Introduction
Colon cancer is one of the most common malignant
tumors worldwide (1). In Europe,
America and other western countries, colon cancer has one of the
highest cancer mortality rates. In 1982, the incidence of colon
cancer in China was (2). However,
with a deterioration in lifestyle and eating habits, including high
fat, high animal protein, high energy intake and lack of fiber
(2), the incidence of colon cancer
has increased year by year and has reached fifth place in rankings
behind lung, liver, stomach and esophageal cancer (1).
Recurrence and metastasis are the primary causes of
mortality in patients with colon cancer. The most common ways for
tumors to metastasize are via the blood and/or lymph nodes,
additionally, angiogenesis is essential for tumor growth and
metastasis (3). Vascular endothelial
growth factor (VEGF) is considered to be one of the most important
angiogenic factors, which can promote the formation of new blood
vessels and lymphatic vessels (4).
VEGF has a number of different physiological functions. For
example, VEGF is able to selectively increase the mitosis of
endothelial cells, and promote proliferation of endothelia, l cells
and angiogenesis. VEGF is also able to strengthen blood vessels by
increasing capillary permeability and therefore promotes the
extravasation of plasma proteins and other macromolecules.
Furthermore, it increases deposition to the extravascular matrix
and supplies nutrition for the establishment of new capillary
networks (5).
Therefore, VEGF is one of the most important growth
factors for angiogenesis, which has a key role in the formation of
blood vessels (6). When VEGF binds
with the VEGF receptor (VEGF-R), the tyrosine protein kinase
pathway is activated, and the corresponding signal transduction
pathways including P44/P42-mitogen-activated protein kinase and
phosphoinositide 3-kinase/protein kinase B are subsequently
activated (7,8). The tyrosine protein kinase pathway is
involved in the normal regulation of cell proliferation and
differentiation amongst other important physiological
processes.
A previous study has reported that VEGF was highly
expressed in numerous malignant tumor tissues including colon and
esophageal cancer, and VEGF expression was closely associated with
tumor metastasis (9,10). Hypoxia-inducible factor 1 (HIF-1) is
an oxygen dependent transcriptional activator, which was discovered
by Ratcliffe et al (11) in
1992. HIF-1 is composed of HIF-1α and HIF-1β subunits. HIF-1α is
the only oxygen regulated subunit, which is mainly expressed in
cells under hypoxic conditions (12).
Hypoxia can increase the accumulation and activity of HIF-1α.
Activated HIF-1α regulates the transcription and expression of
downstream genes, and then stimulates tumor angiogenesis, inhibits
the apoptosis of tumor cells and promotes metastasis (13,14). These
changes enable tumor cells to adapt to hypoxic microenvironment.
The tumor cells can proliferate and invade, and transcription can
take place continuously (13,14). VEGF is one of the target genes of
HIF-1α. HIF-1α promotes the transcription of target gene VEGF, and
regulates angiogenesis in tumors under hypoxic conditions (15,16).
Therefore, HIF-1α may reduce the possibility of recurrence and
metastasis of tumor cells if the formation of new blood vessels in
tumor tissue can be suppressed.
Thalidomide (THD), synthesized by German Chemie
Gruneenthal in 1954, was used in Europe as a sedative treatment for
morning sickness in pregnancy. However, the use of THD during
pregnancy led to phocomelia and consequently the drug was withdrawn
from the market (17). Subsequent
studies have reported that THD has anti-inflammatory,
immunomodulatory and anti-angiogenic effects (18–22). Due
to its anti angiogenic effects, THD is able to affect fetal
development in pregnant women, which leads to fetal deformity.
However, at the same time this effect of THD is one of the
mechanisms for its antitumor activity (18,23). THD
inhibits angiogenesis, stimulates the activity of the immune system
and inhibits the adherence of cancer cells to stromal cells, which
suppresses the occurrence and development of tumors.
There may be several mechanisms of action for THD in
the treatment of malignant tumors (24). As THD is able to mediate
anti-angiogenesis effects through inhibiting the expression of VEGF
and basic fibroblast growth factor (bFGF), a previous study
demonstrated that 150 µg/ml THD was able to reduce tumor blood
supply and inhibit tumor growth (23). THD is also able to can stimulate the
proliferation of natural killer cells and induce lymphocytes to
secrete interferon γ (IFN-γ) and interleukin 2 (IL-2), so as to
kill tumor cells (25). Studies have
also demonstrated that THD may induce tumor cell apoptosis and
arrest cell growth at the G1 phase (26–28).
Additionally, THD is able to inhibit the binding of NF-κB to DNA
and therefore exhibits antitumor effects by blocking
transcriptional activity (9). THD is
also able to downregulate the expression of adhesion molecules in
vascular endothelial cells and therefore inhibit tumor
proliferation (29).
Oxaliplatin (L-OHP) is a platinum anticancer drug
with a broad spectrum of antitumor effects and is used to treat
metastatic colorectal cancer, particularly as an adjuvant treatment
for patients with stage III colon cancer with complete resections
of primary tumor (30). However, if
L-OHP has been used for an extended period time, incidences of
resistance to the drug can increase (31). Furthermore, increasing the dose of
L-OHP results in increased adverse reactions.
Previous studies have indicated that THD, when used
with other chemotherapeutics may result in synergistic antitumor
effects (32,33). However, to the best of our knowledge,
the anti angiogenic effects of THD on SW480 cells has not been
reported. In the present study, SW480 cells were cultured and
treated with various concentrations of THD (12.5, 25, 50, 100 and
200 µg/ml). Optical density values were evaluated by MTT assay, and
inhibition ratio was calculated for all treatment groups. The mRNA
expression of VEGF-A in the treated cells was detected by
quantitative polymerase chain reaction (qPCR), and the levels of
HIF-1α protein were detected by western blotting. The potential
mechanisms of THD on SW480 cells were investigated. Furthermore,
the effect of treatment with a combination of THD and L-OHP on
SW480 cells was analyzed. The results of the present study may
provide experimental basis for future clinical treatment of colon
cancer.
Materials and methods
Cell culture
The SW480 cell line and the HeLa cell lines were
purchased from the Cell Bank of Chinese Academy of Sciences
(Shanghai, China), and cultured in RPMI-1640 (Gibco; Thermo Fisher
Scientific, Inc., Waltham, MA, USA) containing 10% fetal calf serum
(Sijiqing company, Hangzhou, China). SW480 cells and HeLa cells
were cultured in an incubator containing 5% CO2 at
37°C.
Cell treatment
THD (catalog no. 14080431; Changzhou Pharmaceutical
Factory, Changzhou, China) and L-OHP (catalog no. 14091515; Jiangsu
Hengrui Pharmaceutical Factory, Jiangsu, China) were used. The
SW480 cells were treated with 12.5, 25, 50, 100 and 200 µg/ml THD
for 24, 48 or 72 h and 4, 8, 16, 32, 64 and 128 mg/l L-OHP + 5%
glucose for 48 h prior to subsequent assays. The final volume was
200 µl, and the THD control group was treated with DMSO only. The
L-OHP control group was treated with 5% glucose. SW480 cell
proliferation was assayed subsequent to after THD/L-OHP treatment.
Treatment was performed at 37°C. Cell proliferation following THD
treatment and half maximal inhibitory concentration
(IC50) values following L-OHP treatment were determined.
SW480 cells were then treated with 25, 50, 100 µg/ml THD and 64
mg/l L-OHP and 50 µg/ml THD + 64 mg/l L-OHP for 48 h at 37°C. The
control group for mRNA/VEGF detection was SW480 cells that had not
been previously treated with any drugs, and the control group and
positive group for HIF-1α detection were SW480 and HeLa cells,
respectively. Neither group had been previously treated with any
drugs. mRNA, VEGF and the expression levels of HIF-1α were detected
following culture for 48 h.
Cell proliferation assay
Cells were treated as aforementioned. Experiments at
each concentration were repeated 5 times. The MTT
spectrophotometric dye assay (Beijing Dingguo Changsheng
Biotechnology Co., Ltd., Beijing, China) was used to detect cell
proliferation ability following THD treatment and determine
IC50 values following L-OHP treatment. SW480 cells were
seeded in 96-well plates at a density of 2,500 cells/well. At 24,
48 and 72 h post-treatment, cells were incubated with 20 µl MTT for
4 h. Color was developed by incubating the cells with 150 µl DMSO;
and the absorbance was detected at a wavelength of 490 nm using
Microplate Reader (Elx800; BioTek Instruments, Inc., Winooski, VT,
USA).
RNA extraction
The SW480 cells were treated as aforementioned. RNA
extraction from the cells was undertaken using Trizol according to
the manufacturer's protocols (Invitrogen; Thermo Fisher Scientific,
Inc.). RNA was eluted in RNase-free water. The RNA concentration
was determined with the NanoDrop 2000/2000C spectrophotometer (cat.
no. 2000/2000C; NanoDrop Technologies; Thermo Fisher Scientific,
Inc.).
Reverse transcription
To obtain complementary DNA (cDNA), 1 µl Oligo dT
(0.5 µg/µl) and 2.0 µg Total RNA were used according to the
instructions for Promega M-MLV kit (Promega Corporation, Madison,
WI, USA). Following incubation at 70°C for 10 min, the mixture was
immediately placed in ice bath for 2 min. M-MLV RT 5X reaction
buffer, M-MLV Reverse Transcriptase RNase Minus, and 10 mM dNTP
(Promega Corporation) were added to the mixture and then incubated
at 42°C for 1 h, followed by 70°C for 10 min. cDNA was stored at
−80°C.
qPCR
The total RNA was extracted from the SW480 cells and
reverse transcribed to template cDNA. RNA extraction from cells was
performed according to the manufacturers protocol (Invitrogen;
Thermo Fisher Scientific, Inc.). RNA was eluted in RNase-free
water. RNA concentrations were determined using the NanoDrop
2000/2000C spectrophotometer (NanoDrop Technologies; Thermo Fisher
Scientific, Inc.). To obtain cDNA (according to the Promega M-MLV
protocol), 1 µl Oligo dT (0.5 µg/µl; Axygen Scientific Inc., Union
City, CA, USA) and 2.0 µg total RNA were used. Following incubation
at 70°C for 10 min, the mixture was placed in an ice bath
immediately for 2 min. M-MLV RT 5× reaction buffers, M-MLV Reverse
Transcriptase RNase Minus and 10 mM dNTP (Promega Corporation) were
added to the mixture and then incubated at 42°C for 1 h, followed
by 70°C for 10 min. cDNA was stored at −80°C. qPCR was performed on
the Takara PCR thermal cycler using the SYBR Green detection system
(Takara Bio, Inc., Otsu, Japan). Cycling conditions consisted of a
30 sec hot start at 95°C, followed by 45 cycles of denaturation at
95°C for 5 sec, annealing at 60°C for 30 sec, extension at 95°C for
15 sec, 55°C for 30 sec, and then a final inactivation at 95°C for
15 sec. Dissociation curve analyses were performed at the end of
the cycle to confirm that one specific product was measured in each
reaction. This was repeated 3 times. Relative quantification was
performed using the 2−ΔΔCq method (34). The specific primers for each gene are
as follows: VEGF-A (product size, 89 bp) forward
5′-GCTTACTCTCACCTGCTTCTG-3′, reverse 5′-GGCTGCTTCTTCCAACAATG-3′ and
GAPDH (product size, 121 bp) forward 5′-TGACTTCAACAGCGACACCCA-3′
and reverse 5′-CACCCTGTTGCTGTAGCCAAA-3′.
Protein extraction and western blot
analysis
Hela cells and SW480 cells, which had not been
previously treated with any drugs, were used as the positive and
blank controls, respectively. The SW480 cells were treated as
aforementioned. The cells were washed with PBS and lysed in
radioimmunoprecipitation assay buffer (Beyotime Institute of
Biotechnology, Jiangsu, China). The cells were lysed with lysis
buffer and centrifuged at 12,000 × g for 15 min at 4°C. Protein (20
µg) was subjected to 10% SDS-PAGE separation. The proteins were
transferred to PVDF membrane (EMD Millipore, Billerica, MA, USA),
blocked at 4°C overnight using TBST solution containing 5% skimmed
milk. Blots were probed with HIF-1α antibody (mouse anti human;
1:500; catalog no. ab113642; Abcam, Cambridge, MA, USA). All
primary antibodies were incubated 4°C overnight, and washed four
times with TBST, every time 8 min, and then incubated with the
secondary monoclonal antibody anti mouse immunoglobulin G (1:2,000;
catalog no. sc-2005; Santa Cruz Biotechnology, Inc., Dallas, TX,
USA). Blots were visualized using enhanced chemiluminescence-PLUS
kit (Thermo Fisher Scientific, Inc.).
Data analysis
Statistical analysis was performed using SPSS
(version 13.0; SPSS, Inc., Chicago, IL, USA). All quantitative data
are expressed as the mean ± standard deviation. One-way analysis of
variance and Student-Newman-Keuls method were used for analysis.
Student-Newman-Keuls analysis was used to analyze significant
differences between groups, and P<0.05 was considered to
indicate a statistically significant difference.
Results
Effect of THD treatment on SW480
cells
The SW480 cells were treated with different
concentrations of THD (12.5, 25, 50, 100 and 200 µg/ml) for 24, 48
and 72 h. Each concentration of THD was able to inhibit the
proliferation of SW480 cells compared with the control. The growth
inhibition rate of SW480 cells increased significantly (P<0.05)
when the concentration of THD and duration of treatment increased
(Table I).
 | Table I.Effect of THD treatment on human
colon cancer SW480 cells. |
Table I.
Effect of THD treatment on human
colon cancer SW480 cells.
|
| Duration of
treatment, h |
|---|
|
|
|
|---|
| Groups | 24 | 48 | 72 |
|---|
| Control (0
µg/ml) |
0.00±0.00 |
0.00±0.00 |
0.00±0.00 |
| THD (µg/ml) |
|
|
|
|
12.5 |
24.23±1.5 |
27.5±1.37b |
33.24±1.39b |
|
25.0 |
27.73±2.05a |
31.54±1.54a,b |
35.97±1.14a,b |
|
50.0 |
31.54±1.93a |
35.12±2.18a,b |
38.35±1.38a,b |
|
100.0 |
35.24±1.4a |
37.76±1.27a,b |
40.93±0.95a,b |
|
200.0 |
37.07±0.68a |
42.11±2.11a,b |
45.13±0.97a,b |
Determination IC50 of L-OHP
using the MTT method
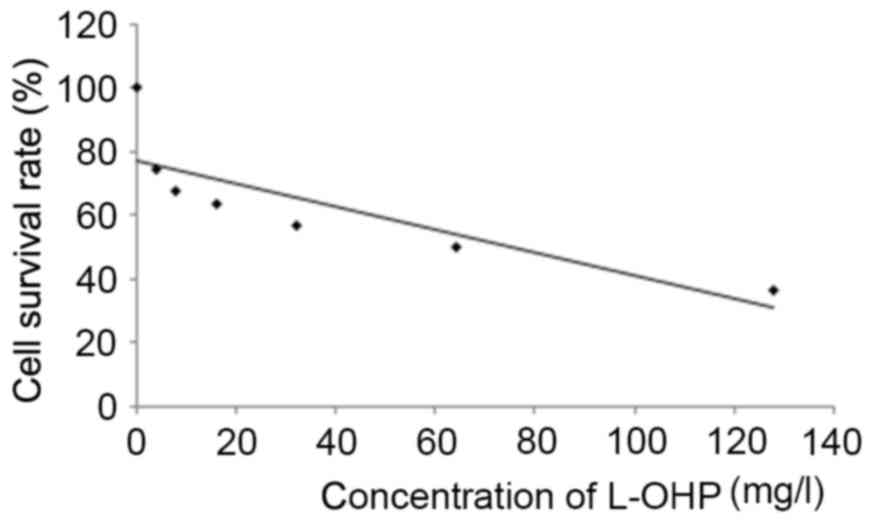
According to Fig. 1,
the IC50 value of L-OHP was 64 mg/l.
mRNA expression of VEGF-A in SW480
cells following treatment with various concentrations of THD, L-OHP
and THD + L-OHP
The qPCR results indicated that VEGF-A mRNA
expression was markedly decreased when the concentration of THD was
increased (Table II). The
2−ΔΔq values for 25, 50 and 100 µg/ml THD treatment
groups were 0.809±0.013, 0.685±0.035 and 0.563±0.034, respectively.
The 2−ΔΔq value for the control group was 1.002±0.074.
The differences between any two groups was significant
(P<0.05).
 | Table II.2−ΔΔq values of VEGF-A
mRNA expression following THD treatment. |
Table II.
2−ΔΔq values of VEGF-A
mRNA expression following THD treatment.
| Control group | 25 µg/ml THD | 50 µg/ml THD | 100 µg/ml THD |
|---|
| 1.002±0.074 |
0.809±0.013a |
0.685±0.035a |
0.563±0.034a |
VEGF-A mRNA expression was also decreased following
the treatment of SW480 cells with 50 µg/ml THD, 64 mg/l L-OHP and
50 µg/ml THD + 64 mg/l L-OHP for 48 h (Table III). The 2−ΔΔq values for
50 µg/ml THD, 64 mg/l L-OHP and 50 µg/ml THD + 64 mg/l L-OHP
treatment groups were 0.685±0.035, 0.281±0.018, 0.167±0.007,
respectively. The differences between the groups were significant
P<0.05. The treatment of SW480 cells with a combination of THD
and L-OHP had a synergistically decreased VEGF-A mRNA
expression.
 | Table III.2−ΔΔq values of VEGF-A
mRNA expression following THD and L-OHP treatment. |
Table III.
2−ΔΔq values of VEGF-A
mRNA expression following THD and L-OHP treatment.
| Control group | 50 µg/ml THD | 64 mg/l L-OHP | 50 µg/ml THD+ 64
mg/l L-OHP |
|---|
| 1.002±0.074 |
0.685±0.035a |
0.281±0.018a |
0.167±0.007a |
Expression of HIF-1α protein
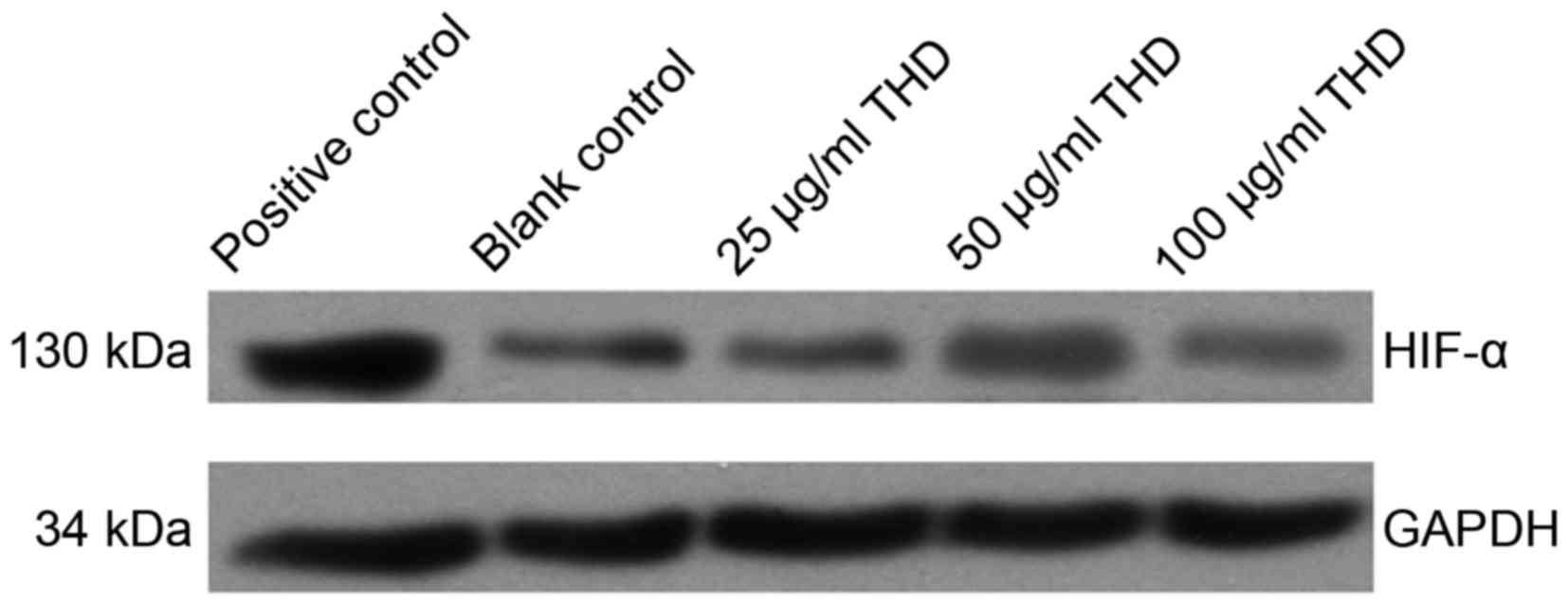
HIF-1α expression was decreased following treatment
of cells with THD. In addition, there was not a significant change
in expression levels as the concentration of THD increased
(Fig. 2). Furthermore, HIF-1α
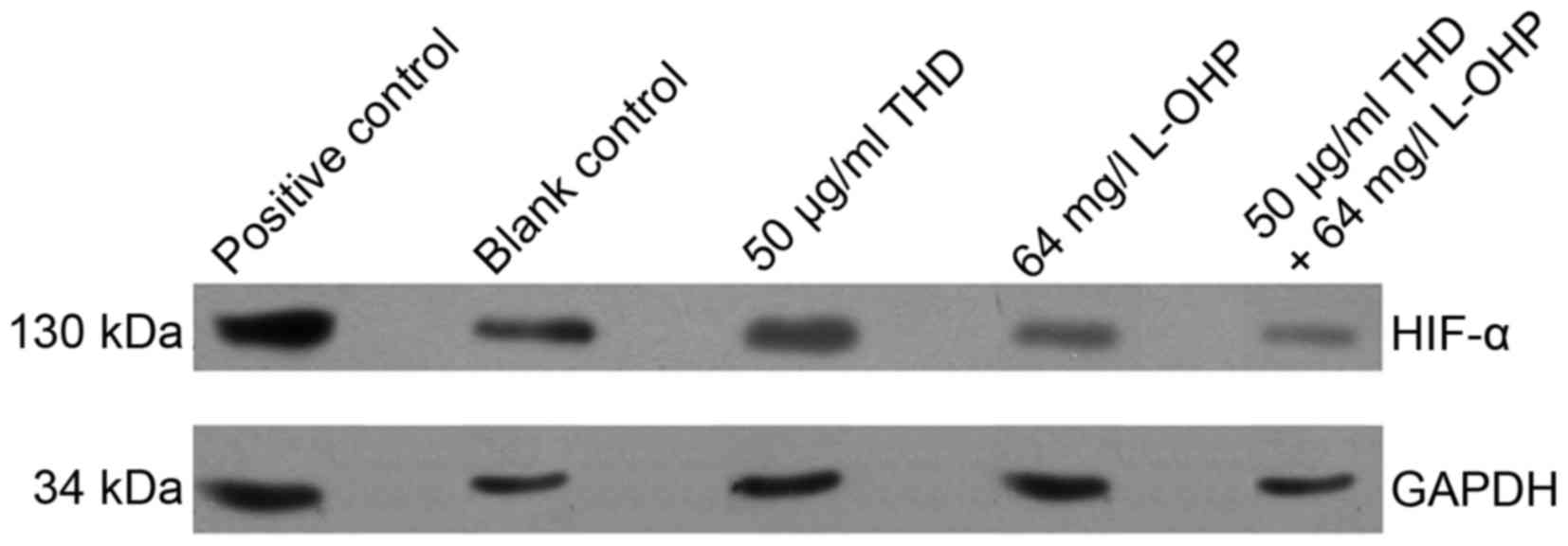
expression was decreased following the treatment of SW480 cells
with 50 µg/ml THD, 64 mg/l L-OHP and 50 µg/ml THD + 64 mg/l OHP for
48 h (Fig. 3). Results indicated that
the treatment of cells with a combination of THD and L-OHP had a
synergistic effect.
Discussion
Colorectal cancer is one of the most common
malignant tumors worldwide. Colorectal cancer has one of the
highest rates of cancer mortality in Europe and other western
countries (1). In 1971, Folkman
(35) reported that angiogenesis has
an important role in the process of tumor growth. Angiogenesis is
necessary for tumor growth and metastasis (3). Subsequently, the study of angiogenesis
attracted extra attention from researchers. VEGF is particularly
important in the process of angiogenesis.
THD, a drug initially synthesized by Chemie
Gruneenthal, was used to treat morning sickness during pregnancy
but was later withdrawn from the market due to serious side effects
of phocomelia (17). However, studies
have also reported that THD has anti-inflammatory,
immunomodulatory, and anti angiogenic effects (16). The effects of THD on proliferation of
colon cancer cells are rarely reported. The present study
investigated the effects of THD treatment on the proliferation of
SW480 cells, and the effects of THD on the expression of VEGF-A and
HIF-1α. Treatment with 12.5, 25, 50, 100 and 200 µg/ml THD was able
to markedly decrease the proliferation of SW480 cells compared with
the control. Furthermore, the growth inhibition rate increased
significantly with the increase in THD concentration and duration
of treatment.
Previous studies have indicated that THD was able to
induce arrest at G1 phase, block DNA synthesis and inhibit the
proliferation of tumor cells [KG-la human acute myelogenous
leukemia cell lines, human pancreatic cancer cells (Patu-8988) and
U251-MG glioma cells] (26–28). Another in vitro study on
ovarian cancer also reported that THD was able to inhibit the
proliferation of ovarian cancer cells SKOV3 (36), which suggested that THD may have a
role in promoting anti-tumor effects by directly inhibiting
proliferation. On the other hand, THD was also able to promote the
apoptosis of transitional cell carcinoma cells (37).
There are three main pro-apoptotic pathways,
including the mitochondrial, death receptor pathway and endoplasmic
reticulum enzyme particles pathway (leading to the activation of
caspase-12 and apoptosis). The B-cell lymphoma 2 (Bcl-2) family has
a key function in the regulation of the mitochondrial pathway
(38,39). According to the differences in
function in apoptosis, the Bcl-2 family is divided into two
categories: Pro-apoptotic genes and anti-apoptotic genes. The
pro-apoptotic genes include BCL2-associated X (Bax), Bcl-2-like
protein 7 (Bak), Bcl2-associated agonist of cell death (Bad) and
BH3-interacting domain death agonist (Bid), and the anti-apoptotic
genes include Bcl-2 and Bcl-xL. Among these genes, the most
important are Bax and Bcl-2.
Qiao et al (40) demonstrated that the expression of Bax
mRNA was upregulated, the expression of Bcl-2 gene was
downregulated following the treatment of SW1990 pancreatic cancer
cells with THD.
Marriott et al (41) used the THD analogue, selective
cytokine inhibitory drug (SelCID-3), to treat tumor cell lines
[colorectal (SW620 and LoVo), pancreatic (BxPc-3 and T3M-4),
melanoma (MJT-3 and SP-1) and prostatic (PC-3 and DU-145)]. The
study demonstrated that SelCID3 was able to suppress the protein
expression of Bcl-2, activate caspase 3 and induce the apoptosis of
cancer cells. Furthermore, THD treatment was able to reduce the
proliferation of tumor cells (41).
VEGF is highly specific. VEGF has a number of
isoforms, including VEGF-A, VEGF-B, VEGF-C, VEGF-D and placental
growth factor (PIGF). VEGF-A can bind with vascular endothelial
growth factor receptor (VEGFR)-1 and VEGFR-2. VEGF has reported to
be a key factor in promoting angiogenesis (42). A number of studies have reported that
VEGF has an important role in promoting proliferation, migration
and chemotactic response in bone, lung, kidney, brain, vascular
endothelial cells, tumor and other tissues (43–48). Under
pathological conditions, hypoxia is the main factor that promotes
the synthesis of VEGF (49). A
previous study has demonstrated that the capacity of VEGF synthesis
in cells under hypoxic conditions would increase 3–12 times
(50). Hypoxia in cells may cause the
release of HIF-1 and promote the transcription of VEGF. When
different concentrations of THD were incubated with human vascular
endothelial cells in vitro, it was observed that the
secretion of VEGF, cell adhesion ability of collagen and cell
migration ability were all inhibited. Therefore, the formation of
new capillaries was also reduced. This may be associated with the
THD-induced downregulation of VEGF (51,52). At
the same time, THD was able to block the NF-κB signaling pathway,
its downstream adhesion molecules and the expression of
inflammatory mediators including TNF-α (53).
It was verified in a previous study that NF-κB
activates IL-6, and IL-6 is able to increase the expression of VEGF
(54). Therefore, if the activation
of NF-κB was inhibited, this may inhibit IL-6 activation, thereby
decreasing VEGF expression. The results of q-PCR indicated that the
VEGF-A mRNA expression was markedly decreased following THD
treatment, and the decrease was concentration dependent. Therefore,
THD may be able to reduce angiogenesis in tumor cells. VEGF is one
of the target genes of HIF-1α (55).
Under hypoxic conditions, signal transducer and activator of
transcription 3 (STAT3) is activated. Activated STAT3 induces the
transcription and expression of HIF-1α, which transcribes and
expresses the target gene VEGF. VEGF participates in tumor
angiogenesis, and HIF-1α can promote the expression of
erythropoietin and inhibit the apoptosis of cancer cells in a
hypoxic environment, ultimately result in the constant growth of
tumor cells (56). Nechemia-Arbely
et al (57) also identified in
an in vitro study that activated STAT3 was able to induce
the upregulation of HIF-1α in mouse renal cells.
The inflammatory microenvironment is an important
factor in promoting tumor progression. The IL-6-mediated downstream
signal STAT3 has an important role in tumor development and
metastasis. The IL-6 receptor binds with soluble IL-6 (sIL-6R), and
the IL-6/sIL-6R compound then activates membrane glycoprotein 130,
and causes the activation of STAT3. This leads to the induction of
tumor cells to proliferate, migrate and undergo angiogenesis
(58). Therefore, blocking the IL-6
signal transduction pathway may inhibit the activation of STAT3,
leading to downregulation of HIF-1α and reduced transcription. This
consequently reduces tumor angiogenesis. In the present study, it
was observed that HIF-1α expression was markedly decreased
following the treatment of SW480 cells with THD for 48 h; however,
as the concentration of THD increased, there was not a significant
change in HIF-1α expression levels. At present, the antitumor
mechanisms of THD that have been reported included changes in the
levels of cytokines e.g., concentration of tumor necrosis factor
(TNF)-α, IL-6 and IFN-γ. In addition, it was indicated that these
changes were able to regulate the immune status of the body
(59). An in vitro study
indicated that treatment with 100 µM THD was able to inhibit the
expression of IL-6, IL-8, TNF-α and VEGF (60). Therefore, this led to the inhibition
of STAT3 activation and decreased HIF-1α expression (60). THD has been demonstrated to inhibit
the expression of HIF-1α through the STAT3 signaling pathway
mediated by IL-6. Furthermore, a number of studies have indicated
that THD was also able to directly inhibit HIF-1α expression
(52,61).
Surgery is the main treatment for colorectal cancer,
and its effectiveness combined with post-surgical radiotherapy
and/or chemotherapy has been widely recognized (62). Systemic chemotherapy is the primary
treatment following surgery, its purpose is to eliminate micro
metastases in vivo, to prolong the life of patients and
improve the quality of life of the patients. At present, the
National Comprehensive Cancer Network recommends FOLFOX, a
combination of drugs which include 5-Fu, leucovorin and L-OHP for
the treatment of colon cancer (30,63). L-OHP
is the third generation platinum compound, and it is a new platinum
group, which contains 1,2-diamine cyclohexane (DACH). L-OHP
overcomes the toxicity of I and II generation platinum drugs.
Cisplatin is a second-generation platinum drug, and
cisplatin-resistant tumor cell lines exhibit varying degrees of
resistance to L-OHP (64). L-OHP is a
type of anticancer drug with good prospects and has attracted the
attention of researchers. The mechanism for L-OHP is to induce
platinum DNA adducts (Pt-GG and Pt-AG) which are intra chain
complexes that block DNA transcription and replication (63). However, if L-OHP was used alone, it
may cause neurotoxicity, hematological toxicity, gastrointestinal
toxicity and drug-resistance, which may lead to the failure of the
chemotherapy.
Studies have reported that the use of a combination
of anti-angiogenic drugs is able to improve the efficacy of
chemotherapy. Cremolini et al (65) discovered that progression-free
survival and the median survival time of patients with metastatic
colorectal cancer treated with bevacizumab plus FOLFOXIRI were
improved compared with patients treated with FOLFOXIRI alone.
However due to the high price of bevacizumab, this hindered the use
of the drug in some patients.
Studies have reported that the use of THD in
combination with other antineoplastic drugs may have a synergistic
antitumor effect.
Murphy et al (66) reported that THD may increase cisplatin
content in intracranial tumor and significantly increase the
efficacy of cisplatin in the rat intracranial glioma model using a
combination of cisplatin with THD. A phase II randomized controlled
trial indicated that patients with metastatic colorectal cancer,
who received L-OHP/capecitabine (XELOX) therapy plus THD as
first-line treatment had good tolerance, and the disease control
rate was significantly improved compared with the control group
receiving XELOX alone) (32).
In the present study, as THD was able to inhibit the
proliferation of SW480 cells, downregulate VEGF-A expression and
inhibit the expression of HIF-1α according to the preceding study,
it was assumed that a combination of THD and L-OHP have a
synergistic effect on SW480 cells. The IC50 of L-OHP in
the SW480 cells was 64 mg/l. Additionally, 50 µg/ml THD and 64 mg/l
L-OHP were selected to treat the SW480 cells. The cells were either
treated with THD and L-OHP separately or together for 48 h. The
expression of VEGF-A mRNA and HIF-1α protein were subsequently
detected. The experiment identified that VEGF-A mRNA expression and
the HIF-1α protein expression were decreased following the
treatment of SW480 cells with 50 µg/ml THD, 64 mg/l L-OHP and 50
µg/ml THD + 64 mg/l L-OHP for 48 h. The combined treatment of two
drugs had a synergistic effect. Research has demonstrated that IL-6
may activate STAT3 in colorectal cancer (67,68), and
it has been reported that L-OHP may downregulate the expression of
HIF-1α through inhibiting the STAT3 signaling pathway mediated by
IL-6 (69). THD has been demonstrated
to inhibit the expression of HIF-1α through the STAT3 signaling
pathway mediated by IL-6 aforementioned (52,60,61). Two
drugs have interacted together in this pathway, and had a
synergistic effect. HIF-1α is an upstream regulatory gene of VEGF
and therefore downregulation of HIF-1α resulted in downregulation
of VEGF-A-mRNA.
Therefore, it was not unexpected to detect the
inhibitory effects of THD on proliferation of cancer cells in the
present study. Additionally, THD is also able to have an inhibitory
effect on the expression of VEGF-A and its upstream gene HIF-1α.
Treatment with a combination of THD and L-OHP has a synergistic
inhibitory effect on the growth of the SW480 cells and this
provides a new strategy for the treatment of advanced colorectal
cancer.
References
|
1
|
International Agency for Research on
Cancer, . Colorectal cancer estimated incidence, mortality and
prevalence worldwide in 2012 [EB/OL]. http://globocan.iarc.fr/Pages/fact_sheets_cancer.aspx
|
|
2
|
Zong-Guang Zhou, Lie Yang, Yuan Li, et al:
The 30 years' changes of colorectal cancer and the strategies in
China. Chin J Practic Surg. 32:693–696. 2012.(In Chinese).
|
|
3
|
Folkman J: What is the evidence that
tumors are angiogenesis dependent? J Natl Cancer Inst. 82:4–6.
1990. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wieck MM, Spurrier RG, Levin DE, Mojica
SG, Hiatt MJ, Reddy R, Hou X, Navarro S, Lee J, Lundin A, et al:
Sequestration of vascular endothelial growth factor (VEGF) induces
late restrictive lung disease. PLoS One. 11:e01483232016.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lin LJ, Zheng CQ, Jin Y, Ma Y, Jiang WG
and Ma T: Expression of survivin protein in human colorectal
carcinogenesis. World J Gastroenterol. 9:974–977. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Willems-Widyastuti A, Vanaudenaerde BM,
Vos R, Dilisen E, Verleden SE, De Vleeschauwer SI, Vaneylen A, Mooi
WJ, de Boer WI, Sharma HS and Verleden GM: Azithromycin attenuates
fibroblast growth induced vascular endothelial growth factor via
p38(MAPK)signaling in human airway smooth muscle cells. Cell
Biochem Biophys. 67:331–339. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zachary I and Gliki G: Signaling
transduction mechanisms mediating biological actions of the
vascular endothelial growth factor family. Cardiov Asc Res.
49:568–581. 2001. View Article : Google Scholar
|
|
8
|
Zeng H, Sanyal S and Mukhopadhyay D:
Tyrosine residues 951 and 1059 of vascular endothelial growth
factor receptor-2 (KDR) are essential for vascular permeability
factor/vascular endothelial growth factor-induced endothelium
migration and proliferation, respectively. Biol Chem.
276:32714–32719. 2001. View Article : Google Scholar
|
|
9
|
Xu XL, Ling ZQ, Chen W, Xu YP and Mao WM:
The overexpression of VEGF in esophageal cancer is associated with
a more advanced TMN stage: A meta-analysis. Cancer Biomark.
13:105–113. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhang Y, Liu X, Zhang J, Li L and Liu C:
The expression and clinical significance of PI3K, pAkt and VEGF in
colon cancer. Oncol Lett. 4:763–766. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ratcliffe PJ: HIF-l and HIF-2: Working
alone or together in hypoxia? J Clin Invest. 117:862–865. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Li W, Xiong ZW, Li HW, Hu HX, Zhang P, Su
H, Huang Y, Zhang HD and Xun FH: Expression of COX-2, VEGF and E
-cad in breast cancer and their clinicopathologic significance.
Chin J Curr Adv Gen Surg. 13:766–771. 2010.(In Chinese).
|
|
13
|
Zhang P, Dong L, Yan K, Long H, Yang TT,
Dong MQ, Zhou Y, Fan QY and Ma BA: CXCR4-mediated osteosarcoma
growth and pulmonary metastasis is promoted by mesenchymal stem
cells through VEGF. Oncol Rep. 30:1753–1761. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wu Y, Jin M, Xu H, Shimin Z, He S, Wang L
and Zhang Y: Clinicopathologic significance of HIF-1α, CXCR4, and
VEGF expression in colon cancer. Clin Dev Immunol.
2010:pii:5375312010. View Article : Google Scholar
|
|
15
|
Ahluwalia A and Tarnawski AS: Critical
role of hypoxia sensor-HIF-1α in VEGF gene activation. Implications
for angiogenesis and tissue injury healing. Curr Med Chem.
19:90–97. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
De Francesco EM, Lappano R, Santolla MF,
Marsico S, Caruso A and Maggiolini M: HIF-1α/GPER signaling
mediates the expression of VEGF induced by hypoxia in breast
cancer-associated fibroblasts (CAFs). Breast Cancer Res.
15:R642013. View
Article : Google Scholar : PubMed/NCBI
|
|
17
|
Decourt B, Drumm-Gurnee D, Wilson J,
Jacobson S, Belden C, Sirrel S, Ahmadi M, Shill H, Powell J, Walker
A, et al: Poor safety and Tolerability Hamper Reaching a
potentially therapeutic dose in the use of thalidomide for
Alzheimer's disease: Results from a Double-Blind Placebo-Controlled
trail. Curr Alzheimer Res. 14:403–411. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Raturi R, Patel AA and Carter JD: Two
cases demonstrating thalidomide's efficacy in refractory lupus
nephritis. Clin Rheumatol. 36:725–728. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Anargyrou K, Dimopoulos MA, Sezer O and
Terpos E: Novel anti-myeloma agents and angiogenesis. Leuk
Lymphoma. 49:677–689. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Bombini G, Canetti C, Rocha FA and Cunha
FQ: Tumour necrosis factor-alpha mediates neutrophil migration to
the knee synovial cavity during immune inflammation. Eur J
Pharmacol. 496:197–204. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Deng L, Ding W and Granstein RD:
Thalidomide inhibits tumor necrosis factor-alpha production and
antigen presentation by Langerhans cells. J Invest Dermatol.
121:1060–1065. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Keifer JA, Guttridge DC, Ashburner BP and
Baldwin AS Jr: Inhibition of NF-kappa B activity by thalidomide
through suppression of IkappaB kinase activity. J Biol Chem.
276:22382–22387. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yu J, Liu F, Sun Z, Sun M and Sun S: The
enhancement of radiosensitivity in human esophageal carcinoma cell
by thalidomide and its potential mechanism. Cancer Biother
Radiopharm. 26:219–227. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Pan J, Lu GP and Yu ZJ: Thalidomide on
anti-tumor research. Chin J Cancer Prev Treat. 19:552–555.
2012.
|
|
25
|
Uach H, Ritchie D, Stewart AK, Neeson P,
Harrison S, Smyth MJ and Prince HM: Mechanism of action of
immunomodulatory drugs (IMiDS) in multiple myeloma. Leukemia.
24:22–32. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Girgis E, Mahoney J, Darling-Reed S and
Soliman M: Arsenic trioxde enhance the cytotoxic effect of
thalidomide in a KG-la human acute mylogenous leukemia cell line.
Oncol Lett. 1:473–479. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Xu X, Xiong G, Li E, Zhu H, Lio H, Shen G
and Wu S: Effect of thalidomide on cytodynamics and VEGF expression
of human pancreatic cancer cells. Chin J Gastroenterol. 9:511–513.
2012.
|
|
28
|
Gao S, Yang XJ, Zhang WG, Ji YW and Pan Q:
Mechanism of thalidomide to enhance cytotoxicity of temozolomide in
U251-MG glioma cells in vitro. Chin Med J (Engl). 122:1260–1266.
2009.PubMed/NCBI
|
|
29
|
Lin YC, Shun CT, Wu MS and Chen CC: A
novel anticancer effect of thalidomide: Inhibition of intercellular
adhesion molecule-1-mediated cell invasion and metastasis through
suppression of nuclear factor-kappaB. Clin Cancer Res.
12:7165–7173. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
André T, Boni C, Navarro M, Tabernero J,
Hickish T, Topham C, Bonetti A, Clingan P, Bridgewater J, Rivera F
and de Gramont A: Improved overall survival with oxaliplatin,
fluorouracil, and leucovorin as adjuvant treatment in stage II or
III colon cancer in the MOSAIC trial. J Clin Oncol. 27:3109–3116.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Dahan L, Sadok A, Formento JL, Seitz JF
and Kovacic H: Modulation of cellular redox state underlies
antagonism between oxaliplatin and cetuximab in human colorectal
cancer cell lines. Br J Pharmacol. 158:610–620. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Lv J, Liu N, Liu KW, Ding AP, Wang H and
Qiu WS: A Randomised controlled phase II trial of the combination
of XELOX with thalidomide for the first-line treatment of
metastatic colorectal cancer. Cancer Biol Med. 9:111–114.
2012.PubMed/NCBI
|
|
33
|
Govindarajan R: Irinotecan/thalidomide in
metastatic colorectal cancer. Oncology (Williston park). 16(4 Suppl
3): S23–S26. 2002.
|
|
34
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Folkman J: Tumor angiogenesis: Therapeutic
implications. N Engl J Med. 285:1182–1186. 1971. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhang MM, Cai L and LA DD: Study on
mechanism of thalidomide in human ovarian cancer cells SKOV3 in
vitro. J Practi Med. 26:2693–2696. 2010.
|
|
37
|
Huang YT, Cheng CC, Chiu TH and Lai PC:
Therapeutic potential of thalidomide for gemcitabine-resistant
bladder cancer. Int J Oncol. 47:1711–1724. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Wang C and Youle RJ: The role of
mitochondria in apoptosis*. Annu Rev Genet. 43:95–118. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Gu W, Zhao XG and Zhang X: Formononetin
induce apoptosis of SKOV3 cells through regulating the expression
of Bax and Bcl-2. Sichuan J Physiol. 36:60–62. 2014.
|
|
40
|
Qiao Z, Yuan J, Shen J, Wang C, He Z, Hu
Y, Zhang M and Xu C: Effect of thalidomide in combination with
gemcitabine on human pancreatic carcinoma SW-1990 cell lines in
vitro and in vivo. Oncol Lett. 9:2353–2360. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Marriott JB, Clarke LA, Czajks A, Dredge
K, Childs K, Man HW, Schafer P, Govinda S, Muller GW, Stirling DI
and Dalgleish AG: A novel subclass of thalidomide analogue with
anti-solid tumor activity in which caspase-dependent apoptosis is
associated with altered expression of bcl-2 family proteins. Cancer
Res. 63:593–599. 2003.PubMed/NCBI
|
|
42
|
Meller S and Bhandari V: VEGF levels in
humans and animal models with RDS and BPD: Temporal relationships.
Exp Lung Res. 38:192–203. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Gerber HP, Vu TH, Ryan AM, Kowalski J,
Werb Z and Ferrara N: VEGF couples hypertrophic cartilage
remodeling, ossification and angiogenesis during endochondral bone
formation. Nat Med. 5:623–628. 1999. View
Article : Google Scholar : PubMed/NCBI
|
|
44
|
Lee CG, Link H, Baluk P, Homer RJ,
Chapoval S, Bhandari V, Kang MJ, Cohn L, Kim YK, McDonald DM and
Elias JA: Vascular endothelial growth factor (VEGF) induces
remodeling and enhances TH2-mediated sensitization and inflammation
in the lung. Nat Med. 10:1095–1103. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zhao T, Zhao W, Meng W, Liu C, Chen Y,
Gerling IC, Weber KT, Bhattacharya SK, Kumar R and Sun Y:
VEGF-C/VEGFR-3 pathway promotes myocyte hypertrophy and survival in
the infracted myocardium. Am J Transl Res. 7:697–709.
2015.PubMed/NCBI
|
|
46
|
Iacovelli R, Sternberg CN, Porta C,
Verzoni E, de Braud F, Escudier B and Procopio G: Inhibition of the
VEGF/VEGFR pathway improves survival in advanced kidney cancer: A
systematic review and meta-analysis. Curr Drug Targets. 16:164–170.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Quittet MS, Touzani O, Sindji L, Cayon J,
Fillesoye F, Toutain J, Divoux D, Marteau L, Lecocq M, Roussel S,
et al: Effects of mesenchymal stem cell therapy, in association
with pharmacologically active microcarriers releasing VEGF, in an
ischaemic stroke model in the rat. Acta Biomater. 15:77–88. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Dzietko M, Derugin N, Wendland MF, Vexler
ZS and Ferriero DM: Delayed VEGF treatment enhances angiogenesis
and recovery after neonatal focal rodent stroke. Transl Stroke Res.
4:189–200. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Yang X, Zhu H, Ge Y, Liu J, Cai J, Qin Q,
Zhan L, Zhang C, Xu L, Liu Z, et al: Melittin enhances
radiosensitivity of hypoxic head and neck squamous cell carcinoma
by suppressing HIF-1α. Tumour Biol. 35:10443–10448. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Wilkinson-Berka JL: Vasoactive factors and
diabetic retinopathy: Vascular endothelial growth factor,
cycoloxygenase-2 and nitric oxide. Curr Pharm Des. 10:3331–3348.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Buckstein R, Kerbel R, Cheung M, Shaked Y,
Chodirker L, Lee CR, Lenis M, Davidson C, Cussen MA, Reis M, et al:
Lenalidomide and metronomic melphalan for CMML and higher risk MDS:
A phase 2 clinical study with biomarkers of angiogenesis. Leuk Res.
38:756–763. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Tan H, Chen H, Xu C, Ge Z, Gao Y, Fang J,
Liu W and Xiao S: Role of vascular endothelial growth factor in
angiodysplasia: An interventional study with thalidomide. J
Gastroenterol Hepatol. 27:1094–1101. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Hernandez Mde O, Fulco Tde O, Pinheiro RO,
Pereira Rde M, Redner P, Sarno EN, Lopes UG and Sampaio EP:
Thalidomide modulates Mycobacterium leprae-induced NF-κB pathway
and lower cytokine response. Eur J Pharmacol. 670:272–279. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Abcouwer SF: Angiogenic factors and
cytokines in diabetic retinopathy. J Clin Cell Immunol. (Suppl
1)2013.PubMed/NCBI
|
|
55
|
Huang YF, Yang CH, Huang CC, Tai MH and
Hsu KS: Pharmacological and genetic accumulation of
hypoxia-inducible factor-1alpha enhances excitatory synaptic
transmission in hippocampal neurons through the production of
vascular endothelial growth factor. J Neurosci. 30:6080–6093. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Carbajo-Pescador S, Ordonez R, Benet M,
Jover R, García-Palomo A, Mauriz JL and González-Gallego J:
Inhibition of VEGF expression through blockade of Hif1α and STAT3
signaling mediates the anti-angiogenic effect of melatonin in HepG2
liver cancer cells. Br J Cancer. 109:83–91. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Nechemia-Arbely Y, Khamaisi M, Rosenberger
C, Koesters R, Shina A, Geva C, Shriki A, Klaus S, Rosen S,
Rose-John S, et al: In vivo evidence suggesting reciprocal renal
hypoxia-inducible factor-1 upregulation and signal transducer and
activator of transcription 3 activation in response to hypoxic and
non-hypoxic stimuli. Clin Exp Pharmacol Physiol. 40:262–272. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Dourlat J, Liu WQ, Sancier F, Edmonds T,
Pamonsinlapatham P, Cruzalegui F and Garbay C: A novel
non-phosphorylated potential antitumoral peptide inhibits STAT3
biological activity. Biochimie. 91:996–1002. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Majumder S, Sreedhara SR, Banerjee S and
Chatterjee S: TNF-α signaling beholds thalidomide saga: A review of
mechanistic role of TNF-α signaling under thalidomide. Curr Top Med
Chem. 12:1456–1467. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
El-Aarag BY, Kasai T, Zahran MA, Zakhary
NI, Shigehiro T, Sekhar SC, Agwa HS, Mizutani A, Murakami H, Kakuta
H and Seno M: In vitro anti-proliferative and anti-angiogenic
activities of thalidomide dithiocarbamate analogs. Int
Immunopharmacol. 21:283–292. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Cook KM and Figg WD: Angiogenesis
inhibitors: Current strategies and future prospects. CA Cancer J
Clin. 60:222–243. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
People's Republic of China national health
and Family Planning Commission of medical affairs authority, Tumor
branch of Chinese Medical Association, . Standard for diagnosis and
treatment of colorectal cancer (2015). Chin J Practi Surg.
35:1177–1191. 2015.
|
|
63
|
Kelland L: The resurgence of
platinum-based cancer chemotherapy. Nat Rev Cancer. 7:573–584.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Voland C, Bord A, Péleraux A, Pénarier G,
Carrière D, Galiègue S, Cvitkovic E, Jbilo O and Casellas P:
Repression of cell cycle-related proteins by oxaliplatin but not
cisplatin in human colon cancer cells. Mol Cancer Ther.
5:2149–2157. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Cremolini C, Loupakis F, Masi G, Lonardi
S, Granetto C, Mancini ML, Chiara S, Moretto R, Rossini D, Vitello
S, et al: FOLFOXIRI or FOLFOXIRI plus bevacizumab as first-line
treatment of metastatic colorectal cancer: A propensity
score-adjusted analysis from two randomized clinical trials. Ann
Oncol. 27:843–849. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Murphy S, Davey RA, Gu XQ, Haywood MC,
McCann LA, Mather LE and Boyle FM: Enhancement of cisplatin
efficacy by thalidomide in a 9L rat gliosarcoma model. J
Neuroancol. 85:181–189. 2007. View Article : Google Scholar
|
|
67
|
Mihara M, Hashizume M, Yoshida H, Suzuki M
and Shiina M: IL-6/IL-6 receptor system and its role in
physiological and pathological conditions. Clin Sci (Lond).
122:143–159. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Heinrich PC, Behrmann I, Muller-Newen G,
Schaper F and Graeve L: Interleukin-6-type cytokine signalling
through the gp130/Jak/STAT pathway. Biochem J. 334:297–314. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Cross-Knorr S, Lu S, Perez K, Guevara S,
Brilliant K, Pisano C, Quesenberry PJ, Resnick MB and Chatterjee D:
RKIP phosphorylation and STAT3 activation is inhibited by
oxaliplatin and camptothecin and are associated with poor prognosis
in stage II colon cancer patients. BMC Cancer. 13:4632013.
View Article : Google Scholar : PubMed/NCBI
|