Introduction
Despite its steadily declining incidence, gastric
cancer (GC) causes more than 723,000 deaths a year and is still the
third-leading cause of cancer-related death worldwide (1). Poor survival of GC is attributed to the
high percentage of patients with an advanced stage of the disease
at the time of first diagnosis. Approximately 35% of GC patients
harbor distant metastasis at first clinical presentation (2). In addition, GC is characterized by a
high rate of locoregional as well as distant recurrence after
surgical resection with curative intend (3). Currently, the basis for treatment of
patients with metastatic or recurrent disease is chemotherapy.
Although some of these regimens demonstrated activity in recent
phase III trials the median overall survival of patients with
advanced GC remains disappointing (4–7). To
improve the outcome of patients with advanced GC novel treatment
options are urgently needed. One promising approach might be the
use of targeted anticancer therapies. Such therapeutic regimes aim
to inhibit cancer development by interfering with distinct
molecules which are involved in pathways that regulate important
cancer cell abilities like resistance to growth inhibition and
apoptosis, cell invasion, metastasis or the induction of
angiogenesis (8). One common
principle to identify such target molecules is to study the
expression and prognostic significance of proteins in cancer tissue
specimens.
In this context, the inhibitor of apoptosis protein
(IAP) family has attracted considerable attention. IAPs
significantly influence essential biological functions during
cancer development as well as resistance to conventional
chemotherapeutic agents by inhibiting cell death pathways and by
promoting cancer cell proliferation, migration and metastasis at
the same time (9–12). The most extensively studied IAP family
members are survivin/BIRC5 and X-linked inhibitor of apoptosis
(XIAP/BIRC4) (13,14). Undoubtedly, both survivin and XIAP are
multifunctional proteins regulating distinct cellular processes
(9,11,13,15–18).
However, survivin and XIAP have been demonstrated to unfold their
antiapoptotic and pro-metastatic activities by direct interaction.
In this context, survivin has been implicated in stabilization of
XIAP thus preventing XIAP from polyubiquitination and subsequent
proteosomal degradation (19).
Consequently, a survivin-XIAP complex synergistically inhibits
caspases and activates NF-κB signaling (11,20). Of
note, NF-κB activation by a survivin-XIAP complex was linked to
tumor cell invasion and formation of metastasis (11). Although the expression of survivin and
XIAP could be correlated with clinicopathological parameters and
was linked to poor survival in GC (21–27), none
of these studies took the pro-invasive and metastatic functions of
a survivin-XIAP complex into account and analyzed the expression of
both IAPs in different tumor compartments such as the tumor center,
invasion front and metastases. Thus, aim of our study was to
specifically investigate the expression of survivin and XIAP in the
tumor center, invasion front and lymph node metastases of
surgically resected specimens of GC. Considering the synergistic
function of XIAP and survivin, we assessed the prognostic value of
the tumor compartment-related expression of both IAPs and its
association with clinicopathological variables in GC patients
according to the REporting recommendations for tumor MARKer
prognostic studies (REMARK) (28).
Materials and methods
Patient selection and
clinicopathological data
Medical charts of patients with primary gastric
adenocarcinoma who had undergone surgical resection at the
Department of Surgery (A) of the University Hospital Duesseldorf
(Duesseldorf, Germany) between 1995 and 2013 were retrospectively
reviewed. All patients were treated in curative intend, using total
or subtotal gastrectomy and D2 lymphadenectomy. Unfortunately, for
most of the patients information on adjuvant chemotherapy was
unavailable. Exclusion criteria for this study were preoperative
chemotherapy, incomplete pathological report and insufficient tumor
material for further analysis. Patients lost to follow up, or with
incomplete resection and patients who died because of postoperative
complications within 30 days after surgery were excluded from our
survival analysis. The study was carried out in accordance to Good
Clinical Practice, the Declaration of Helsinki and local rules as
well as regulations of the country. The Institutional Review Board
(IRB) of the Medical Faculty, Heinrich Heine University Duesseldorf
approved the use of tissue specimens and clinicopathological data
for this study (IRB-no. 3821). Clinicopathological parameters were
extracted from the clinical and histopathological reports.
Surgically resected tumors were staged according to the 7th TNM
edition of the UICC (29). Overall
survival data were retrieved from a prospectively maintained
database at the Department of Surgery (A).
Tissue microarray and
immunohistochemistry
Formalin-fixed and paraffin embedded (FFPE) tumor
specimens were obtained from the Institute of Pathology, University
Hospital Duesseldorf. Tissue microarray (TMA) construction was
performed as recently described (30). If available two cylinders of 1.0 mm
diameter were taken from the tumor center and invasion front, one
from tumor surrounding non-malignant mucosa and one from a lymph
node metastasis. As control for immunohistochemical reaction two
cylinders of normal tonsille tissue and colon cancer were inserted
into the right upper corner of each recipient block. For
immunohistochemistry 2 µm sections were cut from each TMA block and
mounted on superfrost glass slides.
For immunohistochemical staining procedures rabbit
polyclonal anti-survivin antibody (NB500-201; 1:1,000 dilution;
Novus Biologicals, Littleton, CO, USA) and mouse monoclonal
anti-XIAP antibody (Clone 48; 1:50 dilution; BD Biosciences, San
Jose, CA, USA) were used. Isotype control was conducted using mouse
IgG1 kappa (MOPC-21; Abcam, Cambridge, UK) and rabbit
Immunoglobulin Fraction (Code X0903; Dako, Glostrup, Denmark).
Expression levels were estimated according to an immunoreactivity
score (IRS) as described previously (30–32).
Two independent investigators who were blinded to
clinicopathological data examined all sections (LD and MT). In case
of differing results both observers re-examined the slide
simultaneously and a consensus decision was made. For survivin,
nuclear and cytoplasmic protein expression were separately
determined.
Statistical analysis
The mean IRS of both tissue cores from tumor centers
and invasion fronts was calculated for statistical analysis.
Variation of IAP expression in different tumor compartments was
analyzed using the Mann-Whitney U test or Wilcoxon matched pairs
test, as indicated. Spearman's correlation coefficient was used to
test a relationship between survivin and XIAP expression. For
survival analysis and to compare expression levels according to
clinicopathological variables, the median IRS for nuclear and
cytoplasmic survivin as well as for XIAP was combined and
categorized into groups of high (>median IRS=IRS>2) or low
(≤median IRS=IRS≤2) survivin and XIAP expression. This homogenous
cut-off value divided our collective of GC patients into two
approximately equally sized groups for both IAPs, survivin and
XIAP. Categorical data were examined using the Fischer's exact
test. Overall survival was defined as the period from the date of
surgery until the date of last follow up or until death of any
cause. For univariate survival analysis, Kaplan-Meier curves were
generated and assessed using the log-rank (Mantel Cox) test. In
addition, hazard ratios (HR) with 95% confidence intervals (CI)
were estimated and multiple regression analysis was performed with
the proportional hazard Cox's regression. Akaike Information
Criterion (AIC) was applied for variable selection, the model with
the lowest AIC was considered as the model which better fits the
data (33). Statistical analyses were
performed using GraphPad Prism for Windows (Version 5, GraphPad
Software, San Diego, CA, USA), SPSS statistics for Windows (Version
17.0; SPSS, Inc., Chicago, IL, USA) and the R statistical software,
version 3.2.2. P<0.05 was considered to indicate a statistically
significant difference.
Results
Patients and outcome
Using our pre-defined inclusion and exclusion
criteria, a total number of 201 patients who underwent total or
subtotal gastrectomy with D2 lymphadenectomy for GC between 1995
and 2013 could be included in our study. Patient's pathological and
clinical characteristics are summarized in Table I. Only 154 patients with a median
follow-up of 25.3 month (range 1–120 month) and median overall
survival of 29.8 (range 1–120) months could be included into our
survival analysis, as 24 patients with incomplete resection, 14
patients who died within 30 days after surgery and 9 patients who
were lost to follow-up had to be excluded. 103 (66.9%) patients
died during the follow-up period.
 | Table I.Patient characteristics (n=201). |
Table I.
Patient characteristics (n=201).
| Variable | No. of patients
(%) |
|---|
| Total | 201 |
| Age |
|
| Median
(range); years | 71
(26–91) |
| Gender |
|
|
Male | 136 (67.7) |
|
Female | 65
(32.3) |
| Lauren
classification |
|
|
Intestinal type | 76
(37.8) |
| Diffuse
type | 78
(38.8) |
| Mixed
type | 47
(23.4) |
| Tumor stage |
|
|
T1/2 | 124 (61.7) |
|
T3/4 | 77
(38.3) |
| Lymph node
metastasis |
|
| N0 | 56
(27.9) |
|
N1+ | 145 (72.1) |
| Distant
metastasis |
|
| M0 | 182 (90.5) |
| M1 | 19 (9.5) |
| Grading |
|
|
G1/2 | 45
(22.4) |
|
G3/4 | 156 (77.6) |
| UICC stage |
|
| UICC
I/II | 98
(48.8) |
| UICC
III/IV | 103 (51.2) |
| Resection
status |
|
| R0 | 177 (88.1) |
| R+ | 24
(11.9) |
Survivin and XIAP expression in
GC
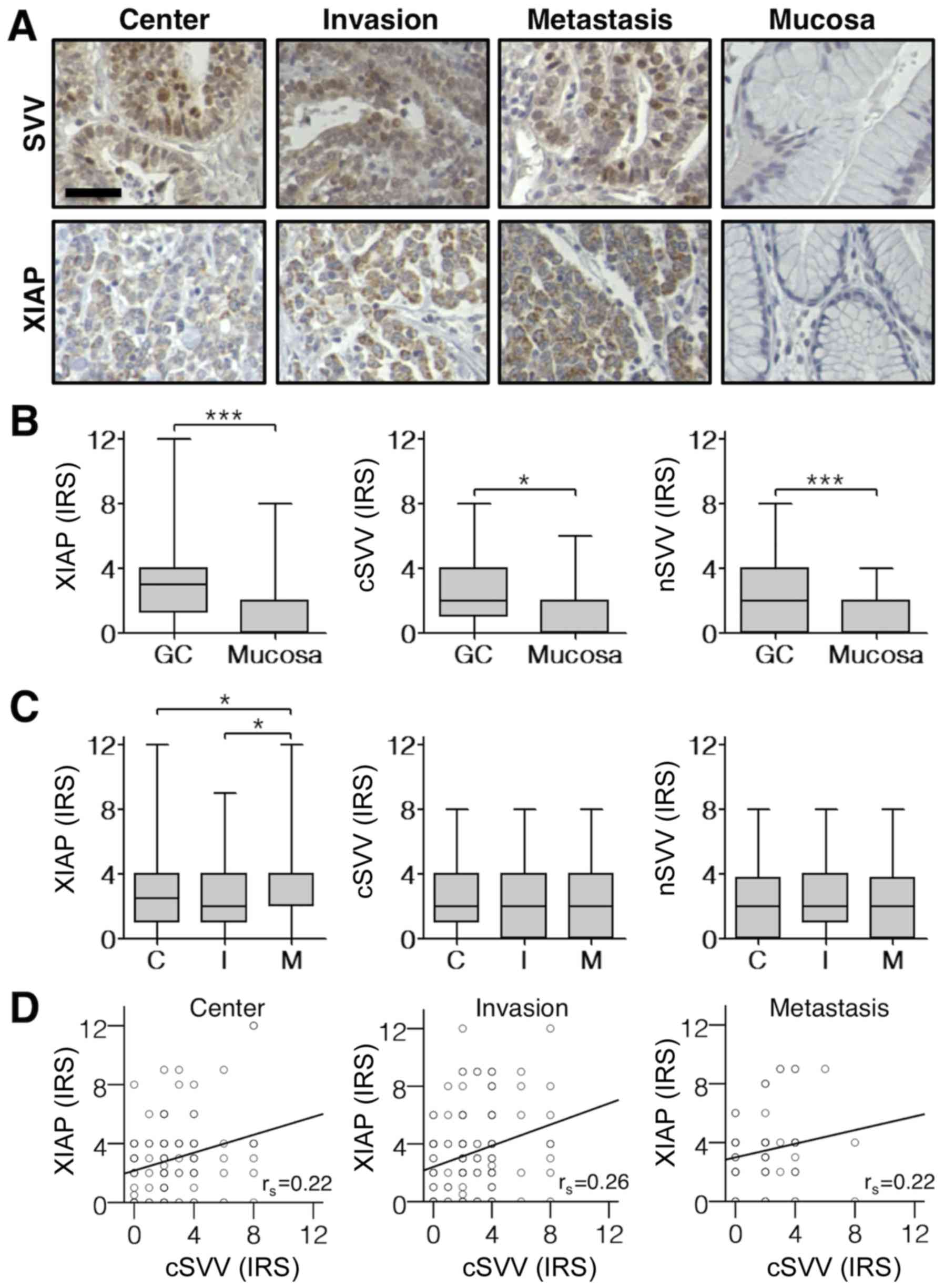
Immunohistochemical staining of TMAs revealed a
cytoplasmic and nuclear expression of survivin, whereas XIAP
expression was exclusively detected in the cytoplasm (Fig. 1A). First, we analyzed expression
patterns of survivin and XIAP in different GC compartments (tumor
center, invasion front and lymph node metastasis) and corresponding
normal mucosa. As shown in Fig. 1B
survivin and XIAP expression levels were significantly elevated in
GC tissues when compared to non-neoplastic mucosa. Furthermore, an
increased expression of XIAP in lymph node metastases became
evident. However, there was no difference in survivin and XIAP
expression levels when comparing tumor centers with tumor invasion
fronts (Fig. 1C). Of note,
cytoplasmic survivin expression correlated positively with XIAP
expression in distinct tumor compartments (Center: P=0.009;
Invasion front: P=0.001; Lymph node metastasis: P=0.047; Fig. 1D).
Correlation between survivin or XIAP
and clinicopathological factors
Next, we categorized the respective IRS of survivin
and XIAP in primary tumors into high (IRS >2) and low (IRS ≤2)
expression and correlated these data with clinicopathological
parameters (Tables II–IV). Using this categorization, we found
that except for nuclear survivin in tumor centers, both IAPs
exhibited higher expression levels in intestinal type GCs when
compared to the diffuse type. In addition, well or moderately
differentiated GC specimens demonstrated higher XIAP expression in
both primary tumor compartments and increased cytoplasmic survivin
in the tumor center. Interestingly, high survivin and XIAP
expression in distinct tumor compartments was associated with less
advanced UICC stages and tumors with an invasion restricted to the
muscularis propria. Surprisingly, tumors without lymph node
metastasis showed higher cytoplasmic survivin expression levels in
the invasion front.
 | Table II.Correlation between XIAP expression
and clinicopathological factors in gastric cancer. |
Table II.
Correlation between XIAP expression
and clinicopathological factors in gastric cancer.
|
| XIAP-center | XIAP-invasion |
|---|
|
|
|
|
|---|
|
| Low n=67 | High n=82 |
| Low n=88 | High n=94 |
|
|---|
|
|
|
|
|
|
|
|
|---|
|
| n | % | n | % | P-value | n | % | n | % | P-value |
|---|
| Age |
|
|
|
| 0.514 |
|
|
|
| 0.302 |
|
≤Median | 33 | 42.3 | 45 | 57.7 |
| 50 | 52.1 | 46 | 47.9 |
|
|
>Median | 34 | 47.9 | 37 | 52.1 |
| 38 | 44.2 | 48 | 55.8 |
|
| Sex |
|
|
|
| 0.369 |
|
|
|
| 0.429 |
|
Men | 17 | 38.6 | 27 | 61.4 |
| 25 | 43.9 | 32 | 56.1 |
|
|
Women | 50 | 47.6 | 55 | 52.4 |
| 63 | 50.4 | 62 | 49.6 |
|
| Lauren |
|
|
|
|
<0.001 |
|
|
|
| 0.005 |
|
diffuse | 34 | 59.6 | 23 | 40.4 |
| 38 | 57.6 | 28 | 42.4 |
|
|
intestinal | 15 | 25.4 | 44 | 74.6 |
| 24 | 33.3 | 48 | 66.7 |
|
|
mixed | 18 | 54.5 | 15 | 45.5 |
| 26 | 59.1 | 18 | 40.9 |
|
| Tumor stage |
|
|
|
| 0.014 |
|
|
|
| 0.049 |
|
T1/T2 | 28 | 35.4 | 51 | 64.6 |
| 47 | 42.3 | 64 | 57.7 |
|
|
T3/T4 | 39 | 55.7 | 31 | 44.3 |
| 41 | 57.7 | 30 | 42.3 |
|
| Lymph nodes |
|
|
|
| 0.074 |
|
|
|
| 0.098 |
|
Negative. N0 | 10 | 30.3 | 23 | 69.7 |
| 19 | 38.0 | 31 | 62.0 |
|
|
Positive. N+ | 57 | 49.1 | 59 | 50.9 |
| 69 | 52.3 | 63 | 47.7 |
|
| Metastasis |
|
|
|
| 0.119 |
|
|
|
| 0.448 |
| M0 | 56 | 42.4 | 76 | 57.6 |
| 78 | 47.3 | 87 | 52.7 |
|
| M1 | 11 | 64.7 | 6 | 35.3 |
| 10 | 58.8 | 7 | 41.2 |
|
| Grading |
|
|
|
| 0.011 |
|
|
|
| 0.023 |
|
G1/G2 | 9 | 25.7 | 26 | 74.3 |
| 14 | 32.6 | 29 | 67.4 |
|
|
G3/G4 | 58 | 50.9 | 56 | 49.1 |
| 74 | 53.2 | 65 | 46.8 |
|
| Resection
margins |
|
|
|
| 0.075 |
|
|
|
| 1.000 |
|
Negative. R0 | 55 | 42.0 | 76 | 58.0 |
| 79 | 48.5 | 84 | 51.5 |
|
|
Positive. R1 | 12 | 66.7 | 6 | 33.3 |
| 9 | 47.4 | 10 | 52.6 |
|
| UICC |
|
|
|
| 0.019 |
|
|
|
| 0.012 |
|
I/II | 20 | 32.8 | 41 | 67.2 |
| 33 | 38.4 | 53 | 61.6 |
|
|
III/IV | 47 | 53.4 | 41 | 46.6 |
| 55 | 57.3 | 41 | 42.7 |
|
 | Table IV.Correlation between nuclear SVV
expression and clinicopathological factors in gastric cancer. |
Table IV.
Correlation between nuclear SVV
expression and clinicopathological factors in gastric cancer.
|
| SVV
(nuc.)-center | SVV
(nuc.)-invasion |
|---|
|
|
|
|
|---|
|
| Low n=95 | High n=53 |
| Low n=120 | High n=54 |
|
|---|
|
|
|
|
|
|
|
|
|---|
|
| n | % | n | % | P-value | n | % | n | % | P-value |
|---|
| Age |
|
|
|
| 0.306 |
|
|
|
| 0.251 |
|
≤Median | 52 | 68.4 | 24 | 31.6 |
| 66 | 73.3 | 24 | 26.7 |
|
|
>Median | 43 | 59.7 | 29 | 40.3 |
| 54 | 64.3 | 30 | 35.7 |
|
| Sex |
|
|
|
| 0.086 |
|
|
|
| 0.163 |
|
Men | 31 | 75.6 | 10 | 24.4 |
| 42 | 76.4 | 13 | 23.6 |
|
|
Women | 64 | 59.8 | 43 | 40.2 |
| 78 | 65.5 | 41 | 34.5 |
|
| Lauren |
|
|
|
| 0.133 |
|
|
|
| 0.005 |
|
diffuse | 40 | 72.7 | 15 | 27.3 |
| 53 | 84.1 | 10 | 15.9 |
|
|
intestinal | 33 | 55.0 | 27 | 45.0 |
| 42 | 60.0 | 28 | 40.0 |
|
|
mixed | 22 | 66.7 | 11 | 33.3 |
| 25 | 61.0 | 16 | 39.0 |
|
| Tumor stage |
|
|
|
| 0.393 |
|
|
|
| 0.503 |
|
T1/T2 | 48 | 60.8 | 31 | 39.2 |
| 70 | 66.7 | 35 | 33.3 |
|
|
T3/T4 | 47 | 68.1 | 22 | 31.9 |
| 50 | 72.5 | 19 | 27.5 |
|
| Lymph nodes |
|
|
|
| 0.839 |
|
|
|
| 0.101 |
|
Negative. N0 | 21 | 61.8 | 13 | 38.2 |
| 29 | 59.2 | 20 | 40.8 |
|
|
Positive. N+ | 74 | 64.9 | 40 | 35.1 |
| 91 | 72.8 | 34 | 27.2 |
|
| Metastasis |
|
|
|
| 1.000 |
|
|
|
| 0.778 |
| M0 | 84 | 64.1 | 47 | 35.9 |
| 108 | 68.4 | 50 | 31.6 |
|
| M1 | 11 | 64.7 | 6 | 35.3 |
| 12 | 75.0 | 4 | 25.0 |
|
| Grading |
|
|
|
| 0.843 |
|
|
|
| 0.089 |
|
G1/G2 | 22 | 62.9 | 13 | 37.1 |
| 25 | 58.1 | 18 | 41.9 |
|
|
G3/G4 | 73 | 64.6 | 40 | 35.4 |
| 95 | 72.5 | 36 | 27.5 |
|
| Resection
margins |
|
|
|
| 0.801 |
|
|
|
| 0.192 |
|
Negative. R0 | 82 | 63.6 | 47 | 36.4 |
| 104 | 67.5 | 50 | 32.5 |
|
|
Positive. R1 | 13 | 68.4 | 6 | 31.6 |
| 16 | 80.0 | 4 | 20.0 |
|
| UICC |
|
|
|
| 0.036 |
|
|
|
| 0.074 |
|
I/II | 32 | 53.3 | 28 | 46.7 |
| 51 | 62.2 | 31 | 37.8 |
|
|
III/IV | 63 | 71.6 | 25 | 28.4 |
| 69 | 75.0 | 23 | 25.0 |
|
XIAP is an independent negative
prognostic marker in diffuse and mixed type GC
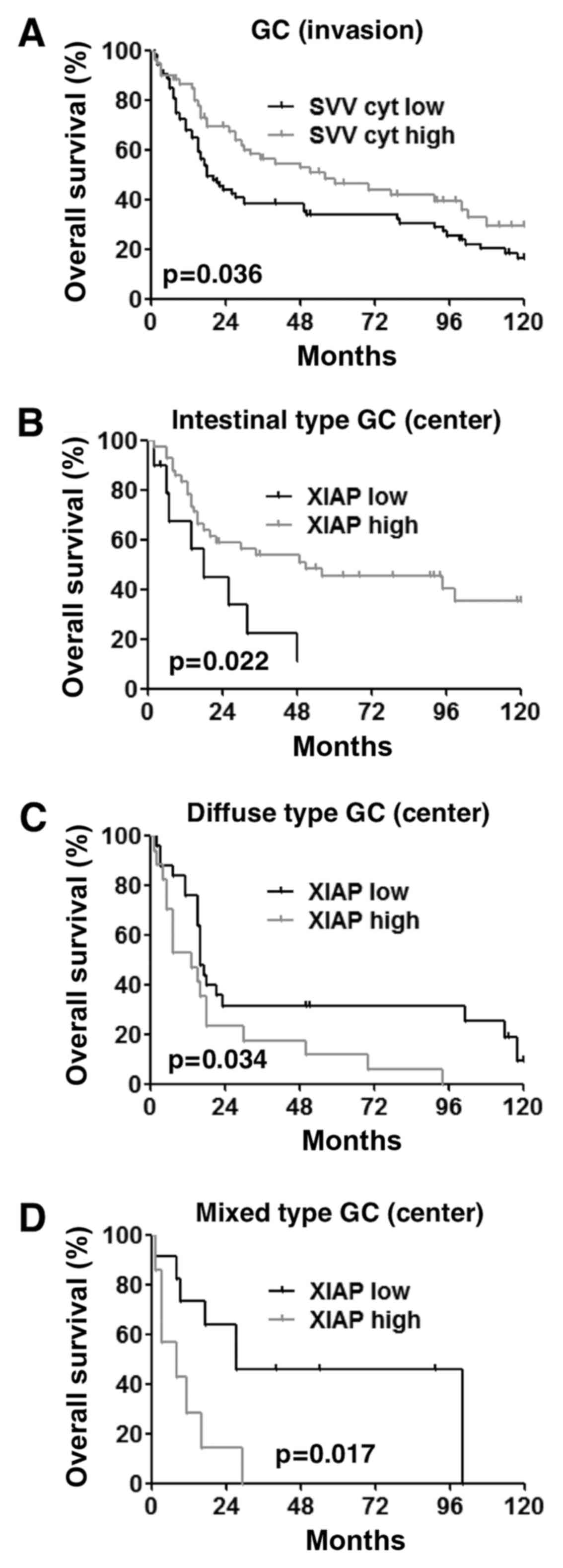
To explore the prognostic value of survivin and XIAP
in GC, we performed survival analysis including all GCs,
irrespective of their histological type. Accordingly, univariate
survival analysis revealed that high tumor stage, lymph node
metastasis, presence of distant metastasis, poor differentiation
(G3+4), advanced UICC stages and low cytoplasmic survivin
expression in the tumor invasion front were significantly
associated with a poor outcome (Table
V, Fig. 2A). However,
multivariate analysis determined only advanced UICC stages, G3/G4
differentiation and male gender as independent predictors of
survival in our study cohort (Table
VI).
 | Table V.Overall survival: Univariate
analysis. |
Table V.
Overall survival: Univariate
analysis.
|
| Gastric cancer | Intestinal
type | Diffuse type | Mixed type |
|---|
|
|
|
|
|
|
|---|
| Variable | HR | 95% CI | P-value | HR | 95% CI | P-value | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| Age | 1.308 | 0.89–1.93 | 0.172 | 1.305 | 0.70–2.45 | 0.406 | 2.067 | 1.14–3.74 | 0.014 | 0.726 | 0.29–1.81 | 0.491 |
| Sex | 1.196 | 0.78–1.83 | 0.409 | 1.194 | 0.57–2.52 | 0.640 | 1.265 | 0.69–2.33 | 0.450 | 1.432 | 0.51–4.01 | 0.492 |
| T 1/2 vs. T
3/4 | 3.024 | 2.02–4.53 |
<0.001 | 2.680 | 1.34–5.36 | 0.004 | 2.921 | 1.58–5.39 |
<0.001 | 3.855 | 1.43–10.4 | 0.005 |
| N0 vs. N+ | 2.066 | 1.32–3.23 | 0.001 | 3.184 | 1.50–6.75 | 0.001 | 1.244 | 0.65–2.38 | 0.508 | 2.059 | 0.67–6.34 | 0.199 |
| M0 vs. M1 | 3.723 | 1.71–8.17 |
<0.001 | 1.848 | 0.25–13.6 | 0.541 | 8.653 | 2.35–31.8 |
<0.001 | 2.927 | 0.82–10.5 | 0.084 |
| G 1/2 vs. G
3/4 | 2.150 | 1.29–3.58 | 0.003 | 2.310 | 1.22–4.39 | 0.009 |
|
|
|
|
|
|
| UICC I/II vs.
III/IV | 3.237 | 2.16–4.85 |
<0.001 | 3.789 | 1.98–7.26 |
<0.001 | 2.279 | 1.25–4.15 | 0.006 | 6.273 | 1.88–20.9 | 0.001 |
| XIAP center | 0.922 | 0.60–1.43 | 0.716 | 0.392 | 0.17–0.90 | 0.022 | 2.048 | 1.04–4.03 | 0.034 | 3.669 | 1.19–11.4 | 0.017 |
| XIAP invasion | 0.716 | 0.48–1.07 | 0.102 | 0.607 | 0.31–1.20 | 0.146 | 0.918 | 0.48–1.74 | 0.794 | 1.229 | 0.46–3.27 | 0.679 |
| SVV center
(cyt.) | 0.824 | 0.52–1.30 | 0.401 | 0.802 | 0.40–1.60 | 0.530 | 0.906 | 0.35–2.35 | 0.840 | 1.089 | 0.36–3.26 | 0.879 |
| SVV invasion
(cyt.) | 0.642 | 0.42–0.98 | 0.036 | 0.639 | 0.34–1.21 | 0.166 | 0.670 | 0.28–1.62 | 0.371 | 0.402 | 0.15–1.08 | 0.062 |
| SVV center
(nuc.) | 0.833 | 0.52–1.34 | 0.447 | 0.737 | 0.36–1.50 | 0.398 | 1.109 | 0.51–2.41 | 0.793 | 1.313 | 0.40–4.29 | 0.651 |
| SVV invasion
(nuc.) | 0.898 | 0.57–1.42 | 0.644 | 0.907 | 0.47–1.77 | 0.773 | 1.398 | 0.58–3.39 | 0.456 | 0.713 | 0.27–1.90 | 0.497 |
 | Table VI.Overall survival: Multivariate
analysis. |
Table VI.
Overall survival: Multivariate
analysis.
|
| Gastric cancer | Intestinal
type | Diffuse type | Mixed type |
|---|
|
|
|
|
|
|
|---|
| Variable | HR | 95% CI | P-value | HR | 95% CI | P-value | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| Age |
| / |
|
| / |
| 3.570 | 1.48–8.61 | 0.005 | 4.104 | 0.84–19.95 | 0.080 |
| Sex | 1.889 | 1.09–3.28 | 0.024 | 4.165 | 1.55–11.19 | 0.005 |
| / |
|
| / |
|
| G 1/2 vs. G
3/4 | 2.094 | 1.12–3.91 | 0.020 | 2.274 | 1.05–4.92 | 0.037 |
| / |
|
| / |
|
| UICC I/II vs.
III/IV | 3.061 | 1.85–5.08 |
<0.001 | 4.658 | 2.04–10.65 |
<0.001 | 5.756 | 2.24–14.80 |
<0.001 |
| / |
|
| XIAP center | 1.608 | 0.97–2.68 | 0.068 |
| / |
| 2.751 | 1.20–6.29 | 0.017 | 3.914 | 1.07–14.33 | 0.039 |
| XIAP invasion |
| / |
|
| / |
|
| / |
| 5.786 | 1.16–28.87 | 0.032 |
The observation that survivin and XIAP expression
are associated with different histologic types, prompted us to
perform a survival analysis for each histologic subtype. Thus,
univariate survival analysis of intestinal type GCs demonstrated
that high tumor stage, lymph node metastasis, poorly differentiated
tumors, advanced UICC stage and low XIAP expression levels in tumor
centers were related to worse overall survival (Table V, Fig.
2B). In contrast, univariate analysis of the diffuse type
revealed, that increased XIAP expression in tumor centers,
patient's age, T3/4 tumors and advanced UICC stages as well as
distant metastasis were significantly associated with a poor
prognosis (Table V, Fig. 2C). In mixed type GCs T3/4 stage, UICC
III/IV stages and high XIAP expression in tumor centers were of
prognostic relevance (Table V,
Fig. 2D). However, multivariate
analysis revealed UICC stage, grading and gender as independent
prognostic factors for intestinal type GCs (Table VI). Interestingly, high XIAP
expression in tumor centers was found to be an independent
prognostic factor in both diffuse type and mixed type GCs (Table VI). Other independent prognostic
factors for the diffuse and mixed type were UICC stage, patient's
age and overexpression of XIAP in the invasion front.
Discussion
In this study we explored survivin and XIAP
expression in a large number of surgically resected GC tissue
specimens. In contrast to previously published studies, we
specifically analyzed survivin and XIAP expression patterns in
distinct compartments (tumor center and invasion front) of the
primary tumor, lymph node metastases and non-neoplastic gastric
mucosa. This accurate protein expression analysis revealed
important results that have not been demonstrated elsewhere.
First and in accordance with previous reports
(24,34–36), we
found an increased survivin and XIAP expression in GC tissue when
compared to adjacent normal mucosa. This is noteworthy, as the
basis for targeted therapy is the identification of a
cancer-specific and druggable protein. The second observation, that
XIAP expression was significantly higher in lymph node metastasis
when compared to corresponding primary tumors supports the theory
that XIAP not only functions as inhibitor of apoptosis but also
plays a distinct role in metastatic tumor disease. This theory was
brought up by Mehrotra et al who could demonstrate that
intermolecular cooperation between XIAP and survivin promotes tumor
cell invasion and metastasis by an activation of cell motility
kinases FAK and Src (11). The
positive correlation between cytoplasmic survivin and XIAP
expression in GC tissue specimens is the third interesting finding
of our analysis. This observation might be explained by evidence
suggesting that mitochondrial survivin released into the cytosol
has the ability to associate with XIAP (37). This survivin-XIAP complex stabilizes
XIAP by protection against ubiquitin-dependent degradation
(11,16,19,38).
Our analysis demonstrated that expression of XIAP
and survivin was associated with intestinal type and
well-differentiated adenocarcinoma, as well as less advanced tumor
stages. In accordance with our findings Nakamura et al and
Zhu et al discovered not only increased survivin levels in
intestinal type GC but also in precancerous lesions. Consequently,
they concluded that survivin occurs as an early event in GC
development (39,40). In contrast, our recently published
meta-analysis investigating the relationship between survivin
expression levels and clinicopathological parameters reported a
significant correlation between increased survivin expression and
GC with lymph node metastasis (21).
However, our meta-analysis investigating the relationship between
survivin expression and lymph node metastases demonstrated a high
heterogeneity among the trials and included also studies reporting
increased survivin expression levels in lymph node-negative GCs
(22,34,41–43).
Consistent with our observation, two previously published studies
found also increased XIAP expression levels in intestinal type and
well-differentiated GC specimens (22,24).
However, in contrast to our results Kim et al (22) discovered increased XIAP expression
levels in advanced GC stages.
Although several studies have addressed the
relationship between high survivin levels and poor prognosis in GC
patients (25–27,35,39,44),
many conflicting findings exist (22,34,40,45).
Of note, our analysis of survivin expression revealed no
significant correlation between survivin expression and patients
overall survival. The observation that XIAP might serve as a
prognostic biomarker in GC was first described by Kim et al
(22). Importantly, our study refined
these observations by performing a detailed survival analysis with
respect to XIAP in the tumor center and invasion front of the
different histological subgroups. Accordingly, our data demonstrate
that in contrast to intestinal type GCs, high XIAP expression
serves as an independent negative prognostic marker for diffuse and
mixed type GCs.
We also observed a tendency towards higher XIAP
expression levels in tumors without lymph node or distant
metastasis. However, these differences were not statistically
significant, but indicate a potential role for XIAP in the early
phase of GC development.
Taken together, our survivin and XIAP expression
analysis in GC tissue specimens revealed that only XIAP might be
regarded as a prognostic marker in diffuse and mixed type GCs.
Thus, our results might suggest that XIAP could be one of the key
molecular targets especially in the subtype of diffuse GC which is
more commonly associated with a very poor prognosis. However, both
IAPs represent promising candidates for targeted therapy
approaches, as they were significantly overexpressed in the vast
majority of all GC specimens investigated. Different strategies to
target IAP proteins for cancer therapy have been investigated thus
far and, among them, small-molecule IAP antagonists and antisense
oligonucleotides have reached the most attention (14,46,47).
Currently, the most intensively investigated survivin antagonist is
the small imidazolium-based compound YM155 (Sepantronium bromide).
This small molecule survivin inhibitor demonstrated impressive
anticancer activity in several preclinical studies using cancer
cell lines originating from various types of cancer, including GC
cells (30,48–50) and
was well tolerated in phase I studies (51,52).
Concerning the potential efficacy of anti-XIAP treatment strategies
in GC initial promising results have been published. Wang et
al demonstrated that the XIAP inhibitor Embelin significantly
reduced GC cell viability, induced apoptosis, and enhanced 5-FU
antitumor activity in vitro (53). Furthermore, Tong et al
established that downregulation of XIAP via antisense RNA can
induce apoptosis and enhance chemotherapeutic agent sensitivity in
GC cells (54). Given these promising
findings future studies should address the question whether or not
survivin and XIAP directed therapy approaches are able to improve
the survival of GC patients.
However, our study might have some limitations. One
limitation is the relatively short follow up period and the design
as a single-institution retrospective analysis. In addition, we
detected survivin by using a polyclonal antibody that might also
bind to the different alternative splice variants of survivin. Of
note, in previous studies using monoclonal as well as
aforementioned polyclonal antibodies, we failed to detect survivin
splice variants on protein levels (55). Importantly, the specificity of the
antibody that we used in our study was also confirmed in knock down
experiments that we recently performed (30).
Despite these limitations, we are the first to
demonstrate that high XIAP expression is a negative prognostic
factor exclusively in the subgroup of diffuse and mixed type GC.
These results are important since they provide a rationale for
future studies investigating the therapeutic efficacy of anti-XIAP
treatment strategies, especially in patients with diffuse and mixed
type GCs.
References
|
1
|
Ferlay J, Soerjomataram I, Ervik M,
Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D and
Bray F: GLOBOCAN 2012 v1.0, Cancer Incidence and Mortality
Worldwide: IARC CancerBase No. 11 (Internet)International Agency
for Research on Cancer. Lyon: 2013 http://globocan.iarc.frAccessed. December
28–2016
|
|
2
|
Howlader N NA, Krapcho M, Miller D, Bishop
K, Altekruse SF, Kosary CL, Yu M, Ruhl J, Tatalovich Z, Mariotto A,
et al: SEER Cancer Statistics Review, 1975–2013National Cancer
Institute. Bethesda, MD: 2016 http://seercancergov/csr/1975_2013/Accessed.
December 28–2016
|
|
3
|
D'Angelica M, Gonen M, Brennan MF,
Turnbull AD, Bains M and Karpeh MS: Patterns of initial recurrence
in completely resected gastric adenocarcinoma. Ann Surg.
240:808–816. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Okines AF, Ashley SE, Cunningham D, Oates
J, Turner A, Webb J, Saffery C, Chua YJ and Chau I: Epirubicin,
oxaliplatin, and capecitabine with or without panitumumab for
advanced esophagogastric cancer: Dose-finding study for the
prospective multicenter, randomized, phase II/III REAL-3 trial. J
Clin Oncol. 28:3945–3950. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Cunningham D, Okines AF and Ashley S:
Capecitabine and oxaliplatin for advanced esophagogastric cancer. N
Engl J Med. 362:858–859. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Roth AD, Fazio N, Stupp R, Falk S,
Bernhard J, Saletti P, Köberle D, Borner MM, Rufibach K, Maibach R,
et al: Docetaxel, cisplatin and fluorouracil; docetaxel and
cisplatin; and epirubicin, cisplatin and fluorouracil as systemic
treatment for advanced gastric carcinoma: A randomized phase II
trial of the Swiss Group for Clinical Cancer Research. J Clin
Oncol. 25:3217–3223. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Giuliani F, Molica S, Maiello E, Battaglia
C, Gebbia V, Di Bisceglie M, Vinciarelli G, Gebbia N and Colucci G:
Gruppo Oncologico dell' Italia Meridionale (prot. 2106): Irinotecan
(CPT-11) and mitomycin-C (MMC) as second-line therapy in advanced
gastric cancer: A phase II study of the Gruppo Oncologico dell'
Italia Meridionale (prot. 2106). Am J Clin Oncol. 28:581–585. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wagner AD and Moehler M: Development of
targeted therapies in advanced gastric cancer: Promising
exploratory steps in a new era. Curr Opin Oncol. 21:381–385. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Mita AC, Mita MM, Nawrocki ST and Giles
FJ: Survivin: Key regulator of mitosis and apoptosis and novel
target for cancer therapeutics. Clin Cancer Res. 14:5000–5005.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Srinivasula SM and Ashwell JD: IAPs:
What's in a name? Mol Cell. 30:123–135. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mehrotra S, Languino LR, Raskett CM,
Mercurio AM, Dohi T and Altieri DC: IAP regulation of metastasis.
Cancer Cell. 17:53–64. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bertrand MJ, Milutinovic S, Dickson KM, Ho
WC, Boudreault A, Durkin J, Gillard JW, Jaquith JB, Morris SJ and
Barker PA: cIAP1 and cIAP2 facilitate cancer cell survival by
functioning as E3 ligases that promote RIP1 ubiquitination. Mol
Cell. 30:689–700. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Altieri DC: New wirings in the survivin
networks. Oncogene. 27:6276–6284. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Fulda S and Vucic D: Targeting IAP
proteins for therapeutic intervention in cancer. Nat Rev Drug
Discov. 11:109–124. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
15
|
Altieri DC: Survivin-The inconvenient IAP.
Semin Cell Dev Biol. 39:91–96. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Dohi T, Beltrami E, Wall NR, Plescia J and
Altieri DC: Mitochondrial survivin inhibits apoptosis and promotes
tumorigenesis. J Clin Invest. 114:1117–1127. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Krieg A, Correa RG, Garrison JB, Le
Negrate G, Welsh K, Huang Z, Knoefel WT and Reed JC: XIAP mediates
NOD signaling via interaction with RIP2. Proc Natl Acad Sci USA.
106:14524–14529. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Krieg A and Reed JC: IAPs and their
emergent role in NLR signaling. Cell Cycle. 9:426–427. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Dohi T, Okada K, Xia F, Wilford CE, Samuel
T, Welsh K, Marusawa H, Zou H, Armstrong R, Matsuzawa S, et al: An
IAP-IAP complex inhibits apoptosis. J Biol Chem. 279:34087–34090.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lu M, Lin SC, Huang Y, Kang YJ, Rich R, Lo
YC, Myszka D, Han J and Wu H: XIAP induces NF-kappaB activation via
the BIR1/TAB1 interaction and BIR1 dimerization. Mol Cell.
26:689–702. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Krieg A, Baseras B, Tomczak M, Verde PE,
Stoecklein NH and Knoefel WT: Role of survivin as prognostic and
clinicopathological marker in gastric cancer: A meta-analysis. Mol
Biol Rep. 40:5501–5511. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kim MA, Lee HE, Lee HS, Yang HK and Kim
WH: Expression of apoptosis-related proteins and its clinical
implication in surgically resected gastric carcinoma. Virchows
Arch. 459:503–510. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Shibata T, Mahotka C, Wethkamp N, Heikaus
S, Gabbert HE and Ramp U: Disturbed expression of the apoptosis
regulators XIAP, XAF1, and Smac/DIABLO in gastric adenocarcinomas.
Diagn Mol Pathol. 16:1–8. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Shibata T, Noguchi T, Takeno S, Gabbert
HE, Ramp U and Kawahara K: Disturbed XIAP and XAF1 expression
balance is an independent prognostic factor in gastric
adenocarcinomas. Ann Surg Oncol. 15:3579–3587. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lee GH, Joo YE, Koh YS, Chung IJ, Park YK,
Lee JH, Kim HS, Choi SK, Rew JS, Park CS and Kim SJ: Expression of
survivin in gastric cancer and its relationship with tumor
angiogenesis. Eur J Gastroenterol Hepatol. 18:957–963. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Song KY, Jung CK, Park WS and Park CH:
Expression of the antiapoptosis gene Survivin predicts poor
prognosis of stage III gastric adenocarcinoma. Jpn J Clin Oncol.
39:290–296. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Meng JR, Tang HZ, Zhou KZ, Shen WH and Guo
HY: TFF3 and survivin expressions associate with a lower survival
rate in gastric cancer. Clin Exp Med. 13:297–303. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
McShane LM, Altman DG, Sauerbrei W, Taube
SE, Gion M and Clark GM: Statistics Subcommittee of the NCI-EORTC
Working Group on Cancer Diagnostics: REporting recommendations for
tumour MARKer prognostic studies (REMARK). Br J Cancer. 93:387–391.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
International Union Against Cancer: TNM
classification of malignant tumours. Sobin LH and GM WC: 7th.
Wiley-Liss; New York, NY: 2009
|
|
30
|
Dizdar L, Oesterwind KA, Riemer JC, Werner
TA, Mersch S, Möhlendick B, Schütte SC, Verde PE, Raba K, Topp SA,
et al: Preclinical assesement of survivin and XIAP as prognostic
biomarkers and therapeutic targets in gastroenteropancreatic
neuroendocrine neoplasia. Oncotarget. 8:8369–8382. 2017.PubMed/NCBI
|
|
31
|
Remmele W and Stegner HE: Recommendation
for uniform definition of an immunoreactive score (IRS) for
immunohistochemical estrogen receptor detection (ER-ICA) in breast
cancer tissue. Pathologe. 8:138–140. 1987.(In German). PubMed/NCBI
|
|
32
|
Werner TA, Tamkan-Ölcek Y, Dizdar L,
Riemer JC, Wolf A, Cupisti K, Verde PE, Knoefel WT and Krieg A:
Survivin and XIAP: Two valuable biomarkers in medullary thyroid
carcinoma. Br J Cancer. 114:427–434. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Burnham KP, Anderson DR and Burnham KP:
Model Selection And Multimodel Inference: A Practical
Information-Theoretic Approach. 2nd. Springer; New York, NY:
2002
|
|
34
|
Lu CD, Altieri DC and Tanigawa N:
Expression of a novel antiapoptosis gene, survivin, correlated with
tumor cell apoptosis and p53 accumulation in gastric carcinomas.
Cancer Res. 58:1808–1812. 1998.PubMed/NCBI
|
|
35
|
Meng H, Lu C, Mabuchi H and Tanigawa N:
Prognostic significance and different properties of survivin
splicing variants in gastric cancer. Cancer Lett. 216:147–155.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Da CL, Xin Y, Zhao J and Luo XD:
Significance and relationship between Yes-associated protein and
survivin expression in gastric carcinoma and precancerous lesions.
World J Gastroenterol. 15:4055–4061. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Dohi T, Xia F and Altieri DC:
Compartmentalized phosphorylation of IAP by protein kinase A
regulates cytoprotection. Mol Cell. 27:17–28. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Arora V, Cheung HH, Plenchette S, Micali
OC, Liston P and Korneluk RG: Degradation of survivin by the
X-linked inhibitor of apoptosis (XIAP)-XAF1 complex. J Biol Chem.
282:26202–26209. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Nakamura M, Tsuji N, Asanuma K, Kobayashi
D, Yagihashi A, Hirata K, Torigoe T, Sato N and Watanabe N:
Survivin as a predictor of cis-diamminedichloroplatinum sensitivity
in gastric cancer patients. Cancer Sci. 95:44–51. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Zhu XD, Lin GJ, Qian LP and Chen ZQ:
Expression of survivin in human gastric carcinoma and gastric
carcinoma model of rats. World J Gastroenterol. 9:1435–1438. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Deng H, Wu RL, Zhou HY, Huang X, Chen Y
and Liu LJ: Significance of Survivin and PTEN expression in full
lymph node-examined gastric cancer. World J Gastroenterol.
12:1013–1017. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Okayama H, Kumamoto K, Saitou K, Hayase S,
Kofunato Y, Sato Y, Miyamoto K, Nakamura I, Ohki S, Sekikawa K and
Takenoshita S: CD44v6, MMP-7 and nuclear Cdx2 are significant
biomarkers for prediction of lymph node metastasis in primary
gastric cancer. Oncol Rep. 22:745–755. 2009.PubMed/NCBI
|
|
43
|
Tsuburaya A, Noguchi Y, Yoshikawa T, Saito
A, Doi C, Okamoto T and Fukuzawa K: An anti-apoptosis gene,
survivin and telomerase expression in gastric cancer.
Hepatogastroenterology. 49:1150–1152. 2002.PubMed/NCBI
|
|
44
|
Bury J, Szumiło J, Dąbrowski A, Ciechański
A, Śliwińska J and Wallner G: Vascular endothelial growth factor
and survivin immunostaining in gastric adenocarcinoma. Pol Przegl
Chir. 84:341–347. 2012.PubMed/NCBI
|
|
45
|
Okada E, Murai Y, Matsui K, Isizawa S,
Cheng C, Masuda M and Takano Y: Survivin expression in tumor cell
nuclei is predictive of a favorable prognosis in gastric cancer
patients. Cancer Lett. 163:109–116. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Ndubaku C, Cohen F, Varfolomeev E and
Vucic D: Targeting inhibitor of apoptosis proteins for therapeutic
intervention. Future Med Chem. 1:1509–1525. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
LaCasse EC, Mahoney DJ, Cheung HH,
Plenchette S, Baird S and Korneluk RG: IAP-targeted therapies for
cancer. Oncogene. 27:6252–6275. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Nakahara T, Kita A, Yamanaka K, Mori M,
Amino N, Takeuchi M, Tominaga F, Hatakeyama S, Kinoyama I,
Matsuhisa A, et al: YM155, a novel small-molecule survivin
suppressant, induces regression of established human
hormone-refractory prostate tumor xenografts. Cancer Res.
67:8014–8021. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Cheng XJ, Lin JC, Ding YF, Zhu L, Ye J and
Tu SP: Survivin inhibitor YM155 suppresses gastric cancer xenograft
growth in mice without affecting normal tissues. Oncotarget.
7:7096–7109. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Ghadimi MP, Young ED, Belousov R, Zhang Y,
Lopez G, Lusby K, Kivlin C, Demicco EG, Creighton CJ, Lazar AJ, et
al: Survivin is a viable target for the treatment of malignant
peripheral nerve sheath tumors. Clin Cancer Res. 18:2545–2557.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Satoh T, Okamoto I, Miyazaki M, Morinaga
R, Tsuya A, Hasegawa Y, Terashima M, Ueda S, Fukuoka M, Ariyoshi Y,
et al: Phase I study of YM155, a novel survivin suppressant, in
patients with advanced solid tumors. Clin Cancer Res. 15:3872–3880.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Tolcher AW, Mita A, Lewis LD, Garrett CR,
Till E, Daud AI, Patnaik A, Papadopoulos K, Takimoto C, Bartels P,
et al: Phase I and pharmacokinetic study of YM155, a small-molecule
inhibitor of survivin. J Clin Oncol. 26:5198–5203. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Wang DG, Sun YB, Ye F, Li W, Kharbuja P,
Gao L, Zhang DY and Suo J: Anti-tumor activity of the X-linked
inhibitor of apoptosis (XIAP) inhibitor embelin in gastric cancer
cells. Mol Cell Biochem. 386:143–152. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Tong QS, Zheng LD, Wang L, Zeng FQ, Chen
FM, Dong JH and Lu GC: Downregulation of XIAP expression induces
apoptosis and enhances chemotherapeutic sensitivity in human
gastric cancer cells. Cancer Gene Ther. 12:509–514. 2005.PubMed/NCBI
|
|
55
|
Mahotka C, Krieg T, Krieg A, Wenzel M,
Suschek CV, Heydthausen M, Gabbert HE and Gerharz CD: Distinct in
vivo expression patterns of survivin splice variants in renal cell
carcinomas. Int J Cancer. 100:30–36. 2002. View Article : Google Scholar : PubMed/NCBI
|