Introduction
Hepatocellular carcinoma (HCC) is the seventh most
common malignant tumor and the second most frequent cause of
cancer-associated mortality worldwide in 2016 (1). Although progress has been made in the
diagnosis of HCC, the treatment and prevention of the disease and
prognosis prediction remain poor (2). At present, the classification and
prognosis evaluation of patients with HCC depend on clinical
staging systems, including Tumor-Node-Metastasis (TNM) stage,
Barcelona Clinic Liver Cancer (BCLC) stage and the Cancer of the
Liver Italian Program stage (3).
Although the clinical stage can predict the risk of tumor
recurrence to a certain extent, it rarely directly reflects the
prognosis of patients with HCC after hepatectomy. It is therefore
crucial to identify an effective prognostic molecular marker to
predict the clinical prognosis of patients with HCC.
A total of 14 subtypes of facilitative glucose
transporters (GLUTs) have been described in humans, of which role
is to transport glucose to different tissues in the body (4). Previous studies have reported that
GLUT-3 is overexpressed in numerous solid tumors, including oral
squamous cell carcinoma, laryngeal carcinoma, nonsmall cell lung
carcinoma and bladder cancer, which may be due to the rapid
proliferation of tumor cells in hypoxic condition (5–9). Since
the rate of ATP produced by glycolysis under anaerobic conditions
is significantly lower than during aerobic metabolism, high GLUTs
expression is required by tumor cells to satisfy the increased need
for glucose (4). GLUT-3 may
therefore be a potential tumor cell marker. To the best of our
knowledge, the expression of GLUT-3 in HCC and its association with
the clinicopathological characteristics of patients have not yet
been identified. In the present study, the association between
GLUT-3 expression in HCC tissues and the clinicopathological
characteristics and clinical prognosis of patients with HCC was
evaluated.
Materials and methods
Patients and tissue specimens
Formalin-fixed paraffin-embedded tissues of 275
patients with HCC who underwent surgical resection between April
2003 and December 2008 at the Shandong Provincial Hospital
Affiliated to Shandong University (Shandong, China) were included
in the training cohort. In parallel, in order to verify the
prognostic efficacy of GLUT-3 as a predictive marker in HCC, 140
formalin-fixed paraffin-embedded tissues of patients with HCC who
underwent surgery during the same period at the Sun Yat-Sen
University Cancer Center (Guangdong, China) were randomly selected
and included in the validation cohort. The inclusion criteria were
as follows: i) Child-Pugh classification (10) was A or B; ii) patients did not
receive antitumor therapy prior to surgery; iii) radical resection
was performed; iv) HCC pathology was confirmed after surgery; v) no
evidence of extrahepatic metastasis or primary cancer of other
organs; and vi) complete follow-up information was available. The
exclusion criteria were as follows: i) Patients received
preoperative antitumor therapy, including radiotherapy or
chemotherapy; ii) preoperative extrahepatic metastasis was
observed; iii) malignant tumors associated with other organs were
identified; and iv) follow-up information was missing.
In the training cohort, the median age of the
patients was 55 years (age range, 24–74 years), 38 patients were
women and 237 patients were men. In the validation cohort, the
median age of the patients was 52 years (age range, 28–72 years),
15 patients were women and 125 patients were men. Clinical baseline
and complete follow-up information were reviewed from the hospital
databases. This study was approved by the Institutional Review
Boards of Sun Yat-Sen University Cancer Center and Shandong
Provincial Hospital Affiliated to Shandong University. Written
informed consent was obtained from all patients included in this
study.
Isolation of RNA and reverse
transcription-quantitative PCR (RT-qPCR)
Total RNA was extracted from the tissue samples
using TRIzol reagent (Invitrogen; Thermo Fisher Scientific, Inc.).
The quality and quantity of RNA were assessed using the Agilent
2100 Bioanalyzer and NanoDrop ND-1000 Spectrophotometer (Agilent
Technologies, Inc.). cDNA was synthesized from immunoprecipitated
RNA using reverse transcriptase followed by second strand synthesis
to generate double-stranded cDNA using SuperScript IV Reverse
Transcriptase kit (Thermo Fisher Scientific, Inc.) under the
following conditions: 25°C for 6 min, 55°C for 20 min, and 80°C for
10 min. The qPCR was performed using SsoFast™ EvaGreen®
Supermix (Bio-Rad Laboratories, Inc.) according to the
manufacturer's protocols. The GAPDH was used as an endogenous
control, and fold changes were calculated via relative
quantification (2−ΔΔCq). Transcripts were assessed using
the following primers: GLUT-3 (forward, CAGCGAGACCCAGAGATGC;
reverse, GACCCCAGTGTTGTAGCCAA) and GAPDH (forward,
TGCACCACCAACTGCTTAGC; reverse, GGCATGGACTGTGGTCATGAG).
IHC staining
The formalin-fixed paraffin-embedded specimens were
cut into 5-µm sections and placed on polylysine-coated slides
(Sigma-Aldrich; Merck KGaA). Sections were deparaffinized in xylene
and rehydrated using a gradient series of alcohol (100% for 5 min;
90% for 5 min; 80% for 5 min; and 70% for 5 min). Antigen retrieval
was performed by heating sections in citrate buffer (pH 6.0; Dako;
Agilent Technologies, Inc.) at 95°C for 10 min. Samples were
blocked with 10% goat serum (Beijing Solarbio Science &
Technology Co., Ltd) at 37°C for 2 h and with Peroxidase-Blocking
Solution (Dako; Agilent Technologies, Inc.) at 37°C for 30 min.
Sections were incubated with the primary antibody against GLUT-3
(1:50; cat. no. ab95256; Abcam) and with an isotype-matched
immunoglobulin G (1:100; cat. no. Ab83567; Abcam) used as a
negative control at room temperature for 2 h. Immunohistochemical
staining was performed using the Dako Envision Plus system [Dako;
Agilent Technologies, Inc. Dako, EnVisio+System/HRP, Mo(DAB+),
K400611-2] according to the manufacturer's instructions
(magnification, ×400). The number of tumor cells with a strong
membrane signal for GLUT-3 was counted in ten low magnification
fields with light microscope, and expressed as a percentage of the
total number of cells. The mean percentage of immunoreactive tumor
cells was calculated and scored according to the following 5-point
scale: 0, 1, 2, 3 or 4 points for 0, 1–25, 26–50, 51–75 or 76–100%
of positively stained cells, respectively. GLUT-3 was considered to
be not expressed if the final score was 0. GLUT-3 expression was
considered to be low if the final score was 1 or 2, and high if the
final score was 3 or 4.
Statistical analysis
Statistical analyses were performed using SPSS 18.0
statistical software for Windows (SPSS, Inc.). GLUT-3 mRNA level in
different types of tissue was analyzed using ANOVA followed by
Scheffe post hoc test. χ2 or Fisher's exact tests were
used to determine the association between GLUT-3 expression levels
and the clinicopathological characteristics of patients.
Disease-free survival (DFS) time was calculated as the time between
surgical resection and the appearance of recurrence evidence at any
site or the last follow-up contact. Overall survival (OS) time was
calculated as the time between surgical resection and the time of
death or the last follow-up.
Receiver operating characteristic (ROC) curves were
used in the training cohort and the validation cohort to validate
the prognostic ability of GLUT-3 expression levels. Survival rate
was calculated using Kaplan-Meier method, and log-rank test was
used to compare differences in survival between groups. The
Kaplan-Meier method was used for univariate analysis, whereas Cox
proportional hazards regression model was used for multivariate
analysis. Variables with P<0.05 in the univariate analysis were
selected as variables for multivariate analysis. A two-tailed
P<0.05 was considered to indicate a statistically significant
difference.
Results
Clinicopathological characteristics
and expression of GLUT-3 in patients with HCC
The clinicopathological characteristics of all
patients in the two cohorts included age, sex, etiology, liver
cirrhosis, Child-Pugh classification, serum α-fetoprotein (AFP)
level, tumor size, tumor number, vascular invasion, histological
differentiation, BCLC stage and TNM stage, and are summarized in
Tables I and II.
 | Table I.Expression of GLUT-3 and its
relationship with clinicopathological characteristics of the
training cohort. |
Table I.
Expression of GLUT-3 and its
relationship with clinicopathological characteristics of the
training cohort.
|
| GLUT-3 expression
(n=275) |
|
|---|
|
|
|
|
|---|
| Characteristic | No and low
(n=193) | High (n=82) | P-value |
|---|
| Age (≥55/<55
years) | 60/133 | 29/53 | 0.489 |
| Sex (F/M) | 28/165 | 10/72 | 0.611 |
| Etiology |
|
| 0.473 |
|
Hepatitis B virus | 176 | 74 |
|
|
Hepatitis C virus | 2 | 1 |
|
|
Other | 15 | 7 |
|
| Cirrhosis |
|
| 0.487 |
|
Yes | 143 | 64 |
|
| No | 50 | 18 |
|
| Child-Pugh
classification |
|
| 0.381 |
| A | 191 | 80 |
|
| B | 2 | 2 |
|
| AFP level |
|
| 0.007 |
| ≤400
ng/ml | 115 | 34 |
|
| >400
ng/ml | 78 | 48 |
|
| Tumor size |
|
| 0.166 |
| ≤5
cm | 100 | 35 |
|
| >5
cm | 93 | 47 |
|
| Tumor number |
|
| 0.039 |
|
Single | 160 | 59 |
|
|
Multiple | 33 | 23 |
|
| Vascular
invasion |
|
| 0.097 |
|
Yes | 40 | 24 |
|
| No | 153 | 58 |
|
| Histological
differentiation |
|
| 0.028 |
|
Well | 33 | 6 |
|
|
Moderate | 125 | 54 |
|
|
Poor | 35 | 22 |
|
| TNM stage |
|
| 0.018 |
| I and
II | 153 | 54 |
|
| III and
IV | 40 | 28 |
|
| BCLC stage |
|
| 0.227 |
| 0 and
A | 154 | 60 |
|
| B and
C | 39 | 22 |
|
 | Table II.Expression of GLUT-3 and its
relationship with clinicopathological characteristics of the
validation cohort. |
Table II.
Expression of GLUT-3 and its
relationship with clinicopathological characteristics of the
validation cohort.
|
| GLUT-3 expression
(n=140) |
|
|---|
|
|
|
|
|---|
| Characteristic | No and low
(n=99) | High (n=41) | P-value |
|---|
| Age (≥55/<55
years) | 31/68 | 17/24 | 0.250 |
| Sex (F/M) | 9/90 | 6/35 | 0.335 |
| Etiology |
|
| 0.473 |
|
Hepatitis B virus | 93 | 37 |
|
|
Hepatitis C virus | 1 | 1 |
|
|
Others | 5 | 3 |
|
| Cirrhosis |
|
| 0.087 |
|
Yes | 74 | 36 |
|
| No | 25 | 5 |
|
| Child-Pugh
classification |
|
| 0.580 |
| A | 92 | 38 |
|
| B | 7 | 3 |
|
| AFP level, |
|
| 0.001 |
| ≤400
ng/ml | 63 | 14 |
|
| >400
ng/ml | 36 | 27 |
|
| Tumor size |
|
| 0.031 |
| ≤5
cm | 56 | 15 |
|
| >5
cm | 43 | 26 |
|
| Tumor number |
|
| 0.660 |
|
Single | 80 | 31 |
|
|
Multiple | 19 | 10 |
|
| Vascular
invasion |
|
| 0.022 |
|
Yes | 8 | 9 |
|
| No | 91 | 32 |
|
| Histological
differentiation |
|
| 0.004 |
|
Well | 21 | 0 |
|
|
Moderate | 60 | 26 |
|
|
Poor | 18 | 15 |
|
| TNM stage |
|
| 0.144 |
| I and
II | 79 | 28 |
|
| III and
IV | 20 | 13 |
|
| BCLC stage |
|
| 0.403 |
| 0 and
A | 81 | 31 |
|
| B and
C | 18 | 10 |
|
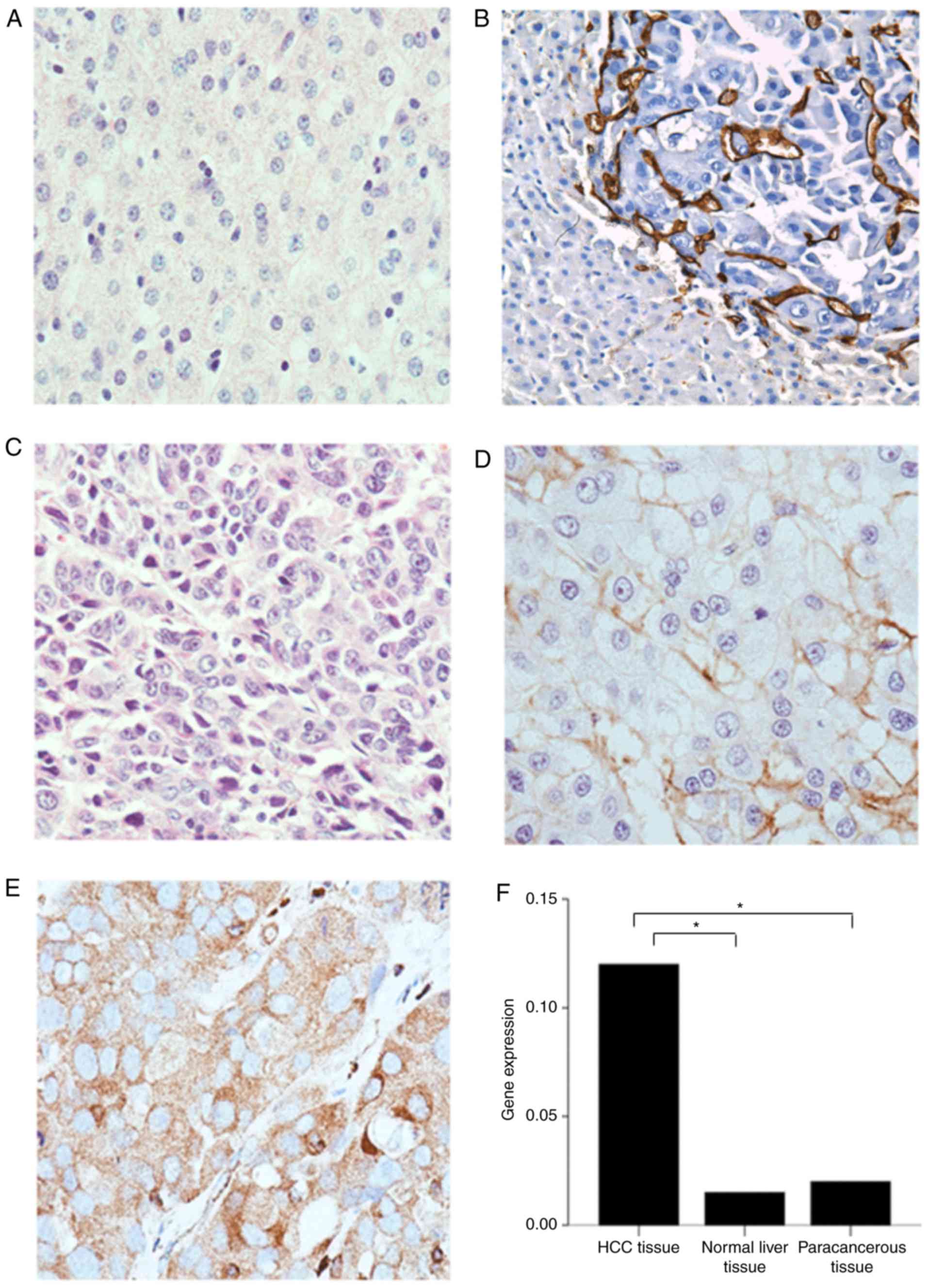
IHC was performed to investigate GLUT-3 expression.
The results demonstrated that GLUT-3 was not expressed in normal
liver (Fig. 1A) and paracancerous
tissues of patients with HCC (Fig.
1B). However, GLUT-3 was expressed in variable ways in HCC
tissues (Fig. 1C-E). Representative
IHC images are presented in Fig.
1.
GLUT-3 expression level in tumor tissues was
significantly higher compared with normal liver tissues (P<0.05)
and paracancerous tissues (P<0.05). However, there was no
statistical difference in GLUT-3 expression level between normal
liver and paracancerous tissues (P>0.05; Fig. 1F).
High GLUT-3 expression tissue score in HCC was
significantly and positively associated with elevated AFP level,
large tumor size, poor histological differentiation and TNM stages
III and IV (P<0.05).
Prognostic values of serum AFP level,
GLUT-3 expression and tumor size for the OS and DFS of patients
with HCC
The area under curves (AUCs) among serum AFP level,
GLUT-3 expression and tumor size in predicting OS and DFS in
patients with HCC were analyzed by ROC curves analysis in the
training and validation cohorts. In the training cohort, the AUCs
for GLUT-3 expression predicting the OS and DFS of patients with
HCC were 0.59 [95% confidence interval (CI), 0.53–0.66] and 0.58
(95% CI, 0.51–0.65), respectively. In the validation cohort, the
AUCs for GLUT-3 expression predicting the OS and DFS of patients
with HCC were 0.61 (95% CI, 0.51–0.71) and 0.61 (95% CI,
0.51–0.70), respectively (Fig.
2).
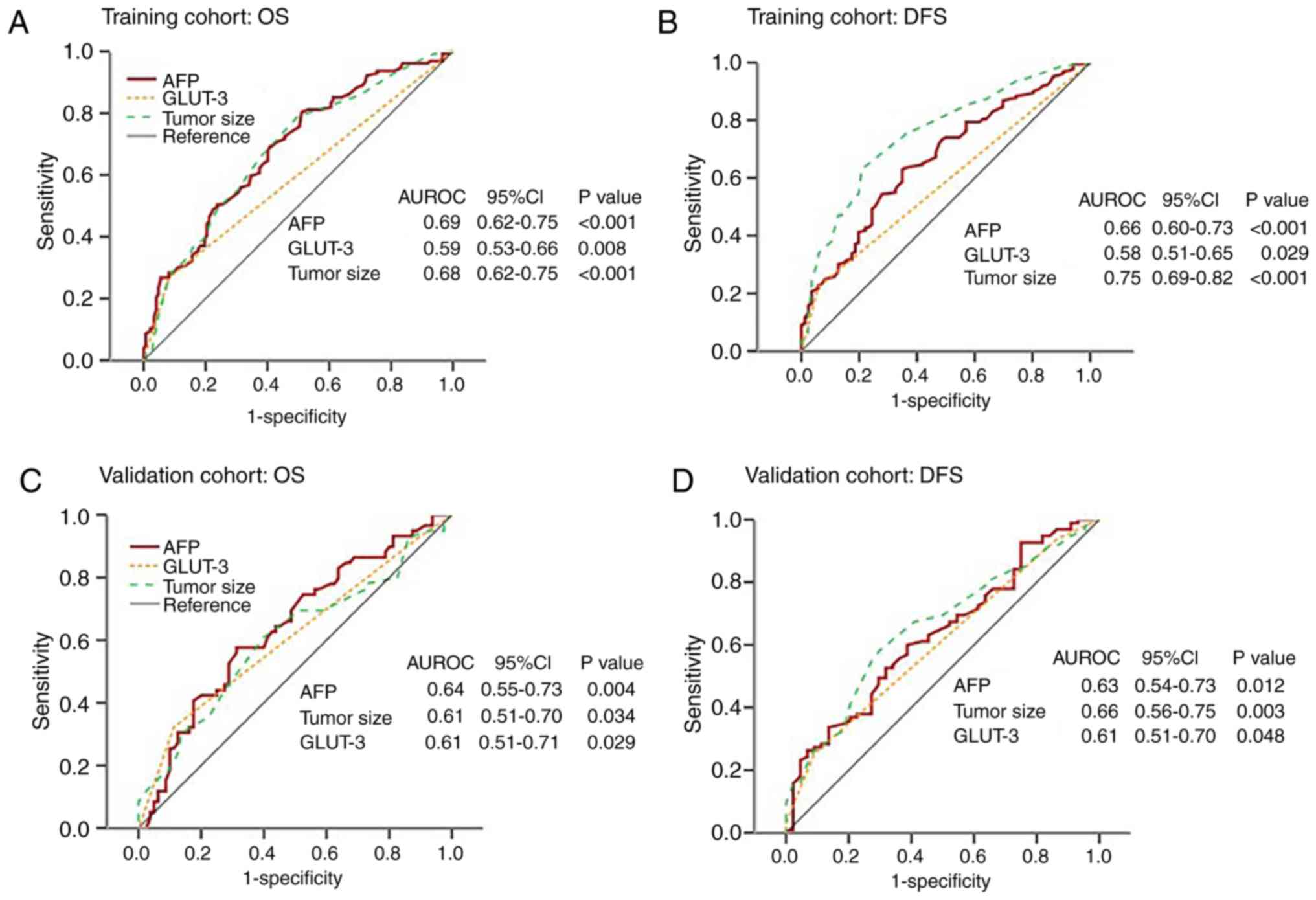 | Figure 2.ROC curves of serum AFP level, GLUT-3
expression level and tumor size for predicting OS and DFS in the
training and validation cohorts. (A) AUCs of AFP, GLUT-3 and tumor
size were 0.69, 0.59 and 0.68, respectively. (B) AUCs of AFP,
GLUT-3 and tumor size were 0.66, 0.58 and 0.75, respectively. (C)
AUCs of AFP, GLUT-3 and tumor size were 0.64, 0.61 and 0.61,
respectively. (D) AUCs of AFP, GLUT-3 and tumor size were 0.63,
0.61 and 0.66, respectively. AUC, area under curve; AFP,
α-fetoprotein; CI, confidence interval; DFS, disease-free survival;
GLUT-3, glucose transporter 3; HCC, hepatocellular carcinoma; OS,
overall survival; ROC, receiver operating characteristic; AUROC,
area under the receiver operating characteristic curve. |
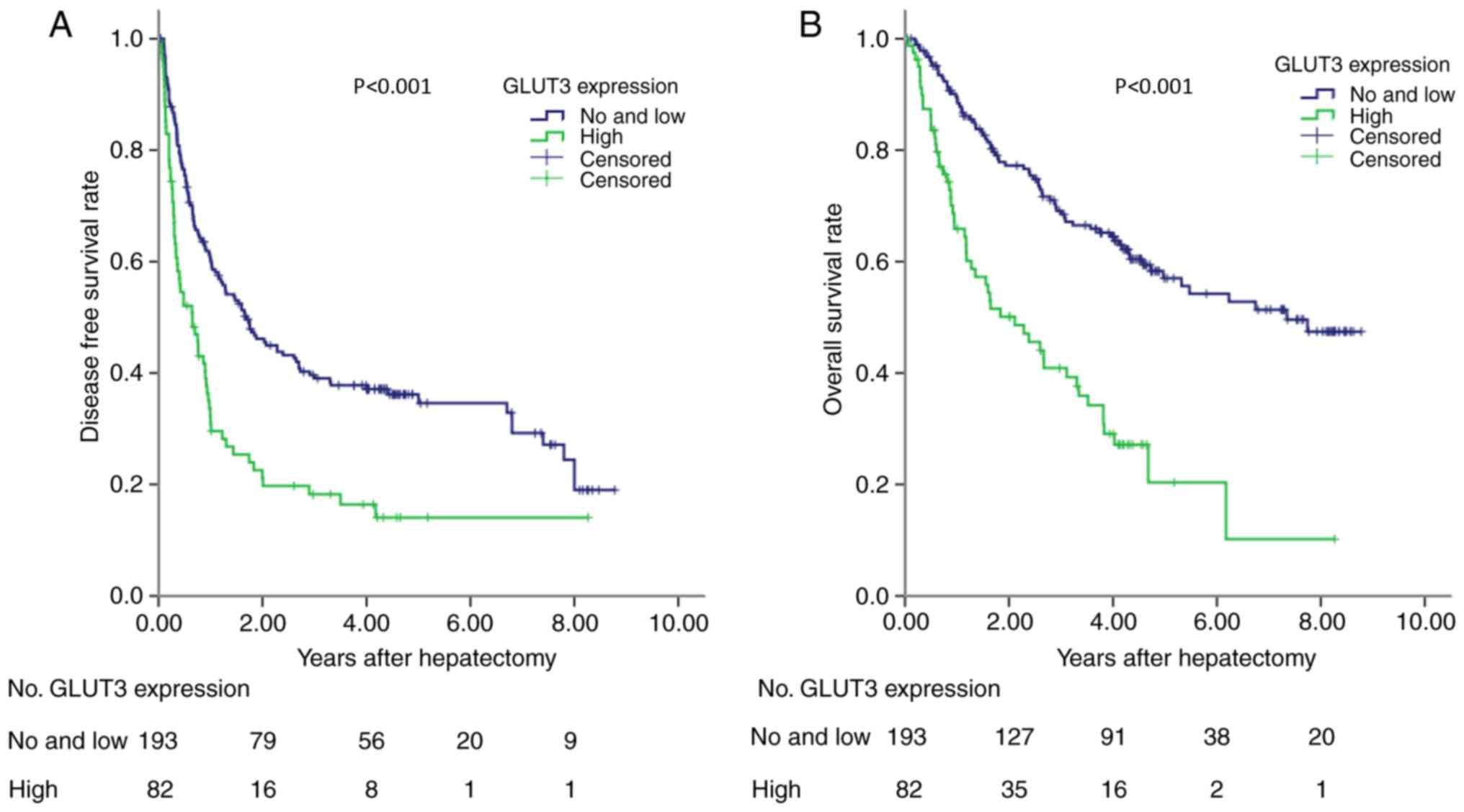
Survival and expression of GLUT-3
In the training cohort, the prognostic ability of
GLUT-3 expression was analyzed in 275 patients with HCC. High
GLUT-3 tissue score was significantly associated with reduced DFS
and OS (P<0.001; Fig. 3). To
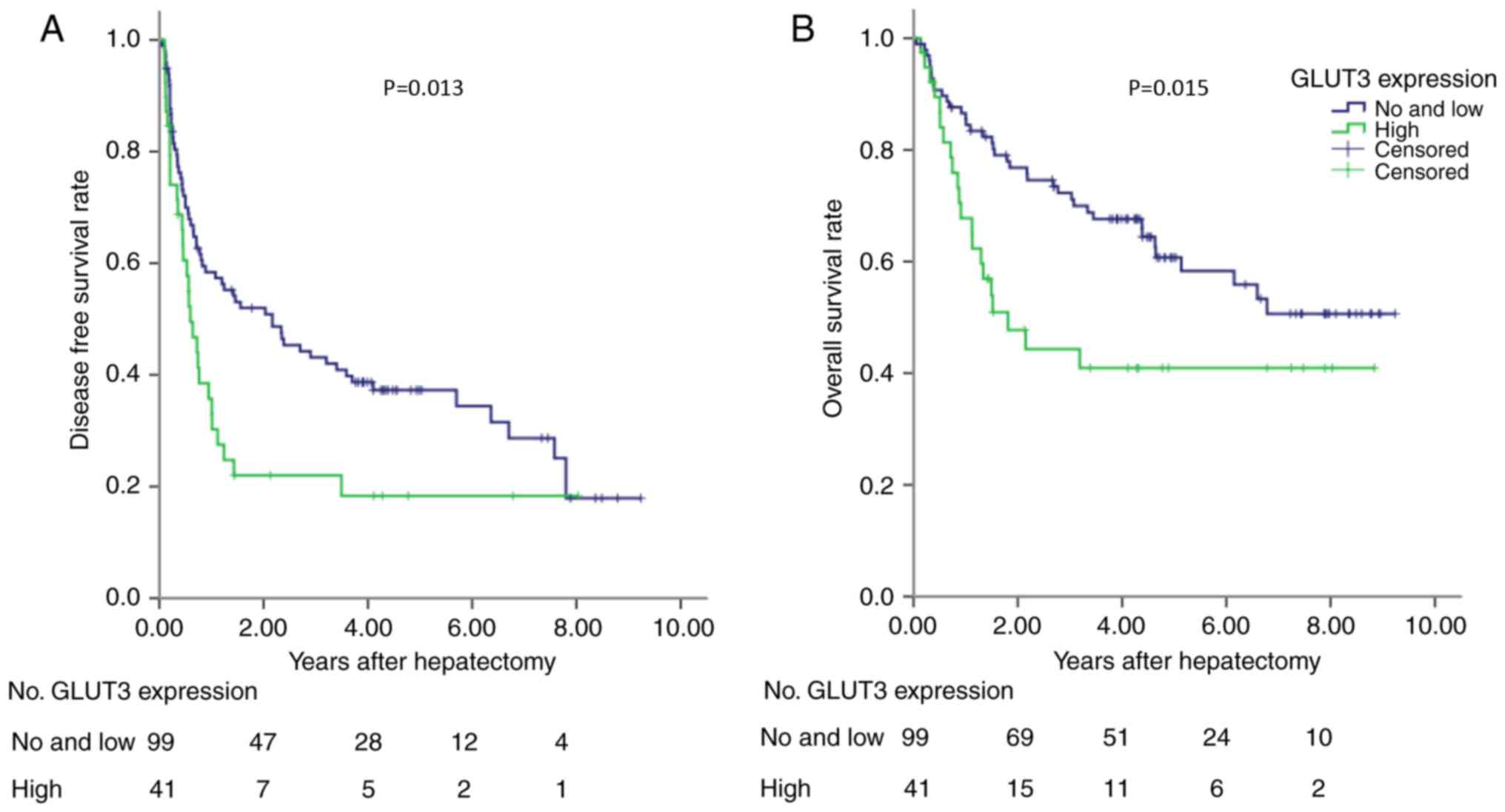
validate these findings, a validation cohort containing patients
with HCC was tested. The results demonstrated that high GLUT-3
expression level in the validation cohort was also associated with
poor DFS and OS (P<0.05; Fig. 4).
The predictive value of GLUT-3 expression in the validation cohort
was therefore validated for OS and DFS. The results from
multivariate Cox regression analysis demonstrated that GLUT-3
expression level, BCLC, vascular invasion and tumor size were
independent prognostic factors for the OS of patients with HCC
(Table III).
 | Table III.Cox regression model analysis in
training cohort. |
Table III.
Cox regression model analysis in
training cohort.
|
|
|
|
|
|
| 95.0% CI for Exp
(B) |
|---|
|
|
|
|
|
|
|
|
|---|
| Characteristic | B | SE | Wald | P-value | Exp (B) | Down | Upper |
|---|
| BCLC stage | 0.671 | 0.232 | 8.368 | 0.004 | 1.957 | 1.242 | 3.084 |
| GLUT-3
expression | 0.891 | 0.208 | 18.388 | <0.001 | 2.436 | 1.622 | 3.660 |
| Vascular
invasion | 0.636 | 0.253 | 6.341 | 0.012 | 1.889 | 1.151 | 3.099 |
| Tumor size | 0.687 | 0.208 | 10.957 | 0.001 | 1.988 | 1.323 | 2.985 |
Discussion
HCC is a common malignant tumor associated with high
mortality rate (11). Surgical
resection is the most effective treatment for patients with liver
cancer; however, the postoperative long-term survival rate of
patients is limited due to tumor recurrence (70% at 5 years)
(12,13). Traditional stratification schemes
that are based on clinical characteristics, including the American
Joint Committee on Cancer (14), TNM
and BCLC stages, provide limited prognostic guidance in the
management of patients with HCC due to disease heterogeneity
(3,15). Specific biomarkers would therefore
allow better stratification of the disease.
High serum AFP levels were associated with poor
prognosis of patients with HCC (16); however, the optimal cut-off value of
serum AFP level that could be used to predict a poor prognosis in
patients with HCC has not yet been determined. To our knowledge, no
molecular profiles have been established to date as the widely
satisfactory prognostic biomarker in HCC, although some biomarkers
have potentially predictive value (EpCAM signature,
G3-proliferation subclass, and SUOX) (17–20).
Prognostic molecular biomarkers should significantly predict the
survival prognosis and be indicated for most patients in clinical
practice. Therefore, more acceptable markers should be explored
according to standard criteria. In the present study, GLUT-3
expression and its prognostic value in patients with HCC were
analyzed. The results demonstrated that increased GLUT-3 expression
level was associated with decreased OS in patients with HCC
following tumor resection. In addition, GLUT-3 expression level was
also associated with elevated serum AFP level, large tumor size,
poor histological differentiation and TNM stages III and IV. Taken
together, these results demonstrated that GLUT-3 overexpression may
be considered as a biomarker for predicting the survival of
patients with HCC.
Increased energy metabolism has been accepted as a
hallmark of cancer, and is widely observed in cancer cells
(21). Increased glucose use by
glycolysis is an exclusive property of invasive cancer cells
(22). In tumor cells, glucose
uptake across the plasma membrane, which is mediated by
facilitative GLUTs, is thought to be the rate-limiting step of
glucose metabolism (23). Enhanced
glucose uptake in tumors can be therefore mediated by
overexpression of GLUTs overexpression (4). Of the 14 subtypes of human GLUTs, the
most closely associated with glucose metabolism are GLUT-1-5, which
have different body distributions under physiological conditions.
For instance, GLUT-l and 3 are the two most widely studied GLUTs,
and the ones that are most strongly associated with malignant
tumors. It has been reported that the upregulation of specific
glucose transporters may represent a key mechanism by which
malignant cells may achieve increased glucose uptake to support the
high rate of glycolysis (24,25). In
addition, GLUT-3 is overexpressed in human brain tumors, oral
tongue carcinoma, endometrial and breast cancers, non-small lung
carcinoma, oral squamous cell carcinoma and laryngeal carcinoma
(5,6,8,26,27). To
the best of our knowledge, the present study was the first to
analyze GLUT-3 expression and its association with the prognosis of
patients with HCC.
Compared with other GLUTs, GLUT-1 and 3 have a
higher affinity for glucose under physiological conditions
(4). GLUT-3 is mainly expressed in
the brain and testicles (28). In
addition, GLUT-3 is present in the intracellular vesicles of
various types of leukocyte and can be transferred to plasma
membrane under the activation of proliferative stimuli. For
instance, in T-lymphocytes, activation is characterized by the
emergence of insulin receptors on the plasma membrane; however,
their physiological significance is unclear (29). As aforementioned, numerous studies
demonstrated that GLUT-3 is also expressed in various types of
tumor tissue. Malignant cells grow faster and require more oxygen
and glucose than normal cells. Although mitochondrial oxidative
phosphorylation is considered to be a more efficient metabolic
process for ATP synthesis compared with glycolysis (30), tumor cells use glycolysis as the main
metabolic mode, even when sufficient oxygen is present. This
phenomenon is known as the Warburg effect (31). Although glycolysis produces less ATP,
a large number of intermediate metabolites can be used to construct
macromolecular structures, including RNA, proteins, lipids and NADP
(30). As tumors grow, cells may
encounter hypoxic conditions that lead to the induction of the
hypoxia inducible factor 1 (HIF-1) transcription factor, which
increases the transcription of glucose transporters (32). The decrease of ATP production
efficiency and the high energy requirement of tumor cells can
stimulate the increase of glucose uptake by malignant tumor cells
as aforementioned (30,32). Furthermore, GLUT-3 overexpression can
participate in the transport of more glucose into tumor cells in
order to satisfy their high metabolism and rapid growth. However,
the mechanism of GLUT-3 overexpression in tumor cells is unknown,
particularly in HCC, which was investigated, to the best of our
knowledge, in only one study to date (33). At present, there are several
hypotheses about the role of GLUT-3 overexpression in tumor cells,
including IL-6/signal transducer and activator of transcription 3
(STAT3), PI3K-Akt and hypoxia-induciblefactor-1 (HIF-1) signaling
pathways. A previous study demonstrated that activation of
IL-6/STAT3 pathway can stimulate expression of GLUT isoforms, and
therefore increase glucose uptake capacity in HCCs (33). STAT3 is a membrane receptor-mediated
nuclear transcription factor (34).
Cytokines, including IL-6, and growth factors (such as epidermal
growth factor and platelet-derived growth factor) activate STAT3
through phosphorylation. Phosphorylated STAT3 enters then the
nucleus, binds to the DNA regulatory regions of target genes and
induces their expression (35). High
expression of GLUT-3 may therefore be facilitated by the activation
of the IL-6/STAT3 pathway. The involvement of PI3K-Akt in GLUTs
regulation suggests that uncontrolled Akt activation, caused by
disturbances in PI3K α subunit or phosphate and tension homolog,
may mediate the increased glucose uptake and overexpression of
GLUTs observed in tumors. A previous study reported that in hypoxic
BeWo choriocarcinoma cells, HIF-1 mediates transcriptional
regulation of glycolytic genes with hypoxia-response elements in
their promoter regions, including GLUT-1 and GLUT-3 (36). GLUT-3 is overexpressed following
HIF-1α complex stabilization in response to hypoxia in BeWo
choriocarcinoma cells (36).
However, the underlying mechanisms of GLUT-3 overexpression in HCC
remain unclear and require further investigation.
The current study presented some limitations.
Firstly, there were inherent biases due to the retrospective nature
of the study. Secondly, the number of patients involved in this
study was relatively small, and results should be confirmed in a
larger patient cohort. Thirdly, the molecular mechanism of GLUT-3
overexpression in liver cancer tissues remains unclear and requires
further investigations.
In conclusion, the present study demonstrated the
association between GLUT-3 expression level and the clinical
prognosis of patients with HCC. Furthermore, the results
demonstrated that increased GLUT-3 expression level was associated
with poor prognosis of patients with HCC, suggesting that GLUT-3
may be considered as a potential prognostic in HCC. This finding
provided a basis for investigating GLUT-3 as a potential target in
the treatment of HCC, which may lead to the development of novel
treatment strategies.
Acknowledgements
Not applicable.
Funding
The research was supported by The Natural Science
Foundation of China Youth Project (grant no. 81802379) and the
Jinan Science and Technology Development Project (grant no.
201805029).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
HG, JL and YH participated in the conception and
design of the study. All authors collected and interpreted the
data. HG, XZ, MC, HL, FL, YH, XS, HZ, ZN and QN performed the
statistical analysis. YH drafted the manuscript, and HG and JL
edited it critically. All authors gave final approval of the
version to be published.
Ethics approval and consent to
participate
This study was approved by the Institutional Review
Board of Sun Yat-Sen University Cancer Center and Shandong
Provincial Hospital Affiliated to Shandong University. Written
informed consent was obtained from all the patients who
participated in this study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
AFP
|
α-fetoprotein
|
|
BCLC
|
Barcelona Clinic Liver Cancer
|
|
DFS
|
disease-free survival
|
|
GLUT
|
glucose transporter
|
|
HCC
|
hepatocellular carcinoma
|
|
HIF-1
|
hypoxia-inducible factor-1
|
|
OS
|
overall survival
|
|
STAT3
|
signal transducer and activator of
transcription3
|
|
TNM
|
Tumor-Node-Metastasis
|
References
|
1
|
Global Burden of Disease Cancer
Collaboration, ; Fitzmaurice C, Akinyemiju TF, Al Lami FH, Alam T,
Alizadeh-Navaei R, Allen C, Alsharif U, Alvis-Guzman N, Amini E,
Anderson BO, et al: Global, regional, and national cancer
incidence, mortality, years of life lost, years lived with
disability, and disability-adjusted life-years for 29 cancer
groups, 1990 to 2016: A systematic analysis for the global burden
of disease study. JAMA Oncol. 4:1553–1568. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
El-Serag HB, Marrero JA, Rudolph L and
Reddy KR: Diagnosis and treatment of hepatocellular carcinoma.
Gastroenterology. 134:1752–1763. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sirivatanauksorn Y and Tovikkai C:
Comparison of staging systems of hepatocellular carcinoma. HPB
Surg. 2011:8182172011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Barron CC, Bilan PJ, Tsakiridis T and
Tsiani E: Facilitative glucose transporters: Implications for
cancer detection, prognosis and treatment. Metabolism. 65:124–139.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ayala FR, Rocha RM, Carvalho KC, Carvalho
AL, da Cunha IW, Lourenço SV and Soares FA: GLUT1 and GLUT3 as
potential prognostic markers for Oral squamous cell carcinoma.
Molecules. 15:2374–2387. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Estilo CL, O-charoenrat P, Talbot S, Socci
ND, Carlson DL, Ghossein R, Williams T, Yonekawa Y, Ramanathan Y,
Boyle JO, et al: Oral tongue cancer gene expression profiling:
Identification of novel potential prognosticators by
oligonucleotide microarray analysis. BMC Cancer. 9:112009.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Baer S, Casaubon L, Schwartz MR,
Marcogliese A and Younes M: Glut3 expression in biopsy specimens of
laryngeal carcinoma is associated with poor survival. Laryngoscope.
112:393–396. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Younes M, Brown RW, Stephenson M, Gondo M
and Cagle PT: Overexpression of Glut1 and Glut3 in stage I nonsmall
cell lung carcinoma is associated with poor survival. Cancer.
80:1046–1051. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Conde VR, Oliveira PF, Nunes AR, Rocha CS,
Ramalhosa E, Pereira JA, Alves MG and Silva BM: The progression
from a lower to a higher invasive stage of bladder cancer is
associated with severe alterations in glucose and pyruvate
metabolism. Exp Cell Res. 335:91–98. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Pugh RN, Murray-Lyon IM, Dawson JL,
Pietroni MC and Williams R: Transection of the oesophagus for
bleeding oesophageal varices. Br J Surg. 60:646–649. 1973.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yang JD, Hainaut P, Gores GJ, Amadou A,
Plymoth A and Roberts LR: A global view of hepatocellular
carcinoma: Trends, risk, prevention and management. Nat Rev
Gastroenterol Hepatol. 16:589–604. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hong SS, Kim TK, Sung KB, Kim PN, Ha HK,
Kim AY and Lee MG: Extrahepatic spread of hepatocellular carcinoma:
A pictorial review. Eur Radiol. 13:874–882. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tabrizian P, Jibara G, Shrager B, Schwartz
M and Roayaie S: Recurrence of hepatocellular cancer after
resection: Patterns, treatments, and prognosis. Ann Surg.
261:947–955. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chun YH, Kim SU, Park JY, Kim DY, Han KH,
Chon CY, Kim BK, Choi GH, Kim KS, Choi JS and Ahn SH: Prognostic
value of the 7th edition of the AJCC staging system as a clinical
staging system in patients with hepatocellular carcinoma. Eur J
Cancer. 47:2568–2575. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Cillo U, Vitale A, Grigoletto F, Farinati
F, Brolese A, Zanus G, Neri D, Boccagni P, Srsen N, D'Amico F, et
al: Prospective validation of the Barcelona Clinic Liver Cancer
staging system. J Hepatol. 44:723–731. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ma WJ, Wang HY and Teng LS: Correlation
analysis of preoperative serum alpha-fetoprotein (AFP) level and
prognosis of hepatocellular carcinoma (HCC) after hepatectomy.
World J Surg Oncol. 11:2122013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yamashita T, Forgues M, Wang W, Kim JW, Ye
Q, Jia H, Budhu A, Zanetti KA, Chen Y, Qin LX, et al: EpCAM and
alpha-fetoprotein expression defines novel prognostic subtypes of
hepatocellular carcinoma. Cancer Res. 68:1451–1461. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Villanueva A, Hoshida Y, Battiston C,
Tovar V, Sia D, Alsinet C, Cornella H, Liberzon A, Kobayashi M,
Kumada H, et al: Combining clinical, pathology, and gene expression
data to predict recurrence of hepatocellular carcinoma.
Gastroenterology. 140:1501–1512.e2. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Jin GZ, Yu WL, Dong H, Zhou WP, Gu YJ, Yu
H, Yu H, Lu XY, Xian ZH, Liu YK, et al: SUOX is a promising
diagnostic and prognostic biomarker for hepatocellular carcinoma. J
Hepatol. 59:510–517. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Nault JC, De Reynies A, Villanueva A,
Calderaro J, Rebouissou S, Couchy G, Decaens T, Franco D, Imbeaud
S, Rousseau F, et al: A hepatocellular carcinoma 5-gene score
associated with survival of patients after liver resection.
Gastroenterology. 145:176–187. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hanahan D and Weinberg RA: Hallmarks of
cancer: The next generation. Cell. 144:646–674. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gatenby RA and Gillies RJ: Why do cancers
have high aerobic glycolysis? Nat Rev Cancer. 4:891–899. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Macheda ML, Rogers S and Best JD:
Molecular and cellular regulation of glucose transporter (GLUT)
proteins in cancer. J Cell Physiol. 202:654–662. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Krzeslak A, Wojcik-Krowiranda K, Forma E,
Jozwiak P, Romanowicz H, Bienkiewicz A and Brys M: Expression of
GLUT1 and GLUT3 glucose transporters in endometrial and breast
cancers. Pathol Oncol Res. 18:721–728. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ha TK, Her NG, Lee MG, Ryu BK, Lee JH, Han
J, Jeong SI, Kang MJ, Kim NH, Kim HJ and Chi SG: Caveolin-1
increases aerobic glycolysis in colorectal cancers by stimulating
HMGA1-mediated GLUT3 transcription. Cancer Res. 72:4097–4109. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Boado RJ, Black KL and Pardridge WM: Gene
expression of GLUT3 and GLUT1 glucose transporters in human brain
tumors. Brain Res Mol Brain Res. 27:51–57. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Starska K, Forma E, Jozwiak P, Brys M,
Lewy-Trenda I, Brzezinska-Blaszczyk E and Krzeslak A: Gene and
protein expression of glucose transporter 1 and glucose transporter
3 in human laryngeal cancer-the relationship with regulatory
hypoxia-inducible factor-1α expression, tumor invasiveness, and
patient prognosis. Tumour Biol. 36:2309–2321. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Haber RS, Weinstein SP, O'Boyle E and
Morgello S: Tissue distribution of the human GLUT3 glucose
transporter. Endocrinology. 132:2538–2543. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Maratou E, Dimitriadis G, Kollias A,
Boutati E, Lambadiari V, Mitrou P and Raptis SA: Glucose
transporter expression on the plasma membrane of resting and
activated white blood cells. Eur J Clin Invest. 37:282–290. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lunt SY and Vander Heiden MG: Aerobic
glycolysis: Meeting the metabolic requirements of cell
proliferation. Annu Rev Cell Dev Biol. 27:441–464. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Warburg O, Wind F and Negelein E: The
metabolism of tumors in the body. J Gen Physiol. 8:519–530. 1927.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
O'Rourke JF, Pugh CW, Bartlett SM and
Ratcliffe PJ: Identification of hypoxically inducible mRNAs in HeLa
cells using differential-display PCR. Role of hypoxia-inducible
factor-1. Eur J Biochem. 241:403–410. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhang HL, Wang MD, Zhou X, Qin CJ, Fu GB,
Tang L, Wu H, Huang S, Zhao LH, Zeng M, et al: Blocking
preferential glucose uptake sensitizes liver tumor-initiating cells
to glucose restriction and sorafenib treatment. Cancer Lett.
388:1–11. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Darnell JE Jr, Kerr IM and Stark GR:
Jak-STAT pathways and transcriptional activation in response to
IFNs and other extracellular signaling proteins. Science.
264:1415–1421. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhong Z, Wen Z and Darnell JE Jr: Stat3: A
STAT family member activated by tyrosine phosphorylation in
response to epidermal growth factor and interleukin-6. Science.
264:95–98. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Baumann MU, Zamudio S and Illsley NP:
Hypoxic upregulation of glucose transporters in BeWo
choriocarcinoma cells is mediated by hypoxia-inducible factor-1. Am
J Physiol Cell Physiol. 293:C477–C485. 2007. View Article : Google Scholar : PubMed/NCBI
|