Introduction
Colorectal cancer (CRC) is the third most common
malignancy in the world (1). Numerous
causes lead to the occurrence of CRC, including the mutation or
alteration of expression of multiple genes, including
proto-oncogenes and tumor suppressor genes. Whether the oncogenes
controlling cell proliferation are highly expressed or the tumor
suppressor genes are deleted or mutated, it can lead to the
occurrence and development of CRC (2). However, the underlying molecular
mechanism causing tumor initiation in CRC remains elusive.
Long non-coding RNAs (lncRNAs) are a class of
non-protein coding transcripts >200 nucleotides in length
(3). Evidence indicates that lncRNAs
play critical roles in the cancer development by regulating the
proliferation, metastasis, drug resistance, and apoptosis of cancer
cells (4–7). The lncRNA taurine up-regulated gene 1
(TUG1) was originally identified as a transcript that is
upregulated by taurine, which is highly conserved in mammals and
was originally detected in a genomic screen in taurine-treated
mouse retinal cells (8). Evidence has
revealed that TUG1 is dysregulated in several cancer types
(9), including bladder carcinoma
(10–12), colon cancer (13,14), lung
cancer (15), osteosarcoma (16), B-cell neoplasms (17) and esophageal squamous cell carcinoma
(18). One recent study has
demonstrated that, double-negative feedback loop between TUG1 and
microRNA-145 (miR-145) promotes epithelial-to-mesenchymal
transition and radioresistance in human bladder cancer cells
(12). The present study aimed to
investigate the biological role and clinical significance of TUG1
in the progression of CRC, providing targets for improving the
therapeutic efficacy of CRC.
Materials and methods
Patient selection
The present study was a case-control pilot study
developed at Nantong Hospital of Traditional Chinese Medicine
(Nantong, China) with the analysis of 90 patients with CRC (38
female and 52 male cases; median age, 62 years old; age range, 48
to 79 years old), and 30 healthy donors (13 female and 17 male
cases; median age, 61 years old; age range, 45 to 72 years old)
were used as the control. Of the specimens, 63 were primary tumor
(stage I and II) and 27 advanced tumor (stage III and IV) CRC. The
protocols for tumor sample collection were approved by the
Institutional Ethics Committee at Nantong Hospital of Traditional
Chinese Medicine. Each patient provided written informed consent
for inclusion in the present study.
Cell culture and lentivirus
infection
SW620 and LoVo cell lines were purchased from the
American Type Culture Collection (Manassas, VA, USA). SW620 cell
lines were cultured in L-15 medium (Gibco; Thermo Fisher
Scientific, Inc., Waltham, MA, USA) and LoVo cell lines were
cultured in F12K medium (Gibco; Thermo Fisher Scientific, Inc.).
All mediums were supplemented with 10% fetal bovine serum (FBS;
Gibco; Thermo Fisher Scientific, Inc.), 100 U/ml penicillin (Gibco;
Thermo Fisher Scientific, Inc.) and 100 µg/ml streptomycin (Gibco;
Thermo Fisher Scientific, Inc.). All cells were cultured at 37°C in
a humidified atmosphere containing 5% CO2. For the
knockdown of TUG1, lentiviral vectors (2E+8 TU/ml) harboring TUG1
short hairpin RNAs (shRNAs) were constructed (Shanghai GeneChem
Co., Ltd., Shanghai, China). The sequences were as follow: shRNA1,
5-GTCTGCATTGAGGATATAG-3; shRNA2, 5-GCCAAATAACTGAAGCTAT-3 and
shRNA3, 5-GTACGTGTCTTGGAAGTCT-3. The negative targeted shRNA was a
scrambled sequence: 5-CCTCTAGGTAAGCATAATTTT-3. Lentiviral particles
were produced by co-transfecting the expression vector
Lenti-KD-shRNA/TUG1 or Lenti-KD-shRNA/NT (Shanghai GeneChem Co.,
Ltd., Shanghai, China) with viral particle packaging helper vector
into 293T cells using Lipofectamine 3000 (Thermo Fisher Scientific,
Inc.). Human 293T cells (American Type Culture Collection) were
cultured in Dulbecco's modified Eagle's medium supplemented with
10% FBS, 100 U/ml penicillin and 100 g/ml streptomycin at 37°C in a
humidified atmosphere containing 5% CO2. The titers of
the viral particles containing Lenti-KD-shRNA/TUG1 and
Lenti-KD-shRNA/NT were determined by performing 10-fold serial
dilutions in Eppendorf tubes for 8 consecutive dilutions. The
dilution method was as follows: A total of 8 EP tubes (1.5 ml) were
prepared for each lentivirus, and 90 µl L-15 medium was added to
each tube. A total of 10 µl lentivirus stock solution was added to
the first tube, mixed, and of this solution 10 µl was then pipetted
into the second tube and mixed again, until a final dilution of
10×10−6. SW620 and LoVo cells were infected with the
aforementioned packaged lentivirus. After 72 h, the efficiencies of
knockdown were determined by reverse transcription-quantitative
polymerase chain reaction (RT-qPCR).
RT-qPCR
Total RNAs were extracted from cells using RNAiso
Plus reagent (Takara Biotechnology Co., Ltd., Dalian, China). The
cDNA was synthesized using PrimeScript™ RT reagent kit with gDNA
Eraser kit (Takara Biotechnology Co., Ltd.). The mRNA level was
determined using SYBR® Premix Ex Taq™ II (Takara
Biotechnology Co., Ltd.) by ABI 7500 real-time PCR system (Applied
Biosystems; Thermo Fisher Scientific, Inc.). GAPDH was used as an
internal control. The thermocycling conditions used were as
follows: 95°C for 5 min, followed by 40 two-step cycles (95°C for
20 sec and 60°C for 30 sec). Relative mRNA levels were calculated
using the 2−ΔΔCq method (19). The sequences of primers are as
follows: TUG1 forward, TAGCAGTTCCCCAATCCTTG and reverse,
CACAAATTCCCATCATTCC; and GAPDH forward, CCACCCATGGCAAATTCCATGGCA
and reverse, TCTAGACGGCAGGTCAGGTCCACC.
Cell proliferation and survival
assays
Cell proliferation was determined using an MTT assay
kit (cat no. C0009; Beyotime Institute of Biotechnology, Haimen,
China) according to manufacturer's instructions. Once the cells
were cultured for 24, 48, 72 and 96 h, MTT solution (0.5%,
dissolved in dimethyl sulfoxide) was added into each well and the
cells were incubated for another 4 h at 37°C, followed by
absorbance detection. The absorbance in each well was measured
using a microplate reader at a wavelength of 490 nm (Bio-Rad
Laboratories, Inc., Hercules, CA, USA).
Tumorigenesis assay
The animal experiments were performed in accordance
with the guidelines by the U.S. National Institute of Health Guide
for the Care and Use of Laboratory Animals (20). The protocol was approved by the Animal
Care and Ethics Committee of Nantong Hospital of Traditional
Chinese Medicine. Scramble shRNA, or TUG1 shRNA-infected SW620 or
LoVo cells (~5×106) were injected subcutaneously in the
female BALB/c nude mice (4–6 weeks old, ~20 g) obtained from
Shanghai SLAC Laboratory Animal Co., Ltd. (Shanghai, China) and
maintained in pathogen-free conditions of temperature (22 ± 2°C),
humidity (55 ± 5%) and a 12 h/12 h light-dark cycle with free
access to food and water. The length (L) and width (W) of each
tumor was measured with calipers and the volume was calculated
using the equation: V=(L × W2) × 0.5 for 42 days after
the injection of cancer cells. Mice were sacrificed after 42 days
using cervical dislocation following CO2 inhalation.
Immunofluorescence and
immunohistochemical staining
The subcutaneous transplantation tumor tissues or
cells (SW620 and LoVo) were fixed with 4% paraformaldehyde for 20
min at room temperature, permeabilized by 0.1% Triton X-100 and
washed with PBS three times followed by blocking with 10% normal
goat serum plus 1% bovine serum albumin for 45 min at room
temperature and incubated with rabbit anti-β-catenin (1:100; cat
no. #9562; Cell Signaling Technology Inc., Danvers, MA, USA) at 4°C
overnight. After washing three times with PBS, the tissue slices
were further incubated with Cy3-conjugated goat anti-rabbit (cat
no. A0516; Beyotime Institute of Biotechnology) or HRP-conjugated
goat anti-rabbit secondary antibodies (cat no. A0208; Beyotime
Institute of Biotechnology) for 1 h at 37°C. Subsequently, the
slides were cover slipped with mounting medium (Dako; Agilent
Technologies, Inc., Santa Clara, CA, USA) containing DAPI to
counterstain the nuclei and imaged with a fluorescence or bright
field microscope (at a magnification of ×100 for immunofluorescence
or ×200 for immunohistochemical staining).
T-cell factor/lymphoid enhancer factor
(TCF/LEF) promoter activity analysis
To test the LEF/TCF promoter activity, SW620 or LoVo
cells were co-transfected with the recombinant plasmid
pGL3-basic-LEF/TCF promoter with a control positive plasmid
pRL-SV40, and the TCF/LEF promoter activity analysis was performed
according to the protocol of Ji et al (21). The promoter activity was analyzed
using a commercial dual-luciferase assay kit (Beyotime Institute of
Biotechnology, China) according to the manufacturer's protocol.
Western blot analysis
The protein expression levels of β-catenin were
analyzed with western blot. Following transfection, cells were
harvested in Radioimmunoprecipitation Assay lysis buffer (Beyotime
Institute of Biotechnology, Shanghai, China) supplemented with
protease inhibitor cocktail (Sigma-Aldrich; Merck KGaA, Darmstadt,
Germany). A total of 40 µg protein from each sample concentration
determined using the BCA protein assay reagent kit (cat no. 23225;
Pierce; Thermo Fisher Scientific, Inc.) and molecular weight
determined by the Prestained Protein Molecular Weight Marker (cat
no. 26612; Fermentas; Thermo Fisher Scientific, Inc., Pittsburgh,
PA, USA, β-catenin: 92kD, β-actin: 42kD] was resolved in 10%
SDS-PAGE and transferred to a polyvinylidene difluoride membrane.
Membranes were blocked with 5% non-fat dry milk in TBS-0.05%
Tween-20 (TBST) buffer for 2 h at room temperature and incubated
with primary antibodies rabbit anti-β-catenin (cat no. #9562; Cell
Signaling Technology, Inc.) and rabbit anti-β-actin (cat no. #4970;
Cell Signaling Technology, Inc.), each diluted to 1:1,000] at 4°C
overnight. After washing 3 times with TBST buffer, the membranes
were incubated HRP-conjugated goat anti-rabbit secondary antibody
(1:5,000; cat no. A0516; Beyotime Institute of Biotechnology) for 1
h at room temperature. Next, the membranes were washed, developed
using Millipore Western Blot chemiluminescence HRP substrate (cat
no. WBKLS0010; Merck KGaA), and images were captured a using
DMI3000B inverted microscope (Leica Microsystems GmbH, Wetzlar,
Germany).
Statistical analysis
Data are presented as mean ± standard error of the
mean of at least three independent experiments. Significance of the
mean between two groups was determined using an unpaired Student's
t-test. Differences at different time points between groups were
evaluated using a one-way analysis of variance followed by a
Dunnett's test. P<0.05 was considered to indicate a
statistically significant difference. Analyses were performed using
SPSS 20.0 software (IBM Corp., Armonk, NY, USA).
Results
Association between the TUG1
expression and clinicopathological characteristics in CRC
Significant upregulation of TUG1 expression was
observed in CRC tissues compared with normal controls (Fig. 1A). Expression of TUG1 expression was
significantly higher in stages III/IV than in stage I/II of CRC,
indicating a positive association between the advanced stage of CRC
and TUG1 expression (Fig. 1B).
Correspondingly, the overall survival (OS) and disease-free
survival (DFS) times were low in CRC patients with high TUG1
expression levels when compared with those expressing low levels of
TUG1 (Fig. 1C and D). These results
indicated that TUG1 might contribute to CRC development.
TUG1 increased the proliferation
ability of CRC cells
To study the contribution of TUG1 to the
proliferation of CRC, the expression of TUG1 was examined in the
SW620 and LoVo cell lines. As depicted in Fig. 2A, the expression level of TUG1 was
significant high in SW620 and LoVo cells in compared with other CRC
cells. To assess the contribution of TUG1 to cellular
proliferation, TUG1 expression levels were successfully knocked
down in SW620 and LoVo cells (Fig.
2B), and it was found that the proliferation rate was
significantly decreased in the two cell lines (Fig. 2C and D). These data indicated that
TUG1 expression contributes to the proliferation of CRC cells.
TUG1 enhanced the activation of the
Wnt/β-catenin signaling pathway
The Wnt/β-catenin signaling pathway is involved in
the promotion of proliferation of CRC cells. To investigate the
mechanism underlying the regulation by TUG1, the expression and
location of β-catenin was examined in CRC cells treated with TUG1
shRNA by immunofluorescence staining. As shown in Fig. 3A and B, TUG1 shRNA significantly
inhibited the expression of β-catenin protein in the nucleus of
SW620 and LoVo cells. Furthermore, the western blot analysis also
revealed that the levels of β-catenin were downregulated in the
nuclear compartment when the CRC cells were treated with TUG1 shRNA
(Fig. 3C and D). However,
transfection with TUG1 shRNA did not affect the total expression of
β-catenin (Fig. 3C and D).
Accordingly, the TCF/LEF reporter assay also demonstrated that the
Wnt/β-catenin signaling was significantly inhibited by TUG1 shRNA
(Fig. 3E and F).
TUG1 promoted the tumorigenicity of
CRC cells
To confirm the aforementioned effects of TUG1 on CRC
cells in vivo, the effect of knockdown of TUG1 on the
tumorigenicity of SW620 and LoVo cells was analyzed using the
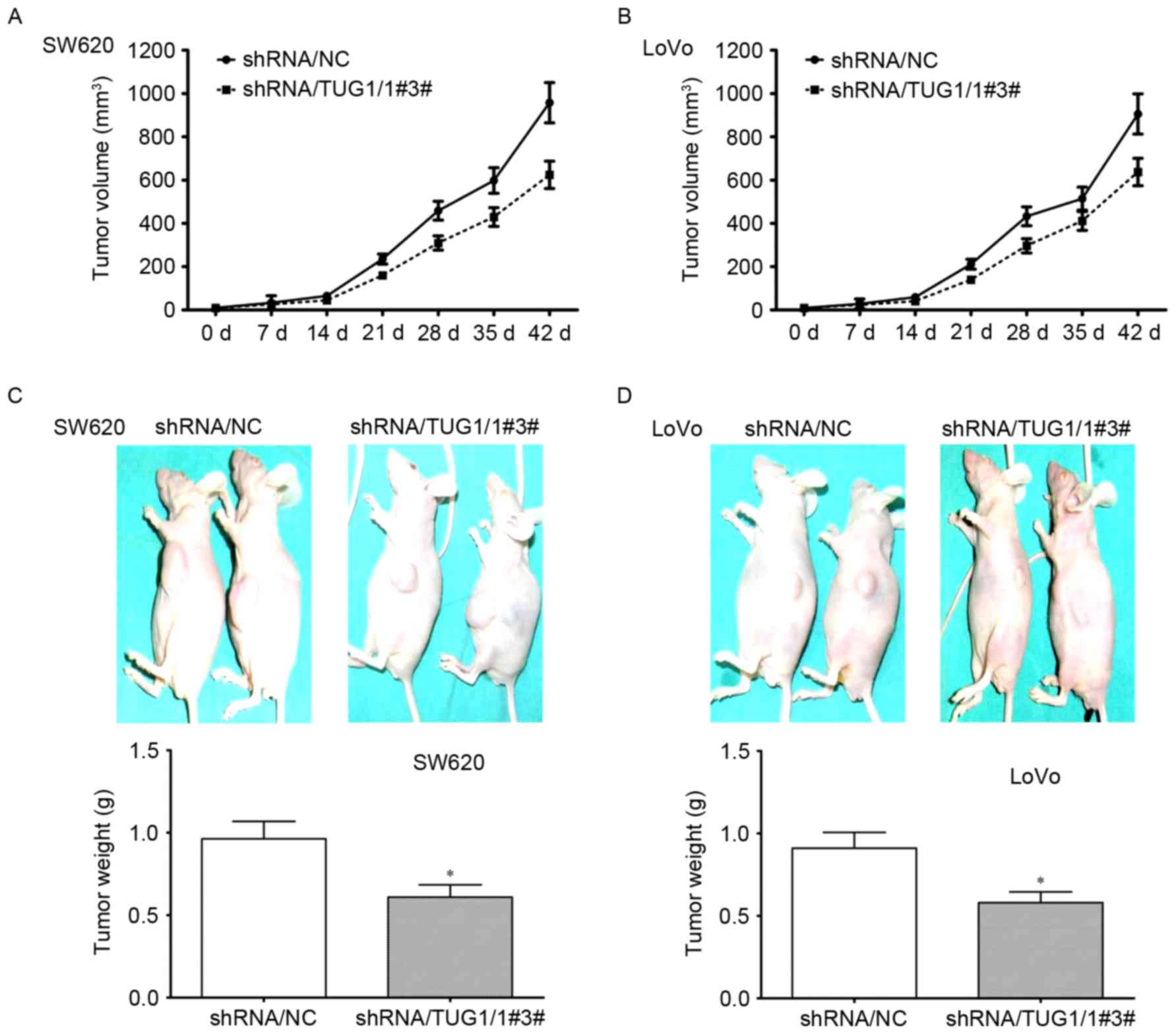
subcutaneous xenograft mouse model. As depicted in Fig. 4A and B, the size of tumors was greatly
reduced at each time point in mice injected with CRC cells
transfected with TUG1 shRNA. At day 42, the tumors were taken out
and measured. The size and weight of tumors were significantly
decreased by knockdown of TUG1 in SW620 and LoVo cells (Fig. 4C and D). These results indicated that
TUG1 could promote the tumorigenicity of CRC cells.
TUG1 promoted the nuclear localization
of β-catenin in patient tissues and the xenograft model
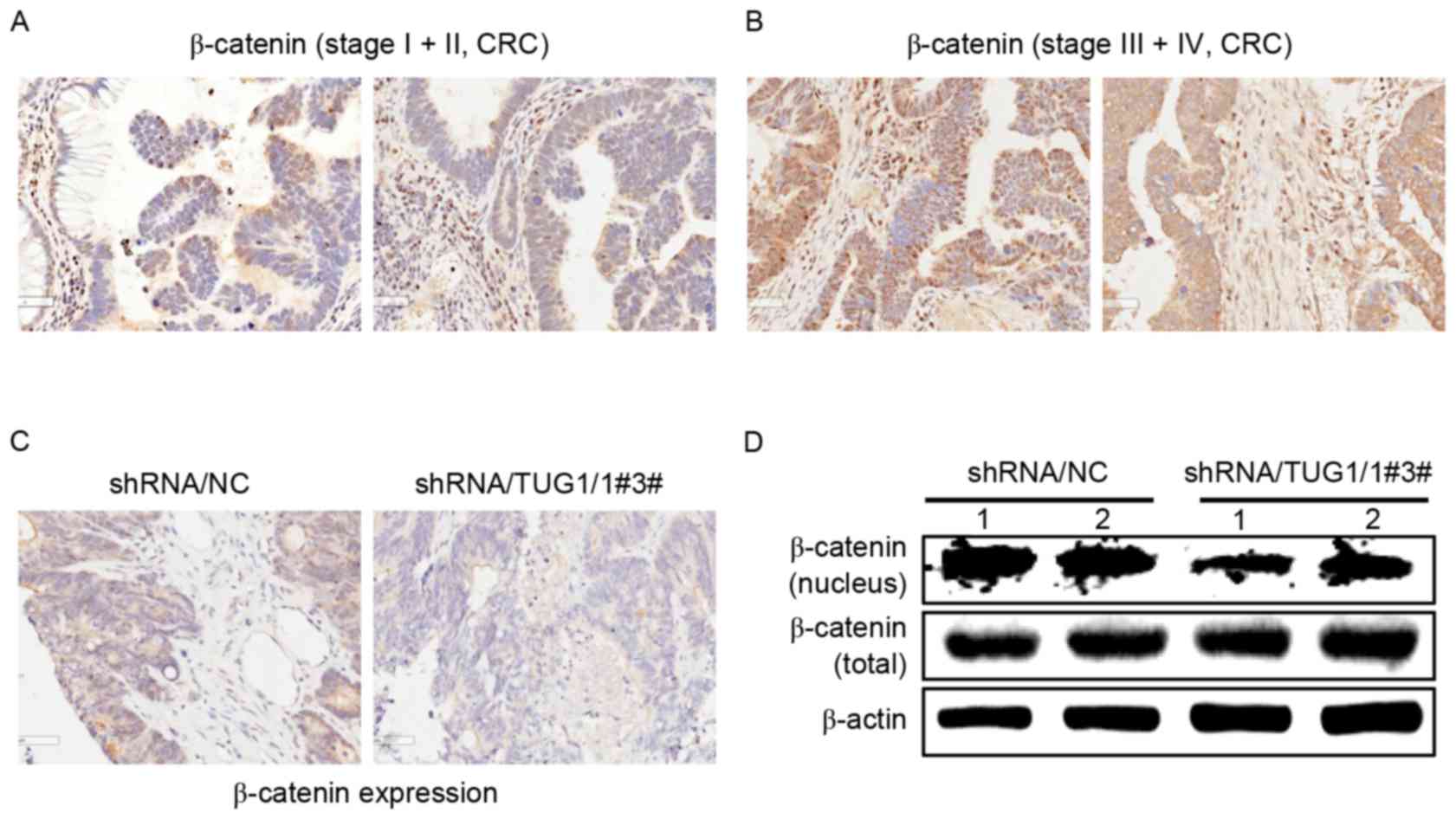
Since TUG1 could regulate the nuclear localization
of β-catenin in CRC cells, β-catenin expression was examined in
different stages of CRC patients. Consistent with the results of
the aforementioned in vitro experiments, immunohistochemical
staining revealed that β-catenin expression was higher in the
nuclei of patients with stage III and IV CRC tumor tissues than
that in stage I and II CRC patients (Fig.
5A and B).
Immunohistochemical staining and western blot
analysis revealed that there was lower β-catenin expression in the
nuclei from the excised tumors in mice generated by CRC cells
transfected with TUG1 shRNA (Fig. 5C and
D). These data indicated that TUG1 promotes the nuclear
translocation of β-catenin in advanced CRC patients (stage III and
IV) and in the in vivo mice model.
Discussion
CRC, as one of the most common malignant tumors in
the world, is typically classified as cancer of the proximal colon,
distal colon or rectum (1). A
considerable number of patients with CRC are diagnosed at advanced
stages and have a poor prognosis (1).
The treatment modalities for CRC include surgical resection,
chemotherapy, immunotherapy and other alternative methods such as
traditional Chinese medicine (22).
Numerous causes can bring about the occurrence of CRC, including
the mutation or altered expression of proto-oncogenes and tumor
suppressor genes. However, to the best of our knowledge, the
underlying molecular mechanism leading to tumor initiation in CRC
remains unclear.
Evidence has demonstrated that lncRNAs serve notable
roles in different aspects of cancer biology and can contribute to
the development of different cancer types (9). The lncRNA TUG1 was originally
characterized as a transcript whose expression was upregulated by
taurine, and was later observed to be overexpressed in a number of
cancer tissues, including bladder carcinoma (10–12), colon
cancer (13,14), lung cancer (15), osteosarcoma (16), B-cell neoplasms (17), and esophageal squamous cell carcinoma
(18). Tan et al (12) demonstrated that TUG1 promoted bladder
cancer cell metastasis and radioresistance via negatively
regulating miR-145 expression by acting as a miRNA sponge. In
esophageal carcinoma, TUG1 promoted proliferation and migration
in vitro (18–22). Gezer et al (23), found that TUG1 were highly enriched in
secreted exosomes of HeLa and MCF-7 cells. These data indicated
that TUG1 might promote cancer progression. Nevertheless, the
function and mechanism of TUG1 in CRC remain unknown. The present
study demonstrated that the expression of TUG1 was upregulated in
the tumor tissues of advanced-stage patients with CRC, and was
positively associated with the mortality of CRC patients. In
addition, high TUG1 expression was also detected in several CRC
cells, including SW620 and LoVo cells. Together, these data
indicated that TUG1 might serve notable roles in the development of
CRC, although the detailed mechanism was unclear.
β-catenin, as the key transcriptional activation
factor of the Wnt/β-catenin signal pathway, can, when activated,
translocate to the nucleus from the cytoplasm in coordination with
other transcription factors, including TCF, LEF, Pygo, and B-cell
CLL/lymphoma 9 to activate its target genes, including cyclin D1,
c-Myc, cluster of differentiation 44, and matrix
metalloproteinase-7 (MMP-7), which serve notable roles in the
initiation and development of tumors (24–27). The
current study demonstrated that the shRNA-mediated knockdown of
TUG1 evidently reduced the proliferation abilities of CRC cells.
Knockdown of TUG1 inhibited the translocation of β-catenin from the
cytoplasm to the nucleus, resulting in decreases in the expression
of c-Myc and MMP-7, whereas overexpression of TUG1 brought about
the contrary. The TCF/LEF family is a group of transcription
factors that is involved in the Wnt/β-catenin signaling pathway,
which bind to DNA through a high-mobility group domain and recruit
the co-activator β-catenin to enhancer elements of target genes
(28). By using LEF/TCF promoter
dual-luciferase reporter construct, it was demonstrated that
knockdown of TUG1 downregulated the promoter activity of LEF/TCF,
whereas it was enhanced by overexpression of TUG1. Together, these
results indicated that, TUG1 might promote the proliferation of CRC
cells by regulating the Wnt/β-catenin signaling and downstream gene
expression.
The in vivo results presented in the current
study also indicated that silence of TUG1 could inhibit the CRC
tumor growth in the subcutaneously transplanted tumor model, and
lower nuclear β-catenin expression was detected in the excised
tumors generated by TUG1 shRNA cells. Furthermore, β-catenin was
also found to be overexpressed in the nuclei of tumor tissues from
patients with advanced (stage III and IV) CRC compared with that of
stage I and II CRC patients. The results of the present study
corresponded with previous studies into CRC (29,30). The
aforementioned data indicated that TUG1 serves a notable in an
in vivo mouse model, in vitro and in advanced CRC
patients.
In conclusion, the results of the present study
revealed that TUG1 altered the nuclear localization of β-catenin,
resulting in lowered Wnt/β-catenin signaling and the eventual
inhibition of proliferation. These results indicated that
prevention and treatment of CRC could be performed by targeting the
TUG1 and Wnt/β-Catenin signaling pathway.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
JFY was responsible for the study design and the
acquisition of data, CYG performed data analysis, CHX and HZY
performed the functional experiments. ZMW and HYC assisted in
designing the experiments and interpret the data. WBL and JFY
performed project design and manuscript revisions.
Ethics approval and consent to
participate
The protocols for tumor sample collection were
approved by the Institutional Ethics Committee at Nantong Hospital
of Traditional Chinese Medicine. Each patient provided written
informed consent for inclusion in the present study.
Patient consent for publication
All the patients provided consent for
publication.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2017. CA Cancer J Clin. 67:7–30. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Watson AJ and Collins PD: Colon cancer: A
civilization disorder. Dig Dis. 29:222–228. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wilusz JE: Long noncoding RNAs: Re-writing
dogmas of RNA processing and stability. Biochim Biophys Acta.
1859:128–138. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Sun M, Nie F, Wang Y, Zhang Z, Hou J, He
D, Xie M, Xu L, De W, Wang Z and Wang J: LncRNA HOXA11-AS Promotes
Proliferation and Invasion of Gastric Cancer by Scaffolding the
Chromatin Modification Factors PRC2, LSD1, and DNMT1. Cancer Res.
76:1–6310. 2016. View Article : Google Scholar
|
|
5
|
Okugawa Y, Toiyama Y, Hur K, Toden S,
Saigusa S, Tanaka K, Inoue Y, Mohri Y, Kusunoki M, Boland CR and
Goel A: Metastasis-associated long non-coding RNA drives gastric
cancer development and promotes peritoneal metastasis.
Carcinogenesis. 35:1–2739. 2014. View Article : Google Scholar
|
|
6
|
Xia H and Hui KM: Mechanism of cancer drug
resistance and the involvement of noncoding RNAs. Curr Med Chem.
21:3029–3041. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Luo HL, Huang MD, Guo JN, Fan RH, Xia XT,
He JD and Chen XF: AFAP1-AS1 is upregulated and promotes esophageal
squamous cell carcinoma cell proliferation and inhibits cell
apoptosis. Cancer Med. 5:1–2885. 2016. View
Article : Google Scholar
|
|
8
|
Xue M, Chen W and Li X: Urothelial cancer
associated 1: A long noncoding RNA with a crucial role in cancer. J
Cancer Res Clin Oncol. 142:1407–1419. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Young TL, Matsuda T and Cepko CL: The
noncoding RNA taurine upregulated gene 1 is required for
differentiation of the murine retina. Curr Biol. 15:1–512. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang XS, Zhang Z, Wang HC, Cai JL, Xu QW,
Li MQ, Chen YC, Qian XP, Lu TJ, Yu LZ, et al: Rapid identification
of TUG1 as a very sensitive and specific unique marker for human
bladder carcinoma. Clin Cancer Res. 12:4851–4858. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Fan Y, Shen B, Tan M, Mu X, Qin Y, Zhang F
and Liu Y: Long non-coding RNA TUG1 increases chemoresistance of
bladder cancer cells by regulating Wnt signaling. FEBS J.
281:1750–1758. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tan J, Qiu K, Li M and Liang Y:
Double-negative feedback loop between long non-coding RNA TUG1 and
miR-145 promotes epithelial to mesenchymal transition and
radioresistance in human bladder cancer cells. FEBS Lett.
589:3175–3181. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tuo YL, Li XM and Luo J: Long noncoding
RNA TUG1 modulates colorectal cancer cell growth and apoptosis
through decreasing tumor suppressive miR-143. Eur Rev Med Pharmacol
Sci. 19:3403–3411. 2015.PubMed/NCBI
|
|
14
|
Huang J, Zhou N, Watabe K, Lu Z, Wu F, Xu
M and Mo YY: Long non-coding RNA TUG1 promotes colorectal tumor
growth by suppression of p27 (Kip1). Cell Death Dis. 5:e10082014.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhang EB, Yin DD, Sun M, Kong R, Liu XH,
You LH, Han L, Xia R, Wang KM, Yang JS, et al: P53-regulated long
non-coding RNA TUG1 affects cell proliferation in human non-small
cell lung cancer, partly through epigenetically regulating HOXB7
expression. Cell Death Dis. 5:e12432014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang Q, Geng PL, Yin P, Wang XL, Jia JP
and Yao J: Down-regulation of long non-coding RNA TUG1 inhibits
osteosarcoma cell proliferation and promotes apoptosis. Asian Pac J
Cancer Prev. 14:1–2315. 2013.PubMed/NCBI
|
|
17
|
Isin M, Ozgur E, Cetin G, Erten N, Aktan
M, Gezer U and Dalay N: Investigation of circulating lncRNAs in
B-cell neoplasms. Clin Chim Acta. 431:1–259. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Xu Y, Wang J, Qiu M and Xu L, Li M, Jiang
F, Yin R and Xu L: Upregulation of the long noncoding RNA TUG1
promotes proliferation and migration of esophageal squamous cell
carcinoma. Tumour Biol. 36:1–1651. 2015.
|
|
19
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:1–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
National Research Council (US) Committee
for the Update of the Guide for the Care and Use of Laboratory
Animals, . Guide for the Care and Use of Laboratory Animals. 8th.
Washington, DC: National Academies Press (US); 2011, PubMed/NCBI
|
|
21
|
Ji Q, Liu X, Fu X, Zhang L, Sui H, Zhou L,
Sun J, Cai J, Qin J, Ren J and Li Q: Resveratrol Inhibits invasion
and metastasis of colorectal cancer cells via MALAT1 mediated
Wnt/β-catenin signal pathway. PLoS One. 8:e787002013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Venook AP: Advances in Adjuvant Therapy
for Colon Cancer: P value or Practical Value. J Clin Oncol.
36:1–1462. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Gezer U, Ozgur E, Cetinkaya M, Isin M and
Dalay N: Long noncoding RNAs with low expression levels in cells
are enriched in secreted exosomes. Cell Biol Int. 38:1–1079.
2014.PubMed/NCBI
|
|
24
|
Li ZR, Wu YF, Ma CY, Nie SD, Mao XH and
Shi YZ: Down-regulation of c-Myc expression inhibits the invasion
of bile duct carcinoma cells. Cell Biol Int. 35:799–802. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhang J, Gill AJ, Issacs JD, Atmore B,
Johns A, Delbridge LW, Lai R and McMullen TP: The Wnt/β-catenin
pathway drives increased cyclin D1 levels in lymph node metastasis
in papillary thyroid cancer. Hum Pathol. 43:1044–1050. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Villar J, Cabrera NE, Valladares F, Casula
M, Flores C, Blanch L, Quilez ME, Santana-Rodríguez N, Kacmarek RM
and Slutsky AS: Activation of the Wnt/β-Catenin signaling pathway
by mechanical ventilation is associated with ventilator-induced
pulmonary fibrosis in healthy lungs. PLoS One. 6:e239142011.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zeilstra J, Joosten SP, Dokter M, Verwiel
E, Spaargaren M and Pals ST: Deletion of the WNT target and cancer
stem cell marker CD44 in Apc(Min/+) mice attenuates intestinal
tumorigenesis. Cancer Res. 68:3655–3661. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Brantjes H, Barker N, van Es J and Clevers
H: TCF: Lady Justice casting the final verdict on the outcome of
Wnt signalling. Biol Chem. 383:1–261. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Brabletz T, Jung A, Reu S, Porzner M,
Hlubek F, Kunz-Schughart LA, Knuechel R and Kirchner T: Variable
betacatenin expression in colorectal cancers indicates tumor
progression driven by the tumor environment. Proc Natl Acad Sci
USA. 98:1–10361. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hlubek F, Brabletz T, Budczies J, Pfeiffer
S, Jung A and Kirchner T: Heterogeneous expression of
Wnt/beta-catenin target genes within colorectal cancer. Int J
Cancer. 121:1–1948. 2007. View Article : Google Scholar : PubMed/NCBI
|