Introduction
Cancer stem cells (CSCs) are a subpopulation of
cancer cells that exhibit self-renewal and pluripotency (1). These traits of CSCs are associated with
their ability to divide asymmetrically and produce an increased
proportion of differentiated progeny cells, respectively, which
enables them to seed tumors (2).
Furthermore, CSCs possess distinctive characteristics, including
high tumor-initiating potential, resistance to therapies and tumor
recurrence (3). The stemness of a
population of cancer cells corresponds to the proportion and
tumorigenicity of CSCs present in this population (1). Furthermore, stemness is associated with
distinct changes in pluripotency gene expression (3). The expression of transcription factors,
including octamer-binding transcription factor 4 (OCT-4),
sex-determining region Y-related high mobility group box gene 2
(SOX2) and Krüppel-like factor 4 (KLF4), is increased in CSCs,
compared with non-CSCs, and confers the ability of self-renewal on
CSCs (4–6).
CSCs have been successfully isolated from various
tumor types, with breast CSCs being the first solid tumor-derived
CSCs (7). Expression of cell-surface
markers, including CD24lowCD44high, may be
used to identify breast CSCs (8).
Alternatively, breast CSCs may be identified on the basis of
aldehyde dehydrogenase (ALDH) activity (9). High pluripotency gene expression has
also been observed in CD24lowCD44high and
ALDH+ CSCs (10).
In breast CSCs, pluripotency gene expression is
regulated by complex signal transduction pathways, including the
signal transducer and activator of transcription 3 (STAT3)
signaling pathway (11,12). Previous studies have identified that
the transcription factor STAT3 serves a significant function in the
expression of pluripotency genes, including OCT-4, SOX2,
KLF4 and ALDH 1 family member A1 (ALDH1A1) (13,14).
Phosphorylation of STAT3 at Tyr705 induces its
activation and enables it to act as a potent activator of
pluripotency gene transcription. Results of in vitro and
in vivo studies have demonstrated that an increased level of
phosphorylated STAT3 (pSTAT3) is associated with
mammosphere-forming capacity, self-renewal, increased invasiveness,
tumor-generating capacity and metastatic potential, and that
targeted STAT3 inhibition suppresses relapse and metastasis in an
animal model (15).
Post-translational modification (PTM) is an
important step in cellular protein maturation and involves the
chemical modification of the protein structure, resulting in the
generation of various modified forms of a protein. Global
inhibition of protein N-glycosylation inhibits the Janus kinase
(JAK)-STAT signaling pathway and other signaling pathways in cancer
cells (16). Owing to targeted
therapy being resisted by CSCs, PTM inhibition is preferred over
targeted therapy for cancer treatment; however, PTM inhibition is
undermined by the problem of toxicity, as it induces considerable
damage by affecting multiple signaling pathways. A previous study
identified that the toxicity was associated with tunicamycin, an
N-glycosylation inhibitor (17).
Glucosamine is a naturally occurring amino
monosaccharide that is primarily located in connective and
cartilage tissues, where it serves as an essential component for
maintaining flexibility and elasticity; therefore, glucosamine is
frequently used for treating osteoarthritis in humans (18). Glucosamine has been indicated to be a
candidate N-glycosylation inhibitor due to its anticancer activity
(16,18). Chesnokov et al (16) indicated that glucosamine decreased the
N-linked glycosylation of gp130, a highly glycosylated interleukin
6 (IL-6) receptor subunit, resulting in the inhibition of the
IL-6-STAT3 signaling pathway. Currently, to the best of our
knowledge, it has not been examined whether glucosamine is able to
modify CSC stemness; therefore, in the present study, the effect of
glucosamine on the stemness of ALDH+ breast CSCs was
investigated.
Materials and methods
Reagents
D-glucosamine hydrochloride was purchased from
Sigma-Aldrich; Merck KGaA (Darmstadt, Germany). Dulbecco's modified
Eagle's medium/Ham's F12 (DMEM-F12) and high-glucose DMEM were
purchased from Gibco; Thermo Fisher Scientific, Inc. (Waltham, MA,
USA). Penicillin/streptomycin/amphotericin B mixture was purchased
from Lonza Group, Ltd. (Basel, Switzerland). An ALDEFLUOR™ kit was
purchased from Stemcell Technologies, Inc. (Vancouver, BC, Canada).
Recombinant human fibroblast growth factor (cat. no. 064-05381) was
purchased from Wako Chemicals USA, Inc. (Richmond, VA, USA). The
antibodies used in the present study were as follows: Mouse
anti-human STAT3 antibody (124H6; cat. no. 9139; Cell Signaling
Technology, Inc., Danvers, MA, USA); mouse anti-human pSTAT3
(Tyr705) antibody (3E2; cat. no. 9138; Cell Signaling
Technology, Inc.); mouse anti-human GAPDH antibody (cat. no.
sc-47724; Santa Cruz Biotechnology Inc., Dallas, TX, USA); and
horseradish peroxidase (HRP)-conjugated goat anti-mouse
immunoglobulin G (IgG; cat. no. sc-2005; Santa Cruz Biotechnology,
Inc.).
Cell culture and glucosamine
treatment
ALDH+ breast CSCs isolated from pleural
effusion of a patient with metastatic breast cancer were provided
by Professor Osamu Ohneda (Laboratory of Regenerative Medicine and
Stem Cell Biology, Graduate School of Comprehensive Human Sciences,
University of Tsukuba, Tsukuba, Japan). Additionally, these cells
have been established as a cell line, as described previously
(19,20). To retain their stemness,
ALDH+ breast CSCs were cultured in serum-free DMEM-F12
supplemented with 1% penicillin/streptomycin/amphotericin B at 37°C
in an atmosphere containing 5% CO2, as described
previously (21,22). Our preliminary experiments performed
using the ALDH+ breast CSCs confirmed that treatment
with 10% fetal bovine serum (FBS; Gibco; Thermo Fisher Scientific,
Inc.) induced differentiation of these CSCs, with notable changes
in their morphology from floating and sphere-like cells to attached
and epithelial-like cells (data not shown).
Human adherent epithelial adenocarcinoma cell line
MCF7 was purchased from the American Type Culture Collection
(Manassas, VA, USA). These cells were cultured in high-glucose DMEM
supplemented with 10% (v/v) FBS and 1%
penicillin/streptomycin/amphotericin B at 37°C in an atmosphere
containing 5% CO2.
ALDH+ breast CSCs and MCF7 cells were
seeded in a 6-well plate (1×105 cells/well) and were
cultured under aforementioned conditions. After 24 h, DMEM-F12 was
replaced with serum-free medium containing D-glucosamine
hydrochloride. The concentrations of D-glucosamine used for
ALDH+ breast CSCs were 0.25, 1, 4, 10 or 16 mM, whereas
for MCF7 cells the concentrations used were 0.25, 1 or 4 mM. The
control was ALDH+ breast CSCs or MCF7 cells without
D-glucosamine treatment. Following 24 h treatment at 37°C, the
cells were harvested for further analysis.
Cell viability assay
Cell viability was determined by performing a trypan
blue exclusion assay. Cell suspension was stained with 0.4% trypan
blue solution (1:1 mixture) and was allowed to stand for 2 min at
room temperature. Viable and dead cells were counted using a Luna™
automated cell counter (Logos Biosystems, Anyang, Gyeonggi, Korea).
Relative viability was calculated using the following formula:
Viability (%)=(number of viable cells/number of total cells)
×100.
Mammosphere formation assay
Breast CSCs were seeded at a density of 100
cells/well in an ultra-low attachment 96-well plate (Corning
Incorporated, New York, NY, USA) and grown in DMEM-F12 supplemented
with 0, 0.25, 1, 4, 10 or 16 mM glucosamine at 37°C in an
atmosphere containing 5% CO2. Formation of mammospheres
from the breast CSCs was determined after 3 days. MCF7 cells were
seeded at a density of 200 cells/well in an ultra-low attachment
96-well plate and grown in high-glucose DMEM supplemented with 20
ng/ml basic fibroblast growth factor at 37°C in an atmosphere
containing 5% CO2. Formation of mammospheres from MCF7
cells was determined after 7 days.
The formation of mammosphere was observed under
inverted microscope at ×100 magnification (model no. IM-3; OPTIKA
Srl, Ponteranica, Italy). Spheres ≥60 µm in diameter were counted
as mammosphere-forming units (MFUs) (23) using OPTIKA Srl software (version 2.7;
OPTIKA Srl). Diameters of irregularly shaped spheres were
determined using the shortest diameter.
RNA extraction and reverse
transcription-quantitative polymerase chain reaction (RT-qPCR)
For performing RT-qPCR, total RNA was extracted from
the cells using the TriPure Isolation Reagent (Roche Diagnostics,
Basel, Switzerland), according to the manufacturer's protocol. RNA
concentration was measured spectrophotometrically by using a
Varioskan Flash Multimode Reader (Thermo Fisher Scientific, Inc.)
and the isolated RNA was stored at −80°C. RT-qPCR was performed
using a KAPA™ SYBR® FAST One-Step qRT-PCR kit (Kapa
Biosystems, Inc., Wilmington, MA, USA) and Exicycler™ 96 thermal
block (Bioneer Corporation, Daejeon, Korea). PCR was performed
using the following primers: OCT-4 forward,
5′-GAGGAGTCCCAGGACATCAAA-3′ and reverse,
5′-AGCTTCCTCCACCCACTTCT-3′; ALDH1A1 forward,
5′-GGAGGAAACCCTGCCTCTTTT-3′ and reverse,
5′-TTGGAAGATAGGGCCTGCAC-3′; KLF4 forward,
5′-CCGCTCCATTACCAAGAG-3′ and reverse, 5′-TTTCTCACCTGTGTGGGTTC-3′;
and 18S rRNA gene forward, 5′-AAACGGCTACCACATCCAAG-3′ and reverse,
5′-CCTCCAATGGATCCTCGTTA-3′. The conditions for PCR are as follows:
Initial denaturation at 42°C for 5 min and 95°C for 5 min; followed
by 40 cycles of denaturation at 95°C for 30 sec, annealing at
optimal temperature (57°C, 59°C, 55°C and 60°C for OCT-4,
ALDH1A1, KLF4, and 18S rRNA, respectively) for 20 sec and
extension at 72°C for 20 sec. Amplicon levels of the target genes
are expressed relative to those of the 18S rRNA gene, which was
used as an internal control, using the ∆∆Cq method
(24).
Western blot analysis
Following treatment with glucosamine, the cells were
washed twice with PBS and were lysed for 10 min in
radioimmunoprecipitation assay buffer (Santa Cruz Biotechnology,
Inc.) containing an anti-protease mixture. Protein concentration
was determined using the Bradford method (25). Protein fractions were suspended in a
Laemmli sample buffer (Bio-Rad Laboratories, Inc.) and were
denatured at 100°C for 5 min. Total protein (20 µg/lane) were
separated by SDS-PAGE (12% gel) and were transferred onto
nitrocellulose membranes (Bio-Rad Laboratories, Inc.). The
membranes were blocked by incubation with 5% bovine serum albumin
(Nacalai Tesque, Inc., Kyoto, Japan) in Tris-buffered saline
containing 0.1% Tween-20 for 1 h at room temperature and were
incubated overnight at 4°C with mouse anti-human monoclonal
antibodies against STAT3 (1:2,000 dilution), pSTAT3 (1:1,000
dilution) and GAPDH (1:200 dilution). The blots were visualized
using HRP-conjugated anti-mouse IgG (1:2,000 dilution) and enhanced
chemiluminescence reagent (cat. no. ab133406; Abcam, Cambridge,
UK). Intensities of bands representing pSTAT3 and STAT3 expression
levels were calculated using ImageJ software (Version 1.50i;
National Institutes of Health, Bethesda, MD, USA), and the
pSTAT3/STAT3 ratio was calculated using the formula: pSTAT3/STAT3
ratio=(pSTAT3 intensity/GAPDH intensity)/(STAT3 intensity/GAPDH
intensity).
Statistical analysis
All results are expressed as mean ± standard
deviation of at least three independent experiments. Statistical
analysis was performed using SPSS 20.0 software (IBM Corp., Armonk,
NY, USA). Statistical differences among the groups were determined
using one-way analysis of variance and Duncan post hoc test.
P<0.05 was considered to indicate a statistically significant
difference.
Results
Glucosamine decreases ALDH+
CSC viability
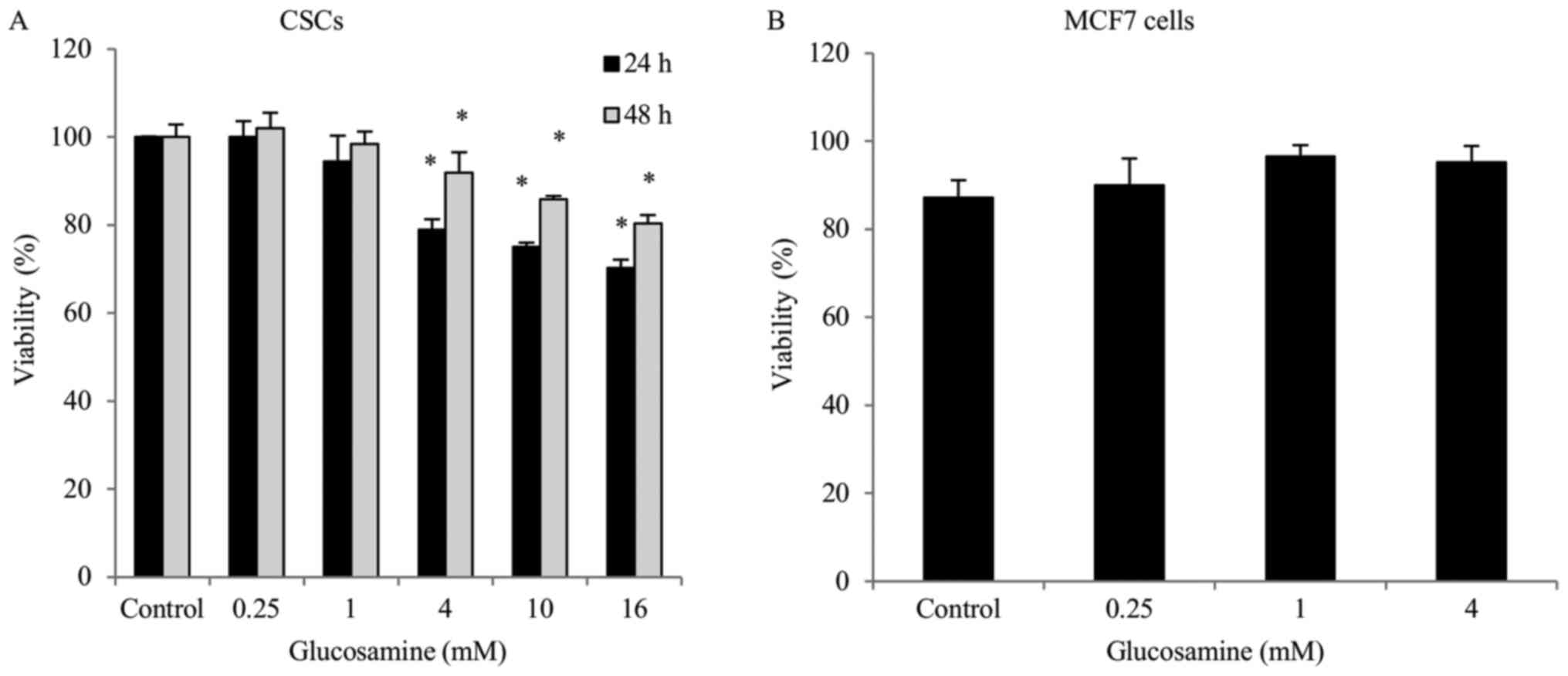
The effects of various concentrations of glucosamine
on the viability of ALDH+ breast CSCs and MCF7 cells was
determined using a trypan blue exclusion assay. Glucosamine
treatment (0.25, 1, 4, 10 or 16 mM) gradually decreased
ALDH+ breast CSC viability in a dose-dependent manner.
Treatment with glucosamine in ALDH+ breast CSCs for
different durations demonstrated that the shorter duration (24 h)
of treatment resulted in a greater decrease in cell viability,
compared with the longer duration (48 h) of treatment (Fig. 1); however, MCF7 cell viability did not
significantly alter following glucosamine treatment (0.25, 1 or 4
mM) (Fig. 1).
Glucosamine downregulates the
expression of stemness genes in ALDH+ breast CSCs and
MCF7 cells
ALDH1A1 is a detoxifying enzyme in the aldehyde
metabolic pathway (9). Furthermore,
ALDH1A1 has been proposed as a marker for identifying and isolating
CSCs from various cancer cells, including breast cancer cells
(9). ALDH1A1 expression was
increased 1.8-fold in untreated ALDH+ breast CSCs,
compared with in MCF7 cells (Fig.
2A). Compared with the control, treatment with 4 mM glucosamine
significantly downregulated ALDH1A1 expression in
ALDH+ breast CSCs (0.7-fold; P<0.05) and MCF7 cells
(0.49-fold; P<0.01) (Fig. 2A).
OCT-4 and KLF4 are transcription factors expressed in embryonic and
adult human stem cells. They are termed Yamanaka factors due to
their major functions in induced pluripotent stem cells (26). OCT-4 and KLF4 expression
was increased 1.8- and 2-fold, respectively, in untreated CSCs,
compared with untreated MCF7 cells (Fig.
2B and C). Glucosamine treatment (4 mM) significantly
downregulated OCT-4 (0.46-fold; P<0.01) and KLF4
expression (0.42-fold; P<0.01) in ALDH+ breast CSCs,
compared with their respective control. The downregulation of
OCT-4 expression following glucosamine treatment
demonstrated a similar pattern in ALDH+ breast CSCs and
MCF7 cells (Fig. 2B); however,
KLF4 expression was upregulated in MCF7 cells (Fig. 2C), compared with the control.
Glucosamine decreases mammosphere
formation in ALDH+ breast CSCs and MCF7 cells
To examine whether glucosamine treatment affected
the self-renewal capacity of CSCs, an in vitro mammosphere
formation assay was performed. Results of our preliminary
experiments demonstrated that mammospheres of ≥60 µm in diameter
were formed in ALDH+ breast CSCs on the third day,
whereas in MCF7 cells these formed on the seventh day following
seeding, indicating increased tumorigenicity of ALDH+
breast CSCs, compared with MCF7 cells. Furthermore, this result
demonstrated that CSCs were more enriched in ALDH+
breast cancer cells, compared with MCF7 cells. The number of
mammospheres formed by untreated ALDH+ breast CSCs was
markedly increased (5.02-fold), compared with MCF7 cells (Fig. 3). Treatment with 4 mM glucosamine
significantly decreased the MFUs in ALDH+ breast CSCs
and MCF7 cells. Notably, the decrease in MFUs in
glucosamine-treated ALDH+ breast CSCs was significantly
increased, compared with glucosamine-treated MCF7 cells (Fig. 3).
Glucosamine suppresses STAT3 signaling
in ALDH+ breast CSCs and MCF7 cells
Subsequently, the activation of the STAT3 pathway, a
key signaling pathway, was examined in ALDH+ breast
CSCs. STAT3 is activated through phosphorylation, and activated
STAT3 induces the expression of target genes, including OCT-4,
SOX2, KLF4 and ALDH1A1 (13,14). STAT3
and pSTAT3 levels were determined by western blot analysis.
Activation of the STAT3 signaling pathway was determined by
calculating the pSTAT3/STAT3 ratio. Glucosamine treatment inhibited
STAT3 phosphorylation in ALDH+ breast CSCs and MCF7
cells (Fig. 4); however, 4 mM
glucosamine-induced inhibition of STAT3 phosphorylation in
ALDH+ breast CSCs was less significant (P<0.05),
compared with that in MCF7 cells (P<0.01).
Discussion
Eradication of cancer cells following therapy has
always been difficult due to their resistance to the anticancer
effects of the therapy. In chemotherapy, cancer cells resist
eradication by pumping out drugs or by preventing the induction of
apoptotic cascades (27). In targeted
therapy, cancer cells resist eradication by compensating for the
decreased or missing activity of a particular protein by activating
other pathways, including IL-6/STAT3 and Notch3, to sustain their
oncogenic state and/or stemness (28).
In the present study, the decision to inhibit PTM as
an approach for non-targeted anticancer therapy was primarily based
on tumor heterogeneity, which is one of the causes of the failure
of cancer treatment. Theoretically, a highly heterogeneous
population of cancer cells can survive targeted therapy due to them
having a high possibility of containing a resistant clone (29). This assumption was confirmed by
reports on the enrichment of CSCs following chemotherapy (30). Residual CSCs adopt numerous mechanisms
to withstand various therapies, including targeted therapy
(27); therefore, to prevent any form
of ‘bounce back’ following targeted therapy, it was considered that
a reasonable approach was to target the most pathways possible
without endangering normal cells. As reported previously, CSCs have
difficulty rewiring important pathways due to PTM inhibition
affecting multiple pathways (16).
Furthermore, due to drug efflux transporters, including multidrug
resistance protein 1 (MDR1), requiring appropriate glycosylation,
cancer cells may not be able to resist the effects of chemotherapy
following PTM inhibition (31);
therefore, it was indicated that PTM inhibition is preferable to
targeted therapy for eradicating CSCs.
As described previously, toxicity is the primary
drawback of the global inhibition of N-glycosylation. This
indicates the requirement for developing a strategy, in which
cancer cells intake increased amount of PTM inhibitor, compared
with normal cells. A possible way to address this issue is to
utilize the concept of cancer cell metabolism, known as the Warburg
effect. To survive under relatively hypoxic conditions, cancer
cells adjust their metabolism to a glycolytic state, thus
increasing lactate production; therefore, cancer cells exhibit high
glucose uptake (32). Glucosamine was
used to inhibit the protein N-glycosylation in the present study
due to its structure mimicking the structure of glucose and it
exhibiting low toxicity (16,33).
The anticancer activity of glucosamine has been
known for >50 years; however, the mechanism underlying the
anticancer activity of glucosamine remains unclear (34). A number of mechanisms, including
autophagy induction, proteasomal activity inhibition, cell cycle
arrest, nuclear factor-κB inhibition and N-glycosylation
inhibition, have been proposed (18).
Glucosamine is indicated to inhibit the activity of JAK/STAT
signaling proteins, including STAT3, by inhibiting the
N-glycosylation of gp130, a subunit of the IL-6 receptor complex
(16); however, to the best of our
knowledge, it has not been examined whether glucosamine affects
stemness. In the present study, it was observed that glucosamine
decreased ALDH1A1, OCT-4 and KLF4 expression and the
mammosphere-forming ability of ALDH+ breast CSCs and
MCF7 cells, indicating that glucosamine decreased the stemness of
these cells. These changes are consistent with the decreased
pSTAT3/STAT3 ratio in these cells. On the basis of results of
previous studies (13,15), we hypothesized that the downregulation
of stemness gene expression in ALDH+ breast CSCs and
MCF7 cells following glucosamine treatment may be a functional
consequence of STAT3 inactivation.
The results from the western blot analysis confirmed
that glucosamine significantly inhibited STAT3 phosphorylation of
CSCs and MCF7 cells to a similar degree. The less significant
inhibitory effect of 4 mM glucosamine in ALDH+ breast
cancer cells may be due to the increased abundance of CSCs in
ALDH+ breast cancer cells, compared with MCF7 cells,
which exerted a density-dependent effect on chemoresistance, as
indicated by He et al (35).
CSCs may also attempt to maintain their stemness by activating
other pathways that contribute to STAT3 activation, including
overactivation of the G-protein-coupled receptor signaling pathway
or overexpression of gp130 (36).
Furthermore, it was observed that the reduction of
the pSTAT3/STAT3 ratio was consistent with the decrease in the
expression of stemness genes OCT-4 and ALDH1A1. These
results confirmed the results of previous studies that reported
that OCT-4 and ALDH1A1 were regulated by the
transcription factor STAT3 (13,14);
however, a STAT3 inhibitor was not used in the present experiments,
which is a limitation of the present study. Lin et al
(13) demonstrated that inhibition of
STAT3 phosphorylation using Stattic and LLL12 decreased the
tumorigenicity and viability of ALDH+ breast CSCs;
therefore, we hypothesized that the downregulation of stemness gene
expression observed in the present study was a functional
consequence of STAT3 inactivation.
Consistent with previous studies (16,37)
involving various prostate cancer and non-small cell lung cancer
cells, the results of the present study demonstrated that treatment
with ≥4 mM glucosamine significantly decreased the viability of
human ALDH+ breast CSCs. This confirmed the requirement
of a high glucosamine concentration for in vitro study. In
the present study, glucosamine treatment in a xenograft mice model
was not investigated owing to circumstances and limitations in
available animal laboratory facilities. In order to reach an
effective concentration in vivo, it is considered that there
will be difficulties. In the study reported by Song et al
(37), using a xenograft mouse lung
tumor model, glucosamine was required to be introduced at a
concentration as high as 500 mg/kg body weight in order to achieve
a significant benefit of glucosamine. Furthermore, Weimer et
al (38) reported that the plasma
level of glucosamine in a mouse model may be increased up to ~2 µM.
Notably, it was observed that the effects of glucosamine on
ALDH+ breast CSC viability are increased after 24 h of
treatment, compared with after 48 h; however, the glucosamine
treatments for longer durations were not performed in the present
study due to MCF7 cells not surviving in serum-free treatment
medium for ≥48 h. If the effect of stemness suppression is only
short-term, it may indicate that glucosamine treatment should be
repeated as necessary to replenish the effect.
In contrast with ALDH+ breast CSCs, a
significant effect of glucosamine on the viability of MCF7 cells
was not observed. Distinct metabolic pathways in CSCs and non-CSCs
may be responsible for different responses to glucosamine
treatment. For example, CD44+CD117+ ovarian
CSCs exhibit increased glucose uptake, compared with
CD44+CD117− ovarian non-CSCs (39). Additionally, the differential effects
of glucosamine on the viability of ALDH+ breast CSCs and
MCF7 cells may depend on the density of CSCs in ALDH+
breast cancer cells, compared with MCF7 cells, due to STAT3
inactivation as aforementioned. Owing to the notably lower density
of CSCs in MCF7 cells, compared with ALDH+ breast cancer
cells, we hypothesized that STAT3 inactivation following
glucosamine treatment may be a mediator of the antiproliferative
effect of glucosamine, which specifically targets CSCs.
Although it was determined that glucosamine
treatment significantly decreased CSC viability, the extent of the
decrease in cell viability was less than that induced by Stattic-
and LLL12-targeted therapy in ALDH+ breast CSCs
(13,15); therefore, we hypothesized that
glucosamine treatment may be improved when applied as an adjuvant
therapy. There are two previous studies that identified that an
N-glycosylation inhibitor acts as a chemosensitizer by affecting
transporter proteins involved in multidrug resistance, including
MDR1 (31). Furthermore, the global
inhibition of PTM may affect the CSC niche (40), thus overcoming problems associated
with CSC targeting, particularly problems associated with
plasticity and epithelial-mesenchymal transition (41).
In conclusion, to the best of our knowledge, the
present study is the first to demonstrate that glucosamine affected
the stemness of human ALDH+ breast CSCs, thus decreasing
their viability. This effect of glucosamine may be associated with
the inhibition of STAT3 phosphorylation; however, further
investigations with xenograft animal models are required to verify
the effects of glucosamine observed in the present study.
Acknowledgements
The authors would like to thank the Laboratory of
Regenerative Medicine & Stem Cell Biology, University of
Tsukuba (Tsukuba, Japan) for providing support for breast CSC
sorting.
Funding
The present study was supported by the Penelitian
Unggulan Perguruan Tinggi 2016 grant from the Ministry of
Research, Technology and Higher Education of the Republic of
Indonesia.
Availability of data and material
The datasets used and/or analyzed in the present
study can be made available by the corresponding author upon
reasonable request.
Authors' contributions
RH and SIW designed all the experiments, and
analyzed and interpreted the study data. RH performed the
experiments and prepared the manuscript. NSH and OO assisted in
conducting cell sorting. SIW and RH edited the manuscript prior to
submission. All the authors have read and approved the final
manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Visvader JE and Lindeman GJ: Cancer stem
cells: Current status and evolving complexities. Cell Stem Cell.
10:717–728. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kreso A and Dick JE: Evolution of the
cancer stem cell model. Cell Stem Cell. 14:275–291. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Batlle E and Clevers H: Cancer stem cells
revisited. Nat Med. 23:1124–1134. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hu T, Liu S, Breiter DR, Wang F, Tang Y
and Sun S: Octamer 4 small interfering RNA results in cancer stem
cell-like cell apoptosis. Cancer Res. 68:6533–6540. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yu F, Li J, Chen H, Fu J, Ray S, Huang S,
Zheng H and Ai W: Kruppel-like factor 4 (KLF4) is required for
maintenance of breast cancer stem cells and for cell migration and
invasion. Oncogene. 30:2161–2172. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Leis O, Eguiara A, Lopez-Arribillaga E,
Alberdi MJ, Hernandez-Garcia S, Elorriaga K, Pandiella A, Rezola R
and Martin AG: Sox2 expression in breast tumours and activation in
breast cancer stem cells. Oncogene. 31:1354–1365. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hamburger AW and Salmon SE: Primary
bioassay of human tumor stem cells. Science. 197:461–463. 1977.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Al-Hajj M, Wicha MS, Benito-Hernandez A,
Morrison SJ and Clarke MF: Prospective identification of
tumorigenic breast cancer cells. Proc Natl Acad Sci USA.
100:3983–3988. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ginestier C, Hur MH, Charafe-Jauffret E,
Monville F, Dutcher J, Brown M, Jacquemier J, Viens P, Kleer CG,
Liu S, et al: ALDH1 is a marker of normal and malignant human
mammary stem cells and a predictor of poor clinical outcome. Cell
Stem Cell. 1:555–567. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ponti D, Costa A, Zaffaroni N, Pratesi G,
Petrangolini G, Coradini D, Pilotti S, Pierotti MA and Daidone MG:
Isolation and in vitro propagation of tumorigenic breast cancer
cells with stem/progenitor cell properties. Cancer Res.
65:5506–5511. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kim SY, Kang JW, Song X, Kim BK, Yoo YD,
Kwon YT and Lee YJ: Role of the IL-6-JAK1-STAT3-Oct-4 pathway in
the conversion of non-stem cancer cells into cancer stem-like
cells. Cell Signal. 25:961–969. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Pattabiraman DR and Weinberg RA: Tackling
the cancer stem cells-what challenges do they pose? Nat Rev Drug
Discov. 13:497–512. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lin L, Hutzen B, Lee HF, Peng Z, Wang W,
Zhao C, Lin HJ, Sun D, Li PK, Li C, et al: Evaluation of STAT3
signaling in ALDH+ and ALDH+/CD44+/CD24- subpopulations of breast
cancer cells. PLoS One. 8:e828212013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yang J, Liao D, Chen C, Liu Y, Chuang TH,
Xiang R, Markowitz D, Reisfeld RA and Luo Y: Tumor-associated
macrophages regulate murine breast cancer stem cells through a
novel paracrine EGFR/Stat3/Sox-2 signaling pathway. Stem Cells.
31:248–258. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Li Y, Rogoff HA, Keates S, Gao Y,
Murikipudi S, Mikule K, Leggett D, Li W, Pardee AB and Li CJ:
Suppression of cancer relapse and metastasis by inhibiting cancer
stemness. Proc Natl Acad Sci USA. 112:1839–1844. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chesnokov V, Gong B, Sun C and Itakura K:
Anti-cancer activity of glucosamine through inhibition of N-linked
glycosylation. Cancer Cell Int. 14:452014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Contessa JN, Bhojani MS, Freeze HH,
Rehemtulla A and Lawrence TS: Inhibition of N-linked glycosylation
disrupts receptor tyrosine kinase signaling in tumor cells. Cancer
Res. 68:3803–3809. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Dalirfardouei R, Karimi G and Jamialahmadi
K: Molecular mechanisms and biomedical applications of glucosamine
as a potential multifunctional therapeutic agent. Life Sci.
152:21–29. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Shiraishi A, Tachi K, Essid N, Tsuboi I,
Nagano M, Kato T, Yamashita T, Bando H, Hara H and Ohneda O:
Hypoxia promotes the phenotypic change of aldehyde dehydrogenase
activity of breast cancer stem cells. Cancer Sci. 108:362–372.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tachi K, Shiraishi A, Bando H, Yamashita
T, Tsuboi I, Kato T, Hara H and Ohneda O: FOXA1 expression affects
the proliferation activity of luminal breast cancer stem cell
populations. Cancer Sci. 107:281–289. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Fang DD, Kim YJ, Lee CN, Aggarwal S,
McKinnon K, Mesmer D, Norton J, Birse CE, He T, Ruben SM and Moore
PA: Expansion of CD133(+) colon cancer cultures retaining stem cell
properties to enable cancer stem cell target discovery. Br J
Cancer. 102:1265–1275. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wanandi SI, Yustisia I, Neolaka GMG and
Jusman SWA: Impact of extracellular alkalinization on the survival
of human CD24-/CD44+ breast cancer stem cells associated with
cellular metabolic shifts. Braz J Med Biol Res. 50:e65382017.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tian J, Hachim MY, Hachim IY, Dai M, Lo C,
Raffa FA, Ali S and Lebrun JJ: Cyclooxygenase-2 regulates
TGFβ-induced cancer stemness in triple-negative breast cancer. Sci
Rep. 7:402582017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Bradford MM: A rapid and sensitive method
for the quantitation of microgram quantities of protein utilizing
the principle of protein-dye binding. Anal Biochem. 72:248–254.
1976. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Takahashi K and Yamanaka S: Induction of
pluripotent stem cells from mouse embryonic and adult fibroblast
cultures by defined factors. Cell. 126:663–676. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Holohan C, Van Schaeybroeck S, Longley DB
and Johnston PG: Cancer drug resistance: An evolving paradigm. Nat
Rev Cancer. 13:714–726. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ying J, Tsujii M, Kondo J, Hayashi Y, Kato
M, Akasaka T, Inoue T, Shiraishi E, Inoue T, Hiyama S, et al: The
effectiveness of an anti-human IL-6 receptor monoclonal antibody
combined with chemotherapy to target colon cancer stem-like cells.
Int J Oncol. 46:1551–1559. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Maley CC, Aktipis A, Graham TA, Sottoriva
A, Boddy AM, Janiszewska M, Silva AS, Gerlinger M, Yuan Y, Pienta
KJ, et al: Classifying the evolutionary and ecological features of
neoplasms. Nat Rev Cancer. 17:605–619. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Shibue T and Weinberg RA: EMT, CSCs, and
drug resistance: The mechanistic link and clinical implications.
Nat Rev Clin Oncol. 14:611–629. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wojtowicz K, Januchowski R, Nowicki M and
Zabel M: Inhibition of protein glycosylation reverses the MDR
phenotype of cancer cell lines. Biomed Pharmacother. 74:49–56.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wanandi SI, Ningsih SS, Asikin H, Hosea R
and Neolaka GMG: Metabolic interplay between tumor cells and
cancer-associated fibroblasts (CAFs) under hypoxia versus normoxia.
Malays J Med Sci. 25:7–16. 2018.
|
|
33
|
Jang BC, Sung SH, Park JG, Park JW, Bae
JH, Shin DH, Park GY, Han SB and Suh SI: Glucosamine hydrochloride
specifically inhibits COX-2 by preventing COX-2 N-glycosylation and
by increasing COX-2 protein turnover in a proteasome-dependent
manner. J Biol Chem. 282:27622–27632. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Quastel JH and Cantero A: Inhibition of
tumour growth by D-glucosamine. Nature. 171:252–254. 1953.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
He Y, Zhu Q, Chen M, Huang Q, Wang W, Li
Q, Huang Y and Di W: The changing 50% inhibitory concentration
(IC50) of cisplatin: A pilot study on the artifacts of
the MTT assay and the precise measurement of density-dependent
chemoresistance in ovarian cancer. Oncotarget. 7:70803–70821. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yu H, Lee H, Herrmann A, Buettner R and
Jove R: Revisiting STAT3 signalling in cancer: New and unexpected
biological functions. Nat Rev Cancer. 14:736–746. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Song KH, Kang JH, Woo JK, Nam JS, Min HY,
Lee HY, Kim SY and Oh SH: The novel oIGF-IR/Akt-dependent
anticancer activities of glucosamine. BMC Cancer. 14:312014.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Weimer S, Priebs J, Kuhlow D, Groth M,
Priebe S, Mansfeld J, Merry TL, Dubuis S, Laube B, Pfeiffer AF, et
al: D-Glucosamine supplementation extends life span of nematodes
and of ageing mice. Nat Commun. 5:35632014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Pastò A, Bellio C, Pilotto G, Ciminale V,
Silic-Benussi M, Guzzo G, Rasola A, Frasson C, Nardo G, Zulato E,
et al: Cancer stem cells from epithelial ovarian cancer patients
privilege oxidative phosphorylation, and resist glucose
deprivation. Oncotarget. 5:4305–4319. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Proffitt KD, Madan B, Ke Z, Pendharkar V,
Ding L, Lee MA, Hannoush RN and Virshup DM: Pharmacological
inhibition of the Wnt acyltransferase PORCN prevents growth of
WNT-driven mammary cancer. Cancer Res. 73:502–507. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Chaffer CL, Brueckmann I, Scheel C,
Kaestli AJ, Wiggins PA, Rodrigues LO, Brooks M, Reinhardt F, Su Y,
Polyak K, et al: Normal and neoplastic nonstem cells can
spontaneously convert to a stem-like state. Proc Natl Acad Sci USA.
108:7950–7955. 2011. View Article : Google Scholar : PubMed/NCBI
|