Introduction
Breast cancer is the most prevalent type of cancer
among women worldwide (1). The
majority of primary breast cancer cases are estrogen receptor (ER)
positive; approximately one-third of patients with breast cancer
are ER− at the time of diagnosis (2). ER status is predictive for the response
to endocrine therapy of breast cancer, and is thus an important
marker of prognosis. Therapies targeting the ER, including
tamoxifen and aromatase inhibitors, have been successfully applied
for treating breast cancer patients with tumors that express ER
(3). However, a fraction of breast
cancers that are initially ER+ lose the expression of ER
during tumor progression, and thus the long-term effectiveness of
endocrine therapies is limited (2).
Methylation of ESR1, the gene encoding ER-α, is associated with ER
status and implicated in the tumor resistance to hormone-based
treatment in breast cancer (4).
MicroRNAs (miRNAs) are a group of highly conserved,
non-coding RNAs that suppress gene expression
post-transcriptionally by binding to the 3′-untranslated region
(3′UTR) of target mRNAs. Dysregulation of miRNAs is associated with
the initiation and progression of a number of types of human
cancer, including breast cancer (5,6). miRNAs
are increasingly regarded as essential regulators of gene
expression in breast cancer, which may act as oncogenes (e.g.,
miR-21) or tumor suppressors (e.g., let-7) (7).
Previous studies have demonstrated that the absence
of ER expression may result from the hypermethylation of CpG
islands in the 5′ region of the ESR1 gene in a proportion of breast
cancer cases (2). DNA methylation is
catalyzed by DNA methyltransferases (DNMTs). It has been reported
that the specific inhibition of DNMT1 expression is sufficient to
induce ER-α re-expression in human breast cancer cell lines
(8). DNMT1 expression has been
demonstrated to be inversely correlated with miRNA-148a expression
in breast cancer tissue, which may imply that there is a negative
feedback regulatory loop between miRNA-148a and DNMT1 (9).
In the present study, it was demonstrated that ER
and miRNA-148a expression in HCC1937 and MCF7 cells were
associated, then further confirmed that miRNA-148a increases the
expression of ER-α through targeting DNMT1.
Materials and methods
Cell culture and transfection
The human breast cancer cell lines MCF7 and HCC1937
were purchased from the Cell Resource Center, Shanghai Institute of
Biochemistry and Cell Biology at the Chinese Academy of Sciences
(Shanghai, China). Cells were maintained at 37°C in a humidified
air atmosphere containing 5% CO2 in RPMI-1640 (MCF7) or
Dulbecco's modified Eagle's medium (HCC1937), supplemented with 10%
fetal bovine serum (all from Gibco; Thermo Fisher Scientific, Inc.,
Waltham, MA, USA).
siRNA against DNMT1 (stB0002746C-1-5), miRNA-148a
mimic (miR10000516-1-5) and miRNA-148a inhibitor (miR20000243-1-5)
were obtained from Guangzhou RiboBio Co., Ltd. (Guangzhou, China).
miRNA mimic/inhibitor control (miR01101-1-5) and siRNA control
(siN05815122147-1-5) were also obtained from Guangzhou RiboBio Co.,
Ltd. Lipofectamine 2000 (Invitrogen; Thermo Fisher Scientific,
Inc.) was used for transfection into MCF-7 cells according to the
manufacturer's protocol. The transfected concentrations of siRNA,
miRNA-148a mimic and inhibitor were 50, 50 and 100 nM,
respectively.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNA was extracted from cells using TRIzol
reagent (Sangon Biotech Co., Ltd., Shanghai, China). ReverTra Ace
qPCR RT kit (Toyobo Co., Ltd., Osaka, Japan) was used to generate
cDNA from mRNA and SYBR Premix Ex Taq (Takara Bio, Inc., Otsu,
Japan) was used to perform qPCR with the ABI 7500 Real-time PCR
system (Applied Biosystems; Thermo Fisher Scientific, Inc.)
according to the manufacturer's protocol. A total of 1 µg cDNA was
used for qPCR. The primer sequences were as follows: DNMT1 forward,
5′-GCACAAACTGACCTGCTTCA-3′ and reverse, 5′-GCCTTTTCACCTCCATCAAA-3′;
ER-α forward, 5′-TTCGGCTCCAACGGCCTGGGGGGTTT-3′ and reverse,
5′-GGTACTGGCCAATCTTTCTCTGCCACCCT-3′; GAPDH forward,
5′-GAAGGTGAAGGTCGGAGTC-3′ and reverse, 5′-GAAGATGGTGATGGGATTTC-3′.
The thermocycling conditions for qPCR were as follows: 95°C for 30
sec, 40 cycles of 95°C for 5 sec and 60°C for 30 sec. The relative
expression level was determined using the 2−ΔΔCq method
(10).
In order to amplify miRNA sequences, total RNA was
extracted using TRIzol reagent (Invitrogen; Thermo Fisher
Scientific, Inc.) and the microRNA qRT-PCR syb kit
(TaqMan™ MicroRNA Assays, Thermo Fisher Scientific,
Inc.) was used for generating cDNA from microRNA and its subsequent
qPCR detection, according to the manufacturer's protocols. Primers
for miRNA-148a and U6 were purchased from Invitrogen (Thermo Fisher
Scientific, Inc.). The miRNA-148a primers were
5′-ATGCTCAGTGCACTACAGAA-3′ (forward) and 5′-GTGCAGGGTCCGAGGT-3′
(reverse). The internal standard was U6 and its primer was
5′-CTCGCTTCGGCAGCACA-3′ (forward) and 5′-AACGCTTCACGAATTTGCGT-3′
(reverse).
Western blot analysis
Cultured cells were harvested and lysed in
radioimmunoprecipitation assay buffer supplemented with complete
protease inhibitor cocktail tablets (Roche Diagnostics GmbH,
Mannheim, Germany). Cell debris was removed by centrifugation at
13,000 × g for 20 min at 4°C. The concentration of proteins were
quantified using Bio-Rad Protein Assay (Bio-Rad Laboratories, Inc.,
Hercules, CA, USA). Lysates (~20 µg) were separated by SDS-PAGE
(10%) and transferred to PVDF membranes (Bio-Rad Laboratories,
Inc.). Subsequently, the membranes were blocked [5% fat-free milk
in Tris buffer saline with 0.1% Tween-20 (TBS-T)] for 1 h at room
temperature. Following blocking, membranes were incubated with
primary antibodies, including ER (dilution, 1:1,000; incubated
overnight at 4°C; cat. no. ab108398; Abcam, Cambridge, MA, USA),
DNMT1 (dilution, 1:1,000; incubated overnight at 4°C; catalog no.
ab13537; Abcam) and GAPDH (dilution, 1:5,000; incubated overnight
at 4°C; cat. no. 5174; Santa Cruz Biotechnology, Inc., Dallas, TX,
USA). Then, the membrane was washed for 5 min 3 times with TBST.
Subsequently, goat anti-rabbit immunoglobulin (for ER, cat. no.
ab150077; Alexa Fluor; Abcam) and goat anti-mouse immunoglobulin
(for DNMT1, cat. no. ab150113; Alexa Fluor; Abcam) were added at a
1:5,000 dilution and incubated with the membrane at room
temperature for 30 min.
Prediction of miRNA-148a targets
TargetScan 5.1 online software (http://www.targetscan.org/; Whitehead Institute for
Biomedical Research, Cambridge, MA, USA) was used to predict the
target genes of miRNA-148a.
Statistical analysis
Student's t-test was used to determine statistical
significance between the groups. SPSS version 17 (IBM Corp.,
Armonk, NY, USA) was used. P<0.05 was considered to indicate a
statistically significant difference.
Results
miRNA-148a is expressed more highly in
ER-α positive cells
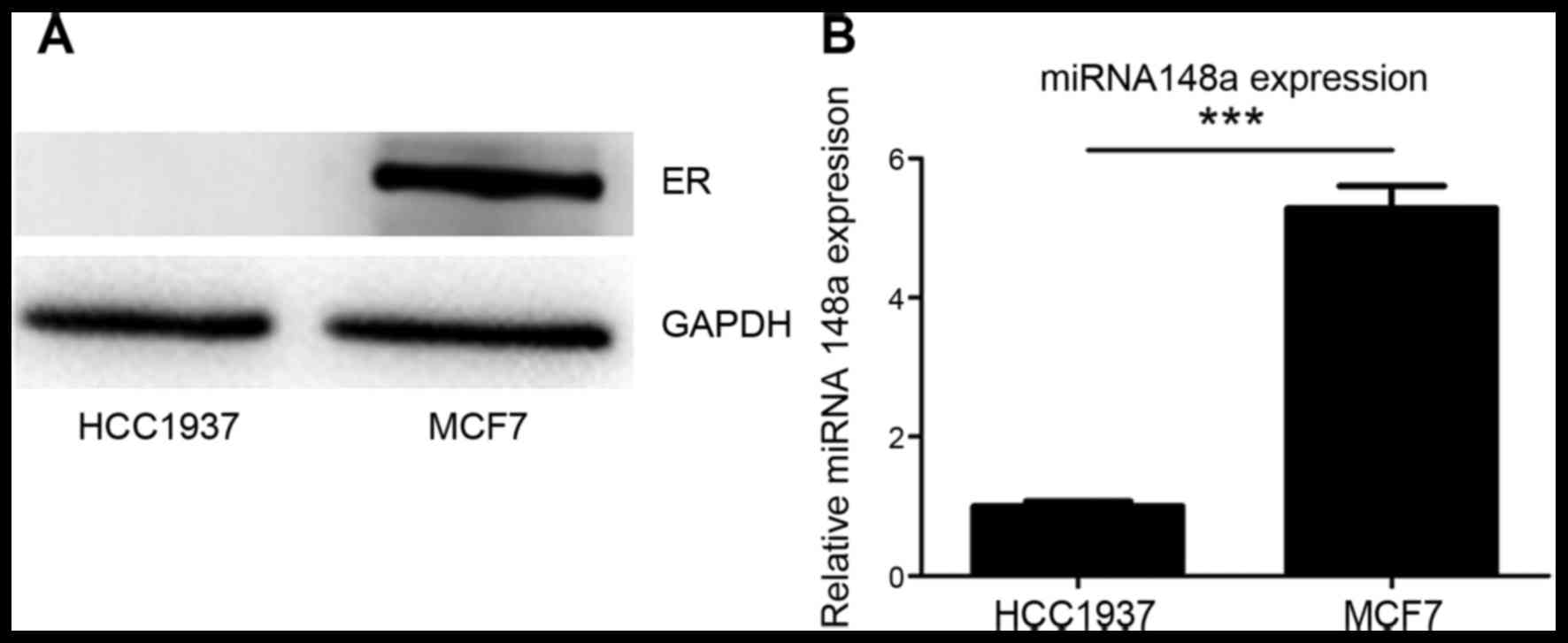
It was confirmed that ER-α expression was negative
in HCC1937 cells and positive in MCF7 cells (Fig. 1A). It was also identified that
miRNA-148a was more highly expressed in MCF7 cells than in HCC1937
cells (Fig. 1B). These data may
indicate an association between ER and miRNA-148a expression in
HCC1937 and MCF7 breast cancer cells.
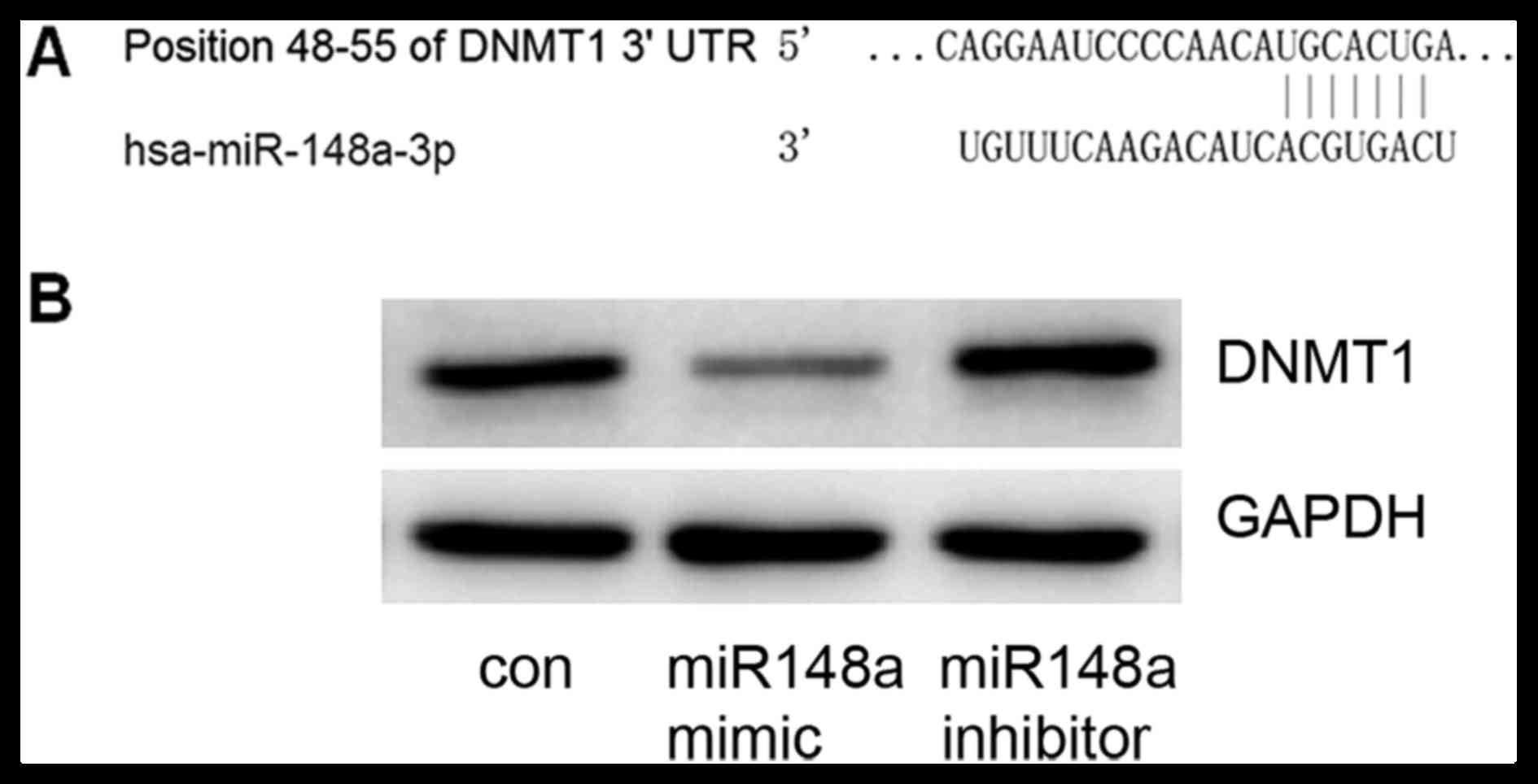
DNMT1 is a target of miRNA-148a
TargetScan was used to predict miRNA-148a target
genes; the DNMT1 gene 3′UTR included 7 sequential pairing bases
with the 5′ region of miRNA-148a (Fig.
2A), indicating that DNMT1 is a potential target for
miRNA-148a. To confirm this prediction, an miRNA-148a mimic or
inhibitor was transfected into MCF7 cells. It was confirmed that
the miRNA-148a mimic could decrease DNMT1 expression and that the
miRNA-148a inhibitor could increase DNMT1 expression (Fig. 2B). These data suggest that DNMT1 is a
target gene of miRNA-148a.
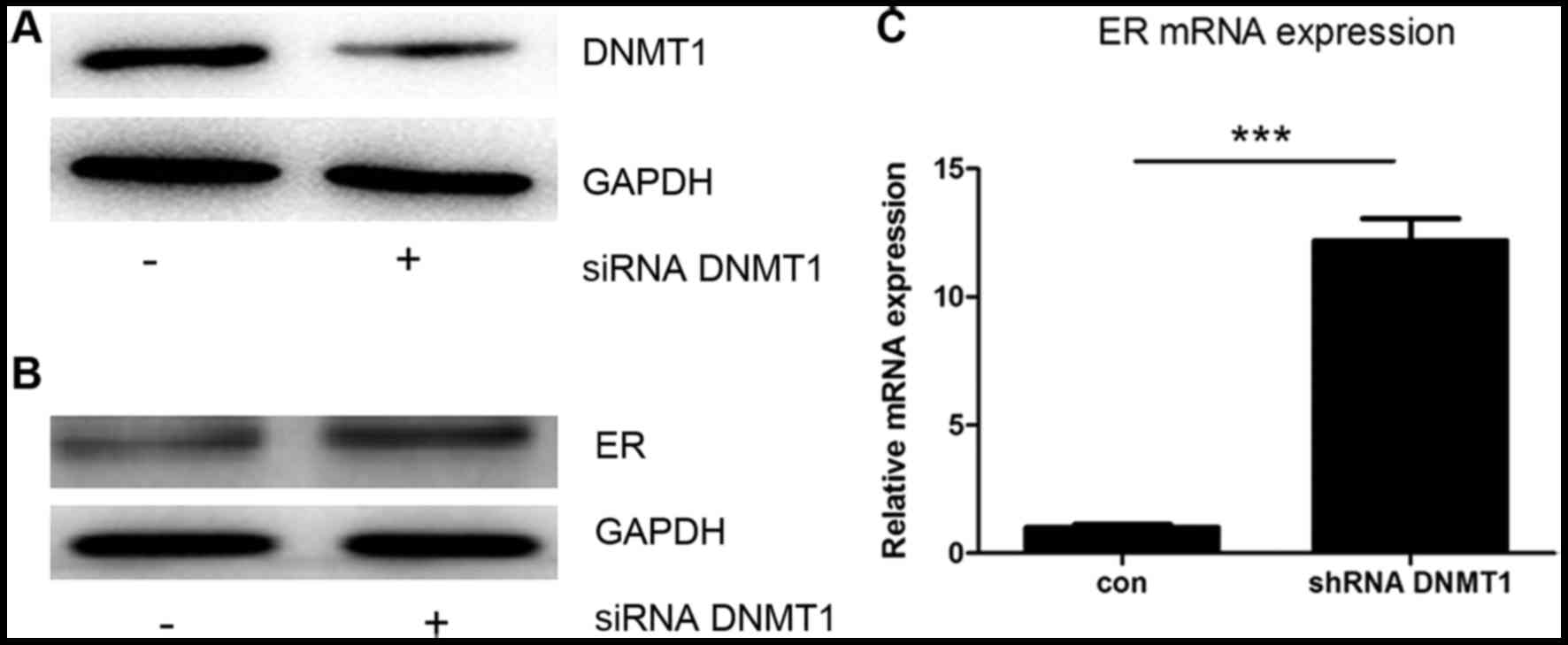
ER-α expression was regulated by
DNMT1
To confirm that DNMT1 can influence ER-α expression
in MCF7 cells, siRNA against DNMT1 was used to knock down DNMT1
expression. Transfection with the siRNA caused a decrease in DNMT1
protein expression (Fig. 3A), and an
increase in the protein and mRNA levels of ER-α (Fig. 3B and C). These data show that
expression of the ER-α gene may be downregulated by the expression
of DNMT1.
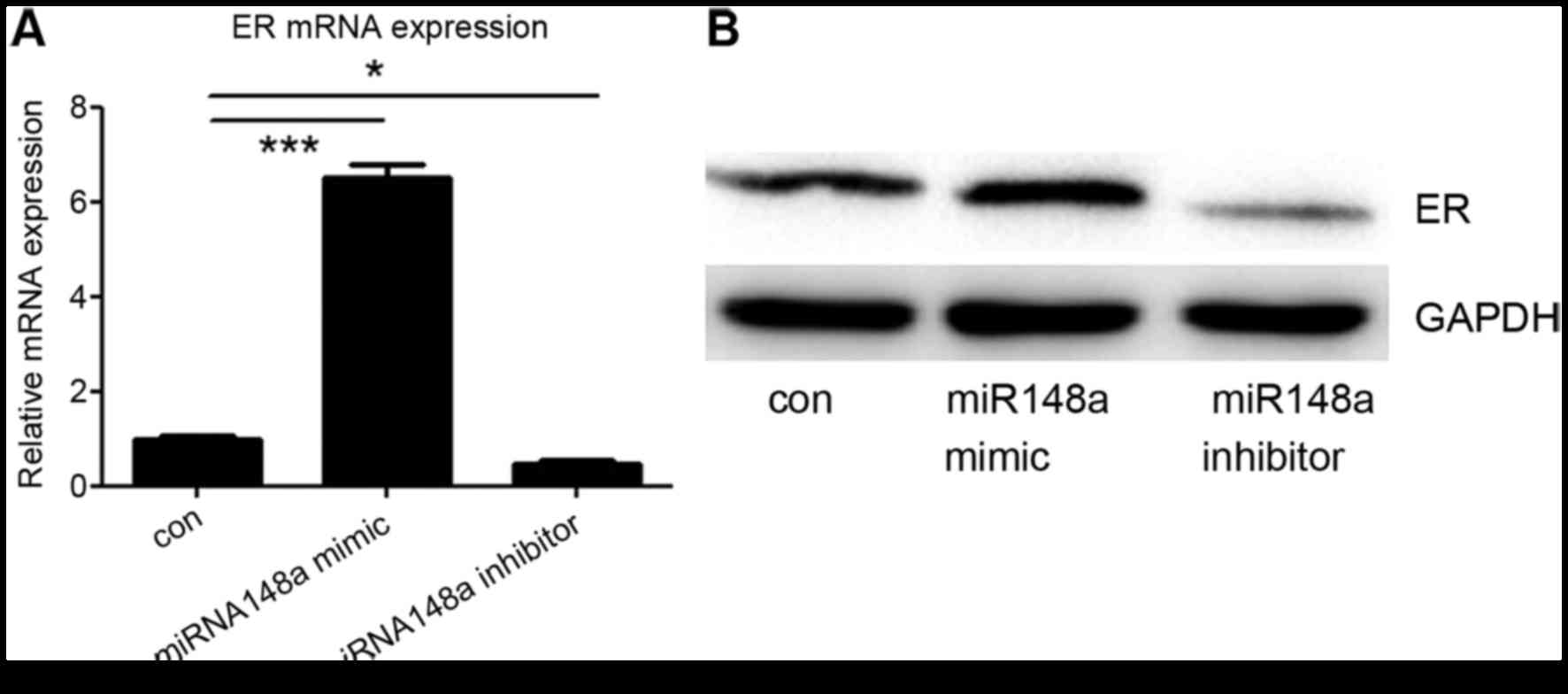
miRNA-148a influences ER-α
expression
Finally, it was investigated whether miRNA-148a
could influence the expression of ER-α in MCF7 cells. An miRNA-148a
mimic or inhibitor was transfected into MCF7 cells, and the protein
and mRNA levels of ER-α were detected. It was demonstrated that
transfection with the miRNA-148a mimic increased ER-α expression,
whereas the miRNA-148a inhibitor decreased ER-α expression,
including at the mRNA and protein level (Fig. 4A and B). These data suggest that
miRNA-148a enhances the expression of ER-α by targeting DNMT1.
Discussion
Although breast cancer is a heterogeneous disease,
two-thirds of breast cancers share the common feature of being
dependent on the presence and interaction of estrogen with the
nuclear ER-α protein (2). Previous
evidence has demonstrated that ER-α is upregulated in mammary
epithelial cells during early stages of breast cancer, and that its
overexpression is an important stimulatory factor for
proliferation, eventually leading to tumor development (11). ER-α is an important therapeutic target
in breast cancer treatment due to its role in orchestrating the
expression of genes from growth-associated pathways (12).
There is a range of breast cancer cell lines
available for research, including MCF-7, HCC1937, T47D, SK-BR3,
MDA-MB-231, MDA-MB-435, HDQ-P1, MCF-10A, MCF-12A. Of these cell
lines, MCF-7 and T47D are ER-α positive and the others, including
HCC1937, are ER-α negative (13). A
previous study investigated the differential expression of miRNAs
in ER-α positive or negative cell lines and identified a number of
differently expressed miRNAs, including miRNA-148a (14). In the present study, it was confirmed
that miRNA-148a was more highly expressed in the ER-α positive
MCF-7 cells than in the ER-α negative HCC1937 cells. It was then
considered whether the elevated expression of miRNA-148a is
associated with the ER-α expression status of MCF-7. A previous
study reported that miRNA-152 caused the downregulation of DNMT1, a
DNA methyltransferase, resulting in the demethylation of the ER-α
gene in human aortic smooth muscle cells (15). As miRNA-152 and miRNA-148a belong to
the same miRNA family and their sequences are similar (16), we hypothesized that miRNA-148a may
also target DNMT1. This hypothesis was confirmed by the present
study.
Methylation is a molecular modification of DNA that
is associated with gene function. The concept that different cell
types have different patterns of methylation was introduced more
than three decades ago (17). A later
study reported that DNA methylation patterns are different between
tissue types, and between tumors and the surrounding tissue
(18). In the context of breast
cancer, a study identified that the number of CpG hypermethylated
islands increased with the decreasing extent of tumor
differentiation, indicating that broad DNA methylation signatures
could be used for differentiating and staging breast cancer
(19). The DNA methylation reaction
is catalyzed by DNMTs including DNMT1, DNMT3a and DNMT3b. DNMT1 is
the most abundant DNA methyltransferase in mammalian cells and the
key maintenance enzyme for hemimethylated DNA during DNA
replication, including for various types of cancer cell (20). Previous studies have demonstrated that
miRNAs may affect the promoter methylation in CpG islands by
targeting the 3′UTR of DNMTs (21–23). In
the present study, it was identified that miRNA-148a targets DNMT1,
which may then regulate ER-α expression. These results indicate a
potential miRNA-based strategy to modulate ER-α expression in
breast cancer cells.
Although the majority of breast cancers are ER-α
positive, ER-α negative breast cancer remains a popular area of
research in oncology. Triple-negative breast cancer (TNBC) is
defined by its lack of ER-α and progesterone-receptor (PR)
expression, along with the absence of human epidermal growth factor
receptor 2 (HER2) overexpression or gene amplification (24). TNBC thus lacks a recognized target for
molecular-oriented therapy. It is a biologically aggressive disease
that is commonly associated with distant recurrence, visceral
metastases and mortality compared with other breast cancer types
(25). Although a number of novel
targeted therapies on TNBC are in development, the effort is not
producing the expected results (26).
miRNAs have been investigated as biomarkers for the diagnosis and
prognosis of cancer, and are potential therapeutic tools against
breast cancer (27). The present
study demonstrated that an miRNA may modulate ER-α expression in
breast cancer cells. This provides a novel perspective for
investigating the role of miRNAs in treating breast cancer.
In conclusion, it was identified in the present
study that miRNA-148a may regulate ER-α expression by inhibiting
DNMT1-mediated DNA methylation in breast cancer cells. This
represents a potential miRNA-based strategy to modulate ER-α
expression, and provides a novel perspective for investigating the
role of miRNAs in treating breast cancer.
References
|
1
|
Parkin DM, Bray F, Ferlay J and Pisani P:
Global cancer statistics, 2002. CA Cancer J Clin. 55:74–108. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Giacinti L, Claudio PP, Lopez M and
Giordano A: Epigenetic information and estrogen receptor alpha
expression in breast cancer. Oncologist. 11:1–8. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Cuzick J, Sestak I, Baum M, Buzdar A,
Howell A, Dowsett M and Forbes JF: ATAC/LATTE investigators: Effect
of anastrozole and tamoxifen as adjuvant treatment for early-stage
breast cancer: 10-year analysis of the atac trial. Lancet Oncol.
11:1135–1141. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Martínez-Galán J, Torres-Torres B, Núñez
MI, López-Peñalver J, Del Moral R, De Almodóvar Ruiz JM, Menjón S,
Concha A, Chamorro C, Ríos S and Delgado JR: Esr1 gene promoter
region methylation in free circulating DNA and its correlation with
estrogen receptor protein expression in tumor tissue in breast
cancer patients. BMC Cancer. 14:592014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Volinia S, Calin GA, Liu CG, Ambs S,
Cimmino A, Petrocca F, Visone R, Iorio M, Roldo C, Ferracin M, et
al: A microRNA expression signature of human solid tumors defines
cancer gene targets. Proc Natl Acad Sci USA. 103:2257–2261. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Takamizawa J, Konishi H, Yanagisawa K,
Tomida S, Osada H, Endoh H, Harano T, Yatabe Y, Nagino M, Nimura Y,
et al: Reduced expression of the let-7 microRNAs in human lung
cancers in association with shortened postoperative survival.
Cancer Res. 64:3753–3756. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yahya SM and Elsayed GH: A summary for
molecular regulations of miRNAs in breast cancer. Clin Biochem.
48:388–396. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yan L, Nass SJ, Smith D, Nelson WG, Herman
JG and Davidson NE: Specific inhibition of DNMT1 by antisense
oligonucleotides induces re-expression of estrogen receptor-alpha
(ER) in ER-negative human breast cancer cell lines. Cancer Biol
Ther. 2:552–556. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Xu Q, Jiang Y, Yin Y, Li Q, He J, Jing Y,
Qi YT, Li W, Lu B, Peiper SS, et al: A regulatory circuit of
mir-148a/152 and DNMT1 in modulating cell transformation and tumor
angiogenesis through IGF-IR and IRS1. J Mol Cell Biol. 5:3–13.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Huang B, Warner M and Gustafsson JÅ:
Estrogen receptors in breast carcinogenesis and endocrine therapy.
Mol Cell Endocrinol 418 Pt. 3:240–244. 2015. View Article : Google Scholar
|
|
12
|
Shoker BS, Jarvis C, Clarke RB, Anderson
E, Hewlett J, Davies MP, Sibson DR and Sloane JP: Estrogen
receptor-positive proliferating cells in the normal and
precancerous breast. Am J Pathol. 155:1811–1815. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Liu Y, Liu R, Fu P, Du F, Hong Y, Yao M,
Zhang X and Zheng S: N1-guanyl-1,7-diaminoheptane sensitizes
estrogen receptor negative breast cancer cells to doxorubicin by
preventing epithelial-mesenchymal transition through inhibition of
eukaryotic translation initiation factor 5A2 activation. Cell
Physiol Biochem. 36:2494–2503. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
de Souza Rocha, Simonini P, Breiling A,
Gupta N, Malekpour M, Youns M, Omranipour R, Malekpour F, Volinia
S, Croce CM, Najmabadi H, et al: Epigenetically deregulated
microRNA-375 is involved in a positive feedback loop with estrogen
receptor alpha in breast cancer cells. Cancer Res. 70:9175–9184.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang YS, Chou WW, Chen KC, Cheng HY, Lin
RT and Juo SH: MicroRNA-152 mediates DNMT1-regulated DNA
methylation in the estrogen receptor α gene. PLoS One.
7:e306352012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chen Y, Song YX and Wang ZN: The
microRNA-148/152 family: Multi-faceted players. Mol Cancer.
12:432013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Razin A and Szyf M: DNA methylation
patterns. Formation and function. Biochim Biophys Acta.
782:331–342. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Szyf M: DNA methylation signatures for
breast cancer classification and prognosis. Genome Med. 4:262012.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yan PS, Perry MR, Laux DE, Asare AL,
Caldwell CW and Huang TH: CpG island arrays: An application toward
deciphering epigenetic signatures of breast cancer. Clin Cancer
Res. 6:1432–1438. 2000.PubMed/NCBI
|
|
20
|
Li E, Bestor TH and Jaenisch R: Targeted
mutation of the DNA methyltransferase gene results in embryonic
lethality. Cell. 69:915–926. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chen KC, Wang YS, Hu CY, Chang WC, Liao
YC, Dai CY and Juo SH: OxLDL up-regulates microRNA-29b, leading to
epigenetic modifications of MMP-2/MMP-9 genes: A novel mechanism
for cardiovascular diseases. FASEB J. 25:1718–1728. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Huang J, Wang Y, Guo Y and Sun S:
Down-regulated microRNA-152 induces aberrant DNA methylation in
hepatitis B virus-related hepatocellular carcinoma by targeting DNA
methyltransferase 1. Hepatology. 52:60–70. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Braconi C, Huang N and Patel T:
MicroRNA-dependent regulation of DNA methyltransferase-1 and tumor
suppressor gene expression by interleukin-6 in human malignant
cholangiocytes. Hepatology. 51:881–890. 2010.PubMed/NCBI
|
|
24
|
Rakha EA, El-Sayed ME, Green AR, Lee AH,
Robertson JF and Ellis IO: Prognostic markers in triple-negative
breast cancer. Cancer. 109:25–32. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Anders CK and Carey LA: Biology,
metastatic patterns, and treatment of patients with triple-negative
breast cancer. Clin Breast Cancer. 9 Suppl 2:S73–S81. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tomao F, Papa A, Zaccarelli E, Rossi L,
Caruso D, Minozzi M, Vici P, Frati L and Tomao S: Triple-negative
breast cancer: New perspectives for targeted therapies. Onco
Targets Ther. 8:177–193. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Bertoli G, Cava C and Castiglioni I:
MicroRNAs: New biomarkers for diagnosis, prognosis, therapy
prediction and therapeutic tools for breast cancer. Theranostics.
5:1122–1143. 2015. View Article : Google Scholar : PubMed/NCBI
|