Introduction
Hepatocellular carcinoma (PHC) is the fifth most
common cancer and the third leading cause of cancer-associated
mortality globally (1). In China, PHC
is a leading cause of cancer-associated mortality (2). PHC is usually diagnosed at an advanced
stage, and early diagnosis is of particular importance. Golgi
glycoprotein 73 (GP73) is a type II Golgi transmembrane protein
(3) and a potential novel marker for
the diagnosis of PHC. A previous study has confirmed GP73 as a
serum marker of liver cancer, and it is possible to use its gene
expression to monitoring early recurrence (2). It is possible to monitor its protein
levels by immunohistochemistry (4).
The expression of GP73 mRNA levels and clinical characteristics are
less well reported. Transcatheter arterial chemoembolization (TACE)
is the preferred treatment for unresectable PHC. A previous study
suggested that tumor necrosis following intervention is more likely
to cause relapse and metastasis, due to the numerous viable tumor
cells remaining following TACE therapy (5). Nevertheless, this evaluation of the
efficacy of TACE was focused on radiological assessment with no
uniform standard determination of residual tumor cells and a lack
of objective laboratory parameters, thereby hindering optimal
opportunity of one more TACE treatment following the first TACE.
This will often induce early and easy recurrence. Serum
α-fetoprotein (AFP) is widely used to detect primary liver tumors,
but this technique demonstrates poor sensitivity and specificity
(6). The present study used reverse
transcription-quantitative polymerase chain reaction (RT-qPCR)
analysis to examine the levels of GP73 mRNA expression in PHC and
its association with clinicopathological characteristics. In
addition, ELISA was used to investigate the importance of GP73 and
the involvement of AFP in assessing the efficacy of TACE in primary
liver cancer.
Materials and methods
Ethical statement
The present study was approved by the Ethics
Committee of the Fourth Hospital of Hebei University (Shijiazhuang,
China). All patients provided written, informed consent.
Clinical data
A total of 40 PHC tissue samples with corresponding
para-cancer tissue samples and 15 normal liver tissue samples were
obtained from 55 patients admitted to the Department of
Hepatobiliary Surgery at the Fourth Hospital of Hebei University
from October 2013 to June 2014. None of the patients were diagnosed
with metastasis prior to surgery. Tissue samples were obtained from
40 patients with pathologically confirmed PHC, and the para-cancer
tissues were obtained within 2.0 cm from the border of the
cancerous tissue. The samples were frozen in liquid nitrogen within
30 min of dissection. The patients with PHC included 29 males and
11 females, aged 39–74 (52±11.3) years. According to the Edmondson
grading system (7), 28 patients
demonstrated low to medium differentiation (level I–III), and the
other 12 patients demonstrated high differentiation (level IV). The
median diameter of the tumors was 6.33 cm (range, 1.9–11.6 cm), and
11 cases demonstrated vascular invasion. All patients had complete
clinicopathological data, and did not receive radiotherapy or
chemotherapy prior to surgery. Histopathological diagnosis revealed
the presence of 31 cases of PHC and 9 cases of bile duct carcinoma.
All patients underwent AFP examination. The 15 normal samples were
obtained from normal liver tissue adjacent to benign liver tumors,
including 12 males and 3 females with an average age of 45.3 years
(range, 32–66 years).
Clinical TACE data comprised of 68 patients with PHC
at the Fourth Hospital of Hebei Medical University, all of whom
were hospitalized for the first time between May 2014 and January
2015), including 45 males and 23 females. Patients were 39–78 years
old, with an average age of 53.2 years. According to the Barcelona
clinic liver cancer (BCLC) staging system (8), 29 cases were stage A, 26 stage B, and 13
stage C. The patients were diagnosed with PHC with a history of
chronic liver disease, following imaging and examination of the
tumor marker AFP. All patients met the criteria of level A or B in
the Child-Pugh staging system (9),
life quality Kamofsky score (10)
>70, and no absolute contraindications to TACE. The patients
demonstrated no significant difference in transaminases, alkaline
phosphatase, γ-glutamine transpeptidase (γ-GT), albumin (PA), blood
routine or kidney function compared with the control group, and had
not received any prior treatment (chemotherapy, radiotherapy or
immunotherapy). The peripheral blood of patients with liver cancer
was isolated 2 days prior to surgery and 7 and 30 days following
surgery, and was stored at −80°C. The data of 29 healthy people
that came to the Fourth Hospital of Hebei Medical University from
September 2014 to January 2015 for health examinations were used as
controls, including 17 males and 12 females with an average age of
41.6 years (range, 23–64 years).
RT-qPCR was used to determine mRNA
expression
As per the manufacturer's protocol, the tissue
samples stored at −80°C were cut into sections (0.3–0.5 cm), placed
in 1.5 m centrifuge tubes, and 1 ml RNA iso Plus reagent (Takara
Bio, Inc., Otsu, Japan) was added, followed by centrifugation at
4°C at 19,185 × g for 15 min. Absorbance (A) at 260 nm and 280 nm
(A260 and A280) was measured to calculate the RNA concentration.
First strand cDNA was synthesized using a reverse transcription kit
(PrimeScript™ RT reagent kit with gDNA Eraser; Takara Bio, Inc.) in
a 36-µl reaction volume, according to the manufacturer's protocol.
Primer sequences used were as follows: GP73 forward,
5′-GTGTGAGGAGCGAATAGAAGAGG-3′ and reverse,
5′-GTCTCTGGTCGTTGTTTTCACT-3′; GAPDH forward,
5′-CTGGGCTACACTGAGCACC-3′ and reverse, 5′-AAGTGGTCGTTGAGGGCAATG-3′.
All primers were synthesized by Sangon Biotech Co., Ltd. (Shanghai,
China). PCR amplification was performed in a 50 µl reaction volume,
using SYBR® Premix EX Taq™ (Takara Bio, Inc.).
Thermocycling conditions were as follows: 95°C for 3 min, and 45
cycles of melting at 95°C for 30 sec, followed by 57°C for 30 sec.
Gel electrophoresis images were acquired and stored (UVP, LLC,
Upland, CA, USA), and A values of the GP73 and GAPDH bands were
calculated using the 2−ΔΔCq method (11). Relative expression of GP73 was
represented as the ratio of AGP73 to
AGAPDH.
TACE
Using a modified Seldinger technique (12), the right femoral artery was punctured
and the guide wire was inserted, followed by the 5F sheath, and
then a 5F-RH catheter was inserted for celiac trunk angiography and
indirect portal venography. Superior mesenteric artery, left
gastric artery and right phrenic artery angiography were also
performed when necessary. Then, the catheter was inserted into the
target vessel (certain patients required the use of Progreat
microcatheters, purchased from Terumo Corporation, Tokyo, Japan),
and 1,000 mg tegafur (Hao Chuang Co., Ltd., Hainan, China), 150 mg
oxaliplatin (Jiangsu Hengrui Co., Ltd., Lianyungang, China) and 200
mg leucovorin (Hainan SIDA Pharmaceutical Co., Ltd., Hainan, China)
were slowly perfused, followed by 8–25 ml ultra-fluid lipiodol
(Jiangsu Hengrui Co., Ltd.) containing 15 mg hydroxycamptothecin
for embolization of the artery supporting the tumor. Choice and
volume of the embolization agent were based on the cross-sectional
area of the embolization and size of the tumor, as well as
preoperative liver function status of the patients.
Assessment of serum GP73 levels in the
blood samples by ELISA and AFP levels by chemiluminescence
immunoassay
Serum GP73 levels were assessed using the Human
Golgi glycoprotein 73 (GP73) ELISA kit (Beijing Hotgen Bioech Co.,
Ltd., Beijing, China) according to the manufacturer's protocol. For
each test, samples (20 µl) were added to a 96-well plate. The
experiment was performed in triplicate and independently repeated
three times. Optical density values at 450 nm and 630 were read
using a multiskan MK3 microplate reader (Thermo Fisher Scientific,
Inc., Waltham, MA, USA). AFP levels were determined using the Cobas
601 enhanced chemiluminescence immunoassay analyzer and supporting
kit [Human αFP (alpha-fetoprotein) kit; YZB/GEM 1177–2011; Roche
Diagnostic GmbH], according to the manufacturer's protocol.
Statistical analysis
The data were analyzed using SPSS 19.0 statistical
software (IBM Corp., Armonk, NY, USA). Quantitative data were
expressed as the mean ± standard deviation. The Mann-Whitney U-test
was used for comparisons between the groups. Spearman correlation
coefficients were used to analyze the associations between GP73
expression levels and clinicopathological data. P<0.05 was
considered to indicate a statistically significant difference.
Results
GP73 mRNA expression levels are higher
in cancer tissues than para-cancer or normal tissues
GP73 mRNA expression was detected in all 40 samples
of primary liver cancer and corresponding para-cancer liver
tissues, as well as in normal liver tissue, but the levels differed
between the PHC tissues, corresponding para-cancer liver tissues
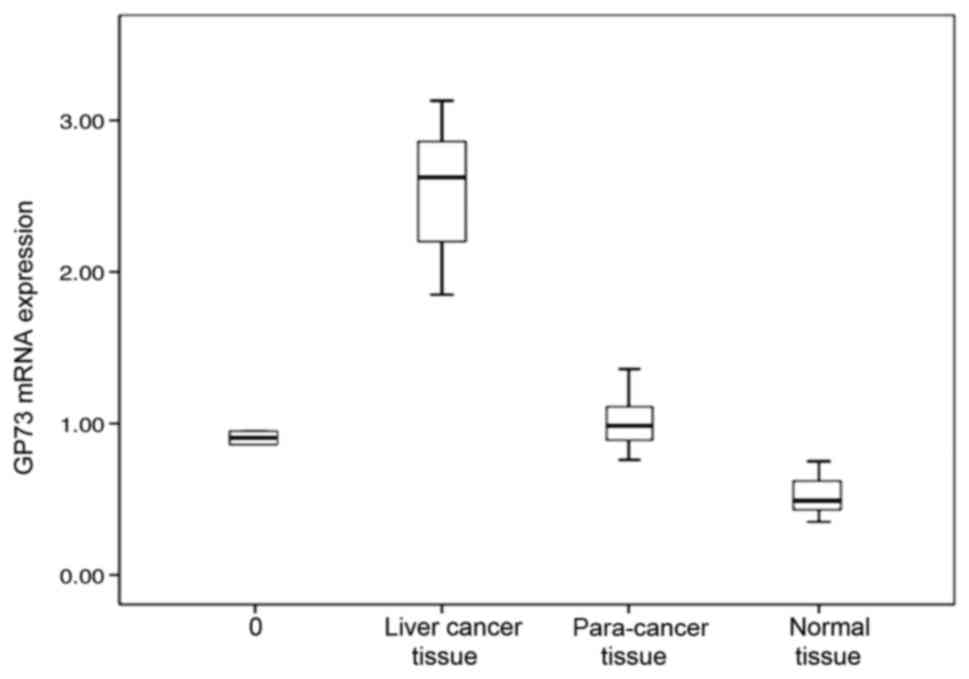
and normal liver tissue. The relative levels of GP73 mRNA were
2.35+0.17 in primary liver cancer, which was significantly higher
than that in para-cancer liver tissue (0.93+0.05) and normal liver
tissue (0.53+0.04; Table I; Figs. 1–4).
 | Table I.Relative expression levels of GP73
mRNA. |
Table I.
Relative expression levels of GP73
mRNA.
| Group | Number of
patients | GP73 mRNA expression
levels |
|---|
| Liver cancer
tissue | 40 | 2.35±0.17 |
| Para-cancer
tissue | 40 |
0.93±0.15a |
| Normal liver
tissue | 15 |
0.53±0.24a |
GP73 mRNA expression levels are
associated with tumor size, vascular invasion and tumor
differentiation
GP73 expression in liver cancer tissue demonstrated
no significant associations with age, sex, number of tumors, serum
AFP or history of hepatitis (P>0.05; Table II), but demonstrated significant
associations with tumor size, vascular invasion and tumor
differentiation (P<0.05; Table
II).
 | Table II.GP73 mRNA expression levels in liver
cancer, and its associations with clinicopathological
characteristics. |
Table II.
GP73 mRNA expression levels in liver
cancer, and its associations with clinicopathological
characteristics.
| Clinicopathological
characteristic | n | GP73 mRNA expression
levels (RU) | P-value |
|---|
| Age |
|
| 0.31 |
| ≤65 | 34 | 2.15±0.13 |
|
|
>65 | 6 | 2.21±0.18 |
|
| Sex |
|
| 0.42 |
| Male | 29 | 2.32±0.08 |
|
|
Female | 11 | 2.33±0.16 |
|
| Tumor size, cm |
|
| 0.01 |
| ≤3 | 21 | 2.17±0.13 |
|
|
>3 | 19 | 2.42±0.09 |
|
| Tumor number |
|
| 0.59 |
| 1 | 31 | 2.36±0.11 |
|
| ≥2 | 9 | 2.39±0.09 |
|
| Differentiation |
|
| 0.02 |
| Well
differentiated | 12 | 2.15±0.13 |
|
|
Moderately and poorly
differentiated | 28 | 2.37±0.08 |
|
| Angioinvasion |
|
| 0.01 |
| Yes | 11 | 2.41±0.15 |
|
| No | 29 | 2.17±0.11 |
|
| AFP, ng/ml |
|
| 0.85 |
| ≤400 | 10 | 2.34±0.11 |
|
|
>400 | 30 | 2.31±0.12 |
|
| HBV |
|
| 0.56 |
| + | 33 | 2.35±0.13 |
|
| − | 7 | 2.33±0.08 |
|
Serum GP73 levels differ between
patients with cancer and controls, differ between BCLC stage, and
are associated with clinical stage
Prior to TACE, serum GP73 levels of the 68 patients
with primary liver cancer [152.5 µg/l (76.4–284.5 µg/l)] were
significantly higher compared with the normal control [49.3 µg/l
(12.6–26.7 µg/l); P<0.01]. Significant differences in GP73
levels were also detected between BCLC stage A, B and C liver
cancers [92.12 µg/l (38.9–135.2 µg/l), 122.9 µg/l (55.2–178.5
µg/l), 162.55 µg/l (110.8–232.9 µg/l), respectively; P<0.05].
These results indicated that serum GP73 levels were correlated with
clinical stage (P<0.05; r=0.27), but were not significantly
affected by the sex or age of the patient.
Serum GP73 levels decreased following
TACE
Serum GP73 levels of the 68 patients with primary
liver cancer significantly decreased to 99.2 µg/l (66.7–150.8 µg/l;
P<0.05) 7 days following TACE; and serum GP73 levels of the 28
patients with BCLC Stage A liver cancer decreased to 76.5 µg/l
(59.2–107.2 µg/l; P<0.01) 7 days following TACE.
AFP-negative patients have a GP73 high
detection rate
Among the 65 patients with primary liver cancer, 43
had AFP levels >400 µg/ml, and the other 22 had AFP levels
<400 µg/ml. Among those with AFP levels <400 µg/l, 49 (75.3%)
patients had a GP73 level ≥132 µg/l, suggesting a high detection
rate of GP73 for AFP-negative patients, which may help to improve
the accuracy of liver cancer diagnosis for patients with negative
AFP.
Patients with a good response to TACE
had lower serum GP73 levels than those with a poor response
Serum GP73 levels were rechecked 30 days following
TACE, and upper abdominal computerized tomography or digital
subtraction hepatic arteriography was performed. The results
indicated that 31 patients had poor lipiodol retention, the lesion
was active with intrahepatic metastasis, and serum GP73 levels were
an average of 183.2 µg/l (79.5–235.2 µg/l). The other 34 cases
achieved a good response to TACE (good lipodol retention and no
indication of intrahepatic metastasis), and their GP73 levels were
115.2 µg/l (63.4–148.2 µg/l), which was significantly lower than
those with a poor response (P<0.05).
Discussion
Studies on cancer recurrence and
metastasis-associated factors are of significance for the
prediction of PHC prognosis and anti-metastatic therapy (13). Early diagnosis and treatment of PHC
are vital for improving the overall survival rate of patients
(14). AFP is an important indicator
for early diagnosis of liver cancer and is used to monitor its
progression, and thus is widely used in clinical practice. However,
30–40% of liver cancers, in particular those originating from the
bile duct, are negative for AFP expression. Furthermore, elevated
AFP levels may be detected in patients with cirrhosis or
exacerbations of chronic hepatitis (15). Prospective studies analyzing the
performance characteristics of AFP for PHC surveillance reported
sensitivities of 39–64%, specificities of 76–91% and positive
predictive values of 9–32% (16,17). Thus,
the clinical value of AFP has been questioned due to its low
sensitivity and specificity (16–18), and
novel serum markers for liver cancer with higher sensitivity and
specificity are being actively sought in current research. GP73 is
a novel tumor marker and was originally described as a resident
Golgi type II transmembrane protein, with a single, N-terminal
transmembrane domain and an extensive, C-terminal coiled-coil
domain located on the luminal surface of the Golgi apparatus
(19). Its mRNA was first identified
in a patient with syncytial giant cell hepatitis (20). Serum GP73 levels gradually increase
with the progression of hepatitis, cirrhosis, and liver cancer
(2), suggesting that it is an
effective indicator for monitoring liver cancer progression.
Using immunohistochemistry, GP73 was previously
demonstrated to be highly expressed in primary liver cancer tissue
and correlated with tumor differentiation (21). However, the semi-quantitative nature
of immunohistochemistry makes it relatively difficult to provide an
accurate diagnosis, and it demonstrates considerable false positive
and false negative results. In the present study, RT-qPCR was used
to determine GP73 mRNA levels in liver cancer tissues, para-cancer
tissues and normal liver tissues, as this method is intuitive and
reliable. Statistical analysis indicated that GP73 expression was
the highest in primary liver cancer, moderate in para-cancer
tissue, and the lowest in normal liver tissue (P<0.05),
suggesting that high GP73 levels may be involved in the progression
of primary liver cancer, and may be used as a marker for the
diagnosis of liver cancer and for monitoring its recurrence. The
associations between GP73 mRNA expression levels and
clinicopathological features of patients with primary liver cancer
were investigated, and GP73 expression was revealed to not be
affected by age, sex, number of tumors, serum AFP levels or history
of hepatitis, but it was associated with tumor size, vascular
invasion and tumor differentiation, inferred as increased primary
liver cancer burden and risk of metastasis and recurrence (22,23). The
association of GP73 expression with these indicators suggested that
GP73 may be involved in the progression and metastasis of primary
liver cancer, and may be used as a novel serum marker for
monitoring primary PHC recurrence following surgery, which may help
clinicians to determine the timing of preventive treatment and to
improve prognosis and patient survival.
There was a high detection rate in patients with
liver cancer who were negative for AFP expression. The combined use
of GP73 and AFP may significantly improve the accuracy of early
diagnosis of liver cancer. In addition, patients with BCLC stage C
had higher serum GP73 levels than patients with BCLC stage B and A,
suggesting the involvement of GP73 in progression and metastasis of
liver cancer. In addition, dynamic changes of serum GP73 levels
also reflected the efficacy of TACE treatment. Serum GP73 levels
significantly decreased 7 days following TACE treatment, suggesting
that TACE significantly reduced liver tumor burden and delayed
tumor progression. Serum GP73 levels in patients with tumor
progression was significantly higher than those in remission 30
days following TACE treatment. Acute liver damage and repeated
liver tissue regeneration may lead to increased GP73 expression
(22), which is then released into
the blood and reflects tumor progression. The combined use of the
two markers may become an important means for the diagnosis of
primary liver cancer. Hu et al (24) studied receiver operating
characteristic (ROC) curves comparing Chinese patients with PHC
admitted to Xiangya Second Hospital from June 2008 to December
2008, and demonstrated that the Hepatitis B virus-associated PHC
area under the (AU) ROC curve for GP73 was 0.89 [95% confidence
interval (CI), 0.82–0.97], with a sensitivity of 77.4% and
specificity of 83.9%, whereas the AUROC for AFP was 0.77 (95% CI,
0.65–0.89), with a sensitivity of 48.4% and specificity of 96.8%.
Zhou et al (25) performed a
meta-analysis, which revealed a sensitivity of 76% (GP73, 95% CI,
51–91%) vs. 70% (AFP, 95% CI, 47–86%) and specificity of 86% (GP73,
95%CI, 65–95%) compared with 89% (AFP, 95% CI, 69–96%),
respectively, which indicated that serum GP73 has a comparable
accuracy to AFP for the diagnosis of PHC. The present study
indicated that serum GP73 levels were significantly increased in
patients with poor prognosis 30 days following surgery, which may
be useful for the assessment of prognosis and disease progression.
It is generally recognized that for patients with low AFP
expression, AFP levels rarely reflect the efficacy of TACE, while
the dynamic changes of GP73 levels accurately reflect the condition
of the patient. These results suggested that for patients with AFP
levels ≤400 µg/l, the dynamic changes in serum GP73 are valuable in
the assessment of TACE efficacy, and should be promoted in clinical
examination.
Combination with AFP may significantly improve the
rate of early diagnosis of PHC, and GP73 levels inpatients with
BCLC stage C PHC were significantly higher compared with patients
with stage B PHC, which indicated that serum GP73 levels are
associated with the progression and metastasis of PHC. Meanwhile,
serum GP73 changes are relevant to the effect of TACE. Serum GP73
levels decreased 7 days post-intervention as compared with those
recorded prior to treatment, given that TACE significantly reduces
the liver tumor load and delays the progression of tumors. GP73
serum levels in patients with disease progression, recorded 30 days
post-operation, were significantly higher than those of patients in
remission. With disease progression and interventional therapy,
normal liver cells are damaged, and acute hepatic injury and
repeated liver tissue remodeling may lead to GP73 being released
into the blood, subsequently leading to disease progression
(26). GP73 sensitivity and
specificity were higher compared with that of detection of AFP, and
the two may be used in combination as an improved method of
diagnosis for primary liver cancer.
The present study indicated that serum GP73 levels
significantly increased in patients with a rapid progression of the
disease 30 days following TACE, and may be useful in evaluating
prognosis and disease progression. For AFP negative patients or
those with low concentrations by AFP, the effect of TACE is
difficult to predict. The dynamic changes of serum GP73 level may
accurately reflect the patient's condition, suggesting its clinical
application value in monitoring the effect of TACE treatment in
AFP-negative patients with PHC or with AFP levels <400 g/l, and
this may be widely applied for clinical detection.
In summary, to the best of our knowledge, this is
the first demonstration of GP73 expression at them RNA and serum
levels in terms of treatment with TACE, and its association with
clinicopathological features of PHC was demonstrated. Manipulation
of GP73 expression in patients with PHC patients may lead to the
development of novel therapies. Further studies of GP73 functions
and mechanisms of its regulation in normal and PHC tissues are
warranted.
References
|
1
|
Llovet JM, Burroughs A and Bruix J:
Hepatocellular carcinoma. Lancet. 362:1907–1917. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mao Y, Yang H, Xu H, Lu X, Sang X, Du S,
Zhao H, Chen W, Xu Y, Chi T, et al: Golgi protein 73 (GOLPH2) is a
valuable serum marker for hepatocellular carcinoma. Gut.
59:1687–1693. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kladney RD, Bulla GA, Guo L, Mason AL,
Tollefson AE, Simon DJ, Koutoubi Z and Fimmel CJ: GP73, a novel
Golgi-localized protein uprcgulated by viral infection. Gene.
249:53–65. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhang F, Gu Y, Li X, Wang W, He J and Peng
T: Upregulated Golgi phosphoprotein 2 (GOLPH2) expression in lung
adenocarcinoma tissue. Clin Biochem. 43:983–991. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Nagasue N, Galizia G, Kohno H, Chang YC,
Hayashi T, Yamanoi A, Nakamura T and Yukaya H: Adverse effects of
preoperative hepatic artery chemoembolization for resectable
hepatocellular carcinoma: A retrospective comparision of 138 liver
resection. Surgery. 106:81–86. 1989.PubMed/NCBI
|
|
6
|
Benowitz S: Liver cancer biomarkers
struggling to succeed. J Natl Cancer Inst. 99:590–591. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Edmondson HA and Steiner PE: Primary
carcinoma of the liver: A study of 100 cases among 48,900
necropsies. Cancer. 7:462–503. 1954. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bruix J, Sherman M, Llovet JM, Beaugrand
M, Lencioni R, Burroughs AK, Christensen E, Pagliaro L, Colombo M
and Rodés J: EASL Panel of Experts on HCC: Clinical management of
hepatocellular carcinoma. Conclusions of the Barcelona-2000 EASL
conference. European association for the study of the liver. J
Hepatol. 35:421–430. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Triantos C, Zisimopoulos K, Tsochatzis E,
Vlachogiannakos J, Manolakopoulos S, Rigamonti C, Goulis J, Manesis
E, Anastasiou J, Papalexi F, et al: 207 Meld vs cps for prognosis
in cirrhosis. Results from a multicentre study. J Hepatol.
52:(Suppl 1). S892010. View Article : Google Scholar
|
|
10
|
Bai H: Manual of tumor diagnosis and
treatment for primary physicians. Peking University Medical Press;
Beijing: pp. 3562008
|
|
11
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Seldinger SI: Catheter replacement of the
needle in percutaneous arteriography; a new technique. Acta Radiol.
39:368–376. 1953. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Saffroy R, Pham P, Reffas M, Takka M,
Lemoine A and Debuire B: New perspectives and strategy research
biomarkers for hepatocellular carcinoma. Clin Chem Lab Med.
45:1169–1179. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Liu X, Wan X, Li Z, Lin C, Zhan Y and Lu
X: Golgi protein 73 (GP73), a useful serum marker in liver
diseases. Clin Chem Lab Med. 49:1311–1316. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bruix J and Sherman M: Practice Guidelines
Committee, American Association for the Study of Liver Diseases:
Management of hepatocellular carcinoma. Hepatology. 42:1208–1236.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Oka H, Tamori A, Kuroki T, Kobayashi K and
Yamamoto S: Prospective study of alpha-fetoprotein in cirrhotic
patients monitored for development of hepatocellular carcinoma.
Hepatol. 19:61–66. 1994. View Article : Google Scholar
|
|
17
|
Pateron D, Ganne N, Trinchet JC,
Aurousseau MH, Mal F, Meicler C, Coderc E, Reboullet P and
Beaugrand M: Prospective study of screening for hepatocellular
carcinoma in Caucasian patients with cirrhosis. J Hepatol.
20:65–71. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zoli M, Magalotti D, Bianchi G, Gueli C,
Marchesini G and Pisi E: Efficacy of a surveillance program for
early detection of hepatocellular carcinoma. Cancer. 78:977–985.
1996. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Marrero JA, Romano PR, Nikolaeva O, Steel
L, Mehta A, Fimmel CJ, Comunale MA, D'Amelio A, Lok AS and Block
TM: GP73, a resident Golgi glycoprotein, is a novel serum marker
for hepatocellular carcinoma. J Hepatol. 43:1007–1012. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Norton PA, Comunale MA, Krakover J,
Rodemich L, Pirog N, D'Amelio A, Philip R, Mehta AS and Block TM:
N-linked glycosylation of the liver cancer biomarker GP73. J Cell
Biochem. 104:136–149. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Riener MO, Stenner F, Liewen H, Soll C,
Breitenstein S, Pestalozzi BC, Samaras P, Probst-Hensch N,
Hellerbrand C, Müllhaupt B, et al: Golgi phosphoprotein 2 (GOLPH2)
expression in liver tumors and its value as a serummarker in
hepatocellular carcinomas. Hepatology. 49:1602–1609. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Poon Tung-Ping R, Fan ST and Wong J: Risk
factors, prevention, and management of postoperative recurrence
after resection of hepatocellular carcinoma. Ann Surg. 232:10–24.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Shimada K, Sano T, Sakamoto Y and Kosuge
T: A long-term follow-up and management study of hepatocellular
carcinoma patients surviving for 10 years or longer after curative
hepatectomy. Cancer. 104:1939–1947. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hu JS, Wu DW, Liang S and Miao XY: GP73, a
resident Golgi glycoprotein, is sensibility and specificity for
hepatocellular carcinoma of diagnosis in a hepatitis B-endemic
Asian population. Med Oncol. 27:339–345. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhou Y, Yin X, Ying J and Zhang B: Golgi
protein 73 versus alpha-fetoprotein as a biomarker for
hepatocellular carcinoma: A diagnostic meta-analysis. BMC Cancer.
12:172012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Iftikhar R, Kladney RD, Havlioglu N,
Schmitt-Gräff A, Gusmirovic I, Solomon H, Luxon BA, Bacon BR and
Fimmel CJ: Disease- and cell-specific expression of GP73 in human
liver disease. Am J Gastroenterol. 99:1087–1095. 2004. View Article : Google Scholar : PubMed/NCBI
|