tumors, in
accordance with The Cancer Genome Atlas cohort.
|
|
| SPARCL1
expression |
|
|---|
|
|
|
|
|
|---|
| Patient's vital
status | No. of
patients | Low (%) | High (%) | P-value |
|---|
| Glioma of lower
grade | 528 |
|
| 0.001 |
|
Deceased | 134 (25.4%) | 84
(62.7) | 50
(37.3) |
|
|
Alive | 394 (74.6%) | 180 (45.7) | 214 (54.3) |
|
| Lung
adenocarcinoma | 516 |
|
| 0.001 |
|
Deceased | 187 (36.2%) | 111 (59.4) | 76
(40.6) |
|
|
Alive | 329 (63.8%) | 146 (44.4) | 183 (55.6) |
|
| Hepatocellular
carcinoma | 372 |
|
| 0.024 |
|
Deceased | 130 (34.9%) | 75
(57.7) | 55
(42.3) |
|
|
Alive | 242 (65.1%) | 110 (45.5) | 132 (54.5) |
|
| Cervical
adenocarcinoma | 305 |
|
| 0.076 |
|
Deceased | 73 (23.9%) | 43
(58.9) | 30
(41.1) |
|
|
Alive | 232 (76.1%) | 109 (47.0) | 123 (53.0) |
|
| Subcutaneous
melanoma | 470 |
|
| 0.42 |
|
Deceased | 222 (47.2%) | 100 (45.0) | 122 (55.0) |
|
|
Alive | 248 (52.8%) | 135 (54.4) | 113 (45.6) |
|
| Acute myeloid
leukemia | 173 |
|
| 0.916 |
|
Deceased | 114 (65.9%) | 57
(50.0) | 57
(50.0) |
|
|
Alive | 59 (34.1%) | 29
(49.2) | 30
(50.8) |
|
| Lymphoma | 27 |
|
| 1 |
|
Deceased | 6 (22.2%) |
3 (50.0) |
3 (50.0) |
|
|
Alive | 21 (77.8%) | 11
(52.4) | 10
(47.6) |
|
| Prostate
adenocarcinoma | 497 |
|
| 0.339 |
|
Deceased | 10 (2.0%) |
7 (70.0) |
3 (30.0) |
|
|
Alive | 487 (98.0%) | 242 (49.7) | 245 (50.3) |
|
| Sarcoma | 261 |
|
| 0.666 |
|
Deceased | 99 (37.9%) | 51
(51.5) | 48
(48.5) |
|
|
Alive | 162 (62.1%) | 79
(48.8) | 83
(51.2) |
|
| Esophageal
carcinoma | 184 |
|
| 0.981 |
|
Deceased | 77 (41.8%) | 38
(49.4) | 39
(50.6) |
|
|
Alive | 107 (58.2%) | 53
(49.5) | 54
(50.5) |
|
 | Table IV.Association of SPARCL1 expression
with neoplasm status and neoplasm histological grade in several
cancers, in accordance with The Cancer Genome Atlas cohort. |
Table IV.
Association of SPARCL1 expression
with neoplasm status and neoplasm histological grade in several
cancers, in accordance with The Cancer Genome Atlas cohort.
|
|
| SPARCL1
expression |
|
|---|
|
|
|
|
|
|---|
| Variables | No. of
patients | Low (%) | High (%) | P-value |
|---|
| Glioma of lower
grade | 442 |
|
| 0.036 |
| With
tumor | 220 (49.8%) | 100 (45.5) | 120 (54.5) |
|
|
Tumor-free | 222 (50.2%) | 123 (55.4) | 99
(44.6) |
|
| Cervical
adenocarcinoma | 263 |
|
| 0.013 |
| With
tumor | 76 (28.9%) | 47
(61.8) | 29
(38.2) |
|
|
Tumor-free | 187 (71.1%) | 84
(44.9) | 103 (50.2) |
|
| Ovarian serous
cystadenocarcinoma | 476 |
|
| 0.004 |
|
Histological grade 2 | 56 (11.8%) | 18
(32.1) | 38
(67.9) |
|
|
Histological grade 3 | 420 (88.2%) | 220 (52.4) | 200 (47.6) |
|
Conclusions
Previous studies on the association between SPARCL1
and tumor progression are relatively limited. One previous study
suggested that SPARCL1 is an oncogene (7), but a number of contradictory studies
have identified SPARCL1 as a potential tumor suppressor gene
(4,12,13,15). A
bioinformatic analysis, on the basis of the data retrieved from
Oncomine and the TCGA cohort, was conducted to identify the
associations between SPARCL1 and tumor progression. Oncomine
included information concerning SPARCL1 expression in almost 20
solid tumors (Fig. 1) and this
identified that downregulation of SPARCL1 is prevalent in the
majority of tumors, suggesting that SPARCL1 is a tumor suppressor
gene.
The present review revealed that the downregulation
of the SPARCL1 was markedly associated with poor OS time of ≥7
solid tumors, which included prostate (19), colorectal (27), gastric (23), liver, lung, glioma and breast cancer
(Table II; Figs. 3 and 4).
In addition, decreased expression of SPARCL1 typically predicted
increased mortality in glioma, lung, liver and cervical cancer
(Table III). Therefore, SPARCL1 may
be a universal prognostic marker of tumors in the clinic.
Previous studies have indicated that SPARCL1 is a
tumor suppressor gene and is involved in tumor cell viability
(13), migration and invasion
(12–16) and cell adhesion (12,16), and
is associated with drug resistance of tumors (17,18).
Consistent with these studies, bioinformatics analysis in the
present review revealed that SPARCL1 was associated with prognosis,
invasiveness, metastasis, recurrence and drug resistance of cancer
(Fig. 2A). It is hypothesized that
SPARCL1 exhibits these aforementioned actions by interactions with
a number of biological processes/signaling pathways including cell
adhesion, cell viability, cell cycle and cell migration (Fig. 2B). These results indicate that SPARCL1
serves important functions in tumor progression.
The present review has elucidated the association
between SAPRCL1 and cancer. SPARCL1 may be an important tumor
suppressor gene in tumor progression and development, and it may be
a therapeutic target for cancer treatment and a potential biomarker
for prognosis.
Acknowledgements
The present review was supported by the National
Natural Science Foundation of China (grant nos. 81302283, 81560424,
81660606 and 81460397), the China Postdoctoral Science Foundation
(grant nos. 2014M552535XB and 2014M552291) and the Natural Science
Foundation of Guangxi (grant nos. 2014GXNSFCA118010,
2015GXNSFBA139115, 2015GXNSFAA139151 and 2014GXNSFBA118155).
References
|
1
|
Johnston IG, Paladino T, Gurd JW and Brown
IR: Molecular cloning of SC1: A putative brain extracellular matrix
glycoprotein showing partial similarity to osteonectin/BM40/SPARC.
Neuron. 4:165–176. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Schraml P, Shipman R, Stulz P and Ludwig
CU: cDNA subtraction library construction using a magnet-assisted
subtraction technique (MAST). Trends Genet. 9:70–71. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Girard JP and Springer TA: Cloning from
purified high endothelial venule cells of hevin, a close relative
of the antiadhesive extracellular matrix protein SPARC. Immunity.
2:113–123. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Claeskens A, Ongenae N, Neefs JM, Cheyns
P, Kaijen P, Cools M and Kutoh E: Hevin is down-regulated in many
cancers and is a negative regulator of cell growth and
proliferation. Br J Cancer. 82:1123–1130. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Socha MJ, Said N, Dai Y, Kwong J,
Ramalingam P, Trieu V, Desai N, Mok SC and Motamed K: Aberrant
promoter methylation of SPARC in ovarian cancer. Neoplasia.
11:126–135. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Tai IT, Dai M, Owen DA and Chen LB:
Genome-wide expression analysis of therapy-resistant tumors reveals
SPARC as a novel target for cancer therapy. J Clin Invest.
115:1492–1502. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lau CP, Poon RT, Cheung ST, Yu WC and Fan
ST: SPARC and Hevin expression correlate with tumour angiogenesis
in hepatocellular carcinoma. J Pathol. 210:459–468. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Rhodes DR, Yu J, Shanker K, Deshpande N,
Varambally R, Ghosh D, Barrette T and Pandey A: ONCOMINE: A cancer
microarray database and integrated data-mining platform. Neoplasia.
6:1–6. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Schlomm T, Luebke AM, Sültmann H,
Hellwinkel OJ, Sauer U, Poustka A, David KA, Chun FK, Haese A,
Graefen M, et al: Extraction and processing of high quality RNA
from impalpable and macroscopically invisible prostate cancer for
microarray gene expression analysis. Int J Oncol. 27:713–720.
2005.PubMed/NCBI
|
|
10
|
Isler SG, Ludwig CU, Chiquet-Ehrismann R
and Schenk S: Evidence for transcriptional repression of SPARC-like
1, a gene downregulated in human lung tumors. Int J Oncol.
25:1073–1079. 2004.PubMed/NCBI
|
|
11
|
Biade S, Marinucci M, Schick J, Roberts D,
Workman G, Sage EH, O'Dwyer PJ, Livolsi VA and Johnson SW: Gene
expression profiling of human ovarian tumours. Br J Cancer.
95:1092–1100. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Esposito I, Kayed H, Keleg S, Giese T,
Sage EH, Schirmacher P, Friess H and Kleeff J: Tumor-suppressor
function of SPARC-like protein 1/hevin in pancreatic cancer.
Neoplasia. 9:8–17. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hu H, Zhang H, Ge W, Liu X, Loera S, Chu
P, Chen H, Peng J, Zhou L, Yu S, et al: Secreted protein acidic and
rich in cysteines-like 1 suppresses aggressiveness and predicts
better survival in colorectal cancers. Clin Cancer Res.
18:5438–5448. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Xiang Y, Qiu Q, Jiang M, Jin R, Lehmann
BD, Strand DW, Jovanovic B, DeGraff DJ, Zheng Y, Yousif DA, et al:
SPARCL1 suppresses metastasis in prostate cancer. Mol Oncol.
7:1019–1030. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yu Y, Chen Y, Ma J, Yu X, Yu G and Li Z:
SPARCL1 is a novel predictor of tumor recurrence and survival in
hilar cholangiocarcinoma. Tumour Biol. 37:4159–4167. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Turtoi A, Musmeci D, Naccarato AG, Scatena
C, Ortenzi V, Kiss R, Murtas D, Patsos G, Mazzucchelli G, De Pauw
E, et al: Sparc-like protein 1 is a new marker of human glioma
progression. J Proteome Res. 11:5011–5021. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mintz MB, Sowers R, Brown KM, Hilmer SC,
Mazza B, Huvos AG, Meyers PA, Lafleur B, McDonough WS, Henry MM, et
al: An expression signature classifies chemotherapy-resistant
pediatric osteosarcoma. Cancer Res. 65:1748–1754. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yin F, Liu X, Li D, Wang Q, Zhang W and Li
L: Bioinformatic analysis of chemokine (C-C motif) ligand 21 and
SPARC-like protein 1 revealing their associations with drug
resistance in ovarian cancer. Int J Oncol. 42:1305–1316.
2013.PubMed/NCBI
|
|
19
|
Hurley PJ, Marchionni L, Simons BW, Ross
AE, Peskoe SB, Miller RM, Erho N, Vergara IA, Ghadessi M, Huang Z,
et al: Secreted protein, acidic and rich in cysteine-like 1
(SPARCL1) is down regulated in aggressive prostate cancers and is
prognostic for poor clinical outcome. Proc Natl Acad Sci USA.
109:14977–14982. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hurley PJ, Hughes RM, Simons BW, Huang J,
Miller RM, Shinder B, Haffner MC, Esopi D, Kimura Y, Jabbari J, et
al: Androgen-Regulated SPARCL1 in the Tumor Microenvironment
Inhibits Metastatic Progression. Cancer Res. 75:4322–4334. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhang H, Widegren E, Wang DW and Sun XF:
SPARCL1: A potential molecule associated with tumor diagnosis,
progression and prognosis of colorectal cancer. Tumour Biol.
32:1225–1231. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Jakharia A, Borkakoty B and Singh S:
Expression of SPARC like protein 1 (SPARCL1), extracellular
matrix-associated protein is down regulated in gastric
adenocarcinoma. J Gastrointest Oncol. 7:278–283. 2016.PubMed/NCBI
|
|
23
|
Li P, Qian J, Yu G, Chen Y, Liu K, Li J
and Wang J: Down-regulated SPARCL1 is associated with clinical
significance in human gastric cancer. J Surg Oncol. 105:31–37.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Cancer Genome Atlas Research Network:
Integrated genomic analyses of ovarian carcinoma. Nature.
474:609–615. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sato T, Arai E, Kohno T, Takahashi Y,
Miyata S, Tsuta K, Watanabe S, Soejima K, Betsuyaku T and Kanai Y:
Epigenetic clustering of lung adenocarcinomas based on DNA
methylation profiles in adjacent lung tissue: Its correlation with
smoking history and chronic obstructive pulmonary disease. Int J
Cancer. 135:319–334. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Mencalha AL, Levinsphul A, Deterling LC,
Pizzatti L and Abdelhay E: SPARC-like1 mRNA is overexpressed in
human uterine leiomyoma. Mol Med Rep. 1:571–574. 2008.PubMed/NCBI
|
|
27
|
Yu SJ, Yu JK, Ge WT, Hu HG, Yuan Y and
Zheng S: SPARCL1, Shp2, MSH2, E-cadherin, p53, ADCY-2 and MAPK are
prognosis-related in colorectal cancer. World J Gastroenterol.
17:2028–2036. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gao J, Aksoy BA, Dogrusoz U, Dresdner G,
Gross B, Sumer SO, Sun Y, Jacobsen A, Sinha R, Larsson E, et al:
Integrative Analysis of Complex Cancer Genomics and Clinical
Profiles Using the cBioPortal. Sci Signal. 6:pl12013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Hedditch EL, Gao B, Russell AJ, Lu Y,
Emmanuel C, Beesley J, Johnatty SE, Chen X, Harnett P, George J, et
al: ABCA transporter gene expression and poor outcome in epithelial
ovarian cancer. J Natl Cancer Inst. 106:pii: dju149. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Gyorffy B, Lánczky A and Szállási Z:
Implementing an online tool for genome-wide validation of
survival-associated biomarkers in ovarian-cancer using microarray
data from 1287 patients. Endocr Relat Cancer. 19:197–208. 2012.
View Article : Google Scholar : PubMed/NCBI
|