Introduction
Breast cancer is a malignant tumor that often occurs
in females, comprising ~7–10%; of all systemic malignant tumors.
Tumor recurrence rate, metastasis rate, and mortality are high in
patients with advanced breast cancer (1). As breast cancer is characterized by high
rates of metastasis due to the presence of lymphatic vessels and
reflux veins in the female breast, it is easy for breast cancer to
transfer to the lung, liver, brain, and other organs during the
early stages, resulting in poor clinical treatment and high
mortality (2). In recent years, with
developments in molecular biology and evidence-based medicine,
treatment methods utilizing a combination of surgical resection
with radiotherapy, chemotherapy, and targeted therapy has been
developed to combat breast cancer. Although this comprehensive
treatment method has a certain curative effect, the recurrence and
metastasis rates for breast cancer patients remain high and
prognosis is generally poor (3–5).
Matrix metalloproteinases (MMPs) are a family of
zinc-dependent proteases, and MMP-2 and MMP-9 are major members of
the MMP family (6,7). MMP-2 and MMP-9 are mainly secreted by
tumor cells and stromal cells in the form of zymogens. After
activation via hydrolysis, MMP-2 and MMP-9 degrade basement
membrane (BM) type IV collagen, affecting the ability of BMs to
impede tumor cell movement (8).
Research has shown that MMP-2 and MMP-9 play key roles in degrading
extracellular matrices and promoting tumor invasion and metastasis
(9,10).
The study aimed to investigate MMP-2 and MMP-9
expression levels in breast cancer and their relationships with
breast cancer clinicopathological parameters and prognosis. In this
study, two breast cancer cell lines (MDA-MB-231 and MCF-7) and one
human normal breast cell line (HS578Bst) were cultured in
vitro. Fluorescence RT-PCR and western blotting were used to
detect the expressions of MMP-2 and MMP-9 in cells at mRNA and
protein levels. MMP-2 and MMP-9 expression levels in breast cancer
tissues and tumor-adjacent normal tissues were detected using
immunohistochemistry (IHC) and the relationship between MMP-2 and
MMP-9 expression and clinicopathological parameters and prognosis
was studied.
Materials and methods
Materials
Breast cancer cell lines MDA-MB-231 and MCF-7 and
human normal breast cell line HS578Bst were acquired from the Cell
Banks of the Chinese Academy of Sciences (Shanghai, China). The
following reagents were used: Roswell Park Memorial Institute-1640
(RPMI-1640) medium and fetal bovine serum (FBS) (Gibco, Carlsbad,
CA, USA); FBS (HyClone Laboratories, Logan, UT, USA); TRIzol kits,
reverse transcription kits, and RT-PCR kits (Invitrogen, Carlsbad,
CA, USA); MMP-2, MMP-9, glyceraldehyde 3-phosphate dehydrogenase
(GAPDH) rabbit anti-human primary polyclonal antibodies (cat. no.
10373-2-AP, 10375-2-AP and 10494-1-AP), and mouse anti-rabbit
horse-radish peroxidase (HRP) secondary monoclonal antibodies (cat.
no. HRP-60004; Proteintech Group, Inc., Wuhan, China);
bicinchoninic acid (BCA) protein quantification kits (Beyotime
Institute of Biotechnology, Jiangsu, China); and IHC kit SP-9001
(Beijing Zhongshan Golden Bridge Biotechnology Co., Ltd., Beijing,
China). Primer synthesis was performed by Takara (Dalian,
China).
A total of 80 female breast cancer patients
accompanied by complete clinical data admitted to Yidu Central
Hospital of Weifang for treatment between January, 2007 and
December, 2010 were selected for this study. Patients were aged
24–78 years (median age, 55 years). All patients were clinically
and pathologically diagnosed with breast cancer and received
surgical treatment for the first time without any prior history of
receiving radiotherapy and chemotherapy. Informed consents were
signed by the patients and/or guardians. The study was approved by
the Ethics Committee of Yidu Central Hospital of Weifang. Breast
cancer tissue samples were extracted from tumors by excision.
Tumor-adjacent normal tissues were extracted from an area 10 cm
away from the tumor edge (no infiltration of tumor cells was
observed by microscopy). Specimens were fixed with 10% formaldehyde
and embedded in conventional paraffin after extraction. All
patients had complete follow-up records, including age, tumor size,
lymph node status, clinical staging and survival condition.
Detection of cellular mRNA MMP-2 and
MMP-9 expression by RT-PCR
HS578Bst, MCF-7 and MDA-MB-231 cells were cultured
in RPMI-1640 medium containing 10% FBS in an incubator at 37°C with
5% CO2. The medium was changed every other day and cells
were digested by Trypsin and then subcultured upon initiation of
cell fusion.
Cells were collected during the logarithmic growth
phase following digestion and centrifugation. Total RNA was
extracted from the sample using TRIzol kits according to
manufacturer's instructions. RNA concentration and purity were
measured, with an A260/A280 value from 1.8–2.0 deemed acceptable.
Reverse transcription was conducted according to the instructions
provided in the reverse transcription kit, and mRNA expression was
detected according to the methods provided in the RT-PCR kit using
the RT-derived cDNA as the template (primer sequences are listed in
Table I). GAPDH was selected as the
internal reference. The reaction conditions were as follows: 94°C
for 5 min; 94°C for 30 sec, 57°C for 30 sec, 72°C for 30 sec for 30
cycles; 72°C for 5 min. Ct values were generated by instrument
software (ABI7300; Thermo Fisher Scientific, Waltham, MA, USA).
Relative expression levels were calculated using the
2−ΔCt method according to the following formula: ΔCt
(target gene) = Ct (target gene)-Ct (control gene).
 | Table I.RT-PCR primer sequences. |
Table I.
RT-PCR primer sequences.
| Gene | Primer sequences |
|---|
| MMP-2 | F:
5′-CTCATCGCAGATGCCTGGAA-3′ |
|
| R:
5′-TTCAGGTAATAGGCACCCTTGAAGA-3′ |
| MMP-9 | F:
5′-ACGCACGACGTCTTCCAGTA-3′ |
|
| R:
5′-CCACCTGGTTCAACTCACTCC-3′ |
| GAPDH | F:
5′-GCACCGTCAAGGCTGAGAAC-3′ |
|
| R:
5′-TGGTGAAGACGCCAGTGGA-3′ |
Detection of cellular MMP-2 and MMP-9
expression by western blot analysis
Digestion and centrifugation were used to collect
cells during the logarithmic growth phase. Cell lysates were
generated and protein concentrations were determined by BCA assay.
Protein samples (50 µg) were then loaded and separated by sodium
dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE).
Isolated proteins were transferred to polyvinylidene difluoride
(PVDF) membranes and blocked with 5% skim milk powder at room
temperature for 2 h. Rabbit anti-human MMP-2, MMP-9, and GAPDH
primary polyclonal antibodies (1:1,000) were added, respectively,
and membranes were incubated overnight at 4°C. Membranes were then
completely washed by Tris-buffered saline with Tween-20 (TTBS) and
mouse anti-rabbit secondary monoclonal antibodies (1:2,000) were
added, with the membrane incubated for 1 h at room temperature.
Enhanced chemiluminescence (ECL) development was conducted in the
dark, with a gel imager (Bio-Rad Laboratories, Hercules, CA, USA)
used to obtain results. GADPH was used as an internal reference and
gray scale analysis was performed.
Detection of MMP-2 and MMP-9 protein
expression in pathological tissues by IHC
The procedure was carried out according to the
instructions of the IHC kit. Briefly, after paraffin sections were
dewaxed, endogenous peroxidases were inactivated with 3%
H2O2. Citrate buffer was used for thermal
remediation and proteins were blocked with 10% goat serum. MMP-2
and MMP-9 primary antibodies (1:100 dilution) were applied,
respectively, and samples were incubated at 4°C overnight. After
washing with phosphate-buffered saline (PBS) three times,
biotin-labeled secondary antibodies were added, with samples
allowed to incubate for 15 min. After that, proteins were washed
with PBS three times and diaminobenzidine (DAB) solution was
applied for color development in the dark. Hematoxylin was used for
restaining, and gum was used for mounting. Finally, pictures were
taken under a microscope (TE2000-U; Nikon, Tokyo, Japan).
MMP-2 and MMP-9 protein expression was noted in the
cytoplasm by the presence of a brown color. The proportion of
positive cells among total cells in the entire visual field were
recorded and divided into four categories: <10% (−); 10–25% (+);
26–75% (++); >75% (+++). Categories - and + represent low
expression while ++ and +++ represent high expression. Scores were
calculated and analyzed.
Examining the relationship between
MMP-2 and MMP-9 expression and breast cancer prognosis
Patients were divided into MMP-2 low expression and
high expression groups as well as MMP-9 low and high expression
groups according to the IHC results. There were no statistically
significant differences in terms of gender, smoking history,
alcohol history, and family history among these groups. Regular
follow-ups were conducted after surgery for 5 years in the form of
phone calls or outpatient reviews. Survival duration was recorded
from the first day after surgery to the patient death or the
follow-up conclusion deadline. MMP-2 and MMP-9 expression effects
on breast cancer patient survival conditions were analyzed
statistically based on follow-up results.
Statistical analysis
Statistical Product and Service Solutions (SPSS)
17.0 (International Business Machines Corp., Armonk, NY, USA) was
used in this study. Measurement data were analyzed by using one-way
analysis of variance (ANOVA). The data were compared between the
two groups using χ2 analysis. Clinical prognostic data
were analyzed by Kaplan-Meier survival analysis. p≤0.05 was
interpreted as a statistically significant difference.
Results
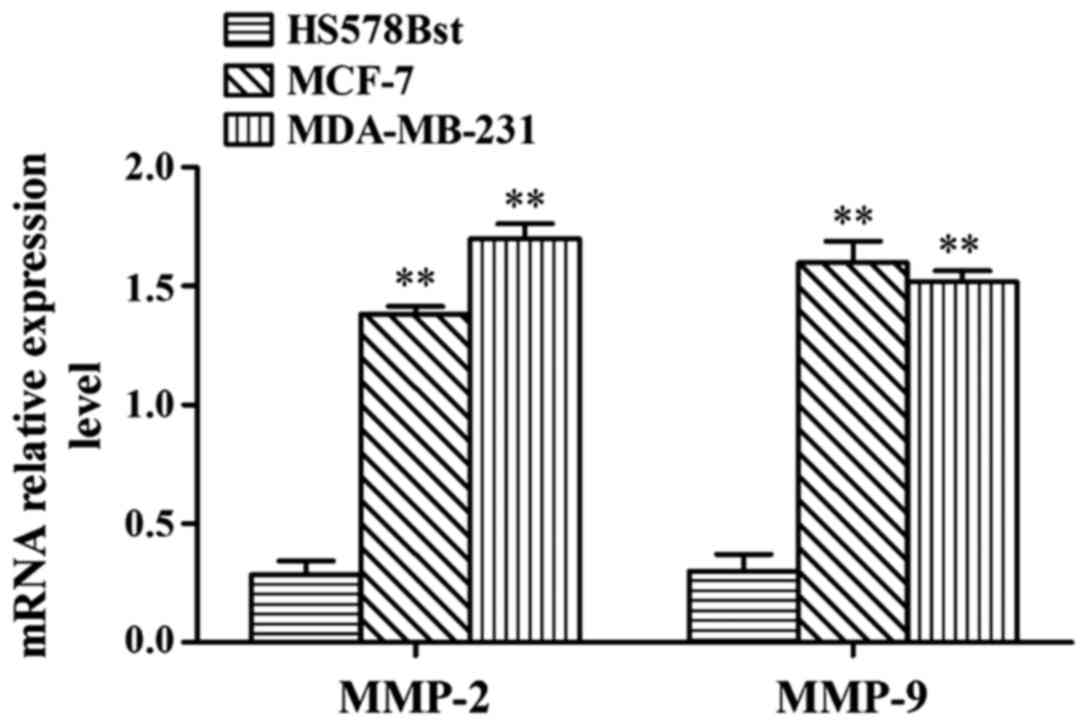
MMP-2 and MMP-9 mRNA expression levels
in MCF-7, MDA-MB-231 and HS578Bst cells
RT-PCR results showed that MMP-2 and MMP-9 mRNA
expression levels in MDA-MB-231 and MCF-7 cells were significantly
higher than in HS578Bst cells (p<0.01) (Fig. 1).
MMP-2 and MMP-9 protein expression in
MCF-7, MDA-MB-231 and HS578Bst cells
Western blot results showed that MMP-2 and MMP-9
protein expression in MDA-MB-231 and MCF-7 cells were significantly
higher than those in HS578Bst cells (p<0.01) (Fig. 2). Representative western blot analyses
for MMP-2 and MMP-9 protein expression (Fig. 2A). Gray-scale densitometry analysis
results for MMP-2 and MMP-9 protein expression (Fig. 2B). MMP-2 and MMP-9 protein expression
in MDA-MB-231 and MCF-7 cells were significantly higher than in
HS578Bst cells, p<0.01.
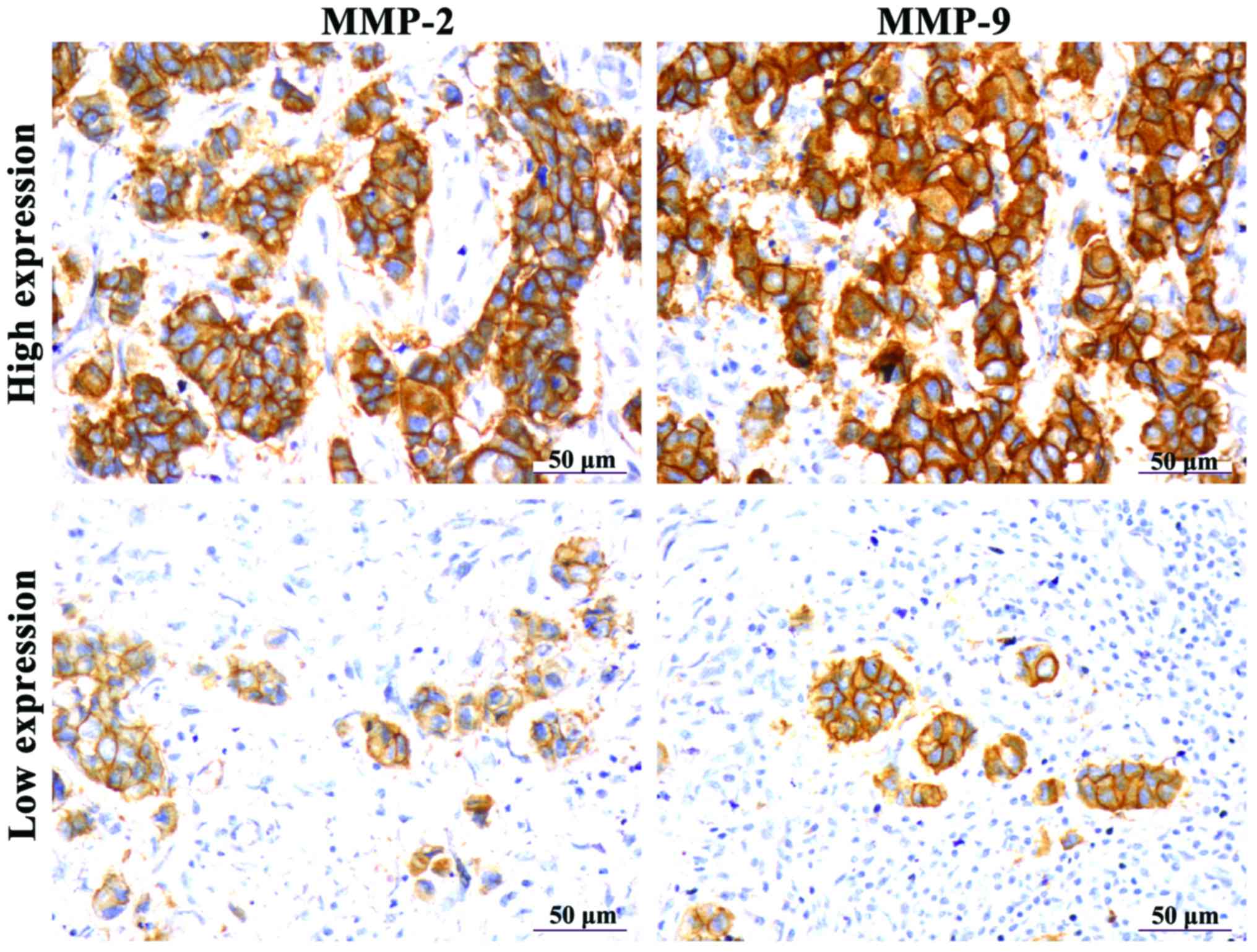
MMP-2 and MMP-9 protein expression in
breast cancer tissues
IHC results showed that the positive expression of
MMP-2 and MMP-9 appeared as brown particles in the cytoplasm
(Fig. 3). Through statistical
analysis, it was found that high expression rates of MMP-2 and
MMP-9 were found in the tumors of 83.75% (67/80) and 78.75% (63/80)
of the patients enrolled in this study, respectively. Differences
in MMP expression between breast cancer tissues and tumor-adjacent
normal tissues were statistically significant (p<0.01) (Table II).
 | Table II.MMP-2 and MMP-9 protein expression
levels in breast cancer and tumor-adjacent normal tissues (cases,
%). |
Table II.
MMP-2 and MMP-9 protein expression
levels in breast cancer and tumor-adjacent normal tissues (cases,
%).
|
|
| MMP-2 | MMP-9 |
|---|
|
|
|
|
|
|---|
| Group | Cases | − and + | ++ and +++ | − and + | ++ and +++ |
|---|
| Breast cancer
tissues | 80 | 13 (16.25) | 67 (83.75) | 17 (21.25) | 63 (78.75) |
| Tumor-adjacent normal
tissues | 40 | 31 (77.50) | 9 (22.50) | 33 (82.50) | 7 (17.50) |
|
χ2-value |
| 43.08 | 41.16 |
| P-value |
| <0.01 | <0.01 |
The relationship between breast cancer
clinicopathological indices and MMP-2 and MMP-9 expression
levels
Table III shows the
analysis results of the relationship between breast cancer
clinicopathological indices and MMP-2 and MMP-9 expression.
Abnormal expression levels of MMP-2 and MMP-9 were correlated with
the occurrence of lymph node metastasis and tumor staging
(p<0.05), but not correlated with patient age and tumor size
(p>0.05).
 | Table III.The relationship between abnormal
MMP-2 and MMP-9 expression and clinicopathological parameters. |
Table III.
The relationship between abnormal
MMP-2 and MMP-9 expression and clinicopathological parameters.
|
|
| MMP-2 positive | MMP-9 positive |
|---|
|
|
|
|
|
|---|
| Clinical
parameter | Cases | Cases, % |
χ2-value | P-value | Cases, % |
χ2-value | P-value |
|---|
| Age (years) |
|
|
|
|
|
|
|
| ≤40 | 33 | 28 (84.85) | 0.05 | >0.05 | 25 (75.76) | 0.30 | >0.05 |
|
>40 | 47 | 39 (82.98) |
|
| 38 (80.85) |
|
|
| Tumor size (cm) |
|
|
|
|
|
|
|
| ≤5 | 49 | 42 (85.71) | 0.36 | >0.05 | 38 (77.55) | 0.11 | >0.05 |
|
>5 | 31 | 25 (80.65) |
|
| 25 (80.65) |
|
|
| Lymphatic
metastasis |
|
|
|
|
|
|
|
| No | 34 | 25 (73.53) | 4.45 | <0.05 | 23 (67.65) | 4.36 | <0.05 |
| Yes | 46 | 42 (91.30) |
|
| 40 (86.96) |
|
|
| Tumor staging |
|
|
|
|
|
|
|
| I–II | 29 | 20 (68.97) | 5.70 | <0.05 | 18 (62.07) | 7.56 | <0.05 |
|
III–IV | 51 | 47 (92.16) |
|
| 45 (88.24) |
|
|
Analysis of survival condition and
breast cancer patient prognosis
A total of 80 patients with breast cancer were
followed up, in which 51 patients survived and 29 patients died
(Table IV). Kaplan-Meier single
factor analysis showed that MMP-2 and MMP-9 expression were
associated with a significant effect on patient prognosis
(p<0.05). Low MMP-2 and MMP-9 expression indicated a relatively
good patient prognosis (Table IV and
Fig. 4).
 | Table IV.Relationship between patient survival
condition and MMP-2 and MMP-9 expression. |
Table IV.
Relationship between patient survival
condition and MMP-2 and MMP-9 expression.
| Group | Cases | 5-year-survival
(cases) | 5-year-survival
(%) | Wald
(log-rank) | P-value |
|---|
| MMP-2 |
|
|
|
|
|
| High
expression | 67 | 39 | 58.21 | 5.007 | <0.05 |
| Low
expression | 13 | 12 | 92.31 |
|
|
| MMP-9 |
|
|
|
|
|
| High
expression | 63 | 36 | 57.14 | 5.584 | <0.05 |
| Low
expression | 17 | 15 | 88.24 |
|
|
Discussion
Breast cancer is a malignant tumor occurring in
breast epithelial tissues, and its pathogenesis is very complex. In
recent years, the incidence rate of breast cancer has increased on
an annual basis, and more young people have developed the disease.
Breast cancer has become an important factor affecting women's
health (11–13).
The MMP-2 gene is located on the human chromosome
16q21. Activated MMP-2 can not only degrade type IV collagen in
BMs, but also can degrade type V, VI and X collagens as well as
gelatins (14). MMP-9 can be secreted
extracellularly in the form of zymogen, and activated MMP-9 can
degrade type IV collagens and fibronectins to destroy BMs and
extracellular matrixes, thereby affecting the adhesion ability of
tumor cells (15). Extracellular
matrixes and BMs are natural barriers in tumor infiltration and
diffusion processes, and cancer cells passing through these
physical barriers is key to tumor cell transfer and migration. It
has been found that type IV collagens are the main components of
the extracellular matrix and BMs. Tumor cells are able to
specifically express MMP-2 and MMP-9 to degrade type IV collagens
and destroy these tissue barriers, thus promoting the invasion and
metastasis of tumor cells (16).
According to studies, MMP-2 and MMP-9 expression levels in breast
cancer tissues were significantly higher than in fibrous adenomas,
and likewise, tumor tissue MMP-2 and MMP-9 expression was
significantly higher in patients with infiltrative breast cancer
and lymph node metastasis than patients with non-infiltrative
cancer and non-lymph node metastasis (17,18). Based
on 168 cases of postmenopausal advanced breast cancer, Rahko et
al found that MMP-9 expression was closely related to clinical
staging, pathology type, and hormone receptor status (19). Li et al (20)used IHC to detect MMP-2 and MMP-9
expression levels in 270 cases of axillary lymph node-negative
breast cancer, finding that MMP-2 and MMP-9 expression were
positively correlated with histological grading.
This study investigated the effects of MMP-2 and
MMP-9 on breast cancer patient pathological parameters and
prognosis. First, the breast cancer cell lines MDA-MB-231 and
MCF-7, as well as the human normal breast cell line HS578Bst were
selected and cultured. MMP-2 and MMP-9 mRNA and protein expression
levels were significantly higher in the breast cancer cell lines
than in HS578Bst cells. Following this, breast cancer tissue
specimens were collected from 80 patients, with tumor-adjacent
normal tissue specimens collected from 40 patients. MMP-2 and MMP-9
expression in breast cancer tissues were significantly higher than
in tumor-adjacent normal tissues. High expression levels of MMP-2
and MMP-9 were correlated with lymph node metastasis and tumor
staging by clinicopathological data analysis. Kaplan-Meier single
factor survival analysis showed that low MMP-2 and MMP-9 expression
indicated relatively good patient prognosis.
In recent years, the relationship between MMPs and
breast cancer has become a research hotspot. Jones et al
found that MMP-2 was mainly expressed in the cytoplasm of breast
cancer cells, and a small amount of these cells were present in the
normal breast duct and the BMs around acini (21). Lebeau et al detected MMP-2 and
MMP-9 expression in 70 infiltrative breast cancer patients, finding
that MMP-2 and MMP-9 were expressed in tumor cells and surrounding
stromal cells, with particularly high expression at the edges of
the tumor infiltration area (22).
This study not only confirmed the presence of high MMP-2 and MMP-9
expression in breast cancer cell lines and breast cancer tissues,
but also confirmed that MMP-2 and MMP-9 expression were associated
with lymph node metastasis, tumor staging and prognosis.
In conclusion, MMP-2 and MMP-9 are highly expressed
in breast cancer and are closely related to lymph node metastasis
and tumor staging. MMP-2 and MMP-9 can be used as reference
indicators for guiding breast cancer treatment and estimating
prognosis.
References
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Garcia-Murillas I, Schiavon G, Weigelt B,
Ng C, Hrebien S, Cutts RJ, Cheang M, Osin P, Nerurkar A, Kozarewa
I, et al: Mutation tracking in circulating tumor DNA predicts
relapse in early breast cancer. Sci Transl Med. 7:302ra1332015.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Im NK, Jang WJ, Jeong CH and Jeong GS:
Delphinidin suppresses PMA-induced MMP-9 expression by blocking the
NF-κB activation through MAPK signaling pathways in MCF-7 human
breast carcinoma cells. J Med Food. 17:855–861. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Shalaby MA, Nounou HA, Ms A, O A, Azzam N
and Saeed HM: Associations between single nucleotide polymorphisms
of COX-2 and MMP-2 genes and colorectal cancer susceptibility in
the Saudi population. Asian Pac J Cancer Prev. 15:4989–4994. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhou R, Xu L, Ye M, Liao M, Du H and Chen
H: Formononetin inhibits migration and invasion of MDA-MB-231 and
4T1 breast cancer cells by suppressing MMP-2 and MMP-9 through
PI3K/AKT signaling pathways. Horm Metab Res. 46:753–760. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chia CY, Kumari U and Casey PJ: Breast
cancer cell invasion mediated by Gα12 signaling involves expression
of interleukins-6 and −8, and matrix metalloproteinase-2. J Mol
Signal. 9:62014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Stoeltzing O, Ahmad SA, Liu W, McCarty MF,
Wey JS, Parikh AA, Fan F, Reinmuth N, Kawaguchi M, Bucana CD, et
al: Angiopoietin-1 inhibits vascular permeability, angiogenesis,
and growth of hepatic colon cancer tumors. Cancer Res.
63:3370–3377. 2003.PubMed/NCBI
|
|
8
|
Zhang W, Wang F, Xu P, Miao C, Zeng X, Cui
X, Lu C, Xie H, Yin H, Chen F, et al: Perfluorooctanoic acid
stimulates breast cancer cells invasion and up-regulates matrix
metalloproteinase-2/-9 expression mediated by activating NF-κB.
Toxicol Lett. 229:118–125. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Iochmann S, Bléchet C, Chabot V, Saulnier
A, Amini A, Gaud G, Gruel Y and Reverdiau P: Transient RNA
silencing of tissue factor pathway inhibitor-2 modulates lung
cancer cell invasion. Clin Exp Metastasis. 26:457–467. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Safranek J, Pesta M, Holubec L, Kulda V,
Dreslerova J, Vrzalova J, Topolcan O, Pesek M, Finek J and Treska
V: Expression of MMP-7, MMP-9, TIMP-1 and TIMP-2 mRNA in lung
tissue of patients with non-small cell lung cancer (NSCLC) and
benign pulmonary disease. Anticancer Res. 29:2513–2517.
2009.PubMed/NCBI
|
|
11
|
Huang S, Chen J and Jia Y: Research on the
correlation of MMP9 and p53 expression with the prognosis of triple
negative breast cancer. Xiandai Shengwu Yixue Jinzhan. 14:881–884.
2014.
|
|
12
|
Choi JS, Baek HM, Kim S, Kim MJ, Youk JH,
Moon HJ, Kim EK and Nam YK: Magnetic resonance metabolic profiling
of breast cancer tissue obtained with core needle biopsy for
predicting pathologic response to neoadjuvant chemotherapy. PLoS
One. 8:e838662013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yari K and Rahimi Z, Moradi MT and Rahimi
Z: The MMP-2 −735 C allele is a risk factor for susceptibility to
breast cancer. Asian Pac J Cancer Prev. 15:6199–6203. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Fernandez-Garcia B, Eiró N, Marín L,
González-Reyes S, González LO, Lamelas ML and Vizoso FJ: Expression
and prognostic significance of fibronectin and matrix
metalloproteases in breast cancer metastasis. Histopathology.
64:512–522. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Li H, Huang J, Yang B, Xiang T, Yin X,
Peng W, Cheng W, Wan J, Luo F, Li H, et al: Mangiferin exerts
antitumor activity in breast cancer cells by regulating matrix
metalloproteinases, epithelial to mesenchymal transition, and
β-catenin signaling pathway. Toxicol Appl Pharmacol. 272:180–190.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Parsons SL, Watson SA, Collins HM, Griffin
NR, Clarke PA and Steele RJ: Gelatinase (MMP-2 and −9) expression
in gastrointestinal malignancy. Br J Cancer. 78:1495–1502. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Bucan V, Mandel K, Bertram C, Lazaridis A,
Reimers K, Park-Simon TW, Vogt PM and Hass R: LEF-1 regulates
proliferation and MMP-7 transcription in breast cancer cells. Genes
Cells. 17:559–567. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Katunina AI, Gershtein ES, Ermilova VD,
Tereshkina IV, Nazarenko AY, Tyleuova AA, Dvorova EK, Karabekova
ZK, Gritskevich MV and Berezov TT: Matrix metalloproteinases 2, 7,
and 9 in tumors and sera of patients with breast cancer. Bull Exp
Biol Med. 151:359–362. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Rahko E, Jukkola A, Melkko J, Paavo P,
Bloigu R, Talvensaari-Mattila A and Turpeenniemi-Hujanen T: Matrix
metalloproteinase-9 (MMP-9) immunoreactive protein has modest
prognostic value in locally advanced breast carcinoma patients
treated with an adjuvant antiestrogen therapy. Anticancer Res.
24:4247–4253. 2004.PubMed/NCBI
|
|
20
|
Li HC, Cao DC, Liu Y, Hou YF, Wu J, Lu JS,
Di GH, Liu G, Li FM, Ou ZL, et al: Prognostic value of matrix
metalloproteinases (MMP-2 and MMP-9) in patients with lymph
node-negative breast carcinoma. Breast Cancer Res Treat. 88:75–85.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Jones JL, Glynn P and Walker RA:
Expression of MMP-2 and MMP-9, their inhibitors, and the activator
MT1-MMP in primary breast carcinomas. J Pathol. 189:161–168. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lebeau A, Müller-Aufdemkamp C, Allmacher
C, Sauer U, Nerlich A, Lichtinghagen R and Löhrs U: Cellular
protein and mRNA expression patterns of matrix
metalloproteinases-2, −3 and −9 in human breast cancer: Correlation
with tumour growth. J Mol Histol. 35:443–455. 2004. View Article : Google Scholar : PubMed/NCBI
|